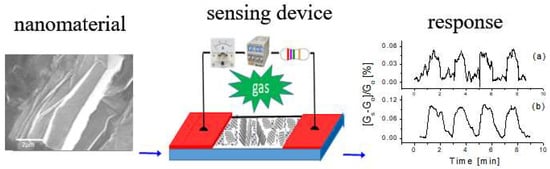
Super Stable Pollution Gas Sensor Based on Functionalized 2D Boron Nitride Nanosheet Materials for High Humidity Environments
Abstract
:1. Introduction
2. Experimental Details
3. Results and Discussions
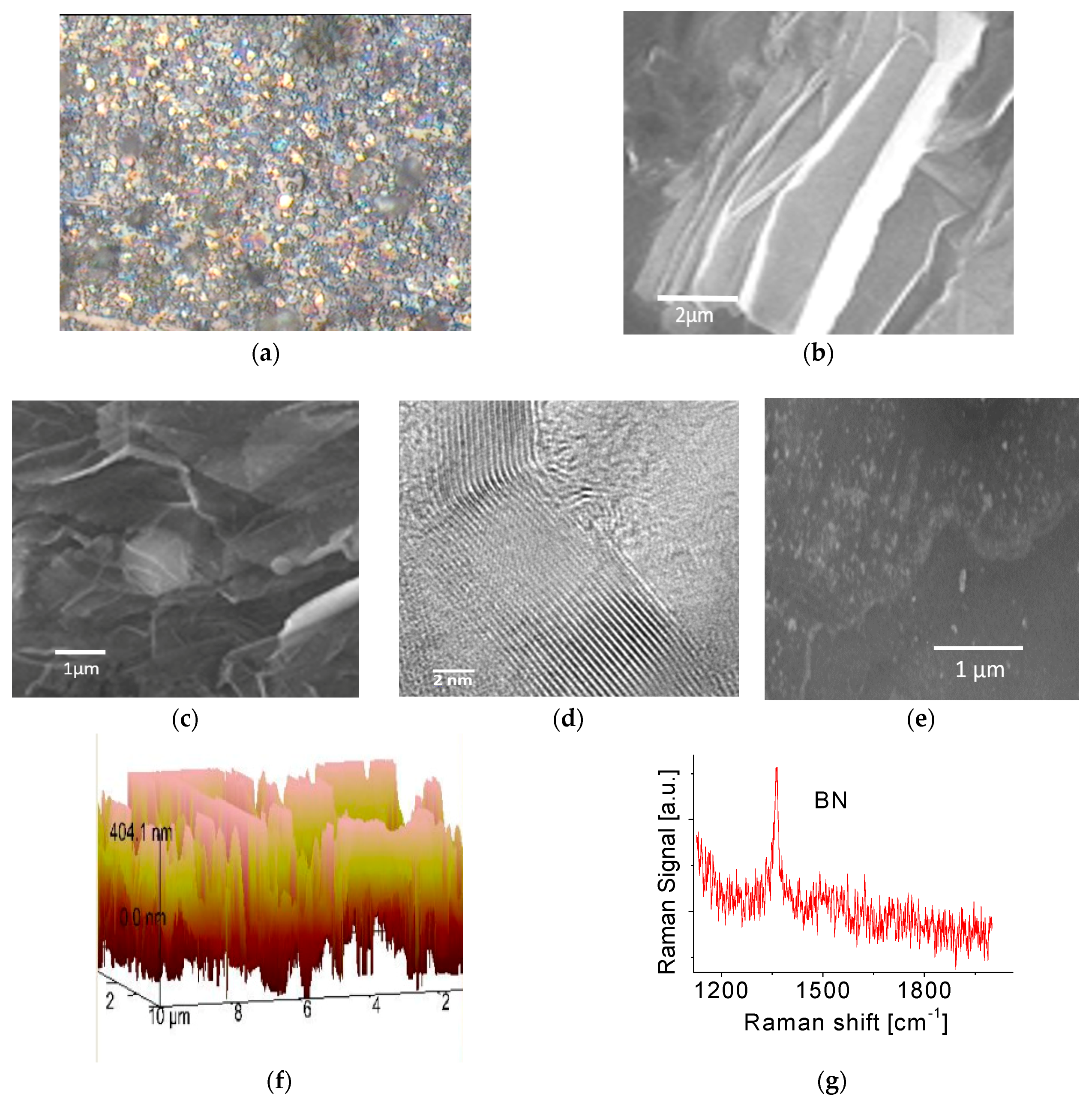
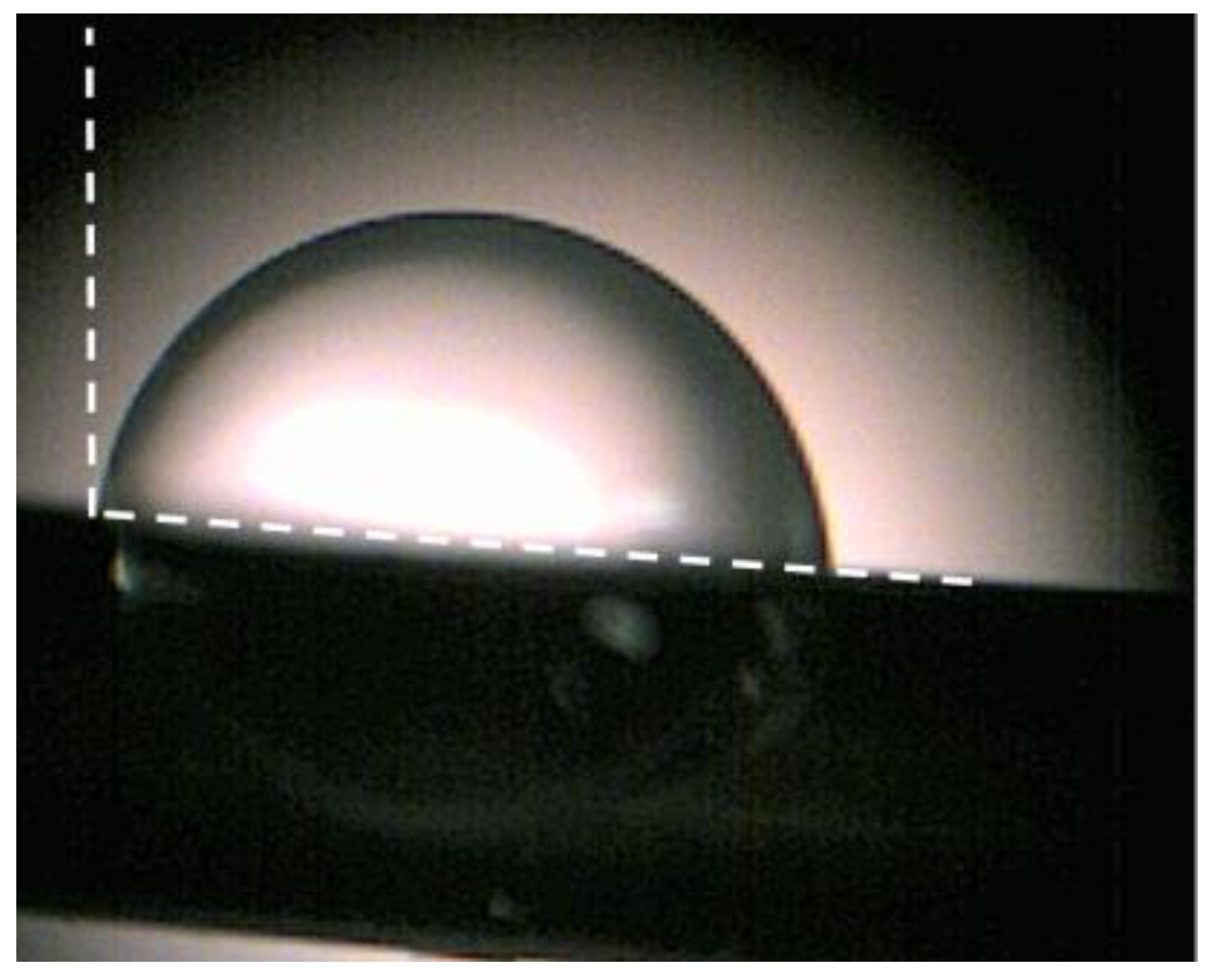
3.1. Basic Characterization of the 2D BNNS Materials
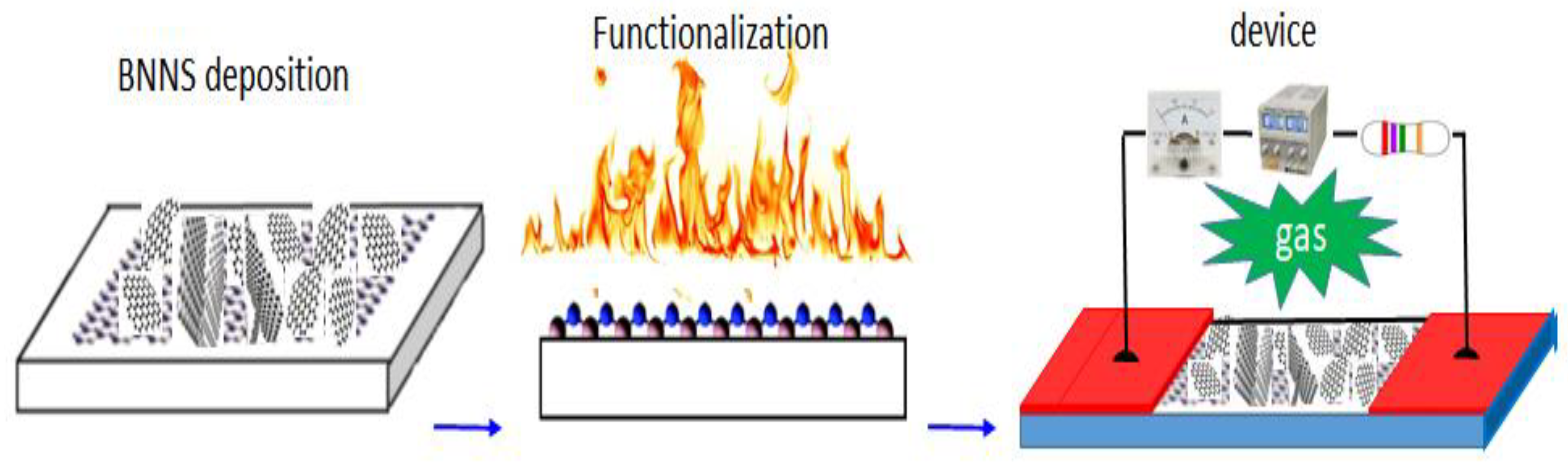
3.2. Fabrication of Prototype and Characterization of Its Electrical Properties
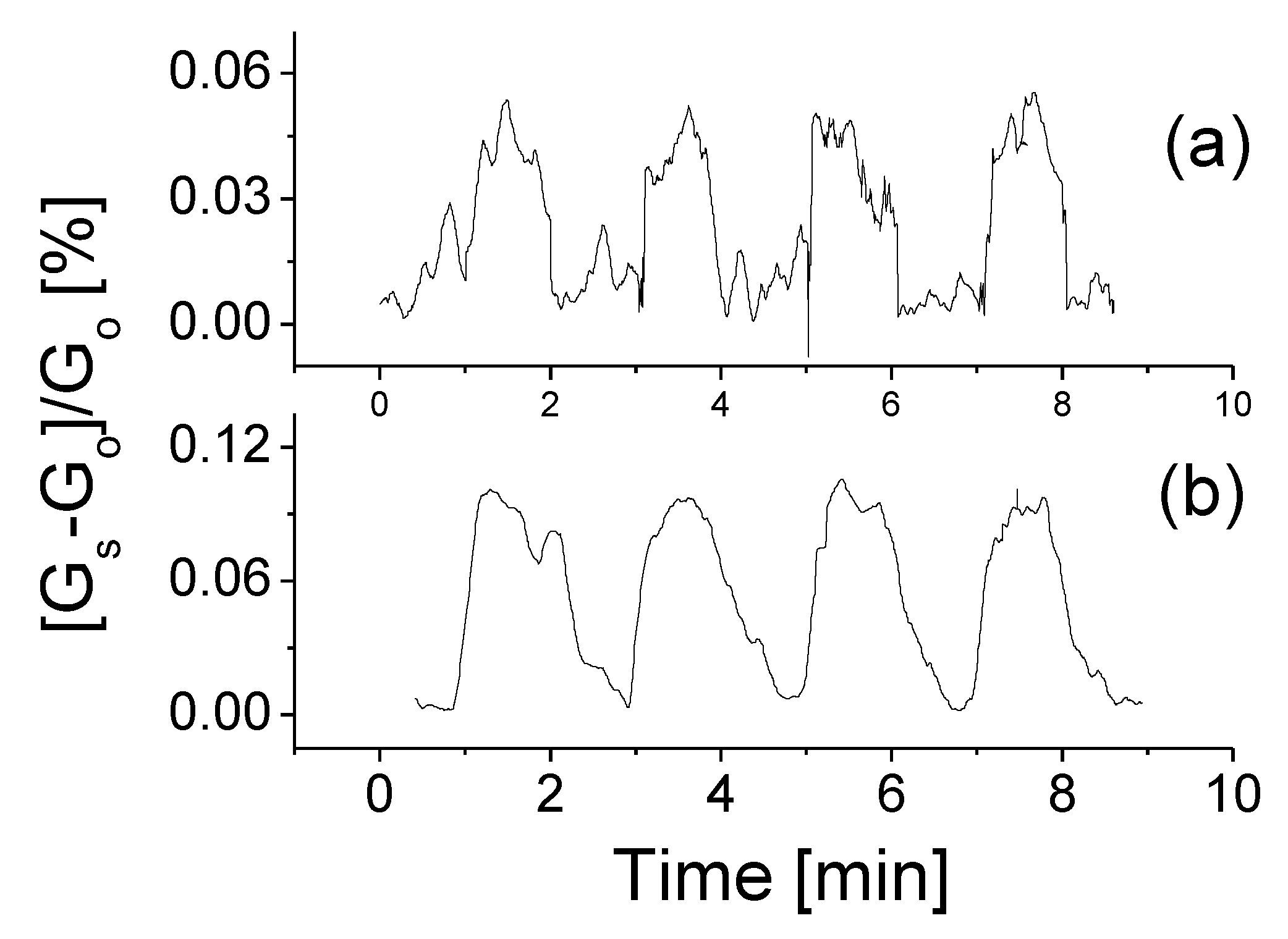
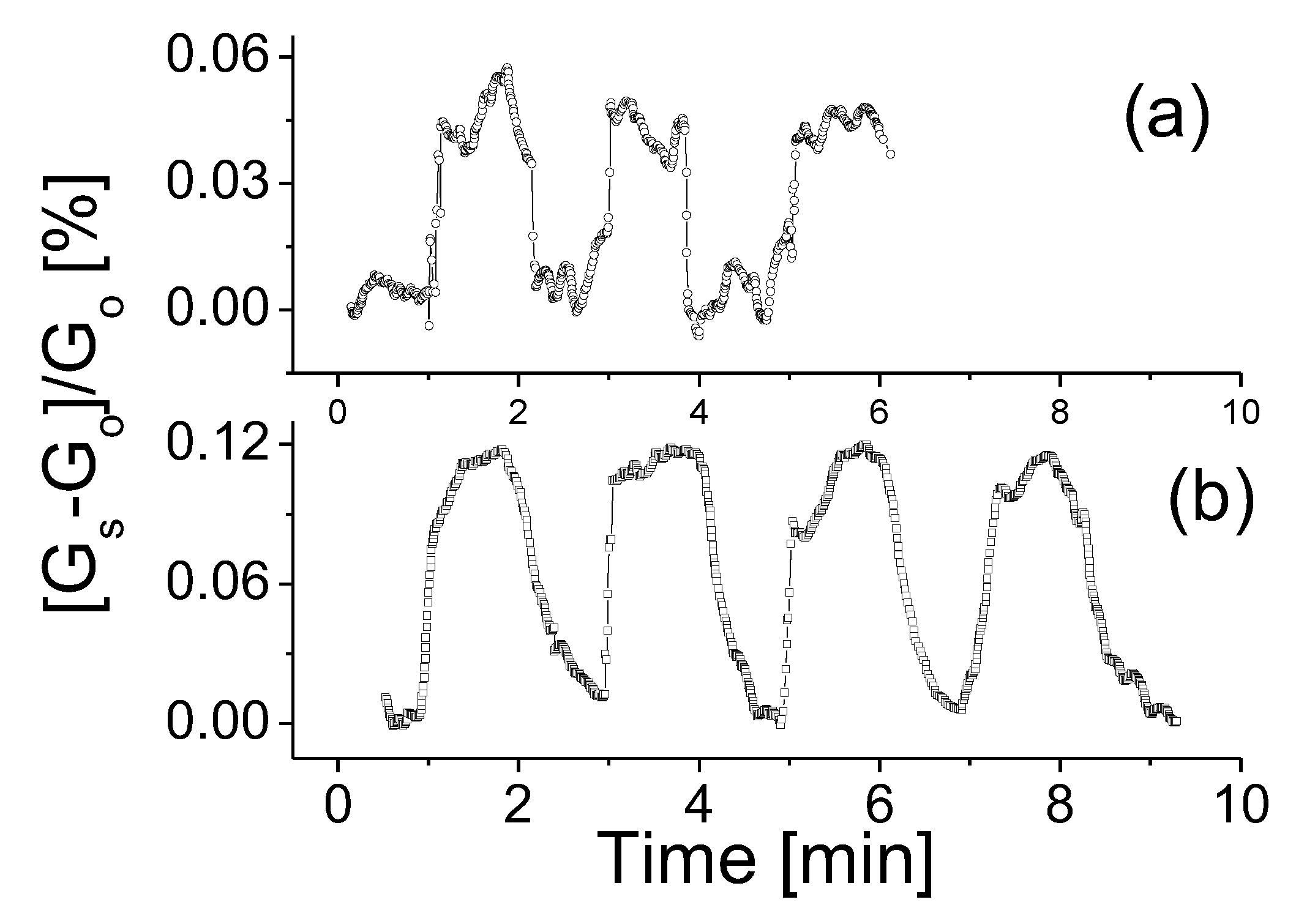
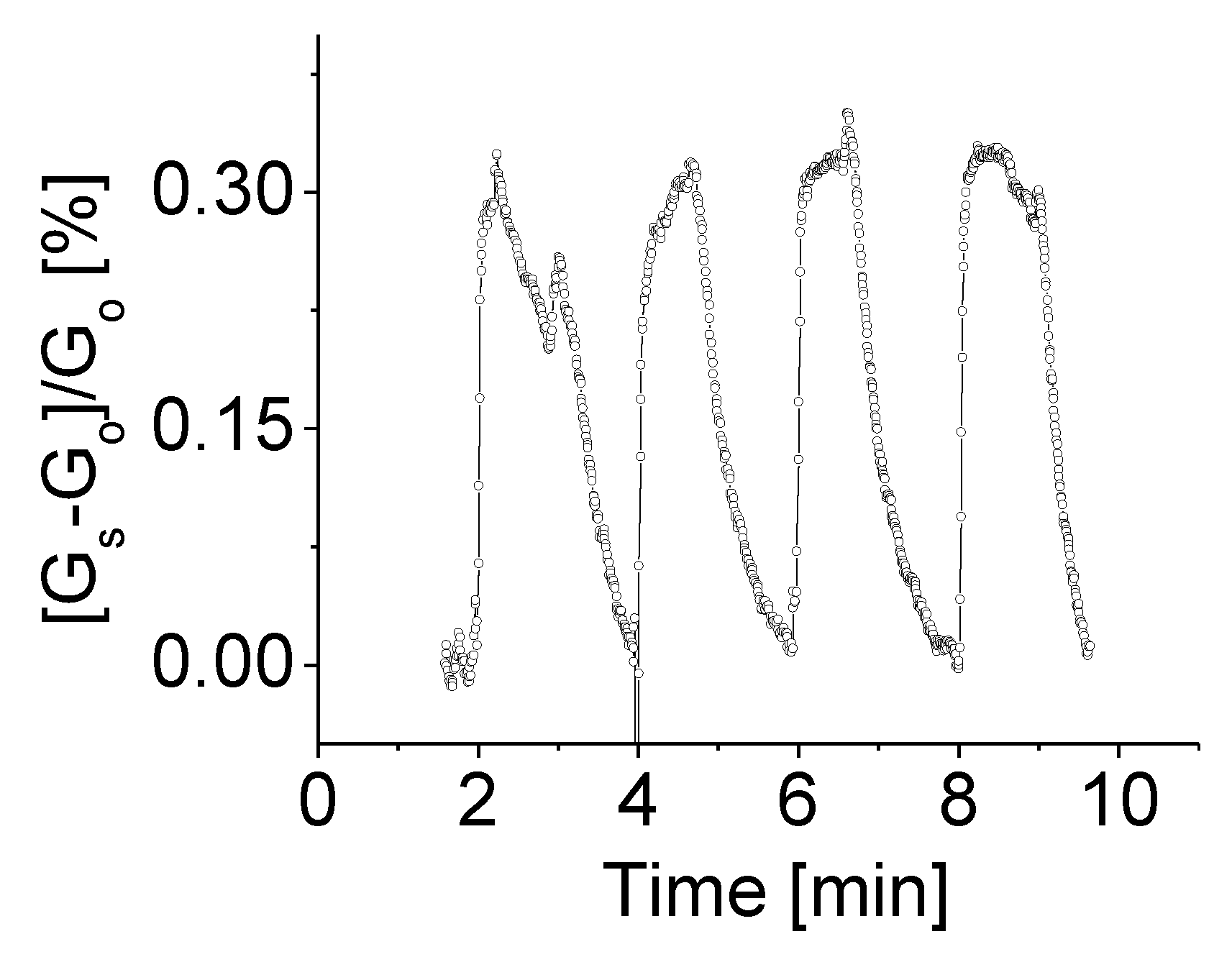
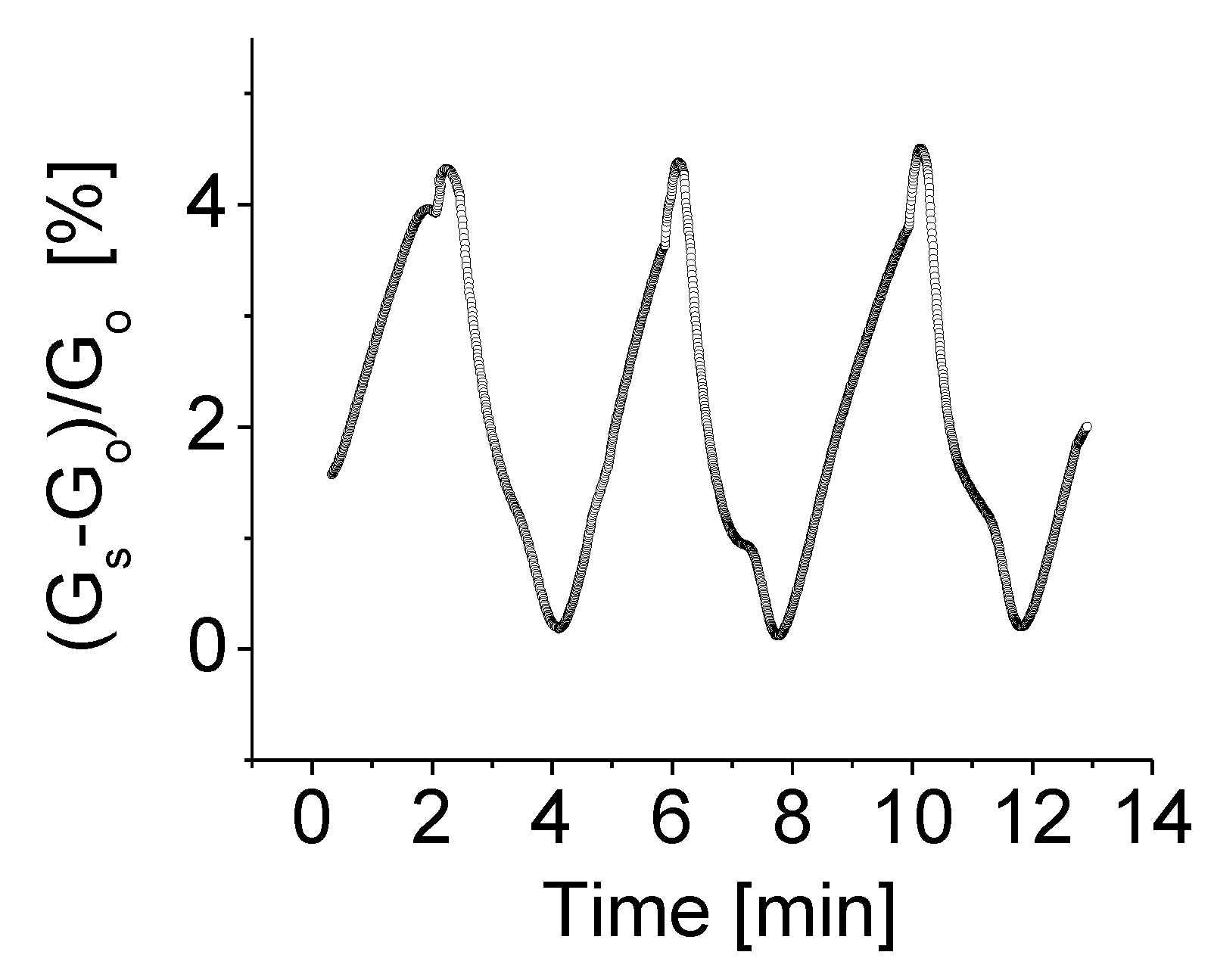
3.3. Response to Target Gases with Different Concentrations
3.4. Bias Voltage Effect
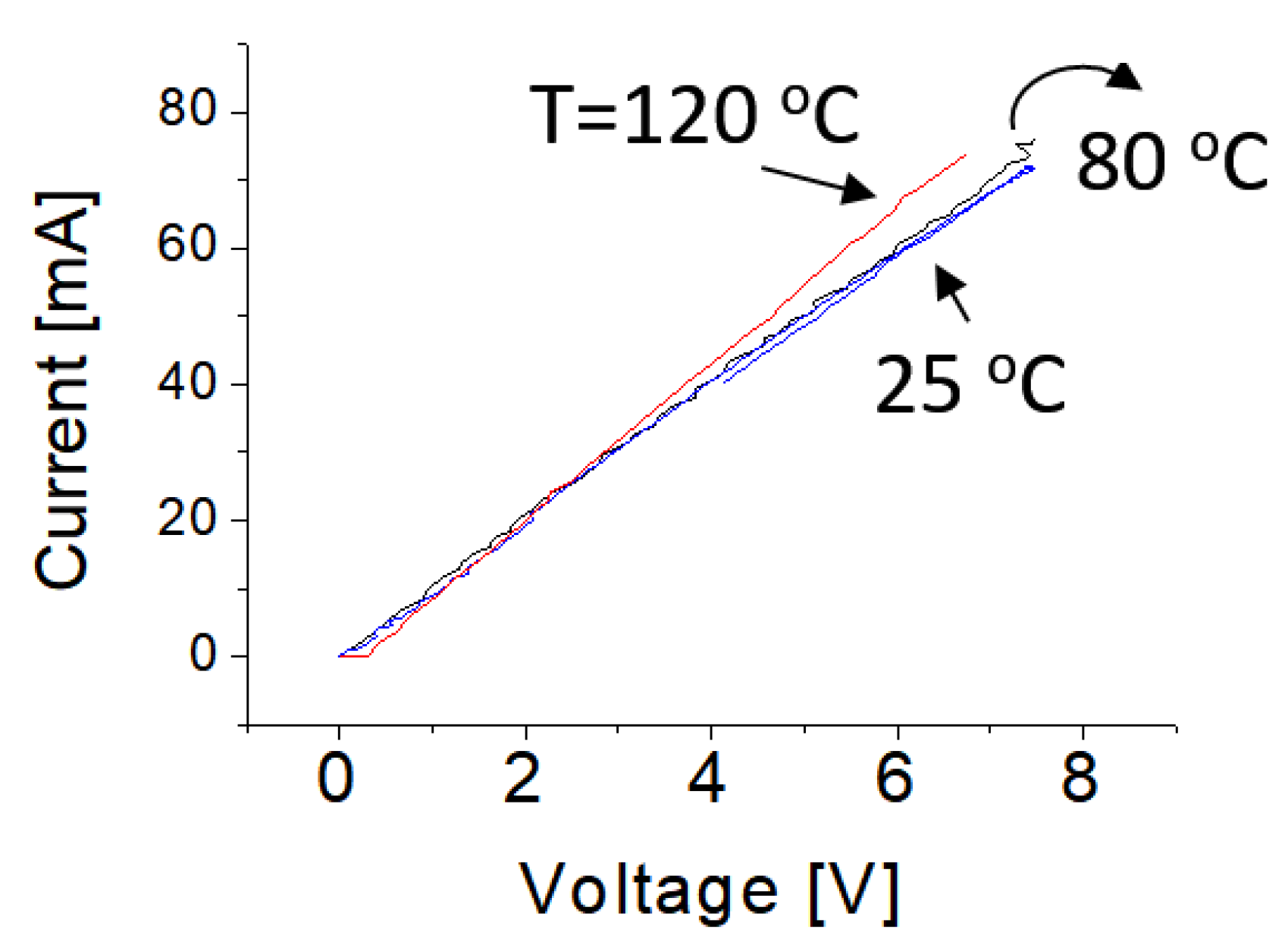
3.5. Temperature Effect
3.6. Sensing Mechanism
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Kim, Y.H.; Phan, D.T.; Ahn, S.; Nam, K.H.; Park, C.M.; Jeon, K.J. Two-dimensional SnS2 materials as high-performance NO2 sensors with fast response and high sensitivity. Sens. Actuators B Chem. 2018, 255, 616–621. [Google Scholar] [CrossRef]
- Han, D.; Yang, W.; Gu, F.; Wang, Z. A low temperature NO2 gas sensor based metal-thiourea complex nanowires. Sens. Actuators B Chem. 2018, 255, 1139–1146. [Google Scholar] [CrossRef]
- Asres, G.A.; Baldoví, J.J.; Dombovari, A.; Järvinen, T.; Lorite, G.S.; Mohl, M.; Shchukarev, A.; Paz, A.P.; Xian, L.; Mikkola, J.P.; et al. Ultrasensitive H2S gas sensors based on p-type WS2 hybrid materials. Nano Res. 2018, 11, 4215–4224. [Google Scholar] [CrossRef]
- Shao, L.; Wua, Z.; Duan, H.; Shaymurat, T. Discriminative and rapid detection of ozone realized by sensor array of Zn2+ doping tailored MoS2 ultrathin nanosheets. Sens. Actuators B Chem. 2018, 258, 937–946. [Google Scholar] [CrossRef]
- Lal, N. Room Temperature Gas Sensing with Ultrathin Au Nanowires. J. Mater. Sci. Eng. 2016, 5, 1000241. [Google Scholar] [CrossRef]
- Joshi, N.; Hayasaka, T.; Liu, Y.; Liu, H.; Oliveira, O.; Lin, L. A review on chemiresistive room temperature gas sensors based on metal oxide nanostructures, graphene and 2D transition metal dichalcogenides. Microchim. Acta 2018, 185, 213. [Google Scholar] [CrossRef] [PubMed]
- Cao, A.; Sudhölter, E.J.R.; de Smet, L. Silicon Nanowire-Based Devices for Gas-Phase Sensing. Sensors 2014, 14, 245–271. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Chu, J.; Wang, L.; Duan, S.; Feng, P. Boron-doped diamond nanowires for CO gas sensing application. Sens. Actuators B Chem. 2017, 241, 383–389. [Google Scholar] [CrossRef]
- Cardoza-Contreras, M.N.; Romo-Herrera, J.M.; Ríos, L.A.; García-Gutiérrez, R.; Zepeda, T.A.; Contreras, O.E. Single ZnO Nanowire-Based Gas Sensors to Detect Low Concentrations of Hydrogen. Sensors 2015, 16, 30539–30544. [Google Scholar] [CrossRef] [PubMed]
- Wan, Q.; Li, Q.H.; Chen, Y.J.; Wang, T.H.; He, X.L.; Li, J.P.; Lin, C.L. Fabrication and ethanol sensing characteristics of ZnO nanowire gas sensors. Appl. Phys. Lett. 2004, 84, 3654–3656. [Google Scholar] [CrossRef]
- Chen, X.; Wong, C.K.Y.; Yuan, C.A.; Zhang, G. Nanowire-based gas sensors. Sens. Actuators B Chem. 2013, 177, 178–195. [Google Scholar] [CrossRef]
- Wang, L.; Wang, S.; Xua, M.; Hua, X.; Zhang, H.; Wang, Y.; Huang, W. An Au-functionalized ZnO nanowire gas sensor for detection of benzene and toluene. Phys. Chem. Chem. Phys. 2013, 15, 17179–17186. [Google Scholar] [CrossRef] [PubMed]
- Lupan, O.; Postica, V.; Labat, F.; Ciofini, I.; Pauporte, T.; Adelung, R. Ultra-sensitive and selective hydrogen nanosensor with fast response at room temperature based on a single Pd/ZnO nanowire. Sens. Actuators B Chem. 2018, 254, 1259–1270. [Google Scholar] [CrossRef]
- Qiang, X.; Hu, M.; Zhao, B.; Qin, Y.; Zhang, T.; Zhou, L.; Liang, J. Preparation of porous silicon/Pd-loaded WO3 nanowires for enhancement of ammonia sensing properties at room temperature. Mater. Sci. Semicond. Process. 2018, 79, 113–118. [Google Scholar] [CrossRef]
- Feng, P.; Shao, F.; Shi, Y.; Wan, Q. Gas Sensors Based on Semiconducting Nanowire Field-Effect Transistors. Sensors 2014, 14, 17406–17429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gupta, A.; Sakthivel, T.; Seal, S. Recent development in 2D materials beyond graphene. Prog. Mater. Sci. 2015, 73, 44–126. [Google Scholar] [CrossRef]
- Frisenda, R.; Navarro-Moratalla, E.; Gant, P.; Pérez De Lara, D.; Jarillo-Herrero, P.; Gorbachev, R.V.; Castellanos-Gomez, A. Recent progress in the assembly of nanodevices and van der Waals heterostructures by deterministic placement of 2D materials. Chem. Soc. Rev. 2018, 47, 53–68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, P.; Ma, R.; Sasaki, T. Recent progress on exploring exceptionally high and anisotropic H+/OH− ion conduction in two-dimensional materials. Chem. Sci. 2018, 9, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Li, Y.; Yu, G.; Li, C.Z.; Zhang, S.B.; Zhou, Z.; Chen, Z. Hydrogenation: A Simple Approach to Realize Semiconductor−Half-Metal−Metal Transition in Boron Nitride Nanoribbon. J. Am. Chem. Soc. 2010, 132, 1699–1705. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.; Pu, H.; Wells, S.A.; Wen, Z.; Mao, S.; Chang, J.; Hersam, M.C.; Chen, J. Ultrahigh sensitivity and layer-dependent sensing performance of phosphorene-based gas sensors. Nat. Commun. 2015, 6, 8632. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sajjad, M.; Feng, X.P. Study the Gas Sensing Properties of Boron Nitride Nanosheets. Mater. Res. Bull. 2013, 49, 35–38. [Google Scholar] [CrossRef]
- Varghese, S.S.; Varghese, S.H.; Swaminathan, S.; Singh, K.K.; Mittal, V. Two-Dimensional Materials for Sensing: Graphene and Beyond. Electronics 2015, 4, 651–687. [Google Scholar] [CrossRef] [Green Version]
- Aldalbahi, A.; Li, E.; Rivera, M.; Velazquez, R.; Altalhi, T.; Peng, X.; Feng, P.X. A new approach for fabrications of SiC based photodetectors. Sci. Rep. 2016, 6, 23457. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, S.F.; Aldalbahi, A.; Feng, P.X. Nanostructured tungsten oxide composite for high-performance gas. Sensors 2015, 15, 27035–27046. [Google Scholar] [CrossRef] [PubMed]
- Gaur, A.P.S.; Sahoo, S.; Ahmadi, M.; Dash, S.P.; Guinel, M.J.-F.; Katiyar, R.S. Surface Energy Engineering for Tunable Wettability through Controlled Synthesis of MoS2. Nano Lett. 2014, 14, 4314–4321. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Qin, L.; Hao, Y.; Kuang, S.; Bai, X.; Chong, Y.-M.; Zhang, W.; Wang, E. Vertically Aligned Boron Nitride Nanosheets: Chemical Vapor Synthesis, Ultraviolet Light Emission, and Superhydrophobicity. ACS Nano 2010, 4, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Guo, W.; Yakobson, B.I. Self-modulated band gap in boron nitride nanoribbons and hydrogenated sheets. Nanoscale 2013, 5, 6381–6387. [Google Scholar] [CrossRef] [PubMed]
- Sajjad, M.; Peng, X.Y.; Chu, J.; Zhang, H.X.; Feng, P.X. Design and Installation of CO2-pulsed laser plasma deposition system for the synthesis of mass product nanostructure. J. Mater. Res. 2013, 28, 1747–1752. [Google Scholar] [CrossRef]
- Bogaerts, A.; Aghaei, M.; Autrique, D.; Lindner, H.; Chen, Z.; Wendelen, W. Computer simulations of laser ablation, plume expansion and plasma formation. Adv. Mater. Res. 2011, 227, 1–10. [Google Scholar] [CrossRef]
- Keidar, M.; Fan, J.; Boyd, I.D.; Beilis, I.I. Vaporization of heated materials into discharge plasmas. J. Appl. Phys. 2001, 89, 3095–3098. [Google Scholar] [CrossRef] [Green Version]
- Aldalbahi, A.; Rivera, M.; Rahaman, M.; Zhou, A.F.; Mohammed, A.W.; Feng, P. High-performance and self-powered deep UV photodetectors based on high quality 2D boron nitride nanosheets. Nanomaterials 2017, 7, 454. [Google Scholar] [CrossRef] [PubMed]
- Sajjad, M.; Ahmadi, M.; Guinel, M.J.-F.; Lin, Y.; Feng, P. Large-scale synthesis of single-crystal and polycrystalline boron nitride nanosheets. J. Mater. Sci. 2013, 48, 2543–2549. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Imamura, S.; Nishio, K.; Fujimoto, K. Influence of temperature and humidity on the electrical sensing of Pt/WO3 thin film hydrogen gas sensor. J. Ceram. Soc. Jpn. 2016, 124, 629–633. [Google Scholar] [CrossRef]
- Wei, P.; Ning, Z.; Ye, S.; Sun, L.; Yang, F.; Wong, K.C.; Westerdahl, D.; Louie, P.K.K. Impact Analysis of Temperature and Humidity Conditions on Electrochemical Sensor Response in Ambient Air Quality Monitoring. Sensors 2018, 18, 59. [Google Scholar] [CrossRef] [PubMed]
- Beheshtian, J.; Peyghan, A.A.; Bagheri, Z.; Kamfiroozi, M. Interaction of small molecules (NO, H2, N2, and CH4) with BN nanocluster surface. Struct. Chem. 2012, 23, 1567–1572. [Google Scholar] [CrossRef]
- Dua, S.K.; Brand, P.; Karg, E.; Heyder, J. Gravitational Transport of Particles in Pure Gases and Gas Mixtures. Aerosol Sci. Technol. 1994, 21, 170–178. [Google Scholar] [CrossRef] [Green Version]
- Matarín, O.; Rimola, A. Influence of Defects in Boron Nitride Nanotubes in the Adsorption of Molecules. Insights from B3LYP-D2* Periodic Simulations. Crystals 2016, 6, 63. [Google Scholar] [CrossRef]
- Wright, J.S.; Lim, W.; Norton, D.P.; Pearton, S.J.; Ren, F.; Johnson, J.L.; Ural, A. Nitride and oxide semiconductor nanostructured hydrogen gas sensors. Semicond. Sci. Technol. 2010, 25, 024002. [Google Scholar] [CrossRef]
- Zhou, Z.; Zhao, J.; Chen, Z.; Gao, X.; Yan, T.; Wen, B.; Schleyer, P.V.R. Comparative Study of Hydrogen Adsorption on Carbon and BN Nanotubes. J. Phys. Chem. B 2006, 110, 13363–13369. [Google Scholar] [CrossRef] [PubMed]
- Ganji, M.D.; Mirnejad, A.; Najafi, A. Theoretical investigation of methane adsorption onto boron nitride and carbon nanotubes. Sci. Technol. Adv. Mater. 2010, 11, 045001. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, B.-Y.; Cho, J.S.; Yoon, J.W.; Na, C.W.; Lee, C.S.; Ahn, J.H.; Kang, Y.C.; Lee, J.H. Extremely sensitive ethanol sensor using Pt-doped SnO2 hollow nanospheres prepared by Kirkendall diffusion. Sens. Actuators B Chem. 2016, 234, 353–360. [Google Scholar] [CrossRef]
- Qadri, M.U.; Diaz, A.F.; Cittadini, M.; Martucci, A.; Pujol, M.C.; Ferré-Borrull, J.; Llobet, E.; Aguiló, M.; Díaz, F. Effect of Pt Nanoparticles on the Optical Gas Sensing Properties of WO3 Thin Films. Sensors 2014, 14, 11427–11443. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giancaterini, L.; Cantalini, C.; Cittadini, M.; Sturaro, M.; Guglielmi, M.; Martucci, A.; Resmini, A.; Anselmi-Tamburini, U. Au and Pt Nanoparticles Effects on the Optical and Electrical Gas Sensing Properties of Sol–Gel-Based ZnO Thin-Film Sensors. IEEE Sens. J. 2015, 15, 1068–1076. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, Q.; Sun, Q.; Jena, P.; Chen, X.S. Electric field enhanced hydrogen storage on polarizable materials substrates. Proc. Natl. Acad. Sci. USA 2010, 107, 2801–2806. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marlid, B.; Larsson, K.; Carlsson, J.-O. Hydrogen and Fluorine Adsorption on the h-BN (001) Plane. J. Phys. Chem. B 1999, 103, 7637–7642. [Google Scholar] [CrossRef]
- Koswattage, K.R.; Shimoyama, I.; Baba, Y.; Sekiguchi, T.; Nakagawa, K. Selective adsorption of atomic hydrogen on a h-BN thin film. J. Chem. Phys. 2011, 135, 014706. [Google Scholar] [CrossRef] [PubMed]
- Weck, P.F.; Kim, E.; Lepp, S.H.; Naduvalath, B.; Sadeghpour, H.R. Dimer-induced stabilization of H adsorbate cluster on BN(0001) surface. Phys. Chem. Chem. Phys. 2008, 10, 5184–5187. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feng, P.X.; Chavez, E.; Malca, C. Super Stable Pollution Gas Sensor Based on Functionalized 2D Boron Nitride Nanosheet Materials for High Humidity Environments. Chemosensors 2018, 6, 49. https://doi.org/10.3390/chemosensors6040049
Feng PX, Chavez E, Malca C. Super Stable Pollution Gas Sensor Based on Functionalized 2D Boron Nitride Nanosheet Materials for High Humidity Environments. Chemosensors. 2018; 6(4):49. https://doi.org/10.3390/chemosensors6040049
Chicago/Turabian StyleFeng, Peter X., Ermides Chavez, and Carlos Malca. 2018. "Super Stable Pollution Gas Sensor Based on Functionalized 2D Boron Nitride Nanosheet Materials for High Humidity Environments" Chemosensors 6, no. 4: 49. https://doi.org/10.3390/chemosensors6040049
APA StyleFeng, P. X., Chavez, E., & Malca, C. (2018). Super Stable Pollution Gas Sensor Based on Functionalized 2D Boron Nitride Nanosheet Materials for High Humidity Environments. Chemosensors, 6(4), 49. https://doi.org/10.3390/chemosensors6040049