A Perspective on Recent Advances in Piezoelectric Chemical Sensors for Environmental Monitoring and Foodstuffs Analysis
Abstract
:1. Introduction
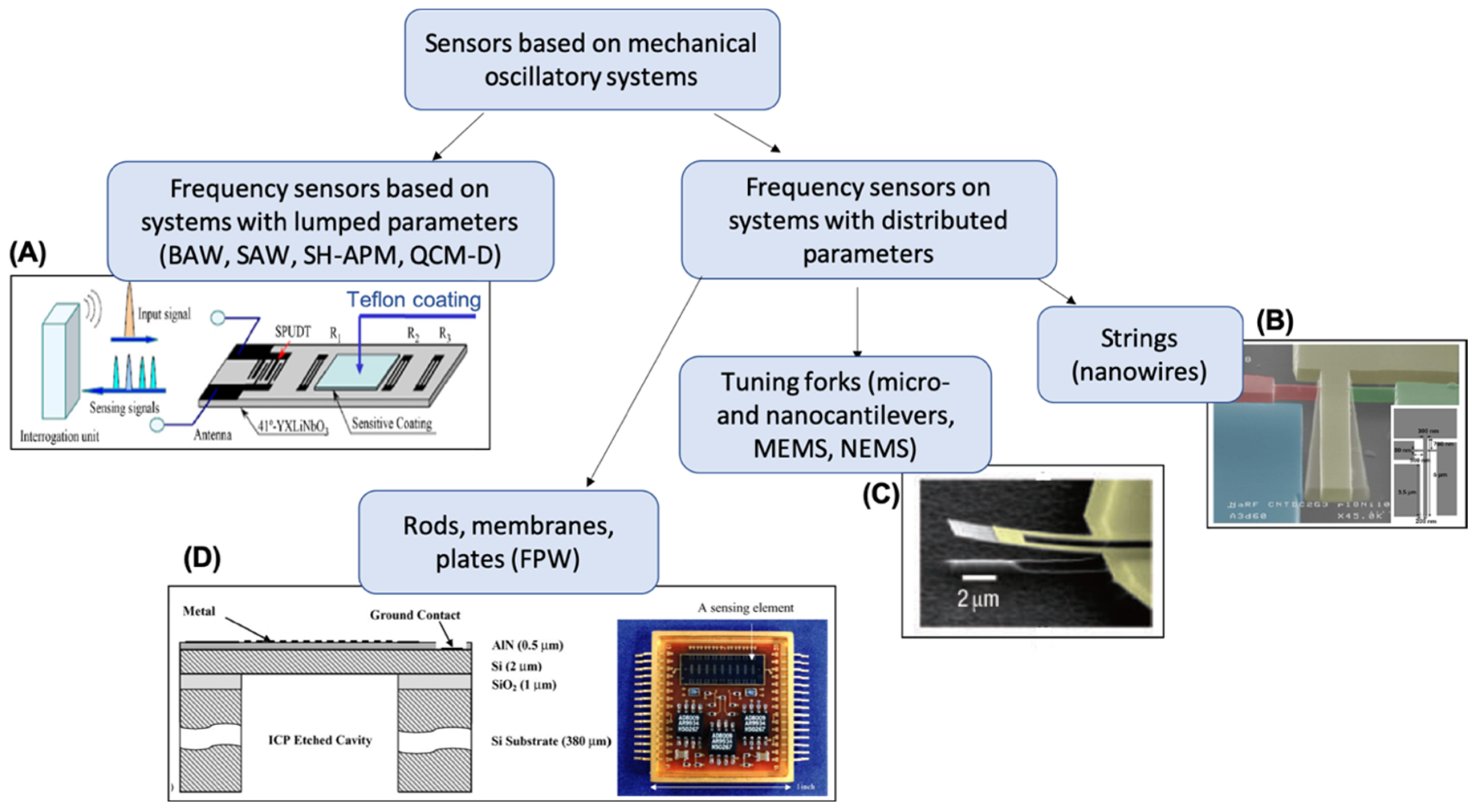
2. Piezoelectric Sensors Based on Gravimetric Resonant Devices
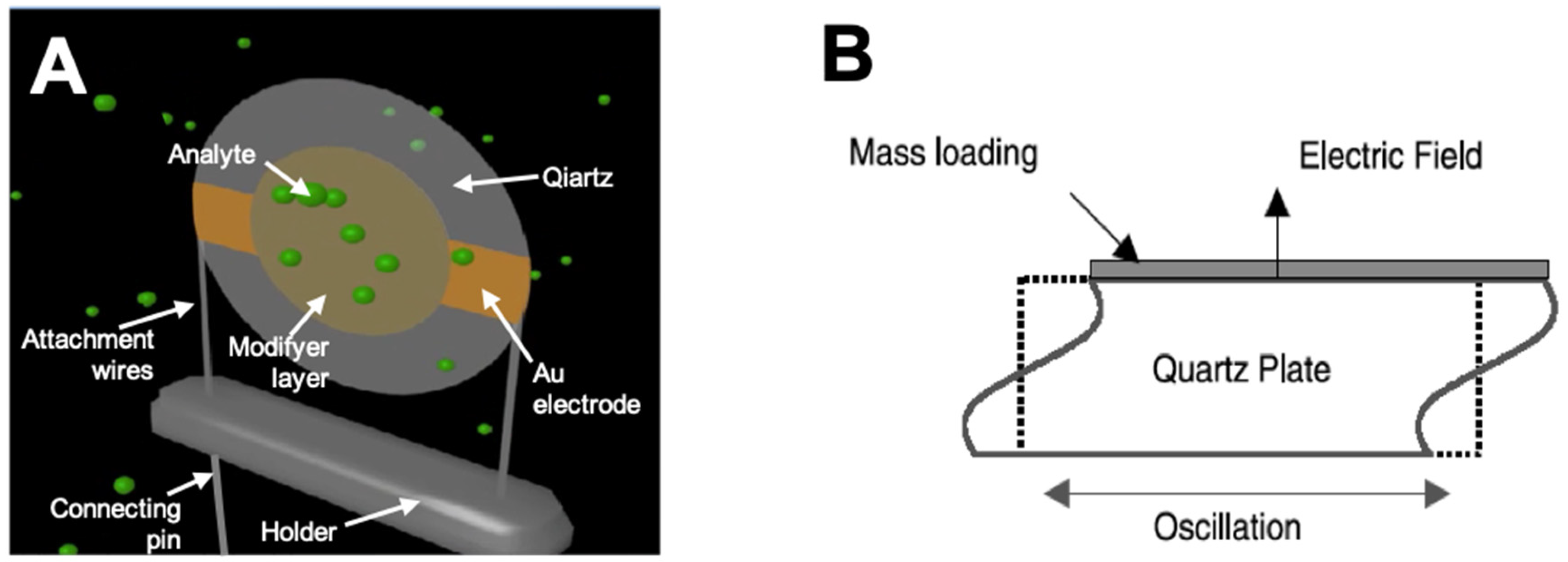
2.1. Design and Operation Principle
2.2. Piezosensors Optimization Strategies
2.3. Coating Selection
3. Single Piezosensor Applications
4. Arrays of Piezosensors as Measuring Elements of Multicomponent Detectors
4.1. Foodstuffs Analysis
4.2. Environmental Monitoring
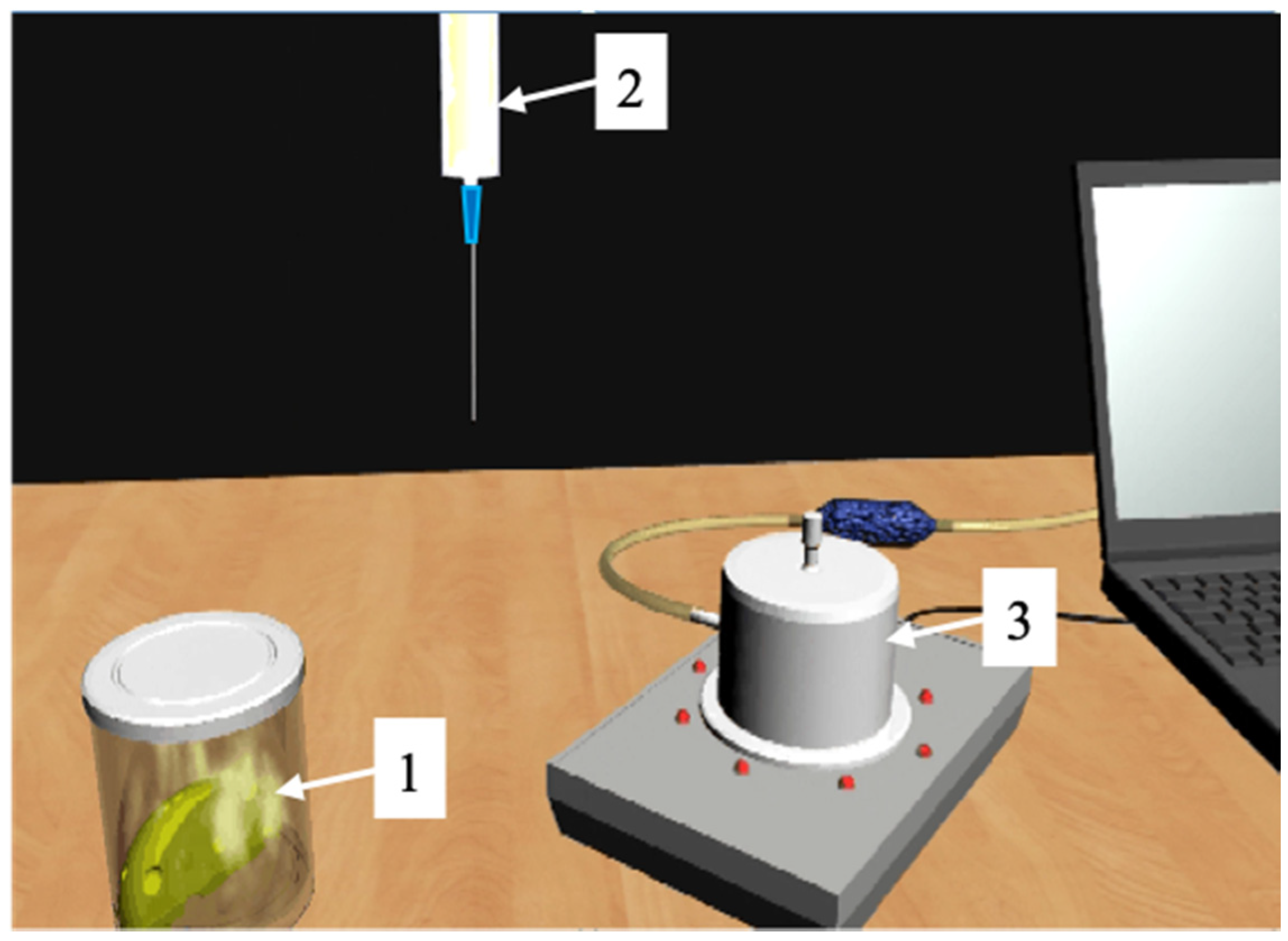
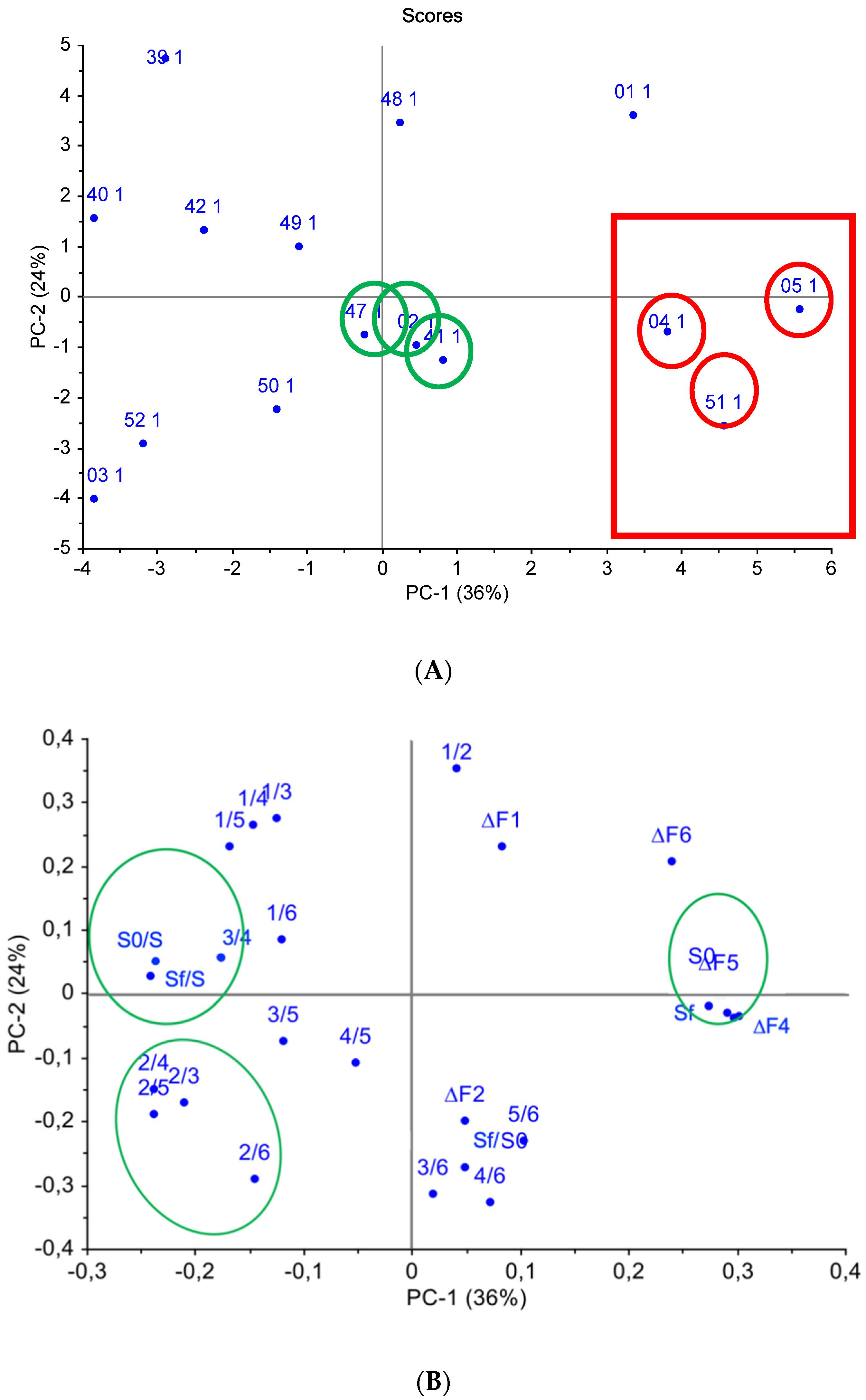
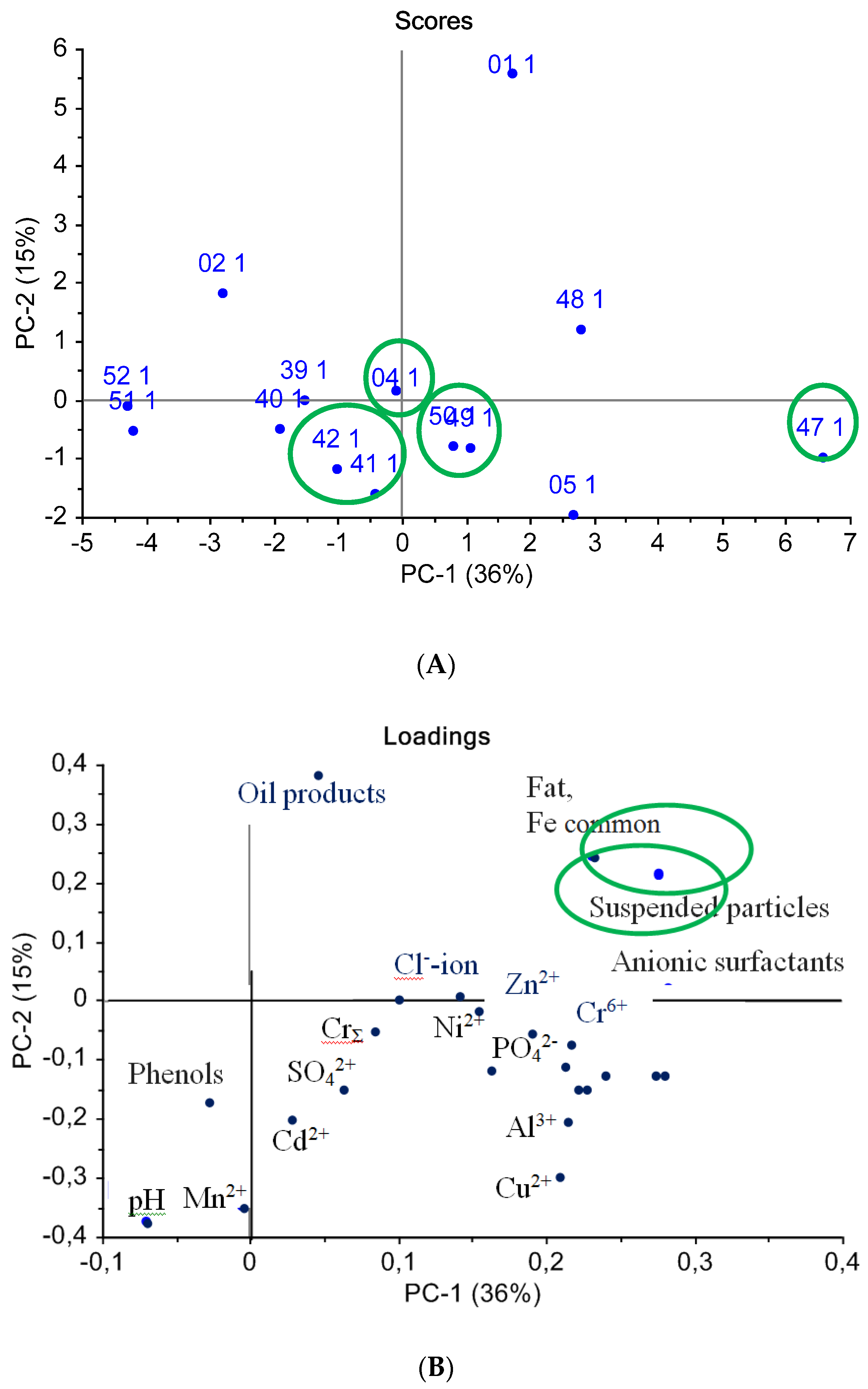
4.3. Case Study: QCM E-Nose for Wastewater Quality Assessment
5. New Opportunities and Trends for Piezosensors Development
Author Contributions
Funding
Conflicts of Interest
References
- Kuchmenko, T.A. Problems of Analytical Chemistry in “Chemical Sensors”; Vlasov, Y.G., Ed.; Scientific Council on Analytical Chemistry OHNM of RAS, Nauka: Moscow, Russia, 2011; Volume 14, p. 127. [Google Scholar]
- Nikolelis, D.P.; Varzakas, T.; Erdem, A.; Nikoleli, G.P. Portable Biosensing of Food Toxicants and Environmental Pollutants; Jones, B., Huang, H., Eds.; CRC Press by Taylor Francis Group: Boca Raton, FL, USA, 2014; p. 71. ISBN 13978-1-4665-7633-9. [Google Scholar]
- Lahcen, A.A.; Amine, A. Recent Advances in Electrochemical Sensors Based on Molecularly Imprinted Polymers and Nanomaterials. Electroanalysis 2019, 31, 188–201. [Google Scholar] [CrossRef]
- Lvova, L.; Pudi, R.; Galloni, P.; Lippolis, V.; Di Natale, C.; Lundstrom, I.; Paolesse, R. Multi-transduction sensing films for Electronic Tongue applications. Sens. Actuators B 2015, 207B, 1076–1086. [Google Scholar] [CrossRef]
- Lvova, L.; Di Natale, C.; Paolesse, R. Chemical Sensors for Water Potability Assessment. In Bottled and Packaged Water; Grumezescu, A.M., Holba, A.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; Volume 4, pp. 177–208. [Google Scholar] [CrossRef]
- Lvova, L.; Nadporozhskaya, M. Chemical sensors for soil analysis: Principles and applications. In New Pesticides and Soil Sensors; Grumezescu, A.M., Ed.; Series Nanotechnology in the Agri-Food Industry; Elsevier: Amsterdam, The Netherlands, 2017; Volume 10, pp. 637–678. [Google Scholar] [CrossRef]
- Pelosi, P.; Zhu, J.; Knoll, W. From Gas Sensors to Biomimetic Artificial Noses. Chemosensors 2018, 6, 32. [Google Scholar] [CrossRef]
- Loutfi, A.; Coradeschi, S.; Mani, G.K.; Shankar, P.; Rayappan, J.B.B. Electronic noses for food quality: A review. J. Food Eng. 2015, 144, 103–111. [Google Scholar] [CrossRef]
- Lvova, L.; Kirsanov, D.; Legin, A.; Di Natale, C. Multisensor Systems for Chemical Analysis: Materials and Sensors; Pan Stanford Publishing: Singapore, 2013; p. 392. ISBN1 978-981441116-5. ISBN2 978-981441115-8. [Google Scholar]
- Lvova, L.; Kirsanov, D. Multisensor Systems for Analysis of Liquids and Gases: Trends and Developments (e-Book); Frontiers Media: Lausanne, Switzerland, 2019. [Google Scholar] [CrossRef]
- Li, Z.; Askim, J.R.; Suslick, K.S. The Optoelectronic Nose: Colorimetric and Fluorometric Sensor Arrays. Chem. Rev. 2019, 119, 231–292. [Google Scholar] [CrossRef] [PubMed]
- Di Natale, C.; Martinelli, E.; Magna, G.; Mandoj, F.; Monti, D.; Nardis, S.; Stefaneli, M.; Paolesse, R. Porphyrins for olfaction mimic: The Rome Tor Vergata approach. JPP 2018, 21, 769–781. [Google Scholar] [CrossRef]
- Dung, T.T.; Oh, Y.; Choi, S.-J.; Kim, I.-D.; Oh, M.-K.; Kim, M. Applications and Advances in Bioelectronic Noses for Odour Sensing. Sensors 2018, 18, 103. [Google Scholar] [CrossRef]
- Pohanka, M. Overview of Piezoelectric Biosensors, Immunosensors and DNA Sensors and Their Applications. Materials 2018, 11, 448. [Google Scholar] [CrossRef]
- Kuchmenko, T.A. Application of Piezoquarz Micro-Weighing Method in Analytical Chemistry; VSUET: Voronezh, Russia, 2001; p. 280. [Google Scholar]
- Fanget, S.; Hentz, S.; Puget, P.; Arcamone, J.; Matheron, M.; Colinet, E.; Andreucci, P.; Duraffourg, L.; Myers, E.; Roukes, M.L. Gas sensors based on gravimetric detection—A review. Sens. Actuators B 2011, 160, 804–821. [Google Scholar] [CrossRef]
- Hauptmann, P. Resonant sensors and applications. Sens. Actuators A 1991, 26, 371–377. [Google Scholar] [CrossRef]
- Wang, W.; Lee, K.; Kim, T.; Park, I.; Yang, S. A novel wireless, passive CO2 sensor incorporating a surface acoustic wave reflective delay line. Smart Mater. Struct. 2007, 16, 1382–1389. [Google Scholar] [CrossRef]
- Glen McHale, G. Generalized concept of shear horizontal acoustic plate mode and Love wave sensors. Meas. Sci. Technol. 2003, 14, 1847–1853. [Google Scholar] [CrossRef]
- Li, M.; Tang, H.X.; Roukes, M.L. Ultra-sensitive NEMS-based cantilevers for sensing, scanned probe and very high-frequency applications. Nat. Nanotechnol. 2007, 2, 114–120. [Google Scholar] [CrossRef]
- Pepper, J.; Noring, R.; Klempner, M.; Cunningham, B.T.; Petrovich, A.; Bousquet, R.; Clapp, C.; Brady, J.; Hugh, B. Detection of proteins and intact microorganisms using microfabricated flexural plate wave resonator arrays. Sens. Actuators B 2003, 96, 565–575. [Google Scholar] [CrossRef]
- Mujahid, A.; Dickert, F.L. Surface Acoustic Wave (SAW) for Chemical Sensing Applications of Recognition Layers. Sensors 2017, 17, 2716. [Google Scholar] [CrossRef]
- Korotcenkov, G. Materials for Piezoelectric-Based Gas Sensors. In Handbook of Gas Sensor Materials: Properties, Advantages and Shortcomings for Applications; Conventional Approaches. Integrated Analytical Systems; Springer: New York, NY, USA, 2013; Volume 1, pp. 307–328. ISBN 978-1-4614-7165-3. [Google Scholar]
- Mile, E.; Jourdan, G.; Bargatin, I.; Marcoux, C.; Labarthe, S.; Kharrat, C.; Andreucci, P.; Hentz, S.; Colinet, E.; Duraffourg, L. In-plane nanoelectromechanical resonators based on silicon nanowire piezoresistive detection. Nanotechnology 2010, 21, 165504. [Google Scholar] [CrossRef] [Green Version]
- Newell, W.E. Miniaturization of Tuning Forks. Science 1964, 161, 1320–1326. [Google Scholar] [CrossRef]
- Carr, D.W.; Evoy, S.; Sekaric, L.; Craighead, H.G.; Parpia, J.M. Measurement of mechanical resonance and losses in nanometer scale silicon wires. Appl. Phys. Lett. 1999, 75, 920–922. [Google Scholar] [CrossRef] [Green Version]
- Hu, Z.; Thundat, T.; Warmack, R.J.J. Investigation of adsorption and absorption-induced stresses using microcantilever sensors. J. Appl. Phys. 2001, 90, 427–431. [Google Scholar] [CrossRef]
- Hillman, A.R. The EQCM: Electrogravimetry with a light touch. J. Solid State Electrochem. 2011, 15, 1647–1660. [Google Scholar] [CrossRef]
- Sauerbrey, G. Verwendung von Schwingquarzen zur Wägung dünner Schichten und zurMikrowägung. Z. Phys. 1959, 155, 206–222. [Google Scholar] [CrossRef]
- Tonda-turo, C.; Carmagnola, I.; Ciardelli, G. Quartz crystal microbalance with dissipation monitoring: A powerful method to predict the in vivo behavior of bioengineered surfaces. Front. Bioeng. Biotechnol. 2018, 6, 158. [Google Scholar] [CrossRef]
- Miller, S.A.; Hiatt, L.A.; Robert, G.; Keil, R.G.; Wright, D.W.; Cliffel, D.E. Multifunctional nanoparticles as simulants for a gravimetric immunoassay. Anal. Bioanal. Chem. 2011, 399, 1021–1029. [Google Scholar] [CrossRef]
- De Giglio, E.; Trapani, A.; Cafagna, D.; Sabbatini, L.; Cometa, S. Dopamine-loaded chitosan nanoparticles: Formulation and analytical characterization. Anal. Bioanal. Chem. 2011, 400, 1997–2002. [Google Scholar] [CrossRef]
- Wang, L.; Gan, X.-X. Biomolecule-functionalized magnetic nanoparticles for flow-through quartz crystal microbalance immunoassay of aflatoxin B1. Bioprocess Biosyst. Eng. 2009, 32, 109–116. [Google Scholar] [CrossRef]
- Nam, D.H.; Lee, J.-O.; Sang, B.-I.; Won, K.; Kim, Y.H. Silaffin Peptides as a Novel Signal Enhancer for Gravimetric Biosensors. Appl. Biochem. Biotechnol. 2013, 170, 25–31. [Google Scholar] [CrossRef]
- Ali, M.G.S.; Elsyed, N.Z.; Abdel Fattah, A.M.; Ali Gharieb, A. Loss mechanisms in piezoceramic materials. J. Comput. Electron. 2012, 11, 196–202. [Google Scholar] [CrossRef]
- Lin, Z.-Q.; Gea, H.C.; Liu, S.-T. Design of piezoelectric energy harvesting devices subjected to broadband random vibrations by applying topology optimization. Acta Mech. Sin. 2011, 27, 730–737. [Google Scholar] [CrossRef]
- Chen, Y.Y.; Wang, J.; Du, J.K.; Yang, J.S. Effects of mass layer imperfect bonding on the electrical impedance of a quartz crystal microbalance. Sci. China Phys. Mech. Astron. 2013, 56, 2186–2191. [Google Scholar] [CrossRef]
- Sharapov, V.; Sotula, Z.; Kunickaya, L. The Technologies of PEAT Synthesis. In Piezo-Electric Electro-Acoustic Transducers; Microtechnology and MEMS; Springer: Cham, Sitzerland, 2014; pp. 91–113. [Google Scholar] [CrossRef]
- Chang, J.W.; Jung, M.; Kim, S.G.; Shim, S.B.; Kim, J.; Lee, S.G.; Yoo, Y.; Kim, B. High-quality nanomechanical resonator based on a defect-free gold nanowire. J. Korean Phys. Soc. 2013, 63, 263–268. [Google Scholar] [CrossRef]
- Naumenko, N.; Solie, L. Optimal cuts of Langasite, La3Ga5SiO14 for SAW devices. IEEE Trans. Ultrason. Ferroelectr. 2001, 48, 530–537. [Google Scholar] [CrossRef]
- Wang, X.; Wang, W.; Li, H.; Fu, C.; Ke, Y.; He, S. Development of a SnO2/CuO-coated surface acoustic wave-based H2S sensor with switch-like response and recovery. Sens. Actuators B 2012, 169, 10–16. [Google Scholar] [CrossRef]
- Hristova-Vasileva, T.; Petkov, K.; Vassilev, V.; Arnaudov, A. Piezoelectric Crystal Sensor for Ammonia Detection. In Nanotechnological Basis for Advanced Sensors; Reithmaier, J., Paunovic, P., Kulisch, W., Popov, C., Petkov, P., Eds.; NATO Science for Peace and Security Series B: Physics and Biophysics; Springer: Dordrecht, The Netherlands, 2011; pp. 439–443. [Google Scholar]
- Wang, X.; Cui, F.; Lin, J.; Ding, B.; Yu, J.; Al-Deyab, S.S. Functionalized nanoporous TiO2 fibers on quartz crystal microbalance platform for formaldehyde sensor. Sens. Actuators B 2012, 171–172, 658–665. [Google Scholar] [CrossRef]
- Latif, U.; Rohrer, A.; Lieberzeit, P.A.; Dickert, F.L. QCM gas phase detection with ceramic materials-VOCs and oil vapors. Anal. Bioanal. Chem. 2011, 400, 2457–2462. [Google Scholar] [CrossRef]
- Bernabei, M.; Pennazza, G.; Santonico, M.; Corsi, C.; Roscioni, C.; Paolesse, R.; Di Natale, C.; D’Amico, A. A preliminary study on the possibility to diagnose urinary tract cancers by an electronic nose. Sens. Actuators B 2008, 131, 1–4. [Google Scholar] [CrossRef]
- D’Amico, A.; Pennazza, G.; Santonico, M.; Martinelli, E.; Roscioni, C.; Galluccio, G.; Paolesse, R.; Di Natale, C. An investigation on electronic nose diagnosis of lung cancer. Lung Cancer 2010, 68, 170–176. [Google Scholar] [CrossRef]
- Bartolazzi, A.; Santonico, M.; Pennazza, G.; Martinelli, E.; Paolesse, R.; D’Amico, A.; Di Natale, C. A sensor array and GC study about VOCs and cancer cells. Sens. Actuators B 2010, 146, 483–488. [Google Scholar] [CrossRef]
- Capuano, R.; Spitalieri, P.; Talarico, R.V.; Catini, A.; Domakoski, A.C.; Martinelli, E.; Scioli, M.G.; Orlandi, A.; Cicconi, R.; Paolesse, R.; et al. Volatile compounds emission from teratogenic human pluripotent stem cells observed during their differentiation in vivo. Sci. Rep. 2018, 1, 11056. [Google Scholar] [CrossRef]
- Stefanelli, M.; Magna, G.; Zurlo, F.; Caso, M.F.; di Bartolomeo, E.; Antonaroli, S.; Venanzi, M.; Paolesse, R.; Di Natale, C.; Monti, D. Chiral Selectivity of Porphyrin-ZnO Nanoparticle Conjugates. ACS Appl. Mater. Interfaces 2019, 11, 12077–12087. [Google Scholar] [CrossRef]
- Lvova, L.; Mastroianni, M.; Pomarico, G.; Santonico, M.; Pennazza, G.; Di Natale, C.; Paolesse, R.; D’Amico, A. Carbon Nanotubes Modified with Porphyrin Units for Chemical Sensing of Gaseous Phase. Sens. Actuators B 2012, 170, 163–171. [Google Scholar] [CrossRef]
- Gao, N.; Dong, J.; Liu, M.; Ning, B.; Cheng, C.; Guo, C.; Zhou, C.; Peng, Y.; Bai, J.; Gao, Z. Development of molecularly imprinted polymer films used for detection of profenofos based on a quartz crystal microbalance sensor. Analyst 2012, 137, 1252–1258. [Google Scholar] [CrossRef]
- Tretjakov, A.; Syritski, V.; Reut, J.; Boroznjak, R.; Volobujeva, O.; Öpik, A. Surface molecularly imprinted polydopamine films for recognition of immunoglobulin G. Microchim. Acta 2013, 180, 1433–1442. [Google Scholar] [CrossRef]
- Wangchareansak, T.; Sangma, C.; Choowongkomon, K.; Dickert, F.; Lieberzeit, P. Surface molecular imprints of WGA lectin as artificial receptors for mass-sensitive binding studies. Anal. Bioanal. Chem 2011, 400, 2499–2506. [Google Scholar] [CrossRef]
- Jenik, M.; Seifner, A.; Lieberzeit, P.; Dickert, F.L. Pollen-imprinted polyurethanes for QCM allergen sensors. Anal. Bioanal. Chem. 2009, 394, 523–528. [Google Scholar] [CrossRef]
- Wangchareansak, T.; Sangma, C.; Ngernmeesri, P.; Thitithanyanont, A.; Lieberzeit, P.A. Self-assembled glucosamine monolayers as biomimetic receptors for detecting WGA lectin and influenza virus with a quartz crystal microbalance. Anal. Bioanal. Chem. 2013, 405, 6471–6478. [Google Scholar] [CrossRef]
- Chunta, S.; Suedee, R.; Lieberzeit, P.A. Low-Density Lipoprotein Sensor Based on Molecularly Imprinted Polymer. Anal. Chem. 2016, 88, 1419–1425. [Google Scholar] [CrossRef]
- Chunta, S.; Suedee, R.; Lieberzeit, P.A. High-density lipoprotein sensor based on molecularly imprinted polymer. Anal. Bioanal. Chem 2018, 410, 875–883. [Google Scholar] [CrossRef]
- Ayad, M.M.; El-Hefnawey, G.; Torad, N.L. A sensor of alcohol vapours based on thin polyaniline base film and quartz crystal microbalance. J. Hazard. Mater. 2009, 168, 85–88. [Google Scholar] [CrossRef]
- Yan, Y.; Lu, D.; Zhou, H.; Hou, H.; Zhang, T.; Wu, L.; Cai, L. Polyaniline-Modified Quartz Crystal Microbalance Sensor for Detection of Formic Acid Gas. Water Air Soil Pollut. 2012, 223, 1275–1280. [Google Scholar] [CrossRef]
- Ayad, M.M.; Torad, N.L. Quartz crystal microbalance sensor for detection of aliphatic amines vapours. Sens. Actuators B 2010, 147, 481–487. [Google Scholar] [CrossRef]
- Sun, P.; Jiang, Y.; Xie, G.; Du, X.; Hu, J. A room temperature supramolecular-based quartz crystal microbalance (QCM) methane gas sensor. Sens. Actuators B 2009, 141, 104–108. [Google Scholar] [CrossRef]
- Shaposhnik, A.V.; Zvyagin, A.A.; Yukish, V.A.; Ryabtsev, S.V.; Domashevskaya, E.P.; Kotov, V.V. Piezogravimetrical Toluene Concentration Sensor. Russian Patent # 2376590, 2007. Available online: https://russianpatents.com/patent/237/2376590 (accessed on 26 August 2019).
- Rusanova, T.Y.; Kalach, A.V.; Rumyantseva, S.S.; Shtykov, S.N.; Ryzhkina, I.S. Determination of Volatile Organic Compounds Using Piezosensors Modified with the Langmuir–Blodgett Films of Calix[4] resorcinarene. J. Anal. Chem. 2009, 64, 1270–1274. [Google Scholar] [CrossRef]
- Kuchmenko, T.A.; Shogenov, Y.K.; Grazhulene, S.S.; Redkin, A.N. Quartz crystal microbalance determination of vapors of volatile organic compounds on carbon nanotubes under batch conditions. J. Anal. Chem. 2012, 67, 21–27. [Google Scholar]
- Lim, C.; Wang, W.; Yang, S.; Lee, K. Development of SAW-based multi-gas sensor for simultaneous detection of CO2 and NO2. Sens. Actuators B 2011, 154, 9–16. [Google Scholar] [CrossRef]
- Kuchmenko, T.A.; Umarkhanov, R.U.; Kochetova, Z.Y.; Bel’skikh, N.V. Development of a Piezosensor-Based Transducer, Gas Analyzer, and Ammonia Detector. J. Anal. Chem. 2012, 67, 930–937. [Google Scholar] [CrossRef]
- Kuchmenko, T.A.; Umarkhanov, R.U. Peculiarities of microweighing of trace quantities of alkylamines on polymer and solid-state thin films. J. Anal. Chem. 2013, 68, 368–375. [Google Scholar] [CrossRef]
- Kuchmenko, T.A.; Mishina, A.A. Sorption specifics of volatile amines on thin films of acid-base indicators. J. Anal. Chem. 2011, 66, 701–708. [Google Scholar] [CrossRef]
- Kim, N.; Park, I.-S.; Kim, D.-K. High-sensitivity detection for model organophosphorus and carbamate pesticide with quartz crystal microbalance-precipitation sensor. Biosens. Bioelectron. 2007, 22, 1593–1599. [Google Scholar] [CrossRef]
- Liu, S.; Zheng, Z.; Li, X. Advances in pesticide biosensors: Current status, challenges, and future perspectives. Anal. Bioanal. Chem. 2013, 405, 63–90. [Google Scholar] [CrossRef]
- Kuchmenko, T.A.; Pogrebnaya, D.A.; Shuba, A.A. Application of principal component analysis and discriminant analysis using latent structure regression to the identification of highly volatile organic compounds in gas mixtures based on piezoelectric quartz microweighing parameters. J. Anal. Chem. 2013, 68, 652–661. [Google Scholar] [CrossRef]
- Reddy, B.S.K.; Kumar, K.R.; Balakrishnaiah, G.; Gopal, K.R.; Reddy, R.R.; Sivakumar, V.; Arafath, S.M.; Lingaswamy, A.P.; Pavankumari, S.; Umadevi, K.; et al. Ground-based in situ measurements of near-surface aerosol mass concentration over Anantapur: Heterogeneity in source impacts. Adv. Atmos. Sci. 2013, 30, 235–246. [Google Scholar] [CrossRef]
- Favrat, O.; Gavoille, J.; Aleya, L.; Monteil, G. Real Time Study of Detergent Concentration Influence on Solid Fatty Acid Film Removal Processes. J. Surfact. Deterg. 2013, 16, 213–219. [Google Scholar] [CrossRef]
- Zhang, C.; Luo, S. Piezoelectric atrazine sensor based on a molecularly imprinted film of titanium dioxide. Microchim. Acta 2011, 175, 63–68. [Google Scholar] [CrossRef]
- Qin, S.; Tang, X.; Du, X.; Zhu, L.; Wei, Y.; Kwon, O.; Fang, J.; Wang, P.; Zhu, D.M. Mechanical and Viscoelastic Properties of Polymer Layers on Solid-Liquid Interfaces. In IUTAM Symposium on Surface Effects in the Mechanics of Nanomaterials and Heterostructures; Cocks, A., Wang, J., Eds.; IUTAM Bookseries (closed); Springer: Dordrecht, The Netherlands, 2013; Volume 31, pp. 217–228. [Google Scholar]
- Simbeck, T.; Hammer, M.M.; Thomaier, S.; Stock, C.; Riedl, E.; Gores Heiner, J. Kinetics of adsorption of poly(vinylimidazole) (PVI) onto copper surfaces investigated by quartz crystal microbalance studies. J. Solid State Electrochem. 2012, 16, 3467–3472. [Google Scholar] [CrossRef]
- Davide, F.A.M.; D’Amico, A. Pattern recognition from sensor arrays: Theoretical considerations. Sens. Actuators A 1992, 32, 507–518. [Google Scholar] [CrossRef]
- Lieberzeit, P.A.; Rehman, A.; Iqbal, N.; Najafi, B.; Dickert, F.L. QCM sensor array for monitoring terpene emissions from odoriferous plants. Monatsh. Chem 2009, 140, 947–952. [Google Scholar] [CrossRef]
- Sun, X.; Zhang, Y.; Shao, J.; Shen, L.; Qian, H.; Zhu, W. A quartz crystal microbalance-based Immunosensor for Shrimp Allergen Determination in Food. Eur. Food Res. Technol. 2010, 231, 563–570. [Google Scholar]
- Kuchmenko, T.A.; Drozdova, E.V. Application of piezosensors array for express analysis of free volatile components from phenolformaldehyde plastics. Anal. Control 2014, 18, 66–75. [Google Scholar]
- Chuikov, A.M.; Peregudov, A.N.; Isaev, A.A.; Kalach, A.V. The possibility of using the “Electronic nose” system for evaluation of gases and vapors toxic level while exploiting some building materials. Technol. Technosphere Saf. 2011, 36, 17–25. [Google Scholar]
- Mujahid, A.; Dickert, F.L. Monitoring automotive oil degradation: Analytical tools and onboard sensing technologies. Anal. Bioanal. Chem. 2012, 404, 1197–1209. [Google Scholar] [CrossRef]
- Chen, S.; Wang, Y.; Choi, S. Applications and Technology of Electronic Nose for Clinical Diagnosis. Open J. Appl. Biosens. 2013, 2, 39–50. [Google Scholar] [CrossRef] [Green Version]
- Lieberzeit, P.A.; Rehman, A.; Najafi, B.; Dickert, F.L. Real-life application of a QCM-based e-nose: Quantitative characterization of different plant-degradation processes. Anal. Bioanal. Chem. 2008, 391, 2897–2903. [Google Scholar] [CrossRef]
- López de Lerma, N.; Moreno, J.; Peinado, R.A. Determination of the Optimum Sun-Drying Time for Vitis vinifera L. cv. Tempranillo Grapes by E-nose Analysis and Characterization of Their Volatile Composition. Food Bioprocess Technol. 2014, 7, 732–740. [Google Scholar] [CrossRef]
- Hui, G.; Wu, Y.; Ye, D.; Ding, W. Fuji Apple Storage Time Predictive Method Using Electronic Nose. Food Anal. Methods 2013, 6, 82–88. [Google Scholar]
- Liu, H.; Zeng, F.K.; Wang, Q.H.; Wu, H.S.; Tan, L.H. Studies on the chemical and flavor qualities of white pepper (Piper nigrum L.) derived from five new genotypes. Eur. Food Res. Technol. 2013, 237, 245–251. [Google Scholar] [CrossRef]
- Jo, D.; Kim, G.R.; Yeo, S.H.; Jeong, Y.J.; Noh, B.S.; Kwon, J.H. Analysis of aroma compounds of commercial cider vinegars with different acidities using SPME/GC-MS, electronic nose, and sensory evaluation. Food Sci. Biotechnol. 2013, 22, 1559–1565. [Google Scholar] [CrossRef]
- Kuchmenko, T.A.; Lisitskaya, R.P.; Khoperskaya, M.A.; Strelnikova Yu, I.; Koblyakova, O.S. Control of the content of food fragrantizers in confectionary masses with the use of sorption gas sensors. Anal. Control 2012, 16, 399–405. [Google Scholar]
- Li, Z.; Wang, N.; Vijaya Raghavan, G.S.; Vigneault, C. Volatiles Evaluation and Dielectric Properties Measurements of Chinese Spirits for Quality Assessment. Food Bioprocess Technol. 2011, 4, 247–253. [Google Scholar] [CrossRef]
- Cotrone, S.; Cafagna, D.; Cometa, S.; De Giglio, E.; Magliulo, M.; Torsi, L.; Sabbatini, L. Microcantilevers and organic transistors: Two promising classes of label-free biosensing devices which can be integrated in electronic circuits. Anal. Bioanal. Chem. 2012, 402, 1799–1811. [Google Scholar] [CrossRef]
- Vera, L.; Aceña, L.; Guasch, J.; Boqué, R.; Mestres, M.; Busto, O. Characterization and classification of the aroma of beer samples by means of an MS e-nose and chemometric tools. Anal. Bioanal. Chem. 2011, 399, 2073–2081. [Google Scholar] [CrossRef]
- Hu, X.; Mallikarjunan, P.; Vaughan, D. Development of non-destructive methods to evaluate oyster quality by electronic nose technology. Sens. Instrumen. Food Qual. 2008, 2, 51–57. [Google Scholar] [CrossRef]
- Delgado-Rodriguez, M.; Ruiz-Montoya, M.; Giraldez, I.; Lopez, R.; Madejon, E.; Diaz, M.J. Use of electronic nose and GC-MS in detection and monitoring some VOC. Atmos. Environ. 2012, 51, 278–285. [Google Scholar] [CrossRef]
- Kuchmenko, T.A.; Pogrebnaya, D.A.; Khripushin, V.V. The new instrumental methods for assessment organoleptic indicators of milk. Anal. Control 2012, 16, 289–298. [Google Scholar]
- Cano, M.; Roales, J.; Castillero, P.; Mendoza, P.; Palma, M.; Calero, A.M.; Jiménez-Ot, C.; Pedrosa, J.M. Improving the training and data processing of an electronic olfactory system for the classification of virgin olive oil into quality categories. Sens. Actuators B 2011, 160, 916–922. [Google Scholar] [CrossRef]
- Kuchmenko, T.A.; Shuba, A.A.; Belskikh, N.V. The identification parameters of organic substances in multisensors piezoquartz microbalance. Anal. Control 2012, 16, 151–161. [Google Scholar]
- Penza, M.; Cassano, G.; Aversa, P.; Antolini, F.; Cusano, A.; Consales, M.; Giordano, M.; Nicolais, L. Carbon nanotubes-coated multi-transducing sensors for VOCs detection. Sens. Actuatots B 2005, 111–112, 171–180. [Google Scholar] [CrossRef]
- Chen, I.S.; Stawasz, M.; Di Mascio, S.K.; Welch, J.J.; Neuner, J.W.; Chen, P.S.H.; Di Meo, F., Jr. Micromachined chemical sensor with dual-transduction mechanisms. Appl. Phys. Lett. 2005, 86, 173510. [Google Scholar] [CrossRef]
- Wang, L.; Swensen, J.S. Dual-transduction-mode sensing approach for chemical detection. Sens. Actuators B 2012, 174, 366–372. [Google Scholar] [CrossRef]
- Kim, J.; Kim, S.; Ohashi, T.; Muramatsu, H.; Chang, S.M.; Kim, W.S. Construction of simultaneous SPR and QCM sensing platform. Bioprocess Biosyst. Eng. 2010, 33, 39–45. [Google Scholar] [CrossRef]
- Alpha MOS: Sensory Analysis Solutions. Available online: https://www.alpha-mos.com/heracles-smell-analysis (accessed on 24 August 2019).
- Sensigent. Available online: http://www.sensigent.com/products/cyranose.html (accessed on 24 August 2019).
- Airsense Analytics. Available online: https://airsense.com/en/products/portable-electronic-nose (accessed on 24 August 2019).
- Electronic Nose for Environmental. Available online: http://e-nose.asia (accessed on 24 August 2019).
- Owlstone Medical. Available online: https://www.owlstonemedical.com/products/lonestar/ (accessed on 24 August 2019).
- Narsaiah, K.; Jha, S.N. Nondestructive methods for quality evaluation of livestock products. J. Food Sci. Technol. 2012, 49, 342–348. [Google Scholar] [CrossRef]
- Lang, H.P.; Gerber, C. Microcantilever Sensors. In STM and AFM Studies on (Bio)molecular Systems: Unravelling the Nanoworld; Samorì, P., Ed.; Topics in Current Chemistry; Springer: Berlin/Heidelberg, Germany, 2008; Volume 285, pp. 1–27. [Google Scholar] [CrossRef]
- Chaudhary, M.; Gupta, A. Microcantilever-based Sensors. Def. Sci. J. 2009, 59, 634–641. [Google Scholar] [CrossRef] [Green Version]
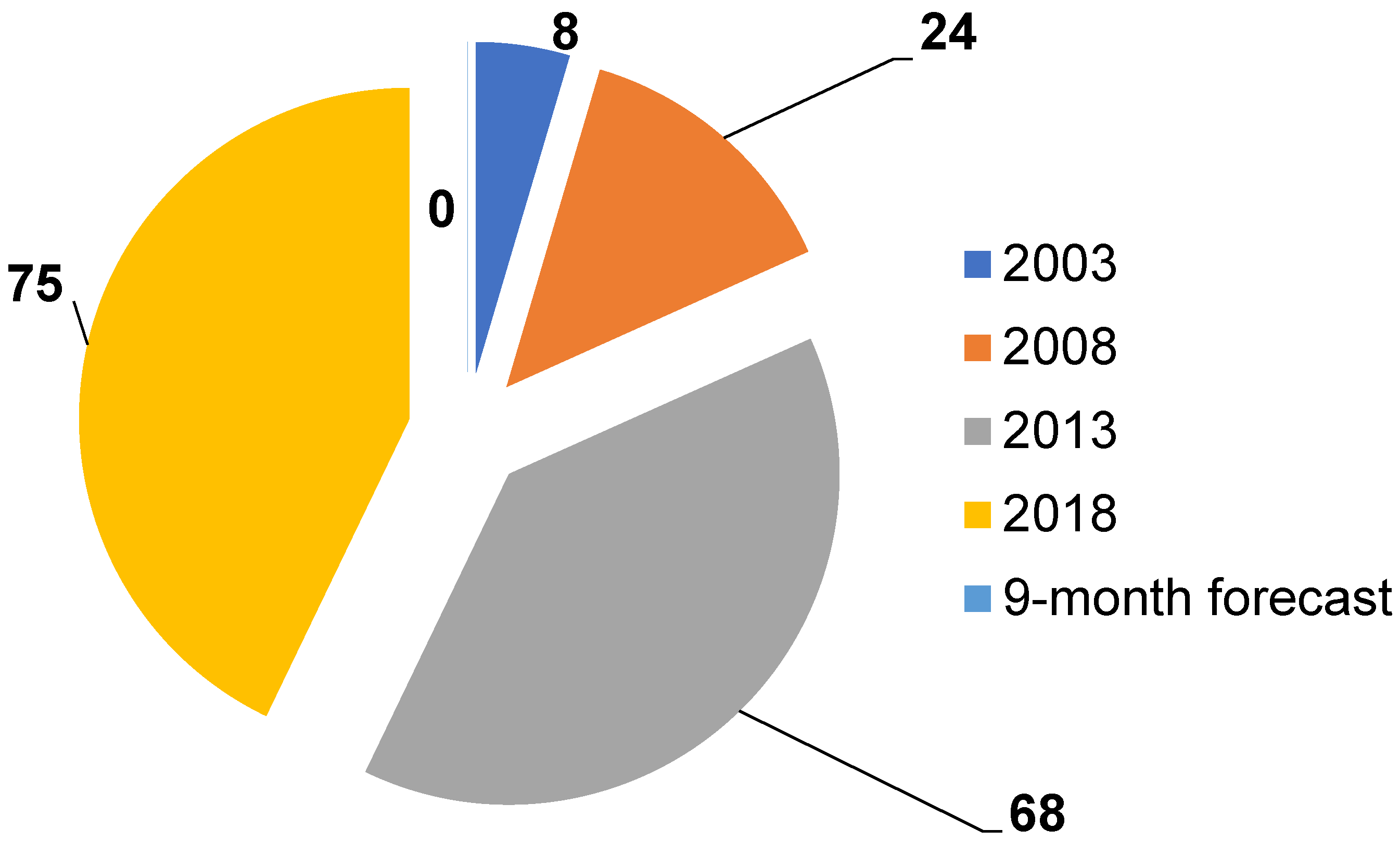
| Analysis Objects | Technique | Sensor Coating Type | Measuring System | Ref. |
|---|---|---|---|---|
| CO2 gas | SAW | Teflon | single sensor | [18] |
| proteins and intact microorganisms | FPW | surface-immobilized coatings of biotinylated anti-dog IgG and dog-IgG antibodies | chemical sensor arrays with differential outputs | [21] |
| cell adhesion, cytotoxicity, cell viability, cell phenomena | QCM-D | cell preincubated QCM-D sensors | ΔD–Δf measurements in the presence of different reagents | [30] |
| hemagglutinin (HA) glycoprotein of influenza virions (H5N1) | QCM | polyepitope-functionalized Au NPs | antibody–antigen binding-based gravimetric immunosensor | [31] |
| dopamine (DA) | QCM-D | chitosan nanoparticles (CSNPs) | piezoelectric sensors array with crown ether coatings | [32] |
| aflatoxin B1 | QCM | biomolecule-functionalized magnetic nanoparticles | immunoassay | [33] |
| different proteins using recombinant DNA technology | QCM | silica particles induced by the GFP-R1 chimeric protein | single immunosensor | [34] |
| H2S vapor | SAW | SnO2/CuO coatings | single sensor | [41] |
| NH3 | QCM | NiCl2 and AgCl films | single sensor | [42,66] |
| formaldehyde | QCM | nanoporous TiO2 fibers | single sensor | [43] |
| VOCs: ethanol, n-propanol, n-butanol, n-hexane, n-heptane, n-/iso-octane, n-decane and monitoring emanation of degraded engine oil | QCM | titanate sol–gel layers imprinted with carbonic acids | single sensor | [44] |
| VOC cancer markers | QCM | porphyrins and their metallic complexes | e-nose composed from 8–12-element chemical sensor array | [45,46,47,48] |
| chiral VOCs | QCM | porphyrin–ZnO nanoparticle conjugates | single sensor | [49] |
| n-butanol | QCM | electropolymerized porphyrin-containing coating | single sensor | [50] |
| profenofos | QCM | MIP based on 11-mercaptoundecanoic acid (MUA) | single sensor | [51] |
| immunoglobulin G (IgG) | QCM | MIP polydopamine films | single sensor | [52] |
| wheat germ agglutinin (WGA) lectin | QCM | MIP polyacrylic film | single sensor | [53] |
| pollen allergens (birch and nettle) | QCM | pollen-imprinted polyurethanes | single sensor | [54] |
| influenza viruses and other biomolecules | QCM | self-assembling glucosamine monolayers | single sensor | [55] |
| low-density (LDLs) and high-density (HDLs) lipoproteins in blood serum | QCM | MIPs from acrylic acid (AA), methacrylic acid (MAA), and N-vinylpyrrolidone (VP) monomers in different ratios | single sensors | [56,57] |
| methanol, ethanol, 2-propanol, and 1-propanol vapors | QCM | thin polyaniline film | single sensor | [58] |
| formic acid gas | QCM | thin polyaniline film | single sensor | [59] |
| aliphatic amine vapors | QCM | polyaniline/emeraldine base (PANI/EB) film | single sensor | [60] |
| CH4 gas | QCM | supramolecular cryptophane-A film deposited via electrospray method | single sensor | [61] |
| toluene | QCM | sensitive film coatings made of fulvic acids isolated from humus | single sensor | [62] |
| VOCs: ethanol, benzene, toluene, ethylbenzene, ethyl acetate, acetone, hexane, and cumene | QCM | Langmuir–Blodgett calix [4] resorcinarene | single sensor | [63] |
| VOCs: benzene, methylbenzene, 1,2-dimethylbenzene, ethylbenzene, isopropylbenzene, 1,2,4-trimethylbenzene, monoatomic aliphatic alcohols (C2—C9) with normal and isomeric structures | QCM | doped and nondoped multilayer carbon nanotubes (CNTs) | MAG-8 gas analyzer using e-nose methodology with an array of eight piezoelectric sensors | [64] |
| simultaneous detection of CO2 and NO2 | SAW | CO2-sensitive film (Teflon AF 2400) and an NO2-sensitive film (indium tin oxide) | multigas sensor | [65] |
| alkylamines | QCM | polymer and solid-state thin films, thin films of acid–base indicators | single sensor, gas analyzer | [67,68] |
| organophosphorus and carbamate pesticides | QCM | acetylcholinesterase (AChE) immobilized on QCM surface | single sensor | [69] |
| chain aliphatic acids | QCM | standard chromatographic coatings: polyethylene glycol PEG-2000 (PEG); the ethers: polyethylene glycol adipate (PEGA), phthalate (PEGP), etc.; the specific sorbents 18-crown-6 (18C6) and propolis (Pr) | MAG-8 analyzer using e-nose methodology with an array of eight piezoelectric sensors | [71] |
| variations in the size and concentration of aerosol particles | QCM | no coating | single sensor | [72] |
| stearic acid and homolog dissolution monitoring | QCM | no coating | single sensor | [73] |
| atrazine sensor | QCM | molecularly imprinted film of titanium dioxide | single sensor | [74] |
| adsorption of poly(vinylimidazole) (PVI) on Cu | QCM | no coating | single sensor | [76] |
| shrimp allergen determination in food | QCM | self-assembly of 1,6-hexanedithiol (HDT) and antishrimp antibodies | e-nose immunosensor system | [79] |
| monitoring terpene emissions from odoriferous plants | QCM | molecularly imprinted polymer (MIP) selectively interacting with alpha-pinene, thymol, estragol, linalool, and camphor | sensor array | [80] |
| free volatile components from phenolformaldehyde plastics | QCM | nonspecified | sensor array | [81] |
| composts of grass and pine characterization, alcohol and terpene detection | QCM | affinity materials and MIPs | sensor array of six piezosensors | [84] |
| volatile food flagrancies in confectionary masses | QCM | nonspecified | sensor array | [89] |
| organoleptic indicators of milk assessments | QCM | nonspecified | sensor array | [95] |
| wastewater quality assessments | QCM | MWCNTs, zirconium(IV) oxynitrate, biohydroxyapatite coatings | MAG-8 gas analyzer with six piezosensors | This work’s case study |
| Sensor | Coating | Coating Weight (µg) |
|---|---|---|
| Sensor 1 | Multiwalled carbon nanotubes (MWCNTs) oxidized by nitric acid | 5.03 |
| Sensor 2 | Zirconium(IV) oxynitrate | 4.03 |
| Sensor 3 | Biohydroxyapatite | 4.03 |
| Sensor 4 | Biohydroxyapatite | 2.15 |
| Sensor 5 | Zirconium(IV) oxynitrate | 2.12 |
| Sensor 6 | Multiwalled carbon nanotubes (MWCNTs) oxidized by nitric acid | 1.96 |
| Sample # | ΔFi | Sensor 1 | Sensor 2 | Sensor 3 | Sensor 4 | Sensor 5 | Sensor 6 | Si (Hz/s) |
|---|---|---|---|---|---|---|---|---|
| Potable Water | X ± ΔX | 6 ± 1 | 7 ± 1 | 12 ± 1 | 7 ± 1 | 6 ± 1 | 11 ± 1 | 125 ± 20 |
| Δ | 0.23 | 0.19 | 0.12 | 0.21 | 0.23 | 0.21 | 0.16 | |
| 04 | X ± ΔX | 12 ± 2 | 14 ± 2 | 25 ± 2 | 12 ± 2 | 12 ± 1 | 20 ± 2 | 485 ± 30 |
| Δ | 0.23 | 0.20 | 0.09 | 0.22 | 0.11 | 0.13 | 0.08 | |
| 01 | X ± ΔX | 13 ± 1 | 15 ± 1 | 26 ± 1 | 15 ± 2 | 12 ± 1 | 24 ± 2 | 480 ± 40 |
| Δ | 0.11 | 0.09 | 0.05 | 0.16 | 0.12 | 0.10 | 0.08 | |
| 51 | X ± ΔX | 12 ± 1 | 15 ± 2 | 24 ± 2 | 15 ± 2 | 12 ± 1 | 21 ± 2 | 470 ± 50 |
| Δ | 0.12 | 0.19 | 0.12 | 0.19 | 0.12 | 0.12 | 0.14 | |
| 05 | X ± ΔX | 12 ± 1 | 15 ± 1 | 24 ± 1 | 15 ± 2 | 12 ± 1 | 21 ± 2 | 445 ± 30 |
| Δ | 0.12 | 0.09 | 0.06 | 0.16 | 0.12 | 0.13 | 0.09 | |
| 47 | X ± ΔX | 12 ± 1 | 14 ± 1 | 25 ± 2 | 14 ± 1 | 11 ± 1 | 19 ± 1 | 420 ± 20 |
| Δ | 0.12 | 0.10 | 0.12 | 0.10 | 0.13 | 0.07 | 0.06 | |
| 49 | X ± ΔX | 12 ± 1 | 14 ± 1 | 24 ± 1 | 14 ± 1 | 12 ± 2 | 19 ± 1 | 430 ± 30 |
| Δ | 0.12 | 0.01 | 0.06 | 0.01 | 0.2 | 0.07 | 0.08 | |
| 48 | X ± ΔX | 11 ± 1 | 12 ± 1 | 24 ± 1 | 13 ± 2 | 11 ± 1 | 18 ± 2 | 420 ± 20 |
| Δ | 0.13 | 0.12 | 0.06 | 0.19 | 0.13 | 0.14 | 0.05 | |
| 50 | X ± ΔX | 12 ± 1 | 14 ± 1 | 24 ± 1 | 14 ± 1 | 11 ± 1 | 19 ± 1 | 435 ± 20 |
| Δ | 0.13 | 0.10 | 0.06 | 0.10 | 0.13 | 0.07 | 0.04 | |
| 52 | X ± ΔX | 10 ± 1 | 14 ± 1 | 24 ± 1 | 13 ± 1 | 12 ± 1 | 19 ± 1 | 350 ± 20 |
| Δ | 0.15 | 0.10 | 0.06 | 0.10 | 0.13 | 0.07 | 0.07 | |
| 02 | X ± ΔX | 10 ± 1 | 14 ± 1 | 24 ± 1 | 14 ± 1 | 11 ± 1 | 19 ± 1 | 420 ± 20 |
| Δ | 0.01 | 0.10 | 0.06 | 0.10 | 0.13 | 0.13 | 0.08 | |
| 03 | X ± ΔX | 13 ± 1 | 14 ± 1 | 23 ± 1 | 13 ± 1 | 11 ± 1 | 19 ± 1 | 440 ± 20 |
| Δ | 0.11 | 0.10 | 0.06 | 0.10 | 0.03 | 0.03 | 0.06 | |
| 39 | X ± ΔX | 14 ± 1 | 14 ± 1 | 24 ± 1 | 14 ± 1 | 11 ± 1 | 19 ± 1 | 460 ± 30 |
| Δ | 0.10 | 0.10 | 0.06 | 0.10 | 0.13 | 0.07 | 0.08 | |
| 40 | X ± ΔX | 14 ± 1 | 14 ± 1 | 23 ± 1 | 13 ± 1 | 11 ± 1 | 19 ± 1 | 420 ± 20 |
| Δ | 0.10 | 0.01 | 0.06 | 0.10 | 0.01 | 0.07 | 0.06 | |
| 42 | X ± ΔX | 13 ± 1 | 14 ± 1 | 23 ± 1 | 13 ± 1 | 11 ± 1 | 19 ± 1 | 400 ± 30 |
| Δ | 0.21 | 0.10 | 0.06 | 0.10 | 0.01 | 0.08 | 0.08 | |
| 41 | X ± ΔX | 12 ± 1 | 14 ± 1 | 23 ± 1 | 15 ± 1 | 11 ± 1 | 20 ± 2 | 490 ± 20 |
| Δ | 0.12 | 0.10 | 0.06 | 0.09 | 0.13 | 0.15 | 0.04 |
| Sample # | Mn | Al | Pb | Cr | Fe | Ni | Zn | Cu | Cd |
|---|---|---|---|---|---|---|---|---|---|
| 01 | 0.062 | 0.086 | 0.0045 | <0.01 | 9.42 | 0.0031 | 0.09 | 0.0089 | <0.0001 |
| 02 | 0.04 | 0.20 | <0.001 | <0.01 | 0.61 | 0.0016 | 0.12 | 0.0097 | <0.0001 |
| 03 | 0.52 | 2,6 | 0.520 | 0.11 | 52 | 0.018 | 0.8 | 0.60 | 0.0027 |
| 04 | 0.102 | 0.64 | <0.001 | <0.01 | 2.4 | 0.0029 | 0.12 | 0.034 | <0.0001 |
| 05 | 0.181 | 2.24 | - | <0.01 | 6.8 | 0.0096 | 0.55 | 0.10 | 0.0004 |
| 39 | 0.095 | 0.56 | 0.0022 | 0.039 | 1.0 | 0.0049 | 0.16 | 0.031 | 0.0002 |
| 40 | 0.16 | 1.46 | 0.0047 | 0.012 | 1.2 | 0.0033 | 0.12 | 0.040 | 0.0002 |
| 41 | 0.092 | 0.24 | 0.0120 | <0.01 | 1.8 | 0.0048 | 0.18 | 0.050 | 0.0015 |
| 42 | 0.17 | 0.63 | 0.0033 | 0.013 | 2.8 | 0.0044 | 0.43 | 0.108 | 0.0003 |
| 47 | 0.118 | 2.12 | 0.011 | 0.025 | 4.4 | 0.030 | 0.33 | 0.096 | 0.0003 |
| 48 | 0.098 | 1.14 | 0.009 | <0.01 | 4.2 | 0.0058 | 0.98 | 0.036 | <0.0001 |
| 49 | 0.17 | 0.33 | 0.0041 | <0.01 | 1.9 | 0.011 | 0.20 | 0.048 | <0.0001 |
| 50 | 0.096 | 0.32 | 0.0029 | <0.01 | 2.7 | 0.061 | 0.11 | 0.053 | 0.0002 |
| 51 | 0.16 | 0.25 | 0.0014 | <0.01 | 0.43 | 0.0016 | 0.013 | 0.0055 | <0.0001 |
| 52 | 0.126 | 0.148 | 0.0026 | <0.01 | 0.26 | 0.0014 | 0.011 | 0.004 | <0.0001 |
| Parameters | A13 | A14 | A15 * | A16 | A23 | A24 | A25 | A26 | A36 | A45 | A56 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Co2+ | 0.595 | 0.509 | - | - | - | - | - | - | - | - | - |
| Cr3+ | 0.536 | 0.474 | - | 0.442 | - | - | - | - | - | - | - |
| Crtotal | - | - | - | - | 0.496 | 0.456 | 0.493 | 0.434 | - | - | - |
| Cd2+ | - | - | - | - | 0.507 | 0.453 | - | 0.436 | - | - | - |
| V3+ | - | - | - | - | - | - | 0.757 | 0.529 | - | - | - |
| Cu2+ | - | - | - | - | - | - | 0.409 | 0.433 | - | - | - |
| As3+ | - | - | - | - | - | - | 0.435 | 0.409 | - | - | - |
| Pb2+ | - | - | - | - | - | - | 0.426 | 0.405 | - | - | - |
| Mn2+ | - | - | - | - | - | - | - | 0.459 | - | - | - |
| Phosphates (P) | 0.478 | 0.597 | - | - | - | - | - | - | - | - | - |
| S2− | - | - | - | - | 0.473 | - | - | 0.482 | - | - | - |
| F− | - | - | - | - | - | - | - | - | 0.797 | - | - |
| Fats | - | - | - | - | - | - | - | - | - | - | 0.422 |
| Phenols | - | - | - | - | - | - | - | - | - | 0.371 | - |
| Oil products | - | - | - | - | - | - | 0.422 | - | - | - | - |
| Projected Indicator | b0 | A12 | A13 | A14 | A23 | A24 | A25 | A26 | A34 | A46 | RMSEP | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Co2+ | β | −0.034 | - * | 0.281 | −0.129 | - | -- | - | - | - | - | 0.0027 |
| p | 0.012 | - | 0.063 | 0.100 | - | - | - | - | - | - | ||
| Cd2+ | β | 0.036 | - | - | - | - | −0.040 | - | 0.060 | - | −0.055 | 0.0007 |
| p | 0.056 | - | - | - | - | 0.05 | - | 0.030 | - | 0.037 | ||
| V3+ | β | 0.172 | - | - | - | - | −0.205 | 0.030 | 0.275 | - | −0.276 | 0.0003 |
| p | 0.001 | - | - | - | - | 0.001 | 0.001 | 0.001 | - | 0.001 | ||
| Phosphates | β | −26.7 | 20.7 | −193 | 131.8 | - | - | - | - | - | - | 2.12 |
| p | 0.011 | 0.017 | 0.032 | 0.009 | - | - | - | - | - | - | ||
| Fats | β | 5046 | - | - | - | −4266 | - | - | 3270 | −1415 | −3420 | 21.7 |
| p | 0.008 | - | - | - | 0.010 | - | - | 0.012 | 0.013 | 0.007 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuchmenko, T.A.; Lvova, L.B. A Perspective on Recent Advances in Piezoelectric Chemical Sensors for Environmental Monitoring and Foodstuffs Analysis. Chemosensors 2019, 7, 39. https://doi.org/10.3390/chemosensors7030039
Kuchmenko TA, Lvova LB. A Perspective on Recent Advances in Piezoelectric Chemical Sensors for Environmental Monitoring and Foodstuffs Analysis. Chemosensors. 2019; 7(3):39. https://doi.org/10.3390/chemosensors7030039
Chicago/Turabian StyleKuchmenko, Tatyana A., and Larisa B. Lvova. 2019. "A Perspective on Recent Advances in Piezoelectric Chemical Sensors for Environmental Monitoring and Foodstuffs Analysis" Chemosensors 7, no. 3: 39. https://doi.org/10.3390/chemosensors7030039
APA StyleKuchmenko, T. A., & Lvova, L. B. (2019). A Perspective on Recent Advances in Piezoelectric Chemical Sensors for Environmental Monitoring and Foodstuffs Analysis. Chemosensors, 7(3), 39. https://doi.org/10.3390/chemosensors7030039






