Monitoring the Italian Home Palliative Care Services
Abstract
1. Introduction
2. Methods
2.1. Data Collection
2.2. Admission Requirements of the Best Practice Panel
- Stage1
- It satisfied the four Regulatory standards (HPCS status)
- Stage2
- It provided all variables of the minimum dataset
- Stage3
- The scores of all indicators K1–K6 are consistent (within the outer fence) with the distributions evaluated for all HPCSs that made it to Stage 3
- Stage4
- The score of weekly visits per deceased cancer patient is consistent with the mean Intensity Care Coefficient (SR within the range ±2.5) evaluated for the HPCSs that made it to this stage (Best Practice Panel status)
2.3. Process/Outcomes Measures
3. Results
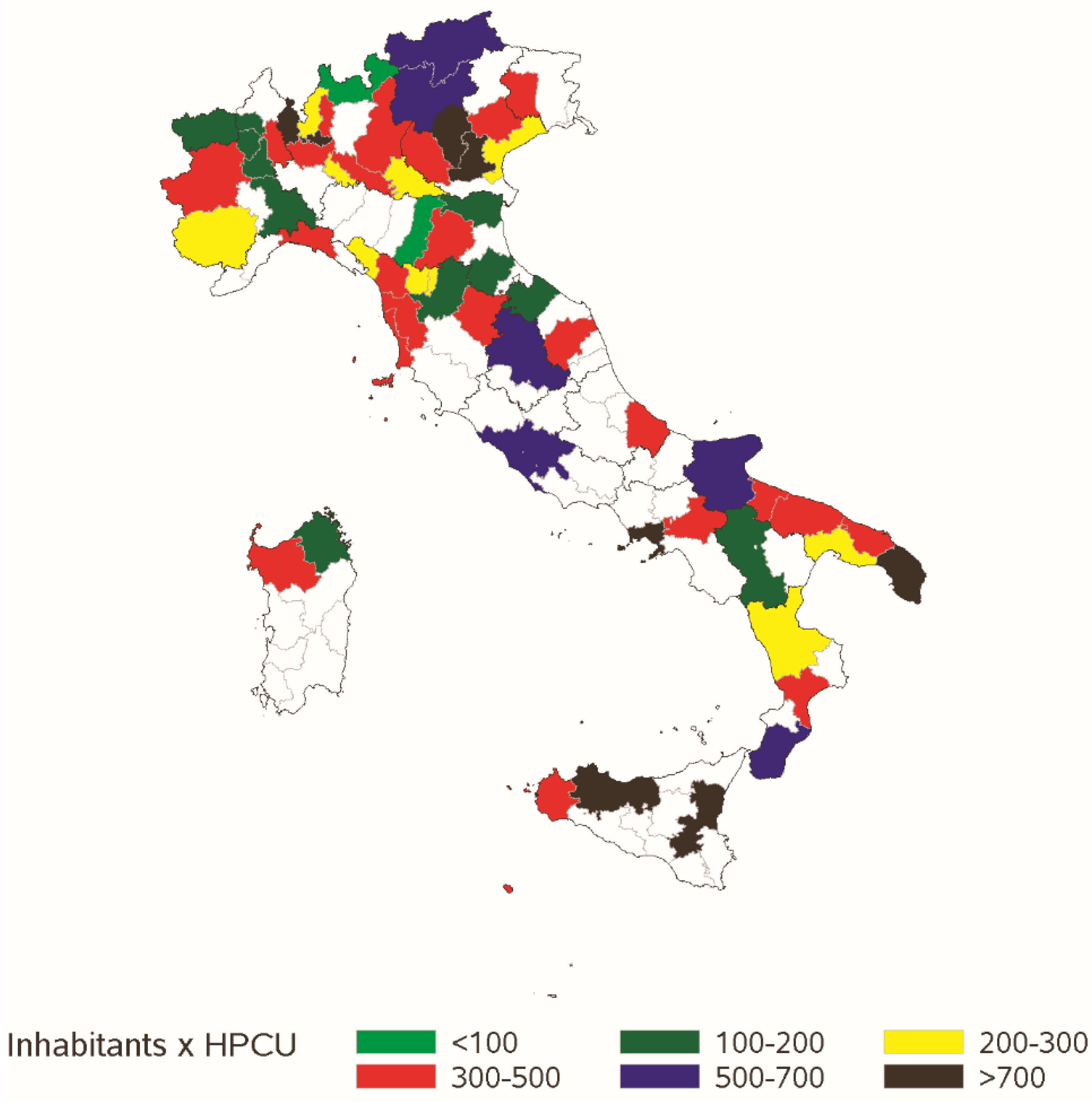
3.1. Registered HPCSs
3.2. Best Practice Panel Admission Process
3.3. Best Practice Panel Process and Outcomes
4. Discussion
Access to Quality At-Home Palliative Care Services
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- ISTAT-Italian Institute of Statistics. Leading Causes of Death in Italy in 2012; Istat Press: Rome, Italy, 2014. [Google Scholar]
- Rosenwax, L.K.; McNamara, B.; Blackmore, A.M.; Holman, C.D.J. Estimating the size of a potential palliative care population. Palliat. Med. 2005, 19, 556–562. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Batiste, X.; Martinez-Munoz, M.; Blay, C.; Jose, E.; Joan, C.C.; Albert, L. Identifying needs and improving palliative care of chronically-ill patients: A community-oriented, population-based, public-health approach. Curr. Opin. Support. Palliat. Care 2012, 6, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Murtagh, F.E.; Bausewein, C.; Verne, J.; Groeneveld, E.I.; Kaloki, Y.E.; Higginson, I.J. How many people need palliative care? A study developing and comparing methods for population-based estimates. Palliat. Med. 2014, 28, 49–58. [Google Scholar] [CrossRef] [PubMed]
- NCP-National Consensus Project for Quality Palliative Care. Clinical Practice Guidelines for quality palliative care, executive summary. J. Palliat. Med. 2004, 7, 611–627. [Google Scholar] [CrossRef] [PubMed]
- Gomes, B.; Calanzani, N.; Curiale, V.; McCrone, F.; Higginson, I.J. Effectiveness and cost-effectiveness of home palliative care services for adults with advanced illness and their caregivers. Cochrane Database Syst. Rev. 2013, 6, CD007760. [Google Scholar] [CrossRef] [PubMed]
- Shepperd, S.; Bee, W.; Straus, S.E. Hospital at home: Home-based end of life care. Cochrane Database Syst. Rev. 2011, 6, CD009231. [Google Scholar]
- Gómez-Batiste, X.; Porta-Sales, J.; Espinosa-Rojas, J.; Pascual-López, A.; Tuca, A.; Rodriguez, J. Effectiveness of palliative care services in symptom control of patients with advanced terminal cancer: A Spanish, multicenter, prospective, quasiexperimental, prepost study. J. Pain Symptom Manag. 2010, 40, 652–660. [Google Scholar] [CrossRef]
- Zimmermann, C.; Riechelmann, R.; Krzyzanowska, M.; Rodin, G.; Tannock, I. Effectiveness of specialized palliative care: A systematic review. JAMA 2008, 299, 1698–1709. [Google Scholar] [CrossRef]
- Lunder, U.; Sauter, S.; Fürst, C.F. Evidence-based palliative care: Beliefs and evidence for changing practice. Palliat. Med. 2004, 18, 265–266. [Google Scholar] [CrossRef]
- National Law. Disposizioni per Garantire L’accesso alle cure Palliative e alla Terapia del Dolore; GU della Repubblica Italiana, Italian Ministry Press: Rome, Italy, 2010. [Google Scholar]
- Tomlinson, S. National Action Planning Workshop on End-of-Life Care; Health Canada Secretariat on Palliative and End-of-Life Care: Winnipeg, MB, Canada, 2002. [Google Scholar]
- Council of Europe. Recommendation Rec 24 of the Committee of Ministers to Member States on the Organization of Palliative Care. 2003. Available online: www.coe.int/t/dg3/health/Source/Rec(2003)24_en.pdf (accessed on 8 September 2009).
- Department of Health. End of Life Care Strategy: Promoting High Quality Care for All Adults at the End of Life; DH: London, 2008. Available online: www.dh.gov.uk/en/Publicationsandstatistics/Publications/PublicationsPolicyAndGuidance/ DH_086277 (accessed on 21 September 2009).
- Palliative Care Australia, Australia. 2014. Available online: www.palliativecare.org.au/ (accessed on 18 July 2015).
- Centeno, C.; Clark, D.; Lynch, T.; Racafort, J.; Praill, D.; De Lima, L.; Greenwood, A.; Flores, L.A.; Brasch, S.; Giordano, A.; et al. EAPC Task Force: Facts and indicators on palliative care development in 52 countries of the WHO European region: Results of an EAPC Task Force. Palliat. Med. 2007, 21, 463–471. [Google Scholar] [CrossRef]
- Centeno, C.; Pons-Izquierdo, J.J.; Lynch, T.; Donea, O.; Rocafort, J.; Clark, D. EAPC Atlas of Palliative Care in Europe 2013; EAPC (European Association for Palliative Care): Milano, Italy, 2013; 385p. [Google Scholar]
- Worldwide Palliative Care Alliance. Global Atlas of Palliative Care at the End of Life. 2014. Available online: www.who.int/nmh/Global_Atlas_of_Palliative_Care.pdf (accessed on 15 November 2015).
- Economist. Quality of Death Index. 2015. Available online: www.economistinsights.com/healthcare/analysis/quality–death–index–2015 (accessed on 12 November 2015).
- Wright, M.; Wood, J.; Lynch, T.; Clark, D. Mapping levels of palliative care development: A global view. J. Pain Symptom Manag. 2008, 35, 469–485. [Google Scholar] [CrossRef]
- Lynch, T.; Connor, S.; Clark, D. Mapping levels of palliative care development: A global update. J. Pain Symptom Manag. 2013, 45, 1094–1106. [Google Scholar] [CrossRef] [PubMed]
- Clark, D.; Wright, M. The international observatory on end of life care: A global view of palliative care development. J. Pain Symptom Manag. 2007, 33, 542–546. [Google Scholar] [CrossRef] [PubMed]
- Clark, D. The International Observatory on End of Life Care: A new initiative to support palliative care development around the world. J. Pain Palliat. Care Pharmacother. 2003, 17, 231–238. [Google Scholar] [CrossRef]
- Lilford, R.; Mohammed, M.A.; Spiegelhalter, D.J.; Thomson, R. Use and misuse of process and outcome data in managing performance of acute medical care: Avoiding institutional stigma. Lancet 2004, 363, 1147–1154. [Google Scholar] [CrossRef]
- Bradley, E.H.; Pallas, S.; Bashyal, C.; Berman, P.; Curry, L. Developing Strategies for Improving Health Care Delivery; The International Bank for Reconstruction and Development, The World Bank: Washington, DC, USA, 2010. [Google Scholar]
- World Health Organization. Global Strategy on People-Centred and Integrated Health Services WHO 2015. Available online: www.who.int/servicedeliverysafety/en (accessed on 12 November 2015).
- National Institute for Clinical Excellence. End of Life Care for Adults; National Institute for Clinical Excellence: London, UK, 2011. [Google Scholar]
- Dawson, R. How significant is a boxplot outlier? J. Stat. Educ. 2011, 19, 1–13. [Google Scholar] [CrossRef]
- Scaccabarozzi, G.; Lovaglio, P.G.; Limonta, F.; Locatelli, W. La Remunerazione Delle Attività Sanitarie: Le Attività di Assistenza Domiciliare; Falcitelli, N., Langiano, T., Eds.; La Remunerazione delle Attività sanitarie, Fondazione Smith-Kline: Il Mulino, NY, USA, 2006; pp. 89–118. [Google Scholar]
- Scaccabarozzi, G.; Lovaglio, P.G.; Limonta, F.; Colombo, C. Caratterizzazione e valorizzazione dei percorsi di cura in Home care: I DRG domiciliari. Tendenze Nuove 2005, 3, 335–350. [Google Scholar]
- Lovaglio, P.G.; Scaccabarozzi, G. Definizione di Sistemi Remunerativi in Home Care. Italy J. Appl. Stat. 2006, 19, 1–19. [Google Scholar]
- Fox, J. Applied Regression Analysis and Generalized Linear Models; Sage: Thousand Oaks, CA, USA, 2008. [Google Scholar]
- Italian Ministry of Health. Report to the Italian Parliament on the Implementation of Law n. 38, on 15th of March 2010 on Measures to Ensure Access to Palliative Care and Pain Therapy; Italian Ministry of Health: Rome, Italy, 2010.
- National Report on Patient Outcome in Palliative Care in Australia. January–June 2015. Available online: http://ahsri.uow.edu.au/content/groups/public/@web/@chsd/@pcoc/documents/doc/uow205078.pdf (accessed on 8 February 2016).
- National Survey of Patient Activity Data for Specialist Palliative Care Sevices, MDS Full Report for the Year 2013–2014. Available online: http://www.ncpc.org.uk/sites/default/files/MDS-2013-14-Summary-report-and-all-settings.pdf (accessed on 8 February 2016).
- Schenker, Y.; Arnold, R. The Next Era of Palliative Care. JAMA 2015, 314, 1565–1566. [Google Scholar] [CrossRef] [PubMed]
- Dzingina, M.D.; Higginson, I.J. Public health and palliative care in 2015. Clin. Geriatr. Med. 2015, 31, 253–263. [Google Scholar] [CrossRef]
- Zheng, L.; Finucane, A.; Oxenham, D. How good is primary care at identifying patients who need palliative care? A mixed methods study. Eur. J. Palliat. Care 2013, 20, 216–222. [Google Scholar]
- Cochrane, E.; Colville, E.; Conway, R. Addressing the needs of patients with advanced non-malignant disease in a hospice day care setting. Int. J. Palliat. Nurs. 2008, 14, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Kelley, A.S.; Morrison, R.S. Palliative Care for the Seriously Ill. N. Engl. J. Med. 2015, 373, 747–755. [Google Scholar] [CrossRef] [PubMed]
- Grol, R.; Wensing, M.; Eccles, M.P. Improving Patient Care: The Implementation of Change in Clinical Practice; Elsevier: Edinburgh, Scotland, 2005. [Google Scholar]
| Stage 1: Regulatory Standards for HPCS Status | Label | %YES | |||
| The HPCS is a structure identified within the organizational plan (simple or complex, as required by law CCNL 1998/2001, art. 27) | I1 | 86% | |||
| The HPCS has a team of full-time palliative care Physicians (75% of the work hours were exclusively dedicated to PC throughout the year) | I3 | 91% | |||
| The HPCS has a team of full-time palliative care Nurses (75% of the work hours were exclusively dedicated to PC throughout the year) | I4 | 92% | |||
| The HPCS adopts the Integrated Individual Care Plan (PAI) to assess the needs, to plan and implement interventions and to evaluate the outcomes of the care provided | I6 | 99% | |||
| Stage 2: Minimum dataset in the Business Year 2013 | Label | N | Total | Mean | Median |
| Assisted patients | E1 | 118 | 40,955 | 347.0 | 236.5 |
| Cancer patients | E2 | 117 | 38,384 | 328.7 | 213.0 |
| Deceased cancer patients | Y2 | 117 | 29,008 | 247.9 | 176.0 |
| Days with at least one home visit (deceased cancer patients) | E8_1 | 108 | 722,744 | 6692.7 | 3683.5 |
| Duration of care, in days (deceased cancer patients) | E8_2 | 108 | 1,679,809 | 15553.7 | 8040.5 |
| Home visits by PC physicians (deceased cancer patients) | E16_1 | 108 | 722,744 | 6692.0 | 3683.5 |
| Home visits by PC nurses (deceased cancer patients) | E16_2 | 104 | 419,201 | 4030.7 | 2299.0 |
| PC physicians active in the HPCS | PCP | 118 | 2593 | 21.9 | 20.0 |
| Yearly work hours of the PC physicians active in the HPCS | W_PCP | 118 | 21,044 | 178.3 | 104.5 |
| PC nurses active in the HPCS | PCN | 118 | 2662 | 22.5 | 20.0 |
| Yearly work hours of the PC nurses active in the HPCS | W_PCN | 118 | 28,133 | 238.4 | 162.0 |
| Main Structure Data | |||||
| Type of organization and structure of the HPCS’s organizational plan | Public 59% (36% Local Health Agencies, 23% hospitals), Private 41% (35% non-profit, 6% private hospitals) | ||||
| Type of PC provided | Specialized PC: 97% (Primary PC too: 65%) | ||||
| The HPCS is an organization that also provides palliative care in other settings (multiple answers) | Hospital and residential facilities (84%), hospices (40%), outpatient settings (58%) | ||||
| Presence of a local Palliative Care network (formalized partnership protocols in the same territorial area with): multiple answers | Local hospitals and hospices (90%), General Practitioners and Pediatricians (64%), informally with GPs for the implementation of an Individual Care Plan (80%) | ||||
| The HPCS provides pediatric care | 76% | ||||
| Formalized support of Non-Profit Organizations | 69% | ||||
| Home Beds Equivalent (total days of care in the year/number of days in the year) | 1–10: 12%, 11–20: 17.8%, 21–30: 7.6%, 31–40: 14.4%, 41–60: 13.6%, 61–100: 15.3%, ≥100: 19.4% (≥200: 7.6%) | ||||
| Continuity of the Care | |||||
| Is the continuity of care guaranteed on weekends and nights? | Not by | Only phone by | Guaranteed by | ||
| Nurse—Doctor | Nurse—Doctor | Nurse—Doctor | |||
| Night: | 69.5%—33.1% | 6.8%—20.3% | 23.7%—46.6% | ||
| Saturday noon: | 8.5%—20.3% | 5.9%—14.4% | 85.6%—65.2% | ||
| Sunday morning: | 8.5%—20.3% | 1.7%—13.6% | 89.8%—66.1% | ||
| Sunday noon: | 14.4%—15.3% | 5.9%—13.6% | 79.6%—71.2% | ||
| Main Process Data | |||||
| Meetings, Training and staff supervision of the HPCSs: 76% have weekly meetings to discuss the Individual Care Plan, 12% depend on prognosis, 12% once a month. 93% organize specific training to the medical and nursing staff, 83% guarantee periodic psychological support to the team. | |||||
| Clinical care procedures of the HPCSs: 93% adopt procedures to evaluate patients’ needs and to modify the Individual Care Plan. 57% rely on a structured interview with families before assisting patients, 40% are not structured and 3% have no interviews. 13% rely on a structured interview directly with the patient, 69% are not structured and 19% have no interviews. HPCSs use standardized scales to evaluate pain (99%, 39% pediatric care), physical symptoms (81%), quality of life (64%, 3% pediatric care) and psychological symptoms (30% patient, 16% family). Patients and families’ non-clinical needs: HPCSs guarantee psychological support (87%), spiritual assistance (38%), social/economic-related support (38%). At the near death stage, HPCSs adopt standardized tools to evaluate patients’ needs (91%), to communicate with the families (88%), to evaluate the symptoms (92%), to provide support (84%). 79% collaborate with an ethical committee, 32% adopts structured procedures for ethical conflicts. 8% does not supply drugs (opioids) at home. | |||||
| Informative systems (ITC) and Quality initiatives: 96% use ICT to record the activity data, 69% use ICT to record and monitor the activity data, 86% use patient satisfaction questionnaires, 80% use risk management procedures, 79% use audit/feedback reports to continuously improve. | |||||
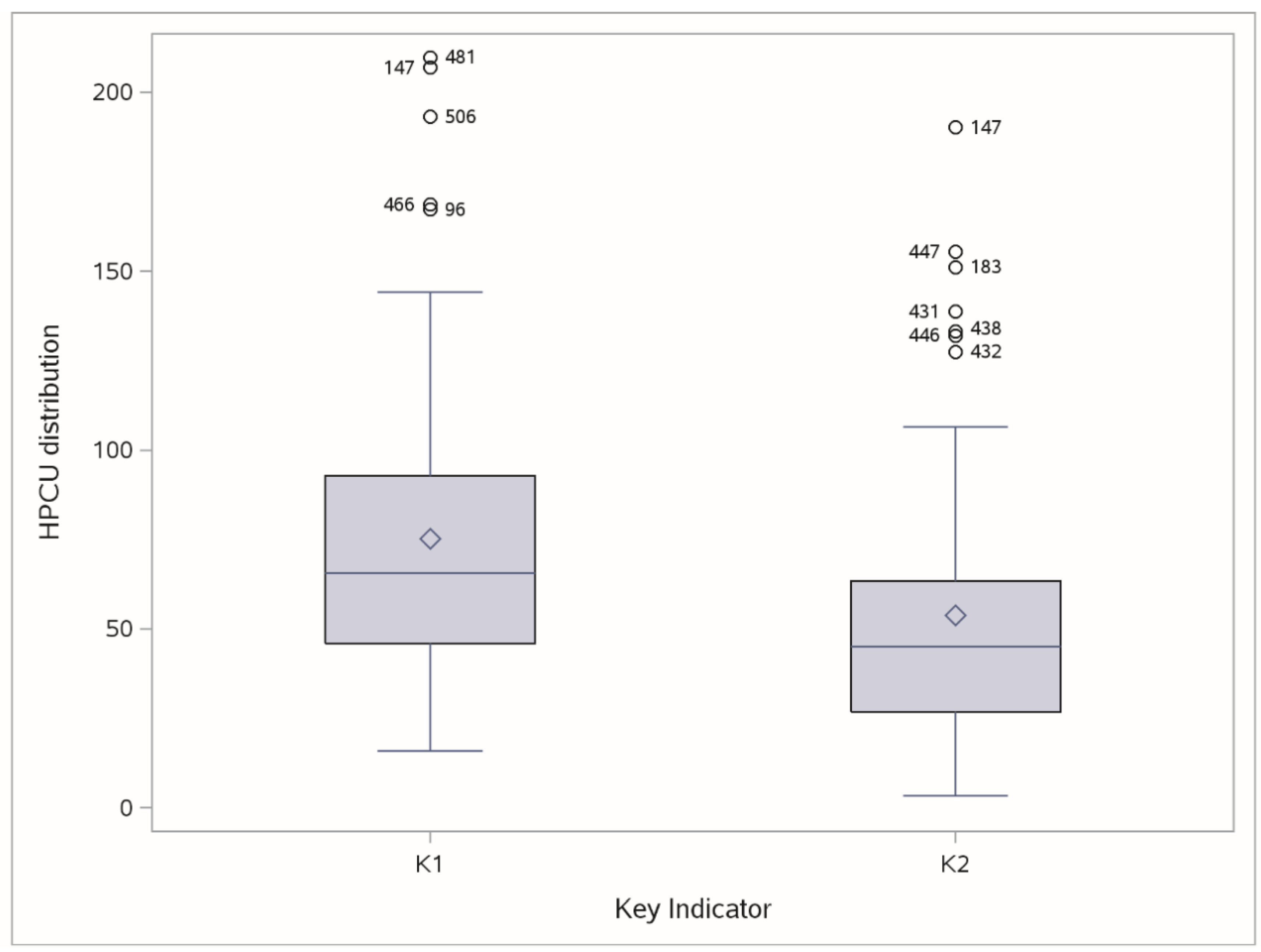
| Stage 3: Key Indicators | Label | Formula | Outlier Check |
| Assisted cancer patients × FTPCP | K1 | E2/FTPCP a | Boxplot, outer fence |
| Assisted cancer patients × FTPCN | K2 | E2/FTPCN b | Boxplot, outer fence |
| FTPCP home visits × 100 deceased cancer patients | K3 | [(E16_1/FTPCP)/Y2 c] × 100 | Boxplot, outer fence |
| FTPCN home visits × 100 deceased cancer patients | K4 | [(E16_2/FTPCN)/Y2 c] × 100 | Boxplot, outer fence |
| Weekly PC Physician visits × deceased cancer patient | K5 | (E16_1/Y2)/TW d | Boxplot, outer fence |
| Weekly PC Nurse visits × deceased cancer patient | K6 | (E16_2/Y2)/TW d | Boxplot, outer fence |
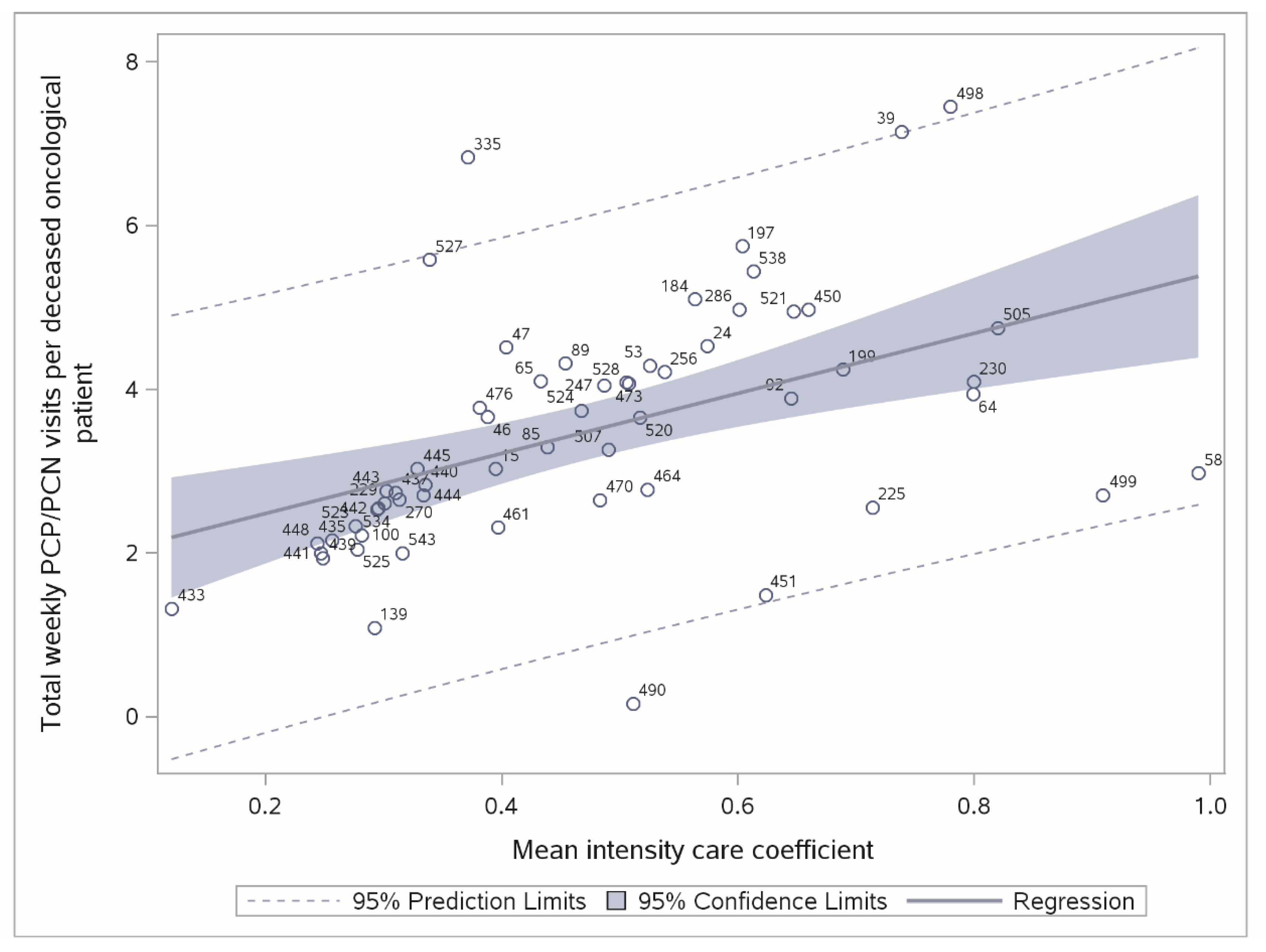
| Stage 4: Relationship to Monitor | Label | Statistical Analysis | Outlier Check |
| Weekly visits x deceased cancer patient (Y = K5 + K6) and mean intensity care coefficient (X = E8_1/ E8_2) c | Y − X | Y vs. predicted value (Polynomial regression) | Prediction bands, studentized residuals |
| Admission Process | HPCS that Passed | ||
|---|---|---|---|
| Stage | Label | N | % of 118 |
| Stage 1 | Regulatory status of the HPCSs | 95 | 80.6% |
| Stage 2 | Provision of the Minimum dataset | 83 | 70.3% |
| Stage 3 | Reliable indicators (K1–K6 no outliers) | 58 | 49.2% |
| Stage 4 | Reliable empirical ratio (Y − X) | 56 | 47.5% |
| Activity Data and Main Indicators | N | Min | Mean | Median | Max |
|---|---|---|---|---|---|
| Home Beds Equivalent Class | 56 | 11–20 | na | 31–40 | ≥200 |
| Assisted patients (N = 19,866) | 56 | 27.0 | 354.6 | 275.5 | 2141.0 |
| % pediatric patients per patients assisted | 53 | 0.0 | 0.4 | 0.0 | 3.7 |
| Cancer patients (N = 18,858) | 56 | 27.0 | 336.8 | 263.0 | 2054.0 |
| % cancer patients per patients assisted | 56 | 38.6 | 94.9 | 100.0 | 100.0 |
| Deceased cancer patients (N = 15,512) | 56 | 14.0 | 259.1 | 210.0 | 1591.0 |
| % deceased cancer patients per cancer patients | 56 | 25.2 | 77.4 | 82.9 | 100.0 |
| Non-cancer patients (N = 1008) | 56 | 0.0 | 17.9 | 0.0 | 208.0 |
| Deceased non-cancer patients (N = 623) | 54 | 0.0 | 11.5 | 0.0 | 184.0 |
| FTPCPs × team | 56 | 0.8 | 3.7 | 3.0 | 12.0 |
| FTPCNs × team | 56 | 1.7 | 5.6 | 4.6 | 24.8 |
| Patients assisted × FTPCP | 56 | 17.1 | 113.8 | 81.0 | 433.0 |
| Patients assisted × FTPCN | 56 | 11.4 | 73.4 | 57.6 | 190.4 |
| Cancer patients × FTPCP | 56 | 17.1 | 103.0 | 77.2 | 337.3 |
| Cancer patients × FTPCN | 56 | 7.6 | 69.5 | 55.0 | 123.5 |
| Deceased cancer patients × FTPCP | 56 | 8.9 | 83.3 | 58.5 | 300.3 |
| Deceased cancer patients × FTPCN | 56 | 5.6 | 53.8 | 41.0 | 115.7 |
| Duration of care in days (deceased cancer patients) | 56 | 17.0 | 60.7 | 49.8 | 126.3 |
| Mean Intensity care coefficient (deceased cancer patients) | 56 | 0.12 | 0.48 | 0.46 | 0.99 |
| Home visits × FTPCP per patient assisted | 55 | 0.0 | 3.4 | 2.7 | 8.9 |
| Home visits × FTPCN per patient assisted | 55 | 0.0 | 4.1 | 3.3 | 13.4 |
| Home visits × FTPCP per deceased cancer patient | 56 | 0.3 | 3.7 | 2.9 | 12.1 |
| Home visits × FTPCN per deceased cancer patient | 56 | 0.8 | 4.0 | 3.5 | 10.1 |
| Weekly visits at home of FTPCPs × deceased cancer patient | 56 | 0.2 | 1.3 | 1.3 | 3.0 |
| Weekly visits at home of FTPCNs × deceased cancer patient | 56 | 0.5 | 2.2 | 2.1 | 5.8 |
| Promptness of take in charge (deceased cancer patients) | |||||
| ≤1 day from the report | 51 | 0.0 | 24.2 | 1.7 | 100.0 |
| ≤2 days from the report | 51 | 0.0 | 17.3 | 6.9 | 100.0 |
| ≤3 days from the report | 51 | 0.0 | 43.0 | 17.3 | 100.0 |
| >3 days from the report | 51 | 0.0 | 15.6 | 1.1 | 100.0 |
| Place of death (deceased cancer patients) | |||||
| Home | 55 | 33.8 | 74.4 | 78.8 | 100.0 |
| Hospice | 55 | 0.0 | 14.2 | 11.1 | 51.5 |
| Hospital | 55 | 0.0 | 10.7 | 9.1 | 37.9 |
| Nursing homes | 55 | 0.0 | 0.7 | 0.0 | 10.1 |
| Place of death (deceased non-cancer patients) | |||||
| Home | 22 | 25.0 | 79.6 | 87.2 | 100.0 |
| Hospice | 22 | 0.0 | 7.6 | 0.0 | 40.0 |
| Hospital | 22 | 0.0 | 10.1 | 2.9 | 75.0 |
| Nursing homes | 22 | 0.0 | 2.7 | 0.0 | 33.3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scaccabarozzi, G.; Lovaglio, P.G.; Limonta, F.; Peruselli, C.; Bellentani, M.; Crippa, M. Monitoring the Italian Home Palliative Care Services. Healthcare 2019, 7, 4. https://doi.org/10.3390/healthcare7010004
Scaccabarozzi G, Lovaglio PG, Limonta F, Peruselli C, Bellentani M, Crippa M. Monitoring the Italian Home Palliative Care Services. Healthcare. 2019; 7(1):4. https://doi.org/10.3390/healthcare7010004
Chicago/Turabian StyleScaccabarozzi, Gianlorenzo, Pietro Giorgio Lovaglio, Fabrizio Limonta, Carlo Peruselli, Mariadonata Bellentani, and Matteo Crippa. 2019. "Monitoring the Italian Home Palliative Care Services" Healthcare 7, no. 1: 4. https://doi.org/10.3390/healthcare7010004
APA StyleScaccabarozzi, G., Lovaglio, P. G., Limonta, F., Peruselli, C., Bellentani, M., & Crippa, M. (2019). Monitoring the Italian Home Palliative Care Services. Healthcare, 7(1), 4. https://doi.org/10.3390/healthcare7010004





