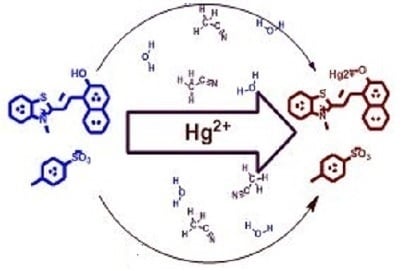
Colorimetric and Fluorescence-Based Detection of Mercuric Ion Using a Benzothiazolinic Spiropyran
Abstract
1. Introduction
2. Materials and Methods
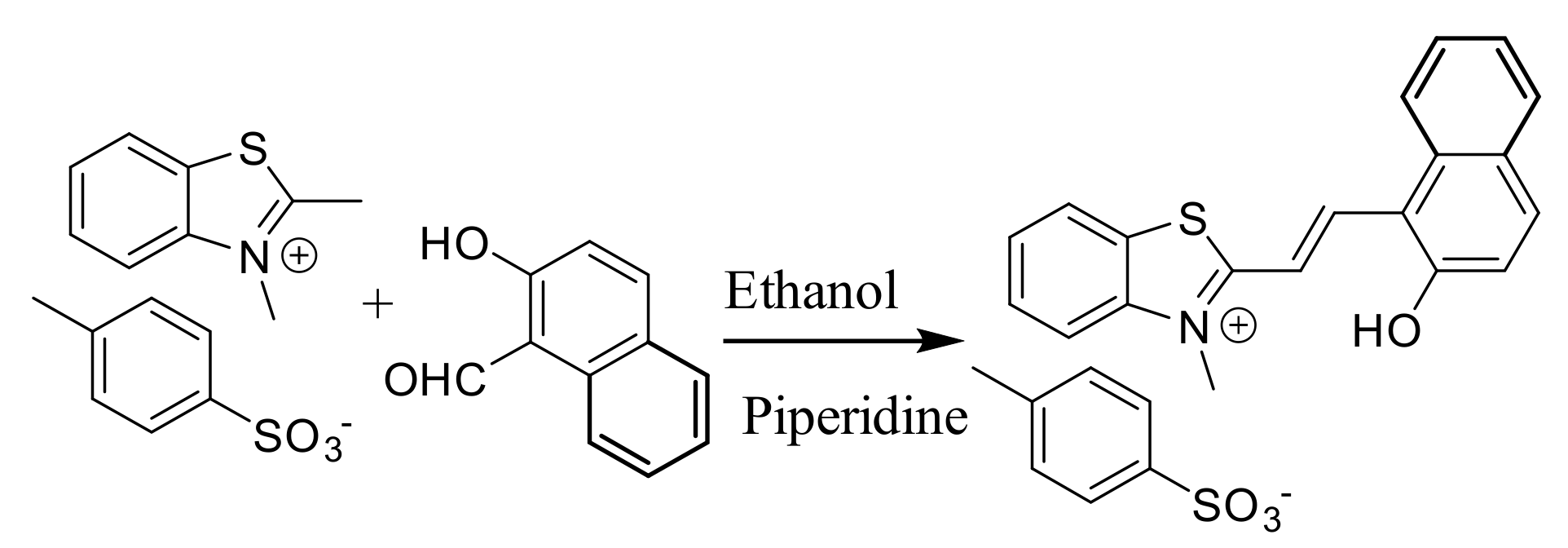
2.1. Synthesis
2.2. UV-Visible Studies
2.3. Procedure for Fluorescence Studies
2.4. Color Intensity Measurement Studies
3. Results and Discussion
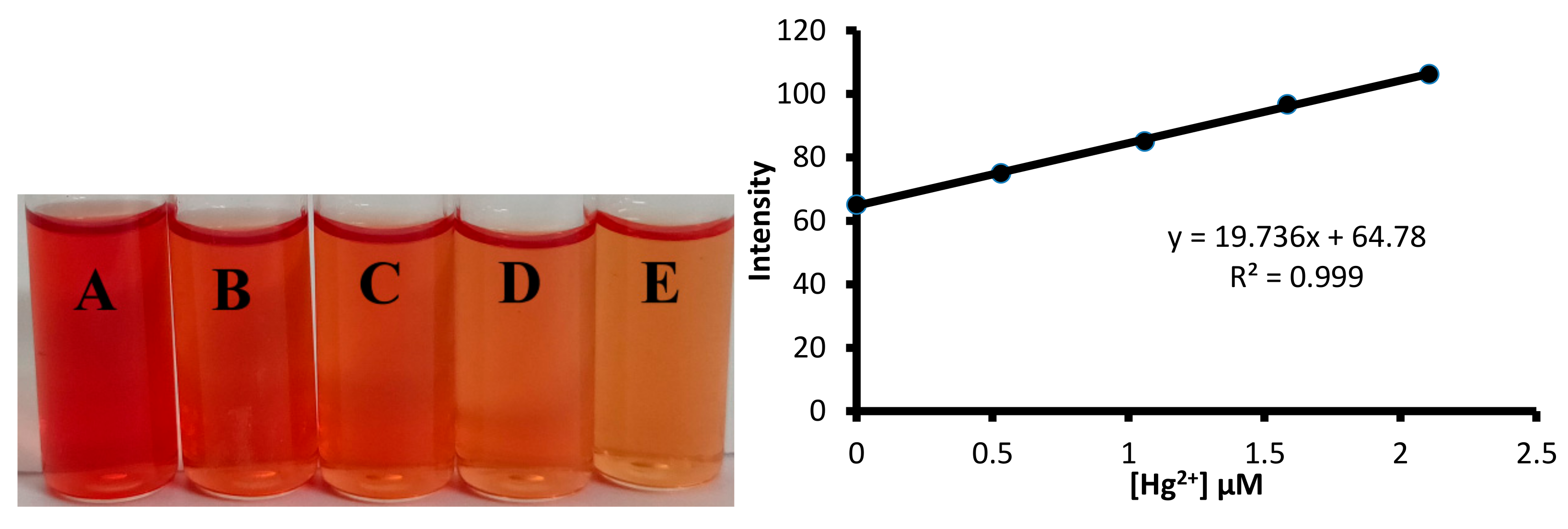
3.1. Naked Eye Detection
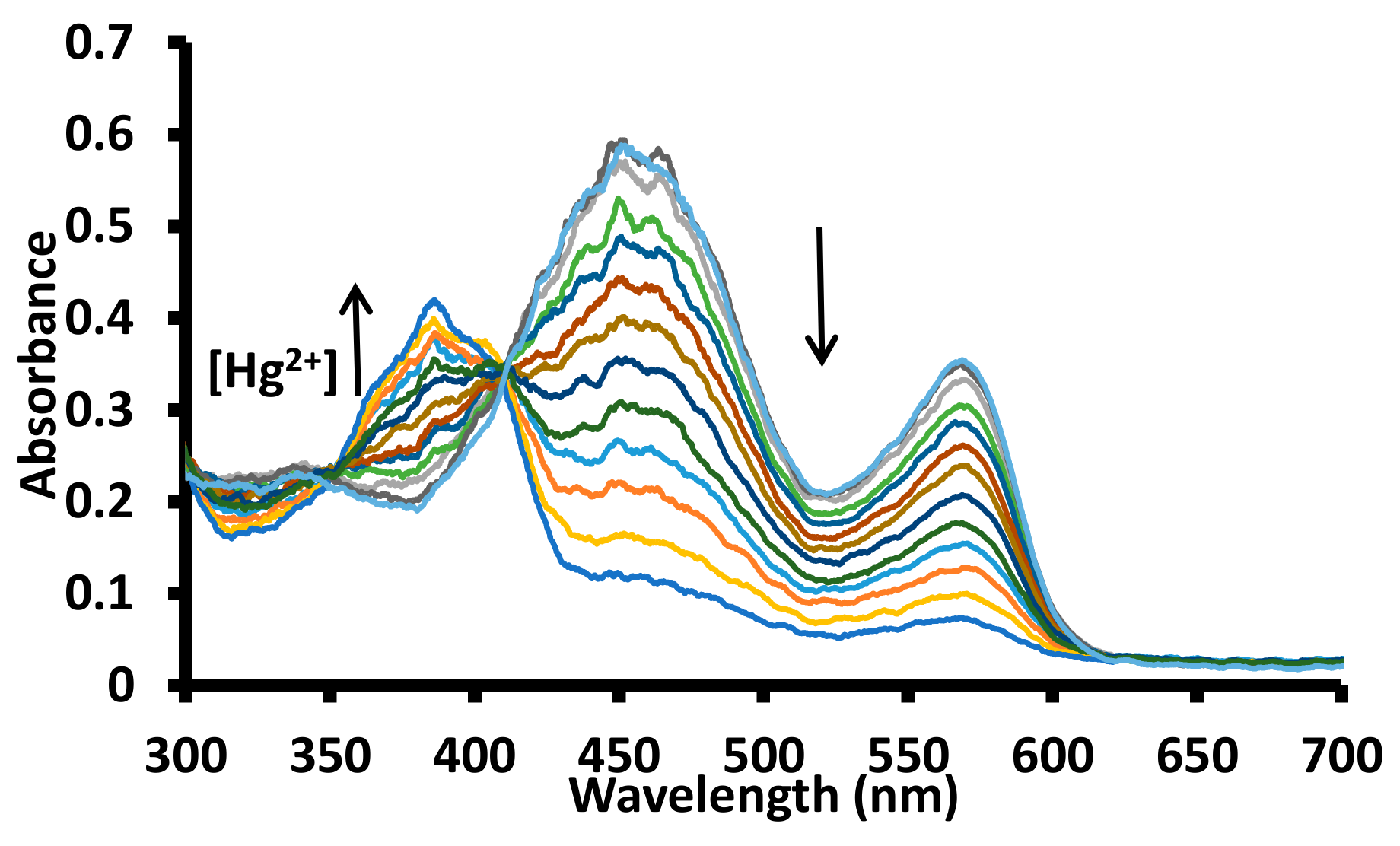
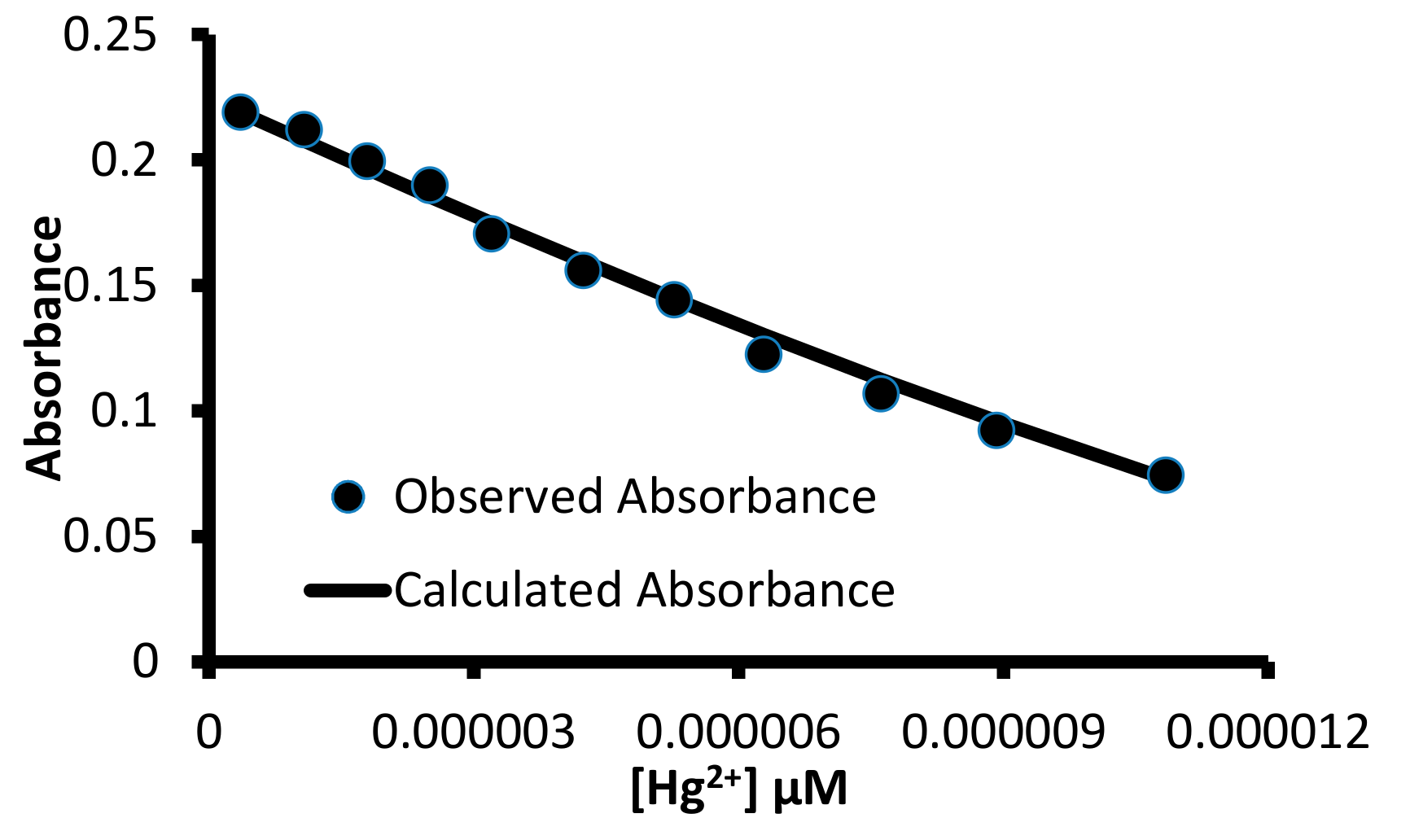
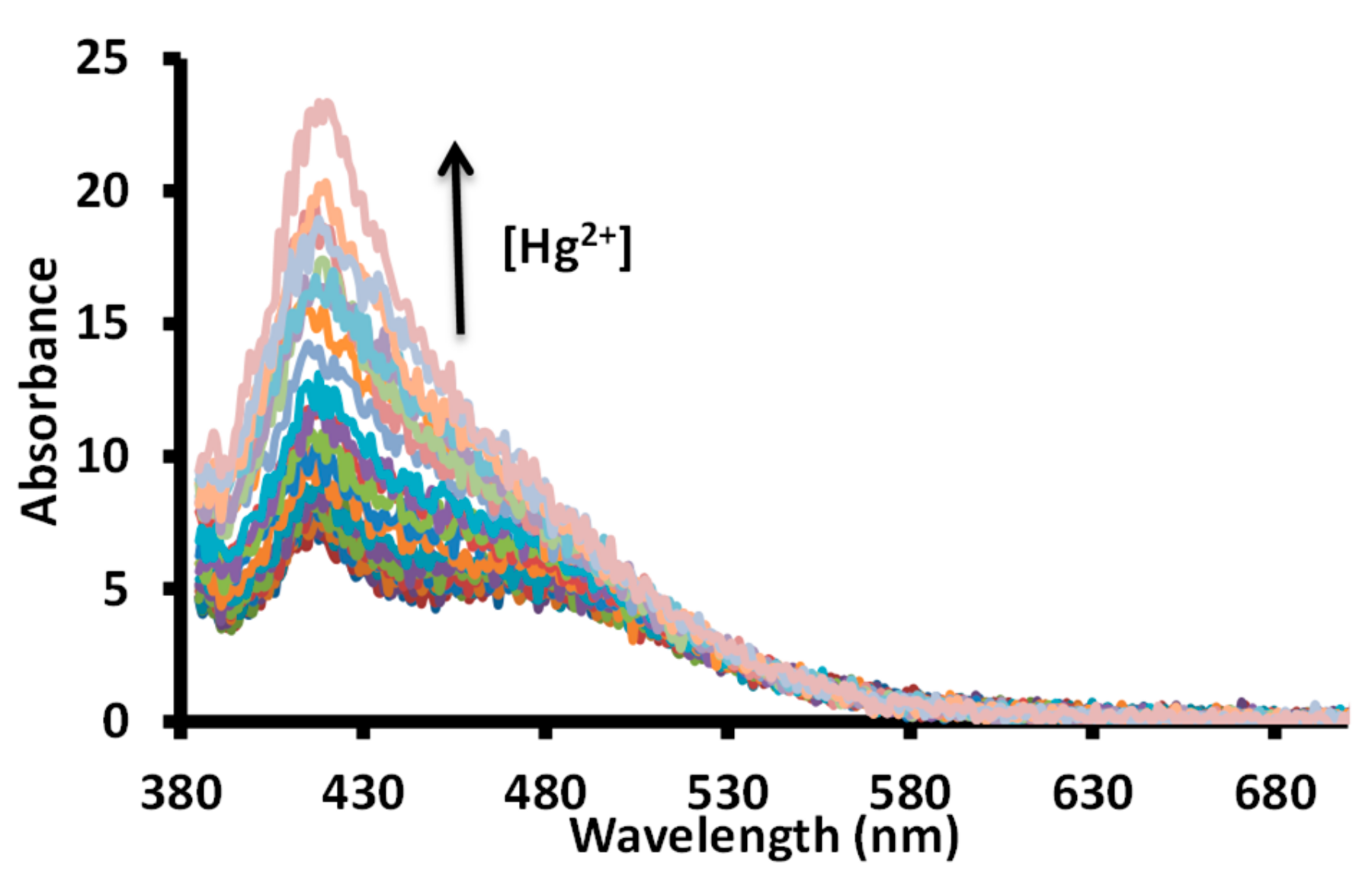
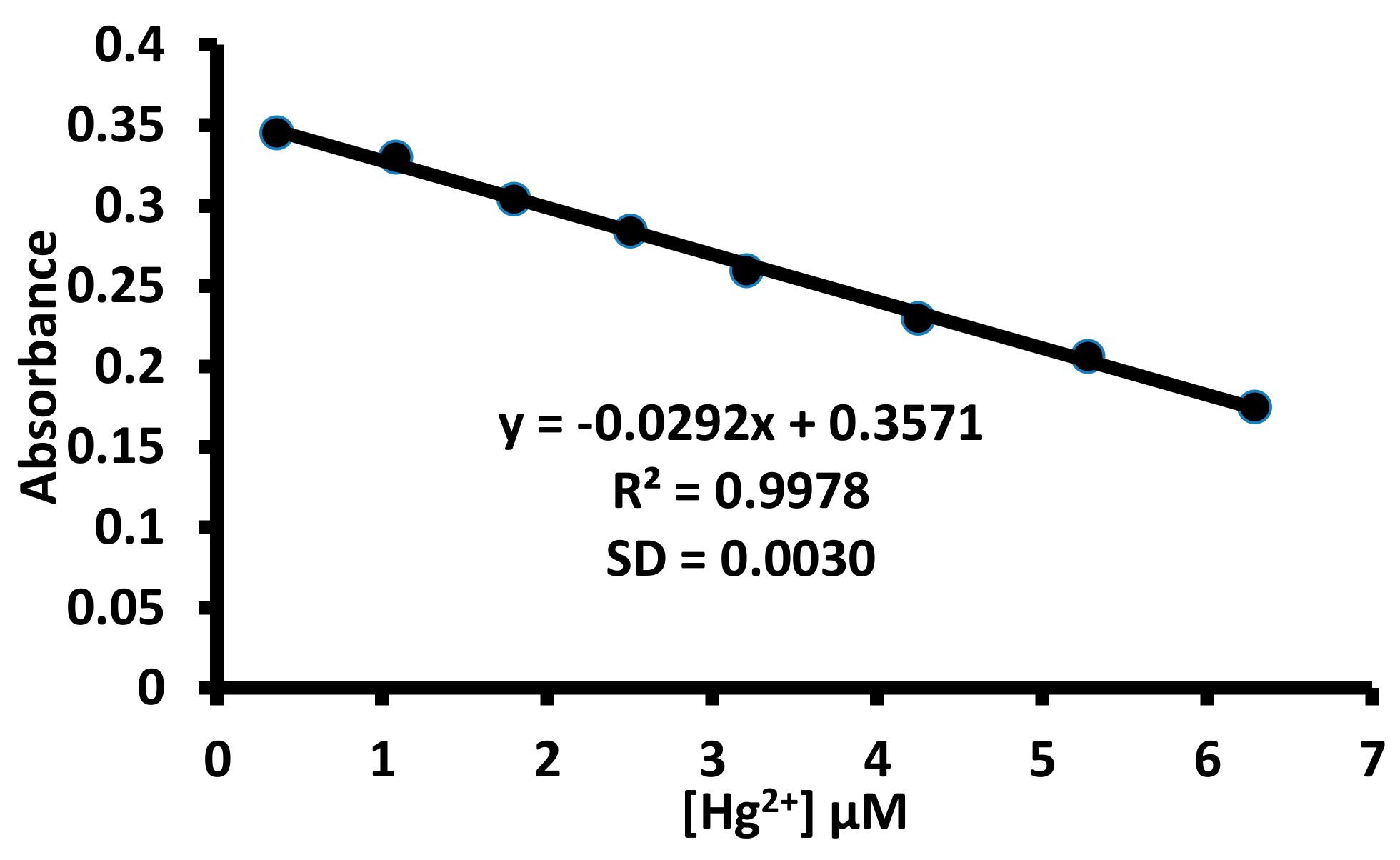
3.2. UV-Visible Studies
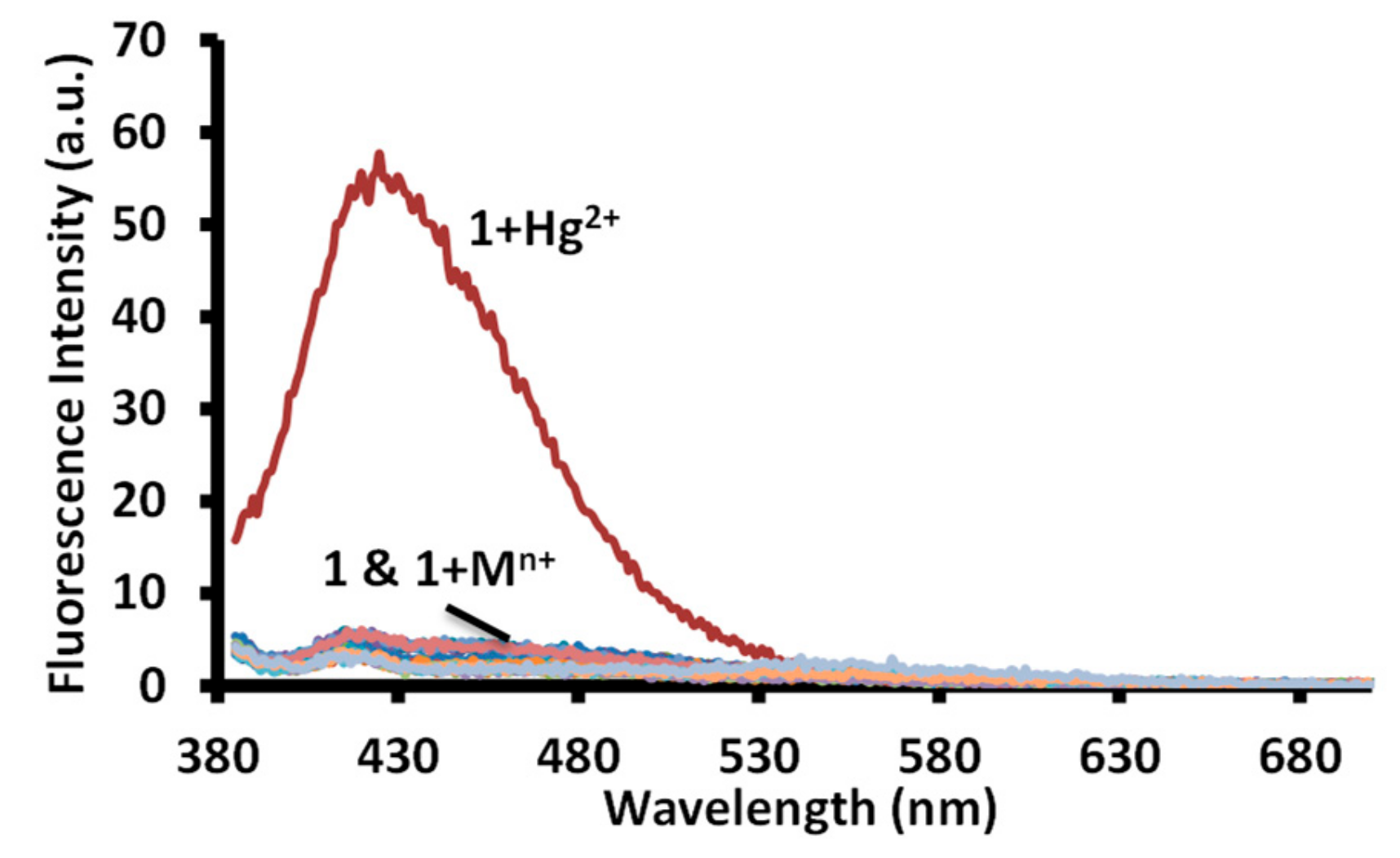
3.3. Fluorescence Studies
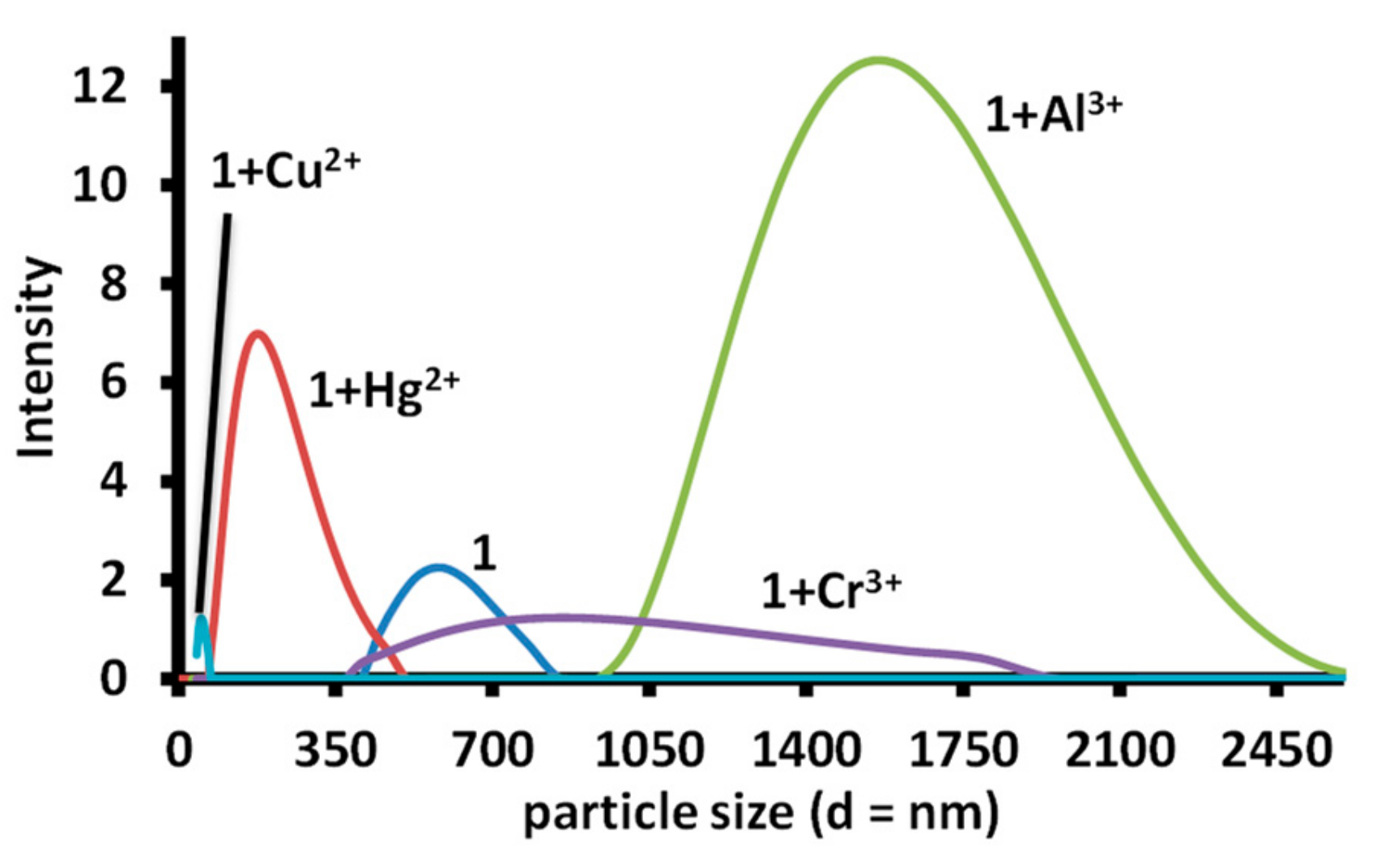
3.4. DLS Studies
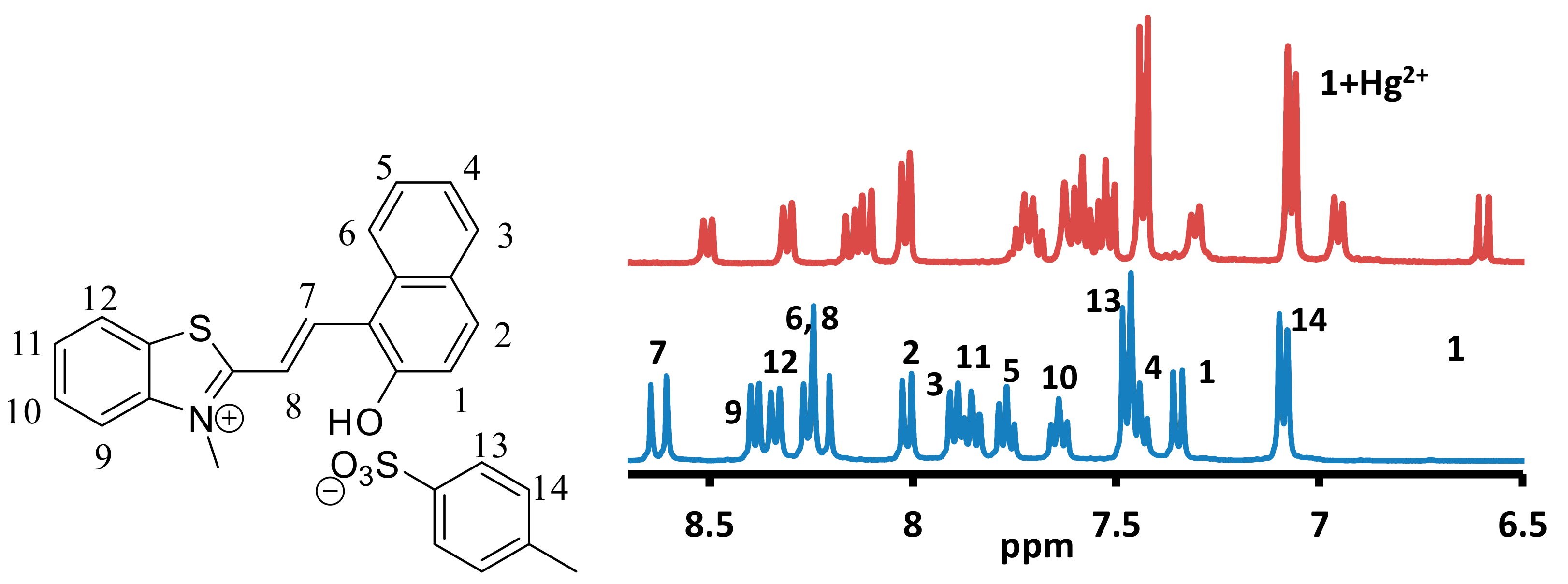
3.5. NMR Studies
3.6. Practical Utility
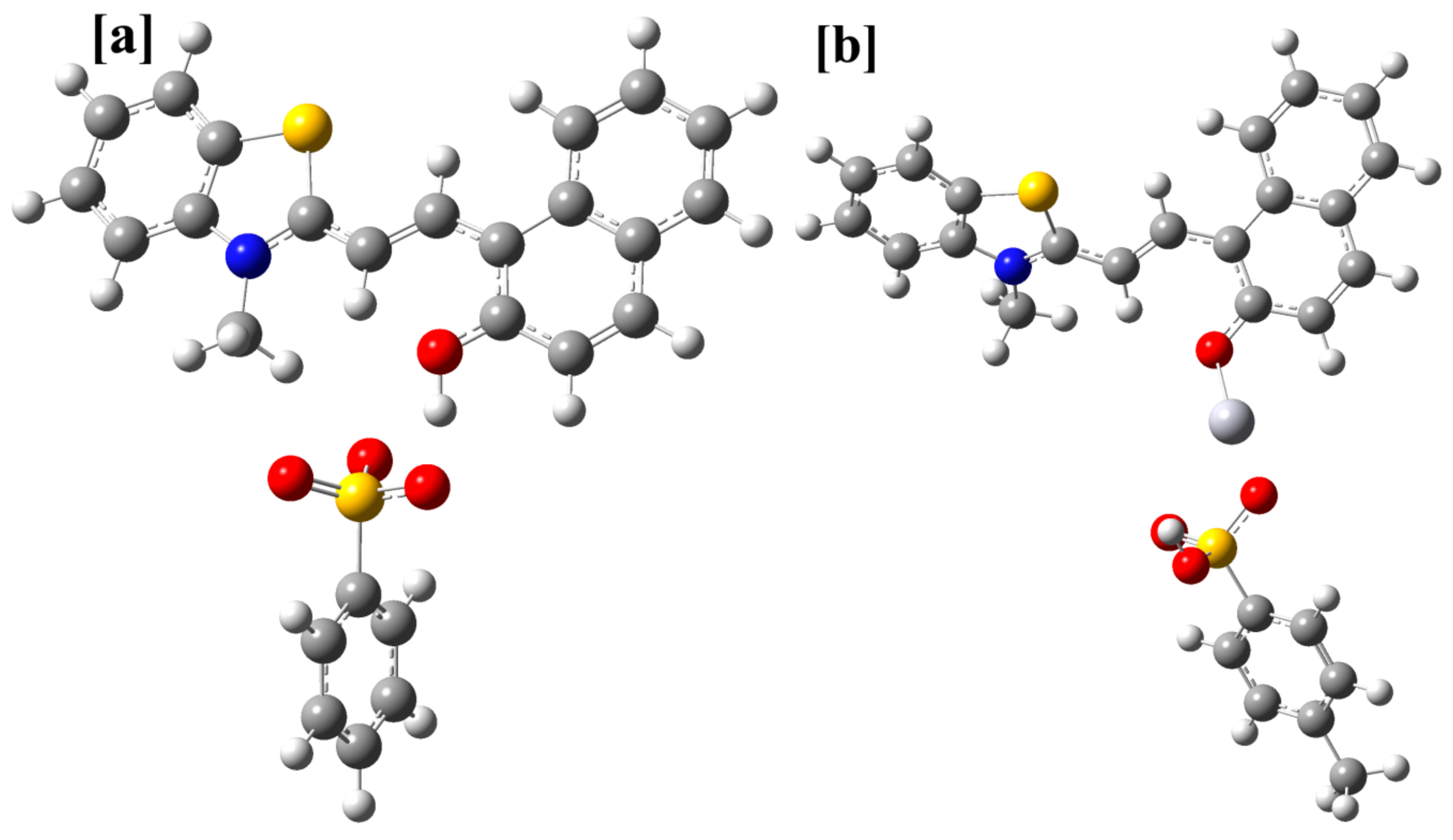
3.7. Computational Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hylander, L.D.; Goodsite, M.E. Environmental costs of mercury pollution. Sci. Total Environ. 2006, 368, 352–370. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Feng, X.; Qiu, G.; Shang, L.; Li, Z. Mercury pollution in Asia: A review of the contaminated sites. J. Hazard. Mater. 2009, 168, 591–601. [Google Scholar] [CrossRef] [PubMed]
- Harris, H.H.; Pickering, I.J.; George, G.N. The chemical form of mercury in fish. Science 2003, 301, 1203. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Li, J.; Qi, J.; Chen, J. Highly selective and effective mercury (II) fluorescent sensors. New J. Chem. 2015, 39, 843–848. [Google Scholar] [CrossRef]
- Kim, H.N.; Ren, W.X.; Kim, J.S.; Yoon, J. Fluorescent and colorimetric sensors for detection of lead, cadmium, and mercury ions. Chem. Soc. Rev. 2012, 41, 3210–3244. [Google Scholar] [CrossRef]
- Boening, D.W. Ecological effects, transport, and fate of mercury: A general review. Chemosphere 2000, 40, 1335–1351. [Google Scholar] [CrossRef]
- Joensuu, O.I. Fossil fuels as a source of mercury pollution. Science 1971, 172, 1027–1028. [Google Scholar] [CrossRef]
- Gonzalez, H. Mercury pollution caused by a chlor-alkali plant. Water Air Soil Pollut. 1991, 56, 83–93. [Google Scholar] [CrossRef]
- Porcella, D.; Ramel, C.; Jernelov, A. Global mercury pollution and the role of gold mining: An overview. Water Air Soil Pollut. 1997, 97, 205–207. [Google Scholar] [CrossRef]
- Boischio, A.A.P.; Henshel, D. Fish Consumption, Fish Lore, and Mercury Pollution—Risk Communication for the Madeira River People. Environ. Res. 2000, 84, 108–126. [Google Scholar] [CrossRef]
- Okpala, C.O.R.; Sardo, G.; Vitale, S.; Bono, G.; Arukwe, A. Hazardous properties and toxicological update of mercury: From fish food to human health safety perspective. Crit. Rev. Food Sci. Nutr. 2018, 58, 1986–2001. [Google Scholar] [CrossRef] [PubMed]
- Obrist, D.; Agnan, Y.; Jiskra, M.; Olson, C.L.; Colegrove, D.P.; Hueber, J.; Moore, C.W.; Sonke, J.E.; Helmig, D. Tundra uptake of atmospheric elemental mercury drives Arctic mercury pollution. Nature 2017, 547, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Krishna, P.; Basha, S.S.; Sathyavani, K.G.; Prabhavathi, K. Heavy metal bioaccumulation in the Channa marulius from Lake Kolleru and human health risk assessment. Int. J. Zool. Stud. 2018, 3, 76–79. [Google Scholar]
- Li, J.; Li, X.; Wang, L.; Duan, Q. Advances in uptake, transportation and bioaccumulation of heavy metal ions in bivalves. Shuichan Kexue 2007, 26, 51–55. [Google Scholar]
- Jackson, A.C. Chronic Neurological Disease Due to Methylmercury Poisoning. Can. J. Neurol. Sci. 2018, 45, 620–623. [Google Scholar] [CrossRef]
- Rice, K.M.; Walker, E.M., Jr.; Wu, M.; Gillette, C.; Blough, E.R. Environmental mercury and its toxic effects. J. Prev. Med. Public Health 2014, 47, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Houston, M.C. Role of mercury toxicity in hypertension, cardiovascular disease, and stroke. J. Clin. Hypertens. 2011, 13, 621–627. [Google Scholar] [CrossRef]
- Dos Santos, A.A.; Chang, L.W.; Guo, G.L.; Aschner, M. Fetal Minamata Disease: A Human Episode of Congenital Methylmercury Poisoning. In Handbook of Developmental Neurotoxicology; Elsevier: San Diego, CA, USA, 2018; pp. 399–406. [Google Scholar]
- Ancora, M.P.; Zhang, L.; Wang, S.; Schreifels, J.J.; Hao, J. Meeting Minamata: Cost-effective compliance options for atmospheric mercury control in Chinese coal-fired power plants. Energy Policy 2016, 88, 485–494. [Google Scholar] [CrossRef]
- Weiss, B. Why methyl mercury remains a conundrum 50 years after Minamata. Toxicol. Sci. 2007, 97, 223–225. [Google Scholar] [CrossRef]
- Nyanza, E.C.; Bernier, F.P.; Manyama, M.; Hatfield, J.; Martin, J.W.; Dewey, D. Maternal exposure to arsenic and mercury in small-scale gold mining areas of Northern Tanzania. Environ. Res. 2019, 173, 432–442. [Google Scholar] [CrossRef]
- Lin, J. Determination of heavy metals in sewages by microwave digestion coupled FAAS. Fujian Fenxi Ceshi 2007, 16, 84–86. [Google Scholar]
- Racki, G.; Rakociński, M.; Marynowski, L. Anomalous Upper Devonian mercury enrichments: Comparison of Inductively Coupled Plasma–Mass Spectrometry (ICP-MS) and Atomic Absorption Spectrometry (AAS) analytical data. Geol. Q. 2018, 62, 487–495. [Google Scholar] [CrossRef]
- Lech, T.; Turek, W. Application of TDA AAS to Direct Mercury Determination in Postmortem Material in Forensic Toxicology Examinations. J. Anal. Toxicol. 2019, 43, 385–391. [Google Scholar] [CrossRef]
- Murkovic, I.; Wolfbeis, O.S. Fluorescence-based sensor membrane for mercury (II) detection. Sens. Actuator B Chem. 1997, 39, 246–251. [Google Scholar] [CrossRef]
- Zhang, Y.; Gao, L.; Wen, L.; Heng, L.; Song, Y. Highly sensitive, selective and reusable mercury(II) ion sensor based on a ssDNA-functionalized photonic crystal film. Phys. Chem. Chem. Phys. 2013, 15, 11943–11949. [Google Scholar] [CrossRef] [PubMed]
- Zheng, G.C.; Wang, J.; Kong, L.T.; Cheng, H.F.; Liu, J.H. Cellular-Like Gold Nanofeet: Synthesis, Functionalization, and Surface Enhanced Fluorescence Detection for Mercury Contaminations. Plasmonics 2012, 7, 487–494. [Google Scholar] [CrossRef]
- Sahoo, P.R.; Prakash, K.; Kumar, S. Light controlled receptors for heavy metal ions. Coord. Chem. Rev. 2018, 357, 18–49. [Google Scholar] [CrossRef]
- Crano, J.C.; Guglielmetti, R.J. Organic Photochromic and Thermochromic Compounds: Volume 2: Physicochemical Studies, Biological Applications, and Thermochromism; Springer: New York, NY, USA, 1999; Volume 2. [Google Scholar]
- Sahoo, P.R.; Prakash, K.; Kumar, A.; Kumar, S. Efficient Reversible Optical Sensing of Water Achieved through the Conversion of H-Aggregates of a Merocyanine Salt to J-Aggregates. Chem. Select 2017, 2, 5924–5932. [Google Scholar] [CrossRef]
- Rösch, U.; Yao, S.; Wortmann, R.; Würthner, F. Fluorescent H-Aggregates of Merocyanine Dyes. Angew. Chem. Int. Ed. 2006, 45, 7026–7030. [Google Scholar] [CrossRef]
- Möbius, D. Scheibe aggregates. Adv. Mater. 1995, 7, 437–444. [Google Scholar] [CrossRef]
- Sahoo, P.R.; Kumar, S. Synthesis of an optically switchable salicylaldimine substituted naphthopyran for selective and reversible Cu2+ recognition in aqueous solution. RSC Adv. 2016, 6, 20145–20154. [Google Scholar] [CrossRef]
- Kumar, S.; Velasco, K.; McCurdy, A. X-ray, kinetics and DFT studies of photochromic substituted benzothiazolinic spiropyrans. J. Mol. Struct. 2010, 968, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Watkins Davita, L.; Fujiwara, T. A tailored spirooxazine dimer as a photoswitchable binding tool. Chem. Commun. 2009, 4369–4371. [Google Scholar] [CrossRef] [PubMed]
- Firdaus, M.L.; Aprian, A.; Meileza, N.; Hitsmi, M.; Elvia, R.; Rahmidar, L.; Khaydarov, R. Smartphone Coupled with a Paper-Based Colorimetric Device for Sensitive and Portable Mercury Ion Sensing. Chemosensors 2019, 7, 25. [Google Scholar] [CrossRef]
- Firdaus, M.L.; Alwi, W.; Trinoveldi, F.; Rahayu, I.; Rahmidar, L.; Warsito, K. Determination of Chromium and Iron Using Digital Image-based Colorimetry. Procedia Environ. Sci. 2014, 20, 298–304. [Google Scholar] [CrossRef]
- Masawat, P.; Harfield, A.; Srihirun, N.; Namwong, A. Green Determination of Total Iron in Water by Digital Image Colorimetry. Anal. Lett. 2017, 50, 173–185. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXT–Integrated space-group and crystal-structure determination. Acta Cryst. A 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Macrae, C.F.; Bruno, I.J.; Chisholm, J.A.; Edgington, P.R.; McCabe, P.; Pidcock, E.; Rodriguez-Monge, L.; Taylor, R.; Streek, J.V.; Wood, P.A. Mercury CSD 2.0–new features for the visualization and investigation of crystal structures. J. Appl. Cryst. 2008, 41, 466–470. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09, Revision A.02; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Wolff, S.; Grimwood, D.; McKinnon, J.; Jayatilaka, D.; Spackman, M. Crystalexplorer 17.5; University of Western Australia: Perth, Australia, 2018. [Google Scholar]
- Thakuria, R.; Nath, N.K.; Roy, S.; Nangia, A. Polymorphism and isostructurality in sulfonylhydrazones. CrystEngComm 2014, 16, 4681–4690. [Google Scholar] [CrossRef]
- Gans, P.; Sabatini, A.; Vacca, A. Investigation of equilibria in solution. Determination of equilibrium constants with the HYPERQUAD suite of programs. Talanta 1996, 43, 1739–1753. [Google Scholar] [CrossRef]
- Klajn, R. Spiropyran-based dynamic materials. Chem. Soc. Rev. 2014, 43, 148–184. [Google Scholar] [CrossRef] [PubMed]
- Adamo, C.; Barone, V. Toward reliable density functional methods without adjustable parameters: The PBE0 model. J. Chem. Phys. 1999, 110, 6158–6170. [Google Scholar] [CrossRef]
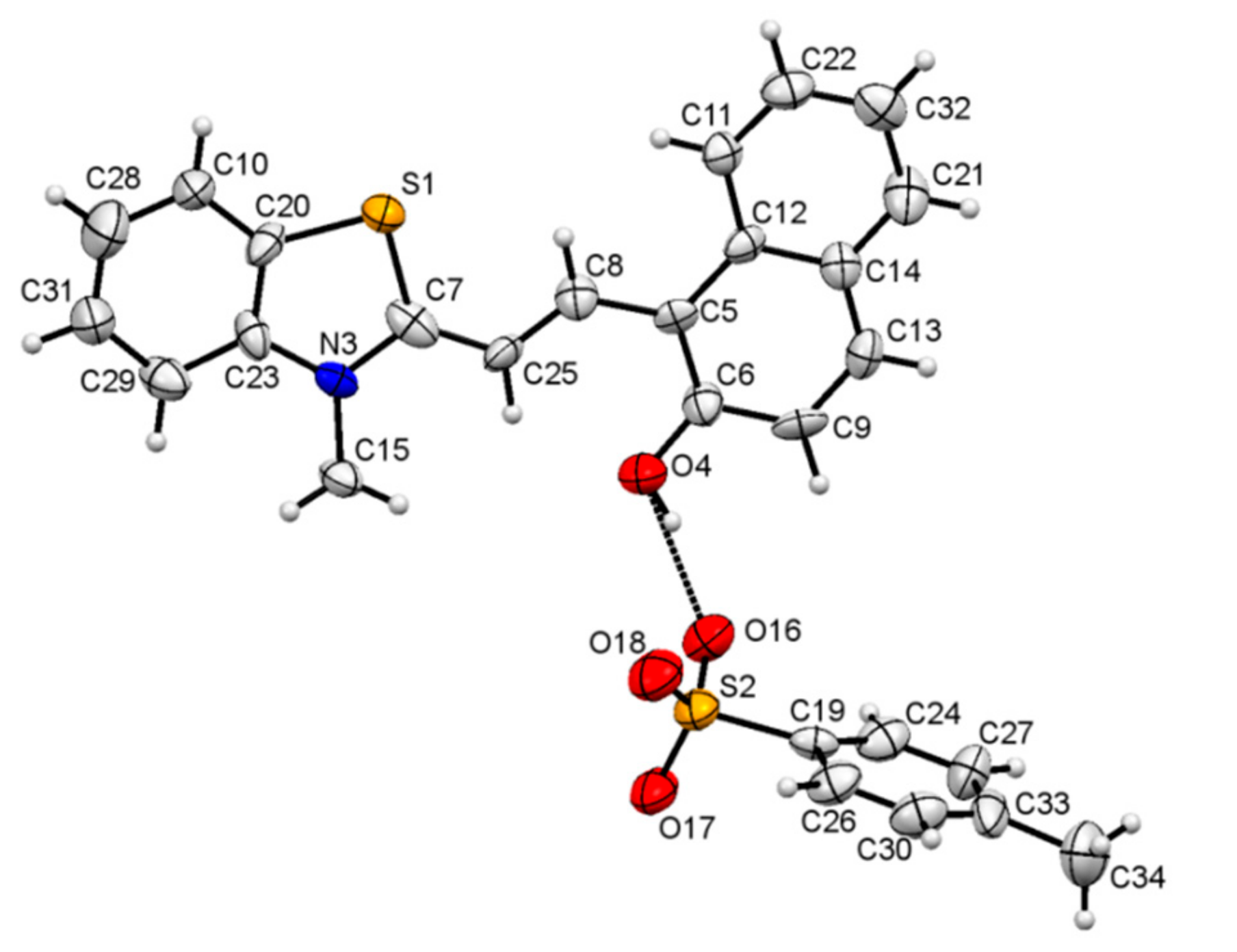
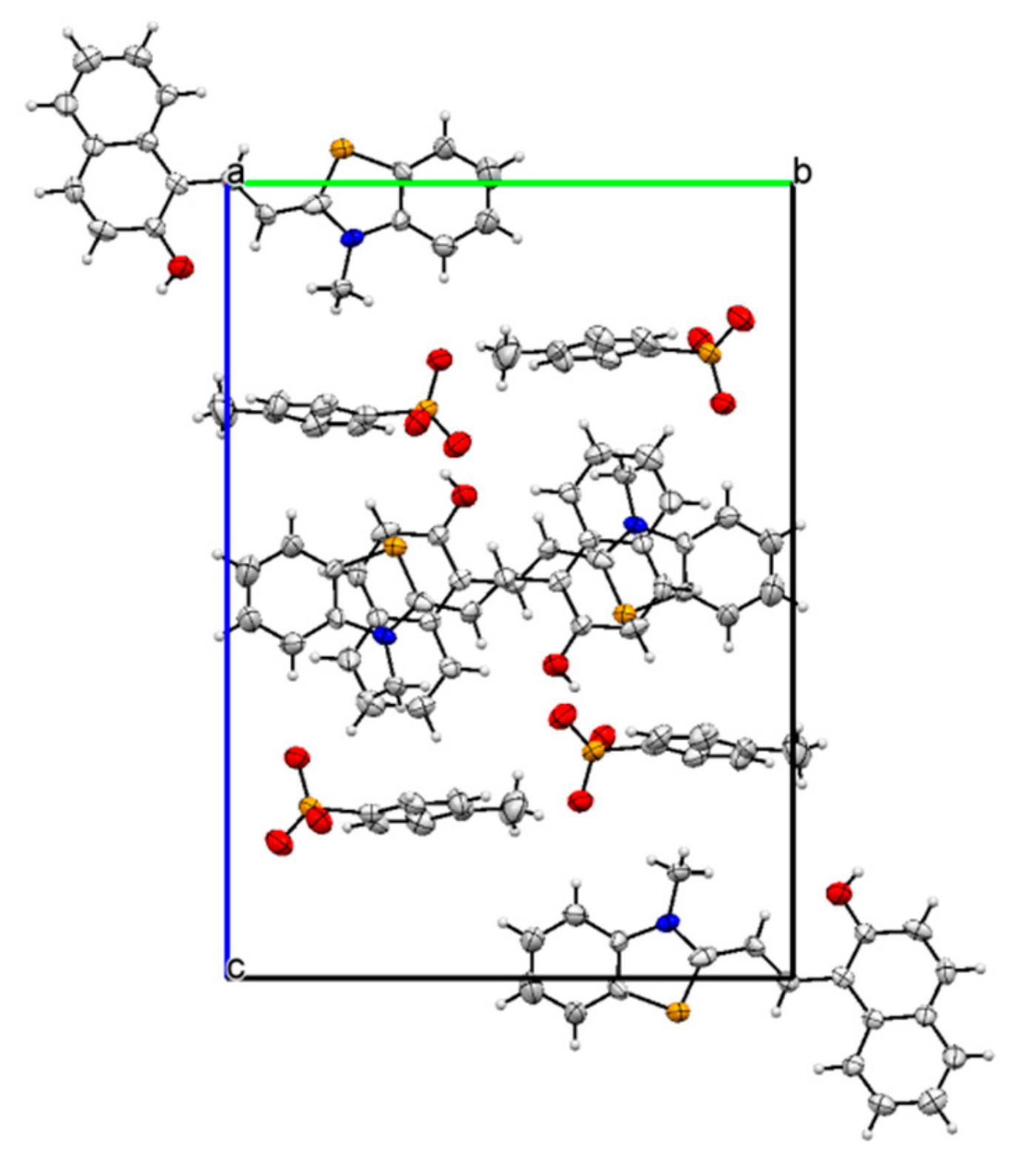
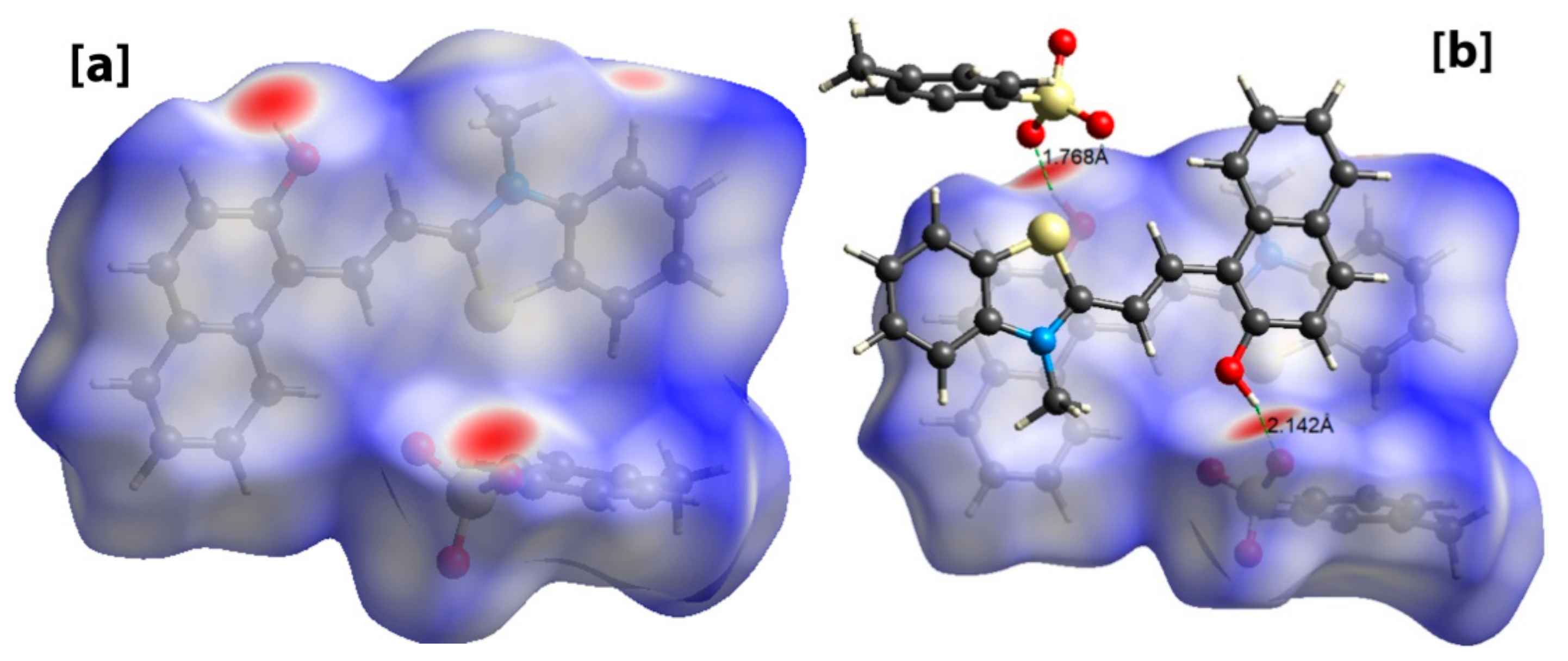
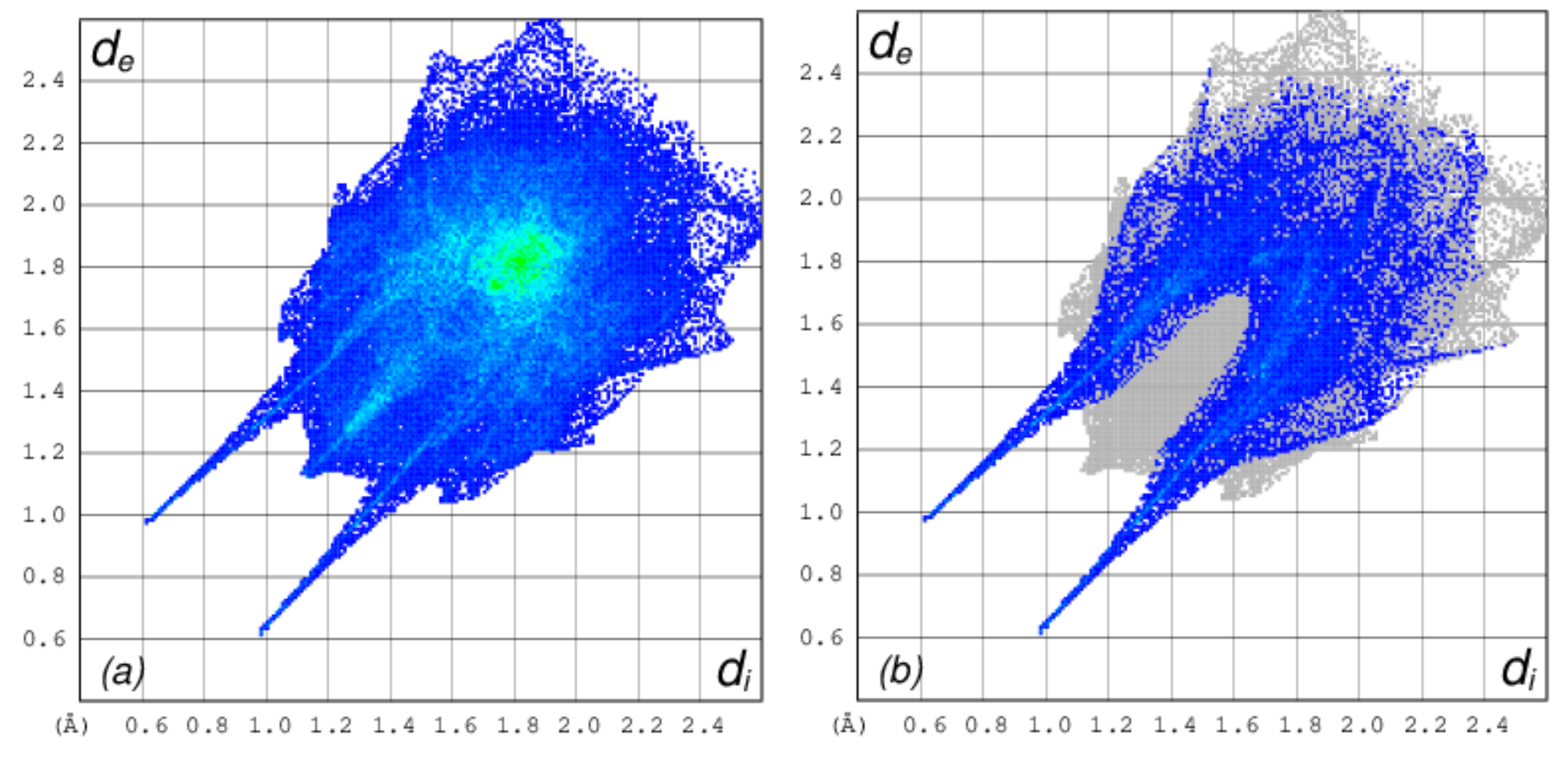
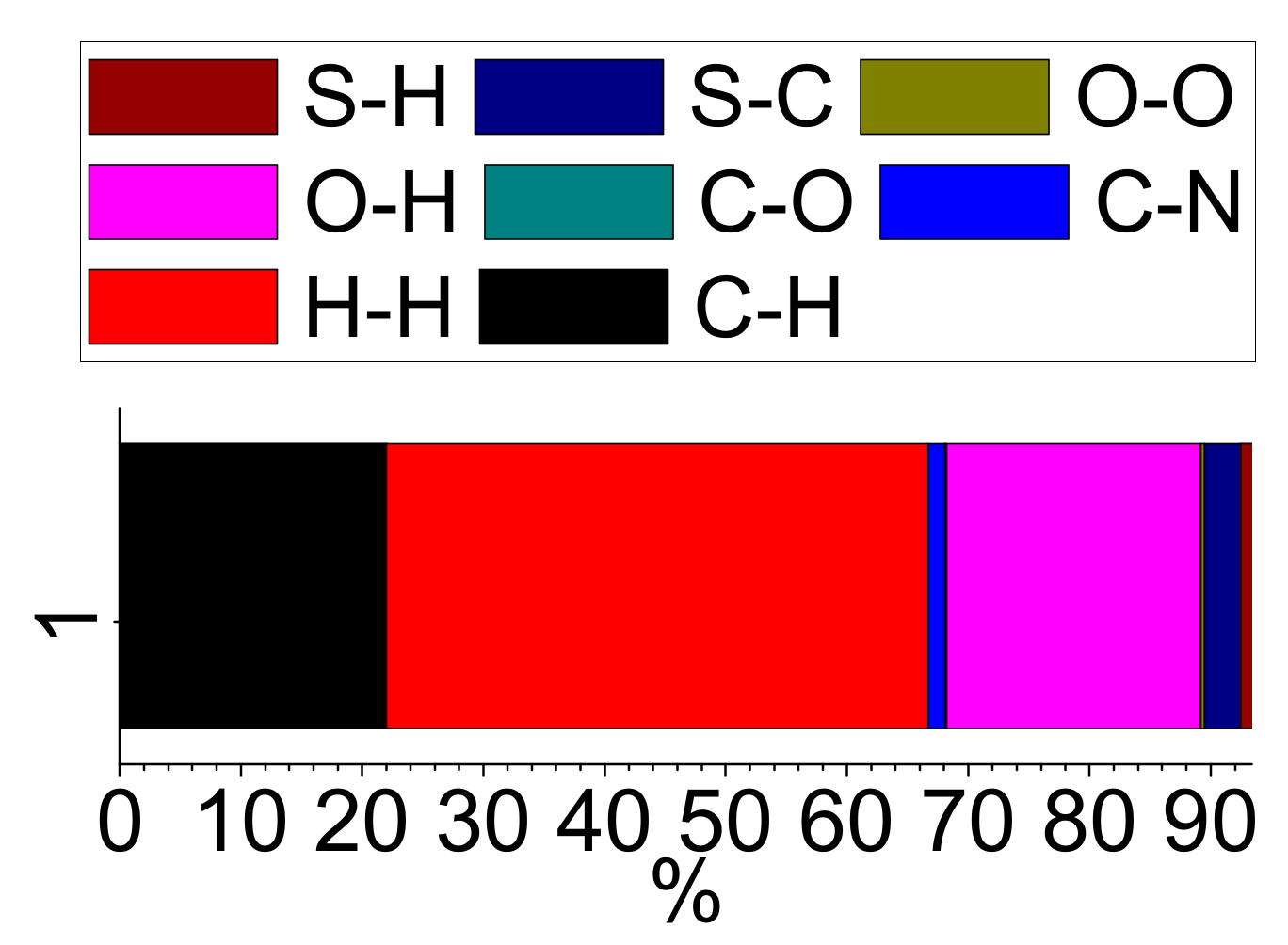





| D—H···A | D—H | H···A | D···A | D—H···A |
|---|---|---|---|---|
| O4—H4···S2i | 0.92 (9) | 2.66 (10) | 3.520 (6) | 155.0 (8) |
| O4—H4···O16i | 0.92 (9) | 1.65 (9) | 2.571 (7) | 176.1 (9) |
| C15—H15B···O17ii | 0.96 | 2.54 | 3.433 (9) | 154.4 |
| C25—H25···O4 | 0.93 | 2.11 | 2.747 (9) | 124.9 |
| C29—H29···O17ii | 0.93 | 2.40 | 3.332 (9) | 178.0 |
| Frag. | N | Symop | R | E_ele | E_pol | E_dis | E_rep | E_tot |
|---|---|---|---|---|---|---|---|---|
| a | 1 | - | 5.42 | −5.8 | −4.3 | −30 | 34 | −14.4 |
| b | 1 | x, y, z | 15.3 | −0.4 | −0.2 | −9.2 | 7.2 | −4.1 |
| c | 1 | - | 13.17 | 0.4 | −0.1 | −0.3 | 0 | 0.2 |
| d | 1 | x, −y + 0.5, z + 0.5 | 12.3 | −0.1 | −0.1 | −0.9 | 0 | −1 |
| e | 1 | - | 12.5 | −0.1 | −0.2 | −0.6 | 0 | −0.8 |
| f | 1 | - | 9.07 | −1.3 | −0.9 | −13 | 7.8 | −8.5 |
| g | 1 | - | 7.8 | −49.8 | −10.1 | −13.2 | 92 | −14.8 |
| h | 1 | −x, −y, −z | 4.07 | −22.9 | −9.1 | −113.6 | 80.7 | −80 |
| Metal | Logβ11 | Change in λmax (nm) at |
|---|---|---|
| Hg2+ | 5.2480 ± 0.0229 | 380, 450, 570 |
| Cu2+ | 5.0172 ± 0.0193 | 455, 564 |
| Al3+ | 4.8992 ± 0.0504 | 563 |
| Cr3+ | 4.8949 ± 0.0075 | 564 |
| Fe3+ | 4.59582 ± 0.0307 | 563 |
| Stereo-Isomer | E1a | E2b | E3c | Erel = ETTC − ESId | E1+Hg | Ercf | ΔE = E1−Hg2+ − (E3 + EHg2+) |
|---|---|---|---|---|---|---|---|
| TTC | −814,597.26 | −814,854.47 | −1,376,038.65 | 0 | −1,402,460.35 | 0 | −195.90 |
| CTT | −814,599.01 | −814,849.28 | −1,376,032.66 | 5.98 | −1,402,457.10 | 3.24 | −198.64 |
| CTC | −814,602.62 | −814,851.30 | −1,376,032.18 | 6.47 | −1,402,457.77 | 2.57 | −199.79 |
| TTT | −814,595.80 | −814,852.95 | −1,376,031.82 | 6.82 | −1,402,460.04 | 0.31 | −202.41 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, A.; Kumar, A.; Sahoo, P.R.; Kumar, S. Colorimetric and Fluorescence-Based Detection of Mercuric Ion Using a Benzothiazolinic Spiropyran. Chemosensors 2019, 7, 35. https://doi.org/10.3390/chemosensors7030035
Kumar A, Kumar A, Sahoo PR, Kumar S. Colorimetric and Fluorescence-Based Detection of Mercuric Ion Using a Benzothiazolinic Spiropyran. Chemosensors. 2019; 7(3):35. https://doi.org/10.3390/chemosensors7030035
Chicago/Turabian StyleKumar, Ajeet, Arvind Kumar, Priya Ranjan Sahoo, and Satish Kumar. 2019. "Colorimetric and Fluorescence-Based Detection of Mercuric Ion Using a Benzothiazolinic Spiropyran" Chemosensors 7, no. 3: 35. https://doi.org/10.3390/chemosensors7030035
APA StyleKumar, A., Kumar, A., Sahoo, P. R., & Kumar, S. (2019). Colorimetric and Fluorescence-Based Detection of Mercuric Ion Using a Benzothiazolinic Spiropyran. Chemosensors, 7(3), 35. https://doi.org/10.3390/chemosensors7030035