Hydrothermal Synthesis and Annealing Effect on the Properties of Gas-Sensitive Copper Oxide Nanowires
Abstract
:1. Introduction
2. Materials and Methods
2.1. Hydrothermal Synthesis
2.2. Gas Sensing Tests
3. Results and Discussions
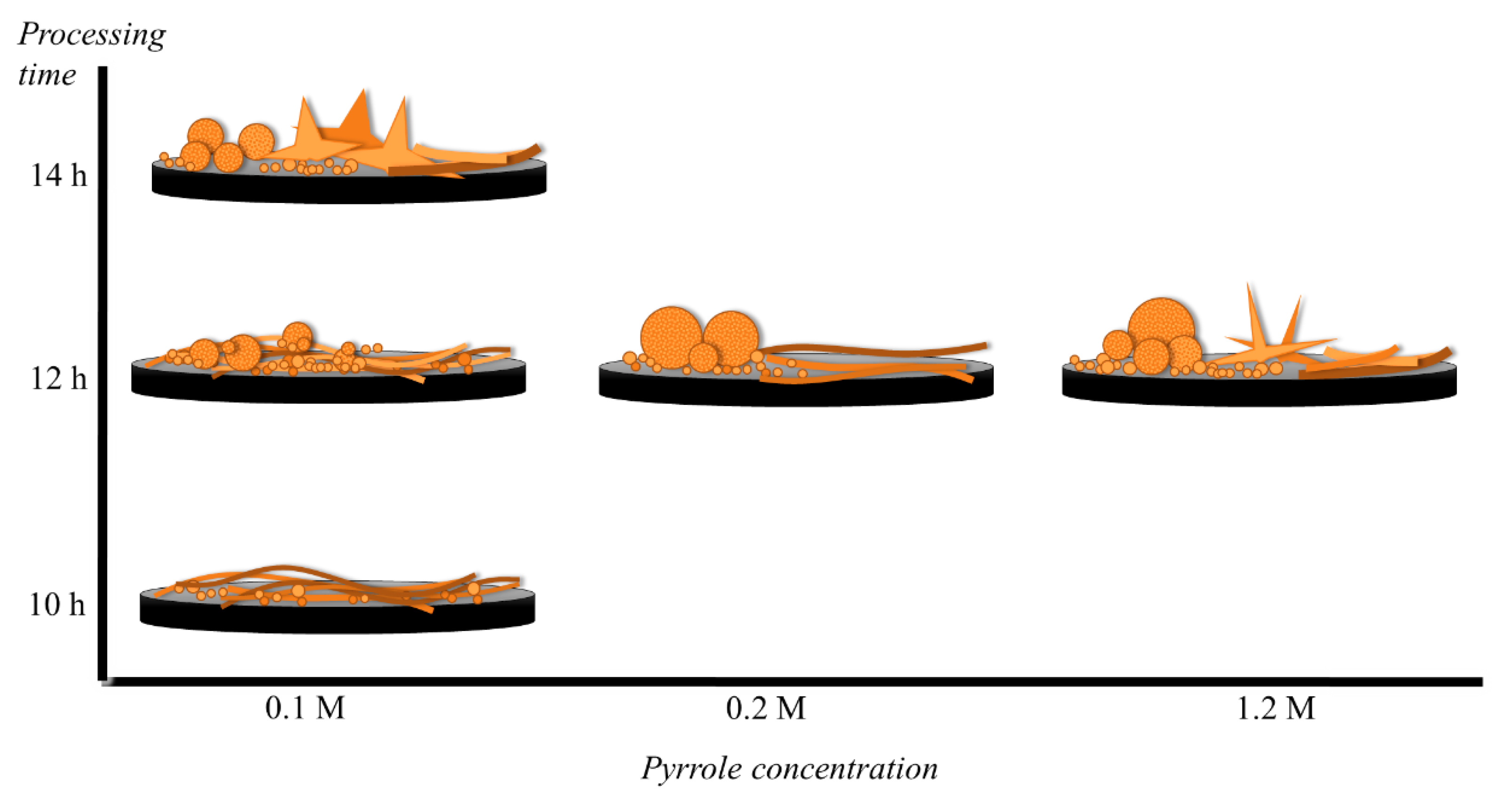
3.1. Copper Oxide Nanowires Formation
3.2. Copper Oxide Hydrothermal Growth
| 2n(C4H5N) + 2Cu2+ + nH2O → nCu2O + 2(2n − 1)H+ + C4H4N  C4H3N C4H3N  2(n−1)C4H4N 2(n−1)C4H4N | (reaction 1) |
| nCu2O + O2 → 2nCuO | (reaction 2) |
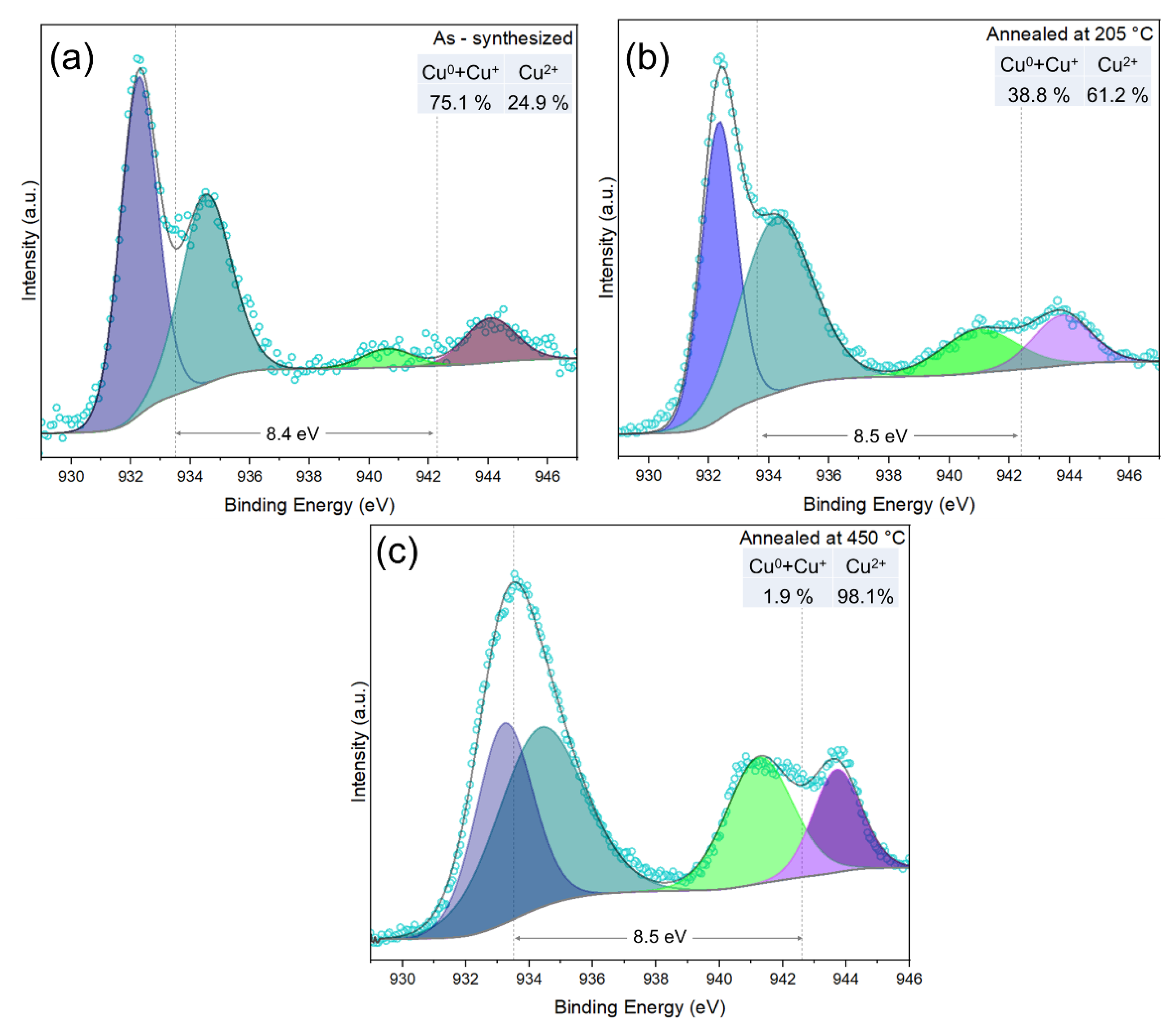
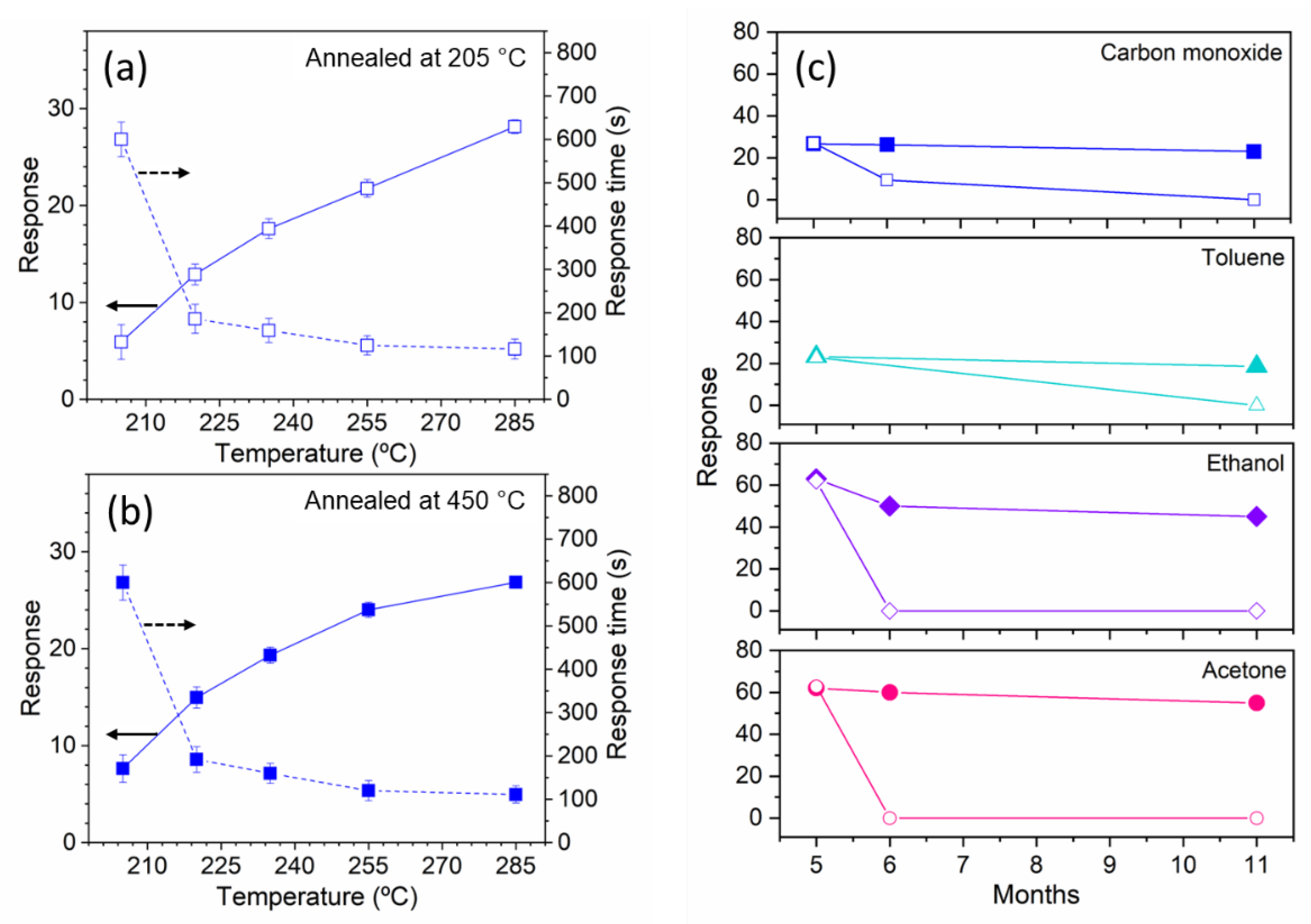
3.3. Annealing Temperature Effect on the Copper Oxide Nanowires
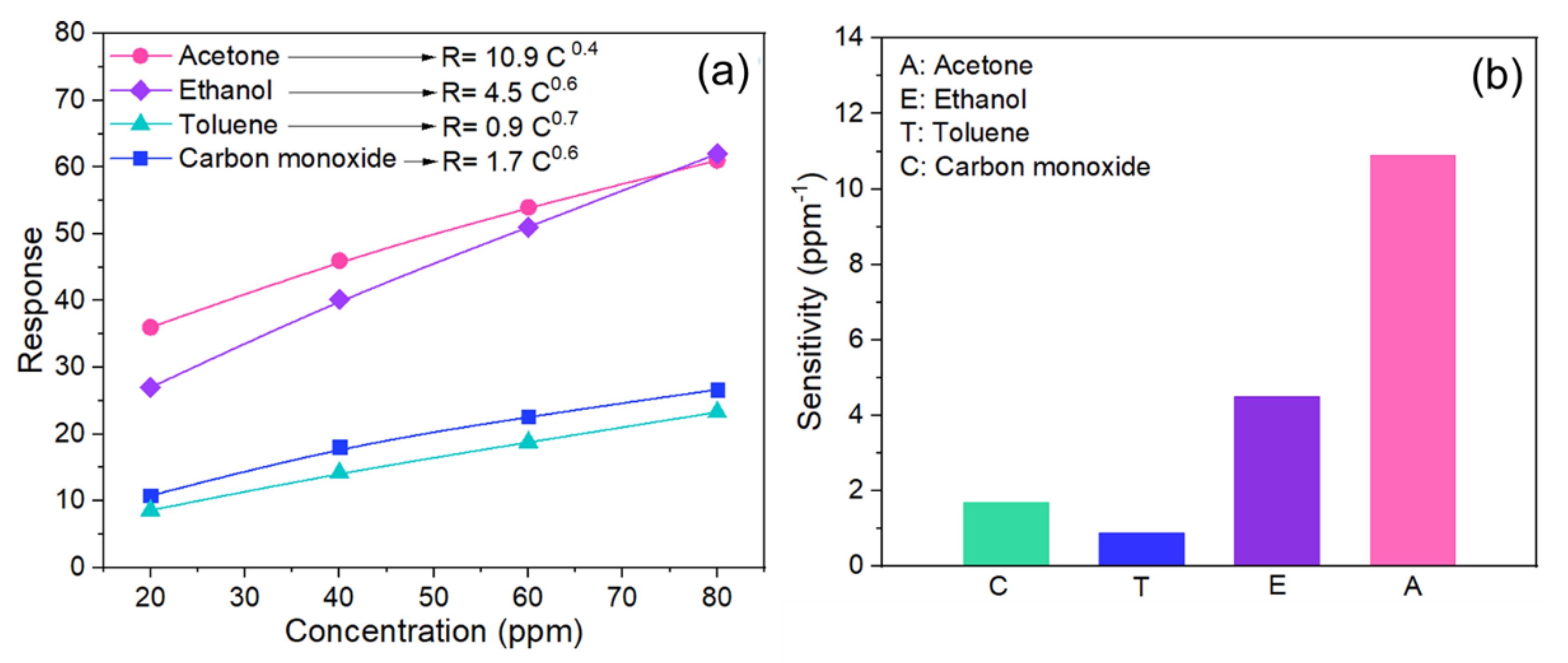
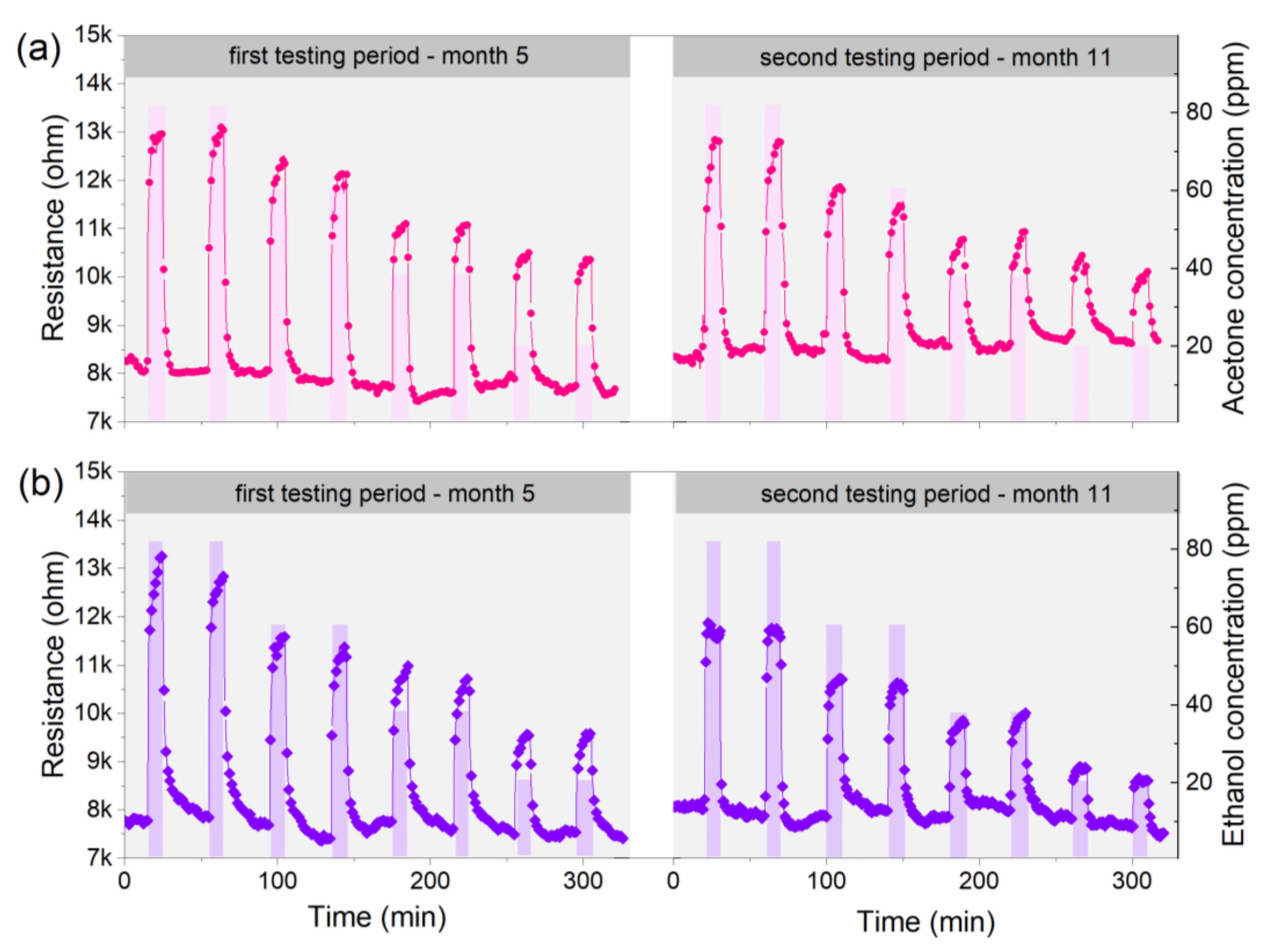
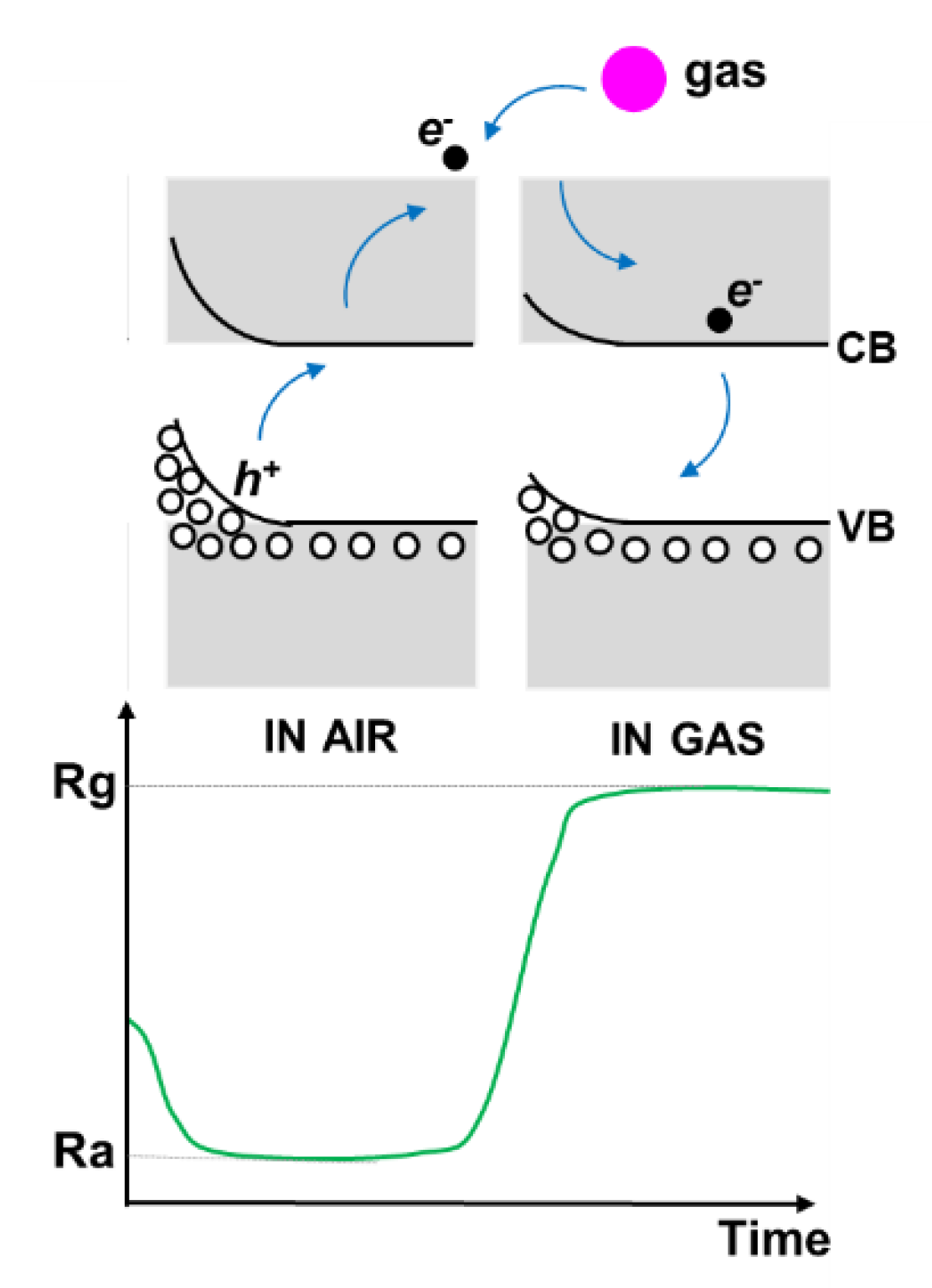
3.4. Gas Sensing Properties of the Copper Oxide Nanowires
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhai, T.; Yao, J. One-Dimensional Nanostructures: Principles and Applications; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013. [Google Scholar] [CrossRef]
- Hontañón, E.; Vallejos, S. One-Dimensional Metal Oxide Nanostructures for Chemical Sensors. In 21st Century Nanostructured Materials—Physics, Chemistry, Classification, and Emerging Applications in Industry, Biomedicine, and Agriculture; IntechOpen: London, UK, 2022; ISBN 978-1-80355-085-5. [Google Scholar] [CrossRef]
- Liang, J.; Kishi, N.; Soga, T.; Jimbo, T. The synthesis of highly aligned cupric oxide nanowires by heating copper foil. J. Nanomater. 2011, 2011, 268508. [Google Scholar] [CrossRef]
- Kang, K.; Park, J.; Kim, B.; Na, K.; Cho, I.; Rho, J.; Yang, D.; Lee, J.Y.; Park, I. Self-Powered Gas Sensor Based on a Photovoltaic Cell and a Colorimetric Film with Hierarchical Micro/Nanostructures. ACS Appl. Mater. Interfaces 2020, 12, 39024–39032. [Google Scholar] [CrossRef] [PubMed]
- Umar, A.; Ibrahim, A.A.; Nakate, U.T.; Albargi, H.; Alsaiari, M.A.; Ahmed, F.; Alharthi, F.A.; Ali Alghamdi, A.; Al-Zaqri, N. Fabrication and characterization of CuO nanoplates based sensor device for ethanol gas sensing application. Chem. Phys. Lett. 2021, 763, 138204. [Google Scholar] [CrossRef]
- Hu, Q.; Zhang, W.; Wang, X.; Wang, Q.; Huang, B.; Li, Y.; Hua, X.; Liu, G.; Li, B.; Zhou, J.; et al. Binder-free CuO nanoneedle arrays based tube-type sensor for H2S gas sensing. Sens. Actuators B Chem. 2021, 326, 128993. [Google Scholar] [CrossRef]
- Umar, A.; Alshahrani, A.A.; Algarni, H.; Kumar, R. CuO nanosheets as potential scaffolds for gas sensing applications. Sens. Actuators B Chem. 2017, 250, 24–31. [Google Scholar] [CrossRef]
- Hien, V.X.; Minh, N.H.; Son, D.T.; Nghi, N.T.; Phuoc, L.H.; Khoa, C.T.; Vuong, D.D.; Chien, N.D.; Heo, Y.W. Acetone sensing properties of CuO nanowalls synthesized via oxidation of Cu foil in aqueous NH4OH. Vacuum 2018, 150, 129–135. [Google Scholar] [CrossRef]
- Yang, F.; Guo, J.; Liu, M.; Yu, S.; Yan, N.; Li, J.; Guo, Z. Design and understanding of a high-performance gas sensing material based on copper oxide nanowires exfoliated from a copper mesh substrate. J. Mater. Chem. A 2015, 3, 20477–20481. [Google Scholar] [CrossRef]
- Şişman, O.; Kılınç, N.; Öztürk, Z.Z. Structural, electrical and H2 sensing properties of copper oxide nanowires on glass substrate by anodization. Sens. Actuators B Chem. 2016, 236, 1118–1125. [Google Scholar] [CrossRef]
- Sondors, R.; Kosmaca, J.; Kunakova, G.; Jasulaneca, L.; Ramma, M.M.; Meija, R.; Kauranens, E.; Antsov, M.; Erts, D. Size distribution, mechanical and electrical properties of cuo nanowires grown by modified thermal oxidation methods. Nanomaterials 2020, 10, 1051. [Google Scholar] [CrossRef]
- Zhong, M.L.; Zeng, D.C.; Liu, Z.W.; Yu, H.Y.; Zhong, X.C.; Qiu, W.Q. Synthesis, growth mechanism and gas-sensing properties of large-scale CuO nanowires. Acta Mater. 2010, 58, 5926–5932. [Google Scholar] [CrossRef]
- Pathiraja, G.; Yarbrough, R.; Rathnayake, H. Fabrication of ultrathin CuO nanowires augmenting oriented attachment crystal growth directed self-assembly of Cu(OH)2 colloidal nanocrystals. Nanoscale Adv. 2020, 2, 2897–2906. [Google Scholar] [CrossRef]
- Tan, J.; Dun, M.; Li, L.; Zhao, J.; Li, X.; Hu, Y.; Huang, G.; Tan, W.; Huang, X. Self-template derived CuO nanowires assembled microspheres and its gas sensing properties. Sens. Actuators B Chem. 2017, 252, 1–8. [Google Scholar] [CrossRef]
- Tan, Y.; Xue, X.; Peng, Q.; Zhao, H.; Wang, T.; Li, Y. Controllable fabrication and electrical performance of single crystalline Cu2O nanowires with high aspect ratios. Nano Lett. 2007, 7, 3723–3728. [Google Scholar] [CrossRef]
- Du, G.H.; Van Tendeloo, G. Cu(OH)2 nanowires, CuO nanowires and CuO nanobelts. Chem. Phys. Lett. 2004, 393, 64–69. [Google Scholar] [CrossRef]
- Shi, G.; Liu, J.; Chen, B.; Bao, Y.; Xu, J. Phase-controlled growth of cubic phase CuO nanoparticles by chemical vapor deposition. IPSS Appl. Mater. Sci. 2017, 214, 1700041. [Google Scholar] [CrossRef]
- Cao, M.; Wang, Y.; Guo, C.; Qi, Y.; Hu, C.; Wang, E. A simple route towards CuO nanowires and nanorods. J. Nanosci. Nanotechnol. 2004, 4, 824–828. [Google Scholar] [CrossRef]
- Gounder Thangamani, J.; Khadheer Pasha, S.K. Hydrothermal synthesis of copper (II) oxide-nanoparticles with highly enhanced BTEX gas sensing performance using chemiresistive sensor. Chemosphere 2021, 277, 130237. [Google Scholar] [CrossRef]
- Mohamed, R.M.; Harraz, F.A.; Shawky, A. CuO nanobelts synthesized by a template-free hydrothermal approach with optical and magnetic characteristics. Ceram. Int. 2014, 40, 2127–2133. [Google Scholar] [CrossRef]
- Kevin, M.; Ong, W.L.; Lee, G.H.; Ho, G.W. Formation of hybrid structures: Copper oxide nanocrystals templated on ultralong copper nanowires for open network sensing at room temperature. Nanotechnology 2011, 22, 235701. [Google Scholar] [CrossRef]
- Filipič, G.; Cvelbar, U. Copper oxide nanowires: A review of growth. Nanotechnology 2012, 23, 194001. [Google Scholar] [CrossRef]
- Zoolfakar, A.S.; Rani, R.A.; Morfa, A.J.; O’Mullane, A.P.; Kalantar-Zadeh, K. Nanostructured copper oxide semiconductors: A perspective on materials, synthesis methods and applications. J. Mater. Chem. C 2014, 2, 5247–5270. [Google Scholar] [CrossRef]
- Rajabzadeh, M.; Khalifeh, R.; Eshghi, H.; Sorouri, M. Design and Preparation of Hallow Mesoporous Silica Spheres Include CuO and Its Catalytic Performance for Synthesis of 1,2,3-Triazole Compounds via the Click Reaction in Water. Catal. Lett. 2019, 149, 1125–1134. [Google Scholar] [CrossRef]
- Arfaoui, L.; Janene, F.; Kouass, S.; Mignard, S.; Touati, F.; Dhaouadi, H. CuO Nanosheets: Synthesis, Characterization, and Catalytic Performance. Russ. J. Inorg. Chem. 2019, 64, 1687–1696. [Google Scholar] [CrossRef]
- Siddiqui, H.; Qureshi, M.S.; Haque, F.Z. Biosynthesis of Flower-Shaped CuO Nanostructures and Their Photocatalytic and Antibacterial Activities. Nano Micro Lett. 2020, 12, 29. [Google Scholar] [CrossRef] [PubMed]
- Arshad, A.; Iqbal, J.; Siddiq, M.; Ali, M.U.; Ali, A.; Shabbir, H.; Nazeer, U.B.; Saleem, M.S. Solar light triggered catalytic performance of graphene-CuO nanocomposite for waste water treatment. Ceram. Int. 2017, 43, 10654–10660. [Google Scholar] [CrossRef]
- Dubal, D.P.; Gund, G.S.; Holze, R.; Jadhav, H.S.; Lokhande, C.D.; Park, C.J. Surfactant-assisted morphological tuning of hierarchical CuO thin films for electrochemical supercapacitors. Dalt. Trans. 2013, 42, 6459–6467. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, Q.; Li, J. Direct electrochemistry of hemoglobin immobilized in CuO nanowire bundles. Talanta 2010, 83, 162–166. [Google Scholar] [CrossRef]
- Stepniowski, W.J.; Misiolek, W.Z. Review of Fabrication Methods, Physical Properties, and Applications of Nanostructured Copper Oxides Formed via Electrochemical Oxidation. Nanomaterials 2018, 8, 379. [Google Scholar] [CrossRef] [Green Version]
- Steinhauer, S. Gas Sensors Based on Copper Oxide Nanomaterials: A Review. Chemosensors 2021, 9, 51. [Google Scholar] [CrossRef]
- Hozák, P.; Vorokhta, M.; Khalakhan, I.; Jarkovská, K.; Cibulková, J.; Fitl, P.; Vlček, J.; Fara, J.; Tomeček, D.; Novotný, M.; et al. New Insight into the Gas-Sensing Properties of CuOx Nanowires by Near-Ambient Pressure XPS. J. Phys. Chem. C 2019, 123, 29739–29749. [Google Scholar] [CrossRef]
- Vallejos, S.; Grácia, I.; Chmela, O.; Figueras, E.; Hubálek, J.; Cané, C. Chemoresistive micromachined gas sensors based on functionalized metal oxide nanowires: Performance and reliability. Sens. Actuators B Chem. 2016, 235, 525–534. [Google Scholar] [CrossRef]
- Kasisomayajula, S.; Jadhav, N.; Gelling, V.J. In situ preparation and characterization of a conductive and magnetic nanocomposite of polypyrrole and copper hydroxychloride. RSC Adv. 2015, 6, 967–977. [Google Scholar] [CrossRef]
- Stoesser, A.; von Seggern, F.; Purohit, S.; Nasr, B.; Kruk, R.; Dehm, S.; Wang, D.; Hahn, H.; Dasgupta, S. Facile fabrication of electrolyte-gated single-crystalline cuprous oxide nanowire field-effect transistors. Nanotechnology 2016, 27, 415205. [Google Scholar] [CrossRef] [PubMed]
- Cao, F.; Jia, S.; Zheng, H.; Zhao, L.; Liu, H.; Li, L.; Zhao, L.; Hu, Y.; Gu, H.; Wang, J. Thermal-induced formation of domain structures in CuO nanomaterials. Phys. Rev. Mater. 2017, 1, 053401. [Google Scholar] [CrossRef]
- Han, Z.; Lu, L.; Zhang, H.W.; Yang, Z.Q.; Wang, F.H.; Lu, K. Comparison of the Oxidation Behavior of Nanocrystalline and Coarse-Grain Copper. Oxid. Met. 2005, 63, 261–275. [Google Scholar] [CrossRef]
- Yabuki, A.; Tanaka, S. Oxidation behavior of copper nanoparticles at low temperature. Mater. Res. Bull. 2011, 46, 2323–2327. [Google Scholar] [CrossRef]
- Mardiansyah, D.; Badloe, T.; Triyana, K.; Mehmood, M.Q.; Raeis-Hosseini, N.; Lee, Y.; Sabarman, H.; Kim, K.; Rho, J. Effect of temperature on the oxidation of Cu nanowires and development of an easy to produce, oxidation-resistant transparent conducting electrode using a PEDOT:PSS coating. Sci. Rep. 2018, 8, 10639. [Google Scholar] [CrossRef]
- Biesinger, M.C. Advanced analysis of copper X-ray photoelectron spectra. Surf. Interface Anal. 2017, 49, 1325–1334. [Google Scholar] [CrossRef]
- Biesinger, M.C.; Payne, B.P.; Grosvenor, A.P.; Lau, L.W.M.W.; Gerson, A.R.; Smart, R.S.C.; StC Smart, R. Resolving surface chemical states in XPS analysis of first row transition metals, oxides and hydroxides: Cr, Mn, Fe, Co and Ni. Appl. Surf. Sci. 2011, 257, 2717–2730. [Google Scholar] [CrossRef]
- Vuong, N.M.; Chinh, N.D.; Huy, B.T.; Lee, Y.I. CuO-decorated ZnO hierarchical nanostructures as efficient and established sensing materials for H2S Gas Sensors. Sci. Rep. 2016, 6, 26736. [Google Scholar] [CrossRef]
- Grosvenor, A.P.; Kobe, B.A.; Biesinger, M.C.; McIntyre, N.S. Investigation of multiplet splitting of Fe 2p XPS spectra and bonding in iron compounds. Surf. Interface Anal. 2004, 36, 1564–1574. [Google Scholar] [CrossRef]
- Claros, M.; Setka, M.; Jimenez, Y.P.; Vallejos, S. AACVD Synthesis and Characterization of Iron and Copper Oxides Modified ZnO Structured Films. Nanomaterials 2020, 10, 471. [Google Scholar] [CrossRef] [PubMed]
- Bejaoui, A.; Guerin, J.; Aguir, K. Modeling of a p-type resistive gas sensor in the presence of a reducing gas. Sens. Actuators B Chem. 2013, 181, 340–347. [Google Scholar] [CrossRef]
- Li, Z.; Li, H.; Wu, Z.; Wang, M.; Luo, J.; Torun, H.; Hu, P.; Yang, C.; Grundmann, M.; Liu, X.; et al. Advances in designs and mechanisms of semiconducting metal oxide nanostructures for high-precision gas sensors operated at room temperature. Mater. Horiz. 2019, 6, 470–506. [Google Scholar] [CrossRef]
- Yang, C.; Su, X.; Xiao, F.; Jian, J.; Wang, J. Gas sensing properties of CuO nanorods synthesized by a microwave-assisted hydrothermal method. Sens. Actuators B Chem. 2011, 158, 299–303. [Google Scholar] [CrossRef]


| Ethanol | Methanol | n-propanol | Acetone | Toluene | Benzene | Xylene | Ethylbenzene | Ethyl Acetate | Benzaldehyde | Benzyl Alcohol | Cyclo Hexane | Hydrogen Sulfide | Carbon Monoxide | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HS–NWS | 80–140 ϕ 18,000 L | ❿ | ❿ | ❸ | ❹ | 285 °C—80 ppm | This work | ||||||||||
| CuFE–NWlls | 45 T | ❹ | ❿ | 320 °C—500 ppm | [8] | ||||||||||||
| CuME–NWs | 50–70 ϕ | ❼ | ❻ | ❸ | ❸ | ❿ | ❼ | 260 °C—100 ppm | [9] | ||||||||
| KR–NW | 15 ϕ | ❼ | ❹ | ❿ | ❼ | ❸ | ❺ | 190 °C—100 ppm | [14] | ||||||||
| MAHS–NRs | 15–20 ϕ 60–80 L | ❿ | ❺ | ❺ | ❶ | ❺ | ❽ | ❸ | 220 °C—100 ppm | [45] | |||||||
| NPs | 20 CS | ❿ | ❷ | ❺ | ❹ | 160 °C—240 ppm | [19] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Claros, M.; Gràcia, I.; Figueras, E.; Vallejos, S. Hydrothermal Synthesis and Annealing Effect on the Properties of Gas-Sensitive Copper Oxide Nanowires. Chemosensors 2022, 10, 353. https://doi.org/10.3390/chemosensors10090353
Claros M, Gràcia I, Figueras E, Vallejos S. Hydrothermal Synthesis and Annealing Effect on the Properties of Gas-Sensitive Copper Oxide Nanowires. Chemosensors. 2022; 10(9):353. https://doi.org/10.3390/chemosensors10090353
Chicago/Turabian StyleClaros, Martha, Isabel Gràcia, Eduard Figueras, and Stella Vallejos. 2022. "Hydrothermal Synthesis and Annealing Effect on the Properties of Gas-Sensitive Copper Oxide Nanowires" Chemosensors 10, no. 9: 353. https://doi.org/10.3390/chemosensors10090353
APA StyleClaros, M., Gràcia, I., Figueras, E., & Vallejos, S. (2022). Hydrothermal Synthesis and Annealing Effect on the Properties of Gas-Sensitive Copper Oxide Nanowires. Chemosensors, 10(9), 353. https://doi.org/10.3390/chemosensors10090353







