Connecting the Mind–Body Split: Understanding the Relationship between Symptoms and Emotional Well-Being in Chronic Pain and Functional Gastrointestinal Disorders
Abstract
1. Introduction
2. Definitions
2.1. Prevalence
2.2. Living with the Symptoms
3. Connections between Mind and Body
3.1. Evidence within the Context of FGID
3.2. Evidence within the Context of Chronic Pain
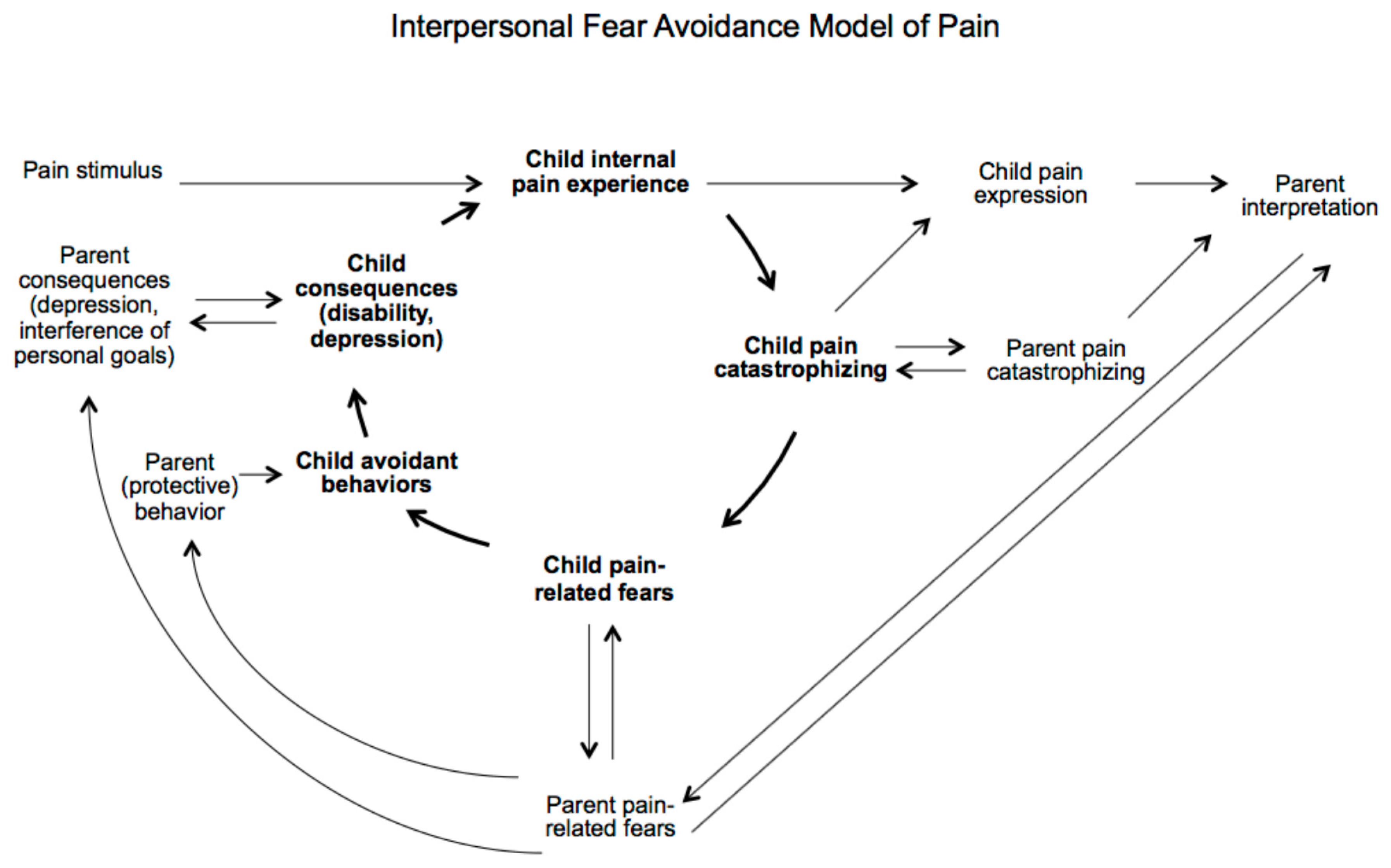
4. Reciprocal Relation between Parents and Children
5. Closing the Mind and Body Split
6. Case Example
7. Promoting the Mind and Body Working Together
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pinquart, M.; Shen, Y. Behavior problems in children and adolescents with chronic physical illness: A meta-analysis. J. Pediatr. Psychol. 2011, 36, 1003–1016. [Google Scholar] [CrossRef] [PubMed]
- Pinquart, M.; Shen, Y. Anxiety in children and adolescents with chronic physical illnesses: A meta-analysis. Acta Paediatr. 2011, 100, 1069–1076. [Google Scholar] [CrossRef] [PubMed]
- Lambert, V.; Keogh, D. Striving to live a normal life: A review of children and young people’s experience of feeling different when living with a longterm condition. J. Pediatr. Nurs. 2015, 30, 63–77. [Google Scholar] [CrossRef] [PubMed]
- Luyckx, K.; Seiffge-Krenke, I.; Schwartz, S.J.; Goossens, L.; Weets, I.; Hendrieckx, C.; Groven, C. Identity development, coping, and adjustment in emerging adults with a chronic illness: The sample case of type 1 diabetes. J. Adolesc. Health 2008, 43, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Pinquart, M.; Teubert, D. Academic, physical, and social functioning of children and adolescents with chronic physical illness: A meta-analysis. J. Pediatr. Psychol. 2011, 37, 376–389. [Google Scholar] [CrossRef] [PubMed]
- Christie, D.; Viner, R. ABC of adolescence: Adolescent development. BMJ 2005, 330, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Friedrichsdorf, S.J.; Giordano, J.; Desai Dakoji, K.; Warmuth, A.; Daughtry, C.; Schulz, C.A. Chronic Pain in children and adolescents: Diagnosis and treatment of primary pain disorders in head, abdomen, muscles and joints. Children 2016, 3, 42. [Google Scholar] [CrossRef] [PubMed]
- Treede, R.D.; Rief, W.; Barke, A.; Aziz, Q.; Bennett, M.I.; Benoliel, R.; Cohen, M.; Evers, S.; Finnerup, N.B.; First, M.B.; et al. A classification of chronic pain for ICD-11. Pain 2015, 156, 1003–1007. [Google Scholar] [CrossRef] [PubMed]
- Goodman, J.E.; McGrath, P.J. The epidemiology of pain in children and adolescents: A review. Pain 1991, 46, 247–264. [Google Scholar] [CrossRef]
- King, S.; Chambers, C.T.; Huguet, A.; MacNevin, R.C.; McGrath, P.J.; Parker, L.; MacDonald, A.J. The epidemiology of chronic pain in children and adolescents revisited: A systematic review. Pain 2011, 152, 2729–2738. [Google Scholar] [CrossRef] [PubMed]
- Chiou, E.; Nurko, S. Management of functional abdominal pain and irritable bowel syndrome in adolescents. Expert Rev. Gastroenterol. 2010, 4, 293–304. [Google Scholar] [CrossRef] [PubMed]
- Sandhu, B.K.; Paul, S.P. Irritable bowel syndrome in children: Pathogenesis, diagnosis and evidence based treatment. World J. Gastroenterol. 2014, 20, 6013–6023. [Google Scholar] [CrossRef] [PubMed]
- Drossman, D.A. Functional gastrointestinal disorders: History, pathophysiology, clinical features, and Rome IV. Gastroenterology 2016, 150, 1262–1279. [Google Scholar] [CrossRef] [PubMed]
- Hyams, J.S.; Di Lorenzo, C. Childhood functional gastrointestinal disorders: Child/adolescent. Gastroenterology 2016, 150, 1456–1468. [Google Scholar] [CrossRef] [PubMed]
- Barakzai, M.D.; Gregory, J.; Fraser, D. The effect of culture on symptom reporting: Hispanics and irritable bowel syndrome. J. Am. Acad. Nurse Pract. 2007, 19, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Zola, I.K. Culture and symptoms—An analysis of patients’ presenting complaints. Am. Soc. Rev. 1966, 31, 615–630. [Google Scholar] [CrossRef]
- Zuckerman, M.J.; Guerra, L.G.; Drossman, D.A.; Foland, J.A.; Gregory, G.G. Health care seeking behaviors related to bowel complaints: Hispanics versus non-Hispanic whites. Dig. Dis. Sci. 1996, 41, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Ferreira-Maia, A.P.; Matijasevich, A.; Wang, Y. Epidemiology of functional gastrointestinal disorders in infants and toddlers: A systematic review. World J. Gastroenterol. 2016, 22, 6547–6558. [Google Scholar] [CrossRef] [PubMed]
- Rajindrajith, S.; Devanarayana, N.M. Subtypes and symptomatology of irritable bowel syndrome in children and adolescents: A school-based survey using Rome III criteria. J. Neurogastroenterol. Motil. 2012, 18, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Dong, L.; Dingguo, L.; Xiaoxing, X.; Hanming, L. An epidemiologic study of irritable bowel syndrome in adolescents and children in China: A school-based study. Pediatrics 2005, 116, e393–e396. [Google Scholar] [CrossRef] [PubMed]
- El-Matary, W.; Spray, C.; Sandhu, B. Irritable bowel syndrome: The commonest cause of recurrent abdominal pain in children. Eur. J. Pediatrics 2004, 163, 584–588. [Google Scholar] [CrossRef] [PubMed]
- Stein, R.; Chelimsky, T.; Li, H.; Chelimsky, G. Functional Gastrointestinal Disorders in a Primary Care Pediatric Clinic. Glob. Pediatr. Health 2015, 2. [Google Scholar] [CrossRef] [PubMed]
- Hechler, T.; Dobe, M.; Zernikow, B. Commentary: A worldwide call for multimodal inpatient treatment for children and adolescents suffering from chronic pain and pain-related disability. J. Pediatr. Psychol. 2009, 35, 138–140. [Google Scholar] [CrossRef] [PubMed]
- Rasquin, A.; Di Lorenzo, C.; Forbes, D.; Guialdes, E.; Hyams, J.S.; Staiano, A.; Walker, L.S. Childhood functional gastrointestinal disorders: Child/Adolescent. Gastroenterology 2006, 130, 1527–1537. [Google Scholar] [CrossRef] [PubMed]
- Gauntlett-Gilbert, J.; Eccleston, C. Disability in adolescents with chronic pain: Patterns and predictors across different domains of functioning. Pain 2007, 131, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Konijnenberg, A.Y.; Uieterwaal, C.S.P.M.; Kimpen, J.L.L.; van der Hoeven, J.; Buitetlaar, J.K.; de Graeff-Meeder, E.R. Children with unexplained chronic pain: Substantial impairment in everyday life. Arch. Dis Child. 2005, 90, 680–688. [Google Scholar] [CrossRef] [PubMed]
- Kridler, J.; Kamat, D. Irritable bowel syndrome: A review for general pediatricians. Pediatr. Ann. 2016, 45, e30–e33. [Google Scholar] [CrossRef] [PubMed]
- Logan, D.E.; Simons, L.E.; Stein, M.J.; Chastain, L. School impairment in adolescents with chronic pain. J. Pain 2008, 9, 407–416. [Google Scholar] [CrossRef] [PubMed]
- Long, A.C.; Krishnamurthy, V.; Palermo, T.M. Sleep disturbances in school-age children with chronic pain. J. Pediatr. Psychol. 2008, 33, 258–268. [Google Scholar] [CrossRef] [PubMed]
- Hunfeld, J.A.M.; Perquin, C.W.; Duivenvoorden, H.J.; Hazebroek-Kampschreur, A.A.J.M.; Passchier, J.; van Suijlekom-Smit, L.W.A.; Van der Wouden, J.C. Chronic pain and its impact on quality of life in adolescents and their families. J. Pediatr. Psychol. 2001, 26, 145–153. [Google Scholar] [CrossRef] [PubMed]
- McOmber, M.; Schulman, R. Recurrent abdominal pain and irritable bowel syndrome in children. Curr. Opin. Pediatr. 2007, 19, 581–585. [Google Scholar] [CrossRef] [PubMed]
- Schurman, J.V.; Friesen, C.A.; Dai, H.; Elder Danda, C.; Hyman, P.E.; Cocjin, J.T. Sleep problems and functional disability in children with functional gastrointestinal disorders: An examination of the potential mediating effects of physical and emotional symptoms. BMC Gastroenterol. 2012. [Google Scholar] [CrossRef] [PubMed]
- Campo, J.V.; Comer, D.M.; Jansen-McWilliams, L.; Gardner, W.; Kelleher, K.J. Recurrent pain, emotional distress, and health service use in childhood. J. Pediatr. 2002, 141, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Campo, J.V.; Bridge, J.; Lucas, A.; Savorelli, S.; Walker, L.; Di Lorenzo, C.; Iyengar, S.; Brent, D.A. Physical and emotional health of mothers of youth with functional abdominal pain. Arch. Pediatr. Adolesc. Med. 2007, 161, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Muscatello, M.R.A.; Bruno, A.; Mento, C.; Pandolfo, G.; Zoccali, R.A. Personality traits and emotional patterns in irritable bowel syndrome. World J. Gastroenterol. 2016, 22, 6402–6415. [Google Scholar] [CrossRef] [PubMed]
- Youssef, N.N.; Murphy, T.G.; Langseder, A.L.; Rosh, J.R. Quality of life for children with functional abdominal pain: A comparison study of patients’ and parents’ perceptions. Pediatrics 2006, 117, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Eccleston, C.; Crombez, G.; Scotford, A.; Clinch, J.; Connell, H. Adolescent chronic pain: Patterns and predictors of emotional distress in adolescents with chronic pain and their parents. Pain 2004, 108, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Varni, J.W.; Rapoff, M.A.; Waldron, S.A.; Gragg, R.A.; Bernstein, B.H.; Lindsley, C.B. Chronic pain and emotional distress in children and adolescents. J. Dev. Behav. Pediatr. 1996, 17, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Reed-Knight, B.; Maddux, M.H.; Deacy, A.D.; Lamparyk, K.; Stone, A.L.; Mackner, L. Brain-gut interactions and maintenance factors in pediatric gastroenterological disorders: Recommendations for clinical care. Clin. Pract. Pediatr. Psychol. 2017, 5, 93–105. [Google Scholar] [CrossRef]
- Vanner, S.J.; Greenwood-Van, M.B.; Mawe, G.M.; Shea-Donohue, T.; Verdu, E.F.; Wood, J.; Grundy, D. Fundamentals of Neurogastroenterology. Gastroenterology 2016, 150, 1280–1291. [Google Scholar] [CrossRef] [PubMed]
- Chogle, A.; Mintjens, S.; Saps, M. Pediatric IBS: An overview on pathophysiology, diagnosis and treatment. Pediatr. Ann. 2014, 43, e76–e82. [Google Scholar] [CrossRef] [PubMed]
- Korterink, J.J.; Diederen, K.; Benninga, M.A.; Tabbers, M.M. Epidemiology of pediatric functional abdominal pain disorders: A meta-analysis. PLoS ONE 2015, 10, e0126982. [Google Scholar] [CrossRef] [PubMed]
- Mackner, L.M.; Greenley, R.N.; Szigethy, E.; Herzer, M.; Deer, K.; Hommel, K.A. Psychosocial issues in pediatric inflammatory bowel disease: Report of the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition. J. Pediatr. Gastroenterol. Nutr. 2013, 56, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.H.; Maletic, V.; Raison, C.L. Inflammation and its discontents: The role of cytokines in the pathophysiology of major depression. Biol. Psychiatry 2009, 65, 732–741. [Google Scholar] [CrossRef] [PubMed]
- Tillisch, K.; Mayer, E.A.; Labus, J.S. Quantitative meta-analysis identifies brain regions activated during rectal distension in irritable bowel syndrome. Gastroenterology 2011, 140, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Van Oudenhove, L.; Levy, R.L.; Crowell, M.D.; Drossman, D.A.; Halpert, A.D.; Keefer, L.K.; Lackner, J.M.; Murphy, T.B.; Naliboff, B.D. Biopsychosocial aspects of functional gastrointestinal disorders: How central and environmental processes contribute to the development and expression of functional gastrointestinal disorders. World J. Gastroenterol. 2016, 22, 6402–6415. [Google Scholar] [CrossRef] [PubMed]
- Shelby, G.D.; Shirkey, K.C.; Sherman, A.L.; Beck, J.E.; Haman, K.; Shears, A.R.; Horst, S.N.; Smith, C.A.; Garber, J.; Walker, L.S. Functional abdominal pain in childhood and long-term vulnerability to anxiety disorders. Pediatrics 2013, 132, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Centre for Economic Performance (2012). How Mental Illness Loses Out in the NHS. Available online: http://cep.lse.ac.uk/pubs/download/special/cepsp26.pdf (accessed on 20 November 2017).
- Prasko, J.; Jelenova, D.; Mihal, V. Psychological aspects and psychotherapy of inflammatory bowel disease and irritable bowel syndrome in children. Biomed. Pap. Med. Fac. Univ. Palacký Olomouc Czech Repub. 2010, 154, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Bonaz, B.L.; Bernstein, C.N. Brain-gut interactions in inflammatory bowel disease. Gastroenterology 2013, 144, 36–49. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.J.; Kim, Y.B.; Kim, J.H.; Kwon, H.C.; Kim, D.K.; Cho, S.W. The alteration of enterochromaffin cell, mast cell, and lamina propria T lymphocyte numbers in irritable bowel syndrome and its relationship with psychological factors. J. Gastroenterol. Hepatol. 2008, 23, 1689–1694. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C. Mental health: Thinking from the gut. Nature 2015, 518, S12–S15. [Google Scholar] [CrossRef] [PubMed]
- Noel, M.; Groenewald, C.B.; Beals-Erickson, S.E.; Gebert, J.T.; Palermo, T.M. Chronic pain in adolescence and internalizing mental health disorders: A nationally representative study. Pain 2016, 157, 1333–1338. [Google Scholar] [CrossRef] [PubMed]
- Stanford, E.A.; Chambers, C.T.; Biesanz, J.C.; Chen, E. The frequency, trajectories and predictors of adolescent recurrent pain: A population-based approach. Pain 2008, 138, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Noel, M.; Vinall, J.; Tomfohr-Madsen, L.; Holley, A.L.; Wilson, A.C.; Palermo, T.M. Sleep Mediates the Association between PTSD Symptoms and Chronic Pain in Youth. J. Pain 2017. [Google Scholar] [CrossRef] [PubMed]
- Fearon, P.; Hotopf, M. Relation between headache in childhood and physical and psychiatric symptoms in adulthood: National birth cohort study. BMJ 2001, 322, 1145–1150. [Google Scholar] [CrossRef] [PubMed]
- Hotopf, M.; Carr, S.; Mayou, R.; Wadsworth, M.; Wessely, S. Why do children have chronic abdominal pain, and what happens to them when they grow up? Population based cohort study. BMJ 1998, 316, 1196–1200. [Google Scholar] [CrossRef] [PubMed]
- Simons, L.E.; Kaczynski, K.J. The Fear Avoidance model of chronic pain: Examination for pediatric application. J. Pain 2012, 13, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Vlaeyen, J.W.; Linton, S.J. Fear-avoidance and its consequences in chronic musculoskeletal pain: A state of the art. Pain 2000, 85, 317–332. [Google Scholar] [CrossRef]
- Caes, L.; Fisher, E.; Clinch, J.; Tobias, J.H.; Eccleston, C. The role of pain-related anxiety in adolescents’ disability and social impairment: ALSPAC data. Eur. J. Pain 2015, 19, 842–851. [Google Scholar] [CrossRef] [PubMed]
- Simons, L.E.; Sieberg, C.B.; Claar, R.L. Anxiety and functional disability in a large sample of children and adolescents with chronic pain. Pain Res. Manag. 2012, 17, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Fisher, E.; Caes, L.; Clinch, J.; Tobias, J.H.; Eccleston, C. Anxiety at 13 and its effect on pain, pain-related anxiety, and pain-related disability at 17: An ALSPAC cohort longitudinal analysis. Psychol. Health Med. 2016, 21, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Daniels, J.A. Adolescent separation-individuation and family transitions. Adolescence 1990, 25, 105–116. [Google Scholar] [PubMed]
- Jordan, A.L.; Eccleston, C.; Osborn, M. Being a parent of the adolescent with complex chronic pain: An interpretative phenomenological analysis. Eur. J. Pain 2007, 11, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Noel, M.; Beals-Erickson, S.E.; Law, E.F.; Alberts, N.; Palermo, T.M. Characterizing the pain narratives of parents of youth with chronic pain. Clin. J. Pain 2016, 32, 849–858. [Google Scholar] [CrossRef] [PubMed]
- Caes, L.; Vervoort, T.; Devos, P.; Verlooy, J.; Benoit, Y.; Goubert, L. Parental distress and catastrophic thoughts about child pain: Implications for parental protective behavior in the context of child leukemia related medical procedures. Clin. J. Pain 2014, 30, 787–799. [Google Scholar] [CrossRef] [PubMed]
- Caes, L.; Vervoort, T.; Eccleston, C.; Vandenhende, M.; Goubert, L. Parental catastrophizing about child’s pain and its relationship with activity restriction: The mediating role of parental distress. Pain 2011, 152, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Caes, L.; Vervoort, T.; Trost, Z.; Goubert, L. The impact of parental catastrophizing and contextual threat on parents’ emotional and behavioral responses to their child’s pain. Pain 2012, 153, 687–695. [Google Scholar] [CrossRef] [PubMed]
- Calvano, C.; Groβ, M.; Warschburger, P. Do mothers benefit from a child-focused cognitive behavioral treatment (CBT) for childhood functional abdominal pain? A randomized controlled pilot trial. Children 2017, 4, 13. [Google Scholar] [CrossRef] [PubMed]
- Claar, R.L.; Simons, L.E.; Logan, D.E. Parental response to children’s pain: The moderating impact of children’s emotional distress on symptoms and disability. Pain 2008, 138, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Peterson, C.C.; Palermo, T.M. Parental reinforcement of recurrent pain: The moderating impact of child depression and anxiety on functional disability. Pain 2004, 131, 132–141. [Google Scholar] [CrossRef]
- Walker, L.S.; Claar, R.L.; Garber, J. Social consequences of children’s pain: When do they encourage symptom maintenance? J. Pediatr. Psychol. 2002, 27, 689–698. [Google Scholar] [CrossRef] [PubMed]
- Levy, R.L.; Whitehead, W.E.; Walker, L.S.; Von Korff, M.; Feld, A.D.; Garner, M.; Christie, D. Increased somatic complaints and health-care utilization in children: Effects of parent IBS status and parent response to gastrointestinal symptoms. Am. J. Gastroenterol. 2004, 99, 2442–2451. [Google Scholar] [CrossRef] [PubMed]
- Fales, J.L.; Essner, B.S.; Harris, M.A.; Palermo, T.M. When helping hurts: Miscarried helping in families of youth with chronic pain. J. Pediatr. Psychol. 2014, 39, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Goubert, L.; Simons, L.E. Cognitive styles and processes in paediatric pain. In Oxford Textbook of Pediatric Pain; Oxford University Press: Oxford, UK, 2013; pp. 95–101. ISBN 9780199642656. [Google Scholar]
- Dinan, T.G.; Cryan, J.; Shanahan, F.; Keeling, P.W.; Quigley, E.M. IBS: An epigenetic perspective. Nat. Rev. Gastroenterol. Hepatol. 2010, 7, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Stone, A.L.; Walker, L.S. Adolescents’ observations of parent pain behaviors: Preliminary measure validation and test of social learning theory in pediatric chronic pain. J. Pediatr. Psychol. 2017, 42, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.E.; Blount, R.L.; Walker, L.S. Children’s pain threat appraisal and catastrophizing moderate the impact of parent verbal behavior on children’s symptom complaints. J. Pediatr. Psychol. 2010, 36, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Kozlowska, K. Good children presenting with conversion disorder. Clin. Child Psychol. Psychiatry 2001, 6, 575–591. [Google Scholar] [CrossRef]
- Carter, B.; McGoldrick, M. The Changing Family Life Cycle; Allyn & Bacon: Boston, MA, USA, 1989. [Google Scholar]
- Griffiths, J.L.; Griffiths, M.E. The Body Speaks: Therapeutic Dialogues for Mind-Body Problems; Basic Books: New York, NY, USA, 1994. [Google Scholar]
- Christie, D.; Griffin, A. Taking the psycho out of psychosomatic: Using systemic approaches in a paediatric setting for the treatment of adolescents with unexplained physical symptoms. Clin. Child Psychol. Psychiatry 2008, 13, 531–542. [Google Scholar] [CrossRef]
- Levy, R.L.; Langer, S.L.; Walker, L.S.; Romano, J.M.; Christie, D.L.; Youssef, N.; DuPen, M.M.; Feld, A.D.; Ballard, S.A.; Welsh, E.M.; et al. Cognitive-behavioral therapy for children with functional abdominal pain and their parents decreases pain and other symptoms. Am. J. Gastroenterol. 2010, 105, 946–956. [Google Scholar] [CrossRef] [PubMed]
- Rutten, J.M.T.M.; Vlieger, A.M.; Frankenhuis, C.; George, E.K.; Groeneweg, M.; Norbruis, O.F.; Tjon a Ten, W.; Van Wering, H.; Dijkgraaf, M.G.W.; Merkus, M.P.; et al. Gut-directed hypnotherapy in children with irritable bowel syndrome or functional abdominal pain (syndrome): A randomized controlled trial on self exercises at home using CD versus individual therapy by qualified therapists. BMC Pediatr. 2014. [Google Scholar] [CrossRef] [PubMed]
- Salt, W.B.; Season, E.H. Fibromyalgia and the Mindbodyspirit Connection; Parkview: Cincinnati, OH, USA, 2000. [Google Scholar]
- Williams, A.C.; Erskine, A. Chronic Pain. In Health Psychology: Processes and Applications; Broome, A., Llewelyn, S., Eds.; Chapman Hall: London, UK, 1995; pp. 353–367. [Google Scholar]
- Dhaliwal, S.K.; Hunt, R.H. Doctor-patient interaction for irritable bowel syndrome in primary care: A systematic perspective. Eur. J. Gastroenterol. Hepatol. 2004, 16, 1161–1166. [Google Scholar] [CrossRef] [PubMed]
- Garralda, M.E. A selective review of child psychiatric syndromes with a somatic presentation. Br. J. Psychiatry 1992, 161, 759–773. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.P.; Barnard, P.; Bigwood, C.; Candy, D.C.A. Challenges in management of Irritable Bowel Syndrome in children. Indian Pediatr. 2013, 50, 1137–1143. [Google Scholar] [CrossRef] [PubMed]
- Grattan-Smith, P.; Fairley, M.; Procopis, P. Clinical features of conversion disorder. Arch. Dis. Child. 1988, 63, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Leslie, S.A. Diagnosis and treatment of hysterical conversion reactions. Arch. Dis. Child. 1988, 63, 506–511. [Google Scholar] [CrossRef] [PubMed]
- Deaton, A.V. Treating conversion disorders: Is a pediatric rehabilitation hospital the place? Rehabil. Psychol. 1998, 43, 56–62. [Google Scholar] [CrossRef]
- Gooch, J.L.; Wolcott, R.; Speed, J. Behavioural management of conversion disorder in children. Arch. Phys. Med. Rehabil. 1997, 78, 264–268. [Google Scholar] [CrossRef]
- Calvert, P.; Jureidini, J. Restrained rehabilitation: An approach to children and adolescents with unexplained signs and symptoms. Arch. Dis. Child. 2003, 88, 399–402. [Google Scholar] [CrossRef] [PubMed]
- Kent, D.A.; Tomasson, K.; Coryell, W. Course and outcome of conversion and somatization disorders: A four year follow-up. Psychosomatics 1995, 36, 138–144. [Google Scholar] [CrossRef]
- Ballou, S.; Keefer, L. Psychological interventions for Irritable Bowel Syndrome and Inflammatory Bowel Diseases. Clin. Transl. Gastroenterol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Eccleston, C.; Fisher, E.; Law, E.; Bartlett, J.; Palermo, T.M. Psychological interventions for parents of children and adolescents with chronic illness. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef]
- Eccleston, C.; Morley, S.; Williams, A.; Yorle, L.; Mastroyannopoulou, K. Systematic review of randomised controlled trials of psychological therapy for chronic pain in children and adolescents, with a subset meta-analysis of pain relief. Pain 2002, 99, 157–165. [Google Scholar] [CrossRef]
- Laird, K.T.; Tanner-Smith, E.E.; Russell, A.C.; Hollon, S.D.; Walker, L.S. Comparative efficacy of psychological therapies for improving mental health and daily functioning in irritable bowel syndrome: A systematic review and meta-analysis. Clin. Psychol. Rev. 2017, 51, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Weydert, J.A.; Ball, T.M.; Davis, M.F. Systematic review of treatment for recurrent abdominal pain. Pediatrics 2003, 111, e1–e11. [Google Scholar] [CrossRef] [PubMed]
- Foxx-Orenstein, A.E. New and emerging therapies for the treatment of irritable bowel syndrome: An update for gastroenterologists. Ther. Adv. Gastroenterol. 2016, 9, 354–375. [Google Scholar] [CrossRef] [PubMed]
- Bagnall, A.M.; Whiting, P.; Wright, K.; Sowden, A. The Effectiveness of Interventions Used in the Treatment/Management of CFS and/or ME in Adults and Children; NHS Centre for Review and Dissemination, University of York: York, UK, 2002. [Google Scholar]
- Eccleston, C.; Malleson, P.N.; Clinch, J.; Connell, H.; Sourbut, C. Chronic pain in adolescents: Evaluation of a programme of interdisciplinary cognitive behaviour therapy. Arch. Dis. Child. 2003, 88, 881–885. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caes, L.; Orchard, A.; Christie, D. Connecting the Mind–Body Split: Understanding the Relationship between Symptoms and Emotional Well-Being in Chronic Pain and Functional Gastrointestinal Disorders. Healthcare 2017, 5, 93. https://doi.org/10.3390/healthcare5040093
Caes L, Orchard A, Christie D. Connecting the Mind–Body Split: Understanding the Relationship between Symptoms and Emotional Well-Being in Chronic Pain and Functional Gastrointestinal Disorders. Healthcare. 2017; 5(4):93. https://doi.org/10.3390/healthcare5040093
Chicago/Turabian StyleCaes, Line, Alex Orchard, and Deborah Christie. 2017. "Connecting the Mind–Body Split: Understanding the Relationship between Symptoms and Emotional Well-Being in Chronic Pain and Functional Gastrointestinal Disorders" Healthcare 5, no. 4: 93. https://doi.org/10.3390/healthcare5040093
APA StyleCaes, L., Orchard, A., & Christie, D. (2017). Connecting the Mind–Body Split: Understanding the Relationship between Symptoms and Emotional Well-Being in Chronic Pain and Functional Gastrointestinal Disorders. Healthcare, 5(4), 93. https://doi.org/10.3390/healthcare5040093




