Sensing Atrazine Herbicide Degradation Products through Their Interactions with Humic Substances by Surface-Enhanced Raman Scattering
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Preparation of Silver Nanoparticles (AgNPs)
2.3. Sample Preparation
2.4. UV-Vis Spectra
2.5. Raman and SERS Sample Preparations
2.6. Quantitative Analysis
2.7. Theoretical Raman Calculations
3. Results and Discussion
3.1. Morphology of AgNPs: Extinction Spectra and TEM
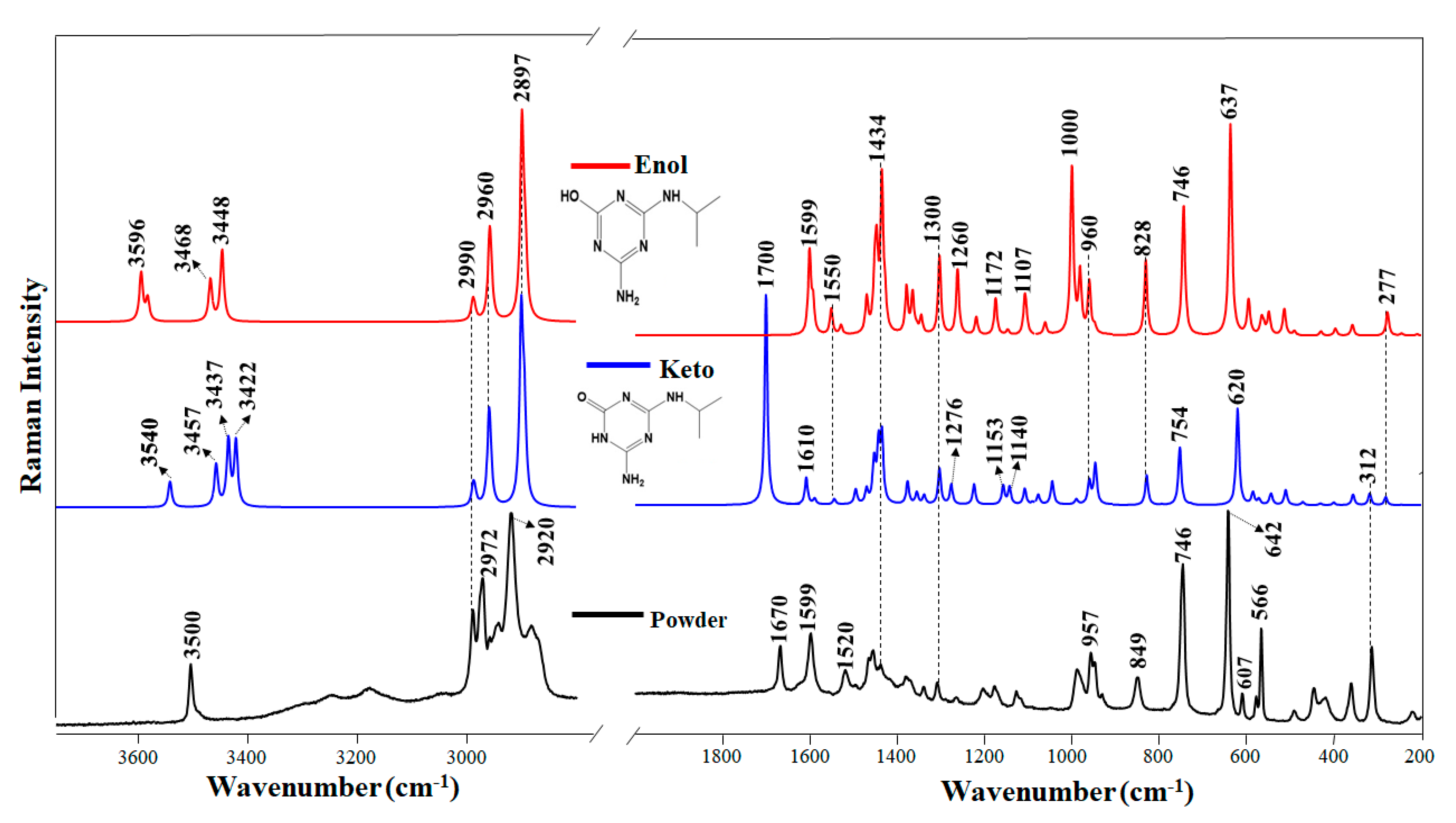
3.2. Experimental and Theoretical Raman Spectra of DEHA
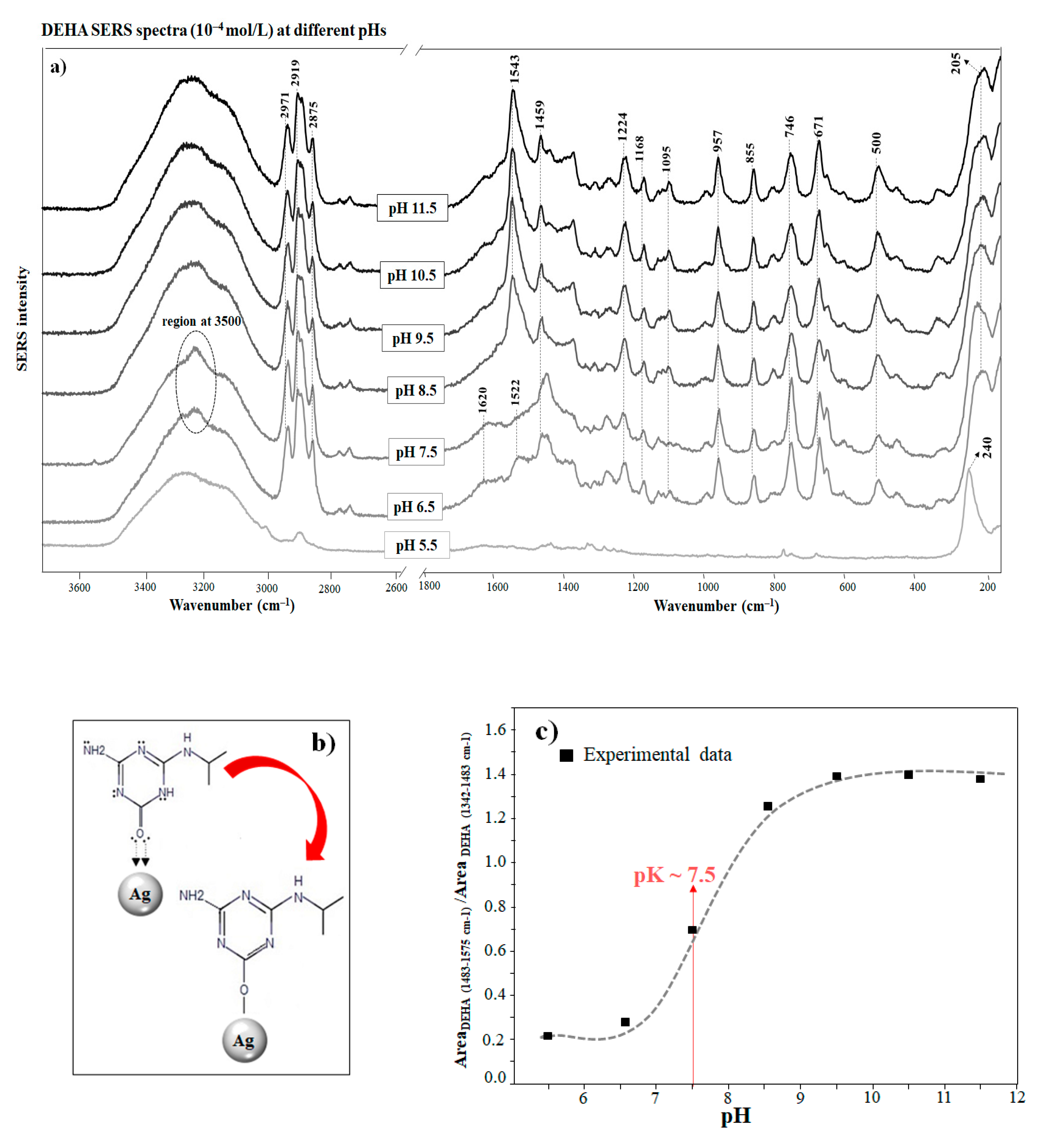
3.3. SERS Spectra of DEHA at Different pHs
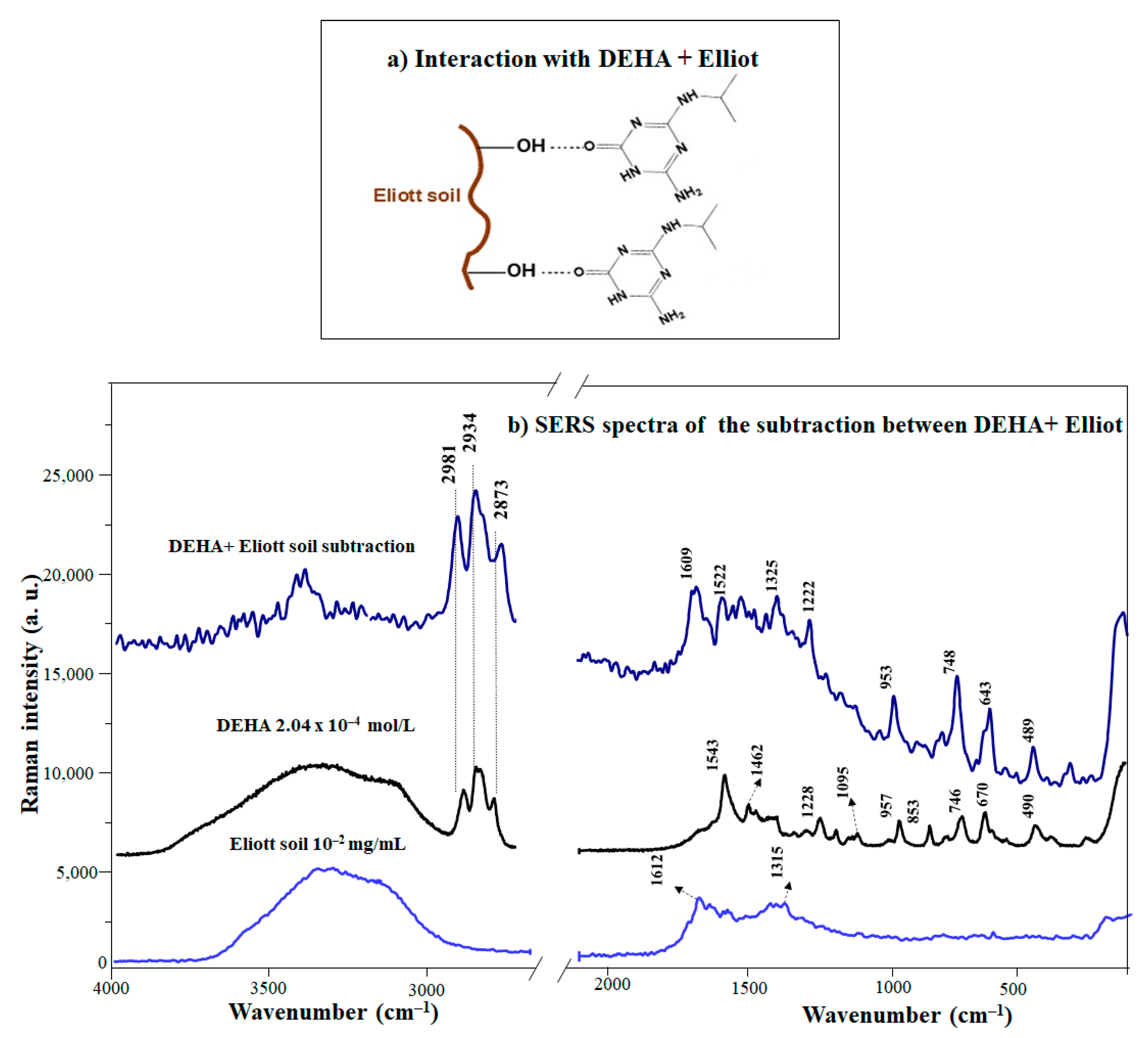
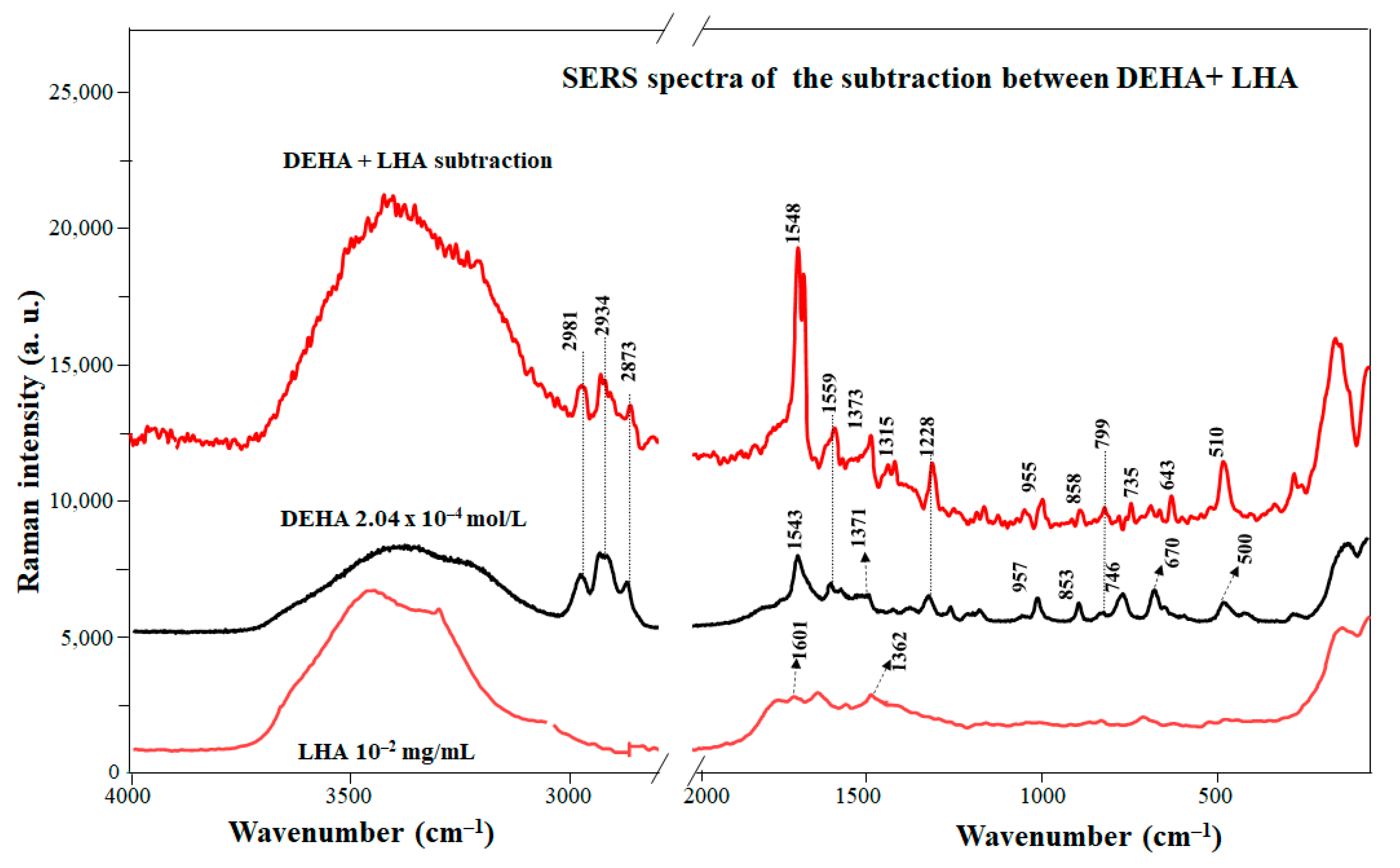
3.4. Interaction of DEHA with Humic Substances
3.5. Quantitative Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Barceló, D. Occurrence, handling and chromatographic determination of pesticides in the aquatic environment. A review. Analyst 1991, 116, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Yaseen, T.; Pu, H.; Sun, D.-W. Functionalization techniques for improving SERS substrates and their applications in food safety evaluation: A review of recent research trends. Trends Food Sci. Technol. 2018, 72, 162–174. [Google Scholar] [CrossRef]
- Rehan, M. Microbial biodegradation of S-triazine herbicides in soil. JCRF 2016, 1, 1–3. [Google Scholar] [CrossRef]
- Mandelbaum, R.T.; Wackett, L.P.; Allan, D.L. Mineralization of the S-triazine by stable bacterial mixed cultures. Appl. Environ. Microbiol. 1993, 59, 1695–1701. [Google Scholar] [CrossRef] [PubMed]
- Almeida Lage, A.L.; Ribeiro, J.M.; de Souza-Fagundes, E.M.; Brugnera, M.F.; Martins, D.C.d.S. Efficient atrazine degradation catalyzed by manganese porphyrins: Determination of atrazine degradation products and their toxicity evaluation by human blood cells test models. J. Hazard. Mater. 2019, 378, 120748. [Google Scholar] [CrossRef] [PubMed]
- Binet, F.; Kersanté, A.; Munier-Lamy, C.; Le Bayon, R.-C.; Belgy, M.-J.; Shipitalo, M.J. Lumbricid macrofauna alter atrazine mineralization and sorption in a silt loam soil. Soil Biol. Biochem. 2006, 38, 1255–1263. [Google Scholar] [CrossRef]
- Ben-Hur, M.; Letey, J.; Farmer, W.J.; Williams, C.F.; Nelson, S.D. Soluble and solid organic matter effects on atrazine adsorption in cultivated soils. Soil Sci. Soc. Am. J. 2003, 67, 1140–1146. [Google Scholar] [CrossRef]
- Piccolo, A.; Conte, P.; Scheunert, I.; Paci, M. Atrazine interactions with soil humic substances of different molecular structure. J. Environ. Qual. 1998, 27, 1324–1333. [Google Scholar] [CrossRef]
- Martin-Neto, L.; Traghetta, D.G.; Vaz, C.M.P.; Crestana, S.; Sposito, G. On the interaction mechanisms of atrazine and hydroxyatrazine with humic substances. J. Environ. Qual. 2001, 30, 520–525. [Google Scholar] [CrossRef] [PubMed]
- Abate, G.; Penteado, J.C.; Cuzzi, J.D.; Vitti, G.C.; Lichtig, J.; Masini, J.C. Influence of Humic acid on adsorption and desorption of atrazine, hydroxyatrazine, deethylatrazine, and deisopropylatrazine onto a clay-rich soil sample. J. Agric. Food Chem. 2004, 52, 6747–6754. [Google Scholar] [CrossRef]
- Tao, Q.-H.; Tang, H.-X. Effect of dye compounds on the adsorption of atrazine by natural sediment. Chemosphere 2004, 56, 31–38. [Google Scholar] [CrossRef]
- Celano, G.; Šmejkalová, D.; Spaccini, R.; Piccolo, A. Interactions of three s-triazines with humic acids of different structure. J. Agric. Food Chem. 2008, 56, 7360–7366. [Google Scholar] [CrossRef] [PubMed]
- Mulbry, W.W. Purification and characterization of an inducible s-triazine hydrolase from Rhodococcus corallinus NRRL B-15444R. Appl. Environ. Microbiol. 1994, 60, 613–618. [Google Scholar] [CrossRef]
- Lin, C.H.; Lerch, R.N.; Garrett, H.E.; George, M.F. Bioremediation of Atrazine-contaminated soil by forage grasses: Transformation, uptake, and detoxification. J. Environ. Qual. 2008, 37, 196–206. [Google Scholar] [CrossRef]
- Lovley, D.R.; Coates, J.D.; Blunt-Harris, E.L.; Phillips, E.J.P.; Woodward, J.C. Humic substances as electron acceptors for microbial respiration. Nature 1996, 382, 445–448. [Google Scholar] [CrossRef]
- Olu-Owolabi, B.I.; Diagboya, P.N.; Okoli, C.P.; Adebowale, K.O. Sorption behaviour of pentachlorophenol in sub-Saharan tropical soils: Soil types sorption dynamics. Environ. Earth Sci. 2016, 75, 1494. [Google Scholar] [CrossRef]
- Barrett, M.R. The Environmental Impact of Pesticide Degradates in Groundwater. In ACS Symposium Series; Meyer, M.T., Thurman, E.M., Eds.; American Chemical Society: Washington, DC, USA, 1996; Volume 630, pp. 200–225. [Google Scholar] [CrossRef]
- LeBaron, H.M.; McFarland, J.E.; Burnside, O.C.; Farland, M.; Burnside, O. The Triazine Herbicides: A Milestone in the Development of Weed Control Technology. In The Triazine Herbicides: 50 Years Revolutionizing Agriculture, 1st ed.; LeBaron, H.M., McFarland, J.E., Burnside, O.C., Eds.; Elsevier: San Diego, CA, USA, 2008; pp. 1–12. [Google Scholar]
- Steelink, C.; Tollin, G. Free radicals in soil. In Soil Biochemistry; McLaren, A.D., Peterson, G.M., Eds.; Dekker: New York, NY, USA, 1967; pp. 147–169. [Google Scholar]
- Crosby, D.G. Nonbiological degradation of herbicides in the soil. In Herbicides: Physiology, Biochemistry, and Ecology; Audus, L.J., Ed.; Academic Press: New York, NY, USA, 1976; Volume 2, pp. 65–97. [Google Scholar]
- Laird, D.A.; Koskinen, W. Triazine Soil Interactions. The Triazine Herbicides: 50 Years Revolutionizing Agriculture, 1st ed.; LeBaron, H.M., McFarland, J.E., Burnside, O.C., Eds.; Elsevier: San Diego, CA, USA, 2008; pp. 275–299. [Google Scholar]
- Oliveira, M.J.S.; Rubira, R.J.G.; Furini, L.N.; Batagin-Neto, A.; Constantino, C.J.L. Detection of thiabendazole fungicide/parasiticide by SERS: Quantitative analysis and adsorption mechanism. Appl. Surf. Sci. 2020, 517, 145786. [Google Scholar] [CrossRef]
- Mandelbaum, R.T.; Wackett, L.P.; Allan, D.L. Rapid hydrolysis of atriazine to hydroxyatrazine by soil bacteria. Environ. Sci. Technol. 1993, 27, 1943–1946. [Google Scholar] [CrossRef]
- Rubira, R.J.G.; Constantino, C.J.L.; Otero, J.C.; Sanchez-Cortes, S. Abiotic degradation of s-triazine pesticides analyzed by surface-enhanced Raman scattering. J. Raman Spectrosc. 2020, 51, 264–273. [Google Scholar] [CrossRef]
- Klementova, S.; Piskova, V. UV Photodegradation of triazines atrazine, simazine and propazine and of atrazine metabolites desethylatrazine and desisopropylatrazine. Res. J. Chem. Environ. 2005, 4, 20–23. [Google Scholar]
- Calderbank, A. The occurrence and significance of bound pesticide residues in soil. Rev. Environ. Contam. Toxicol. 1989, 108, 71–103. [Google Scholar] [CrossRef]
- Johnson, R.M.; Halaweish, F.; Furmann, J.J. Analysis of atrazine and associated metabolites by reverse-phase high performance thin layer chromatography. J. Liq. Chromatogr. 1992, 15, 2941–2957. [Google Scholar] [CrossRef]
- Aroca, R. Surface-Enhanced Vibrational Spectroscopy; John Wiley & Sons, Ltd.: Chichester, UK, 2006. [Google Scholar]
- Moskovits, M. Surface-enhanced spectroscopy. Rev. Mod. Phys. 1985, 57, 783–826. [Google Scholar] [CrossRef]
- Wokaun, A. Surface enhancement of optical fields: Mechanism and applications. Mol. Phys. 1985, 56, 1–33. [Google Scholar] [CrossRef]
- Guerrini, L.; Campos-Vallette, M.; Domingo, C.; Garcia-Ramos, J.V.; Sanchez-Cortes, S.L.P. Detection of persistent organic pollutants using SERS sensors based on organically functionalized Ag nanoparticles. In Surface Enhanced Raman Spectroscopy; Schlücker, S., Ed.; Wiley: Hoboken, NJ, USA, 2010; pp. 103–128. [Google Scholar]
- Cañamares, M.V.; Garcia-Ramos, J.V.; Gómez-Varga, J.D.; Domingo, C.; Sanchez-Cortes, S. Comparative Study of the Morphology, Aggregation, Adherence to Glass, and Surface-Enhanced Raman Scattering Activity of Silver Nanoparticles Prepared by Chemical Reduction of Ag + Using Citrate and Hydroxylamine. Langmuir 2005, 21, 8546–8553. [Google Scholar] [CrossRef]
- Furini, L.N.; Sanchez-Cortes, S.; López-Tocón, I.; Otero, J.C.; Aroca, R.F.; Constantino, C.J.L. Detection and quantitative analysis of carbendazim herbicide on Ag nanoparticles via surface-enhanced Raman scattering. J. Raman Spectrosc. 2015, 46, 1095–1101. [Google Scholar] [CrossRef]
- International Union of Pure and Applied Chemistry (IUPAC). Available online: https://iupac.org/greeniupac2016/ (accessed on 1 December 2017).
- Zheng, G.; Lundberg, M.; Jakowski, J.; Vreven, T.; Frisch, M.J.; Morokuma, K. Implementation and benchmark tests of the DFTB method and its application in the ONIOM method. Int. J. Quantum Chem. 2009, 109, 1841–1854. [Google Scholar] [CrossRef]
- Merrick, J.P.; Moran, D.; Radom, L. An evaluation of harmonic vibrational frequency scale factors. J. Phys. Chem. A 2007, 111, 11683–11700. [Google Scholar] [CrossRef] [PubMed]
- Rubira, R.J.G.; Furini, L.N.; Constantino, C.J.L.; Sanchez-Cortes, S. SERS detection of prometryn herbicide based on its optimized adsorption on Ag nanoparticles. Vib. Spectrosc. 2021, 114, 103245. [Google Scholar] [CrossRef]
- Skipper, H.D.; Volk, V.V.; Mortland, M.M.; Raman, K.V. Hydrolysis of atrazine on soil colloids. Weed Sci. 1978, 26, 46–51. [Google Scholar] [CrossRef]
- Schmidt, M.; Bréchignac, C. Silver and oxygen: Transition from clusters to nanoparticles. Comptes Rendus Phys. 2016, 17, 481–484. [Google Scholar] [CrossRef]
- Dhafer, C.E.B.; Mezni, A.; Smiri, L.S. Surface-enhanced Raman scattering study of Ag-PVP interactions in the biocompatible Ag@PVP nanoparticles. J. Tunis. Chem. Soc. 2017, 19, 152–157. [Google Scholar]
- Gao, Y.; Jiang, P.; Liu, D.F.; Yuan, H.J.; Yan, X.Q.; Zhou, Z.P.; Wang, J.X.; Song, L.; Liu, L.F.; Zhou, W.Y.; et al. Evidence for the Monolayer Assembly of Poly(vinylpyrrolidone) on the surfaces of silver nanowires. J. Phys. Chem. B 2004, 108, 12877–12881. [Google Scholar] [CrossRef]
- Mdluli, P.S.; Sosibo, N.M.; Revaprasadu, N.; Karamanis, P.; Leszczynski, J. Surface enhanced Raman spectroscopy (SERS) and density functional theory (DFT) study for understanding the regioselective adsorption of pyrrolidinone on the surface of silver and gold colloids. J. Mol. Struct. 2009, 935, 32–38. [Google Scholar] [CrossRef]
- Chowdhury, U.K.; Biswas, B.K.; Chowdhury, T.R.; Samanta, G.; Mandal, B.K.; Basu, G.C.; Chanda, C.R.; Lodh, D.; Saha, K.C.; Mukherjee, S.K.; et al. Groundwater arsenic contamination in Bangladesh and West Bengal, India. Environ. Health Perspect. 2000, 108, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Baia, M.; Astilean, S.; Iliescu, T. Raman and SERS Investigations of Pharmaceuticals; Springer: Berlin/Heidelberg, Germany, 2008; ISBN 9783540782827. [Google Scholar]
- Hirt, R.C.; Schmitt, R.G. Ultraviolet absorption spectra ot derivatives of symmetric triazine—II. Spectrochim. Acta 1958, 12, 127–138. [Google Scholar] [CrossRef]
- Boitsov, E.N.; Finkel’shtein, A.I. Optical investigation of the molecular structure of s-triazine derivatives. VI. Ultraviolet absorption spectra of aqueous solution of cyanuric acid derivatives at various pH. Opt. Spectrosc. 1960, 9, 26–28. [Google Scholar]
- Ward, T.M.; Weber, J.B. Aqueous solubility of alkylamino-s-triazine as a function of pH and molecular structure. J. Agric. Food Chem. 1968, 16, 959–961. [Google Scholar] [CrossRef]
- Skipper, H.D.; Volk, V.V. Biological and chemical degradation of atrazine in three oregon soils. Weed Sci. 1972, 20, 344–347. [Google Scholar] [CrossRef]
- Mircescu, N.E.; Oltean, M.; Chiş, V.; Leopold, N. FTIR, FT-Raman, SERS and DFT study on melamine. Vib. Spectrosc. 2012, 62, 165–171. [Google Scholar] [CrossRef]
- Hayes, M.H.B. Adsorption of triazine herbicides on soil organic matter, including a short review on soil organic matter chemistry. In Single Pesticide Volume: The Triazine Herbicides; Gunther, F.A., Gunther, J.D., Eds.; Springer Science and Business Media LLC: New York, NY, USA, 1970; pp. 131–174. [Google Scholar] [CrossRef]
- Stevenson, F.J. Organic matter reactions involving herbicides in soil. J. Environ. Qual. 1972, 1, 333–343. [Google Scholar] [CrossRef]
- Senesi, N.; D’Orazio, V.; Miano, T.M. Adsorption mechanisms of s-triazine and bipyridylium herbicides on humic acids from hop field soils. Geoderma 1995, 66, 273–283. [Google Scholar] [CrossRef]
- Gorup, L.F. Nanopartículas Coloidais de ouro e Prata e sua Funcionalização com Dibutil-Doicalgenetos. Master’s Thesis, Universidade de São Paulo, São Carlos, Brazil, 2010. [Google Scholar]
- Korak, J.; Rosario-Ortiz, F.L.; Summers, R.S. Fluorescence Characterization of Humic Substance Coagulation: Application of New Tools to an Old Process. In Advances in the Physicochemical Characterization of Organic Matter, ACS Symposium Serie; Rosario-Ortiz, F.L., Ed.; American Chemical Society: Washington, DC, USA, 2014; Volume 1160, pp. 281–300. [Google Scholar] [CrossRef]
- Ritchie, J.D.; Perdue, E.M. Proton-binding study of standard and reference fulvic acids, humic acids, and natural organic matter. Geochim. Cosmochim. Acta 2003, 67, 85–96. [Google Scholar] [CrossRef]
- Thorn, K.A.; Folan, D.W.; McCarthy, P. Characterization of the International Humic Substances Society Standard and Reference Fulvic and Humic Acids by Solution State Carbon-13 (13c) and Hydrogen-1 (1h) Nuclear Magnetic Resonance Spectrometry; Water-Resources Investigations Report 89-4196; U.S. Geological Survey: Denver, CO, USA, 1989.
- Kalouskova, N. Adsorption of atrazine on humic acids. J. Environ. Sci. Health Part B 1989, 24, 599–617. [Google Scholar] [CrossRef]
- Corrado, G.; Sanchez-Cortes, S.; Francioso, O.; Garcia-Ramos, J.V. Surface-enhanced Raman and fluorescence joint analysis of soil humic acids. Anal. Chim. Acta 2008, 616, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Langmuir, I. The constitution and fundamental properties of solids and liquids. II. Liquids. 1. J. Am. Chem. Soc. 1917, 39, 1848–1906. [Google Scholar] [CrossRef]
- Langmuir, I. The constitution and fundamental properties of solids and liquids. Part I. Solids. J. Am. Chem. Soc. 1916, 38, 2221–2295. [Google Scholar] [CrossRef]
- Izquierdo-Lorenzo, I.; Sanchez-Cortes, S.; Garcia-Ramos, J.V. Adsorption of beta-adrenergic agonists used in sport doping on metal nanoparticles: A detection study based on surface-enhanced raman scattering. Langmuir 2010, 26, 14663–14670. [Google Scholar] [CrossRef] [PubMed]
- Saleh, N.; Al-Rawashdeh, N.A.F. Fluorescence enhancement of carbendazim fungicide in cucurbit[6]uril. J. Fluoresc. 2006, 16, 487–493. [Google Scholar] [CrossRef]
- Liu, B.; Zhou, P.; Liu, X.; Sun, X.; Li, H.; Lin, M. Detection of pesticides in fruits by surface-enhanced raman spectroscopy coupled with gold nanostructures. Food Bioprocess. Technol. 2013, 6, 710–718. [Google Scholar] [CrossRef]
- Luo, S.; Wu, Y.; Gou, H. A voltammetric sensor based on GO–MWNTs hybrid nanomaterial-modified electrode for determination of carbendazim in soil and water samples. Ionics 2013, 19, 673–680. [Google Scholar] [CrossRef]
- Janči, T.; Mikac, L.; Ivanda, M.; Marušić Radovčić, N.; Medić, H.; Vidaček, S. Optimization of parameters for histamine detection in fish muscle extracts by surface-enhanced Raman spectroscopy using silver colloid SERS substrates. J. Raman Spectrosc. 2017, 48, 64–72. [Google Scholar] [CrossRef]
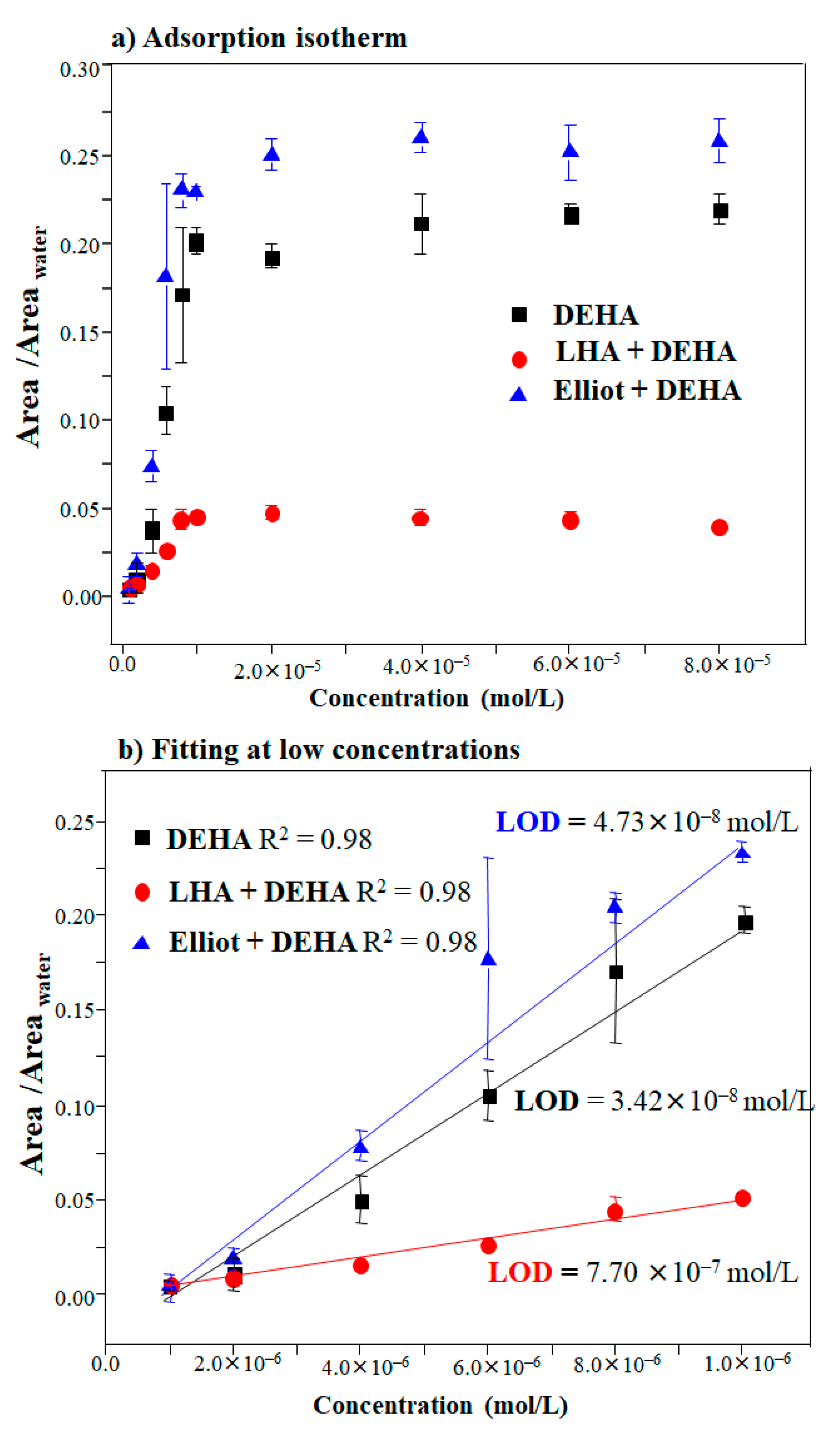
| Sample | Sensitivity (IsmKad) | Limit of Detection | |
|---|---|---|---|
| EHA + DEHA | 3273 | 4.73 × 10−8 mol/L | 8 ppb |
| DEHA | 2149 | 3.42 × 10−8 mol/L | 6 ppb |
| LHA + DEHA | 33 | 7.70 × 10−7 mol/L | 130 ppb |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zanasi, G.; Rubira, R.J.G.; Francioso, O.; Cañamares, M.V.; Constantino, C.J.L.; Sanchez-Cortes, S. Sensing Atrazine Herbicide Degradation Products through Their Interactions with Humic Substances by Surface-Enhanced Raman Scattering. Chemosensors 2021, 9, 148. https://doi.org/10.3390/chemosensors9060148
Zanasi G, Rubira RJG, Francioso O, Cañamares MV, Constantino CJL, Sanchez-Cortes S. Sensing Atrazine Herbicide Degradation Products through Their Interactions with Humic Substances by Surface-Enhanced Raman Scattering. Chemosensors. 2021; 9(6):148. https://doi.org/10.3390/chemosensors9060148
Chicago/Turabian StyleZanasi, Giulia, Rafael J. G. Rubira, Ornella Francioso, Maria Vega Cañamares, Carlos J. L. Constantino, and Santiago Sanchez-Cortes. 2021. "Sensing Atrazine Herbicide Degradation Products through Their Interactions with Humic Substances by Surface-Enhanced Raman Scattering" Chemosensors 9, no. 6: 148. https://doi.org/10.3390/chemosensors9060148
APA StyleZanasi, G., Rubira, R. J. G., Francioso, O., Cañamares, M. V., Constantino, C. J. L., & Sanchez-Cortes, S. (2021). Sensing Atrazine Herbicide Degradation Products through Their Interactions with Humic Substances by Surface-Enhanced Raman Scattering. Chemosensors, 9(6), 148. https://doi.org/10.3390/chemosensors9060148








