Classification and Identification of Essential Oils from Herbs and Fruits Based on a MOS Electronic-Nose Technology
Abstract
1. Introduction
2. Materials and Methods
2.1. Essential Oil Samples
2.2. Electronic Nose Instrument
2.3. Instrument Settings and Run Parameters
2.3.1. Baseline Phase
2.3.2. Sample Aroma-Injection Phase
2.3.3. Purge and Air-Cleanse Phase
2.3.4. Data Collection Cycle
2.4. Sample E-Nose Analyses
2.5. Post-Analysis Data Processing and Statistical Comparisons
3. Results
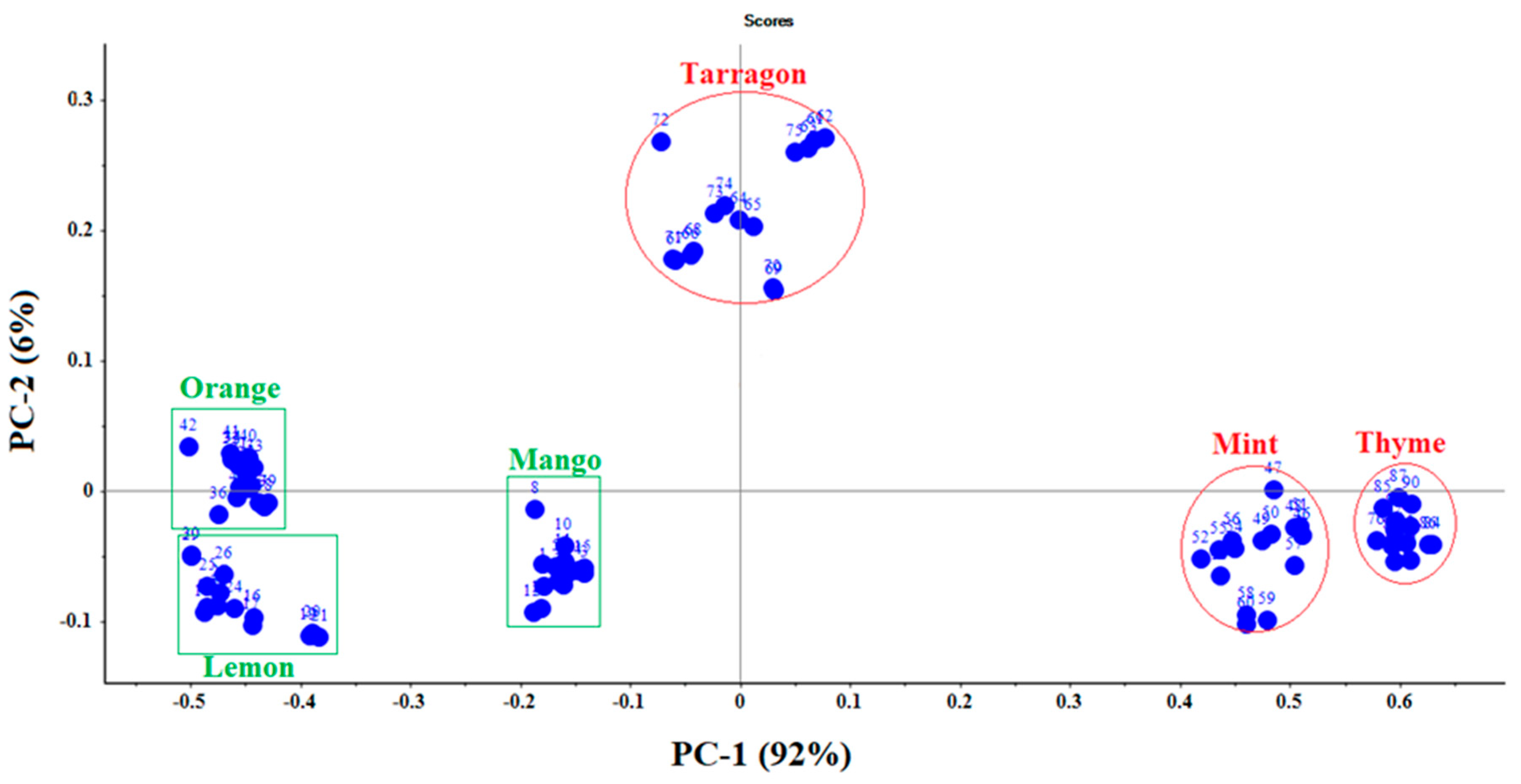
3.1. Principal Component Analyses
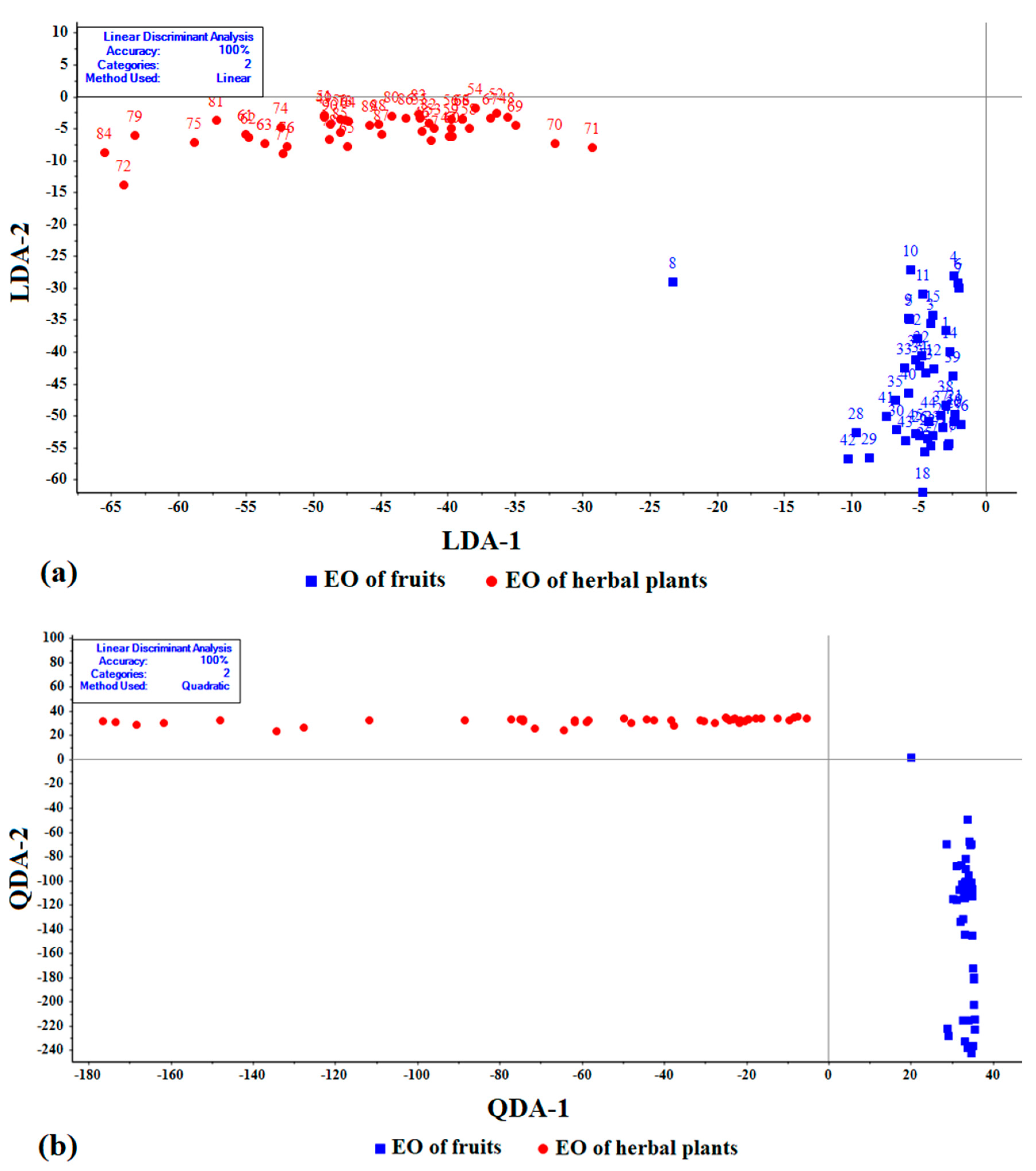
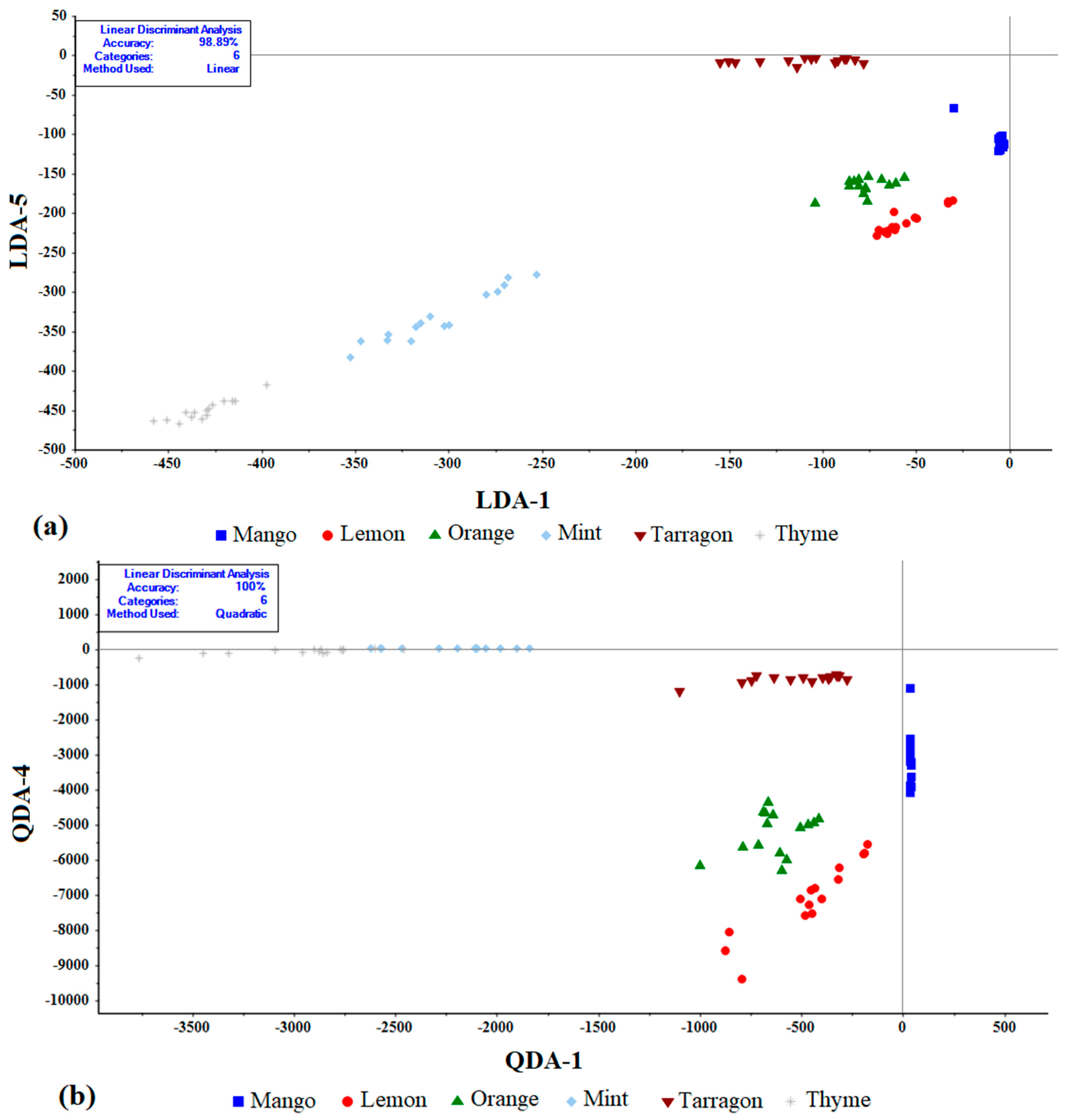
3.2. Linear Discriminate Analyses
3.3. Kernal Analysis of Model Performance
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Model | Type of Fruit Juice | Accuracy | Precision | Sensitivity | Specificity | AUC |
|---|---|---|---|---|---|---|
| LDA | EO of fruits | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 |
| EO of herbal plants | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |
| Average per class | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |
| QDA | EO of fruits | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 |
| EO of herbal plants | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |
| Average per class | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 |
References
- Baby, R.; Cabezas, M.; Castro, E.; Filip, R.; Walsöe de Reca, N.E. Quality control of medicinal plants with an electronic nose. Sens. Actuators B Chem. 2005, 106, 24–28. [Google Scholar] [CrossRef]
- Gorji-Chakespari, A.; Nikbakht, A.M.; Sefidkon, F.; Ghasemi-Varnamkhasti, M.; Valero, E.L. Classification of essential oil composition in Rosa damascena Mill. genotypes using an electronic nose. JARMAP 2017, 4, 27–34. [Google Scholar] [CrossRef]
- Sharmeen, J.B.; Mahomoodally, F.M.; Zengin, G.; Maggi, F. Essential oils as natural sources of fragrance compounds for cosmetics and cosmeceuticals. Molecules 2021, 26, 666. [Google Scholar] [CrossRef] [PubMed]
- Tigrine-Kordjani, N.; Meklati, B.Y.; Chemat, F. Microwave ‘dry’ distillation as a useful tool for extraction of edible essential oils. Int. J. Aromath. 2006, 16, 141–147. [Google Scholar] [CrossRef]
- Valenti, M. Strawberry Essential Oil, and Its Shocking Greenwashing Truth. Valenti Organics. Available online: https://www.byvalenti.com/blog/journal/beauty/strawberry-essential-oil-and-its-shocking-greenwashing-truth/ (accessed on 29 April 2021).
- Karami, H.; Rasekh, M.; Mirzaee-Ghaleh, E. Qualitative analysis of edible oil oxidation using an olfactory machine. J. Food Meas. Charact. 2020, 14, 2600–2610. [Google Scholar] [CrossRef]
- Karami, H.; Rasekh, M.; Mirzaee-Ghaleh, E. Application of the E-nose machine system to detect adulterations in mixed edible oils using chemometrics methods. J. Food Process. Preserv. 2020, 44, e14696. [Google Scholar] [CrossRef]
- Gancarz, M.; Malaga-Toboła, U.; Oniszczuk, A.; Tabor, S.; Oniszczuk, T.; Gawrysiak-Witulska, M.; Rusinek, R. Detection and measurement of aroma compounds with the electronic nose and a novel method for MOS sensor signal analysis during the wheat bread making process. Food Bioprod. Process. 2021. [Google Scholar] [CrossRef]
- Rusinek, R.; Gawrysiak-Witulska, M.; Siger, A.; Oniszczuk, A.; Ptaszyńska, A.A.; Knaga, J.; Malaga-Toboła, U.; Gancarz, M. Effect of supplementation of flour with fruit fiber on the volatile compound profile in bread. Sensors 2021, 21, 2812. [Google Scholar] [CrossRef]
- Karami, H.; Rasekh, M.; Mirzaee-Ghaleh, E. Comparison of chemometrics and AOCS official methods for predicting the shelf life of edible oil. Chemometr. Intell. Lab. Syst. 2020, 206, 104165. [Google Scholar] [CrossRef]
- Wilson, A.D.; Baietto, M. Applications and advances in electronic nose technologies. Sensors 2009, 9, 5099–5148. [Google Scholar] [CrossRef]
- Hernández-Mesa, M.; Escourrou, A.; Monteau, F.; Le Bizec, B.; Dervilly-Pinel, G. Current applications and perspectives of ion mobility spectrometry to answer chemical food safety issues. TrAC 2017, 94, 39–53. [Google Scholar] [CrossRef]
- Müller, P.; Salminen, K.; Nieminen, V.; Kontunen, A.; Karjalainen, M.; Isokoski, P.; Rantala, J.; Savia, M.; Väliaho, J.; Kallio, P.; et al. Scent classification by K nearest neighbors using ion-mobility spectrometry measurements. Expert Syst. Appl. 2019, 115, 593–606. [Google Scholar] [CrossRef]
- Kafle, G.K.; Khot, L.R.; Sankaran, S.; Bahlol, H.Y.; Tufariello, J.A.; Hill, H.H. State of ion mobility spectrometry and applications in agriculture: A review. Eng. Agric. Environ. Food 2016, 9, 346–357. [Google Scholar] [CrossRef]
- Wilson, A.D. Diverse applications of electronic nose technologies in agriculture and forestry. Sensors 2013, 13, 2295–2348. [Google Scholar] [CrossRef]
- Dai, C.; Huang, X.; Lv, R.; Zhang, Z.; Sun, J.; Aheto, J.H. Analysis of volatile compounds of Tremella aurantialba fermentation via electronic nose and HS-SPME-GC-MS. J. Food Saf. 2018, 38, 1–8. [Google Scholar] [CrossRef]
- Delgado-Rodríguez, M.; Ruiz-Montoya, M.; Giraldez, I.; López, R.; Madejón, E.; Díaz, M.J. Use of electronic nose and GC-MS in detection and monitoring some VOC. Atmos. Environ. 2012, 51, 278–285. [Google Scholar] [CrossRef]
- Marek, G.; Dobrzański, B.; Oniszczuk, T.; Combrzyński, M.; Ćwikła, D.; Rusinek, R. Detection and differentiation of volatile compound profiles in roasted coffee arabica beans from different countries using an electronic nose and GC-MS. Sensors 2020, 20, 2124. [Google Scholar] [CrossRef]
- Camardo Leggieri, M.; Mazzoni, M.; Fodil, S.; Moschini, M.; Bertuzzi, T.; Prandini, A.; Battilani, P. An electronic nose supported by an artificial neural network for the rapid detection of aflatoxin B1 and fumonisins in maize. Food Control 2021, 123, 107722. [Google Scholar] [CrossRef]
- Ottoboni, M.; Pinotti, L.; Tretola, M.; Giromini, C.; Fusi, E.; Rebucci, R.; Grillo, M.; Tassoni, L.; Foresta, S.; Gastaldello, S.; et al. Combining e-nose and lateral flow immunoassays (LFIAs) for rapid occurrence/co-occurrence aflatoxin and fumonisin detection in maize. Toxins 2018, 10, 416. [Google Scholar] [CrossRef] [PubMed]
- Timsorn, K.; Lorjaroenphon, Y.; Wongchoosuk, C. Identification of adulteration in uncooked jasmine rice by a portable low-cost artificial olfactory system. Measurement 2017, 108, 67–76. [Google Scholar] [CrossRef]
- Russo, M.; Serra, D.; Suraci, F.; Postorino, S. Effectiveness of electronic nose systems to detect bergamot (Citrus bergamia Risso et Poiteau) essential oil quality and genuineness. J. Essent. Oil Res. 2012, 24, 137–151. [Google Scholar] [CrossRef]
- Graboski, A.M.; Zakrzevski, C.A.; Shimizu, F.M.; Paschoalin, R.T.; Soares, A.C.; Steffens, J.; Paroul, N.; Steffens, C. Electronic nose based on carbon nanocomposite sensors for clove essential oil detection. ACS Sens. 2020, 5, 1814–1821. [Google Scholar] [CrossRef]
- Lias, S.; Mohamad Ali, N.A.; Jamil, M.; Tolmanan, M.S.Y.; Misman, M.A. A study on the application of electronic nose coupled with DFA and statistical analysis for evaluating the relationship between sample volumes versus sensor intensity of agarwood essential oils blending ratio. MATEC Web Conf. 2018, 201, 02008. [Google Scholar] [CrossRef]
- Wu, S.-Q.; Li, R.; Jiang, Z.-T.; Wang, Y.; Tan, J.; Tang, S.-H. Evaluation of antioxidant active ingredients of spikenard essential oil by ultra-fast gas chromatography electronic nose and radical scavenging mechanism. Ind. Crops Prod. 2020, 151, 112489. [Google Scholar] [CrossRef]
- Wilson, A.D.; Lester, D.G.; Oberle, C.S. Application of conductive polymer analysis for wood and woody plant identifications. For. Ecol. Managem. 2005, 209, 207–224. [Google Scholar] [CrossRef]
- Baietto, M.; Wilson, A.D. Electronic-nose applications for fruit identification, ripeness, and quality grading. Sensors 2015, 15, 899–931. [Google Scholar] [CrossRef]
- Wilson, A.D. Application of a conductive polymer electronic-nose device to identify aged woody samples. In Proceedings of the Third International IARIA Conference on Sensor Device Technologies and Applications, Rome, Italy, 19–24 August 2012; Yurish, S., Chilibon, I., Carvalho, V., Gervais-Ducouret, S., Eds.; Xpert Publishing Services: Wilmington, DE, USA, 2012; pp. 77–82. [Google Scholar]
- Siqueira, L.F.S.; Araújo Júnior, R.F.; de Araújo, A.A.; Morais, C.L.M.; Lima, K.M.G. LDA vs. QDA for FT-MIR prostate cancer tissue classification. Chemom. Intell. Lab. Syst. 2017, 162, 123–129. [Google Scholar] [CrossRef]
- Basri, K.N.; Hussain, M.N.; Bakar, J.; Sharif, Z.; Khir, M.F.A.; Zoolfakar, A.S. Classification and quantification of palm oil adulteration via portable NIR spectroscopy. Spectrochim. Acta A 2017, 173, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Okur, S.; Sarheed, M.; Huber, R.; Zhang, Z.; Heinke, L.; Kanbar, A.; Wöll, C.; Nick, P.; Lemmer, U. Identification of mint scents using a QCM based e-nose. Chemosensors 2021, 9, 31. [Google Scholar] [CrossRef]
- Gómez, A.H.; Wang, J.; Hu, G.; Pereira, A.G. Electronic nose technique potential monitoring mandarin maturity. Sens. Actuators B Chem. 2006, 113, 347–353. [Google Scholar] [CrossRef]
- Rasekh, M.; Karami, H. Application of electronic nose with chemometrics methods to the detection of juices fraud. J. Food Process. Preserv. 2021, 45, e15432. [Google Scholar] [CrossRef]
- Lashgari, M.; MohammadiGol, R. Discrimination of Golab apple storage time using acoustic impulse response and LDA and QDA discriminant analysis techniques. Iran Agr. Res. 2016, 35, 65–70. [Google Scholar]
- Hong, X.; Wang, J.; Qi, G. E-nose combined with chemometrics to trace tomato-juice quality. J. Food Eng. 2015, 149, 38–43. [Google Scholar] [CrossRef]
- Tohidi, M.; Ghasemi-Varnamkhasti, M.; Ghafarinia, V.; Saeid Mohtasebi, S.; Bonyadian, M. Identification of trace amounts of detergent powder in raw milk using a customized low-cost artificial olfactory system: A novel method. Measurement 2018, 124, 120–129. [Google Scholar] [CrossRef]
- Huang, L.; Liu, H.; Zhang, B.; Wu, D. Application of electronic nose with multivariate analysis and sensor selection for botanical origin identification and quality determination of honey. Food Bioproc. Tech. 2015, 8, 359–370. [Google Scholar] [CrossRef]
- Faal, S.; Loghavi, M.; Kamgar, S. Physicochemical properties of Iranian ziziphus honey and emerging approach for predicting them using electronic nose. Measurement 2019, 148, 106936. [Google Scholar] [CrossRef]
- Ghasemi-Varnamkhasti, M.; Mohammad-Razdari, A.; Yoosefian, S.H.; Izadi, Z.; Rabiei, G. Selection of an optimized metal oxide semiconductor sensor (MOS) array for freshness characterization of strawberry in polymer packages using response surface method (RSM). Postharvest Biol. Technol. 2019, 151, 53–60. [Google Scholar] [CrossRef]
- Rasekh, M.; Karami, H. E-nose coupled with an artificial neural network to detection of fraud in pure and industrial fruit juices. Int. J. Food Prop. 2021, 24, 592–602. [Google Scholar] [CrossRef]
- Wei, P.; Ning, Z.; Ye, S.; Sun, L.; Yang, F.; Wong, K.C.; Westerdahl, D.; Louie, P.K.K. Impact analysis of temperature and humidity conditions on electrochemical sensor response in ambient air quality monitoring. Sensors 2018, 18, 59. [Google Scholar] [CrossRef]
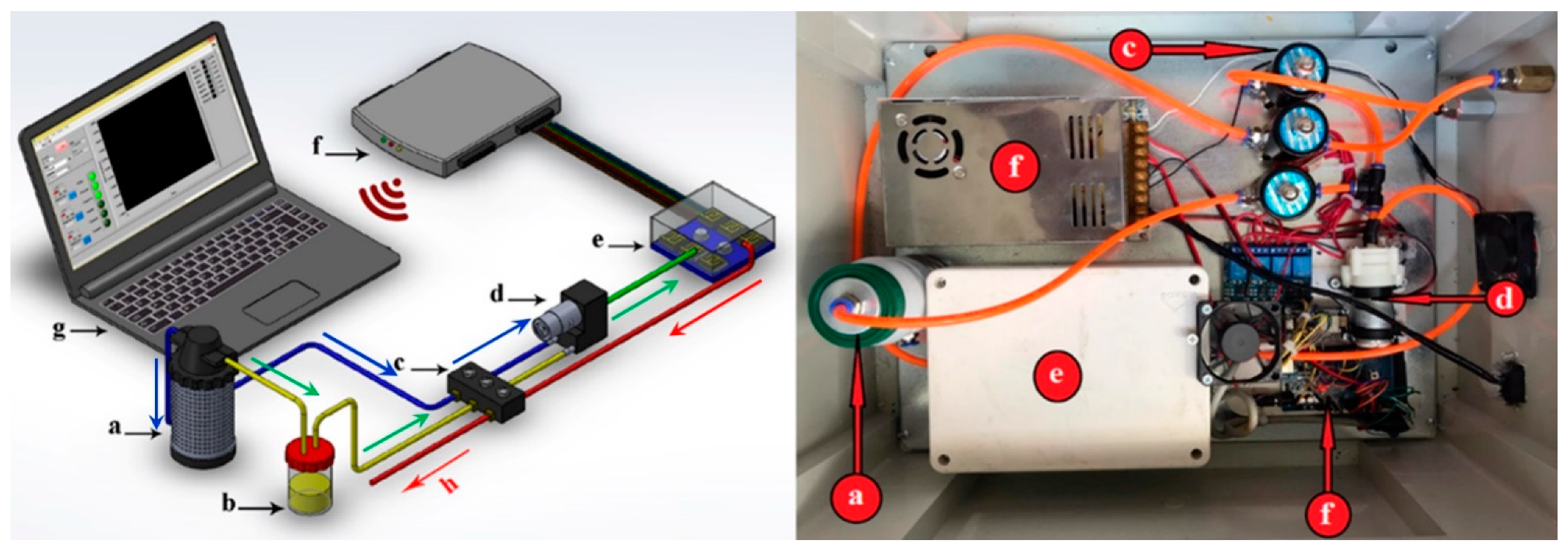

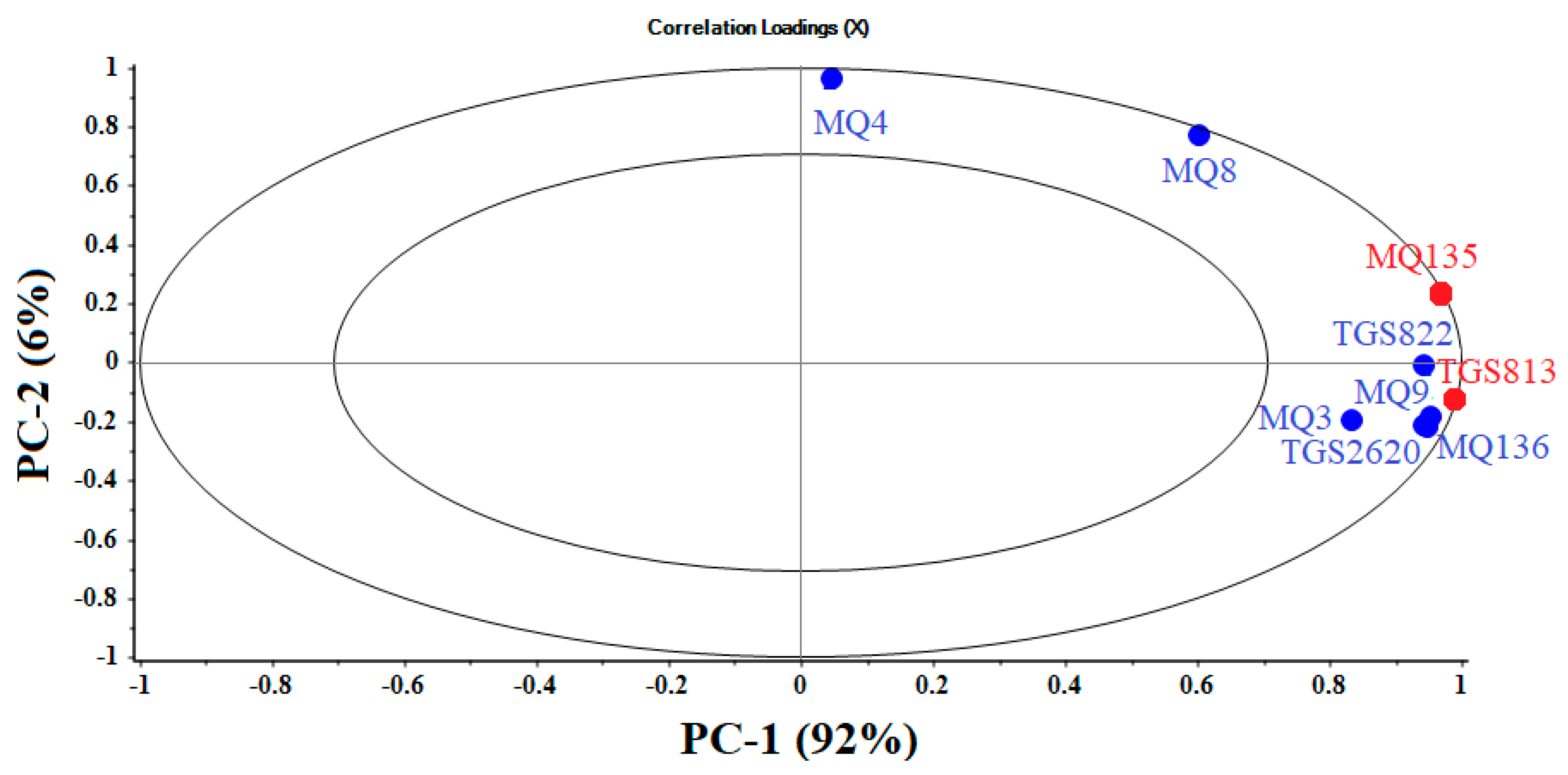
| Sensor | Sensor Name | Detection Ranges (ppm) | Main Applications (Gases Detected) |
|---|---|---|---|
| 1 | MQ9 | 10–10,000 | CO, combustible gases |
| 2 | MQ4 | 300–100 | Urban gases, methane (CH4) |
| 3 | MQ135 | 10–10,000 | Ammonia, benzene, sulfides |
| 4 | MQ8 | 100–1000 | Hydrogen (H2) |
| 5 | TGS2620 | 50–5000 | Alcohols, organic solvents |
| 6 | MQ136 | 1–200 | Sulfur dioxide (SO2) |
| 7 | TGS813 | 500–10,000 | CH4, C3H8, C4H10 (hydrocarbons) |
| 8 | TGS822 | 50–5000 | Organic solvents |
| 9 | MQ3 | 10–300 | Alcohols |
| Model 1 | Type of Fruit Juice 2 | EO of Herbal Plants | EO of Fruits | Accuracy |
|---|---|---|---|---|
| LDA | EO of fruits | 45 50.0% | 0 0.0% | 100% 0.0% |
| EO of herbal plants | 0 0.0% | 45 50.0% | 100% 0.0% | |
| 100% 0.0% | 100% 0.0% | 100% 0.0% | ||
| QDA | EO of fruits | 45 50.0% | 0 0.0% | 100% 0.0% |
| EO of herbal plants | 0 0.0% | 45 50.0% | 100% 0.0% | |
| 100% 0.0% | 100% 0.0% | 100% 0.0% |
| Model | Type of EO. | Mango | Lemon | Orange | Mint | Tarragon | Thyme | Accuracy |
|---|---|---|---|---|---|---|---|---|
| LDA | Mango | 15 16.7% | 0 0.0% | 0 0.0% | 0 0.0% | 0 0.0% | 0 0.0% | 100% 0.0% |
| Lemon | 0 0.0% | 15 16.7% | 0 0.0% | 0 0.0% | 0 0.0% | 0 0.0% | 100% 0.0% | |
| Orange | 0 0.0% | 0 0.0% | 15 16.7% | 0 0.0% | 0 0.0% | 0 0.0% | 100% 0.0% | |
| Mint | 0 0.0% | 0 0.0% | 0 0.0% | 13 14.4% | 0 0.0% | 0 0.0% | 100% 0.0% | |
| Tarragon | 0 0.0% | 0 0.0% | 0 0.0% | 0 0.0% | 15 16.7% | 0 0.0% | 100% 0.0% | |
| Thyme | 0 0.0% | 0 0.0% | 0 0.0% | 2 2.2% | 0 0.0% | 15 16.7% | 86.7% 13.3% | |
| 100% 0.0% | 100% 0.0% | 100% 0.0% | 86.7% 13.3% | 100% 0.0% | 100% 0.0% | 97.8% 2.2% | ||
| QDA | Mango | 15 16.66% | 0 0.0% | 0 0.0% | 0 0.0% | 0 0.0% | 0 0.0% | 100% 0.0% |
| Lemon | 0 0.0% | 15 16.66% | 0 0.0% | 0 0.0% | 0 0.0% | 0 0.0% | 100% 0.0% | |
| Orange | 0 0.0% | 0 0.0% | 15 16.66% | 0 0.0% | 0 0.0% | 0 0.0% | 100% 0.0% | |
| Mint | 0 0.0% | 0 0.0% | 0 0.0% | 15 16.66% | 0 0.0% | 0 0.0% | 100% 0.0% | |
| Tarragon | 0 0.0% | 0 0.0% | 0 0.0% | 0 0.0% | 15 16.66% | 0 0.0% | 100% 0.0% | |
| Thyme | 0 0.0% | 0 0.0% | 0 0.0% | 0 0.0% | 0 0.0% | 15 16.66% | 100% 0.0% | |
| 100% 0.0% | 100% 0.0% | 100% 0.0% | 100% 0.0% | 100% 0.0% | 100% 0.0% | 100% 0.0% |
| Model | Type of EO. | Accuracy | Precision | Sensitivity | Specificity | AUC |
|---|---|---|---|---|---|---|
| LDA | Mango | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 |
| Lemon | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |
| Orange | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |
| Mint | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |
| Tarragon | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |
| Thyme | 0.990 | 0.882 | 1.000 | 0.990 | 0.995 | |
| Average per class | 0.998 | 0.980 | 1.000 | 0.998 | 0.999 | |
| QDA | Mango | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 |
| Lemon | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |
| Orange | 1.000 | 1.000 | 1.000 | 0.995 | 0.997 | |
| Mint | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |
| Tarragon | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |
| Thyme | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |
| Average per class | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | |
| Kernel Function | C-SVM * | Nu-SVM * | ||||||
|---|---|---|---|---|---|---|---|---|
| c | γ | Train | Validation | Nu | γ | Train | Validation | |
| Categories 6 | ||||||||
| linear | 0.01 | 1 | 96.7 | 96.7 | 0.01 | 1 | 100 | 98.9 |
| Polynomial | 10 | 1 | 98.9 | 98.9 | 0.50 | 1 | 96.7 | 98.9 |
| Radial basis function | 100 | 1 | 98.9 | 98.9 | 0.26 | 0.1 | 100 | 98.9 |
| sigmoid | 0.01 | 0.01 | 96.7 | 96.7 | 0.26 | 0.1 | 98.9 | 98.9 |
| Categories 2 | ||||||||
| linear | 1 | 1 | 100 | 100 | 0.01 | 1 | 100 | 100 |
| Polynomial | 1 | 1 | 100 | 100 | 0.01 | 0.01 | 100 | 100 |
| Radial basis function | 1 | 1 | 100 | 100 | 0.01 | 0.01 | 100 | 100 |
| sigmoid | 10 | 0.1 | 100 | 100 | 0.01 | 0.26 | 100 | 100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rasekh, M.; Karami, H.; Wilson, A.D.; Gancarz, M. Classification and Identification of Essential Oils from Herbs and Fruits Based on a MOS Electronic-Nose Technology. Chemosensors 2021, 9, 142. https://doi.org/10.3390/chemosensors9060142
Rasekh M, Karami H, Wilson AD, Gancarz M. Classification and Identification of Essential Oils from Herbs and Fruits Based on a MOS Electronic-Nose Technology. Chemosensors. 2021; 9(6):142. https://doi.org/10.3390/chemosensors9060142
Chicago/Turabian StyleRasekh, Mansour, Hamed Karami, Alphus Dan Wilson, and Marek Gancarz. 2021. "Classification and Identification of Essential Oils from Herbs and Fruits Based on a MOS Electronic-Nose Technology" Chemosensors 9, no. 6: 142. https://doi.org/10.3390/chemosensors9060142
APA StyleRasekh, M., Karami, H., Wilson, A. D., & Gancarz, M. (2021). Classification and Identification of Essential Oils from Herbs and Fruits Based on a MOS Electronic-Nose Technology. Chemosensors, 9(6), 142. https://doi.org/10.3390/chemosensors9060142









