Patients Taking Direct Oral Anticoagulants (DOAC) Undergoing Oral Surgery: A Review of the Literature and a Proposal of a Peri-Operative Management Protocol
Abstract
1. Introduction
2. Methods
2.1. What are DOACs?
2.2. How do DOACs Work?
2.3. What are DOACs Used For?
2.4. Half-Life, Daily Administration, and Posology
2.5. Which Tests are Used for Measuring Anticoagulation?
2.6. Drug Interactions
2.7. Antidots
2.8. Elimination Pathway
2.9. Renal Function
2.10. Hepatic Impairment
2.11. Elderly Patients
2.12. Local Risk Factors
2.13. Continuing or Discontinuing the Treatment?
2.14. Reintroduction of the Discontinued Medication
2.15. Peri-Operative Diet
2.16. Surgical Planning
2.17. Local Anesthesia
2.18. Improvement of Coagulation
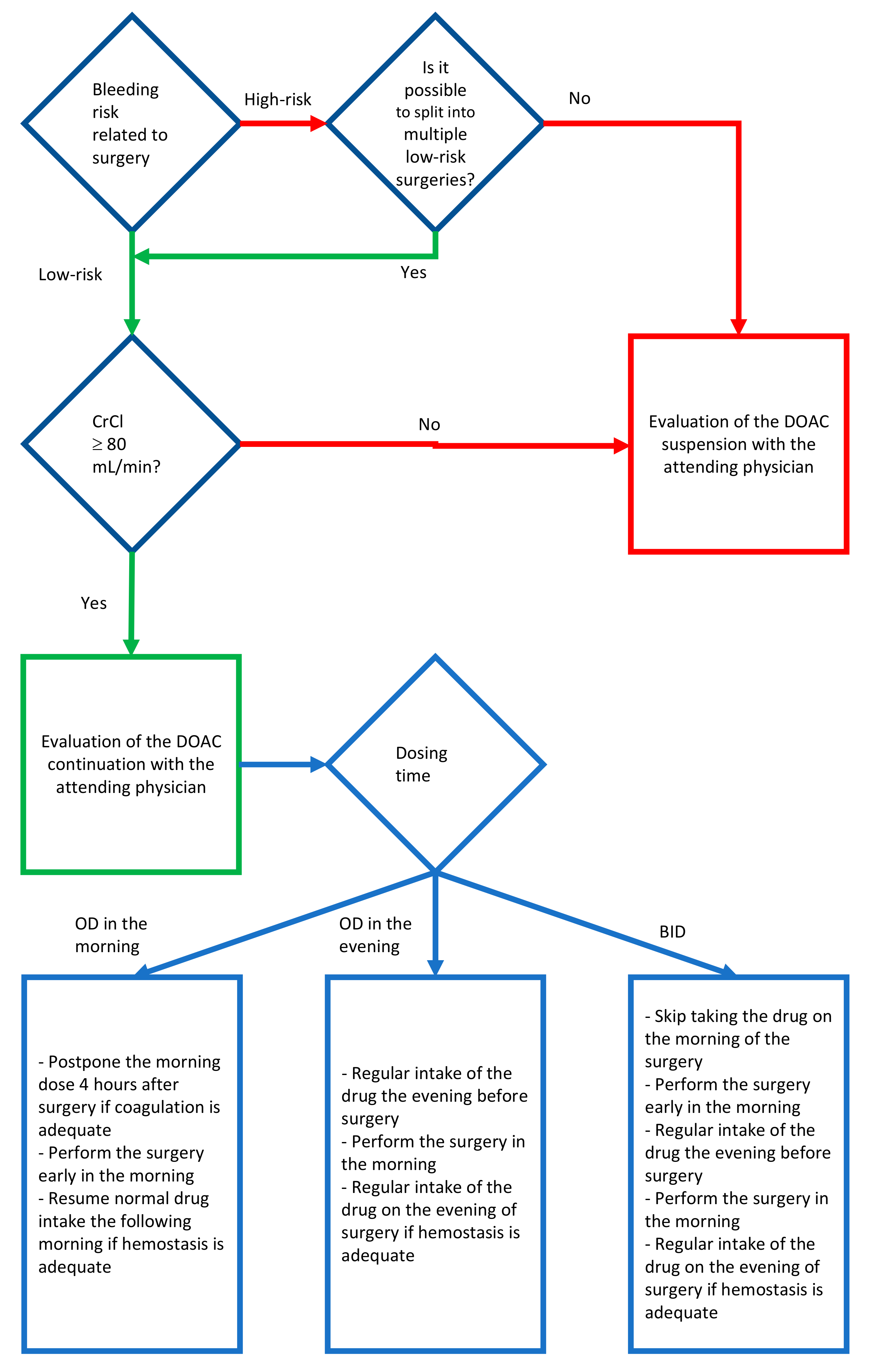
2.19. Guideline Proposal for the Management of the DOAC Assuming Patient Undergoing Oral Surgery
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hirsh, J.; Dalen, J.E.; Anderson, D.R.; Poller, L.; Bussey, H.; Ansell, J.; Deykin, D. Oral anticoagulants: Mechanism of action, clinical effectiveness, and optimal therapeutic range. Chest 2001, 119, 8S–21S. [Google Scholar] [CrossRef] [PubMed]
- Loo, S.Y.; Dell’Aniello, S.; Huiart, L.; Renoux, C. Trends in the prescription of novel oral anticoagulants in UK primary care. Br. J. Clin. Pharmacol. 2017, 83, 2096–2106. [Google Scholar] [CrossRef] [PubMed]
- Miranda, M.; Martinez, L.; Franco, R.; Forte, V.; Barlattani, A.; Bollero, P. Differences between warfarin and new oral anticoagulants in dental clinical practice. Oral Implant. 2016, 9, 151–156. [Google Scholar]
- Miclotte, I.; Vanhaverbeke, M.; Agbaje, J.O.; Legrand, P.; Vanassche, T.; Verhamme, P.; Politis, C. Pragmatic approach to manage new oral anticoagulants in patients undergoing dental extractions: A prospective case-control study. Clin. Oral Investig. 2016, 21, 2183–2188. [Google Scholar] [CrossRef]
- Precht, C.; Demirel, Y.; Assaf, A.T.; Pinnschmidt, H.O.; Knipfer, C.; Hanken, H.; Friedrich, R.E.; Wikner, J. Perioperative management in patients with undergoing direct oral anticoagulant therapy in oral surgery—A multicentric questionnaire survey. Vivo 2019, 33, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Lababidi, E.; Breik, O.; Savage, J.; Engelbrecht, H.; Kumar, R.; Crossley, C. Assessing an oral surgery specific protocol for patients on direct oral anticoagulants: A retrospective controlled cohort study. Int. J. Oral Maxillofac. Surg. 2018, 47, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.P.; Woolcombe, S.A.; Patel, R.K.; Obisesan, O.; Roberts, L.N.; Bryant, C.; Arya, R. Managing direct oral anticoagulants in patients undergoing dentoalveolar surgery. Br. Dent. J. 2017, 222, 245–249. [Google Scholar] [CrossRef]
- Cabbar, F.; Cabbar, A.T.; Coşansu, K.; Cekirdekci, E.I. Effects of direct oral anticoagulants on quality of life during periprocedural management for dental extractions. J. Oral Maxillofac. Surg. 2019, 77, 904–911. [Google Scholar] [CrossRef]
- Stangier, J.; Rathgen, K.; Stähle, H.; Gansser, D.; Roth, W. The pharmacokinetics, pharmacodynamics and tolerability of dabigatran etexilate, a new oral direct thrombin inhibitor, in healthy male subjects. Br. J. Clin. Pharmacol. 2007, 64, 292–303. [Google Scholar] [CrossRef]
- Gross, P.L.; Weitz, J.I. New anticoagulants for treatment of venous thromboembolism. Arter. Thromb. Vasc. Biol. 2008, 28, 380–386. [Google Scholar] [CrossRef]
- Poulakos, M.N.; Walker, J.N.; Baig, U.; David, T. Edoxaban: A direct oral anticoagulant. Am. J. Health Pharm. 2017, 74, 117–129. [Google Scholar] [CrossRef] [PubMed]
- Stangier, J.; Clemens, A. Pharmacology, pharmacokinetics, and pharmacodynamics of dabigatran etexilate, an oral direct thrombin inhibitor. Clin. Appl. Thromb. 2009, 15, 9S–16S. [Google Scholar] [CrossRef] [PubMed]
- Hauel, N.H.; Nar, H.; Priepke, H.; Ries, U.; Stassen, J.M.; Wienen, W. Structure-based design of novel potent nonpeptide thrombin inhibitors. J. Med. Chem. 2002, 45, 1757–1766. [Google Scholar] [CrossRef] [PubMed]
- Stassen, J.-M.; Priepke, H.; Ries, U.J.; Hauel, N.; Wienen, W. In-vitro profile and ex-vivo anticoagulant activity of the direct thrombin inhibitor dabigatran and its orally active prodrug, dabigatran etexilate. Thromb. Haemost. 2007, 98, 155–162. [Google Scholar] [CrossRef]
- Firriolo, F.J.; Hupp, W.S. Beyond warfarin: The new generation of oral anticoagulants and their implications for the management of dental patients. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2012, 113, 431–441. [Google Scholar] [CrossRef]
- Food and Drug Administration Approved Drug Label. Available online: http://www.accessdata.fda.gov/drugsatfda_docs/label/2019/202155s021lbl.pdf (accessed on 10 June 2019).
- Food and Drug Administration Approved Drug Label. Available online: http://www.accessdata.fda.gov/drugsatfda_docs/label/2019/202439s029lbl.pdf (accessed on 10 June 2019).
- Food and Drug Administration Approved Drug Label. Available online: http://www.accessdata.fda.gov/drugsatfda_docs/label/2018/022512s035lbl.pdf (accessed on 19 June 2010).
- Food and Drug Administration Approved Drug Label. Available online: http://www.accessdata.fda.gov/drugsatfda_docs/label/2017/206316s012lbl.pdf (accessed on 19 June 2010).
- Hanken, H.; Gröbe, A.; Heiland, M.; Smeets, R.; Kluwe, L.; Wikner, J.; Koehnke, R.; Al-Dam, A.; Eichhorn, W. Postoperative bleeding risk for oral surgery under continued rivaroxaban anticoagulant therapy. Clin. Oral Investig. 2015, 20, 1279–1282. [Google Scholar] [CrossRef]
- Eriksson, B.I.; Dahl, O.E.; Rosencher, N.; Kurth, A.A.; Van Dijk, C.N.; Frostick, S.P.; Prins, M.H.; Hettiarachchi, R.; Hantel, S.; Schnee, J.; et al. Dabigatran etexilate versus enoxaparin for prevention of venous thromboembolism after total hip replacement: A randomised, double-blind, non-inferiority trial. Lancet 2007, 370, 949–956. [Google Scholar] [CrossRef]
- Eriksson, B.O.; Dahl, O.E.; Rosencher, N.; Kurth, A.A.; Van Dijk, C.N.; Frostick, S.P.; Kälebo, P.; Christiansen, A.V.; Hantel, S.; Hettiarachchi, R.; et al. subcutaneous enoxaparin for the prevention of venous thromboembolism after total knee replacement: The Re-model randomized trial. J. Thromb. Haemost. 2007, 5, 2178–2185. [Google Scholar] [CrossRef]
- Eriksson, B.O.; Borris, L.C.; Friedman, R.J.; Haas, S.; Huisman, M.V.; Kakkar, A.K.; Bandel, T.J.; Beckmann, H.; Muehlhofer, E.; Misselwitz, F.; et al. Rivaroxaban versus enoxaparin for thromboprophylaxis after hip arthroplasty. N. Engl. J. Med. 2008, 358, 2765–2775. [Google Scholar] [CrossRef]
- Ginsberg, J.S.; Davidson, B.L.; Comp, P.C.; Francis, C.W.; Friedman, R.J.; Huo, M.H.; Lieberman, J.R.; Muntz, J.E.; Raskob, G.E.; Clements, M.L.; et al. north american enoxaparin regimen for prevention of venous thromboembolism after knee arthroplasty surgery. J. Arthroplast. 2009, 24, 1–9. [Google Scholar]
- Stangier, J.; Stähle, H.; Rathgen, K.; Fuhr, R. Pharmacokinetics and pharmacodynamics of the direct oral thrombin inhibitor dabigatran in healthy elderly subjects. Clin. Pharmacokinet. 2008, 47, 47–59. [Google Scholar] [CrossRef] [PubMed]
- Stangier, J.; Stähle, H.; Rathgen, K.; Roth, W.; Shakeri-Nejad, K. Pharmacokinetics and pharmacodynamics of dabigatran etexilate, an oral direct thrombin inhibitor, are not affected by moderate hepatic impairment. J. Clin. Pharmacol. 2008, 48, 1411–1419. [Google Scholar] [CrossRef] [PubMed]
- Castellone, D.D.; Van Cott, E.M. Laboratory monitoring of new anticoagulants. Am. J. Hematol. 2010, 85, 185–187. [Google Scholar] [CrossRef] [PubMed]
- Heidbuchel, H.; Verhamme, P.; Alings, M.; Antz, M.; Diener, H.C.; Hacke, W.; Oldgren, J.; Sinnaeve, P.; Camm, A.J.; Kirchhof, P.; et al. Updated European Heart Rhythm Association Practical Guide on the use of non-vitamin K antagonist anticoagulants in patients with non-valvular atrial fibrillation. Europace 2015, 17, 1467–1507. [Google Scholar] [CrossRef]
- Mauprivez, C.; Khonsari, R.H.; Razouk, O.; Goudot, P.; Lesclous, P.; Descroix, V.; Lesclous, P. Management of dental extraction in patients undergoing anticoagulant oral direct treatment: A pilot study. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2016, 122, e146–e155. [Google Scholar] [CrossRef]
- Carlsson, S.C.; Mattsson, C.; Eriksson, U.G.; Sarich, T.C.; Wåhlander, K.; Eliasson, Å.; Karlson, B.W.; Sheth, S.B.; Held, P. A review of the effects of the oral direct thrombin inhibitor ximelagatran on coagulation assays. Thromb. Res. 2005, 115, 9–18. [Google Scholar] [CrossRef]
- Yoshikawa, H.; Yoshida, M.; Yasaka, M.; Murasato, Y.; Fukunaga, D.; Shintani, A.; Okada, Y.; Yoshida, H. Safety of tooth extraction in patients receiving direct oral anticoagulant treatment versus warfarin: A prospective observation study. Int. J. Oral Maxillofac. Surg. 2019, 48, 1102–1108. [Google Scholar] [CrossRef]
- Blech, S.; Ebner, T.; Ludwig-Schwellinger, E.; Stangier, J.; Roth, W. The metabolism and disposition of the oral direct thrombin inhibitor, dabigatran, in humans. Drug Metab. Dispos. 2007, 36, 386–399. [Google Scholar] [CrossRef]
- Gnoth, M.J.; Buetehorn, U.; Muenster, U.; Schwarz, T.; Sandmann, S. In vitro and in vivo p-glycoprotein transport characteristics of rivaroxaban. J. Pharmacol. Exp. Ther. 2011, 338, 372–380. [Google Scholar] [CrossRef]
- Mehta, R.S. Novel oral anticoagulants. Part II: Direct thrombin inhibitors. Expert Rev. Hematol. 2010, 3, 351–361. [Google Scholar] [CrossRef]
- Clemm, R.; Neukam, F.W.; Rusche, B.; Bauersachs, A.; Musazada, S.; Schmitt, C. Management of anticoagulated patients in implant therapy: A clinical comparative study. Clin. Oral Implant. Res. 2015, 27, 1274–1282. [Google Scholar] [CrossRef]
- Berton, F.; Costantinides, F.; Rizzo, R.; Franco, A.; Contarin, J.; Stacchi, C.; Maglione, M.; Visintini, E.; Di Lenarda, A.; Di Lenarda, R. Should we fear direct oral anticoagulants more than vitamin K antagonists in simple single tooth extraction? A prospective comparative study. Clin. Oral Investig. 2018, 23, 3183–3192. [Google Scholar] [CrossRef] [PubMed]
- Yagyuu, T.; Kawakami, M.; Ueyama, Y.; Imada, M.; Kurihara, M.; Matsusue, Y.; Imai, Y.; Yamamoto, K.; Kirita, T. Risks of postextraction bleeding after receiving direct oral anticoagulants or warfarin: A retrospective cohort study. BMJ Open 2017, 7, e015952. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, Y.; Yokoe, C.; Imai, Y.; Sugihara, M.; Futatsuki, T. Tooth extraction in patients taking nonvitamin K antagonist oral anticoagulants. J. Dent. Sci. 2016, 11, 59–64. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cocero, N.; Basso, M.; Grosso, S.; Carossa, S. Direct oral anticoagulants and medical comorbidities in patients needing dental extractions: Management of the risk of bleeding. J. Oral Maxillofac. Surg. 2019, 77, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Galliazzo, S.; Donadini, M.; Ageno, W. Antidotes for the direct oral anticoagulants: What news? Thromb. Res. 2018, 164, S119–S123. [Google Scholar] [CrossRef] [PubMed]
- Mühlberg, W.; Platt, D. Age-dependent changes of the kidneys: Pharmacological implications. Gerontology 1999, 45, 243–253. [Google Scholar] [CrossRef]
- Müller, M.; Schlittler, F.; Schaller, B.; Nagler, M.; Exadaktylos, A.K.; Sauter, T.C. Characteristics, treatment and outcome of bleeding after tooth extraction in patients on DOAC and phenprocoumon compared to non-anticoagulated patients—A retrospective study of emergency department consultations. Clin. Oral Investig. 2018, 23, 2273–2278. [Google Scholar] [CrossRef]
- Zeevi, I.; Allon, D.M.; Rosenfeld, E.; Avishai, G.; Gilman, L.; Nissan, J.; Chaushu, G. Four-year cross-sectional study of bleeding risk in dental patients on direct oral anticoagulants. Quintessence Int. 2017, 48, 503–509. [Google Scholar]
- Abayon, M.; Kolokythas, A.; Harrison, S.; Elad, S. Dental management of patients on direct oral anticoagulants: Case series and literature review. Quintessence Int. 2016, 47, 687–696. [Google Scholar]
- Breik, O.; Cheng, A.; Sambrook, P.; Goss, A. Protocol in managing oral surgical patients taking dabigatran. Aust. Dent. J. 2014, 59, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Moreno, G.; Fernández-Cejas, E.; Aguilar-Salvatierra, A.; De Carlos, F.; Ruiz, R.A.D.; Calvo-Guirado, J.L. Dental implant surgery in patients in treatment by dabigatran. Clin. Oral Implant. Res. 2016, 29, 644–648. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Moreno, G.; Aguilar-Salvatierra, A.; Fernández-Cejas, E.; Ruiz, R.A.D.; Marković, A.; Calvo-Guirado, J.L. Dental implant surgery in patients in treatment with the anticoagulant oral rivaroxaban. Clin. Oral Implant. Res. 2015, 27, 730–733. [Google Scholar] [CrossRef] [PubMed]
- Dézsi, C.A.; Dézsi, B.B.; Dézsi, A.D. Management of dental patients receiving antiplatelet therapy or chronic oral anticoagulation: A review of the latest evidence. Eur. J. Gen. Pract. 2017, 23, 197–202. [Google Scholar] [CrossRef]
- Galletti, G.; Alfonsi, F.; Raffaele, A.; Valente, N.A.; Chatelain, S.; Kolerman, R.; Cinquini, C.; Romeggio, S.; Iezzi, G.; Barone, A. Implant placement in patients under treatment with rivaroxaban: A retrospective clinical study. Int. J. Environ. Res. Public Health 2020, 17, 4607. [Google Scholar] [CrossRef]
- Nieto, J.A.; Mora, D.; Bikdeli, B.; Pinar, M.P.; Valle, R.; Pedrajas, J.M.; Mahé, I.; González-Martínez, J.; Díaz-Pedroche, M.D.C.; Monreal, M.; et al. Thirty-day outcomes in patients with proximal deep vein thrombosis who discontinued anticoagulant therapy prematurely. Thromb. Res. 2020, 189, 61–68. [Google Scholar] [CrossRef]
- Jackson, L.R.; Kim, S.; Blanco, R.; Thomas, L.; Ansell, J.; Fonarow, G.C.; Gersh, B.J.; Go, A.S.; Kowey, P.R.; Mahaffey, K.W.; et al. Discontinuation rates of warfarin versus direct acting oral anticoagulants in US clinical practice: Results from outcomes registry for better informed treatment of atrial fibrillation II (ORBIT-AF II). Am. Heart J. 2020, 226, 85–93. [Google Scholar] [CrossRef]
- Wahl, M.J. Dental surgery in anticoagulated patients. Arch. Intern. Med. 1998, 158, 1610–1616. [Google Scholar] [CrossRef]
- Healey, J.S.; Eikelboom, J.W.; Douketis, J.; Wallentin, L.; Oldgren, J.; Yang, S.; Themeles, E.; Heidbuchel, H.; Avezum, A.; Reilly, P.; et al. Periprocedural bleeding and thromboembolic events with dabigatran compared with warfarin. Circulation 2012, 126, 343–348. [Google Scholar] [CrossRef]
- Sherwood, M.W.; Douketis, J.D.; Patel, M.R.; Piccini, J.P.; Hellkamp, A.S.; Lokhnygina, Y.; Spyropoulos, A.C.; Hankey, G.J.; Singer, D.E.; Nessel, C.C.; et al. Outcomes of temporary interruption of rivaroxaban compared with warfarin in patients with nonvalvular atrial fibrillation: Results from the rivaroxaban once daily, oral, direct factor Xa inhibition compared with vitamin K antagonism for prevention of stroke and embolism trial in atrial fibrillation (ROCKET AF). Circulation 2014, 129, 1850–1859. [Google Scholar]
- Miller, S.; Miller, C.S. Direct oral anticoagulants: A retrospective study of bleeding, behavior, and documentation. Oral Dis. 2018, 24, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Heidbuchel, H.; Verhamme, P.; Alings, M.; Antz, M.; Diener, H.C.; Hacke, W.; Oldgren, J.; Sinnaeve, P.R.; Camm, A.J.; Kirchhof, P.; et al. Updated European Heart Rhythm Association Practical Guide on the use of non-vitamin-K antagonist anticoagulants in patients with non-valvular atrial fibrillation: Executive summary. Eur. Heart J. 2017, 38, 2137–2149. [Google Scholar] [PubMed]
- Lip, G.Y.H.; Agnelli, G. Edoxaban: A focused review of its clinical pharmacology. Eur. Heart J. 2014, 35, 1844–1855. [Google Scholar] [CrossRef] [PubMed]
- Stangier, J.; Haertter, S.; Liesenfeld, K.-H.; Wienen, W.; Feuring, M.; Clemens, A.; Van Ryn, J. Dabigatran etexilate—A novel, reversible, oral direct thrombin inhibitor: Interpretation of coagulation assays and reversal of anticoagulant activity. Thromb. Haemost. 2010, 103, 1116–1127. [Google Scholar] [CrossRef] [PubMed]
| Indications | Dabigatran | Rivaroxaban | Apixaban | Edoxaban |
|---|---|---|---|---|
| Prevention | ||||
| Stroke and systemic embolism in patients with NVAF | Yes | Yes | Yes | Yes |
| Recurrence of DVT and PE in patients who have been previously treated | Yes | Yes | ||
| DVT and PE in patients who have undergone hip/knee replacement surgery | Yes | Yes | Yes | |
| Recurrence DVT and/or PE in patients at continued risk for recurrent DVT and/or PE after completion of initial treatment lasting at least 6 months | Yes | |||
| Major cardiovascular events (CV death, MI and stroke) in patients with chronic CAD or PAD | Yes (in combination with aspirin) | |||
| Treatment | ||||
| DVT and PE in patients who have been treated with a parenteral anticoagulant for 5–10 days | Yes | Yes | Yes |
| Drug Feature | Dabigatran | Rivaroxaban | Apixaban | Edoxaban |
|---|---|---|---|---|
| Prodrug | Yes | No | No | No |
| Target | Thrombin | FXa | FXa | FXa |
| Absolute bioavailability | 3–7% | 66% (Almost 100% with food) | >50% | 61.8% |
| Time to maximum plasma concentration | 1.5–4 h | 2.5–4 h | 2 h | 1–3 h |
| Daily administration | BID | OD | BID | OD |
| Renal clearance | 80–85% | 35% | 25–27% | 35–50% |
| Plasma protein binding | 35% | 90% | 87% | 40–59% |
| Test | ECT, TT | FXa inhibition assay | FXa inhibition assay, dilute prothrombin time | anti-factor Xa activity |
| Drug | Via | Dabigatran | Rivaroxaban | Apixaban | Edoxaban |
|---|---|---|---|---|---|
| Antacids (H2B; PPI; Al-Mg-hydroxide) | GI absorption | −12/30% PDC (no clinical effect) | No effect | No effect | No effect |
| Itraconazole Ketoconazole Posaconazole Voriconazole | potent P-gp and BCRP competition; CYP3A4 inhibition | +140/150% PDC CONTRA-INDICATED | Up to +160% PDC CONTRA-INDICATED | +100% PDC CONTRA-INDICATED | +87–95% PDC Reduce DOAC dose |
| Fluconazole | Moderate CYP3A4 inhibition | No data | +42% PDC Consider DOAC dose reduction | No data | No data |
| Clarithromycin Erythromycin | moderate P-gp competition and CYP3A4 inhibition | +15–20% PDC Consider DOAC dose reduction | +30–54% PDC Consider DOAC dose reduction | No data | +90% PDC Reduce DOAC dose |
| Rifampicin | P-gp/ BCRP and CYP3A4 inducer | −66% PDC CONTRA-INDICATED | Up to −50% PDC CONTRA-INDICATED | −54% PDC CONTRA-INDICATED | −35% PDC, but with compensatory Increase of Active Metabolites AVOID IF POSSIBLE |
| Non-COX-selective NSAIDs (Naproxen Ibuprofen, Ketoprofen, Fenoprofen, Flurbiprofen) and salicylates | P-gp competition and pharmaco-dynamic interaction | GI and peptic ulceration and/or perforation, +60% BR AVOID IF POSSIBLE | |||
| NSAIDs | Pharmacodynamic Interaction | Increased BR AVOID IF POSSIBLE | Increased BR AVOID IF POSSIBLE | Increased BR AVOID IF POSSIBLE | Increased BR AVOID IF POSSIBLE |
| Opioid | Increased BR AVOID IF POSSIBLE | ||||
| Renal Function (CRCL, mL/min) | Dabigatran | Rivaroxaban | Apixaban | Edoxaban |
|---|---|---|---|---|
| >80 | 12–17 h | 5–9 h (young) 11–13 h (elderly) | 9–12 h | 10–14 h |
| >50 to ≤80 | 17 h | 8.7 h | 14.6 h | 8.6 h |
| >30 to ≤50 | 19 h | 9 h | 17.6 h | 9.4 h |
| >15 to ≤30 | 28 h | 9.5 h | 17.3 h | 16.9 h |
| ≤15 | No data | |||
| Surgical | Metabolic |
|---|---|
| Extraction of one to three single-rooted teeth Extraction of one multi-rooted teeth Positioning of one to three implants in the anterior region Positioning of one to two implants in the posterior region Periodontal surgery Incision of abscess Flap limited to the attached gingiva and not involving the free gingiva | Good renal function |
| Renal Function (CLCR mL/min) | Dabigatran | Rivaroxaban–Apixaban–Edoxaban | ||
|---|---|---|---|---|
| Low-Risk Surgery | High-Risk Surgery | Low-Risk Surgery | High-Risk Surgery | |
| ≥80 | 24 h | 48 h | 24 h | 48 h |
| 50–79 | 36 h | 72 h | 24 h | 48 h |
| 30–49 | 48 h | 96 h | 24 h | 48 h |
| 15–29 | Not indicated | Not indicated | 36 h | 48 h |
| <15 | No official indication for use | |||
| Study | N of Procedures | Bleeding (%) | Degree | Dabigatran | Rivaroxaban | Apixaban | Edoxaban |
|---|---|---|---|---|---|---|---|
| Miranda et al. 2016 [3] | 12 | 0 | N.A. | N.A. | N.A. | N.A. | |
| Clemm et al. 2016 [35] | 16 | 0 | 0/6 | 0/8 | 0/2 | ||
| Hanken et al. 2016 [20] | 52 | 11.5 | Two local compressions, four reoperations | 6/52 | |||
| Mauprivez et al. 2016 [29] | 32 | 17.8 | 85.7% local compression, 14.3% reoperation | 2/9 | 1/21 | 2/2 | |
| Morimoto et al. 2016 [38] | 19 | 31.6 | five local compression, one reoperation | 0/4 | 3/9 | 3/6 | |
| Yagyuu et al. 2017 [37] | 41 | 9.7 | Minor bleeding | N.A. | N.A. | N.A. | N.A. |
| Lababidi et al. 2018 [6] | 38 | 10.5 | Minor bleeding | N.A. | N.A. | N.A. | N.A. |
| Yoshikawa et al. 2019 [31] | 128 | 3.1 | 50% local compression, 50% reoperation | 1/37 | 0/34 | 2/39 | 1/18 |
| Berton et al. 2019 [36] | 65 | 18.5 | Local compression | 1/11 | 3/28 | 1/22 | 0/4 |
| Accurate Suturing Technique |
|---|
| Irrigation of the alveoli by antifibrinolytic agents such us tranexamic acid together with gelatin sponges |
| Collagen and resorbable oxycellulose applied into the extraction socket |
| Acrylic splint for wound protection |
| Application of fibrin glue and secondary sutures |
| Accurate alveolar bone cleaning to remove bleeding granulation tissue |
| Post-operative wound compression with gauze soaked in tranexamic acid |
| Ice pack |
| Mouth rinses with a 10 mL of 5% tranexamic acid aqueous solution for 2 min, repeated four times daily for seven days |
| Cold food and avoid mouthwashes |
| Avoid vertical releasing incisions if possible |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lupi, S.M.; Rodriguez y Baena, A. Patients Taking Direct Oral Anticoagulants (DOAC) Undergoing Oral Surgery: A Review of the Literature and a Proposal of a Peri-Operative Management Protocol. Healthcare 2020, 8, 281. https://doi.org/10.3390/healthcare8030281
Lupi SM, Rodriguez y Baena A. Patients Taking Direct Oral Anticoagulants (DOAC) Undergoing Oral Surgery: A Review of the Literature and a Proposal of a Peri-Operative Management Protocol. Healthcare. 2020; 8(3):281. https://doi.org/10.3390/healthcare8030281
Chicago/Turabian StyleLupi, Saturnino Marco, and Arianna Rodriguez y Baena. 2020. "Patients Taking Direct Oral Anticoagulants (DOAC) Undergoing Oral Surgery: A Review of the Literature and a Proposal of a Peri-Operative Management Protocol" Healthcare 8, no. 3: 281. https://doi.org/10.3390/healthcare8030281
APA StyleLupi, S. M., & Rodriguez y Baena, A. (2020). Patients Taking Direct Oral Anticoagulants (DOAC) Undergoing Oral Surgery: A Review of the Literature and a Proposal of a Peri-Operative Management Protocol. Healthcare, 8(3), 281. https://doi.org/10.3390/healthcare8030281






