Novel Insight into the Mechanisms of the Bidirectional Relationship between Diabetes and Periodontitis
Abstract
1. Periodontitis
2. Diabetes Mellitus
3. Bidirectional Relationship between Periodontitis and DM
4. Pathogenesis of Periodontitis
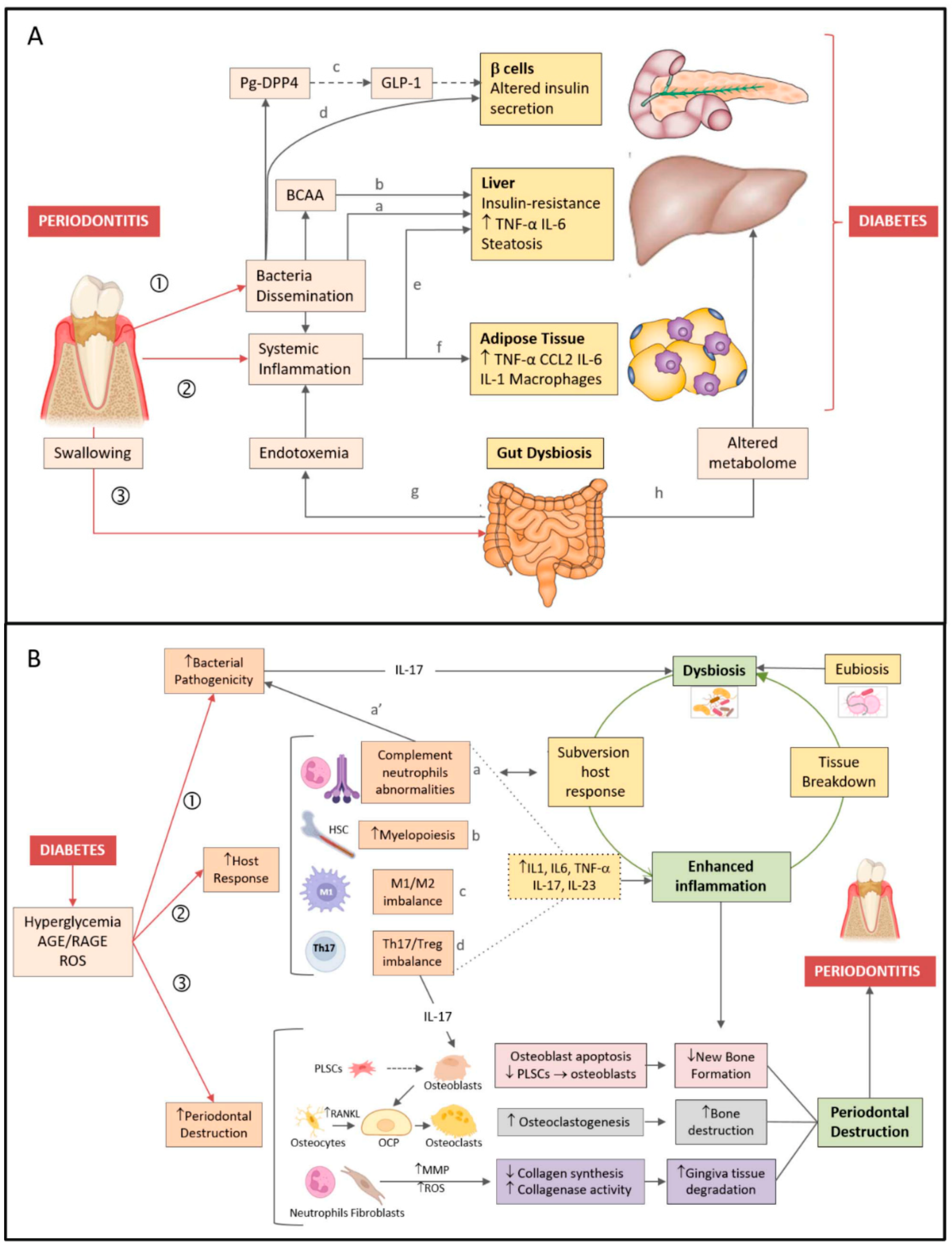
5. Mechanisms Linking Periodontitis and DM
5.1. Periodontitis—Type 2 DM Direction
5.1.1. Dissemination of Periodontal Bacteria
5.1.2. Inflammation
5.1.3. Oral-Gut Axis
5.2. DM-Periodontitis Direction
5.2.1. Changes in the Microbiota
5.2.2. Inflammatory Host Response
5.2.3. Immune Host Response
5.2.4. Periodontal Tissue Destruction
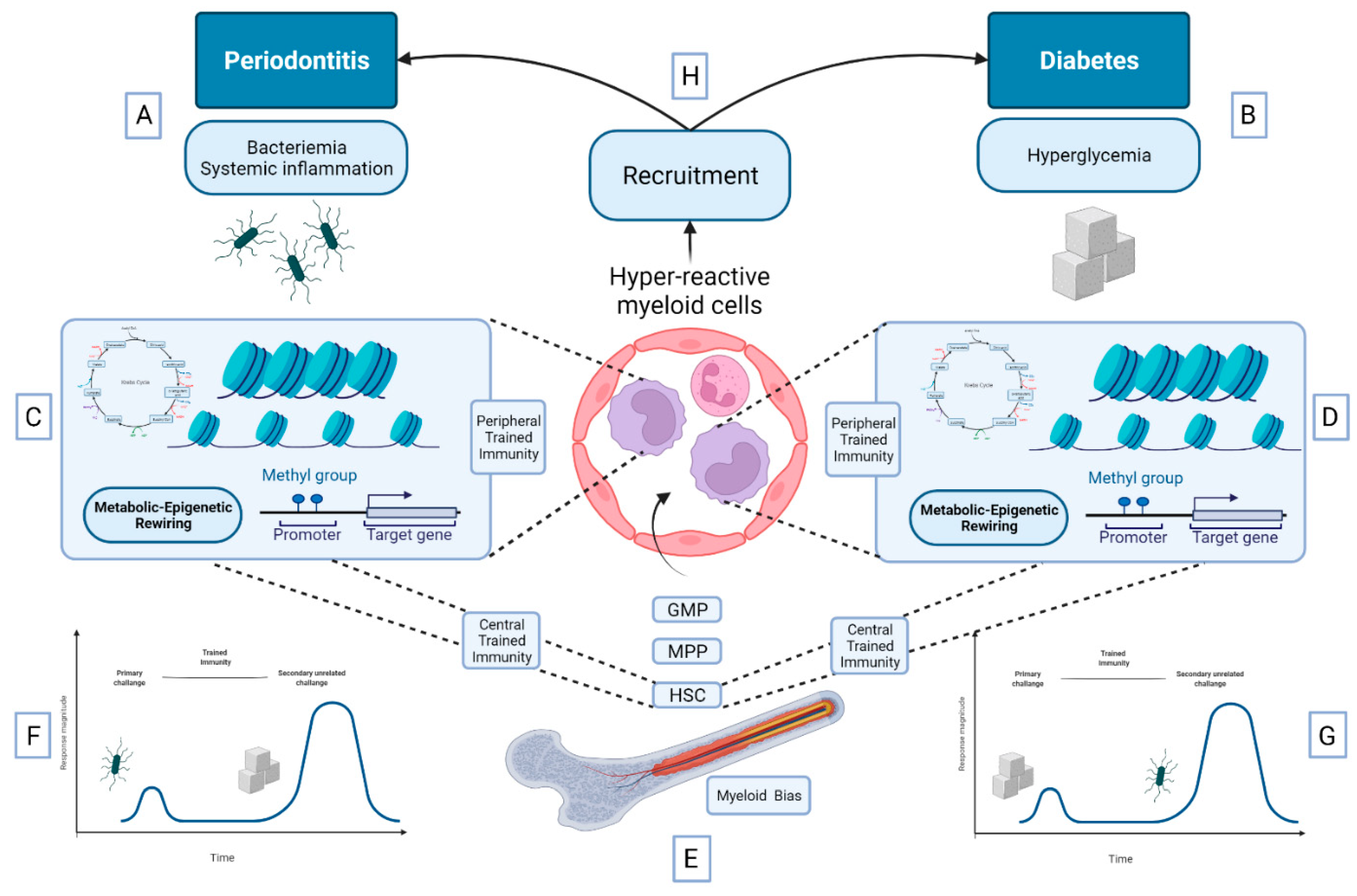
5.3. Bidirectional Relationship between DM and Periodontitis: A Role for Trained Immunity?
6. Conclusions and Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
References
- Eke, P.I.; Thornton-Evans, G.O.; Wei, L.; Borgnakke, W.S.; Dye, B.A.; Genco, R.J. Periodontitis in US Adults: National Health and Nutrition Examination Survey 2009–2014. J. Am. Dent. Assoc. 2018, 149, 576–588.e6. [Google Scholar] [CrossRef] [PubMed]
- Kassebaum, N.J.; Bernabé, E.; Dahiya, M.; Bhandari, B.; Murray, C.J.L.; Marcenes, W. Global Burden of Severe Periodontitis in 1990–2010: A Systematic Review and Meta-Regression. J. Dent. Res. 2014, 93, 1045–1053. [Google Scholar] [CrossRef] [PubMed]
- Papapanou, P.N.; Sanz, M.; Buduneli, N.; Dietrich, T.; Feres, M.; Fine, D.H.; Flemmig, T.F.; Garcia, R.; Giannobile, W.V.; Graziani, F.; et al. Periodontitis: Consensus Report of Workgroup 2 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Periodontol. 2018, 89 (Suppl. 1), S173–S182. [Google Scholar] [CrossRef] [PubMed]
- Jepsen, S.; Suvan, J.; Deschner, J. The Association of Periodontal Diseases with Metabolic Syndrome and Obesity. Periodontology 2000 2020, 83, 125–153. [Google Scholar] [CrossRef]
- Charupinijkul, A.; Arunyanak, S.; Rattanasiri, S.; Vathesatogkit, P.; Thienpramuk, L.; Lertpimonchai, A. The Effect of Obesity on Periodontitis Progression: The 10-Year Retrospective Cohort Study. Clin. Oral. Investig. 2021, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gobin, R.; Tian, D.; Liu, Q.; Wang, J. Periodontal Diseases and the Risk of Metabolic Syndrome: An Updated Systematic Review and Meta-Analysis. Front. Endocrinol. 2020, 11, 336. [Google Scholar] [CrossRef]
- Sanz, M.; Marco Del Castillo, A.; Jepsen, S.; Gonzalez-Juanatey, J.R.; D’Aiuto, F.; Bouchard, P.; Chapple, I.; Dietrich, T.; Gotsman, I.; Graziani, F.; et al. Periodontitis and Cardiovascular Diseases: Consensus Report. J. Clin. Periodontol. 2020, 47, 268–288. [Google Scholar] [CrossRef]
- IDF Diabetes Atlas, Tenth Edition. Available online: https://diabetesatlas.org/ (accessed on 19 December 2021).
- American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2021. Diabetes Care 2021, 44, S15–S33. [Google Scholar] [CrossRef] [PubMed]
- James, D.E.; Stöckli, J.; Birnbaum, M.J. The Aetiology and Molecular Landscape of Insulin Resistance. Nat. Rev. Mol. Cell Biol. 2021, 22, 751–771. [Google Scholar] [CrossRef]
- Petersen, M.C.; Vatner, D.F.; Shulman, G.I. Regulation of Hepatic Glucose Metabolism in Health and Disease. Nat. Rev. Endocrinol. 2017, 13, 572–587. [Google Scholar] [CrossRef] [PubMed]
- Leto, D.; Saltiel, A.R. Regulation of Glucose Transport by Insulin: Traffic Control of GLUT4. Nat. Rev. Mol. Cell Biol. 2012, 13, 383–396. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Bao, W.; Liu, J.; Ouyang, Y.-Y.; Wang, D.; Rong, S.; Xiao, X.; Shan, Z.-L.; Zhang, Y.; Yao, P.; et al. Inflammatory Markers and Risk of Type 2 Diabetes: A Systematic Review and Meta-Analysis. Diabetes Care 2013, 36, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Kaminga, A.C.; Wen, S.W.; Liu, A. Chemokines in Prediabetes and Type 2 Diabetes: A Meta-Analysis. Front. Immunol. 2021, 12, 622438. [Google Scholar] [CrossRef] [PubMed]
- Vozarova, B.; Weyer, C.; Lindsay, R.S.; Pratley, R.E.; Bogardus, C.; Tataranni, P.A. High White Blood Cell Count Is Associated with a Worsening of Insulin Sensitivity and Predicts the Development of Type 2 Diabetes. Diabetes 2002, 51, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Forbes, J.M.; Cooper, M.E. Mechanisms of Diabetic Complications. Physiol. Rev. 2013, 93, 137–188. [Google Scholar] [CrossRef] [PubMed]
- Taylor, G.W. Bidirectional Interrelationships between Diabetes and Periodontal Diseases: An Epidemiologic Perspective. Ann. Periodontol. 2001, 6, 99–112. [Google Scholar] [CrossRef]
- Löe, H. Periodontal disease: The sixth complication of diabetes mellitus. Diabetes Care 1993, 16, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.; Wang, C.; Ali, A.; Shih, Y.A.; Xie, Q.; Guo, C. Prevalence of Periodontitis in People Clinically Diagnosed with Diabetes Mellitus: A Meta-Analysis of Epidemiologic Studies. Acta Diabetol. 2021, 58, 1307–1327. [Google Scholar] [CrossRef]
- Wu, C.-Z.; Yuan, Y.-H.; Liu, H.-H.; Li, S.-S.; Zhang, B.-W.; Chen, W.; An, Z.-J.; Chen, S.-Y.; Wu, Y.-Z.; Han, B.; et al. Epidemiologic Relationship between Periodontitis and Type 2 Diabetes Mellitus. BMC Oral. Health 2020, 20, 204. [Google Scholar] [CrossRef]
- Kocher, T.; König, J.; Borgnakke, W.S.; Pink, C.; Meisel, P. Periodontal Complications of Hyperglycemia/Diabetes Mellitus: Epidemiologic Complexity and Clinical Challenge. Periodontology 2000 2018, 78, 59–97. [Google Scholar] [CrossRef]
- Genco, R.J.; Borgnakke, W.S. Diabetes as a Potential Risk for Periodontitis: Association Studies. Periodontology 2000 2020, 83, 40–45. [Google Scholar] [CrossRef]
- Romano, F.; Perotto, S.; Mohamed, S.E.O.; Bernardi, S.; Giraudi, M.; Caropreso, P.; Mengozzi, G.; Baima, G.; Citterio, F.; Berta, G.N.; et al. Bidirectional Association between Metabolic Control in Type-2 Diabetes Mellitus and Periodontitis Inflammatory Burden: A Cross-Sectional Study in an Italian Population. J. Clin. Med. 2021, 10, 1787. [Google Scholar] [CrossRef]
- Tonetti, M.S.; Greenwell, H.; Kornman, K.S. Staging and Grading of Periodontitis: Framework and Proposal of a New Classification and Case Definition. J. Clin. Periodontol. 2018, 45 (Suppl. 20), S149–S161. [Google Scholar] [CrossRef]
- Borgnakke, W.S.; Poudel, P. Diabetes and Oral Health: Summary of Current Scientific Evidence for Why Transdisciplinary Collaboration Is Needed. Front. Dent. Med. 2021, 2, 50. [Google Scholar] [CrossRef]
- Romano, F.; Perotto, S.; Mohamed, S.E.O.; Giraudi, M.; Bernardi, S.; Durazzo, M.; Gruden, G.; Aimetti, M. Type 2 Diabetes Mellitus and Periodontitis: Are Diabetic Patients Aware about This Bidirectional Association? Acta Diabetol. 2021, 58, 1277–1280. [Google Scholar] [CrossRef] [PubMed]
- Ziukaite, L.; Slot, D.E.; Van der Weijden, F.A. Prevalence of Diabetes Mellitus in People Clinically Diagnosed with Periodontitis: A Systematic Review and Meta-Analysis of Epidemiologic Studies. J. Clin. Periodontol. 2018, 45, 650–662. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Lee, J.S.; Lee, K.-J.; Woo, H.G.; Song, T.-J. Improved Oral Hygiene Is Associated with Decreased Risk of New-Onset Diabetes: A Nationwide Population-Based Cohort Study. Diabetologia 2020, 63, 924–933. [Google Scholar] [CrossRef] [PubMed]
- Borgnakke, W.S.; Ylöstalo, P.V.; Taylor, G.W.; Genco, R.J. Effect of Periodontal Disease on Diabetes: Systematic Review of Epidemiologic Observational Evidence. J. Clin. Periodontol. 2013, 40 (Suppl. 14), S135–S152. [Google Scholar] [CrossRef]
- Graziani, F.; Gennai, S.; Solini, A.; Petrini, M. A Systematic Review and Meta-Analysis of Epidemiologic Observational Evidence on the Effect of Periodontitis on Diabetes An Update of the EFP-AAP Review. J. Clin. Periodontol. 2018, 45, 167–187. [Google Scholar] [CrossRef]
- Song, T.-J.; Jeon, J.; Kim, J. Cardiovascular Risks of Periodontitis and Oral Hygiene Indicators in Patients with Diabetes Mellitus. Diabetes Metab. 2021, 47, 101252. [Google Scholar] [CrossRef]
- Van Dyke, T.E.; Kholy, K.E.; Ishai, A.; Takx, R.A.P.; Mezue, K.; Abohashem, S.M.; Ali, A.; Yuan, N.; Hsue, P.; Osborne, M.T.; et al. Inflammation of the Periodontium Associates with Risk of Future Cardiovascular Events. J. Periodontol. 2021, 92, 348–358. [Google Scholar] [CrossRef]
- Nguyen, A.T.M.; Akhter, R.; Garde, S.; Scott, C.; Twigg, S.M.; Colagiuri, S.; Ajwani, S.; Eberhard, J. The Association of Periodontal Disease with the Complications of Diabetes Mellitus. A Systematic Review. Diabetes Res. Clin. Pract. 2020, 165, 108244. [Google Scholar] [CrossRef] [PubMed]
- Borgnakke, W.S.; Anderson, P.F.; Shannon, C.; Jivanescu, A. Is There a Relationship between Oral Health and Diabetic Neuropathy? Curr. Diabetes Rep. 2015, 15, 93. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Khawaja, A.T.; Jin, L.; Chan, K.W.; Tonetti, M.; Tang, S.C.W.; Pelekos, G. Effect of Non-Surgical Periodontal Therapy on Renal Function in Chronic Kidney Disease Patients with Periodontitis: A Systematic Review and Meta-Analysis of Interventional Studies. Clin. Oral. Investig. 2020, 24, 1607–1618. [Google Scholar] [CrossRef]
- Alvarenga, M.O.P.; Miranda, G.H.N.; Ferreira, R.O.; Saito, M.T.; Fagundes, N.C.F.; Maia, L.C.; Lima, R.R. Association Between Diabetic Retinopathy and Periodontitis—A Systematic Review. Front. Public Health 2020, 8, 550614. [Google Scholar] [CrossRef] [PubMed]
- Genco, R.J.; Graziani, F.; Hasturk, H. Effects of Periodontal Disease on Glycemic Control, Complications, and Incidence of Diabetes Mellitus. Periodontology 2000 2020, 83, 59–65. [Google Scholar] [CrossRef]
- Sharma, P.; Dietrich, T.; Ferro, C.J.; Cockwell, P.; Chapple, I.L.C. Association between Periodontitis and Mortality in Stages 3-5 Chronic Kidney Disease: NHANES III and Linked Mortality Study. J. Clin. Periodontol. 2016, 43, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Stöhr, J.; Barbaresko, J.; Neuenschwander, M.; Schlesinger, S. Bidirectional Association between Periodontal Disease and Diabetes Mellitus: A Systematic Review and Meta-Analysis of Cohort Studies. Sci. Rep. 2021, 11, 13686. [Google Scholar] [CrossRef] [PubMed]
- Teshome, A.; Yitayeh, A. The Effect of Periodontal Therapy on Glycemic Control and Fasting Plasma Glucose Level in Type 2 Diabetic Patients: Systematic Review and Meta-Analysis. BMC Oral. Health 2016, 17, 31. [Google Scholar] [CrossRef]
- Chen, Y.-F.; Zhan, Q.; Wu, C.-Z.; Yuan, Y.-H.; Chen, W.; Yu, F.-Y.; Li, Y.; Li, L.-J. Baseline HbA1c Level Influences the Effect of Periodontal Therapy on Glycemic Control in People with Type 2 Diabetes and Periodontitis: A Systematic Review on Randomized Controlled Trails. Diabetes Ther. 2021, 12, 1249–1278. [Google Scholar] [CrossRef]
- Cao, R.; Li, Q.; Wu, Q.; Yao, M.; Chen, Y.; Zhou, H. Effect of Non-Surgical Periodontal Therapy on Glycemic Control of Type 2 Diabetes Mellitus: A Systematic Review and Bayesian Network Meta-Analysis. BMC Oral. Health 2019, 19, 176. [Google Scholar] [CrossRef]
- Baeza, M.; Morales, A.; Cisterna, C.; Cavalla, F.; Jara, G.; Isamitt, Y.; Pino, P.; Gamonal, J. Effect of Periodontal Treatment in Patients with Periodontitis and Diabetes: Systematic Review and Meta-Analysis. J. Appl. Oral. Sci. 2020, 28, e20190248. [Google Scholar] [CrossRef]
- Montero, E.; López, M.; Vidal, H.; Martínez, M.; Virto, L.; Marrero, J.; Herrera, D.; Zapatero, A.; Sanz, M. Impact of Periodontal Therapy on Systemic Markers of Inflammation in Patients with Metabolic Syndrome: A Randomized Clinical Trial. Diabetes Obes. Metab. 2020, 22, 2120–2132. [Google Scholar] [CrossRef]
- D’Aiuto, F.; Gkranias, N.; Bhowruth, D.; Khan, T.; Orlandi, M.; Suvan, J.; Masi, S.; Tsakos, G.; Hurel, S.; Hingorani, A.D.; et al. Systemic Effects of Periodontitis Treatment in Patients with Type 2 Diabetes: A 12 Month, Single-Centre, Investigator-Masked, Randomised Trial. Lancet Diabetes Endocrinol. 2018, 6, 954–965. [Google Scholar] [CrossRef]
- de Araújo, A.A.; de Morais, H.B.; de Medeiros, C.A.C.X.; de Castro Brito, G.A.; Guedes, P.M.M.; Hiyari, S.; Pirih, F.Q.; de Araújo Júnior, R.F. Gliclazide Reduced Oxidative Stress, Inflammation, and Bone Loss in an Experimental Periodontal Disease Model. J. Appl. Oral. Sci. 2019, 27, e20180211. [Google Scholar] [CrossRef]
- Kawahara, Y.; Kaneko, T.; Yoshinaga, Y.; Arita, Y.; Nakamura, K.; Koga, C.; Yoshimura, A.; Sakagami, R. Effects of Sulfonylureas on Periodontopathic Bacteria-Induced Inflammation. J. Dent. Res. 2020, 99, 830–838. [Google Scholar] [CrossRef]
- Sawada, N.; Adachi, K.; Nakamura, N.; Miyabe, M.; Ito, M.; Kobayashi, S.; Miyajima, S.-I.; Suzuki, Y.; Kikuchi, T.; Mizutani, M.; et al. Glucagon-Like Peptide-1 Receptor Agonist Liraglutide Ameliorates the Development of Periodontitis. J. Diabetes Res. 2020, 2020, 8843310. [Google Scholar] [CrossRef] [PubMed]
- Gu, M.; Wang, P.; Xiang, S.; Xu, D.; Jin, C.; Jiang, Z.; Hu, N. Effects of Type 2 Diabetes and Metformin on Salivary Microbiota in Patients with Chronic Periodontitis. Microb. Pathog. 2021, 161, 105277. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Li, M.; Xia, L.; Fang, Z.; Yu, S.; Gao, J.; Feng, Q.; Yang, P. Alteration of Salivary Microbiome in Periodontitis with or without Type-2 Diabetes Mellitus and Metformin Treatment. Sci. Rep. 2020, 10, 15363. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Zhang, P.; Wang, Q.; Ji, N.; Xia, S.; Ding, Y.; Wang, Q. Metformin Ameliorates Experimental Diabetic Periodontitis Independently of Mammalian Target of Rapamycin (MTOR) Inhibition by Reducing NIMA-Related Kinase 7 (Nek7) Expression. J. Periodontol. 2019, 90, 1032–1042. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yuan, X.; Wu, Y.; Pei, M.; Yang, M.; Wu, X.; Pang, Y.; Wang, J. Liraglutide Regulates Bone Destruction and Exhibits Anti-Inflammatory Effects in Periodontitis in Vitro and in Vivo. J. Dent. 2020, 94, 103310. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, A.R.; Rao, N.S.; Naik, S.B.; Kumari, M. Efficacy of Varying Concentrations of Subgingivally Delivered Metformin in the Treatment of Chronic Periodontitis: A Randomized Controlled Clinical Trial. J. Periodontol. 2013, 84, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Nicolini, A.C.; Grisa, T.A.; Muniz, F.W.M.G.; Rösing, C.K.; Cavagni, J. Effect of Adjuvant Use of Metformin on Periodontal Treatment: A Systematic Review and Meta-Analysis. Clin. Oral. Investig. 2019, 23, 2659–2666. [Google Scholar] [CrossRef] [PubMed]
- Akram, Z.; Vohra, F.; Javed, F. Locally Delivered Metformin as Adjunct to Scaling and Root Planing in the Treatment of Periodontal Defects: A Systematic Review and Meta-Analysis. J. Periodontal Res. 2018, 53, 941–949. [Google Scholar] [CrossRef]
- Van Dyke, T.E.; Bartold, P.M.; Reynolds, E.C. The Nexus Between Periodontal Inflammation and Dysbiosis. Front. Immunol. 2020, 11, 511. [Google Scholar] [CrossRef]
- Lamont, R.J.; Koo, H.; Hajishengallis, G. The Oral Microbiota: Dynamic Communities and Host Interactions. Nat. Rev. MicroBiol. 2018, 16, 745–759. [Google Scholar] [CrossRef]
- Hajishengallis, G.; Chavakis, T. Local and Systemic Mechanisms Linking Periodontal Disease and Inflammatory Comorbidities. Nat. Rev. Immunol. 2021, 21, 426–440. [Google Scholar] [CrossRef]
- Mark Welch, J.L.; Dewhirst, F.E.; Borisy, G.G. Biogeography of the Oral Microbiome: The Site-Specialist Hypothesis. Annu. Rev. MicroBiol. 2019, 73, 335–358. [Google Scholar] [CrossRef]
- Curtis, M.A.; Diaz, P.I.; Van Dyke, T.E. The Role of the Microbiota in Periodontal Disease. Periodontology 2000 2020, 83, 14–25. [Google Scholar] [CrossRef]
- Socransky, S.S.; Haffajee, A.D. Periodontal Microbial Ecology. Periodontology 2000 2005, 38, 135–187. [Google Scholar] [CrossRef]
- Hajishengallis, G.; Lamont, R.J. Beyond the Red Complex and into More Complexity: The Polymicrobial Synergy and Dysbiosis (PSD) Model of Periodontal Disease Etiology. Mol. Oral. MicroBiol. 2012, 27, 409–419. [Google Scholar] [CrossRef]
- Popadiak, K.; Potempa, J.; Riesbeck, K.; Blom, A.M. Biphasic Effect of Gingipains from Porphyromonas Gingivalis on the Human Complement System. J. Immunol. 2007, 178, 7242–7250. [Google Scholar] [CrossRef] [PubMed]
- Potempa, M.; Potempa, J.; Kantyka, T.; Nguyen, K.-A.; Wawrzonek, K.; Manandhar, S.P.; Popadiak, K.; Riesbeck, K.; Eick, S.; Blom, A.M. Interpain A, a Cysteine Proteinase from Prevotella Intermedia, Inhibits Complement by Degrading Complement Factor C3. PLoS Pathog. 2009, 5, e1000316. [Google Scholar] [CrossRef]
- Wang, M.; Krauss, J.L.; Domon, H.; Hosur, K.B.; Liang, S.; Magotti, P.; Triantafilou, M.; Triantafilou, K.; Lambris, J.D.; Hajishengallis, G. Microbial Hijacking of Complement-Toll-like Receptor Crosstalk. Sci. Signal 2010, 3, ra11. [Google Scholar] [CrossRef]
- Jusko, M.; Potempa, J.; Karim, A.Y.; Ksiazek, M.; Riesbeck, K.; Garred, P.; Eick, S.; Blom, A.M. A Metalloproteinase Karilysin Present in the Majority of Tannerella Forsythia Isolates Inhibits All Pathways of the Complement System. J. Immunol. 2012, 188, 2338–2349. [Google Scholar] [CrossRef] [PubMed]
- Maekawa, T.; Krauss, J.L.; Abe, T.; Jotwani, R.; Triantafilou, M.; Triantafilou, K.; Hashim, A.; Hoch, S.; Curtis, M.A.; Nussbaum, G.; et al. Porphyromonas Gingivalis Manipulates Complement and TLR Signaling to Uncouple Bacterial Clearance from Inflammation and Promote Dysbiosis. Cell Host Microbe 2014, 15, 768–778. [Google Scholar] [CrossRef] [PubMed]
- Taxman, D.J.; Swanson, K.V.; Broglie, P.M.; Wen, H.; Holley-Guthrie, E.; Huang, M.T.-H.; Callaway, J.B.; Eitas, T.K.; Duncan, J.A.; Ting, J.P.Y. Porphyromonas Gingivalis Mediates Inflammasome Repression in Polymicrobial CultuRes. through a Novel Mechanism Involving Reduced Endocytosis. J. Biol. Chem. 2012, 287, 32791–32799. [Google Scholar] [CrossRef] [PubMed]
- Hajishengallis, G.; Liang, S.; Payne, M.A.; Hashim, A.; Jotwani, R.; Eskan, M.A.; McIntosh, M.L.; Alsam, A.; Kirkwood, K.L.; Lambris, J.D.; et al. Low-Abundance Biofilm Species Orchestrates Inflammatory Periodontal Disease through the Commensal Microbiota and Complement. Cell Host Microbe 2011, 10, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Hajishengallis, G. The Inflammophilic Character of the Periodontitis-Associated Microbiota. Mol. Oral. MicroBiol. 2014, 29, 248–257. [Google Scholar] [CrossRef] [PubMed]
- Proctor, D.M.; Shelef, K.M.; Gonzalez, A.; Davis, C.L.; Dethlefsen, L.; Burns, A.R.; Loomer, P.M.; Armitage, G.C.; Ryder, M.I.; Millman, M.E.; et al. Microbial Biogeography and Ecology of the Mouth and Implications for Periodontal Diseases. Periodontology 2000 2020, 82, 26–41. [Google Scholar] [CrossRef] [PubMed]
- Stabholz, A.; Soskolne, W.A.; Shapira, L. Genetic and Environmental Risk Factors for Chronic Periodontitis and Aggressive Periodontitis. Periodontology 2000 2010, 53, 138–153. [Google Scholar] [CrossRef] [PubMed]
- Hajishengallis, E.; Hajishengallis, G. Neutrophil Homeostasis and Periodontal Health in Children and Adults. J. Dent. Res. 2014, 93, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Moutsopoulos, N.M.; Konkel, J.; Sarmadi, M.; Eskan, M.A.; Wild, T.; Dutzan, N.; Abusleme, L.; Zenobia, C.; Hosur, K.B.; Abe, T.; et al. Defective Neutrophil Recruitment in Leukocyte Adhesion Deficiency Type I Disease Causes Local IL-17-Driven Inflammatory Bone Loss. Sci. Transl. Med. 2014, 6, 229ra40. [Google Scholar] [CrossRef]
- Eskan, M.A.; Jotwani, R.; Abe, T.; Chmelar, J.; Lim, J.-H.; Liang, S.; Ciero, P.A.; Krauss, J.L.; Li, F.; Rauner, M.; et al. The Leukocyte Integrin Antagonist Del-1 Inhibits IL-17-Mediated Inflammatory Bone Loss. Nat. Immunol. 2012, 13, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Ryder, M.I. Comparison of Neutrophil Functions in Aggressive and Chronic Periodontitis. Periodontology 2000 2010, 53, 124–137. [Google Scholar] [CrossRef]
- Hajishengallis, G. Immunomicrobial Pathogenesis of Periodontitis: Keystones, Pathobionts, and Host Response. Trends Immunol. 2014, 35, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Petermann, F.; Rothhammer, V.; Claussen, M.C.; Haas, J.D.; Blanco, L.R.; Heink, S.; Prinz, I.; Hemmer, B.; Kuchroo, V.K.; Oukka, M.; et al. Γδ T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-DepenDent. Mechanism. Immunity 2010, 33, 351–363. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, M.; Maggi, L.; Micheletti, A.; Lazzeri, E.; Tamassia, N.; Costantini, C.; Cosmi, L.; Lunardi, C.; Annunziato, F.; Romagnani, S.; et al. Evidence for a Cross-Talk between Human Neutrophils and Th17 Cells. Blood 2010, 115, 335–343. [Google Scholar] [CrossRef]
- Zhang, S.; Gang, X.; Yang, S.; Cui, M.; Sun, L.; Li, Z.; Wang, G. The Alterations in and the Role of the Th17/Treg Balance in Metabolic Diseases. Front. Immunol. 2021, 12, 678355. [Google Scholar] [CrossRef]
- Huang, Z.; Pei, X.; Graves, D.T. The Interrelationship Between Diabetes, IL-17 and Bone Loss. Curr. Osteoporos Rep. 2020, 18, 23–31. [Google Scholar] [CrossRef]
- Bunte, K.; Beikler, T. Th17 Cells and the IL-23/IL-17 Axis in the Pathogenesis of Periodontitis and Immune-Mediated Inflammatory Diseases. Int. J. Mol. Sci. 2019, 20, 3394. [Google Scholar] [CrossRef]
- Graves, D.T.; Ding, Z.; Yang, Y. The Impact of Diabetes on Periodontal Diseases—PubMed. Periodontology 2000 2020, 82, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Liu, C.; Zheng, X.; Jia, X.; Peng, X.; Yang, R.; Zhou, X.; Xu, X. Porphyromonas Gingivalis Induces Insulin Resistance by Increasing BCAA Levels in Mice. J. Dent. Res. 2020, 99, 839–846. [Google Scholar] [CrossRef] [PubMed]
- Komazaki, R.; Katagiri, S.; Takahashi, H.; Maekawa, S.; Shiba, T.; Takeuchi, Y.; Kitajima, Y.; Ohtsu, A.; Udagawa, S.; Sasaki, N.; et al. Periodontal Pathogenic Bacteria, Aggregatibacter Actinomycetemcomitans Affect Non-Alcoholic Fatty Liver Disease by Altering Gut Microbiota and Glucose Metabolism. Sci. Rep. 2017, 7, 13950. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K.; Petro, B.J.; Shlimon, A.E.; Unterman, T.G. Effect of Periodontitis on Insulin Resistance and the Onset of Type 2 Diabetes Mellitus in Zucker Diabetic Fatty Rats. J. Periodontol. 2008, 79, 1208–1216. [Google Scholar] [CrossRef] [PubMed]
- Blasco-Baque, V.; Garidou, L.; Pomié, C.; Escoula, Q.; Loubieres, P.; Le Gall-David, S.; Lemaitre, M.; Nicolas, S.; Klopp, P.; Waget, A.; et al. Periodontitis Induced by Porphyromonas Gingivalis Drives Periodontal Microbiota Dysbiosis and Insulin Resistance via an Impaired Adaptive Immune Response. Gut 2017, 66, 872–885. [Google Scholar] [CrossRef] [PubMed]
- Schenkein, H.A.; Papapanou, P.N.; Genco, R.; Sanz, M. Mechanisms Underlying the Association between Periodontitis and Atherosclerotic Disease. Periodontology 2000 2020, 83, 90–106. [Google Scholar] [CrossRef] [PubMed]
- Horliana, A.C.R.T.; Chambrone, L.; Foz, A.M.; Artese, H.P.C.; de Sousa Rabelo, M.; Pannuti, C.M.; Romito, G.A. Dissemination of Periodontal Pathogens in the Bloodstream after Periodontal Procedures: A Systematic Review. PLoS ONE 2014, 9, e98271. [Google Scholar] [CrossRef] [PubMed]
- Forner, L.; Larsen, T.; Kilian, M.; Holmstrup, P. Incidence of Bacteremia after Chewing, Tooth Brushing and Scaling in Individuals with Periodontal Inflammation. J. Clin. Periodontol. 2006, 33, 401–407. [Google Scholar] [CrossRef]
- Rafferty, B.; Jönsson, D.; Kalachikov, S.; Demmer, R.T.; Nowygrod, R.; Elkind, M.S.V.; Bush, H.; Kozarov, E. Impact of Monocytic Cells on Recovery of Uncultivable Bacteria from Atherosclerotic Lesions. J. Intern Med. 2011, 270, 273–280. [Google Scholar] [CrossRef]
- Benedyk, M.; Mydel, P.M.; Delaleu, N.; Płaza, K.; Gawron, K.; Milewska, A.; Maresz, K.; Koziel, J.; Pyrc, K.; Potempa, J. Gingipains: Critical Factors in the Development of Aspiration Pneumonia Caused by Porphyromonas Gingivalis. J. Innate Immun. 2016, 8, 185–198. [Google Scholar] [CrossRef]
- Gnanasekaran, J.; Binder Gallimidi, A.; Saba, E.; Pandi, K.; Eli Berchoer, L.; Hermano, E.; Angabo, S.; Makkawi, H.A.; Khashan, A.; Daoud, A.; et al. Intracellular Porphyromonas Gingivalis Promotes the Tumorigenic Behavior of Pancreatic Carcinoma Cells. Cancers 2020, 12, 2331. [Google Scholar] [CrossRef] [PubMed]
- Rangé, H.; Labreuche, J.; Louedec, L.; Rondeau, P.; Planesse, C.; Sebbag, U.; Bourdon, E.; Michel, J.-B.; Bouchard, P.; Meilhac, O. Periodontal Bacteria in Human Carotid Atherothrombosis as a Potential Trigger for Neutrophil Activation. Atherosclerosis 2014, 236, 448–455. [Google Scholar] [CrossRef] [PubMed]
- Belstrøm, D.; Holmstrup, P.; Damgaard, C.; Borch, T.S.; Skjødt, M.-O.; Bendtzen, K.; Nielsen, C.H. The Atherogenic Bacterium Porphyromonas Gingivalis Evades Circulating Phagocytes by Adhering to Erythrocytes. Infect. Immun. 2011, 79, 1559–1565. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Shakhatreh, M.-A.K.; James, D.; Liang, S.; Nishiyama, S.-I.; Yoshimura, F.; Demuth, D.R.; Hajishengallis, G. Fimbrial Proteins of Porphyromonas Gingivalis Mediate in Vivo Virulence and Exploit TLR2 and Complement Receptor 3 to Persist in Macrophages. J. Immunol. 2007, 179, 2349–2358. [Google Scholar] [CrossRef] [PubMed]
- Slocum, C.; Coats, S.R.; Hua, N.; Kramer, C.; Papadopoulos, G.; Weinberg, E.O.; Gudino, C.V.; Hamilton, J.A.; Darveau, R.P.; Genco, C.A. Distinct Lipid a Moieties Contribute to Pathogen-Induced Site-Specific Vascular Inflammation. PLoS Pathog. 2014, 10, e1004215. [Google Scholar] [CrossRef] [PubMed]
- Carey, I.M.; Critchley, J.A.; DeWilde, S.; Harris, T.; Hosking, F.J.; Cook, D.G. Risk of Infection in Type 1 and Type 2 Diabetes Compared With the General Population: A Matched Cohort Study. Diabetes Care 2018, 41, 513–521. [Google Scholar] [CrossRef]
- Toniolo, A.; Cassani, G.; Puggioni, A.; Rossi, A.; Colombo, A.; Onodera, T.; Ferrannini, E. The Diabetes Pandemic and Associated Infections: Suggestions for Clinical Microbiology. Rev. Med. MicroBiol. 2019, 30, 1–17. [Google Scholar] [CrossRef]
- Abu-Ashour, W.; Twells, L.K.; Valcour, J.E.; Gamble, J.-M. Diabetes and the Occurrence of Infection in Primary Care: A Matched Cohort Study. BMC Infect. Dis. 2018, 18, 67. [Google Scholar] [CrossRef] [PubMed]
- Jafar, N.; Edriss, H.; Nugent, K. The Effect of Short-Term Hyperglycemia on the Innate Immune System. Am. J. Med. Sci. 2016, 351, 201–211. [Google Scholar] [CrossRef]
- Ghosh, P.; Vaidya, A.; Sahoo, R.; Goldfine, A.; Herring, N.; Bry, L.; Chorev, M.; Halperin, J.A. Glycation of the Complement Regulatory Protein CD59 Is a Novel Biomarker for Glucose Handling in Humans. J. Clin. Endocrinol. Metab. 2014, 99, E999–E1006. [Google Scholar] [CrossRef]
- Hu, R.; Xia, C.-Q.; Butfiloski, E.; Clare-Salzler, M. Effect of High Glucose on Cytokine Production by Human Peripheral Blood Immune Cells and Type I Interferon Signaling in Monocytes: Implications for the Role of Hyperglycemia in the Diabetes Inflammatory Process and Host Defense against Infection. Clin. Immunol. 2018, 195, 139–148. [Google Scholar] [CrossRef]
- Wang, X.; Ota, N.; Manzanillo, P.; Kates, L.; Zavala-Solorio, J.; Eidenschenk, C.; Zhang, J.; Lesch, J.; Lee, W.P.; Ross, J.; et al. Interleukin-22 Alleviates Metabolic Disorders and RestoRes. Mucosal Immunity in Diabetes. Nature 2014, 514, 237–241. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, P.; Gronke, K.; Diefenbach, A. A Catch-22: Interleukin-22 and Cancer. Eur. J. Immunol. 2018, 48, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Montoya-Rosales, A.; Castro-Garcia, P.; Torres-Juarez, F.; Enciso-Moreno, J.A.; Rivas-Santiago, B. Glucose Levels Affect LL-37 Expression in Monocyte-Derived Macrophages Altering the Mycobacterium Tuberculosis Intracellular Growth Control. Microb. Pathog. 2016, 97, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.S.; Lee, K.O.; Low, K.C.; Gamage, A.M.; Liu, Y.; Tan, G.-Y.G.; Koh, H.Q.V.; Alonso, S.; Gan, Y.-H. Glutathione Deficiency in Type 2 Diabetes Impairs Cytokine Responses and Control of Intracellular Bacteria. J. Clin. Invest. 2012, 122, 2289–2300. [Google Scholar] [CrossRef] [PubMed]
- Delamaire, M.; Maugendre, D.; Moreno, M.; Le Goff, M.C.; Allannic, H.; Genetet, B. Impaired Leucocyte Functions in Diabetic Patients. Diabet Med. 1997, 14, 29–34. [Google Scholar] [CrossRef]
- Zykova, S.N.; Jenssen, T.G.; Berdal, M.; Olsen, R.; Myklebust, R.; Seljelid, R. Altered Cytokine and Nitric Oxide Secretion in Vitro by Macrophages from Diabetic Type II-like Db/Db Mice. Diabetes 2000, 49, 1451–1458. [Google Scholar] [CrossRef]
- Llorente, L.; De La Fuente, H.; Richaud-Patin, Y.; Alvarado-De La Barrera, C.; Diaz-Borjón, A.; López-Ponce, A.; Lerman-Garber, I.; Jakez-Ocampo, J. Innate Immune Response Mechanisms in Non-Insulin DepenDent. Diabetes Mellitus Patients Assessed by Flow Cytoenzymology. Immunol. Lett. 2000, 74, 239–244. [Google Scholar] [CrossRef]
- Thomsen, R.W.; Hundborg, H.H.; Lervang, H.-H.; Johnsen, S.P.; Schønheyder, H.C.; Sørensen, H.T. Diabetes Mellitus as a Risk and Prognostic Factor for Community-Acquired Bacteremia Due to Enterobacteria: A 10-Year, Population-Based Study among Adults. Clin. Infect. Dis. 2005, 40, 628–631. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Amano, A.; Nakagawa, I.; Okahashi, N.; Hamada, N. Variations of Porphyromonas Gingivalis Fimbriae in Relation to Microbial Pathogenesis. J. Periodontal Res. 2004, 39, 136–142. [Google Scholar] [CrossRef]
- Ojima, M.; Takeda, M.; Yoshioka, H.; Nomura, M.; Tanaka, N.; Kato, T.; Shizukuishi, S.; Amano, A. Relationship of Periodontal Bacterium Genotypic Variations with Periodontitis in Type 2 Diabetic Patients. Diabetes Care 2005, 28, 433–434. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Takamura, H.; Yoshida, K.; Okamura, H.; Fujiwara, N.; Ozaki, K. Porphyromonas Gingivalis Attenuates the Insulin-Induced Phosphorylation and Translocation of Forkhead Box Protein O1 in Human Hepatocytes. Arch. Oral. Biol. 2016, 69, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Kashiwagi, Y.; Aburaya, S.; Sugiyama, N.; Narukawa, Y.; Sakamoto, Y.; Takahashi, M.; Uemura, H.; Yamashita, R.; Tominaga, S.; Hayashi, S.; et al. Porphyromonas Gingivalis Induces Entero-Hepatic Metabolic Derangements with Alteration of Gut Microbiota in a Type 2 Diabetes Mouse Model. Sci. Rep. 2021, 11, 18398. [Google Scholar] [CrossRef] [PubMed]
- Seyama, M.; Yoshida, K.; Yoshida, K.; Fujiwara, N.; Ono, K.; Eguchi, T.; Kawai, H.; Guo, J.; Weng, Y.; Haoze, Y.; et al. Outer Membrane Vesicles of Porphyromonas Gingivalis Attenuate Insulin Sensitivity by Delivering Gingipains to the Liver. Biochim. Biophys. Acta Mol. Basis Dis. 2020, 1866, 165731. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, M.; Yoshida, K.; Okamura, H.; Ochiai, K.; Takamura, H.; Fujiwara, N.; Ozaki, K. Oral Porphyromonas Gingivalis Translocates to the Liver and Regulates Hepatic Glycogen Synthesis through the Akt/GSK-3β Signaling Pathway. Biochim. Biophys. Acta 2013, 1832, 2035–2043. [Google Scholar] [CrossRef]
- Yoon, M.-S. The Emerging Role of Branched-Chain Amino Acids in Insulin Resistance and Metabolism. Nutrients 2016, 8, 405. [Google Scholar] [CrossRef]
- White, P.J.; McGarrah, R.W.; Herman, M.A.; Bain, J.R.; Shah, S.H.; Newgard, C.B. Insulin Action, Type 2 Diabetes, and Branched-Chain Amino Acids: A Two-Way Street. Mol. Metab. 2021, 52, 101261. [Google Scholar] [CrossRef]
- Newgard, C.B.; An, J.; Bain, J.R.; Muehlbauer, M.J.; Stevens, R.D.; Lien, L.F.; Haqq, A.M.; Shah, S.H.; Arlotto, M.; Slentz, C.A.; et al. A Branched-Chain Amino Acid-Related Metabolic Signature That Differentiates Obese and Lean Humans and Contributes to Insulin Resistance. Cell Metab. 2009, 9, 311–326. [Google Scholar] [CrossRef]
- Menni, C.; Fauman, E.; Erte, I.; Perry, J.R.B.; Kastenmüller, G.; Shin, S.-Y.; Petersen, A.-K.; Hyde, C.; Psatha, M.; Ward, K.J.; et al. Biomarkers for Type 2 Diabetes and Impaired Fasting Glucose Using a Nontargeted Metabolomics Approach. Diabetes 2013, 62, 4270–4276. [Google Scholar] [CrossRef]
- Nawaz, S.S.; Siddiqui, K. The Emerging Role of Branch Chain Amino Acids in the Prediction of Diabetes: A Brief Review. Curr. Diabetes Rev. 2020, 16, 532–537. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, H.K.; Gudmundsdottir, V.; Nielsen, H.B.; Hyotylainen, T.; Nielsen, T.; Jensen, B.A.H.; Forslund, K.; Hildebrand, F.; Prifti, E.; Falony, G.; et al. Human Gut MicrObes. Impact Host Serum Metabolome and Insulin Sensitivity. Nature 2016, 535, 376–381. [Google Scholar] [CrossRef]
- Ilievski, V.; Bhat, U.G.; Suleiman-Ata, S.; Bauer, B.A.; Toth, P.T.; Olson, S.T.; Unterman, T.G.; Watanabe, K. Oral Application of a Periodontal Pathogen Impacts SerpinE1 Expression and Pancreatic Islet Architecture in Prediabetes. J. Periodontal Res. 2017, 52, 1032–1041. [Google Scholar] [CrossRef] [PubMed]
- Ilievski, V.; Toth, P.T.; Valyi-Nagy, K.; Valyi-Nagy, T.; Green, S.J.; Marattil, R.S.; Aljewari, H.W.; Wicksteed, B.; O’Brien-Simpson, N.M.; Reynolds, E.C.; et al. Identification of a Periodontal Pathogen and Bihormonal Cells in Pancreatic Islets of Humans and a Mouse Model of Periodontitis. Sci. Rep. 2020, 10, 9976. [Google Scholar] [CrossRef] [PubMed]
- Diomede, F.; Thangavelu, S.R.; Merciaro, I.; D’Orazio, M.; Bramanti, P.; Mazzon, E.; Trubiani, O. Porphyromonas Gingivalis Lipopolysaccharide Stimulation in Human Periodontal Ligament Stem Cells: Role of Epigenetic Modifications to the Inflammation. Eur. J. HistoChem. 2017, 61, 2826. [Google Scholar] [CrossRef]
- Solini, A.; Suvan, J.; Santini, E.; Gennai, S.; Seghieri, M.; Masi, S.; Petrini, M.; D’Aiuto, F.; Graziani, F. Periodontitis Affects Glucoregulatory Hormones in Severely Obese Individuals. Int. J. Obes. 2019, 43, 1125–1129. [Google Scholar] [CrossRef]
- Kumagai, Y.; Konishi, K.; Gomi, T.; Yagishita, H.; Yajima, A.; Yoshikawa, M. Enzymatic Properties of Dipeptidyl Aminopeptidase IV Produced by the Periodontal Pathogen Porphyromonas Gingivalis and Its Participation in Virulence. Infect. Immun. 2000, 68, 716–724. [Google Scholar] [CrossRef][Green Version]
- Ohara-Nemoto, Y.; Nakasato, M.; Shimoyama, Y.; Baba, T.T.; Kobayakawa, T.; Ono, T.; Yaegashi, T.; Kimura, S.; Nemoto, T.K. Degradation of Incretins and Modulation of Blood Glucose Levels by Periodontopathic Bacterial Dipeptidyl Peptidase 4. Infect. Immun. 2017, 85, e00277-17. [Google Scholar] [CrossRef]
- Konkel, J.E.; O’Boyle, C.; Krishnan, S. Distal Consequences of Oral Inflammation. Front. Immunol. 2019, 10, 1403. [Google Scholar] [CrossRef] [PubMed]
- Loos, B.G.; Craandijk, J.; Hoek, F.J.; Wertheim-van Dillen, P.M.; van der Velden, U. Elevation of Systemic Markers Related to Cardiovascular Diseases in the Peripheral Blood of Periodontitis Patients. J. Periodontol. 2000, 71, 1528–1534. [Google Scholar] [CrossRef]
- Roca-Millan, E.; González-Navarro, B.; Sabater-Recolons, M.-M.; Marí-Roig, A.; Jané-Salas, E.; López-López, J. Periodontal Treatment on Patients with Cardiovascular Disease: Systematic Review and Meta-Analysis. Med. Oral. Patol. Oral. Cir. Bucal 2018, 23, e681–e690. [Google Scholar] [CrossRef]
- Türer, Ç.C.; Durmuş, D.; Balli, U.; Güven, B. Effect of Non-Surgical Periodontal Treatment on Gingival Crevicular Fluid and Serum Endocan, Vascular Endothelial Growth Factor-A, and Tumor Necrosis Factor-Alpha Levels. J. Periodontol. 2017, 88, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, Y.; Kato, T.; Takahashi, N.; Nakajima, M.; Arimatsu, K.; Minagawa, T.; Sato, K.; Ohno, H.; Yamazaki, K. Ligature-Induced Periodontitis in Mice Induces Elevated Levels of Circulating Interleukin-6 but Shows Only Weak Effects on Adipose and Liver Tissues. J. Periodontal Res. 2016, 51, 639–646. [Google Scholar] [CrossRef]
- O’Boyle, C.; Haley, M.J.; Lemarchand, E.; Smith, C.J.; Allan, S.M.; Konkel, J.E.; Lawrence, C.B. Ligature-Induced Periodontitis Induces Systemic Inflammation but Does Not Alter Acute Outcome after Stroke in Mice. Int. J. Stroke 2020, 15, 175–187. [Google Scholar] [CrossRef]
- Sasaki, N.; Katagiri, S.; Komazaki, R.; Watanabe, K.; Maekawa, S.; Shiba, T.; Udagawa, S.; Takeuchi, Y.; Ohtsu, A.; Kohda, T.; et al. Endotoxemia by Porphyromonas Gingivalis Injection Aggravates Non-Alcoholic Fatty Liver Disease, Disrupts Glucose/Lipid Metabolism, and Alters Gut Microbiota in Mice. Front. MicroBiol. 2018, 9, 2470. [Google Scholar] [CrossRef] [PubMed]
- Mei, F.; Xie, M.; Huang, X.; Long, Y.; Lu, X.; Wang, X.; Chen, L. Porphyromonas Gingivalis and Its Systemic Impact: Current Status. Pathogens 2020, 9, 944. [Google Scholar] [CrossRef] [PubMed]
- Arimatsu, K.; Yamada, H.; Miyazawa, H.; Minagawa, T.; Nakajima, M.; Ryder, M.I.; Gotoh, K.; Motooka, D.; Nakamura, S.; Iida, T.; et al. Oral Pathobiont Induces Systemic Inflammation and Metabolic Changes Associated with Alteration of Gut Microbiota. Sci. Rep. 2014, 4, 4828. [Google Scholar] [CrossRef]
- Hatasa, M.; Ohsugi, Y.; Katagiri, S.; Yoshida, S.; Niimi, H.; Morita, K.; Tsuchiya, Y.; Shimohira, T.; Sasaki, N.; Maekawa, S.; et al. Endotoxemia by Porphyromonas Gingivalis Alters Endocrine Functions in Brown Adipose Tissue. Front. Cell Infect. MicroBiol. 2020, 10, 580577. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, Q. Periodontitis Aggravated Pancreatic β-Cell Dysfunction in Diabetic Mice through Interleukin-12 Regulation on Klotho. J. Diabetes Investig. 2016, 7, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Utsugi, T.; Ohno, T.; Ohyama, Y.; Uchiyama, T.; Saito, Y.; Matsumura, Y.; Aizawa, H.; Itoh, H.; Kurabayashi, M.; Kawazu, S.; et al. Decreased Insulin Production and Increased Insulin Sensitivity in the Klotho Mutant Mouse, a Novel Animal Model for Human Aging. Metabolism 2000, 49, 1118–1123. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, T.S.; Hayward, M.R.; Coelho, L.P.; Li, S.S.; Costea, P.I.; Voigt, A.Y.; Wirbel, J.; Maistrenko, O.M.; Alves, R.J.; Bergsten, E.; et al. Extensive Transmission of MicrObes. along the Gastrointestinal Tract. Elife 2019, 8, e42693. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, K.; Kato, T.; Tsuboi, Y.; Miyauchi, E.; Suda, W.; Sato, K.; Nakajima, M.; Yokoji-Takeuchi, M.; Yamada-Hara, M.; Tsuzuno, T.; et al. Oral Pathobiont-Induced Changes in Gut Microbiota Aggravate the Pathology of Nonalcoholic Fatty Liver Disease in Mice. Front. Immunol. 2021, 12, 766170. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Bibiloni, R.; Knauf, C.; Waget, A.; Neyrinck, A.M.; Delzenne, N.M.; Burcelin, R. Changes in Gut Microbiota Control Metabolic Endotoxemia-Induced Inflammation in High-Fat Diet-Induced Obesity and Diabetes in Mice. Diabetes 2008, 57, 1470–1481. [Google Scholar] [CrossRef]
- Ohtsu, A.; Takeuchi, Y.; Katagiri, S.; Suda, W.; Maekawa, S.; Shiba, T.; Komazaki, R.; Udagawa, S.; Sasaki, N.; Hattori, M.; et al. Influence of Porphyromonas Gingivalis in Gut Microbiota of Streptozotocin-Induced Diabetic Mice. Oral. Dis. 2019, 25, 868–880. [Google Scholar] [CrossRef]
- Kato, T.; Yamazaki, K.; Nakajima, M.; Date, Y.; Kikuchi, J.; Hase, K.; Ohno, H.; Yamazaki, K. Oral Administration of Porphyromonas Gingivalis Alters the Gut Microbiome and Serum Metabolome. mSphere 2018, 3, e00460-18. [Google Scholar] [CrossRef] [PubMed]
- Polak, D.; Shapira, L. An Update on the Evidence for Pathogenic Mechanisms That May Link Periodontitis and Diabetes. J. Clin. Periodontol. 2018, 45, 150–166. [Google Scholar] [CrossRef] [PubMed]
- Sanz, M.; Ceriello, A.; Buysschaert, M.; Chapple, I.; Demmer, R.T.; Graziani, F.; Herrera, D.; Jepsen, S.; Lione, L.; Madianos, P.; et al. Scientific Evidence on the Links between Periodontal Diseases and Diabetes: Consensus Report and Guidelines of the JoInt. Workshop on Periodontal Diseases and Diabetes by the International Diabetes Federation and the European Federation of Periodontology. J. Clin. Periodontol. 2018, 45, 138–149. [Google Scholar] [CrossRef] [PubMed]
- Shi, B.; Lux, R.; Klokkevold, P.; Chang, M.; Barnard, E.; Haake, S.; Li, H. The Subgingival Microbiome Associated with Periodontitis in Type 2 Diabetes Mellitus. ISME J. 2020, 14, 519–530. [Google Scholar] [CrossRef]
- Śmiga, M.; Smalley, J.W.; Ślęzak, P.; Brown, J.L.; Siemińska, K.; Jenkins, R.E.; Yates, E.A.; Olczak, T. Glycation of Host Proteins Increases Pathogenic Potential of Porphyromonas Gingivalis. Int. J. Mol. Sci. 2021, 22, 12084. [Google Scholar] [CrossRef]
- Xiao, E.; Mattos, M.; Vieira, G.H.A.; Chen, S.; Corrêa, J.D.; Wu, Y.; Albiero, M.L.; Bittinger, K.; Graves, D.T. Diabetes Enhances IL-17 Expression and Alters the Oral Microbiome to Increase Its Pathogenicity. Cell Host Microbe 2017, 22, 120–128.e4. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.J.; Preshaw, P.M.; Lalla, E. A Review of the Evidence for Pathogenic Mechanisms That May Link Periodontitis and Diabetes. J. Periodontol. 2013, 84, S113–S134. [Google Scholar] [CrossRef]
- Kang, J.; de Brito Bezerra, B.; Pacios, S.; Andriankaja, O.; Li, Y.; Tsiagbe, V.; Schreiner, H.; Fine, D.H.; Graves, D.T. Aggregatibacter Actinomycetemcomitans Infection Enhances Apoptosis in Vivo through a Caspase-3-DepenDent. Mechanism in Experimental Periodontitis. Infect. Immun. 2012, 80, 2247–2256. [Google Scholar] [CrossRef] [PubMed]
- Andriankaja, O.M.; Galicia, J.; Dong, G.; Xiao, W.; Alawi, F.; Graves, D.T. Gene Expression Dynamics during Diabetic Periodontitis. J. Dent. Res. 2012, 91, 1160–1165. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Bal, H.S.; Desta, T.; Krothapalli, N.; Alyassi, M.; Luan, Q.; Graves, D.T. Diabetes Enhances Periodontal Bone Loss through Enhanced Resorption and Diminished Bone Formation. J. Dent. Res. 2006, 85, 510–514. [Google Scholar] [CrossRef] [PubMed]
- Graves, D.T.; Naguib, G.; Lu, H.; Leone, C.; Hsue, H.; Krall, E. Inflammation Is More Persistent in Type 1 Diabetic Mice. J. Dent. Res. 2005, 84, 324–328. [Google Scholar] [CrossRef] [PubMed]
- Naguib, G.; Al-Mashat, H.; Desta, T.; Graves, D.T. Diabetes Prolongs the Inflammatory Response to a Bacterial Stimulus through Cytokine Dysregulation. J. Investig. Dermatol. 2004, 123, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Chiu, H.-C.; Fu, M.M.-J.; Yang, T.-S.; Fu, E.; Chiang, C.-Y.; Tu, H.-P.; Chin, Y.-T.; Lin, F.-G.; Shih, K.-C. Effect of High Glucose, Porphyromonas Gingivalis Lipopolysaccharide and Advanced Glycation End-Products on Production of Interleukin-6/-8 by Gingival Fibroblasts. J. Periodontal Res. 2017, 52, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Bender, O.; Weinberg, E.; Moses, O.; Nemcovsky, C.E.; Weinreb, M. Porphyromonas Gingivalis Lipopolysaccharide and Glycated Serum Albumin Increase the Production of Several Pro-Inflammatory Molecules in Human Gingival Fibroblasts via NFκB. Arch. Oral. Biol. 2020, 116, 104766. [Google Scholar] [CrossRef]
- Hiroshima, Y.; Sakamoto, E.; Yoshida, K.; Abe, K.; Naruishi, K.; Yamamoto, T.; Shinohara, Y.; Kido, J.-I.; Geczy, C.L. Advanced Glycation End-Products and Porphyromonas Gingivalis Lipopolysaccharide Increase Calprotectin Expression in Human Gingival Epithelial Cells. J. Cell BioChem. 2018, 119, 1591–1603. [Google Scholar] [CrossRef]
- Amir, J.; Waite, M.; Tobler, J.; Catalfamo, D.L.; Koutouzis, T.; Katz, J.; Wallet, S.M. The Role of Hyperglycemia in Mechanisms of Exacerbated Inflammatory Responses within the Oral Cavity. Cell Immunol. 2011, 272, 45–52. [Google Scholar] [CrossRef][Green Version]
- Koh, T.J.; DiPietro, L.A. Inflammation and Wound Healing: The Role of the Macrophage. Expert Rev. Mol. Med. 2011, 13, e23. [Google Scholar] [CrossRef]
- Wang, M.; Chen, F.; Wang, J.; Zeng, Z.; Yang, Q.; Shao, S. Th17 and Treg Lymphocytes in Obesity and Type 2 Diabetic Patients. Clin. Immunol. 2018, 197, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Van Dyke, T.E. Pro-Resolving Mediators in the Regulation of Periodontal Disease. Mol. Aspects Med. 2017, 58, 21–36. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N.; Levy, B.D. Resolvins in Inflammation: Emergence of the pro-Resolving Superfamily of Mediators. J. Clin. Investig. 2018, 128, 2657–2669. [Google Scholar] [CrossRef] [PubMed]
- Hasturk, H.; Kantarci, A.; Goguet-Surmenian, E.; Blackwood, A.; Andry, C.; Serhan, C.N.; Van Dyke, T.E. Resolvin E1 Regulates Inflammation at the Cellular and Tissue Level and RestoRes. Tissue Homeostasis in Vivo. J. Immunol. 2007, 179, 7021–7029. [Google Scholar] [CrossRef]
- Sima, C.; Rhourida, K.; Van Dyke, T.E.; Gyurko, R. Type 1 Diabetes Predisposes to Enhanced Gingival Leukocyte Margination and Macromolecule Extravasation in Vivo. J. Periodontal Res. 2010, 45, 748–756. [Google Scholar] [CrossRef]
- Domingueti, C.P.; Dusse, L.M.S.; das Graças Carvalho, M.; de Sousa, L.P.; Gomes, K.B.; Fernandes, A.P. Diabetes Mellitus: The Linkage between Oxidative Stress, Inflammation, Hypercoagulability and Vascular Complications. J. Diabetes Complicat. 2016, 30, 738–745. [Google Scholar] [CrossRef]
- Sun, X.; Mao, Y.; Dai, P.; Li, X.; Gu, W.; Wang, H.; Wu, G.; Ma, J.; Huang, S. Mitochondrial Dysfunction Is Involved in the Aggravation of Periodontitis by Diabetes. J. Clin. Periodontol. 2017, 44, 463–471. [Google Scholar] [CrossRef]
- Kajiura, Y.; Bando, M.; Inagaki, Y.; Nagata, T.; Kido, J. Glycated Albumin and Calprotectin Levels in Gingival Crevicular Fluid from Patients with Periodontitis and Type 2 Diabetes. J. Periodontol. 2014, 85, 1667–1675. [Google Scholar] [CrossRef]
- Akram, Z.; Alqahtani, F.; Alqahtani, M.; Al-Kheraif, A.A.; Javed, F. Levels of Advanced Glycation End Products in Gingival Crevicular Fluid of Chronic Periodontitis Patients with and without Type-2 Diabetes Mellitus. J. Periodontol. 2020, 91, 396–402. [Google Scholar] [CrossRef]
- Zizzi, A.; Tirabassi, G.; Aspriello, S.D.; Piemontese, M.; Rubini, C.; Lucarini, G. Gingival Advanced Glycation End-Products in Diabetes Mellitus-Associated Chronic Periodontitis: An Immunohistochemical Study. J. Periodontal Res. 2013, 48, 293–301. [Google Scholar] [CrossRef]
- Chang, P.-C.; Chien, L.-Y.; Yeo, J.F.; Wang, Y.-P.; Chung, M.-C.; Chong, L.Y.; Kuo, M.Y.-P.; Chen, C.-H.; Chiang, H.-C.; Ng, B.N.; et al. Progression of Periodontal Destruction and the Roles of Advanced Glycation End Products in Experimental Diabetes. J. Periodontol. 2013, 84, 379–388. [Google Scholar] [CrossRef]
- Yi, X.; Zhang, L.; Lu, W.; Tan, X.; Yue, J.; Wang, P.; Xu, W.; Ye, L.; Huang, D. The Effect of NLRP Inflammasome on the Regulation of AGEs-Induced Inflammatory Response in Human Periodontal Ligament Cells. J. Periodontal Res. 2019, 54, 681–689. [Google Scholar] [CrossRef]
- Nonaka, K.; Kajiura, Y.; Bando, M.; Sakamoto, E.; Inagaki, Y.; Lew, J.H.; Naruishi, K.; Ikuta, T.; Yoshida, K.; Kobayashi, T.; et al. Advanced Glycation End-Products Increase IL-6 and ICAM-1 Expression via RAGE, MAPK and NF-ΚB Pathways in Human Gingival Fibroblasts. J. Periodontal Res. 2018, 53, 334–344. [Google Scholar] [CrossRef]
- Lalla, E.; Lamster, I.B.; Feit, M.; Huang, L.; Spessot, A.; Qu, W.; Kislinger, T.; Lu, Y.; Stern, D.M.; Schmidt, A.M. Blockade of RAGE Suppresses Periodontitis-Associated Bone Loss in Diabetic Mice. J. Clin. Investig. 2000, 105, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Tella, E.; Aldahlawi, S.; Eldeeb, A.; El Gazaerly, H. The Effect of Systemic Delivery of Aminoguanidine versus Doxycycline on the Resorptive Phase of Alveolar Bone Following Modified Widman Flap in Diabetic Rats: A Histopathological and Scanning Electron Microscope (SEM) Study. Int. J. Health Sci. 2014, 8, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Du, Z.; Xie, X.; Zhang, Y.; Liu, H.; Zhou, Z.; Zhao, J.; Lee, R.S.; Xiao, Y.; Ivanoviski, S.; et al. Epigenetic Changes Caused by Diabetes and Their Potential Role in the Development of Periodontitis. J. Diabetes Investig. 2021, 12, 1326–1335. [Google Scholar] [CrossRef] [PubMed]
- Hajishengallis, G.; Kajikawa, T.; Hajishengallis, E.; Maekawa, T.; Reis, E.S.; Mastellos, D.C.; Yancopoulou, D.; Hasturk, H.; Lambris, J.D. Complement-DepenDent. Mechanisms and Interventions in Periodontal Disease. Front. Immunol. 2019, 10, 406. [Google Scholar] [CrossRef]
- Gursoy, U.K.; Marakoglu, I.; Oztop, A.Y. Relationship between Neutrophil Functions and Severity of Periodontitis in Obese and/or Type 2 Diabetic Chronic Periodontitis Patients. Quintessence Int. 2008, 39, 485–489. [Google Scholar] [PubMed]
- Manosudprasit, A.; Kantarci, A.; Hasturk, H.; Stephens, D.; Van Dyke, T.E. Spontaneous PMN Apoptosis in Type 2 Diabetes and the Impact of Periodontitis. J. Leukoc. Biol. 2017, 102, 1431–1440. [Google Scholar] [CrossRef]
- Marhoffer, W.; Stein, M.; Schleinkofer, L.; Federlin, K. Evidence of Ex Vivo and in Vitro Impaired Neutrophil Oxidative Burst and Phagocytic Capacity in Type 1 Diabetes Mellitus. Diabetes Res. Clin. Pract. 1993, 19, 183–188. [Google Scholar] [CrossRef]
- Wong, S.L.; Wagner, D.D. Peptidylarginine Deiminase 4: A Nuclear Button Triggering Neutrophil Extracellular Traps in Inflammatory Diseases and Aging. FASEB J. 2018, 32, fj201800691R. [Google Scholar] [CrossRef]
- Wang, J.; Zhou, Y.; Ren, B.; Zou, L.; He, B.; Li, M. The Role of Neutrophil Extracellular Traps in Periodontitis. Front. Cell Infect. MicroBiol. 2021, 11, 639144. [Google Scholar] [CrossRef] [PubMed]
- Miao, Y.; He, L.; Qi, X.; Lin, X. Injecting Immunosuppressive M2 Macrophages Alleviates the Symptoms of Periodontitis in Mice. Front. Mol. BioSci. 2020, 7, 603817. [Google Scholar] [CrossRef] [PubMed]
- Russo, S.; Kwiatkowski, M.; Govorukhina, N.; Bischoff, R.; Melgert, B.N. Meta-Inflammation and Metabolic Reprogramming of Macrophages in Diabetes and Obesity: The Importance of Metabolites. Front. Immunol. 2021, 12, 746151. [Google Scholar] [CrossRef] [PubMed]
- Stienstra, R.; Netea-Maier, R.T.; Riksen, N.P.; Joosten, L.A.B.; Netea, M.G. Specific and Complex Reprogramming of Cellular Metabolism in Myeloid Cells during Innate Immune Responses. Cell Metab. 2017, 26, 142–156. [Google Scholar] [CrossRef] [PubMed]
- Flynn, M.C.; Kraakman, M.J.; Tikellis, C.; Lee, M.K.S.; Hanssen, N.M.J.; Kammoun, H.L.; Pickering, R.J.; Dragoljevic, D.; Al-Sharea, A.; Barrett, T.J.; et al. Transient Intermittent Hyperglycemia Accelerates Atherosclerosis by Promoting Myelopoiesis. Circ. Res. 2020, 127, 877–892. [Google Scholar] [CrossRef]
- Huang, N.; Dong, H.; Luo, Y.; Shao, B. Th17 Cells in Periodontitis and Its Regulation by A20. Front. Immunol. 2021, 12, 742925. [Google Scholar] [CrossRef]
- Ramamurthy, N.S.; Golub, L.M. Diabetes Increases Collagenase Activity in Extracts of Rat Gingiva and Skin. J. Periodontal Res. 1983, 18, 23–30. [Google Scholar] [CrossRef]
- Sasaki, T.; Ramamurthy, N.S.; Yu, Z.; Golub, L.M. Tetracycline Administration Increases Protein (Presumably Procollagen) Synthesis and Secretion in Periodontal Ligament Fibroblasts of Streptozotocin-Induced Diabetic Rats. J. Periodontal Res. 1992, 27, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Balci Yuce, H.; Karatas, Ö.; Tulu, F.; Altan, A.; Gevrek, F. Effect of Diabetes on Collagen Metabolism and Hypoxia in Human Gingival Tissue: A Stereological, Histopathological, and Immunohistochemical Study. Biotech. HistoChem. 2019, 94, 65–73. [Google Scholar] [CrossRef]
- Ren, L.; Fu, Y.; Deng, Y.; Qi, L.; Jin, L. Advanced Glycation End Products Inhibit the Expression of Collagens Type I and III by Human Gingival Fibroblasts. J. Periodontol. 2009, 80, 1166–1173. [Google Scholar] [CrossRef] [PubMed]
- Zhan, D.; Guo, L.; Zheng, L. Inhibition of the Receptor for Advanced Glycation Promotes Proliferation and Repair of Human Periodontal Ligament Fibroblasts in Response to High Glucose via the NF-ΚB Signaling Pathway. Arch. Oral. Biol. 2018, 87, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Checchi, V.; Maravic, T.; Bellini, P.; Generali, L.; Consolo, U.; Breschi, L.; Mazzoni, A. The Role of Matrix Metalloproteinases in Periodontal Disease. Int. J. Environ. Res. Public Health 2020, 17, 4923. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.S.; Vamsi, G.; Sripriya, R.; Sehgal, P.K. Expression of Matrix Metalloproteinases (MMP-8 and -9) in Chronic Periodontitis Patients with and without Diabetes Mellitus. J. Periodontol. 2006, 77, 1803–1808. [Google Scholar] [CrossRef] [PubMed]
- Santos, V.R.; Lima, J.A.; Gonçalves, T.E.D.; Bastos, M.F.; Figueiredo, L.C.; Shibli, J.A.; Duarte, P.M. Receptor Activator of Nuclear Factor-Kappa B Ligand/Osteoprotegerin Ratio in Sites of Chronic Periodontitis of Subjects with Poorly and Well-Controlled Type 2 Diabetes. J. Periodontol. 2010, 81, 1455–1465. [Google Scholar] [CrossRef] [PubMed]
- Graves, D.T.; Alshabab, A.; Albiero, M.L.; Mattos, M.; Corrêa, J.D.; Chen, S.; Yang, Y. Osteocytes Play an Important Role in Experimental Periodontitis in Healthy and Diabetic Mice through Expression of RANKL. J. Clin. Periodontol. 2018, 45, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Pacios, S.; Xiao, W.; Mattos, M.; Lim, J.; Tarapore, R.S.; Alsadun, S.; Yu, B.; Wang, C.-Y.; Graves, D.T. Osteoblast Lineage Cells Play an Essential Role in Periodontal Bone Loss Through Activation of Nuclear Factor-Kappa B. Sci. Rep. 2015, 5, 16694. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Flegler, A.; Kozlov, A.; Stern, P.H. Direct Inhibitory and Indirect Stimulatory Effects of RAGE Ligand S100 on SRANKL-Induced Osteoclastogenesis. J. Cell BioChem. 2009, 107, 917–925. [Google Scholar] [CrossRef]
- Kim, J.-H.; Kim, A.R.; Choi, Y.H.; Jang, S.; Woo, G.-H.; Cha, J.-H.; Bak, E.-J.; Yoo, Y.-J. Tumor Necrosis Factor-α Antagonist Diminishes Osteocytic RANKL and Sclerostin Expression in Diabetes Rats with Periodontitis. PLoS ONE 2017, 12, e0189702. [Google Scholar] [CrossRef]
- Fu, Y.-W.; He, H.-B. Apoptosis of Periodontium Cells in Streptozototocin- and Ligature-Induced Experimental Diabetic Periodontitis in Rats. Acta Odontol. Scand. 2013, 71, 1206–1215. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Bal, H.S.; Desta, T.; Behl, Y.; Graves, D.T. Tumor Necrosis Factor-Alpha Mediates Diabetes-Enhanced Apoptosis of Matrix-Producing Cells and Impairs Diabetic Healing. Am. J. Pathol. 2006, 168, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Mei, Y.-M.; Li, L.; Wang, X.-Q.; Zhang, M.; Zhu, L.-F.; Fu, Y.-W.; Xu, Y. AGEs Induces Apoptosis and Autophagy via Reactive Oxygen Species in Human Periodontal Ligament Cells. J. Cell BioChem. 2019, 121, 3764–3779. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Sun, Y.; Liu, H.; Wang, W.; Wang, J.; Zhang, F. Selective Adipogenic Differentiation of Human Periodontal Ligament Stem Cells Stimulated with High Doses of Glucose. PLoS ONE 2018, 13, e0199603. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Taguchi, Y.; Tominaga, K.; Kimura, D.; Yamawaki, I.; Noguchi, M.; Yamauchi, N.; Tamura, I.; Tanaka, A.; Umeda, M. High Glucose Concentrations Suppress the Proliferation of Human Periodontal Ligament Stem Cells and Their Differentiation Into Osteoblasts. J. Periodontol. 2016, 87, e44–e51. [Google Scholar] [CrossRef]
- Netea, M.G.; Domínguez-Andrés, J.; Barreiro, L.B.; Chavakis, T.; Divangahi, M.; Fuchs, E.; Joosten, L.A.B.; van der Meer, J.W.M.; Mhlanga, M.M.; Mulder, W.J.M.; et al. Defining Trained Immunity and Its Role in Health and Disease. Nat. Rev. Immunol. 2020, 20, 375–388. [Google Scholar] [CrossRef] [PubMed]
- Penkov, S.; Mitroulis, I.; Hajishengallis, G.; Chavakis, T. Immunometabolic Crosstalk: An Ancestral Principle of Trained Immunity? Trends Immunol. 2019, 40, 1–11. [Google Scholar] [CrossRef]
- Patel, A.A.; Zhang, Y.; Fullerton, J.N.; Boelen, L.; Rongvaux, A.; Maini, A.A.; Bigley, V.; Flavell, R.A.; Gilroy, D.W.; Asquith, B.; et al. The Fate and Lifespan of Human Monocyte Subsets in Steady State and Systemic Inflammation. J. Exp. Med. 2017, 214, 1913–1923. [Google Scholar] [CrossRef]
- Chavakis, T.; Mitroulis, I.; Hajishengallis, G. Hematopoietic Progenitor Cells as Integrative Hubs for Adaptation to and Fine-Tuning of Inflammation. Nat. Immunol. 2019, 20, 802–811. [Google Scholar] [CrossRef]
- Mitroulis, I.; Ruppova, K.; Wang, B.; Chen, L.-S.; Grzybek, M.; Grinenko, T.; Eugster, A.; Troullinaki, M.; Palladini, A.; Kourtzelis, I.; et al. Modulation of Myelopoiesis Progenitors Is an Integral Component of Trained Immunity. Cell 2018, 172, 147–161. [Google Scholar] [CrossRef]
- Kaufmann, E.; Sanz, J.; Dunn, J.L.; Khan, N.; Mendonça, L.E.; Pacis, A.; Tzelepis, F.; Pernet, E.; Dumaine, A.; Grenier, J.-C.; et al. BCG Educates Hematopoietic Stem Cells to Generate Protective Innate Immunity against Tuberculosis. Cell 2018, 172, 176–190. [Google Scholar] [CrossRef] [PubMed]
- Donohoe, D.R.; Bultman, S.J. Metaboloepigenetics: Interrelationships between Energy Metabolism and Epigenetic Control of Gene Expression. J. Cell Physiol. 2012, 227, 3169–3177. [Google Scholar] [CrossRef] [PubMed]
- Cheng, S.-C.; Quintin, J.; Cramer, R.A.; Shepardson, K.M.; Saeed, S.; Kumar, V.; Giamarellos-Bourboulis, E.J.; Martens, J.H.A.; Rao, N.A.; Aghajanirefah, A.; et al. MTOR- and HIF-1α-Mediated Aerobic Glycolysis as Metabolic Basis for Trained Immunity. Science 2014, 345, 1250684. [Google Scholar] [CrossRef] [PubMed]
- Saeed, S.; Quintin, J.; Kerstens, H.H.D.; Rao, N.A.; Aghajanirefah, A.; Matarese, F.; Cheng, S.-C.; Ratter, J.; Berentsen, K.; van der Ent, M.A.; et al. Epigenetic Programming of Monocyte-to-Macrophage Differentiation and Trained Innate Immunity. Science 2014, 345, 1251086. [Google Scholar] [CrossRef]
- Bekkering, S.; Arts, R.J.W.; Novakovic, B.; Kourtzelis, I.; van der Heijden, C.D.C.C.; Li, Y.; Popa, C.D.; Ter Horst, R.; van Tuijl, J.; Netea-Maier, R.T.; et al. Metabolic Induction of Trained Immunity through the Mevalonate Pathway. Cell 2018, 172, 135–146.e9. [Google Scholar] [CrossRef]
- Ishai, A.; Osborne, M.T.; El Kholy, K.; Takx, R.A.P.; Ali, A.; Yuan, N.; Hsue, P.; Van Dyke, T.E.; Tawakol, A. Periodontal Disease Associates With Arterial Inflammation Via Potentiation of a Hematopoietic-Arterial Axis. JACC Cardiovasc. Imaging 2019, 12, 2271–2273. [Google Scholar] [CrossRef]
- Wright, H.J.; Matthews, J.B.; Chapple, I.L.C.; Ling-Mountford, N.; Cooper, P.R. Periodontitis Associates with a Type 1 IFN Signature in Peripheral Blood Neutrophils. J. Immunol. 2008, 181, 5775–5784. [Google Scholar] [CrossRef]
- Ling, M.R.; Chapple, I.L.C.; Matthews, J.B. Peripheral Blood Neutrophil Cytokine Hyper-Reactivity in Chronic Periodontitis. Innate Immun. 2015, 21, 714–725. [Google Scholar] [CrossRef]
- Arts, R.J.W.; Joosten, L.A.B.; Netea, M.G. Immunometabolic Circuits in Trained Immunity. Semin. Immunol. 2016, 28, 425–430. [Google Scholar] [CrossRef]
- Christ, A.; Günther, P.; Lauterbach, M.A.R.; Duewell, P.; Biswas, D.; Pelka, K.; Scholz, C.J.; Oosting, M.; Haendler, K.; Baßler, K.; et al. Western Diet Triggers NLRP3-DepenDent. Innate Immune Reprogramming. Cell 2018, 172, 162–175.e14. [Google Scholar] [CrossRef]
- Alrdahe, S.; Al Sadoun, H.; Torbica, T.; McKenzie, E.A.; Bowling, F.L.; Boulton, A.J.M.; Mace, K.A. Dysregulation of Macrophage Development and Phenotype in Diabetic Human Macrophages Can Be Rescued by Hoxa3 Protein Transduction. PLoS ONE 2019, 14, e0223980. [Google Scholar] [CrossRef] [PubMed]
- Edgar, L.; Akbar, N.; Braithwaite, A.T.; Krausgruber, T.; Gallart-Ayala, H.; Bailey, J.; Corbin, A.L.; Khoyratty, T.E.; Chai, J.T.; Alkhalil, M.; et al. Hyperglycemia Induces Trained Immunity in Macrophages and Their Precursors and Promotes Atherosclerosis. Circulation 2021, 144, 961–982. [Google Scholar] [CrossRef] [PubMed]
- Ayala, T.S.; Tessaro, F.H.G.; Jannuzzi, G.P.; Bella, L.M.; Ferreira, K.S.; Martins, J.O. High Glucose Environments Interfere with Bone Marrow-Derived Macrophage Inflammatory Mediator Release, the TLR4 Pathway and Glucose Metabolism. Sci. Rep. 2019, 9, 11447. [Google Scholar] [CrossRef]
- Choudhury, R.P.; Edgar, L.; Rydén, M.; Fisher, E.A. Diabetes and Metabolic Drivers of Trained Immunity: New Therapeutic Targets Beyond Glucose. Arterioscler Thromb Vasc Biol. 2021, 41, 1284–1290. [Google Scholar] [CrossRef] [PubMed]
- Araújo, A.A.; Pereira, A.D.; Medeiros, C.A.; Brito, G.A.; Leitão, R.F.; Araújo, L.D.; Guedes, P.M.; Hiyari, S.; Pirih, F.Q.; Araújo Júnior, R.F. Effects of Metformin on Inflammation, Oxidative Stress, and Bone Loss in a Rat Model of Periodontitis. PLoS ONE 2017, 12, e0183506. [Google Scholar] [CrossRef] [PubMed]
- Frantzis, T.G.; Reeve, C.M.; Brown, A.L. The Ultrastructure of Capillary Basement Membranes in the Attached Gingiva of Diabetic and Nondiabetic Patients with Periodontal Disease. J. Periodontol. 1971, 42, 406–411. [Google Scholar] [CrossRef]
- Scardina, G.A.; Cacioppo, A.; Messina, P. Periodontal Microcirculation in Diabetics: An in Vivo Non-Invasive Analysis by Means of Videocapillaroscopy. Med. Sci. Monit. 2012, 18, CR58–CR64. [Google Scholar] [CrossRef] [PubMed]
- Develioglu, H.; Özdemir, H.; Bostanci, V. Comparative Analysis of the Blood Flow Values of Patients with Type 2 Diabetes Mellitus Presenting with Chronic Periodontitis, Patients with Chronic Periodontitis Only and Healthy Individuals. West Indian Med. J. 2014, 63, 359–363. [Google Scholar] [CrossRef][Green Version]
- Scardina, G.; Citarrella, R.; Messina, P. Diabetic Microagiopathy of Oral Mucosa Depends on Disease Duration and Therapy. Med. Sci. Monit. 2017, 23, 5613–5619. [Google Scholar] [CrossRef]
- Yasuda, K.; Uemura, M.; Suwa, F. Morphological Study of the Palatal Gingiva of the Maxillary First Molar in the Type 2 Diabetes Mellitus Model Rat. Okajimas Folia ANat. Jpn 2011, 88, 65–74. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nishikawa, T.; Suzuki, Y.; Sawada, N.; Kobayashi, Y.; Nakamura, N.; Miyabe, M.; Miyajima, S.-I.; Adachi, K.; Minato, T.; Mizutani, M.; et al. Therapeutic Potential for Insulin on Type 1 Diabetes-Associated Periodontitis: Analysis of Experimental Periodontitis in Streptozotocin-Induced Diabetic Rats. J. Diabetes Investig. 2020, 11, 1482–1489. [Google Scholar] [CrossRef] [PubMed]
- Seppälä, B.; Sorsa, T.; Ainamo, J. Morphometric Analysis of Cellular and Vascular Changes in Gingival Connective Tissue in Long-Term Insulin-DepenDent. Diabetes. J. Periodontol. 1997, 68, 1237–1245. [Google Scholar] [CrossRef] [PubMed]
- Nardi, G.M.; Ferrara, E.; Converti, I.; Cesarano, F.; Scacco, S.; Grassi, R.; Gnoni, A.; Grassi, F.R.; Rapone, B. Does Diabetes Induce the Vascular Endothelial Growth Factor (VEGF) Expression in Periodontal Tissues? A Systematic Review. Int. J. Environ. Res. Public Health 2020, 17, 2765. [Google Scholar] [CrossRef]
- Needleman, I.; Garcia, R.; Gkranias, N.; Kirkwood, K.L.; Kocher, T.; Iorio, A.D.; Moreno, F.; Petrie, A. Mean Annual Attachment, Bone Level, and Tooth Loss: A Systematic Review. J. Clin. Periodontol. 2018, 45 (Suppl. 20), S112–S129. [Google Scholar] [CrossRef]
- Dietrich, T.; Ower, P.; Tank, M.; West, N.X.; Walter, C.; Needleman, I.; Hughes, F.J.; Wadia, R.; Milward, M.R.; Hodge, P.J.; et al. Periodontal Diagnosis in the Context of the 2017 Classification System of Periodontal Diseases and Conditions—Implementation in Clinical Practice. Br. Dent. J. 2019, 226, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Estrich, C.G.; Araujo, M.W.B.; Lipman, R.D. Prediabetes and Diabetes Screening in Dental Care Settings: NHANES 2013 to 2016. JDR Clin. Trans. Res. 2019, 4, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Eke, P.I.; Dye, B. Assessment of Self-Report MeasuRes. for Predicting Population Prevalence of Periodontitis. J. Periodontol. 2009, 80, 1371–1379. [Google Scholar] [CrossRef]
- Boyko, E.J.; Fujimoto, W.Y.; Leonetti, D.L.; Newell-Morris, L. Visceral Adiposity and Risk of Type 2 Diabetes: A Prospective Study among Japanese Americans. Diabetes Care 2000, 23, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Wernicke, K.; Grischke, J.; Stiesch, M.; Zeissler, S.; Krüger, K.; Bauer, P.; Hillebrecht, A.; Eberhard, J. Influence of Physical Activity on Periodontal Health in Patients with Type 2 Diabetes Mellitus. A Blinded, Randomized, Controlled Trial. Clin. Oral. Investig. 2021, 25, 6101–6107. [Google Scholar] [CrossRef]
- Zhang, Y.; He, J.; He, B.; Huang, R.; Li, M. Effect of Tobacco on Periodontal Disease and Oral Cancer. Tob Induc Dis. 2019, 17, 40. [Google Scholar] [CrossRef] [PubMed]
- Bibars, A.R.M.; Obeidat, S.R.; Khader, Y.; Mahasneh, A.M.; Khabour, O.F. The Effect of Waterpipe Smoking on Periodontal Health. Oral. Health PRev. Dent. 2015, 13, 253–259. [Google Scholar] [CrossRef]
- Kulkarni, V.; Uttamani, J.R.; Bhatavadekar, N.B. Comparison of Clinical Periodontal Status among Habitual Smokeless-Tobacco Users and Cigarette Smokers. Int. Dent. J. 2016, 66, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Giovannoni, M.L.; Valdivia-Gandur, I.; Lozano de Luaces, V.; Varela Véliz, H.; Balasubbaiah, Y.; Chimenos-Küstner, E. Betel and Tobacco Chewing Habit and Its Relation to Risk Factors for Periodontal Disease. Oral. Dis. 2018, 24, 829–839. [Google Scholar] [CrossRef]
- Geisinger, M.L.; Geurs, N.C.; Ogdon, D.; Reddy, M.S. Commentary: Targeting Underlying Biologic Mechanisms in Selecting Adjunctive Therapies to Improve Periodontal Treatment in Smokers: A Commentary. J. Periodontol. 2017, 88, 703–710. [Google Scholar] [CrossRef] [PubMed]
- Kotsakis, G.A.; Javed, F.; Hinrichs, J.E.; Karoussis, I.K.; Romanos, G.E. Impact of Cigarette Smoking on Clinical Outcomes of Periodontal Flap Surgical Procedures: A Systematic Review and Meta-Analysis. J. Periodontol. 2015, 86, 254–263. [Google Scholar] [CrossRef] [PubMed]
- Bunaes, D.F.; Lie, S.A.; Enersen, M.; Aastrøm, A.N.; Mustafa, K.; Leknes, K.N. Site-Specific Treatment Outcome in Smokers Following Non-Surgical and Surgical Periodontal Therapy. J. Clin. Periodontol. 2015, 42, 933–942. [Google Scholar] [CrossRef]
- Papantonopoulos, G.H. Smoking Influences Decision Making in Periodontal Therapy: A Retrospective Clinical Study. J. Periodontol. 1999, 70, 1166–1173. [Google Scholar] [CrossRef] [PubMed]
- Silva, H. Tobacco Use and Periodontal Disease-The Role of Microvascular Dysfunction. Biology 2021, 10, 441. [Google Scholar] [CrossRef]
- Obradović, R.; Kesić, L.J.; Gasić, J.; Petrović, M.; Zivković, N. Role of Smoking in Periodontal Disease among Diabetic Patients. West Indian Med. J. 2012, 61, 98–101. [Google Scholar]
- Hajishengallis, G.; Chavakis, T.; Lambris, J.D. Current Understanding of Periodontal Disease Pathogenesis and Targets for Host-Modulation Therapy. Periodontology 2000 2020, 84, 14–34. [Google Scholar] [CrossRef]
- Hasturk, H.; Schulte, F.; Martins, M.; Sherzai, H.; Floros, C.; Cugini, M.; Chiu, C.-J.; Hardt, M.; Van Dyke, T. Safety and Preliminary Efficacy of a Novel Host-Modulatory Therapy for Reducing Gingival Inflammation. Front. Immunol. 2021, 12, 704163. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barutta, F.; Bellini, S.; Durazzo, M.; Gruden, G. Novel Insight into the Mechanisms of the Bidirectional Relationship between Diabetes and Periodontitis. Biomedicines 2022, 10, 178. https://doi.org/10.3390/biomedicines10010178
Barutta F, Bellini S, Durazzo M, Gruden G. Novel Insight into the Mechanisms of the Bidirectional Relationship between Diabetes and Periodontitis. Biomedicines. 2022; 10(1):178. https://doi.org/10.3390/biomedicines10010178
Chicago/Turabian StyleBarutta, Federica, Stefania Bellini, Marilena Durazzo, and Gabriella Gruden. 2022. "Novel Insight into the Mechanisms of the Bidirectional Relationship between Diabetes and Periodontitis" Biomedicines 10, no. 1: 178. https://doi.org/10.3390/biomedicines10010178
APA StyleBarutta, F., Bellini, S., Durazzo, M., & Gruden, G. (2022). Novel Insight into the Mechanisms of the Bidirectional Relationship between Diabetes and Periodontitis. Biomedicines, 10(1), 178. https://doi.org/10.3390/biomedicines10010178






