Mobile Self-Monitoring ECG Devices to Diagnose Arrhythmia that Coincide with Palpitations: A Scoping Review
Abstract
1. Introduction
1.1. Overview of Mobile ECG Devices
1.2. Background Literature
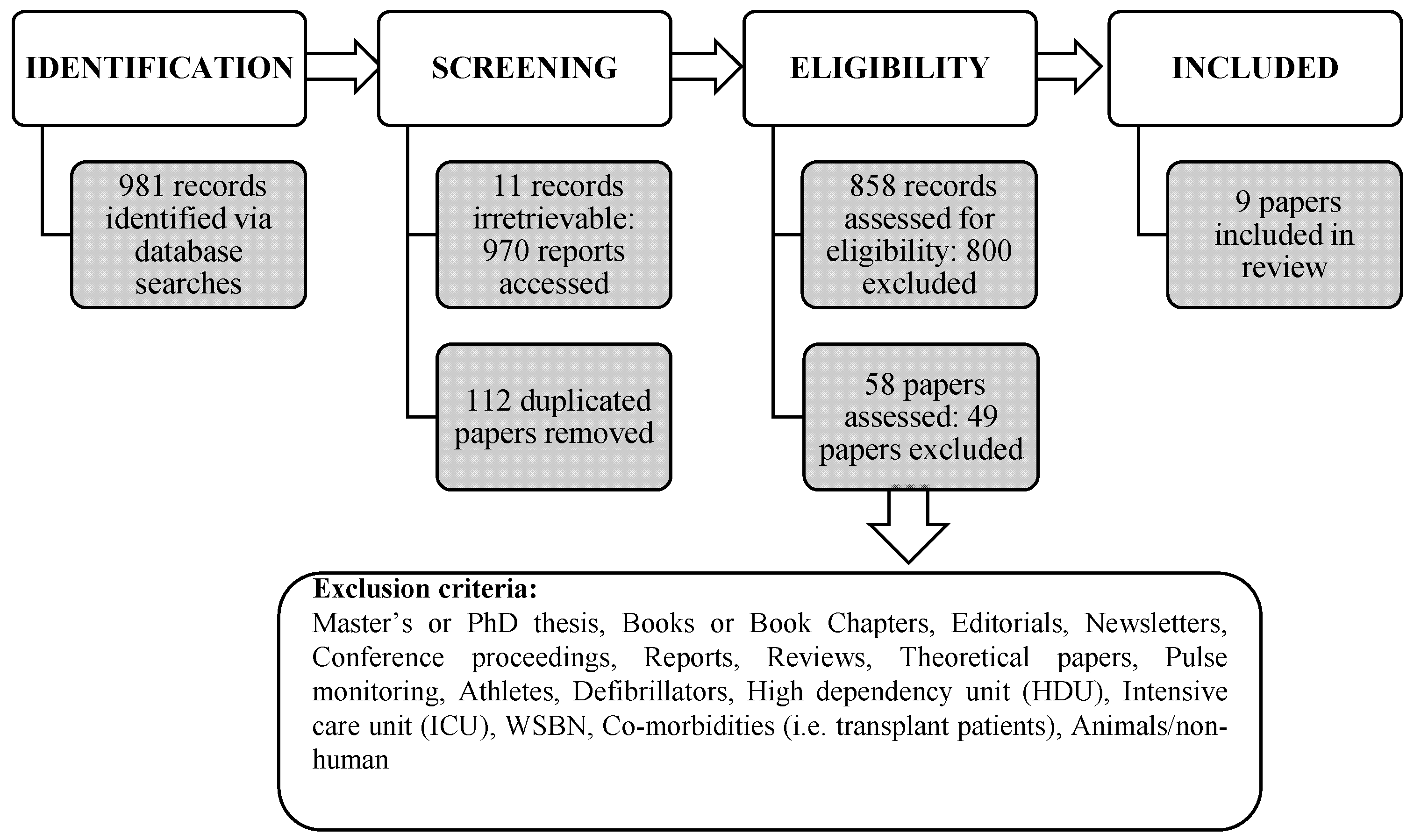
2. Methods
2.1. Objectives
2.2. Search Strategy
2.3. Selection Criteria
3. Results
3.1. General Characteristics of Studies
3.2. Themes
3.3. Primary Themes
4. Discussion
Principle Findings
5. Limitations
6. Future Research
- Future work may wish to consider undertaking a systematic review in order to synthesise existing and recently published work. This systematic review could include development features, accuracy, algorithms, utility and reproducibility, in addition to diagnostics and user/patient experience(s).
- Following the work of Evans et al. [40], clinicians and researchers alike should consider exploring the use of mobile ECG devices from the standpoint of health practitioners working in the delivery of primary and community care.
- Implementing and conducting qualitative data collection in future studies would provide a greater insight and understanding of the needs, apprehensions, and expectations of patients and primary care practitioners. Simultaneously, this would provide the opportunity to examine the role of patient’s and support networks. Evans et al. [40] illustrated the potential opportunities for mobile ECG monitoring in low, middle income countries (LMICs), and by their approach has the potential to offer substantial changes in developed and developing regions.
- Future investigations should explore the adherence and adoption of mobile ECG devices, learning from previous health, gerontological and ICT studies [61,62,63,64,65,66,67,68]. Existing research in different fields has demonstrated how technology has been used and evaluated by community dwelling adults living in different geographic locations. Understanding people’s motivations and behaviour in relation to technology would significantly support future work in this field. In addition, the impact of technology efficacy by health practitioners on service delivery could be assessed.
- Privacy and security issues and concerns surrounding data need to be addressed from a multi-disciplinary standpoint. Further work is needed to explore the use of wearable devices from a clinical environment and conducting qualitative data to gain an in-depth insight into the concerns of patients, support networks and practitioners.
- Future studies should determine the exact cost effectiveness of deploying mobile ECG devices with the aim of providing evidence to health care strategists, governments and managers of the benefits of this form of technology in the community. Such studies could have a significant impact in the care pathways following the detection of AR and AF.
- To ascertain how mobile ECG devices could affect the delivery of primary care, we suggest that a large-scale feasibility study, encompassing variable populations (i.e., age range, ethnicity and socio economic), should be conducted to provide results to different actors (i.e., government, health care practitioners, health care strategists, researchers, patients and support networks). It is important that such studies include as full a range of actors as possible from primary care physicians, cardiologists, patients, lay people, patients’ support networks, health organizations (i.e., NICE), and government funding agencies.
- Future scoping reviews should follow the recent extension to the existing PRISMA protocol—the PRISMA Extension for Scoping Reviews (PRISMA-ScR) [68].
7. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| ACM | Association of Computing Machinery |
| AF | Atrial Fibrillation |
| AFEQT | Atrial Fibrillation Effect on Quality of Life |
| AFKS | Atrial Fibrillation Knowledge Scale |
| AR | Arrhythmia |
| CAS-R | Control Attitudes Scale-Revised |
| CCS-SAF | Canadian Cardiovascular Society Severity in Atrial Fibrillation scale |
| CE | Cost Effectiveness |
| ECG/EKG | Electrocardiogram |
| EE | Energy Expenditure |
| EQ-5D | European Questionnaire 5 Dimensions |
| HR | Heart rate |
| mApp | Mobile Apps |
| NHS | National Health Service |
| NICE | National Institute for Health Care Excellence |
| PHQ-9 | Patient Health Questionnaire |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| QALY | Quality-Adjusted Life Years |
| QOL | Quality of Life |
| RCT | Randomised controlled trial |
| SEAMS | Self-Efficacy for Appropriate Medication Use Scale |
| STAI | State Trait Anxiety Inventory |
| UK | United Kingdom |
| USA | United States of America |
| VA | Veteran Affairs |
References
- Public Health England (PHE). Atrial Fibrillation Prevalence Estimates in England: Application of Recent Population Estimates of AF in Sweden; Public Health England: London, UK, 2017; Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/644869/atrial_fibrillation_AF_briefing.pdf and http://www.webcitation.org/77eZ6R3Y7; (accessed on 14 February 2019).
- United Nations. World Population Prospects: The 2017 Revision, Key Findings and Advance Tables; Report No.: ESA/P/WP/248; United Nations: New York, NY, USA, 2017; Available online: https://esa.un.org/unpd/wpp/Publications/Files/WPP2017_KeyFindings.pdf and http://www.webcitation.org/754kCtnkX; (accessed on 14 February 2019). [Google Scholar]
- Alliance, A. Atrial Fibrillation. Available online: http://www.heartrhythmalliance.org/aa/uk/atrial-fibrillation. (accessed on 14 February 2019).
- Alliance, A. What Is an Arrhythmia? Available online: http://www.heartrhythmalliance.org/aa/uk/what-is-an-arrhythmia?gclid=Cj0KCQiA6JjgBRDbARIsANfu58E53eiuxOFo9nYKJcxOQNi0iIn_T6ZnEU28WcZQcprn6AqNtvyOM7MaAg2bEALw_wcB (accessed on 14 February 2019).
- Odutayo, A.; Wong, C.X.; Hsiao, A.J.; Hopewell, S.; Altman, D.G.; Emdin, C.A. Atrial fibrillation and risks of cardiovascular disease, renal disease, and death: systematic review and meta-analysis. BMJ 2016, 354, i4482. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.J.; Larson, M.G.; Levy, D.; Vasan, R.S.; Leip, E.P.; Wolf, P.A.; D’Agostino, R.B.; Murabito, J.M.; Kannel, W.B.; Benjamin, E.J. Temporal relations of atrial fibrillation and congestive heart failure and their joint influence on mortality: The Framingham heart study. Circulation 2003, 107, 2920–2925. [Google Scholar] [CrossRef] [PubMed]
- Thrall, G.; Lane, D.; Carroll, D.; Lip, G.Y.H. Quality of life in patients with atrial fibrillation: A systematic review. Am. J. Med. 2006, 119, 448.e1–448.e19. [Google Scholar] [CrossRef] [PubMed]
- Kannel, W.; Wolf, P.; Benjamin, E.; Levy, D. Prevalence, incidence, prognosis, and predisposing conditions for atrial fibrillation: population-based estimates. Am. J. Cardiol. 1998, 82, 2N–9N. [Google Scholar] [CrossRef]
- Cheung, C.C.; Krahn, A.D.; Andrade, J.G. The emerging role of wearable technologies in detection of arrhythmia. Can. J. Cardiol. 2018, 34, 1083–1087. [Google Scholar] [CrossRef] [PubMed]
- National Institute for Clinical Excellence (NICE). AliveCor Heart Monitor and AliveECG app (Kardia Mobile) for Detecting Atrial Fibrillation. 2015. Available online: https://www.nice.org.uk/advice/mib35 (accessed on 14 February 2019).
- Campbell, B.; Richley, D.; Ross, C.; Eggett, C.J. Clinical Guidelines by Consensus: Recording a Standard 12-Lead Electrocardiogram. An Approved Method by the Society for Cardiological Science and Technology (SCST). 2017. Available online: http://www.scst.org.uk/resources/SCST_ECG_Recording_Guidelines_20171.pdf (accessed on 30 July 2019).
- Marston, H.R.; Hall, A.K. Gamification: Application for Health and Health Information Technology Engagement. In Handbook of Research on Holistic Perspectives in Gamification for Clinical Practice; Novak, D., Tulu, B., Brendryen, H., Eds.; Medical Information Science Reference: Hershey, PA, USA, 2015; pp. 78–104. [Google Scholar]
- Hendrikx, T.; Rosenqvist, M.; Wester, P.; Sandström, H.; Hörnsten, R. Intermittent short ECG recording is more effective than 24-hour Holter ECG in detection of arrhythmias. BMC Cardiovasc. Disord. 2014, 14, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Usadel, L.; Haverkämper, G.; Herrmann, S.; Löber, R.; Weiss, K.; Opgen-Rhein, B.; Berger, F.; Will, J.C. Arrhythmia detection in pediatric patients: ECG quality and diagnostic yield of a patient-triggered Einthoven lead-I event recorder (Zenicor EKG-2TM). Pediatr. Cardiol. 2016, 37, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Dahlqvist, J.A.; Karlsson, M.; Wiklund, U.; Hörnsten, R.; Rydberg, A. Handheld ECG in analysis of arrhythmia and heart rate variability in children with Fontan circulation. J. Electrocardiol. 2014, 47, 374–382. [Google Scholar] [CrossRef]
- HeartCheck™ device. Available online: https://www.theheartcheck.com/consumer.html (accessed on 14 February 2019).
- Quinn, F.R.; Gladstone, D.J.; Ivers, N.M.; Sandhu, R.K.; Dolovich, L.; Ling, A.; Nakamya, J.; Ramasundarahettige, C.; Frydrych, P.A.; Henein, S.; et al. Diagnostic accuracy and yield of screening tests for atrial fibrillation in the family practice setting: A multicenter cohort study. CMAJ Open 2018, 6, 308–315. [Google Scholar] [CrossRef][Green Version]
- EMAY Handheld ECG/EKG. Available online: http://www.emayltd.com/index.php/Home/Index/detail/id/17 (accessed on 14 February 2019).
- ClinicalTrials Search Term—EMAY device. Results (n = 0). Available online: : http://www.webcitation.org/77hmzZ5BS (accessed on 14 February 2019).
- Beurer Mobile ECG Device. Available online: https://www.beurer.com/web/gb/products/medical/ecg-and-pulse-oximeter/mobile-ecg-device (accessed on 14 February 2019).
- AliveCor. KardiaMobile. 2019. Available online: https://shop.gb.alivecor.com/products/kardiamobile (accessed on 14 February 2019).
- AliveCor. Clinical Research. 2019. Available online: https://alivecor.com/research (accessed on 14 February 2019).
- Pevnick, J.M.; Birkeland, K.; Zimmer, R.; Elad, Y.; Kedan, I. Wearable technology for cardiology: An update and framework for the future. Trends Cardiovasc. Med. 2018, 28, 144–150. [Google Scholar] [CrossRef]
- Lau, J.; Lowres, N.; Neubeck, L.; Neubeck, L.; Brieger, D.B.; Sy, R.W.; Galloway, C.D.; Albert, D.E.; Freedman, S.B. iPhone ECG application for community screening to detect silent atrial fibrillation: A novel technology to prevent stroke. Int. J. Cardiol. 2013, 165, 193–194. [Google Scholar] [CrossRef] [PubMed]
- Lowres, N.; Neubeck, L.; Salkeld, G.; Krass, I.; McLachlan, A.J.; Redfern, J.; Bennett, A.A.; Briffa, T.; Bauman, A.; Martinez, C.; et al. Feasibility and cost-effectiveness of stroke prevention through community screening for atrial fibrillation using iPhone ECG in pharmacies. The Searchaf study. Thromb. Haemost. 2014, 111, 1167–1176. [Google Scholar] [CrossRef] [PubMed]
- Haberman, Z.C.; Jahn, R.T.; Bose, R.; Tun, H.; Shinbane, J.S.; Doshi, R.N.; Chang, P.M.; Saxon, L.A. Wireless smartphone ECG enables large-scale screening in diverse populations. J. Cardiovasc. Electrophysiol. 2015, 26, 520–526. [Google Scholar] [CrossRef] [PubMed]
- Tarakji, K.G.; Wazni, O.M.; Callahan, T.; Kanj, M.; Hakim, A.H.; Wolski, K.; Wilkoff, B.L.; Saliba, W.; Lindsay, B.D. Using a novel wireless system for monitoring patients after the atrial fibrillation ablation procedure: The iTransmit study. Heart Rhythm 2015, 12, 554–559. [Google Scholar] [CrossRef] [PubMed]
- Zenicor Medical Systems. 2019. Available online: https://zenicor.com/our-contact-information (accessed on 14 February 2019).
- Zenicor Medical Systems. Zenicor-ECG. 2019. Available online: https://zenicor.com/zenicor-ekg (accessed on 14 February 2019).
- Reed, M.J.; Grubb, N.R.; Lang, C.C.; O’Brien, R.; Simpson, K.; Padarenga, M.; Grant, A.; Tuck, S. Multi-centre randomised controlled trial of a smartphone-based event recorder alongside standard care versus standard care for patients presenting to the emergency department with palpitations and pre-syncope: The IPED (Investigation of Palpitations in the ED) study. Trials 2018, 19, 7–11. [Google Scholar]
- ClinicalTrials. Available online: http://www.webcitation.org/77hmrlWkq (accessed on 14 February 2019).
- ClinicalTrials. Search Term—AliveCor: Results (n = 25). 2019. Available online: https://clinicaltrials.gov/ct2/results?cond=&term=aliveCor&cntry=&state=&city=&dist= (accessed on 14 February 2019).
- ClinicalTrials. Search Term—Zenicor: Results (n = 5). 2019. Available online: https://clinicaltrials.gov/ct2/results?cond=&term=zenicor&cntry=&state=&city=&dist= (accessed on 14 February 2019).
- ClinicalTrials. Search Term—Atrial Fibrillation, Wearable Devices: Results (n = 17). 2019. Available online: https://clinicaltrials.gov/ct2/results?cond=&term=atrial+fibrillation%2C+wearable+devices&cntry=&state=&city=&dist= (accessed on 14 February 2019).
- ClinicalTrials. Search Term—HeartCheck Hand-Held ECG Device: Results (n = 1). 2019. Available online: https://clinicaltrials.gov/ct2/results?cond=&term=HeartCheck+Hand-held+ECG+device&cntry=&state=&city=&dist= (accessed on 14 February 2019).
- Arksey, H.; O’Malley, L. Scoping studies: Towards a methodological framework. Int. J. Soc. Res. Methodol. Theory Pract. 2005, 8, 19–32. [Google Scholar] [CrossRef]
- Aronsson, M.; Svennberg, E.; Rosenqvist, M.; Engdahl, J.; Al-Khalili, F.; Friberg, L.; Frykman-Kull, V.; Levin, L.A. Cost-effectiveness of mass screening for untreated atrial fibrillation using intermittent ECG recording. Europace 2015, 17, 1023–1029. [Google Scholar] [CrossRef]
- Doliwa, P.S.; Rosenqvist, M.; Frykman, V. Paroxysmal atrial fibrillation with silent episodes: Intermittent versus continuous monitoring. Scand. Cardiovasc. J. 2012, 46, 144–148. [Google Scholar] [CrossRef]
- Boudreaux, B.D.; Hebert, E.P.; Hollander, D.B.; Williams, B.M.; Cormier, C.L.; Naquin, M.R.; Gillan, W.W.; Gusew, E.E.; Kraemer, R.R. Validity of wearable activity monitors during cycling and resistance exercise. Med. Sci. Sports Exerc. 2017, 50, 624–633. [Google Scholar] [CrossRef]
- Evans, G.F.; Shirk, A.; Muturi, P.; Soliman, E.Z. Feasibility of using mobile ECG recording technology to detect atrial fibrillation in low-resource settings. Glob. Heart 2017, 12, 285–289. [Google Scholar] [CrossRef]
- Hickey, A.M.; Hauser, N.R.; Valente, L.E.; Riga, T.C.; Frulla, A.P.; Masterson Creber, R.; Whang, W.; Garan, H.; Jia, H.; Csiacca, R.R.; et al. A single-center randomized, controlled trial investigating the efficacy of a mHealth ECG technology intervention to improve the detection of atrial fibrillation: The iHEART study protocol. BMC Cardiovasc. Disord. 2016, 16, 152. [Google Scholar] [CrossRef] [PubMed]
- Halcox, J.P.J.; Wareham, K.; Cardew, A.; Gilmore, M.; Barry, J.P.; Phillips, C.; Gravenor, M.B. Assessment of remote heart rhythm sampling using the AliveCor heart monitor to screen for atrial fibrillation the REHEARSE-AF study. Circulation 2017, 136, 1784–1794. [Google Scholar] [CrossRef] [PubMed]
- McManus, D.D.; Chong, J.W.; Soni, A.; Saczynski, J.S.; Esa, N.; Napolitano, C.; Darling, C.E.; Boyer, E.; Roselle, R.K.; Floyd, K.C.; et al. Pulse-Smart: Pulse-based arrhythmia discrimination using a novel smartphone application. J. Cardiovasc. Electrophysiol. 2016, 27, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Turakhia, M.P.; Ullal, A.J.; Hoang, D.D.; Than, C.T.; Miller, J.D.; Friday, K.J.; Perez, M.V.; Freeman, J.V.; Wang, P.J.; Heidenreich, P.A.; et al. Feasibility of extended ambulatory electrocardiogram monitoring to identify silent atrial fibrillation in high-risk patients: The screening study for undiagnosed atrial fibrillation (STUDY-AF). Clin. Cardiol. 2015, 38, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Friberg, L.; Bergfeldt, L. Atrial fibrillation prevalence revisited. J. Intern. Med. 2013, 274, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Hendriks, J.M.L.; Crijns, H.J.G.M.; Tieleman, R.G.; Vrijhoef, H.J.M. The atrial fibrillation knowledge scale: Development, validation and results. Int. J. Cardiol. 2013, 168, 1422–1428. [Google Scholar] [CrossRef] [PubMed]
- Dorian, P.; Cvitkovic, S.S.; Kerr, C.R.; Crystal, E.; Gillis, A.M.; Guerra, P.G.; Mitchell, L.B.; Roy, D.; Skanes, A.C.; Wyse, D.G. A novel, simple scale for assessing the symptom severity of atrial fibrillation at the bedside: The CCS-SAFE Scale. Can. J. Cardiol. 2006, 22, 383–386. [Google Scholar] [CrossRef]
- Aliot, E.; Botto, G.L.; Crijns, H.J.; Kirchhof, P. Quality of life in patients with atrial fibrillation: How to assess it and how to improve it. Europace 2014, 16, 787–796. [Google Scholar] [CrossRef]
- Moser, D.K.; Riegel, B.; McKinley, S.; Doering, L.V.; Meischke, H.; Heo, S.; Lennie, T.A.; Dracup, K. The control attitudes scale-revised: Psychometric evaluation in three groups of patients with cardiac illness. Nurs. Res. 2009, 58, 42–51. [Google Scholar] [CrossRef]
- Moon, S.J.; Lee, W.Y.; Hwang, J.S.; Hong, Y.P.; Morisky, D.E. Accuracy of a screening tool for medication adherence: A systematic review and meta-analysis of the Morisky medication adherence scale-8. PLoS ONE 2017, 12, 1–18. [Google Scholar] [CrossRef]
- Morisky, D.; Green, L.; Levine, D. Concurrent and predictive validity of a self-reported measure of medication adherence. Med. Care 1986, 24, 67–74. [Google Scholar] [CrossRef]
- Risser, J.; Jacobson, T.A.; Kripalani, S. Development and psychometric evaluation of the self-efficacy for appropriate medication use scale (SEAMS) in low-literacy patients with chronic disease. J. Nurs. Meas. 2007, 15, 203–219. [Google Scholar] [CrossRef]
- Jenkinson, C.; Wright, L.; Coulter, A. Criterion validity and reliability of the SF-36 in a population sample. Qual. Life Res. 1994, 3, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Jenkinson, C.; Layte, R.; Coulter, A.; Wright, L. Evidence for the sensitivity of the SF-36 health status measure to inequalities in health: Results from the Oxford healthy lifestyles survey. J. Epidemiol. Community Health 1996, 50, 377–380. [Google Scholar] [CrossRef][Green Version]
- Brooks, R. EuroQol: The current state of play. Health Policy 1996, 37, 53–72. [Google Scholar] [CrossRef]
- Group, T.E. EuroQol—A new facility for the measurement of health-related quality of life. Health Policy 1990, 16, 199–208. [Google Scholar]
- Kroenke, K.; Spitzer, R.L.; Williams, J.B.W. The PHQ-9: Validity of a brief depression severity measure. J. Gen. Intern. Med. 2001, 16, 606–613. [Google Scholar] [CrossRef]
- Tluczek, A.; Henriques, J.B.; Brown, R.L. Support for the reliability and validity of a six-item state anxiety scale derived from the state-trait anxiety inventory. J. Nurs. Meas. 2009, 17, 19–28. [Google Scholar] [CrossRef]
- Zenicor Medical Systems AB. Research-Arrhythmia Investigations. Available online: https://zenicor.com/arrhythmia-investigations (accessed on 14 February 2019).
- Hendrikx, T.; Rosenqvist, M.; Sandström, H.; Persson, M.; Hörnsten, R. Identification of paroxysmal, transient arrhythmias: Intermittent registration more efficient than the 24-hour Holter monitoring. Lakartidningen 2015, 112, C6SE. [Google Scholar]
- Marston, H.R.; Freeman, S.; Genoe, R.; Kulcyzki, C.; Musselwhite, C. The Cohesiveness of Technology in Later Life: Findings From the Technology in Later Life (TILL) Project Knowledge Exchange Seminar Series 2017–2018; Stormont: Belfast, UK, 2018. [Google Scholar]
- Waights, V.; Bamidis, P.; Almeida, R. Technologies for care—The imperative for upskilling carers. In Knowledge Exchange Seminar Series 2017–18; Stormont: Belfast, UK, 2018. [Google Scholar]
- Genoe, R.; Kulczycki, C.; Marston, H.; Freeman, S.; Musselwhite, C.; Rutherford, H. E-leisure and older adults: Findings from an international exploratory study. Ther. Recreat. J. 2018, 52, 1–18. [Google Scholar] [CrossRef]
- Marston, H.R.; Kroll, M.; Fink, D.; de Rosario, H.; Gschwind, Y.J. Technology use, adoption and behavior in older adults: Results from the iStoppFalls project. Educ. Gerontol. 2016, 42, 371–387. [Google Scholar] [CrossRef]
- Marston, H.R.; Smith, S.T. Interactive videogame technologies to support independence in the elderly: A narrative review. Games Health J. 2012, 1, 139–152. [Google Scholar] [CrossRef] [PubMed]
- Marston, H.R.; Smith, S.T. Understanding the digital game classification system: A review of the current classification system & its implications for use within games for health. Hum. Fact. Comput. Inform. LNCS 2013, 7946, 314–331. [Google Scholar]
- Mantovani, E.; Bocos, P.C. Are mHealth apps safe? The intended purpose rule, its shortcomings and the regulatory options under the EU medical device framework. In Mobile e-Health. Human–Computer Interaction Series; Marston, H., Freeman, S., Musselwhite, C., Eds.; Springer: Cham, Switzerland, 2017; ISBN 978-3-319-60672-9. [Google Scholar]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA extension for scoping reviews (PRISMA-ScR): Checklist and explanation the PRISMA-ScR statement. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Wiersinga, J. Regulation of medical digital technologies. In Mobile e-Health. Human–Computer Interaction Series; Marston, H., Freeman, S., Musselwhite, C., Eds.; Springer: Cham, Switzerland, 2017; ISBN 978-3-319-60672-9. [Google Scholar]
- Chugh, S.S.; Havmoeller, R.; Narayanan, K.; Singh, D.; Rienstra, M.; Benjamin, E.J.; Gillum, R.F.; Kim, Y.-H.; McAnulty, J.H., Jr.; Zheng, Z.J.; et al. Worldwide epidemiology of atrial fibrillation: A global burden of disease 2010 study. Circulation 2014, 129, 837–847. [Google Scholar] [CrossRef] [PubMed]
| Inclusion | Exclusion |
|---|---|
| Mobile apps (mApps) | Master’s and PhD thesis |
| Electrocardiogram (ECG/EKG) | Conference proceedings |
| Cardiogram | Book Chapters |
| Wearables | Reports |
| Atrial Fibrillation (AF) | Reviews |
| Heart | Pulse monitoring |
| Human | Theoretical papers |
| ECG Wearable Devices/Patches | Athletes |
| Mobile Health (mHealth) | Defibrillators |
| Security/Privacy | Intensive care unit (ICU) or high dependency unit (HDU) |
| Smart Fabric/textiles | WSBN |
| Papers published in Journals | Animals/non-human |
| Commercial technologies | Co-morbidities (i.e., transplant patients) |
| Purpose-built technologies | Newsletters |
| Encryption | Editorials |
| Big Data | PhD, MSc & BSc Thesis |
| Human | |
| Study designs: (randomised control trial (RCT), Exploratory, Cohort, Prospective, Feasibility) |
| Database | Search Term Used | Adaptions |
|---|---|---|
| Association for Computing Machinery (ACM) | (Arrhythmia Atrial Fibrillation ECG EKG Palpitations wearables) AND (-Algorithms -map -sensor -consumer -mathematical -statistical) AND keywords. author. keyword:(Arrhythmia Atrial Fibrillation ECG EKG Palpitations wearables -wavelet -brain -skin -posture -music -grasp -grip -sonic -speculative) AND record Abstract: (Arrhythmia Atrial Fibrillation ECG EKG Palpitations wearables) | Manufacturers’/generic names not recognised. NOT any: Algorithms map sensor consumer mathematical statistical. Keyword NOT: wavelet brain skin posture music grasp grip sonic |
| CINHAL | (TX (“Palpitations” OR “Arrhythmia” OR “Atrial Fibrillation”)) AND (TX “Wearable ECG”) AND (TX “Wearable EKG”) OR (TX (“Wearable technologies” OR Wearable devices)) NOT (TX (“Catheter” OR “Surgery” OR “Ablation” OR “Catheter ablation” OR “Nursing Practice” OR “Gait”)) NOT (TX “Students”) | Manufacturers’/generic names not recognised. NOT “Catheter” OR “Surgery” OR “Ablation” OR “Catheter ablation” OR “Nursing Practice” NOT “Students” |
| Google scholar (wearable device) | ECG EKG Alive OR Cor OR Zoe OR Patch OR Scanadu OR Scout OR Perminova OR CoVa OR necklace OR Kardia OR ECG OR Necklace OR Cardio OR Analytics OR Heal OR Force OR Smart OR Cardio OR Beurer OR ME80 OR Beurer OR PM2 “wearable device” | Excluded patents |
| Google scholar (wearable technology) | ECG EKG Alive OR Cor OR Zoe OR Patch OR Scanadu OR Scout OR Perminova OR CoVa OR necklace OR Kardia OR ECG OR Necklace OR Cardio OR Analytics OR Heal OR Force OR Smart OR Cardio OR Beurer OR ME80 OR Beurer OR PM2 “wearable technology” | Excluded patents |
| PubMed | Palpitations OR Arrhythmia OR Atrial Fibrillation And (ECG) AND (EKG) OR Wearable technologies OR Wearable devices)) AND (Alive Cor OR Zoe Patch OR Scanadu Scout OR Perminova CoVa necklace OR Kardia OR ECG Necklace OR Cardio Analytics OR Heal Force OR Smart Cardio OR Beurer ME80 OR Beurer PM25 OR Prince 180B OR Cardea SOLO OR Spyder Pro OR Spyder Personal OR MiCor A100)) NOT (sport AND algorithms)) | AND NOT sport AND algorithms |
| PubMed MESH | Wearable devices OR Wearable technologies AND (ECG OR EKG) AND (Palpitations OR Arrhythmia OR Atrial Fibrillation) | Manufacturers’/generic names not recognised. AND NOT sport AND algorithms |
| Scopus | Palpitations OR Arrhythmia OR Atrial Fibrillation And {ECG} AND {EKG} OR Wearable* AND techonolo* OR device AND NOT algorithms | Manufacturers’/generic names not used Use wildcard* AND NOT algorithms |
| Scopus | Alive Cor” OR “Zoe Patch” OR “Scanadu Scout” OR “Perminova CoVa Necklace” OR “QardioCore” OR “Kardia” OR “ECG Necklace” OR “Cardio Analytics” OR “Heal Force” OR “Smart Cardio” Or “ChoiceMMed” OR “Beurer ME80” OR “Beurer PM25” OR “Zodore” OR “Prince 180B” OR “Cardea SOLO” OR “Spyder Pro” OR “Spyder Personal” OR “MiCor A100”) | Dropped: “Palpitations” OR “Arrhythmia” OR “Atrial Fibrillation” AND “ECG” OR “EKG” |
| 1st Author Year Country | Objectives | Participants | Study Design | Assessment(s) | Technology | Main Findings |
|---|---|---|---|---|---|---|
| Aronsson et al. [40] 2015 Sweden | To estimate the cost effectiveness of 2 weeks of intermittent screening for asymptomatic atrial fibrillation (AF) in 75/76-year-old individuals. | n = 25,415 Aged 75–76 years Female 55.9% | Observational Cohort study | In total, 30-s recordings taken twice daily, or when symptoms of palpitations for 2 weeks. | Zenicor EKG device | With the use of a decision analytic simulation model, it has been shown that screening for asymptomatic AF in 75/76-year-old individuals is cost effective. |
| Doliwa, Rosenqvist, and Frykman [41] 2012 Sweden | To compare short intermittent heart rhythm recording with or without symptoms with continuous ECG recordings for 30 days, with two registrations of 10 s per day. | n = 22 Aged 46–77 years Females 27% Median age 63 years | Experimental study, randomised controlled blinded trial | Recordings were taken twice daily; once in the morning and once in the evening for a 30-day period. Participants were asked to record when experiencing arrhythmia symptoms (recorded as symptomatic). | Zenicor EKG device | AF episodes were diagnosed in 18 (82%) patients compared with seven (32%) patients using continuous ECG, (p = 0.001. Short-term ECG registrations over extended periods of time seem to be a more sensitive tool, compared with short continuous ECG recordings, for the detection of AF episodes. |
| Boudreau et al. [42] 2017 USA | To determine the validity of eight monitors for Heart Rate (HR) compared with an ECG and seven monitors for Energy Expenditure (EE) compared with a metabolic analyser during graded cycling and resistance exercise. | n = 50 Aged 18–35 years Female n = 28 (56%) Mean age 22.71 ± 2.99 | Experimental comparative study | Session 1: Performed a graded exercise test on a cycle ergometer. Session 2: Performed a graded exercise test of four different strength training exercises on a resistance exercise machine. Repeated 3 days later in the laboratory. Exclusion: Cardiovascular disease or musculoskeletal injury in the last 6 months. | Apple Watch Series 2, Fitbit Blaze, Fitbit Charge 2, Polar H7, Polar A360, Garmin Vivosmart HR, TomTom Touch, and Bose SoundSport Pulse (BSP) headphones | This study revealed that both HR and EE differed among the eight wearable devices during both cycling and resistance exercise and had varying levels of validity when compared with a six-lead ECG and metabolic analyser. It was also observed that HR measures from wearable devices were more accurate at rest and lower exercise intensities than at higher intensities. Among tested devices, HR accuracy, as reflected by intraclass correlation and MAPE values, was highest in the PH7, BSP, and AWS2. The PH7 and AWS2 also proved to provide more accurate caloric estimations than other devices. HR from wearable devices differed at different exercise intensities; EE estimates from wearable devices were inaccurate. |
| Evans et al. [43] 2017 Kenya | To examine the feasibility of using mobile ECG recording technology to detect AF. | n = 50 Mean age 54.3 ± 20.5. Females 66% | Prospective observational study | Of 2-week duration. In a rural community. Health practitioners (physicians, clinical officers, nurse) completed a self-assessment of a 4-item scale relating to ICT access, knowledge/interpretation of results and perception of AF in the community. | AliveCor Kardia Mobile ECG device | ECG tracings of four of the 50 patients who completed the study showed AF (8% AF yield), and none had been previously diagnosed with AF. Using mobile ECG technology in screening for AF in low-resource settings is feasible and can detect a significant proportion of AF cases that will otherwise go undiagnosed. Further study is needed to examine the cost effectiveness of this approach for the detection of AF and its effect on reducing the risk of stroke in developing countries. |
| Haberman et al. [26] 2015 USA | Compare the standard 12-lead ECG to the smartphone ECG in healthy young adults, elite athletes, and cardiology clinic patients. Accuracy for determining baseline ECG intervals and rate and rhythm was assessed. | n = 335 Mean age 35 ± 20 Female 51% | Experimental comparative study | Using an iPhone case or iPad, 30-s lead iECG waveforms were obtained. Standard 12-lead ECGs were acquired immediately after the smartphone tracing was obtained. De-identified ECGs were interpreted by automated algorithms and adjudicated by two board-certified electrophysiologists | AliveCor device (30-s ECG wireless reading). Patients trained over 1–2 min to take their own readings | This study provides evidence that wireless ECG devices can be used on a large scale to detect rate, conduction intervals and AF. Incorporation of automated discrimination, with enhanced smartphone features with notification capability and decision support. Both smartphone and standard ECGs detected atrial rate and rhythm, AV block, and QRS delay with equal accuracy. Sensitivities ranged from 72% (QRS delay) to 94% (atrial fibrillation). Specificities were all above 94% for both modalities. |
| Hickey et al. [44] 2016 USA | The primary aims of the iHEART study are to: (1) document AF using real-time ECG capture; (2) evaluate the impact on AF treatment and Quality-Adjusted Life Years (QALYs); and (3) evaluate the effectiveness of text messaging on AF knowledge and promoting proactive self-management of multiple chronic conditions | n = 300 Aged > 18 years | Study protocol, observational study. Single-centre prospective | ECG reading taken at baseline. Complete all questionnaires at baseline and at 6 months. Questionnaires included the Atrial Fibrillation Knowledge Scale, the Canadian Cardiovascular Society Severity in Atrial Fibrillation scale, the Atrial Fibrillation Effect on Quality of Life, the Control Attitudes Scale-Revised, the Morisky 4-item Self-Report Measure of Medication-Taking Behaviour, the Self-Efficacy for Appropriate Medication Use Scale, the Short Form Health Survey, European Questionnaire 5 Dimensions, the Patient Health Questionnaire, and the State Trait Anxiety Inventory. | iPhone, AliveCor Mobile ECG Kardia app | This will be the first study to investigate the utility of a mobile health intervention in a “real world” setting. We will evaluate the ability of the iHEART intervention to improve the detection and treatment of recurrent atrial fibrillation and assess the intervention’s impact on improving clinical outcomes, quality of life, quality-adjusted life-years and disease-specific knowledge. |
| Halcox et al. [45] 2017 UK | n = 1001, Mean age 72.6 ± 5.4 Females 53.34% | Experimental study | Baseline characteristics. Participant experience survey (completed at the end of the study). Questions included anxiety about their heart rhythm problems, more likely to visit their doctor, or prefer to switch to a study group (responses reported via a 10-point visual analogue scale). iECG patients were asked about ease of use, restriction of activities, anxiety, concern about data security and a general satisfaction with the device (via 5-point Likert scale). Health economics were estimated from the UK National Health Service (NHS) and personal social services, using data from the study activity and relevant costs. | AliveCor Kardia device | Screening with twice-weekly single-lead iECG with remote interpretation in ambulatory patients ≥65 years of age at increased risk of stroke is significantly more likely to identify incident AF than RC over a 12-month period. This approach is also highly acceptable to this group of patients, supporting further evaluation in an appropriately powered, event-driven clinical trial. | |
| McManus et al. [46] 2016 USA | To test whether an enhanced smartphone app for AF detection can discriminate between sinus rhythm (SR), AF, premature atrial contractions (PACs), and premature ventricular contractions (PVCs). | AF—n = 98 65.9 ± 12.2 Male—n = 70 (71.4%) White n = 91(92.9) PAC—n = 15 73.1 ± 5.9 Male—n = 11 (73.3%) White n = 14(93.3) PVC—n = 15 62.8 ± 13.8 Male—n = 9 (60%) White n = 13(86.7) Sinus Rhythm—n = 91 66 ± 11.9 Male—n = 63 (69.2%) White n = 86(94.5) | Experimental study | Analysis of 219 2-min pulse recordings. Usability questionnaire to sub-group of ns = 65 app users. Examined the sensitivity, specificity, and predictive accuracy of the app for AF, PAC, and PVC discrimination from sinus rhythm using the 12-lead EKG or 3-lead telemetry as the gold standard. | PULSE-SMART prototype App used via the iPhone 4S | The smartphone-based app demonstrated excellent sensitivity (0.970), specificity (0.935), and accuracy (0.951) for real-time identification of an irregular pulse during AF. The app also showed good accuracy for PAC (0.955) and PVC discrimination (0.960). The vast majority of surveyed app users (83%) reported that it was “useful” and “not complex” to use. |
| Turakhia et al. 2015a [47] USA | To detect silent AF in asymptomatic patients with known risk factors through screening for AF using continuous ambulatory ECG. | n = 75, Mean age 69 ± 8.0 years. Male only | Observational study, single centre | Records up to 14 days of monitoring on a single vector. Participants press the symptomatic trigger on the device if symptoms presented. Patient diary, detailing symptoms. Baseline characteristics: demographics, medical history, ECG parameters, health behaviours were abstract from patient medical record by two trained investigators. | Zio wearable patch-based device | AF was detected in four subjects (5.3%; AF burden 28–48%). Atrial tachycardia (AT) was present in 67% (≥4 beats), 44% (≥8 beats), and 6.7% (≥60 s) of subjects. The combined diagnostic yield of sustained AT/AF was 11%. In subjects without sustained AT/AF, 11 (16%) had ≥30 supraventricular ectopic complexes per hour Outpatient extended ECG screening for asymptomatic AF is feasible, with AF identified in one in 20 subjects and sustained AT/AF identified in one in nine subjects, respectively. We also found a high prevalence of asymptomatic AT and frequent supraventricular ectopic complexes, which may be relevant to development of AF or stroke. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marston, H.R.; Hadley, R.; Banks, D.; Duro, M.D.C.M. Mobile Self-Monitoring ECG Devices to Diagnose Arrhythmia that Coincide with Palpitations: A Scoping Review. Healthcare 2019, 7, 96. https://doi.org/10.3390/healthcare7030096
Marston HR, Hadley R, Banks D, Duro MDCM. Mobile Self-Monitoring ECG Devices to Diagnose Arrhythmia that Coincide with Palpitations: A Scoping Review. Healthcare. 2019; 7(3):96. https://doi.org/10.3390/healthcare7030096
Chicago/Turabian StyleMarston, Hannah Ramsden, Robin Hadley, Duncan Banks, and María Del Carmen Miranda Duro. 2019. "Mobile Self-Monitoring ECG Devices to Diagnose Arrhythmia that Coincide with Palpitations: A Scoping Review" Healthcare 7, no. 3: 96. https://doi.org/10.3390/healthcare7030096
APA StyleMarston, H. R., Hadley, R., Banks, D., & Duro, M. D. C. M. (2019). Mobile Self-Monitoring ECG Devices to Diagnose Arrhythmia that Coincide with Palpitations: A Scoping Review. Healthcare, 7(3), 96. https://doi.org/10.3390/healthcare7030096






