Modeling-Based Assessment of 3D Printing-Enabled Meniscus Transplantation
Abstract
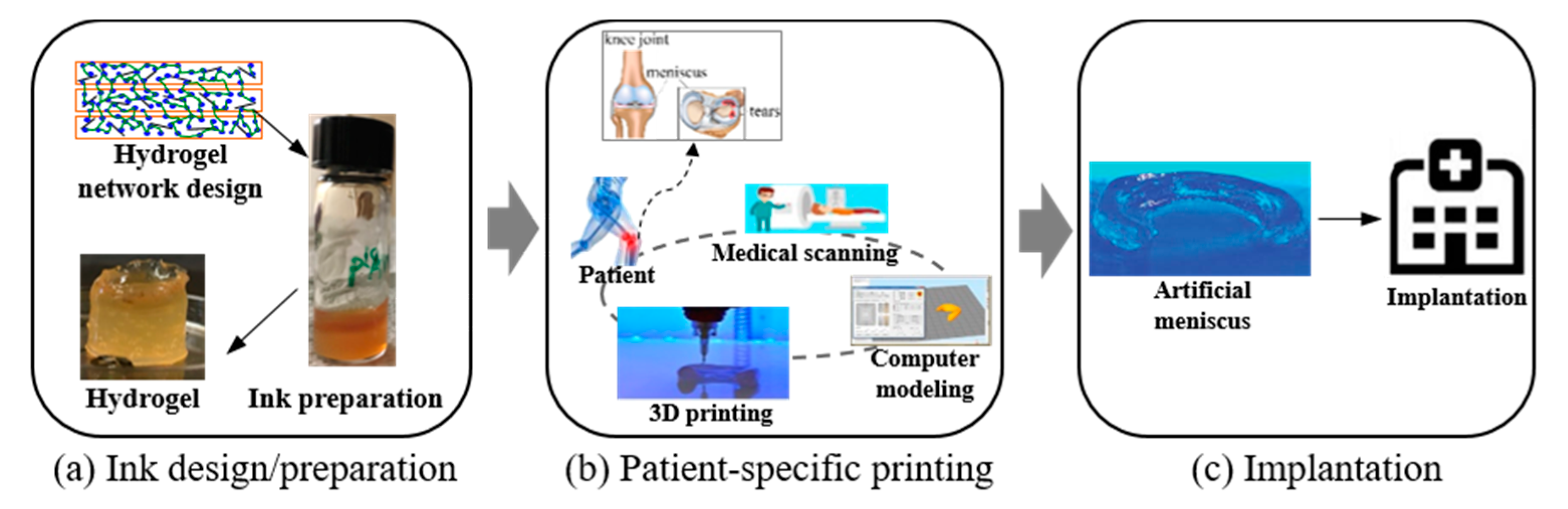
1. Introduction
2. Methods
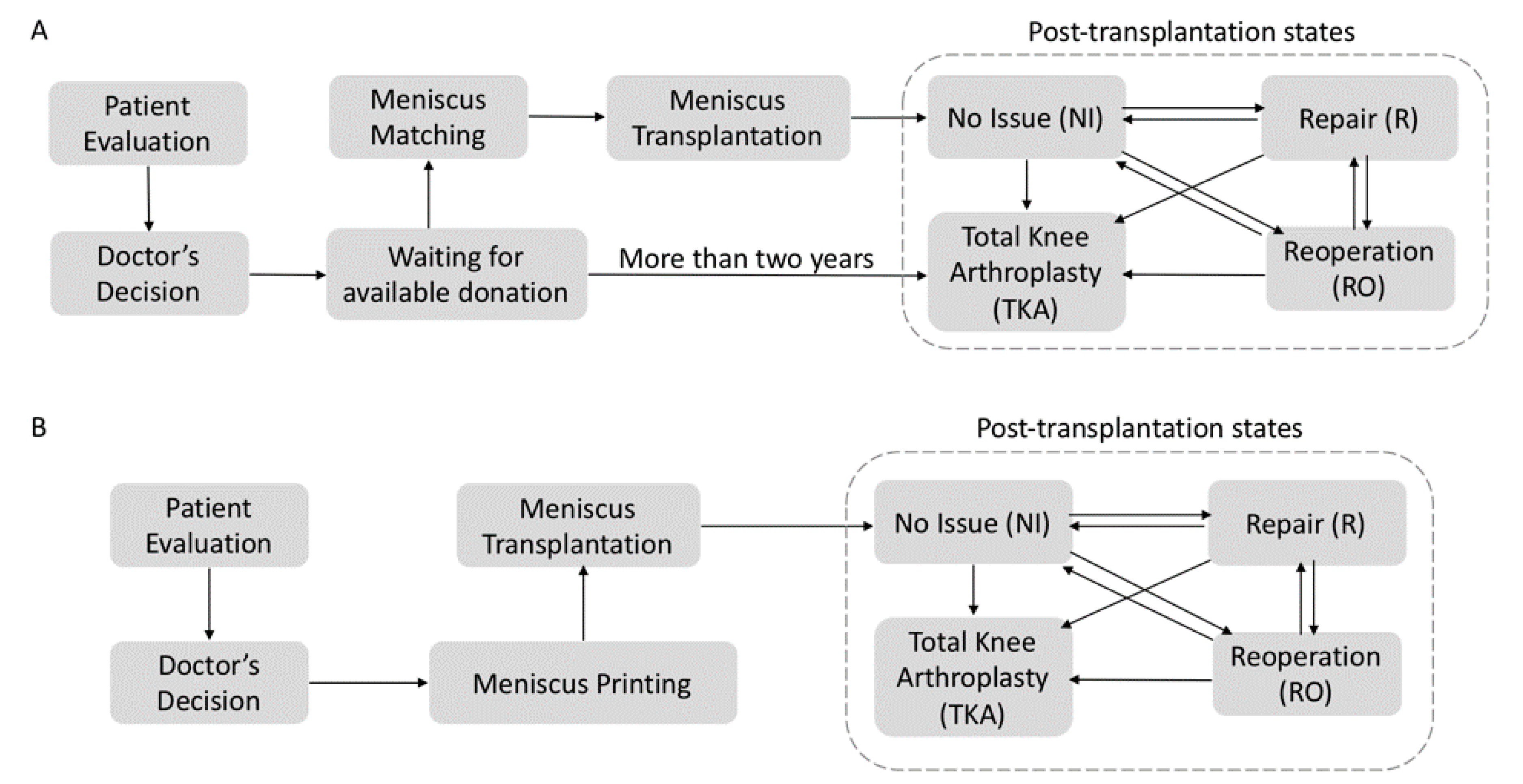
2.1. Model
2.2. Assumptions
- The arrival of patients to the waiting queue follows an exponential distribution with an average interarrival time of 1 week; the arrival of donors follows a mixed exponential distribution with the average interarrival time being a random sample from uniform (1,5). Here, a random interarrival time is used to reflect the uncertainty in donor arrivals that exists in reality [29].
- The queue waiting for allografts in the traditional transplantation model follows the first-come, first-served rule [30]. The capacity of the queue is set to infinity. If a patient stays in the queue for too long (exceeding the threshold of waiting time), it is reasonable to assume that the condition of the meniscus will deteriorate, forcing the patient to have TKA.
- A delay in transplantation caused by other factors, such as acquiring the donated meniscus and transportation in traditional transplantation, or access to a printer and preparation of the 3D-printed meniscus in 3DP-enabled transplantation, are negligible compared to the waiting time for an allograft (months or years) [31], so those factors are ignored in this study.
- The patients have no pre-existing osteoarthritic changes, synovial disease, or inflammatory arthritis. This population comprises the most typical candidates for meniscus allograft and has been considered in related studies (e.g., [32]).
- After the initial transplantation, a patient can enter the intermediate states (R or RO) twice at most; if he/she enters that kind of state for the third time, then he/she will be forced to TKA. This assumption is similar to what is used in existing studies [4,36], which leads to a survival rate of meniscus transplantation in our model close to the ones reported in the literature.
2.3. Model Parameters of Traditional Meniscus Transplantation
2.4. Model Parameters of 3D Printing-Enabled Transplantation
2.5. Simulation Outputs
3. Results
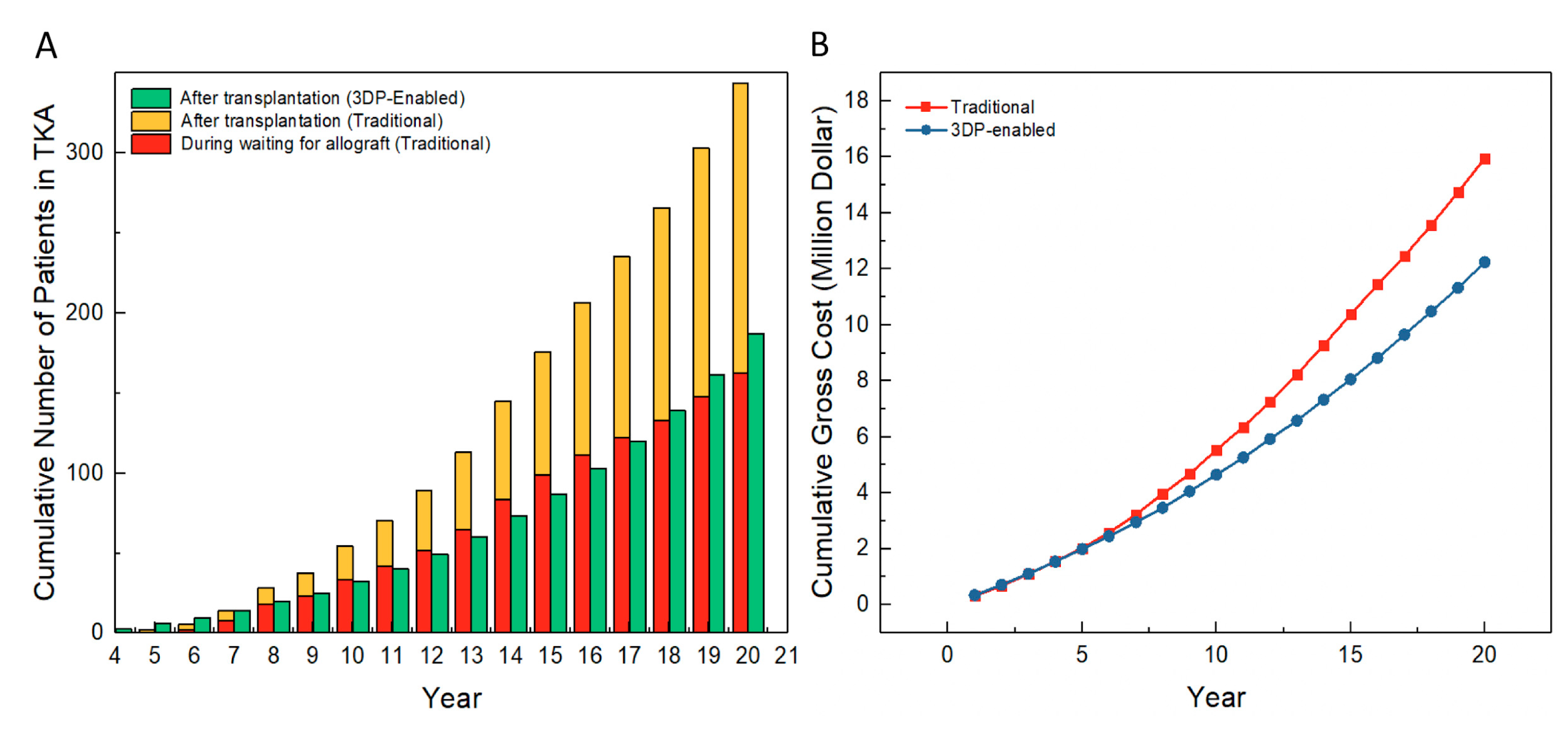
3.1. Number of Patients in TKA
3.2. Cost Analysis
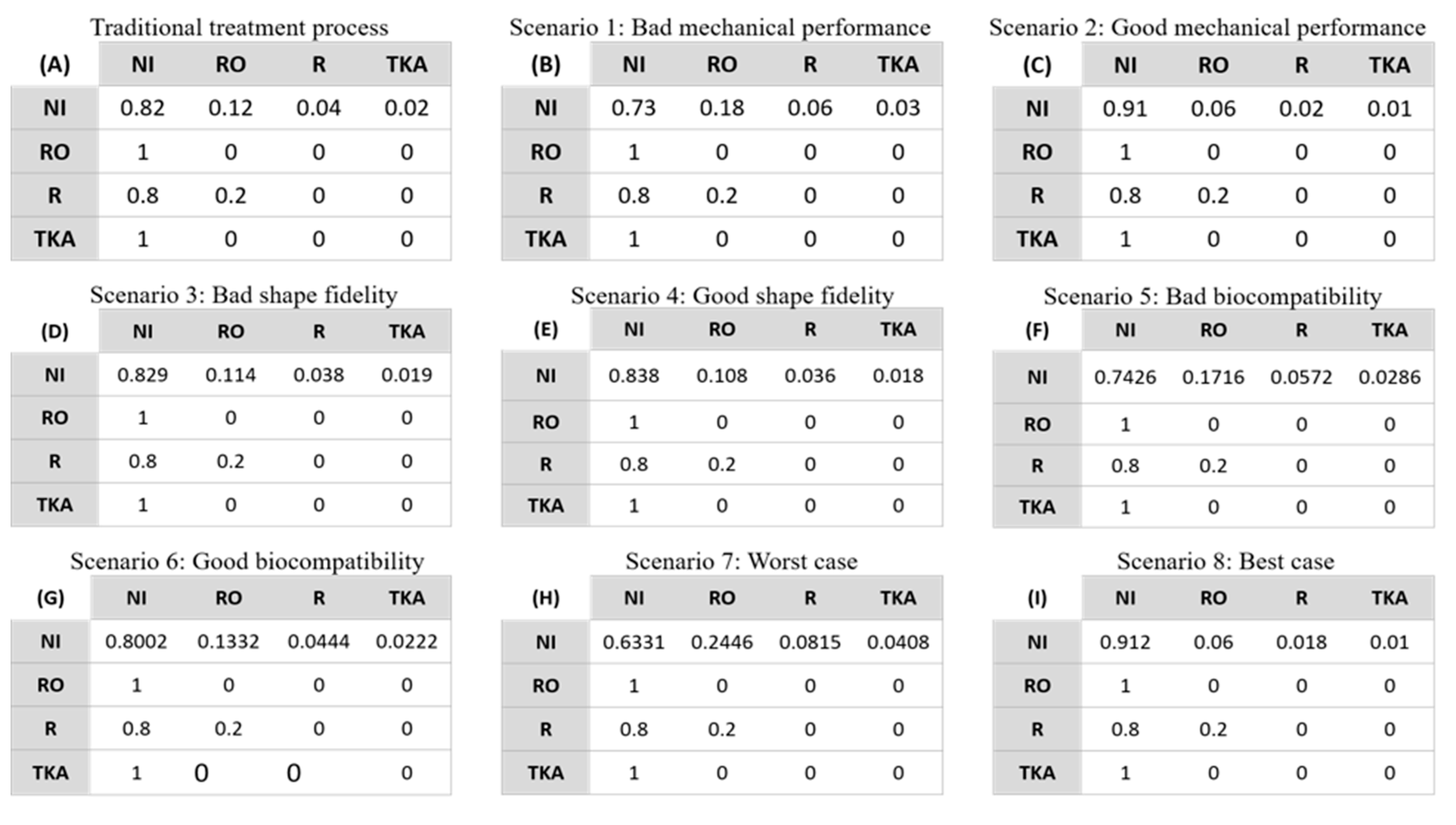
3.3. Sensitivity Analysis
3.3.1. Effect of Mechanical Performance of 3D-Printed Meniscus
3.3.2. Effect of Shape Fidelity of 3D-Printed Meniscus
3.3.3. Effect of Biocompatibility of 3D-Printed Meniscus
3.3.4. Simultaneous Effect of Mechanical Performance, Shape Fidelity and Biocompatibility
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Gelber, P.E.; Verdonk, P.; Getgood, A.M.; Monllau, J.C. Meniscal transplantation: State of the art. J. ISAKOS Jt. Disord. Orthop. Sports Med. 2017, 2, 339–349. [Google Scholar] [CrossRef]
- Kaplan, D.J.; Glait, S.A.; Ryan, W.E., Jr.; Alaia, M.J.; Campbell, K.A.; Strauss, E.J.; Jazrawi, L.M. Meniscal Allograft Transplantation Made Simple: Bridge and Slot Technique. Arthrosc. Tech. 2017, 6, e2129–e2135. [Google Scholar] [CrossRef]
- Lee, S.R.; Kim, J.G.; Nam, S.W. The tips and pitfalls of meniscus allograft transplantation. Knee Surg. Relat. Res. 2012, 24, 137–145. [Google Scholar] [CrossRef]
- Ramme, A.J.; Strauss, E.J.; Jazrawi, L.; Gold, H.T. Cost effectiveness of meniscal allograft for torn discoid lateral meniscus in young women. Phys. Sportsmed. 2016, 44, 278–282. [Google Scholar] [CrossRef]
- Shriram, D.; Praveen Kumar, G.; Cui, F.; Lee, Y.H.D.; Subburaj, K. Evaluating the effects of material properties of artificial meniscal implant in the human knee joint using finite element analysis. Sci. Rep. 2017, 7, 6011. [Google Scholar] [CrossRef] [PubMed]
- McNulty, A.L.; Guilak, F. Mechanobiology of the Meniscus. J. Biomech. 2015, 48, 1469–1478. [Google Scholar] [CrossRef] [PubMed]
- Ventola, C.L. Medical applications for 3D printing: Current and projected uses. Pharm. Ther. 2014, 39, 704–711. [Google Scholar]
- Verdonk, R.; Mendes, J.E.; Monllau, J.C. Meniscal Transplantation; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Korpershoek, J.V.; de Windt, T.S.; Hagmeijer, M.H.; Vonk, L.A.; Saris, D.B.F. Cell-Based Meniscus Repair and Regeneration: At the Brink of Clinical Translation?: A Systematic Review of Preclinical Studies. Orthop. J. Sports Med. 2017, 5, 2325967117690131. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.H.; Rodeo, S.A.; Fortier, L.A.; Lu, C.; Erisken, C.; Mao, J.J. Protein-releasing polymeric scaffolds induce fibrochondrocytic differentiation of endogenous cells for knee meniscus regeneration in sheep. Sci. Transl. Med. 2014, 6, 266ra171. [Google Scholar] [CrossRef]
- Vaquero, J.; Forriol, F. Meniscus tear surgery and meniscus replacement. Muscles Ligaments Tendons J. 2016, 6, 71–89. [Google Scholar] [CrossRef]
- Loh, Q.L.; Choong, C. Three-dimensional scaffolds for tissue engineering applications: Role of porosity and pore size. Tissue Eng. Part B Rev. 2013, 19, 485–502. [Google Scholar] [CrossRef]
- Prasad, A.; Sankar, M.R.; Katiyar, V. State of Art on Solvent Casting Particulate Leaching Method for Orthopedic ScaffoldsFabrication. Mater. Today Proc. 2017, 4, 898–907. [Google Scholar] [CrossRef]
- Thadavirul, N.; Pavasant, P.; Supaphol, P. Development of polycaprolactone porous scaffolds by combining solvent casting, particulate leaching, and polymer leaching techniques for bone tissue engineering. J. Biomed. Mater. Res. Part A 2014, 102, 3379–3392. [Google Scholar] [CrossRef]
- Fereshteh, Z. Freeze-drying technologies for 3D scaffold engineering. In Functional 3D Tissue Engineering Scaffolds; Elsevier: Amsterdam, The Netherlands, 2018; pp. 151–174. [Google Scholar]
- Liu, X.; Ma, P.X. Phase separation, pore structure, and properties of nanofibrous gelatin scaffolds. Biomaterials 2009, 30, 4094–4103. [Google Scholar] [CrossRef]
- Murphy, S.V.; Atala, A. 3D bioprinting of tissues and organs. Nat. Biotechnol. 2014, 32, 773. [Google Scholar] [CrossRef]
- Buma, P.; Ramrattan, N.N.; van Tienen, T.G.; Veth, R.P.H. Tissue engineering of the meniscus. Biomaterials 2004, 25, 1523–1532. [Google Scholar] [CrossRef]
- Ozbolat, I.T.; Yu, Y. Bioprinting toward organ fabrication: Challenges and future trends. IEEE Trans. Biomed. Eng. 2013, 60, 691–699. [Google Scholar] [CrossRef]
- Wei, J.; Wang, J.; Su, S.; Wang, S.; Qiu, J.; Zhang, Z.; Christopher, G.; Ning, F.; Cong, W. 3D printing of an extremely tough hydrogel. RSC Adv. 2015, 5, 81324–81329. [Google Scholar] [CrossRef]
- Zopf, D.A.; Hollister, S.J.; Nelson, M.E.; Ohye, R.G.; Green, G.E. Bioresorbable airway splint created with a three-dimensional printer. N. Engl. J. Med. 2013, 368, 2043–2045. [Google Scholar] [CrossRef]
- Han, H.H.; Shim, J.-H.; Lee, H.; Kim, B.Y.; Lee, J.-S.; Jung, J.W.; Yun, W.-S.; Baek, C.H.; Rhie, J.-W.; Cho, D.-W. Reconstruction of Complex Maxillary Defects Using Patient-specific 3D-printed Biodegradable Scaffolds. Plast. Reconstr. Surg. Glob. Open 2018, 6, e1975. [Google Scholar] [CrossRef]
- Albanna, M.; Binder, K.W.; Murphy, S.V.; Kim, J.; Qasem, S.A.; Zhao, W.; Tan, J.; El-Amin, I.B.; Dice, D.D.; Marco, J. In Situ Bioprinting of Autologous Skin Cells Accelerates Wound Healing of Extensive Excisional Full-Thickness Wounds. Sci. Rep. 2019, 9, 1856. [Google Scholar] [CrossRef] [PubMed]
- Osterhoff, G.; O’Hara, N.N.; D’Cruz, J.; Sprague, S.A.; Bansback, N.; Evaniew, N.; Slobogean, G.P. A Cost-Effectiveness Analysis of Reverse Total Shoulder Arthroplasty versus Hemiarthroplasty for the Management of Complex Proximal Humeral Fractures in the Elderly. Value Health 2017, 20, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Bohensky, M.A.; Pasupathi, K.; Gorelik, A.; Kim, H.; Harrison, J.P.; Liew, D. A Cost-Effectiveness Analysis of Nivolumab Compared with Ipilimumab for the Treatment of BRAF Wild-Type Advanced Melanoma in Australia. Value Health 2016, 19, 1009–1015. [Google Scholar] [CrossRef]
- Graf, K.W., Jr.; Sekiya, J.K.; Wojtys, E.M. Long-term results after combined medial meniscal allograft transplantation and anterior cruciate ligament reconstruction: Minimum 8.5-year follow-up study. Arthrosc. J. Arthrosc. Relat. Surg. 2004, 20, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Stone, K.R.; Adelson, W.S.; Pelsis, J.R.; Walgenbach, A.W.; Turek, T.J. Long-term survival of concurrent meniscus allograft transplantation and repair of the articular cartilage: A prospective two-to 12-year follow-up report. J. Bone Jt. Surg. Br. Vol. 2010, 92, 941–948. [Google Scholar] [CrossRef] [PubMed]
- Van Arkel, E.R.A.; De Boer, H.H. Survival analysis of human meniscal transplantations. J. Bone Jt. Surg. Br. Vol. 2002, 84, 227–231. [Google Scholar] [CrossRef]
- Ghosh, J.K. Introduction to modeling and analysis of stochastic systems, by VG Kulkarni. Int. Stat. Rev. 2012, 80, 487. [Google Scholar] [CrossRef]
- Madwar, S. United States officials propose further retreat from first-come, first-served organ donation. Can. Med. Assoc. 2011, 183, E639–E640. [Google Scholar] [CrossRef] [PubMed]
- Prodromos, C.C.; Joyce, B.T.; Keller, B.L.; Murphy, B.J.; Shi, K. Magnetic resonance imaging measurement of the contralateral normal meniscus is a more accurate method of determining meniscal allograft size than radiographic measurement of the recipient tibial plateau. Arthrosc. J. Arthrosc. Relat. Surg. 2007, 23, 1174–1179. [Google Scholar] [CrossRef]
- Bendich, I.; Rubenstein, W.; Diab, M.M.; Feeley, B. Evaluating meniscus allograft transplant using a cost-effectiveness threshold analysis. Knee 2018, 25, 1171–1180. [Google Scholar] [CrossRef]
- Noyes, F.R.; Heckmann, T.P.; Barber-Westin, S.D. Meniscus repair and transplantation: A comprehensive update. J. Orthop. Sports Phys. Ther. 2012, 42, 274–290. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, I.D.; Moran, C.J.; Potter, H.G.; Warren, R.F.; Rodeo, S.A. Restoration of the meniscus: Form and function. Am. J. Sports Med. 2014, 42, 987–998. [Google Scholar] [CrossRef] [PubMed]
- Linssen, A.M.; Anteunis, L.J.; Joore, M.A. The Cost-Effectiveness of Different Hearing Screening Strategies for 50- to 70-Year-Old Adults: A Markov Model. Value Health 2015, 18, 560–569. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, S. Patient decision making for traditional vs. 3D printing-based meniscus transplantation. In Proceedings of the 2017 Industrial and Systems Engineering Conference, Pittsburgh, PA, USA, 20–23 May 2017. [Google Scholar]
- Nepple, J.J.; Dunn, W.R.; Wright, R.W. Meniscal repair outcomes at greater than five years: A systematic literature review and meta-analysis. J. Bone Jt. Surg. Am. 2012, 94, 2222–2227. [Google Scholar] [CrossRef]
- Mascarenhas, R.; Yanke, A.B.; Frank, R.M.; Butty, D.C.; Cole, B.J. Meniscal allograft transplantation: Preoperative assessment, surgical considerations, and clinical outcomes. J. Knee Surg. 2014, 27, 443–458. [Google Scholar] [CrossRef] [PubMed]
- Yanke, A.B.; Chalmers, P.N.; Frank, R.M.; Friel, N.A.; Karas, V.; Cole, B.J. Clinical outcome of revision meniscal allograft transplantation: Minimum 2-year follow-up. Arthroscopy 2014, 30, 1602–1608. [Google Scholar] [CrossRef] [PubMed]
- McCormick, F.; Harris, J.D.; Abrams, G.D.; Hussey, K.E.; Wilson, H.; Frank, R.; Gupta, A.K.; Bach, B.R., Jr.; Cole, B.J. Survival and reoperation rates after meniscal allograft transplantation: Analysis of failures for 172 consecutive transplants at a minimum 2-year follow-up. Am. J. Sports Med. 2014, 42, 892–897. [Google Scholar] [CrossRef] [PubMed]
- Sgaglione, N.A.; Steadman, J.R.; Shaffer, B.; Miller, M.D.; Fu, F.H. Current concepts in meniscus surgery: Resection to replacement. Arthroscopy 2003, 19, 161–188. [Google Scholar] [CrossRef] [PubMed]
- Chia, H.N.; Wu, B.M. Recent advances in 3D printing of biomaterials. J. Biol. Eng. 2015, 9, 4. [Google Scholar] [CrossRef]
- Knoepfler Lab Stem Cell Blog. How Much Do Stem Cell Treatments Really Cost? Available online: https://ipscell.com/2015/02/stemcelltreatmentcost/ (accessed on 22 February 2015).
- Thermofisher Scientific. 2018. Available online: https://www.thermofisher.com/search/results?query=cell%20culture&focusarea=Search%20All (assessed on 9 May 2019).
- Sigma Aldrich. 2018. Available online: https://www.sigmaaldrich.com/united-states.html (assessed on 9 May 2019).
- VWR. 2018. Available online: https://us.vwr.com/store/product?keyword=TGF-%CE%B2%20I (assessed on 9 May 2019).
- He, Y.; Xue, G.-H.; Fu, J.-Z. Fabrication of low cost soft tissue prostheses with the desktop 3D printer. Sci. Rep. 2014, 4, 6973. [Google Scholar] [CrossRef]
- Barrera Oro, F.; Sikka, R.S.; Wolters, B.; Graver, R.; Boyd, J.L.; Nelson, B.; Swiontkowski, M.F. Autograft versus allograft: An economic cost comparison of anterior cruciate ligament reconstruction. Arthroscopy 2011, 27, 1219–1225. [Google Scholar]
- Liu, R.; Liang, S.; Tang, X.-Z.; Yan, D.; Li, X.; Yu, Z.-Z. Tough and highly stretchable graphene oxide/polyacrylamide nanocomposite hydrogels. J. Mater. Chem. 2012, 22, 14160–14167. [Google Scholar] [CrossRef]
- Gong, J.P.; Katsuyama, Y.; Kurokawa, T.; Osada, Y. Double-Network Hydrogels with Extremely High Mechanical Strength. Adv. Mater. 2003, 15, 1155–1158. [Google Scholar] [CrossRef]
- Billiet, T.; Gevaert, E.; De Schryver, T.; Cornelissen, M.; Dubruel, P. The 3D printing of gelatin methacrylamide cell-laden tissue-engineered constructs with high cell viability. Biomaterials 2014, 35, 49–62. [Google Scholar] [CrossRef]
- Yang, F.; Tadepalli, V.; Wiley, B.J. 3D Printing of a Double Network Hydrogel with a Compression Strength and Elastic Modulus Greater than those of Cartilage. ACS Biomater. Sci. Eng. 2017, 3, 863–869. [Google Scholar] [CrossRef]
- Winkler, R.; Schmidt, F.P.; Haselmann, U.; Fowlkes, J.D.; Lewis, B.B.; Kothleitner, G.; Rack, P.D.; Plank, H. Direct-Write 3D Nanoprinting of Plasmonic Structures. ACS Appl. Mater. Interfaces 2017, 9, 8233–8240. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, B.; Hui, D.; Qiu, J.; Wang, S. 3D bioprinting of soft materials-based regenerative vascular structures and tissues. Compos. Part B Eng. 2017, 123, 279–291. [Google Scholar] [CrossRef]
- Zhang, Y.; Yu, Y.; Ozbolat, I.T. Direct Bioprinting of Vessel-Like Tubular Microfluidic Channels. J. Nanotechnol. Eng. Med. 2013, 4, 020902. [Google Scholar] [CrossRef]
- Goyanes, A.; Scarpa, M.; Kamlow, M.; Gaisford, S.; Basit, A.W.; Orlu, M. Patient acceptability of 3D printed medicines. Int. J. Pharm. 2017, 530, 71–78. [Google Scholar] [CrossRef]

| Parameter | Value | Unit | Source |
|---|---|---|---|
| Cost of no issue (NI) | 0 | $ per person | Zhang et al. [36] |
| Cost of repair (R) | 2760 | $ per person | Ramme et al. [4] |
| Cost of reoperation (RO) | 1770 | $ per person | Ramme et al. [4] |
| Cost of total knee arthroplasty (TKA) | 14,167 | $ per person | Ramme et al. [4] |
| Cost of waiting | 30 | $ per person per week | Assumption |
| Cost of meniscus transplantation | 8875 | $ per person | Ramme et al. [4] |
| State/Transition | Estimated Probability | Probability Used in Simulation | Source |
|---|---|---|---|
| Meniscal repair | 4% | 4% | Ramme et al. [4] |
| Total knee arthroplasty | 0–18% | 2% | Mascarenhas et al. [38]; Yanke et al. [39]; McCormick et al. [40] |
| Reoperation | 2–32% | 12% | McCormick et al. [40] |
| “No issue” | 71–85% | 82% | Sgaglione et al. [41] |
| Successful repair | 70–90% | 80% | Sgaglione et al. [41] |
| Successful reoperation | - | 100% | Ramme et al. [4] |
| Successful total knee arthroplasty | - | 100% | Ramme et al. [4] |
| Repair to reoperation | - | 20% | Ramme et al. [4] |
| Item | Amount | Cost | Source |
|---|---|---|---|
| Stem cell | $1000 | [43] | |
| Cell culture | 500 mL | $213 | [44] |
| Human collagen-II | 22.5 mL | $805.5 | [45] |
| Alginate | 5 g | $0.7 | [45] |
| Polycaprolactone (PCL) | 1 g | $0.4 | [45] |
| TGF-β I | 0.1 mg | $1076.5 | [46] |
| ABS | 63.5 g | $0.8 | He et al. [47] |
| Acetone | 10 mL | $0.1 | He et al. [47] |
| Others (container, beaker, machine, electricity and labor cost etc.) | - | $27 | He et al. [47] |
| Total | - | $3124 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Z.; Wu, Q.; Zeng, L.; Wang, S. Modeling-Based Assessment of 3D Printing-Enabled Meniscus Transplantation. Healthcare 2019, 7, 69. https://doi.org/10.3390/healthcare7020069
Zhang Z, Wu Q, Zeng L, Wang S. Modeling-Based Assessment of 3D Printing-Enabled Meniscus Transplantation. Healthcare. 2019; 7(2):69. https://doi.org/10.3390/healthcare7020069
Chicago/Turabian StyleZhang, Zimeng, Qian Wu, Li Zeng, and Shiren Wang. 2019. "Modeling-Based Assessment of 3D Printing-Enabled Meniscus Transplantation" Healthcare 7, no. 2: 69. https://doi.org/10.3390/healthcare7020069
APA StyleZhang, Z., Wu, Q., Zeng, L., & Wang, S. (2019). Modeling-Based Assessment of 3D Printing-Enabled Meniscus Transplantation. Healthcare, 7(2), 69. https://doi.org/10.3390/healthcare7020069





