Effect of Facial Skin Temperature on the Perception of Anxiety: A Pilot Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Data Collection and Instruments
2.3. Data Analysis
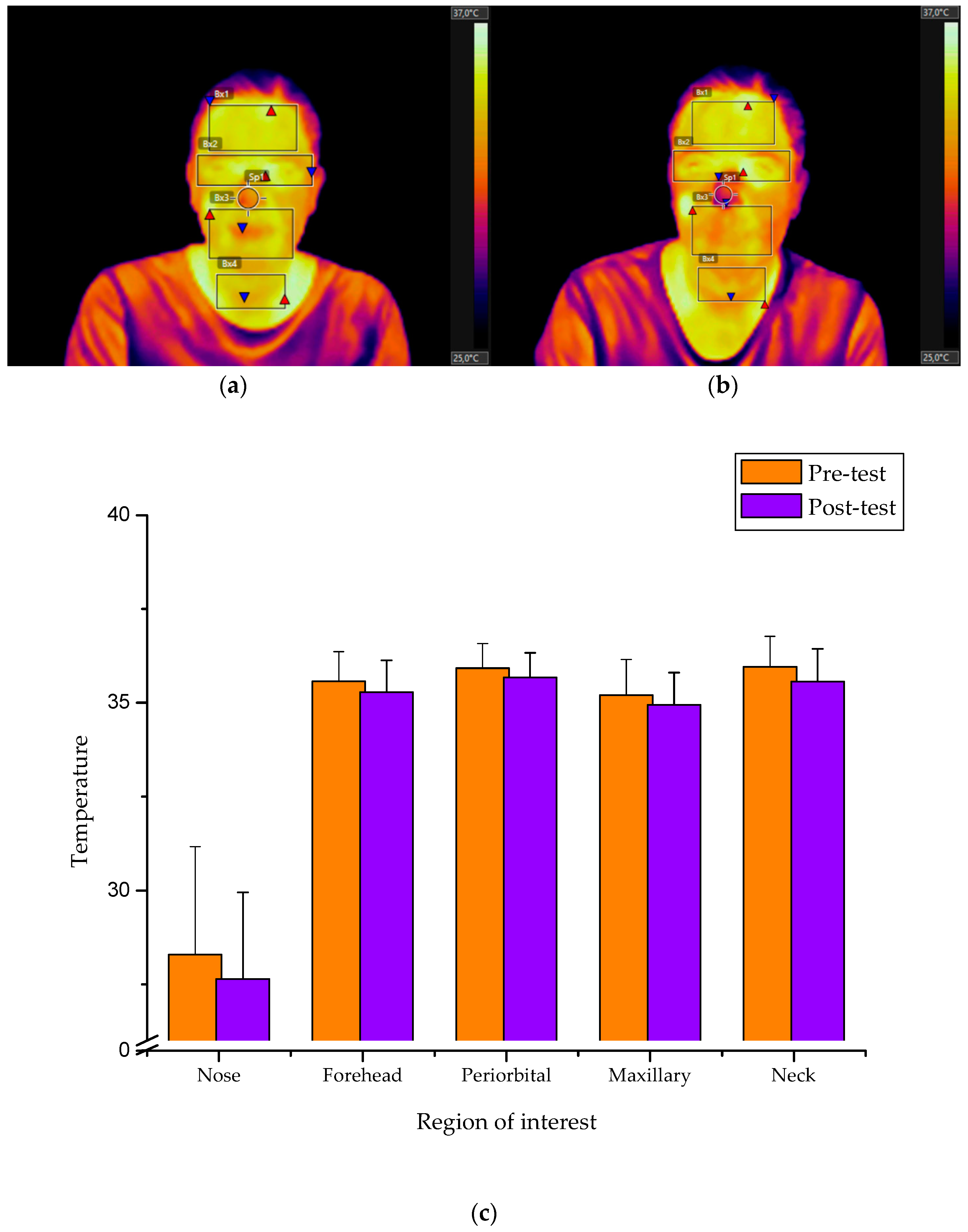
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gamble, A.S. Simulation in undergraduate paediatric nursing curriculum: Evaluation of a complex ‘ward for a day’ education program. Nurse Educ. Pract. 2017, 23, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Park, J.-H.; Shin, S. Effectiveness of simulation-based nursing education depending on fidelity: A meta-analysis. BMC Med. Educ. 2016, 16, 152. [Google Scholar] [CrossRef] [PubMed]
- Cabrera-Mino, C.; Shinnick, M.A.; Moye, S. Task-Evoked Pupillary Responses in Nursing Simulation as an Indicator of Stress and Cognitive Load. Clin. Simul. Nurs. 2019, 31, 21–27. [Google Scholar] [CrossRef]
- Bhanji, F.; Donoghue, A.J.; Wolff, M.S.; Flores, G.E.; Halamek, L.P.; Berman, J.M.; Sinz, E.H.; Cheng, A. Part 14: Education: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2015, 132, S561–S573. [Google Scholar] [CrossRef] [PubMed]
- Judd, B.K.; Currie, J.; Dodds, K.L.; Fethney, J.; Gordon, C.J. Registered nurses psychophysiological stress and confidence during high-fidelity emergency simulation: Effects on performance. Nurse Educ. Today 2019, 78, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Plemmons, C.; Clark, M.; Feng, D. Comparing student clinical self-efficacy and team process outcomes for a DEU, blended, and traditional clinical setting: A quasi-experimental research study. Nurse Educ. Today 2018, 62, 107–111. [Google Scholar] [CrossRef]
- Davey, P.; Whatman, C.; Dicker, B. Comparison of Chest Compressions Metrics Measured Using the Laerdal Skill Reporter and Q-CPR: A Simulation Study. Simul. Healthc. 2015, 10, 257–262. [Google Scholar] [CrossRef]
- Beltrán-Velasco, A.I.; Ruisoto-Palomera, P.; Bellido-Esteban, A.; García-Mateos, M.; Clemente-Suárez, V.J. Analysis of Psychophysiological Stress Response in Higher Education Students Undergoing Clinical Practice Evaluation. J. Med. Syst. 2019, 43, 68. [Google Scholar] [CrossRef]
- Ayuso-Murillo, D.; Colomer-Sánchez, A.; Santiago-Magdalena, C.R.; Lendínez-Mesa, A.; Gracia, E.B.D.; López-Peláez, A.; Herrera-Peco, I. Effect of Anxiety on Empathy: An Observational Study Among Nurses. Healthcare 2020, 8, 140. [Google Scholar] [CrossRef]
- Brugnera, A.; Zarbo, C.; Adorni, R.; Tasca, G.A.; Rabboni, M.; Bondi, E.; Compare, A.; Sakatani, K. Cortical and cardiovascular responses to acute stressors and their relations with psychological distress. Int. J. Psychophysiol. 2017, 114, 38–46. [Google Scholar] [CrossRef]
- Judd, B.K.; Alison, J.A.; Waters, D.; Gordon, C.J. Comparison of Psychophysiological Stress in Physiotherapy Students Undertaking Simulation and Hospital-Based Clinical Education. Simul. Healthc. 2016, 11, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Mills, B.; Carter, O.; Rudd, C.; Claxton, L.; O’Brien, R. An experimental investigation into the extent social evaluation anxiety impairs performance in simulation-based learning environments amongst final-year undergraduate nursing students. Nurse Educ. Today 2016, 45, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Rudland, J.R.; Golding, C.; Wilkinson, T.J. The stress paradox: How stress can be good for learning. Med. Educ. 2020, 54, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Wearne, T.A.; Lucien, A.; Trimmer, E.M.; Logan, J.A.; Rushby, J.; Wilson, E.; Filipčíková, M.; McDonald, S. Anxiety sensitivity moderates the subjective experience but not the physiological response to psychosocial stress. Int. J. Psychophysiol. 2019, 141, 76–83. [Google Scholar] [CrossRef]
- Tempesta, D.; Mazza, M.; Serroni, N.; Moschetta, F.S.; Di Giannantonio, M.; Ferrara, M.; De Berardis, D. Neuropsychological functioning in young subjects with generalized anxiety disorder with and without pharmacotherapy. Prog. Neuropsychopharmacol. Biol. Psychiatry 2013, 45, 236–241. [Google Scholar] [CrossRef]
- Bjørshol, C.A.; Myklebust, H.; Nilsen, K.L.; Hoff, T.; Bjørkli, C.; Illguth, E.; Søreide, E.; Sunde, K. Effect of socioemotional stress on the quality of cardiopulmonary resuscitation during advanced life support in a randomized manikin study. Crit. Care Med. 2011, 39, 300–304. [Google Scholar] [CrossRef]
- Fernández-Ayuso, D.; Fernández-Ayuso, R.; Del-Campo-Cazallas, C.; Pérez-Olmo, J.L.; Matías-Pompa, B.; Fernández-Carnero, J.; Calvo-Lobo, C. The Modification of Vital Signs According to Nursing Students’ Experiences Undergoing Cardiopulmonary Resuscitation Training via High-Fidelity Simulation: Quasi-Experimental Study. JMIR Serious Games 2018, 6, e11061. [Google Scholar] [CrossRef]
- Shinnick, M.A. Validating Eye Tracking as an Objective Assessment Tool in Simulation. Clin. Simul. Nurs. 2016, 12, 438–446. [Google Scholar] [CrossRef]
- Cruz-Albarran, I.A.; Benitez-Rangel, J.P.; Osornio-Rios, R.A.; Morales-Hernandez, L.A. Human emotions detection based on a smart-thermal system of thermographic images. Infrared Phys. Technol. 2017, 81, 250–261. [Google Scholar] [CrossRef]
- Mauriz, E.; Ferradal-Villa, P.; Caloca-Amber, S.; Sánchez-Valdeón, L.; Vázquez-Casares, A.M. Effectiveness of infrared thermography in monitoring ventilation performance during cardiopulmonary resuscitation training: A cross-sectional simulation study in nursing students. In Proceedings of the Seventh International Conference on Technological Ecosystems for Enhancing Multiculturality, Salamanca, Spain, 24–26 October 2018; pp. 361–367. [Google Scholar]
- Ghahramani, A.; Castro, G.; Becerik-Gerber, B.; Yu, X. Infrared thermography of human face for monitoring thermoregulation performance and estimating personal thermal comfort. Build. Environ. 2016, 109, 1–11. [Google Scholar] [CrossRef]
- Haddad, D.S.; Brioschi, M.L.; Baladi, M.G.; Arita, E.S. A new evaluation of heat distribution on facial skin surface by infrared thermography. Dentomaxillofacial Radiol. 2016, 45, 20150264. [Google Scholar] [CrossRef] [PubMed]
- Ioannou, S.; Gallese, V.; Merla, A. Thermal infrared imaging in psychophysiology: Potentialities and limits: Thermal infrared imaging in psychophysiology. Psychophysiology 2014, 51, 951–963. [Google Scholar] [CrossRef] [PubMed]
- Koprowski, R.; Wilczyński, S.; Martowska, K.; Gołuch, D.; Wrocławska-Warchala, E. Dedicated tool to assess the impact of a rhetorical task on human body temperature. Int. J. Psychophysiol. 2017, 120, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Moliné, A.; Gálvez-García, G.; Fernández-Gómez, J.; De la Fuente, J.; Iborra, O.; Tornay, F.; Mata Martín, J.L.; Puertollano, M.; Gómez Milán, E. The Pinocchio effect and the Cold Stress Test: Lies and thermography. Psychophysiology 2017, 54, 1621–1631. [Google Scholar] [CrossRef] [PubMed]
- Spielberger, C.D.; Gorsuch, R.L.; Lushene, R.E.; Vagg, P.R.; Jacobs, G.A. Manual for the State-Trait Anxiety Inventory; Consulting Psychologists Press: Palo Alto, CA, USA, 1983. [Google Scholar]
- Ludwig, N.; Formenti, D.; Rossi, A.; Trecroci, A.; Gargano, M.; Alberti, G. Assessing Facial Skin Temperature Asymmetry with Different Methods. In Proceedings of the 2016 International Conference on Quantitative InfraRed Thermography, QIRT Council, Gdansk, Poland, 4–8 July 2016. [Google Scholar]
- Singh, J.; Arora, A.S. An automated approach to enhance the thermographic evaluation on orofacial regions in lateral facial thermograms. J. Therm. Biol. 2018, 71, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Moliné, A.; Dominguez, E.; Salazar-López, E.; Gálvez-García, G.; Fernández-Gómez, J.; De la Fuente, J.; Iborra, O.; Tornay, F.J.; Gómez Milán, E. The mental nose and the Pinocchio effect: Thermography, planning, anxiety, and lies. J. Investig. Psychol. Offender Profiling 2018, 15, 234–248. [Google Scholar] [CrossRef]
- Chernorizov, A.M.; Isaychev, S.A.; Zinchenko, Y.P.; Galatenko, V.V.; Znamenskaya, I.A.; Zakharov, P.N.; Khakhalin, A.V.; Gradoboeva, O.N. Psychophysiological methods for the diagnostics of human functional states: New approaches and perspectives. Psychol. Russ. State Art 2016, 9, 23–36. [Google Scholar] [CrossRef]
- Cardone, D.; Merla, A. New Frontiers for Applications of Thermal Infrared Imaging Devices: Computational Psychopshysiology in the Neurosciences. Sensors 2017, 17, 1042. [Google Scholar] [CrossRef]
- Khan, M.M.; Ward, R.D.; Ingleby, M. Classifying pretended and evoked facial expressions of positive and negative affective states using infrared measurement of skin temperature. ACM Trans. Appl. Percept. 2009, 6, 1–22. [Google Scholar] [CrossRef]
- Salazar-López, E.; Domínguez, E.; Juárez Ramos, V.; de la Fuente, J.; Meins, A.; Iborra, O.; Gálvez, G.; Rodríguez-Artacho, M.A.; Gómez-Milán, E. The mental and subjective skin: Emotion, empathy, feelings and thermography. Conscious. Cogn. 2015, 34, 149–162. [Google Scholar] [CrossRef]
- Clay-Warner, J.; Robinson, D.T. Infrared Thermography as a Measure of Emotion Response. Emot. Rev. 2015, 7, 157–162. [Google Scholar] [CrossRef]
- Monsieurs, K.G.; Nolan, J.P.; Bossaert, L.L.; Greif, R.; Maconochie, I.K.; Nikolaou, N.I.; Perkins, G.D.; Soar, J.; Truhlář, A.; Wyllie, J.; et al. European Resuscitation Council Guidelines for Resuscitation 2015. Resuscitation 2015, 95, 1–80. [Google Scholar] [CrossRef] [PubMed]
- Neumar, R.W.; Shuster, M.; Callaway, C.W.; Gent, L.M.; Atkins, D.L.; Bhanji, F.; Brooks, S.C.; de Caen, A.R.; Donnino, M.W.; Ferrer, J.M.E.; et al. 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2015, 132, S315–S367. [Google Scholar] [CrossRef] [PubMed]
- Seixas, A.; Ammer, K.; Carvalho, R.; Vilas-Boas, J.P.; Mendes, J.; Vardasca, R. Relationship between skin temperature and soft tissue hardness in diabetic patients: An exploratory study. Physiol. Meas. 2019, 40, 074007. [Google Scholar] [CrossRef]
| Sociodemographic Characteristics | Undergraduate Bachelor Students (BS) | Postgraduate Master Students (MS) | Statistic Values χ2/t | p Value | ||
|---|---|---|---|---|---|---|
| N = 21 mean ± SD (%) | N = 19 mean ± SD (%) | |||||
| Sex | Female | 18 (85.7) | 16 (84.2) | 0.018 b | 0.894 b | |
| Male | 3 (14.3) | 3 (15.8) | ||||
| Age | 21.0 (4) | 23.85 (1.61) | −2.890 a | 0.006 *,a | ||
| Educational level | Baccalaureate | 17 (81) | 0 | 0.000 *,b | ||
| Professional training | 3 (14.3) | 0 | 36.190 b | |||
| Other Bachelor of Science | 1 (4.8) | 19 (100) | ||||
| Practicum in special health services | Yes | 0 | 19 (100) | 0.000 *,b | ||
| No | 21 (100) | 0 | 40.0 b | |||
| Number of special health services in practicum | 0 | 1.84 (0.83) | −9.625 a | 0.000 a | ||
| Work in special health services | Yes | 0 | 10 (52.6) | 14.737 b | 0.000 *,b | |
| No | 21 (100) | 9 (47.4) | ||||
| Number of special health services working | 0 | 0 | 0.84 (1.05) | −3.618 a | 0.002 *,a | |
| Training on basic CPR (basic life support) | Yes | Last two years | 1 (4.8) | 6 (31.6) | 6.686 b | 0.010 *,b |
| More than two years | 2 (9.5) | 2 (10.5) | ||||
| No | 18 (85.7) | 9 (47.4) | ||||
| Duration basic CPR training | 37.67 (46.11) | 56.33 (37.67) | 6.750 b | 0.455 a | ||
| Training on advanced CPR (advanced life support) | Yes | 0 | 4 (21.1) | 4.912 b | 0.027 *,b | |
| No | 21 (100) | 15 (78.9) | ||||
| Facial Region | Temperature Value | Moment | Temperature Mean (SD) | Undergraduate Bachelor Students (BS) | Temperature Mean (SD) | Postgraduate Master Students (MS) | t | p Value Groups | ||
|---|---|---|---|---|---|---|---|---|---|---|
| t-Paired | Significance | t-Paired | Significance | |||||||
| Nose | Average | Pre-test | 27.87 (2.56) | −1.014 | 0.323 | 28.76 (3.20) | 4.095 | 0.001 ** | −0.972 | 0.337 |
| Post-test | 28.17 (2.31) | 27.06 (2.21) | −0.961 | 0.129 | ||||||
| Difference | 0.30 (1.36) | - | −1.70 (1.81) | - | 1.553 | 0.000 * | ||||
| Forehead | Maximum | Pre-test | 35.79 (0.76) | 0.982 | 0.338 | 35.79 (0.76) | 2.362 | 0.030 * | 1.557 | 0.072 |
| Post-test | 35.59 (0.89) | - | 34.95 (0.66) | 3.980 | 0.014 * | |||||
| Difference | −0.20 (0.91) | - | −0.39 (0.72) | - | 3.923 | 0.462 | ||||
| Average | Pre-test | 34.90 (0.77) | 1.291 | 0.211 | 34.13 (1.34) | 2.939 | 0.009 ** | 1.851 | 0.031 * | |
| Post-test | 34.60 (1.10) | 33.52 (1.26) | 1.849 | 0.006 * | ||||||
| Difference | −0.30 (−0.30) | −0.61 (0.91) | 2.578 | 0.317 | ||||||
| Minimum | Pre-test | 32.62 (1.64) | −0.432 | 0.671 | 30.40 (2.75) | 0.789 | 0.440 | 2.617 | 0.005 * | |
| Post-test | 32.77 (1.03) | 30.09 (2.54) | 0.743 | 0.000 * | ||||||
| Difference | 0.15 (1.57) | −0.31 (1.72) | 0.752 | 0.383 | ||||||
| Periorbital | Maximum | Pre-test | 35.91 (0.63) | 1.142 | 0.267 | 35.93 (0.70) | 2.560 | 0.020 * | 2.247 | 0.935 |
| Post-test | 35.67 (0.64) | 35.57 (0.67) | 2.190 | 0.346 | ||||||
| Difference | −0.15 (0.59) | −0.36 (0.62) | 2.906 | 0.267 | ||||||
| Average | Pre-test | 34.01 (0.90) | 0.294 | 0.771 | 33.93 (0.82) | 3.670 | 0.002 ** | 2.886 | 0.764 | |
| Post-test | 33.96 (0.85) | 33.35 (0.59) | 1.013 | 0.012 * | ||||||
| Difference | −0.05 (0.81) | −0.58 (.69) | 1.021 | 0.033 * | ||||||
| Minimum | Pre-test | 28.84 (2.10) | 0.366 | 0.718 | 29.28 (2.07) | 3.596 | 0.002 ** | 3.137 | 0.509 | |
| Post-test | 28.73 (1.77) | 28.18 (1.66) | 3.062 | 0.323 | ||||||
| Difference | −0.11 (1.43) | −1.10 (1.33) | 4.443 | 0.030 * | ||||||
| Maxillary | Maximum | Pre-test | 35.29 (0.99) | 0.579 | 0.569 | 35.11 (0.94) | 2.109 | 0.049 * | 4.282 | 0.548 |
| Post-test | 35.177 (0.90) | 34.70 (0.73) | 0.883 | 0.074 | ||||||
| Difference | −0.11 (0.90) | −0.41 (0.85) | 0.879 | 0.294 | ||||||
| Average | Pre-test | 33.27 (1.30) | 1.285 | 0.214 | 33.10 (1.25) | 2.872 | 0.010 * | −0.082 | 0.681 | |
| Post-test | 32.93 (1.08) | 32.42 (1.12) | −0.082 | 0.153 | ||||||
| Difference | −0.34 (1.21) | −0.68 (1.03) | 0.953 | 0.345 | ||||||
| Minimum | Pre-test | 27.59 (2.25) | 0.287 | 0.777 | 28.20 (2.55) | 4.011 | 0.001 ** | 0.951 | 0.424 | |
| Post-test | 27.46 (1.74) | 26.57 (2.08) | 1.126 | 0.148 | ||||||
| Difference | −0.12 (1.98) | −1.63 (1.77) | 1.123 | 0.016 * | ||||||
| Neck/ Upper chest | Maximum | Pre-test | 36.05 (0.92) | 2.177 | 0.042 * | 35.85 (0.71) | 2.189 | 0.042 * | 0.302 | 0.436 |
| Post-test | 35.652 (0.99) | 35.47 (0.74) | 0.304 | 0.514 | ||||||
| Difference | −0.40 (0.84) | −0.38 (0.75) | 2.635 | 0.934 | ||||||
| Average | Pre-test | 34.50 (0.91) | 1.711 | 0.103 | 34.23 (0.62) | 2.547 | 0.020 * | 2.681 | 0.279 | |
| Post-test | 34.21 (0.93) | 33.84 (0.62) | 2.210 | 0.158 | ||||||
| Difference | −0.30 (0.79) | −0.38 (0.66) | 2.228 | 0.703 | ||||||
| Minimum | Pre-test | 31.21 (1.91) | −0.054 | 0.957 | 30.69 (2.01) | 0.763 | 0.455 | −0.667 | 0.407 | |
| Post-test | 31.23 (1.65) | 30.17 (1.93) | −0.668 | 0.069 | ||||||
| Difference | 0.02 (2.01) | −0.51 (2.94) | 1.001 | 0.499 | ||||||
| Dependent Variable: STAI Pre-Test | Unstandardized Coefficients | Standardized Coefficients | |
|---|---|---|---|
| B | Standard Error | Beta | |
| Constant | −63.284 | 37.913 | |
| Nose temperature | 0.182 | 0.278 | 0.107 |
| Forehead temperature | 6.555 | 1.826 | 1.056 |
| Periorbital temperature | −2.310 | 2.096 | −0.309 |
| Maxillary temperature | −0.806 | 1.175 | −0.157 |
| Neck/upper chest temperature | −1.117 | 1.098 | −0.187 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mauriz, E.; Caloca-Amber, S.; Vázquez-Casares, A.M. Effect of Facial Skin Temperature on the Perception of Anxiety: A Pilot Study. Healthcare 2020, 8, 206. https://doi.org/10.3390/healthcare8030206
Mauriz E, Caloca-Amber S, Vázquez-Casares AM. Effect of Facial Skin Temperature on the Perception of Anxiety: A Pilot Study. Healthcare. 2020; 8(3):206. https://doi.org/10.3390/healthcare8030206
Chicago/Turabian StyleMauriz, Elba, Sandra Caloca-Amber, and Ana M. Vázquez-Casares. 2020. "Effect of Facial Skin Temperature on the Perception of Anxiety: A Pilot Study" Healthcare 8, no. 3: 206. https://doi.org/10.3390/healthcare8030206
APA StyleMauriz, E., Caloca-Amber, S., & Vázquez-Casares, A. M. (2020). Effect of Facial Skin Temperature on the Perception of Anxiety: A Pilot Study. Healthcare, 8(3), 206. https://doi.org/10.3390/healthcare8030206






