Feasibility of Ultrasound-Guided Trigger Point Injection in Patients with Myofascial Pain Syndrome
Abstract
1. Introduction
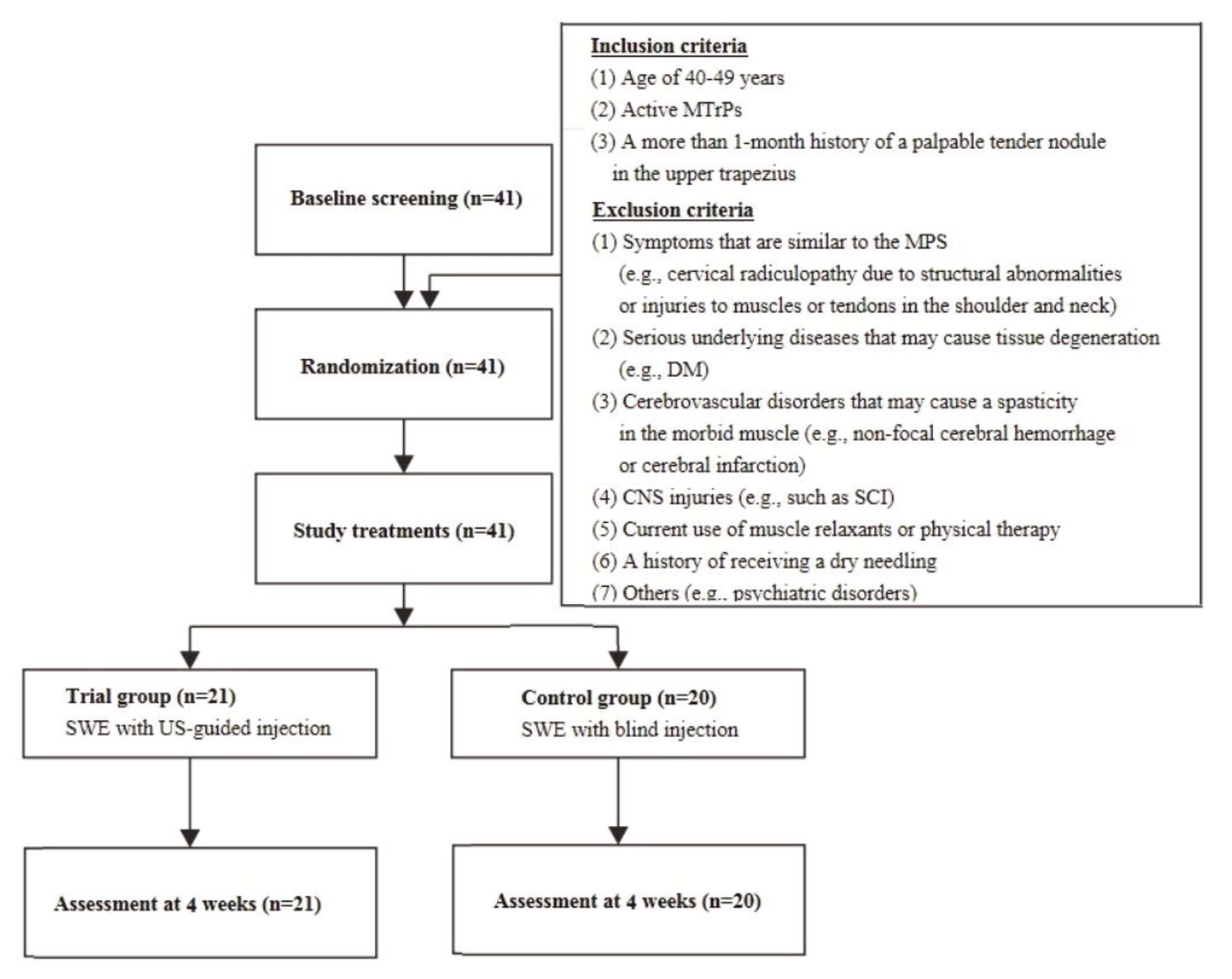
2. Materials and Methods
2.1. Study Patients and Setting
- (1)
- The patients aged between 40 and 49 years
- (2)
- The patients with active MTrPs presenting with spontaneous pain and tender nodules
- (3)
- The patients with a more than 1-month history of a palpable tender nodule in the upper trapezius [20].
- (1)
- The patients presenting with symptoms that are similar to the MPS (e.g., cervical radiculopathy due to structural abnormalities or injuries to muscles or tendons in the shoulder and neck) [20].
- (2)
- The patients with serious underlying diseases that may cause tissue degeneration (e.g., diabetes mellitus)
- (3)
- The patients with cerebrovascular disorders that may cause spasticity in the morbid muscle (e.g., non-focal cerebral hemorrhage or cerebral infarction)
- (4)
- The patients with central nervous system injuries (e.g., spinal cord injury)
- (5)
- The patients who were currently taking muscle relaxants or physical therapy
- (6)
- The patients who received a dry needling
- (7)
- The patients who are deemed to be ineligible for study participation according to our judgment (e.g., those with psychiatric disorders).
2.2. Patient Evaluation and Criteria
2.3. Evaluation Tools
2.4. Statistical Analysis of the Patient Data
3. Results
3.1. Baseline Characteristics of the Patients
3.2. Efficacy Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Leite, F.M.; Atallah, Á.N.; El Dib, R.; Grossmann, E.; Januzzi, E.; Andriolo, R.B.; da Silva, E.M. Cyclobenzaprine for the treatment of myofascial pain in adults. Cochrane Database Syst. Rev. 2009, 3, CD006830. [Google Scholar] [CrossRef] [PubMed]
- Desai, M.J.; Saini, V.; Saini, S. Myofascial pain syndrome: A treatment review. Pain Ther. 2013, 2, 21–36. [Google Scholar] [CrossRef] [PubMed]
- Chou, L.W.; Kao, M.J.; Lin, J.G. Probable mechanisms of needling therapies for myofascial pain control. Evid. Based Complement. Altern. Med. 2012, 2012, 705327. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.Z. Treatment of myofascial pain syndrome. Curr. Pain Headache Rep. 2006, 10, 345–349. [Google Scholar] [CrossRef]
- Simons, D.G. Understanding effective treatments of myofascial trigger points. J. Bodyw. Mov. Ther. 2002, 6, 81–88. [Google Scholar] [CrossRef]
- Simons, D.G. Clinical and etiological update of myofascial pain from trigger points. J. Musculoskelet. Pain 1996, 4, 93–122. [Google Scholar] [CrossRef]
- Fleckenstein, J.; Zaps, D.; Rüger, L.J.; Lehmeyer, L.; Freiberg, F.; Lang, P.M.; Irnich, D. Discrepancy between prevalence and perceived effectiveness of treatment methods in myofascial pain syndrome: Results of a cross-sectional, nationwide survey. BMC Musculoskelet. Disord. 2010, 11, 32. [Google Scholar] [CrossRef]
- Meyer, H.P. Myofascial pain syndrome and its suggested role in the pathogenesis and treatment of fibromyalgia syndrome. Curr. Pain Headache Rep. 2002, 6, 274–283. [Google Scholar] [CrossRef]
- Gerwin, R.D. Diagnosis of myofascial pain syndrome. Phys. Med. Rehabil. Clin. N. Am. 2014, 25, 341–355. [Google Scholar] [CrossRef]
- Yildirim, M.A.; Öneş, K.; Gökşenoğlu, G. Effectiveness of Ultrasound Therapy on Myofascial Pain Syndrome of the Upper Trapezius: Randomized, Single-Blind, Placebo-Controlled Study. Arch. Rheumatol. 2018, 33, 418–423. [Google Scholar] [CrossRef]
- Shah, J.P.; Phillips, T.M.; Danoff, J.V.; Gerber, L.H. An in vivo microanalytical technique for measuring the local biochemical milieu of human skeletal muscle. J. Appl. Physiol. 2005, 99, 1977–1984. [Google Scholar] [CrossRef] [PubMed]
- Narvani, A.A.; Tsiridis, E.; Kendall, S.; Chaudhuri, R.; Thomas, P. A preliminary report on prevalence of acetabular labrum tears in sports patients with groin pain. Knee Surg. Sports Traumatol. Arthrosc. 2003, 11, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Bubnov, R.V. Evidence-based pain management: Is the concept of integrative medicine applicable? EPMA J. 2012, 3, 13. [Google Scholar] [CrossRef] [PubMed]
- Condon, B. Magnetic resonance imaging and spectroscopy: How useful is it for prediction and prognosis? EPMA J. 2011, 2, 403–410. [Google Scholar] [CrossRef]
- Chen, Q.; Bensamoun, S.; Basford, J.R.; Thompson, J.M.; An, K.N. Identification and quantification of myofascial taut bands with magnetic resonance elastography. Arch. Phys. Med. Rehabil. 2007, 88, 1658–1661. [Google Scholar] [CrossRef]
- Ballyns, J.J.; Turo, D.; Otto, P.; Shah, J.P.; Hammond, J.; Gebreab, T.; Gerber, L.H.; Sikdar, S. Office-based elastographic technique for quantifying mechanical properties of skeletal muscle. J. Ultrasound Med. 2012, 31, 1209–1219. [Google Scholar] [CrossRef]
- Nightingale, K.; Soo, M.S.; Nightingale, R.; Trahey, G. Acoustic radiation force impulse imaging: In vivo demonstration of clinical feasibility. Ultrasound Med. Biol. 2002, 28, 227–235. [Google Scholar] [CrossRef]
- Lin, Y.C.; Yu, N.Y.; Jiang, C.F.; Chang, S.H. Characterizing the SEMG patterns with myofascial pain using a multi-scale wavelet model through machine learning approaches. J. Electromyogr. Kinesiol. 2018, 41, 147–153. [Google Scholar] [CrossRef]
- Jiang, C.F.; Lin, Y.C.; Yu, N.Y. Multi-scale surface electromyography modeling to identify changes in neuromuscular activation with myofascial pain. IEEE. Trans. Neural Syst. Rehabil. Eng. 2013, 21, 88–95. [Google Scholar] [CrossRef]
- Shah, J.P.; Thaker, N.; Heimur, J.; Aredo, J.V.; Sikdar, S.; Gerber, L. Myofascial Trigger Points Then and Now: A Historical and Scientific Perspective. PM R 2015, 7, 746–761. [Google Scholar] [CrossRef]
- Vanhoutte, E.K.; Faber, C.G.; Van Nes, S.I.; Jacobs, B.C.; Van Doorn, P.A.; Van Koningsveld, R.; Cornblath, D.R.; Van Der Kooi, A.J.; Cats, E.A.; Van Den Berg, L.H.; et al. Modifying the Medical Research Council grading system through Rasch analyses. Brain 2012, 135, 1639–1649. [Google Scholar] [CrossRef] [PubMed]
- Wilk, K.E.; Reinold, M.M.; Macrina, L.C.; Porterfield, R.; Devine, K.M.; Suarez, K.; Andrews, J.R. Glenohumeral internal rotation measurements differ depending on stabilization techniques. Sports Health 2009, 1, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Haefeli, M.; Elfering, A. Pain assessment. Eur. Spine J. 2006, 15, S17–S24. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.L.; Lester, S.; Taylor, A.W.; Shanahan, M.E.; Gill, T.K. Factor structure and validity of the shoulder pain and disability index in a population-based study of people with shoulder symptoms. BMC Musculoskelet. Disord. 2011, 12, 8. [Google Scholar] [CrossRef]
- Seo, H.D.; Lee, K.W.; Chung, K.S.; Chung, I.J. Reliability and Validity of the Korean version of Shoulder Pain and Disability Index. J. Spec. Educ. Rehabil. Sci. 2012, 51, 319–336. [Google Scholar]
- Song, K.J.; Choi, B.W.; Choi, B.R.; Seo, G.B. Cross-cultural adaptation and validation of the Korean version of the neck disability index. Spine 2010, 35, E1045–E1049. [Google Scholar] [CrossRef]
- Maher, R.M.; Hayes, D.M.; Shinohara, M. Quantification of dry needling and posture effects on myofascial trigger points using ultrasound shear-wave elastography. Arch. Phys. Med. Rehabil. 2013, 94, 2146–2150. [Google Scholar] [CrossRef]
- Vulfsons, S.; Ratmansky, M.; Kalichman, L. Trigger point needling: Techniques and outcome. Curr. Pain Headache Rep. 2012, 16, 407–412. [Google Scholar] [CrossRef]
- Lalitha, P.; Reddy, M.; Reddy, K.J. Musculoskeletal applications of elastography: A pictorial essay of our initial experience. Korean J. Radiol. 2011, 12, 365–375. [Google Scholar] [CrossRef][Green Version]
- Jones, D.A.; Newham, D.J.; Clarkson, P.M. Skeletal muscle stiffness and pain following eccentric exercise of the elbow flexors. Pain 1987, 30, 233–242. [Google Scholar] [CrossRef]
- McHugh, M.P.; Connolly, D.A.; Eston, R.G.; Kremenic, I.J.; Nicholas, S.J.; Gleim, G.W. The role of passive muscle stiffness in symptoms of exercise-induced muscle damage. Am. J. Sports Med. 1999, 27, 594–599. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, R.I. Shortening of muscle fibres during stretch of the active cat medial gastrocnemius muscle: The role of tendon compliance. J. Physiol. 1991, 436, 219–236. [Google Scholar] [CrossRef] [PubMed]
- Shalabi, N.; Persson, M.; Månsson, A.; Vengallatore, S.; Rassier, D.E. Sarcomere Stiffness during Stretching and Shortening of Rigor Skeletal Myofibrils. Biophys. J. 2017, 113, 2768–2776. [Google Scholar] [CrossRef] [PubMed]
- So, J.I.; Song, D.H.; Park, J.H.; Choi, E.; Yoon, J.Y.; Yoo, Y.; Chung, M.E. Accuracy of Ultrasound-Guided and Non-Ultrasound-Guided Botulinum Toxin Injection into Cadaver Salivary Glands. Ann. Rehabil. Med. 2017, 41, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.S.; Wong, S.H. A new look at trigger point injections. Anesthesiol. Res. Pract. 2012, 2012, 492452. [Google Scholar] [CrossRef] [PubMed]
- Lamplot, J.D.; Lillegraven, O.; Brophy, R.H. Outcomes from Conservative Treatment of Shoulder Idiopathic Adhesive Capsulitis and Factors Associated with Developing Contralateral Disease. Orthop. J. Sports Med. 2018, 6, 2325967118785169. [Google Scholar] [CrossRef] [PubMed]
- Muir, J.J.; Curtiss, H.M.; Hollman, J.; Smith, J.; Finnoff, J.T. The accuracy of ultrasound-guided and palpation-guided peroneal tendon sheath injections. Am. J. Phys. Med. Rehabil. 2011, 90, 564–571. [Google Scholar] [CrossRef]
- Daniels, E.W.; Cole, D.; Jacobs, B.; Phillips, S.F. Existing Evidence on Ultrasound-Guided Injections in Sports Medicine. Orthop. J. Sports Med. 2018, 6, 2325967118756576. [Google Scholar] [CrossRef]
- Koh, S.H.; Lee, S.C.; Lee, W.Y.; Kim, J.; Park, Y. Ultrasound-guided intra-articular injection of hyaluronic acid and ketorolac for osteoarthritis of the carpometacarpal joint of the thumb: A retrospective comparative study. Medicine 2019, 98, e15506. [Google Scholar] [CrossRef]
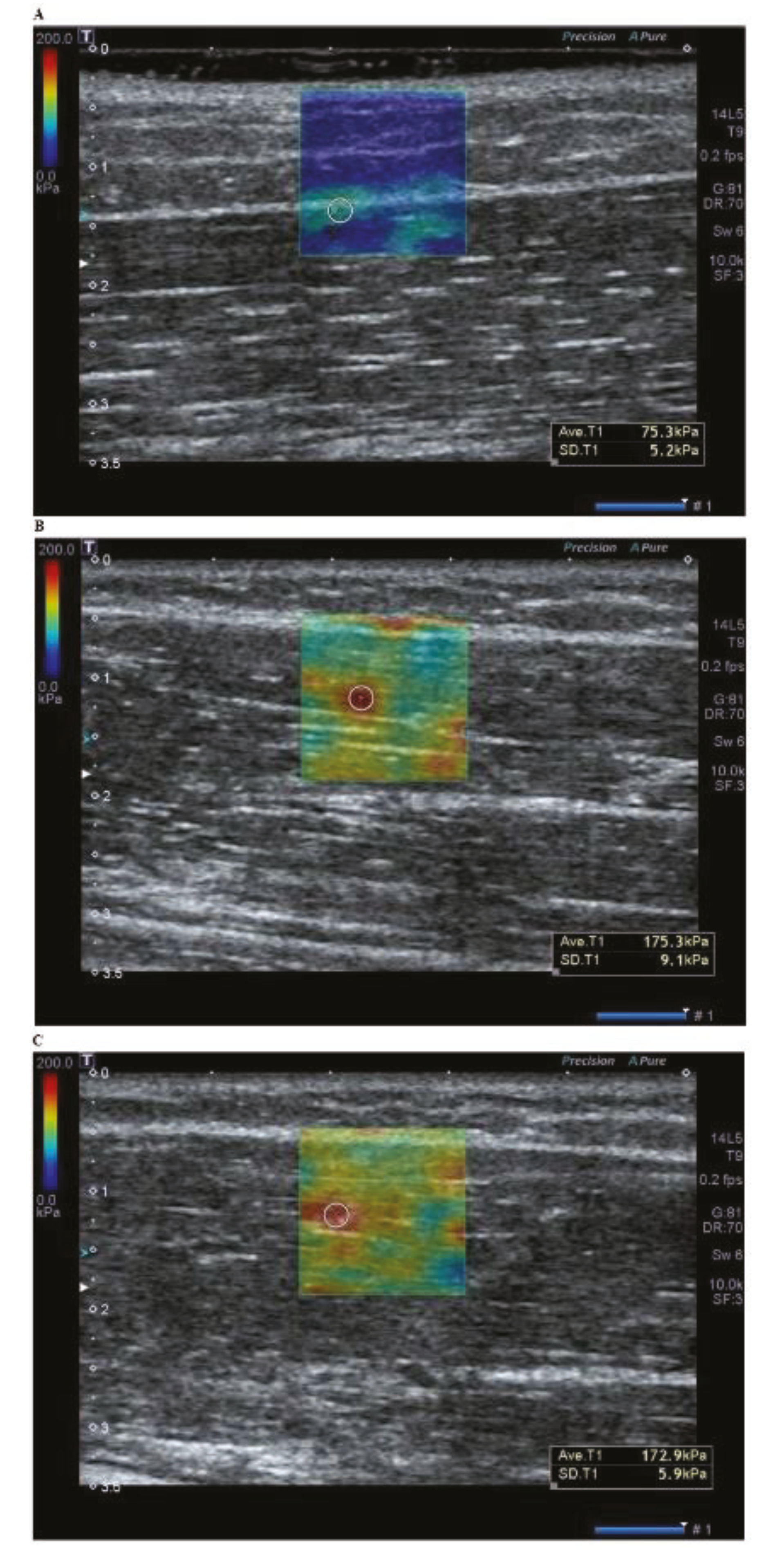
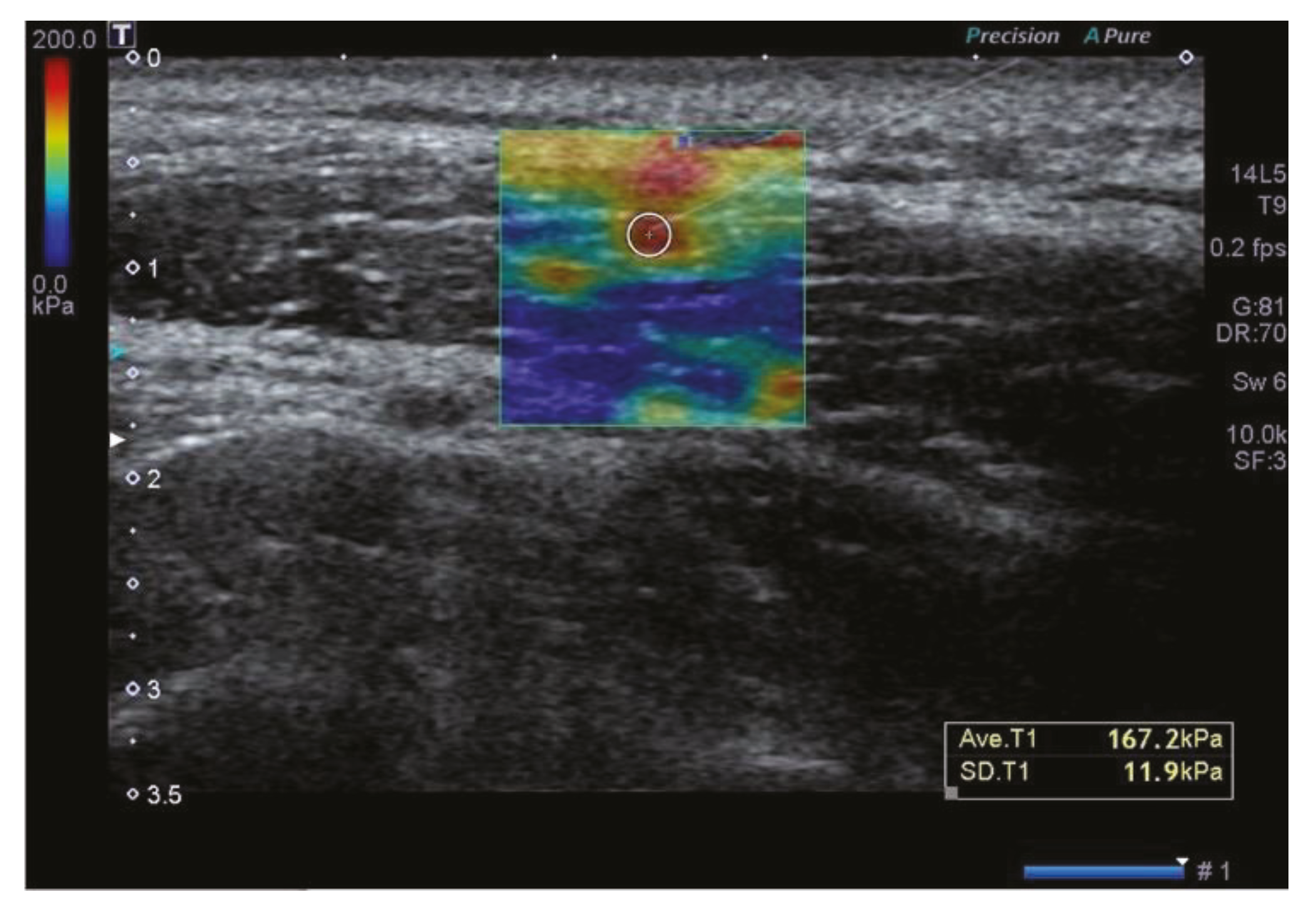
| Variables | Values | |
|---|---|---|
| Age (years) | 44.27 ± 2.22 | |
| Sex | ||
| Men | 18 | |
| Women | 23 | |
| Daily use of analgesics | 1.78 ± 0.80 | |
| ROM | ||
| Abduction | 154.51 ± 7.40° | |
| Adduction | 154.40 ± 6.34° | |
| Flexion | 157.56 ± 6.14° | |
| Extension | 64.36 ± 4.66° | |
| External rotation | 92.44 ± 4.89° | |
| Internal rotation | 96.34 ± 5.48° | |
| MMT | ||
| Abduction | 4.95 ± 0.22 | |
| Adduction | 4.95 ± 0.22 | |
| Flexion | 4.9 5 ± 0.22 | |
| Extension | 4.95 ± 0.22 | |
| External rotation | 4.93 ± 0.26 | |
| Internal rotation | 4.93 ± 0.26 | |
| Pain VAS scores | 7.05 ± 0.62 | |
| NDI scores | 20.54 ± 3.23 | |
| SPADI scores | 38.32 ± 6.50 | |
| Number of palpable tender nodules | 2.37 ± 0.80 | |
| Spasticity on SWE | 159.63 ± 9.72 | |
| Variables | Values | p-Value | ||
|---|---|---|---|---|
| Trial Group (n = 21) | Control Group (n = 20) | |||
| Age (years) | 43.95 ± 2.18 | 44.60 ± 2.33 | 0.652 | |
| Sex | ||||
| Men | 9 | 10 | ||
| Women | 12 | 11 | ||
| Variables | Values | p-Value | ||||||
|---|---|---|---|---|---|---|---|---|
| Trial Group (n = 21) | Control Group (n = 20) | |||||||
| 0 week | 4 weeks | Δ | 0 week | 4 weeks | Δ | |||
| ROM | ||||||||
| Abduction | 154.52 ± 7.57° | 155.00 ± 7.25° | 0.48 ± 1.50° | 154.50 ± 7.42° | 154.50 ± 7.42° | 0.00 ± 0.00° | 0.162 | |
| Adduction | 154.40 ± 6.34° | 154.76 ± 6.98° | 0.00 ± 0.00 | 154.40 ± 6.34° | 154.25 ± 5.45° | 0.25 ± 1.12° | 0.330 | |
| Flexion | 157.62 ± 6.25° | 158.10 ± 5.80° | 0.48 ± 1.50° | 157.50 ± 6.18° | 157.75 ± 5.73° | 0.25 ± 1.12° | 0.276 | |
| Extension | 64.52 ± 4.98° | 64.76 ± 5.12° | 0.24 ± 1.09° | 64.75 ± 4.44° | 65.00 ± 4.30° | 0.25 ± 1.12° | 0.945 | |
| External rotation | 92.14 ± 5.38° | 92.38 ± 5.15° | 0.24 ± 1.09° | 92.75 ± 4.44° | 93.00 ± 4.41° | 0.25 ± 1.12° | 0.945 | |
| Internal rotation | 96.67 ± 5.55° | 96.90 ± 5.58° | 0.24 ± 1.09° | 96.00 ± 5.53° | 96.25 ± 5.35° | 0.25 ± 1.12° | 0.945 | |
| MMT | ||||||||
| Abduction | 4.95 ± 0.22 | 5 ± 0 | 0.05 ± 0.22 | 4.95 ± 0.22 | 4.95 ± 0.22 | 0.00 ± 0.00 | 0.329 | |
| Adduction | 4.95 ± 0.22 | 4.95 ± 0.22 | 0.00 ± 0.00 | 4.95 ± 0.22 | 5.00 ± 0.00 | 0.05 ± 0.22 | 0.330 | |
| Flexion | 4.90 ± 0.30 | 4.95 ± 0.22 | 0.05 ± 0.22 | 4.95 ± 0.22 | 5.00 ± 0.00 | 0.05 ± 0.22 | 0.945 | |
| Extension | 4.95 ± 0.22 | 4.95 ± 0.22 | 0.00 ± 0.00 | 4.95 ± 0.22 | 5.00 ± 0.00 | 0.05 ± 0.22 | 0.330 | |
| External rotation | 4.95 ± 0.22 | 4.95 ± 0.22 | 0.00 ± 0.00 | 4.90 ± 0.31 | 4.90 ± 0.31 | 0.00 ± 0.00 | 0.207 | |
| Internal rotation | 4.90 ± 0.30 | 4.90 ± 0.30 | 0.00 ± 0.00 | 4.95 ± 0.22 | 4.95 ± 0.22 | 0.00 ± 0.00 | 0.276 | |
| Pain VAS scores | 7.06 ± 0.61 | 5.14 ± 0.74 | 1.92 ± 0.56 | 7.03 ± 0.65 | 5.83 ± 1.24 | 1.20 ± 0.85 | * 0.003 | |
| NDI scores | 20.67 ± 3.28 | 9.52 ± 3.48 | 11.14 ± 4.19 | 20.40 ± 3.25 | 14.55 ± 7.32 | 5.85 ± 7.80 | * 0.012 | |
| SPADI scores | 38.67 ± 6.20 | 18.52 ± 8.86 | 20.14 ± 8.90 | 37.95 ± 6.95 | 28.25 ± 15.39 | 9.70 ± 16.39 | * 0.018 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, J.J.; Kim, J.; Park, S.; Paek, S.; Kim, T.H.; Kim, D.K. Feasibility of Ultrasound-Guided Trigger Point Injection in Patients with Myofascial Pain Syndrome. Healthcare 2019, 7, 118. https://doi.org/10.3390/healthcare7040118
Kang JJ, Kim J, Park S, Paek S, Kim TH, Kim DK. Feasibility of Ultrasound-Guided Trigger Point Injection in Patients with Myofascial Pain Syndrome. Healthcare. 2019; 7(4):118. https://doi.org/10.3390/healthcare7040118
Chicago/Turabian StyleKang, Jung Joong, Jungin Kim, Seunghun Park, Sungwoo Paek, Tae Hee Kim, and Dong Kyu Kim. 2019. "Feasibility of Ultrasound-Guided Trigger Point Injection in Patients with Myofascial Pain Syndrome" Healthcare 7, no. 4: 118. https://doi.org/10.3390/healthcare7040118
APA StyleKang, J. J., Kim, J., Park, S., Paek, S., Kim, T. H., & Kim, D. K. (2019). Feasibility of Ultrasound-Guided Trigger Point Injection in Patients with Myofascial Pain Syndrome. Healthcare, 7(4), 118. https://doi.org/10.3390/healthcare7040118





