Predictors of Pregnancy Termination among Young Women in Ghana: Empirical Evidence from the 2014 Demographic and Health Survey Data
Abstract
1. Background
2. Materials and Methods
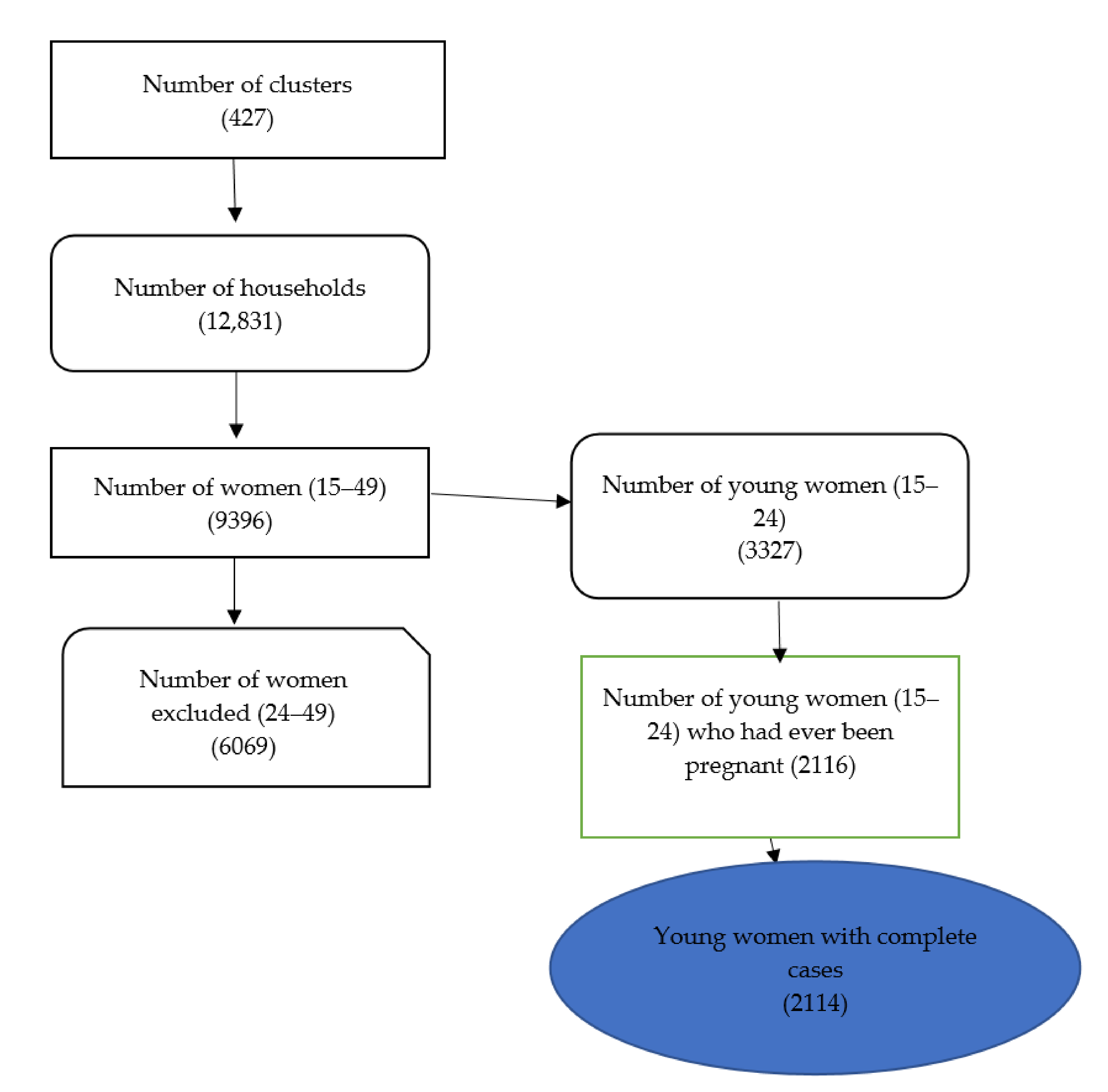
2.1. Data Source
2.2. Study Variables
2.2.1. Dependent Variable
2.2.2. Explanatory Variables
2.3. Statistical Analyses
2.4. Ethics Approval
3. Results
3.1. Descriptive Results on the Prevalence of Pregnancy Termination across Socio-Demographic Characteristics
3.2. Binary Logistic Regression Analysis on the Predictors of Pregnancy Termination
4. Discussion
Strengths and Limitations
5. Conclusions and Policy Implications
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Abortion Rates Drop in More Developed Regions But Fail to Improve in Developing Regions; World Health Organization: Geneva, Switzerland, 2016; Available online: http://www.who.int/reproductivehealth/news/abortion-rates/en/ (accessed on 12 May 2017).
- Zahan, R.; Feng, C.X. Bayesian spatial analysis of socio-demographic factors influencing pregnancy termination and its residual geographic variation among ever-married women of reproductive age in Bangladesh. BMC Public Health 2020, 20, 1348. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Spontaneous and Induced Abortion: Report of a WHO Scientific Group; WHO Technical Report Series No. 461; World Health Organization: Geneva, Switzerland, 1970; pp. 5–11. [Google Scholar]
- Cunnigham, F.G.; Leveno, K.L.; Bloom, S.L.; Hauth, J.C.; Rouse, D.; Spong, C.Y. Williams Obstetrics, 23rd ed.; McGraw-Hill: New York, NY, USA, 2010. [Google Scholar]
- Bulletti, C.; Flamigni, C.; Giacomucci, E. Reproductive failure due to spontaneous abortion and recurrent miscarriage. Hum. Reprod. Update 1996, 2, 118–136. [Google Scholar] [CrossRef]
- World Health Organisation. Definitions and Indicators in Family Planning, Maternal & Child Health and Reproductive Health. Reproductive. Maternal and Child Health European Regional Office; World Health Organisation: Geneva, Switzerland, 2001; Available online: http://www.euro.who.int/document/e68459.pdf (accessed on 6 April 2020).
- WHO. Neonatal and Perinatal Mortality for the Year 2000: Country, Regional and Global Estimates; World Health Organization: Geneva, Switzerland, 2006; Available online: http://whqlibdoc.who.int/publications/2006/9241563206_eng.pdf (accessed on 6 April 2020).
- World Health Organization. Reproductive health indicators: Guidelines for their generation, interpretation and analysis for global monitoring; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- Ganatra, B.; Gerdts, C.; Rossier, C.; Johnson, B.R., Jr.; Tunçalp, Ö.; Assifi, A.; Sedgh, G.; Singh, S.; Bankole, A.; Popinchalk, A.; et al. Global, regional, and subregional classification of abortions by safety, 2010–2014: Estimates from a Bayesian hierarchical model. Lancet 2017, 390, 2372–2381. [Google Scholar] [CrossRef]
- World Health Organisation. Fact Sheet: Preventing Unsafe Abortion; World Health Organisation: Geneva, Switzerland, 2016; Available online: http://www.who.int/mediacentre/factsheets/fs388/en/ (accessed on 6 April 2020).
- Allan Guttmacher Institute. Facts on Abortion in Africa. Available online: http://www.guttmacher.org/pubs/IB_AWW-Africa.pdf (accessed on 18 June 2014).
- Sedgh, G.; Bearak, J.; Singh, S.; Bankole, A.; Popinchalk, A.; Ganatra, B.; Johnston, H.B. Abortion incidence between 1990 and 2014: Global, regional, and subregional levels and trends. Lancet 2016, 388, 258–267. [Google Scholar] [CrossRef]
- World Health Organization. Unsafe Abortion: Global and Regional Estimates of the Incidence and Mortality in 2008, 6th ed.; Department of Reproductive Health and Research, World Health Organization: Geneva, Switzerland, 2011; Available online: http://www.who.int/reproductivehealth/publications/unsafe_abortion (accessed on 18 June 2014).
- Cousens, S.; Blencowe, H.; Stanton, C.; Chou, D.; Ahmed, S.; Steinhardt, L.; Creanga, A.A.; Tunçalp, O.; Balsara, Z.P.; Gupta, S.; et al. National, regional, and worldwide estimates of stillbirth rates in 2009 with trends since 1995: A systematic analysis. Lancet 2011, 377, 1319–1330. [Google Scholar] [CrossRef]
- Blencowe, H.; Cousens, S.; Jassir, F.B.; Say, L.; Chou, D.; Mathers, C.; Hogan, D.; Shiekh, S.; Qureshi, Z.U.; You, D.; et al. National, regional, and worldwide estimates of stillbirth rates in 2015, with trends from 2000: A systematic analysis. Lancet Glob. Health 2016, 4, 98–108. [Google Scholar] [CrossRef]
- Ghana Statistical Service; Ghana Health Service; Macro International. Ghana Maternal Health Survey 2007; GSS, GHS, and ICF International: Rockville, MD, USA, 2009. [Google Scholar]
- Ghana Statistical Service (GSS); Ghana Health Service (GHS); ICF Macro. Ghana Demographic and Health Survey 2008; GSS, GHS, and ICF International: Rockville, MD, USA, 2009. [Google Scholar]
- Chalumeau, M.; Bouvier-Colle, M.-H.; Breart, G. Can clinical risk factors for late stillbirth in West Africa be detected during antenatal care or only during labour? Int. J. Epidemiol. 2002, 31, 661–668. [Google Scholar] [CrossRef]
- Engmann, C.; Walega, P.; Aborigo, R.A.; Adongo, P.; Moyer, C.A.; Lavasani, L.; Williams, J.; Bose, C.; Binka, F.; Hodgson, A. Stillbirths and early neonatal mortality in rural Northern Ghana. Trop. Med. Int. Health 2012, 17, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Ha, Y.P.; Hurt, L.S.; Tawiah-Agyemang, C.; Kirkwood, B.R.; Edmond, K.M. Effect of socioeconomic deprivation and health service utilisation on antepartum and intrapartum stillbirth: Population cohort study from rural Ghana. PLoS ONE 2012, 7, e39050. [Google Scholar] [CrossRef]
- Dassah, E.T.; Odoi, A.T.; Opoku, B.K. Stillbirths and very low Apgar scores among vaginal births in a tertiary hospital in Ghana: A retrospective cross-sectional analysis. BMC Pregnancy Childbirth 2014, 14, 289. [Google Scholar] [CrossRef]
- Amadi, C.N.; Igweze, Z.N.; Orisakwe, O.E. Heavy metals in miscarriages and stillbirths in developing nations. Middle East Fertil. Soc. J. 2017, 22, 91–100. [Google Scholar] [CrossRef]
- Yatich, N.J.; Funkhouser, E.; Ehiri, J.E.; Agbenyega, T.; Stiles, J.K.; Rayner, J.C.; Turpin, A.; Ellis, W.O.; Jiang, Y.; Williams, J.H.; et al. Malaria, intestinal helminths and other risk factors for stillbirth in Ghana. Infect. Dis. Obstet. Gynecol. 2010, 2010, 350763. [Google Scholar] [CrossRef]
- World Health Organization. Safe Abortion: Technical and Policy Guidance for Health Systems; WHO: Geneva, Switzerland, 2012. [Google Scholar]
- World Health Organization. Facts on Induced Abortion Worldwide; World Health Organization: Geneva, Switzerland, 2012; Available online: http://www.who.int/reproductivehealth/publications/unsafe_abortion/induced_abortion_2012.pdf (accessed on 6 April 2020).
- Sundaram, A.; Juarez, F.; Bankole, A.; Singh, S. Factors associated with abortion-seeking and obtaining a safe abortion in Ghana. Stud. Fam. Plan. 2012, 43, 273–286. [Google Scholar] [CrossRef]
- United Nations Department of Economic and Social Affairs (UNDESA). Population Policy Data Bank; Population Division UNDESA: New York, NY, USA, 2003. [Google Scholar]
- Morhee, R.A.S.; Morhee, E.S.K. Overview of the law and availability of abortion services in Ghana. Ghana Med. J. 2006, 40, 80–86. [Google Scholar] [CrossRef]
- Salifu, M.G.; Mohammed, K. Prevalence and Predictors of Contraceptives Use among Women Aged (15–49 years) with Induced Abortion History in Ghana. Adv. Prev. Med. 2020, 2020, 2630905. [Google Scholar] [CrossRef]
- Adisah-Atta, I.; Dim, E.E. Justification of Abortion in West Africa and Interplay of Sociodemographic Predictors: A Comparative Study of Ghana and Nigeria. SAGE Open 2019, 9, 2158244019834368. [Google Scholar] [CrossRef]
- Ahiadeke, C. Incidence of induced abortion in southern Ghana. Int. Fam. Plan. Perspect. 2001, 27, 96–101. [Google Scholar] [CrossRef]
- Lithur, N.O. Advocacy for safe abortion, information dissemination, anti-abortion activities in Ghana. In In Proceedings of the International Consortium for Medical Abortion, Muldersdrift, South Africa, 17–20 October 2004. [Google Scholar]
- Ghana Statistical Service (GSS); Ghana Health Service (GHS); ICF Macro. Accra: Ghana Maternal and Health Survey; Ghana Statistical Service: Accra, Ghana, 2017. [Google Scholar]
- Keogh, S.C.; Otupiri, E.; Chiu, D.W.; Polis, C.B.; Hussain, R.; Bell, S.O.; Nakua, E.K.; Larsen-Reindorf, R. Estimating the incidence of abortion: A comparison of five approaches in Ghana. BMJ Glob. Health 2020, 5, e002129. [Google Scholar] [CrossRef]
- World Health Organization (WHO); UNICEF; UNFPA; World Bank. Trends in Maternal Mortality: 1990 to 2010; WHO: Geneva, Switzerland, 2012. [Google Scholar]
- Mills, S.; Williams, J.E.; Wak, G.; Hodgson, A. Maternal mortality decline in the Kassena-Nankana district of Northern Ghana. Matern. Child Health J. 2008, 12, 577–585. [Google Scholar] [CrossRef]
- Keogh, S.C.; Stillman, M.; Awusabo-Asare, K.; Sidze, E.; Monzón, A.S.; Motta, A.; Leong, E. Challenges to implementing national comprehensive sexuality education curricula in low-and middle-income countries: Case studies of Ghana, Kenya, Peru and Guatemala. PLoS ONE 2018, 13, e0200513. [Google Scholar] [CrossRef]
- Adanu, R.M.; Ntumy, M.N.; Tweneboah, E. Profile of women with abortion complications in Ghana. Trop. Dr. 2005, 35, 139–142. [Google Scholar] [CrossRef]
- Klutsey, E.E.; Ankomah, A. Factors associated with induced abortion at selected hospitals in the Volta Region, Ghana. Int. J. Women’s Health 2014, 6, 809. [Google Scholar]
- Biney, A.A. Exploring contraceptive knowledge and use among women experiencing induced abortion in the Greater Accra Region, Ghana. Afr. J. Reprod. Health 2011, 15, 37–46. [Google Scholar] [PubMed]
- Payne, C.M.; Debbink, M.P.; Steele, E.A.; Buck, C.T.; Martin, L.A.; Hassinger, J.A.; Harris, L.H. Why women are dying from unsafe abortion: Narratives of Ghanaian abortion providers. Afr. J. Reprod. Health 2013, 17, 118–128. [Google Scholar] [PubMed]
- Patel, C.J.; Myeni, M.C. Attitudes toward abortion in a sample of South African female university students. J. Appl. Soc. Psychol. 2008, 38, 736–750. [Google Scholar] [CrossRef]
- Ajzen, I. The theory of planned behavior. Organ. Behav. Hum. Decis. Process. 1991, 50, 179–211. [Google Scholar] [CrossRef]
- Ghana Statistical Service (GSS); Ghana Health Service (GHS); ICF International. Ghana Demographic and Health Survey 2014; GSS, GHS, and ICF International: Rockville, MA, USA, 2015. [Google Scholar]
- Souza, M.G.; Fusco, C.L.; Andreoni, S.A.; Silva, R.D.S. Prevalence and sociodemographic characteristics of women with induced abortion in a population sample of São Paulo, Brazil. Rev. Bras. Epidemiol. 2014, 17, 297–312. [Google Scholar] [CrossRef]
- Seidu, A.A.; Ahinkorah, B.O.; Ameyaw, E.K.; Hubert, A.; Agbemavi, W.; Armah-Ansah, E.K.; Budu, E.; Sambah, F.; Tackie, V. What has women’s reproductive health decision-making capacity and other factors got to do with pregnancy termination in sub-Saharan Africa? evidence from 27 cross-sectional surveys. PLoS ONE 2020, 15, e0235329. [Google Scholar] [CrossRef]
- Chae, S.; Desai, S.; Crowell, M.; Sedgh, G.; Singh, S. Characteristics of women obtaining induced abortions in selected low-and middle-income countries. PLoS ONE 2017, 12, e0172976. [Google Scholar]
- Dickson, K.S.; Adde, K.S.; Ahinkorah, B.O. Socio–economic determinants of abortion among women in Mozambique and Ghana: Evidence from demographic and health survey. Arch. Public Health 2018, 76, 37. [Google Scholar] [CrossRef]
- Seidu, A.A.; Ahinkorah, B.O.; Agbemavi, W.; Amu, H.; Bonsu, F. Reproductive health decision-making capacity and pregnancy termination among Ghanaian women: Analysis of the 2014 Ghana demographic and health survey. J. Public Health 2019, 29, 85–94. [Google Scholar] [CrossRef]
- Dos Santos, A.P.V.; Coelho, E.D.A.C.; Gusmão, M.E.N.; da Silva, D.O.; Marques, P.F.; Almeida, M.S. Factors associated with abortion in women of reproductive age. Rev. Bras. Ginecol. Obs. RBGO Gynecol. Obstet. 2016, 38, 273–279. [Google Scholar] [CrossRef]
- Mote, C.V.; Otupiri, E.; Hindin, M.J. Factors associated with induced abortion among women in Hohoe, Ghana. Afr. J. Reprod. Health 2010, 14, 115–121. [Google Scholar]
- Biddlecom, A. Unsafe abortion in Kenya. Issues Brief 2008, 4, 1–4. [Google Scholar]
- Maina, B.W.; Mutua, M.M.; Sidze, E.M. Factors associated with repeat induced abortion in Kenya. BMC Public Health 2015, 15, 1048. [Google Scholar] [CrossRef]
- Bankole, A.; Oye-Adeniran, B.A.; Singh, S.; Adewole, I.F.; Wulf, D.; Sedgh, G.; Hussain, R. Unwanted Pregnancy and Induced Abortion in Nigeria: Causes and Consequences. 2006. Available online: http://ir.library.ui.edu.ng/handle/123456789/2693 (accessed on 6 April 2020).
- Bain, L.E.; Zweekhorst, M.B.; Amoakoh-Coleman, M.; Muftugil-Yalcin, S.; Omolade, A.I.; Becquet, R.; de Cock Buning, T. To keep or not to keep? Decision making in adolescent pregnancies in Jamestown, Ghana. PLoS ONE 2019, 14, e0221789. [Google Scholar]
- Aniteye, P.; O’Brien, B.; Mayhew, S.H. Stigmatized by association: Challenges for abortion service providers in Ghana. BMC Health Serv. Res. 2016, 16, 486. [Google Scholar] [CrossRef] [PubMed]
- Schwandt, H.M.; Creanga, A.A.; Danso, K.A.; Adanu, R.M.; Agbenyega, T.; Hindin, M.J. A comparison of women with induced abortion, spontaneous abortion and ectopic pregnancy in Ghana. Contraception 2011, 84, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Calves, A.E. Abortion risk and decision making among young people in rural Cameroon. Stud. Fam. Plan. 2012, 33, 249–260. [Google Scholar] [CrossRef]
- Bankole, A.; Singh, S.; Haas, T. Characteristics of women who obtain induced abortion: A worldwide review. Int. Fam. Plan. Perspect. 1999, 25, 68–77. [Google Scholar] [CrossRef]
- Guttmacher Institute. Abortion in Ghana; Guttmacher Institute: New York, NY, USA, 2013; Available online: https://www.guttmacher.org/pubs/FB-Abortion-in-Ghana.pdf (accessed on 6 April 2020).
- Adjei, G.; Enuameh, Y.; Asante, K.P.; Baiden, F.; Nettey, O.E.A.; Abubakari, S.; Mahama, E.; Gyaase, S.; Owusu-Agyei, S. Predictors of abortions in rural Ghana: A cross-sectional study. BMC Public Health 2015, 15, 202. [Google Scholar] [CrossRef]
- Ouédraogo, R.; Sundby, J. Social determinants and access to induced abortion in Burkina Faso: From two case studies. Obstet. Gynecol. Int. 2014, 2014, 402456. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, I.A.; Onwudiegwu, U. Sociodemographic determinants of complicated unsafe abortions in a semi-urban Nigerian town: A four-year review. West Indian Med. J. 2012, 61, 163–167. [Google Scholar] [PubMed]
- Puri, M.; Lamichhane, P.; Harken, T.; Blum, M.; Harper, C.C.; Darney, P.D.; Henderson, J.T. Sometimes they used to whisper in our ears: Health care workers’ perceptions of the effects of abortion legalization in Nepal. BMC Public Health 2012, 12, 297. [Google Scholar] [CrossRef]
- Oliveras, E.; Ahiadeke, C.; Adanu, R.M.; Hill, A.G. Clinic-based surveillance of adverse pregnancy outcomes to identify induced abortions in Accra, Ghana. Stud. Fam. Plan. 2008, 39, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Ahinkorah, B.O.; Seidu, A.A.; Appiah, F.; Baatiema, L.; Sambah, F.; Budu, E.; Ameyaw, E.K. What has reproductive health decision-making capacity got to do with unintended pregnancy? Evidence from the 2014 Ghana Demographic and Health Survey. PLoS ONE 2019, 14, e0223389. [Google Scholar] [CrossRef]
- Ahinkorah, B.O. Individual and contextual factors associated with mistimed and unwanted pregnancies among adolescent girls and young women in selected high fertility countries in sub-Saharan Africa: A multilevel mixed effects analysis. PLoS ONE 2020, 15, e0241050. [Google Scholar] [CrossRef]
- Bankole, A.; Malarcher, S. Removing barriers to adolescents’ access to contraceptive information and services. Stud. Fam. Plan. 2010, 41, 117–124. [Google Scholar] [CrossRef]
- Chandra-Mouli, V.; McCarraher, D.R.; Phillips, S.J.; Williamson, N.E.; Hainsworth, G. Contraception for adolescents in low and middle income countries: Needs, barriers, and access. Reprod. Health 2014, 11, 1. [Google Scholar] [CrossRef]
- Challa, S.; Manu, A.; Morhe, E.; Dalton, V.K.; Loll, D.; Dozier, J.; Zochowski, M.K.; Boakye, A.; Adanu, R.; Hall, K.S. Multiple levels of social influence on adolescent sexual and reproductive health decision-making and behaviors in Ghana. Women Health 2018, 58, 434–450. [Google Scholar] [CrossRef]
- Munakampe, M.N.; Zulu, J.M.; Michelo, C. Contraception and abortion knowledge, attitudes and practices among adolescents from low and middle-income countries: A systematic review. BMC Health Serv. Res. 2018, 18, 909. [Google Scholar] [CrossRef]
- Rossier, C.; Guiella, G.; Ouédraogo, A.; Thiéba, B. Estimating clandestine abortion with the confidants method—Results from Ouagadougou, Burkina Faso. Soc. Sci. Med. 2006, 62, 254–266. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, O. Access to health care in developing countries: Breaking down demand side barriers. Cad. Saude Publica 2007, 23, 2820–2834. [Google Scholar] [CrossRef] [PubMed]
- Mariolis, A.; Mihas, C.; Alevizos, A.; Mariolis-Sapsakos, T.; Marayiannis, K.; Papathanasiou, M.; Gizlis, V.; Karanasios, D.; Merkouris, B. Comparison of primary health care services between urban and rural settings after the introduction of the first urban health centre in Vyronas, Greece. BMC Health Serv. Res. 2008, 8, 124. [Google Scholar] [CrossRef]
- Alhassan, R.K.; Nketiah-Amponsah, E. Frontline staff motivation levels and health care quality in rural and urban primary health facilities: A baseline study in the Greater Accra and Western regions of Ghana. Health Econ. Rev. 2016, 6, 39. [Google Scholar] [CrossRef] [PubMed][Green Version]
| Variables | Weighted n | Percentage | Pregnancy Termination |
|---|---|---|---|
| Age | |||
| 15–19 | 700 | 33.1 | 7.7 |
| 20–24 | 1414 | 66.9 | 21.7 |
| Wealth Quintile | |||
| Poorest | 356 | 16.8 | 11.4 |
| Poorer | 433 | 20.5 | 14.8 |
| Middle | 521 | 24.6 | 17.9 |
| Richer | 463 | 21.9 | 22.6 |
| Richest | 343 | 16.2 | 17.2 |
| Occupation | |||
| Not working | 868 | 41.1 | 10.7 |
| Working | 1246 | 58.9 | 21.5 |
| Educational level | |||
| No education | 221 | 10.4 | 16.7 |
| Primary | 380 | 18.0 | 19.2 |
| Secondary | 1432 | 67.7 | 16.5 |
| Higher | 82 | 3.9 | 17.7 |
| Religion | |||
| Christian | 1717 | 81 | 17.4 |
| Islam | 311 | 14.7 | 13.9 |
| Traditional/no religion | 86 | 1.3 | 22.8 |
| Marital Status | |||
| Never married | 1314 | 62.2 | 13.7 |
| Married | 351 | 16.6 | 20.4 |
| Cohabiting | 362 | 17.1 | 23.0 |
| Divorced/separated | 87 | 4.1 | 29.2 |
| Age at first sex | |||
| Less than 15 years | 304 | 14.4 | 17.5 |
| 15–19 years | 1370 | 64.8 | 18.6 |
| 20–24 years | 188 | 8.9 | 7.1 |
| At first union | 251 | 11.9 | 16.0 |
| Parity | |||
| No births | 1157 | 54.7 | 15.5 |
| One birth | 580 | 27.5 | 17.6 |
| Two births | 264 | 12.5 | 22.1 |
| Three or more births | 97 | 4.6 | 19.2 |
| Exposure to media | |||
| No | 143 | 6.8 | 12.6 |
| Yes | 1971 | 9.3 | 17.4 |
| Place of Residence | |||
| Urban | 1022 | 48.4 | 20.5 |
| Rural | 1092 | 51.6 | 13.9 |
| Region | |||
| Western | 284 | 13.4 | 19.8 |
| Central | 217 | 10.3 | 12.0 |
| Greater Accra | 349 | 16.5 | 21.5 |
| Volta | 168 | 8.0 | 14.8 |
| Eastern | 225 | 10.7 | 16.7 |
| Ashanti | 346 | 16.4 | 22.8 |
| Brong Ahafo | 226 | 10.7 | 16.1 |
| Northern | 169 | 8.0 | 8.7 |
| Upper East | 84 | 4.0 | 5.1 |
| Upper West | 46 | 2.2 | 15.1 |
| Variables | Model 1 COR (95% CI) | Model 2 AOR (95% CI) |
|---|---|---|
| Age | ||
| 15–19 | Ref | Ref |
| 20–24 | 3.36 *** (.42–4.66) | 3.81 *** (2.62–5.54) |
| Wealth Quintile | ||
| Poorest | Ref | Ref |
| Poorer | 1.28 (0.77–1.81) | 0.84 (0.50–1.41) |
| Middle | 1.71 ** (1.17–2.48) | 0.92 (0.53–1.59) |
| Richer | 2.40 *** (1.64–3.52) | 10.2 (0.56–1.86) |
| Richest | 1.70 * (1.08–2.65) | 0.67 (0.33–1.36) |
| Occupation | ||
| Not working | Ref | Ref |
| Working | 1.99 *** (1.54–2.59) | 1.60 *** (1.19–2.14) |
| Educational level | ||
| No education | Ref | Ref |
| Primary | 1.41 (0.90–2.19) | 1.39 (0.82–2.34) |
| Secondary | 1.09 (0.74–1.62) | 0.98 (0.59–1.64) |
| Higher | 1.23 (0.59–2.59) | 1.07 (0.44–2.60) |
| Religion | ||
| Christian | Ref | Ref |
| Islam | 0.69 * (0.48–0.97) | 0.71 (0.47–1.07) |
| Traditional/no religion | 1.11 (0.62–2.01) | 1.07 (0.57–2.01) |
| Marital Status | ||
| Never Married | Ref | Ref |
| Married | 1.42 * (1.04–1.93) | 1.92 ** (1.26–2.93) |
| Cohabiting | 2.07 *** (1.51–2.84) | 2.18 *** (1.43–3.31) |
| Divorced/Separated | 2.53 ** (1.44–4.41) | 2.17 * (1.13–4.18) |
| Age at first sex | ||
| Less than 15 years | Ref | Ref |
| 15–19 years | 0.98 (0.69–1.39) | 0.69 (0.46–1.04) |
| 20–24 years | 0.45 * (0.24–0.84) | 0.19 *** (0.10–0.39) |
| At first union | 0.95 (0.61–1.50) | 0.57 * (0.34–0.96) |
| Parity | ||
| No births | Ref | Ref |
| One birth | 1.15 (0.86–1.53) | 0.64 * (0.44–0.91) |
| Two births | 1.46 * (1.02–2.08) | 0.51 ** (0.32–0.81) |
| Three or more births | 1.38 (0.83–2.30) | 0.39 ** (0.21–0.75) |
| Exposure to media | ||
| No | Ref | Ref |
| Yes | 1.41 (0.87–2.31) | 1.06 (0.60–1.90) |
| Place of Residence | ||
| Urban | Ref | Ref |
| Rural | 0.68 ** (0.53–0.86) | 0.65 * (0.46–0.92) |
| Region | ||
| Western | Ref | Ref |
| Central | 0.38 ** (0.22–0.67) | 0.35 ** (0.20–0.62) |
| Greater Accra | 0.92 (0.57–1.46) | 0.71 (0.43–1.18) |
| Volta | 0.66 (0.39–1.11) | 0.67 (0.39–1.16) |
| Eastern | 0.82 (0.52–1.28) | 0.75 (0.46–1.21) |
| Ashanti | 1.19 (0.76–1.84) | 1.09 (0.69–1.73) |
| Brong Ahafo | 0.61 * (0.39–0.96) | 0.61 (0.37–1.01) |
| Northern | 0.35 *** (0.20–0.63) | 0.29 *** (0.14–0.59) |
| Upper East | 0.20 *** (0.10–0.40) | 0.18 *** (0.08–0.39) |
| Upper West | 0.52 * (0.29–0.92) | 0.48 * (0.24–0.95) |
| pseudo R2 | 0.114 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahinkorah, B.O.; Seidu, A.-A.; Hagan, J.E., Jr.; Archer, A.G.; Budu, E.; Adoboi, F.; Schack, T. Predictors of Pregnancy Termination among Young Women in Ghana: Empirical Evidence from the 2014 Demographic and Health Survey Data. Healthcare 2021, 9, 705. https://doi.org/10.3390/healthcare9060705
Ahinkorah BO, Seidu A-A, Hagan JE Jr., Archer AG, Budu E, Adoboi F, Schack T. Predictors of Pregnancy Termination among Young Women in Ghana: Empirical Evidence from the 2014 Demographic and Health Survey Data. Healthcare. 2021; 9(6):705. https://doi.org/10.3390/healthcare9060705
Chicago/Turabian StyleAhinkorah, Bright Opoku, Abdul-Aziz Seidu, John Elvis Hagan, Jr., Anita Gracious Archer, Eugene Budu, Faustina Adoboi, and Thomas Schack. 2021. "Predictors of Pregnancy Termination among Young Women in Ghana: Empirical Evidence from the 2014 Demographic and Health Survey Data" Healthcare 9, no. 6: 705. https://doi.org/10.3390/healthcare9060705
APA StyleAhinkorah, B. O., Seidu, A.-A., Hagan, J. E., Jr., Archer, A. G., Budu, E., Adoboi, F., & Schack, T. (2021). Predictors of Pregnancy Termination among Young Women in Ghana: Empirical Evidence from the 2014 Demographic and Health Survey Data. Healthcare, 9(6), 705. https://doi.org/10.3390/healthcare9060705







