The Effects of Concurrent Training Versus Aerobic or Resistance Training Alone on Body Composition in Middle-Aged and Older Adults: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Search Strategy and Study Selection
2.2. Identification and Selection Criteria
2.3. Data Extraction and Synthesis
2.4. Quality Assessment
2.5. Statistical Analysis
3. Results
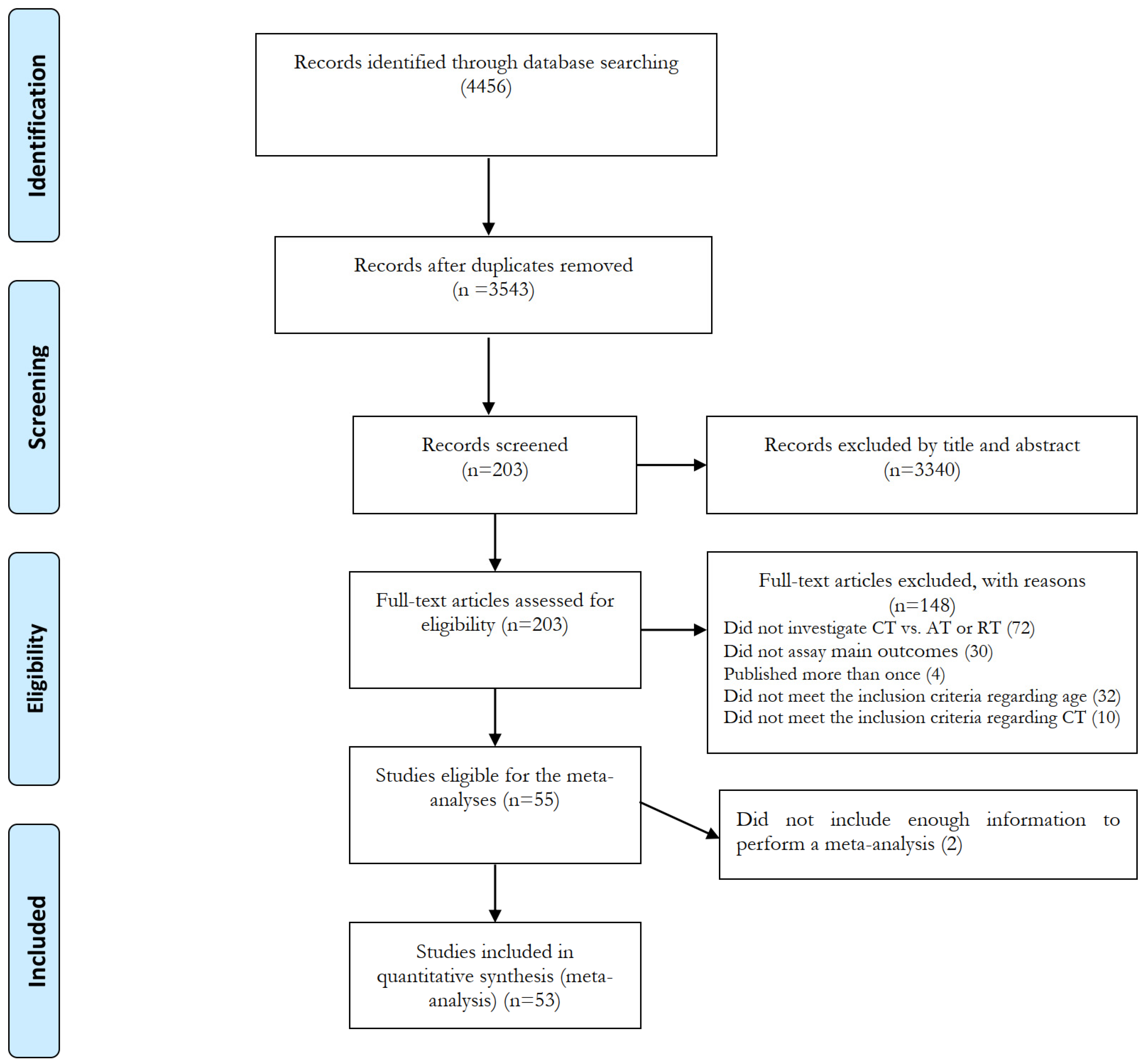
3.1. Search Results
3.2. Study Characteristics
3.3. Meta-Analysis
3.3.1. CT vs. AT
3.3.2. CT vs. RT
3.4. Heterogeneity
3.5. Publication Bias
3.6. Subgroup Analysis
4. Discussion
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194–1217. [Google Scholar] [PubMed]
- Kuk, J.L.; Saunders, T.J.; Davidson, L.E.; Ross, R. Age-related changes in total and regional fat distribution. Ageing Res. Rev. 2009, 8, 339–348. [Google Scholar] [PubMed]
- Zamboni, M.; Armellini, F.; Harris, T.; Turcato, E.; Micciolo, R.; Bergamo-Andreis, I.A.; Bosello, O. Effects of age on body fat distribution and cardiovascular risk factors in women. Am. J. Clin. Nutr. 1997, 66, 111–115. [Google Scholar] [CrossRef]
- Wannamethee, S.G.; Shaper, A.G.; Whincup, P.H. Body fat distribution, body composition, and respiratory function in elderly men. Am. J. Clin. Nutr. 2005, 82, 996–1003. [Google Scholar] [PubMed]
- Janssen, I.; Ross, R. Linking age-related changes in skeletal muscle mass and composition with metabolism and disease. J. Nutr. Health Aging 2005, 9, 408. [Google Scholar]
- Volpi, E.; Nazemi, R.; Fujita, S. Muscle tissue changes with aging. Curr. Opin. Clin. Nutr. Metab. Care 2004, 7, 405–410. [Google Scholar]
- Cruz-Jentoft, A.J.; Sayer, A.A. Sarcopenia. Lancet 2019, 393, 2636–2646. [Google Scholar]
- Larsson, L.; Degens, H.; Li, M.; Salviati, L.; Lee, Y.I.; Thompson, W.; Kirkland, J.L.; Sandri, M. Sarcopenia: Aging-related loss of muscle mass and function. Physiol. Rev. 2019, 99, 427–511. [Google Scholar]
- Yeung, S.S.; Reijnierse, E.M.; Pham, V.K.; Trappenburg, M.C.; Lim, W.K.; Meskers, C.G.; Maier, A.B. Sarcopenia and its association with falls and fractures in older adults: A systematic review and meta-analysis. J. Cachexia Sarcopenia Muscle 2019, 10, 485–500. [Google Scholar] [CrossRef]
- Gao, K.; Cao, L.-F.; Ma, W.-Z.; Gao, Y.-J.; Luo, M.-S.; Zhu, J.; Li, T.; Zhou, D. Association between sarcopenia and cardiovascular disease among middle-aged and older adults: Findings from the China health and retirement longitudinal study. EClinicalMedicine 2022, 44, 101264. [Google Scholar]
- Damluji, A.A.; Alfaraidhy, M.; AlHajri, N.; Rohant, N.N.; Kumar, M.; Al Malouf, C.; Bahrainy, S.; Ji Kwak, M.; Batchelor, W.B.; Forman, D.E. Sarcopenia and Cardiovascular Diseases. Circulation 2023, 147, 1534–1553. [Google Scholar] [CrossRef] [PubMed]
- Mesinovic, J.; Zengin, A.; De Courten, B.; Ebeling, P.R.; Scott, D. Sarcopenia and type 2 diabetes mellitus: A bidirectional relationship. Diabetes Metab. Syndr. Obes. Targets Ther. 2019, 12, 1057–1072. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.N.; Choi, K.M. The implications of sarcopenia and sarcopenic obesity on cardiometabolic disease. J. Cell. Biochem. 2015, 116, 1171–1178. [Google Scholar] [CrossRef]
- Nishikawa, H.; Asai, A.; Fukunishi, S.; Nishiguchi, S.; Higuchi, K. Metabolic syndrome and sarcopenia. Nutrients 2021, 13, 3519. [Google Scholar] [CrossRef]
- Ponti, F.; Santoro, A.; Mercatelli, D.; Gasperini, C.; Conte, M.; Martucci, M.; Sangiorgi, L.; Franceschi, C.; Bazzocchi, A. Aging and imaging assessment of body composition: From fat to facts. Front. Endocrinol. 2020, 10, 861. [Google Scholar] [CrossRef]
- Beaufrere, B.; Morio, B. Fat and protein redistribution with aging: Metabolic considerations. Eur. J. Clin. Nutr. 2000, 54, S48–S53. [Google Scholar] [CrossRef] [PubMed]
- Tchkonia, T.; Morbeck, D.E.; Von Zglinicki, T.; Van Deursen, J.; Lustgarten, J.; Scrable, H.; Khosla, S.; Jensen, M.D.; Kirkland, J.L. Fat tissue, aging, and cellular senescence. Aging Cell 2010, 9, 667–684. [Google Scholar] [CrossRef]
- Di Lorito, C.; Long, A.; Byrne, A.; Harwood, R.H.; Gladman, J.R.F.; Schneider, S.; Logan, P.; Bosco, A.; van der Wardt, V. Exercise interventions for older adults: A systematic review of meta-analyses. J. Sport. Health Sci. 2021, 10, 29–47. [Google Scholar] [CrossRef]
- Khalafi, M.; Malandish, A.; Rosenkranz, S.K.; Ravasi, A.A. Effect of resistance training with and without caloric restriction on visceral fat: A systemic review and meta-analysis. Obes. Rev. 2021, 22, e13275. [Google Scholar] [CrossRef]
- Wewege, M.A.; Desai, I.; Honey, C.; Coorie, B.; Jones, M.D.; Clifford, B.K.; Leake, H.B.; Hagstrom, A.D. The effect of resistance training in healthy adults on body fat percentage, fat mass and visceral fat: A systematic review and meta-analysis. Sports Med. 2022, 52, 287–300. [Google Scholar] [CrossRef]
- Willis, L.H.; Slentz, C.A.; Bateman, L.A.; Shields, A.T.; Piner, L.W.; Bales, C.W.; Houmard, J.A.; Kraus, W.E. Effects of aerobic and/or resistance training on body mass and fat mass in overweight or obese adults. J. Appl. Physiol. 2012, 113, 1831–1837. [Google Scholar] [CrossRef] [PubMed]
- Ismail, I.; Keating, S.; Baker, M.; Johnson, N. A systematic review and meta-analysis of the effect of aerobic vs. resistance exercise training on visceral fat. Obes. Rev. 2012, 13, 68–91. [Google Scholar] [PubMed]
- Hagstrom, A.D.; Marshall, P.W.; Halaki, M.; Hackett, D.A. The effect of resistance training in women on dynamic strength and muscular hypertrophy: A systematic review with meta-analysis. Sports Med. 2020, 50, 1075–1093. [Google Scholar] [PubMed]
- Grgic, J.; Garofolini, A.; Orazem, J.; Sabol, F.; Schoenfeld, B.J.; Pedisic, Z. Effects of resistance training on muscle size and strength in very elderly adults: A systematic review and meta-analysis of randomized controlled trials. Sports Med. 2020, 50, 1983–1999. [Google Scholar]
- Liao, C.D.; Chen, H.C.; Kuo, Y.C.; Tsauo, J.Y.; Huang, S.W.; Liou, T.H. Effects of muscle strength training on muscle mass gain and hypertrophy in older adults with osteoarthritis: A systematic review and meta-analysis. Arthritis Care Res. 2020, 72, 1703–1718. [Google Scholar]
- Cheema, B.S.; Chan, D.; Fahey, P.; Atlantis, E. Effect of progressive resistance training on measures of skeletal muscle hypertrophy, muscular strength and health-related quality of life in patients with chronic kidney disease: A systematic review and meta-analysis. Sports Med. 2014, 44, 1125–1138. [Google Scholar]
- Faigenbaum, A.D.; Kraemer, W.J.; Blimkie, C.J.; Jeffreys, I.; Micheli, L.J.; Nitka, M.; Rowland, T.W. Youth resistance training: Updated position statement paper from the national strength and conditioning association. J. Strength Cond. Res. 2009, 23, S60–S79. [Google Scholar] [CrossRef]
- Al-Mhanna, S.B.; Franklin, B.A.; Jakicic, J.M.; Stamatakis, E.; Pescatello, L.S.; Riebe, D.; Thompson, W.R.; Skinner, J.; Colberg, S.R.; Alkhamees, N.H.; et al. Impact of resistance training on cardiometabolic health-related indices in patients with type 2 diabetes and overweight/obesity: A systematic review and meta-analysis of randomised controlled trials. Br. J. Sports Med. 2025. [Google Scholar] [CrossRef]
- Izquierdo, M.; Merchant, R.; Morley, J.; Anker, S.; Aprahamian, I.; Arai, H.; Aubertin-Leheudre, M.; Bernabei, R.; Cadore, E.; Cesari, M. International exercise recommendations in older adults (ICFSR): Expert consensus guidelines. J. Nutr. Health Aging 2021, 25, 824–853. [Google Scholar] [CrossRef]
- Bull, F.C.; Al-Ansari, S.S.; Biddle, S.; Borodulin, K.; Buman, M.P.; Cardon, G.; Carty, C.; Chaput, J.-P.; Chastin, S.; Chou, R. World Health Organization 2020 guidelines on physical activity and sedentary behaviour. Br. J. Sports Med. 2020, 54, 1451–1462. [Google Scholar]
- Leveritt, M.; Abernethy, P.J.; Barry, B.K.; Logan, P.A. Concurrent strength and endurance training: A review. Sports Med. 1999, 28, 413–427. [Google Scholar]
- Batrakoulis, A.; Jamurtas, A.Z.; Metsios, G.S.; Perivoliotis, K.; Liguori, G.; Feito, Y.; Riebe, D.; Thompson, W.R.; Angelopoulos, T.J.; Krustrup, P.; et al. Comparative Efficacy of 5 Exercise Types on Cardiometabolic Health in Overweight and Obese Adults: A Systematic Review and Network Meta-Analysis of 81 Randomized Controlled Trials. Circ. Cardiovasc. Qual. Outcomes 2022, 15, e008243. [Google Scholar] [CrossRef] [PubMed]
- Al-Mhanna, S.B.; Batrakoulis, A.; Norhayati, M.N.; Mohamed, M.; Drenowatz, C.; Irekeola, A.A.; Afolabi, H.A.; Gulu, M.; Alkhamees, N.H.; Wan Ghazali, W.S. Combined Aerobic and Resistance Training Improves Body Composition, Alters Cardiometabolic Risk, and Ameliorates Cancer-Related Indicators in Breast Cancer Patients and Survivors with Overweight/Obesity: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Sports Sci. Med. 2024, 23, 366–395. [Google Scholar] [CrossRef]
- Ahtiainen, J.P.; Hulmi, J.J.; Kraemer, W.J.; Lehti, M.; Pakarinen, A.; Mero, A.A.; Karavirta, L.; Sillanpää, E.; Selänne, H.; Alen, M.; et al. Strength, [corrected] endurance or combined training elicit diverse skeletal muscle myosin heavy chain isoform proportion but unaltered androgen receptor concentration in older men. Int. J. Sports Med. 2009, 30, 879–887. [Google Scholar] [CrossRef] [PubMed]
- AminiLari, Z.; Fararouei, M.; Amanat, S.; Sinaei, E.; Dianatinasab, S.; AminiLari, M.; Daneshi, N.; Dianatinasab, M. The Effect of 12 Weeks Aerobic, Resistance, and Combined Exercises on Omentin-1 Levels and Insulin Resistance among Type 2 Diabetic Middle-Aged Women. Diabetes Metab. J. 2017, 41, 205–212. [Google Scholar] [CrossRef]
- Cadore, E.L.; Pinto, R.S.; Lhullier, F.L.; Correa, C.S.; Alberton, C.L.; Pinto, S.S.; Almeida, A.P.; Tartaruga, M.P.; Silva, E.M.; Kruel, L.F. Physiological effects of concurrent training in elderly men. Int. J. Sports Med. 2010, 31, 689–697. [Google Scholar] [CrossRef] [PubMed]
- Campos, A.L.P.; Del Ponte, L.d.S.; Cavalli, A.S.; Afonso, M.d.R.; Schild, J.F.G.; Reichert, F.F. Effects of concurrent training on health aspects of elderly women. Rev. Bras. De Cineantropometria Desempenho Hum. 2013, 15, 437–447. [Google Scholar]
- Chen, H.T.; Chung, Y.C.; Chen, Y.J.; Ho, S.Y.; Wu, H.J. Effects of Different Types of Exercise on Body Composition, Muscle Strength, and IGF-1 in the Elderly with Sarcopenic Obesity. J. Am. Geriatr. Soc. 2017, 65, 827–832. [Google Scholar] [CrossRef]
- Church, T.S.; Blair, S.N.; Cocreham, S.; Johannsen, N.; Johnson, W.; Kramer, K.; Mikus, C.R.; Myers, V.; Nauta, M.; Rodarte, R.Q. Effects of aerobic and resistance training on hemoglobin A1c levels in patients with type 2 diabetes: A randomized controlled trial. JAMA 2010, 304, 2253–2262. [Google Scholar]
- Davidson, L.E.; Hudson, R.; Kilpatrick, K.; Kuk, J.L.; McMillan, K.; Janiszewski, P.M.; Lee, S.; Lam, M.; Ross, R. Effects of exercise modality on insulin resistance and functional limitation in older adults: A randomized controlled trial. Arch. Intern. Med. 2009, 169, 122–131. [Google Scholar] [CrossRef]
- Ho, S.S.; Dhaliwal, S.S.; Hills, A.P.; Pal, S. The effect of 12 weeks of aerobic, resistance or combination exercise training on cardiovascular risk factors in the overweight and obese in a randomized trial. BMC Public Health 2012, 12, 704. [Google Scholar] [CrossRef]
- Irving, B.A.; Lanza, I.R.; Henderson, G.C.; Rao, R.R.; Spiegelman, B.M.; Nair, K.S. Combined training enhances skeletal muscle mitochondrial oxidative capacity independent of age. J. Clin. Endocrinol. Metab. 2015, 100, 1654–1663. [Google Scholar] [CrossRef]
- Izquierdo, M.; Ibañez, J.; Häkkinen, K.; Kraemer, W.J.; Larrión, J.L.; Gorostiaga, E.M. Once weekly combined resistance and cardiovascular training in healthy older men. Med. Sci. Sports Exerc. 2004, 36, 435–443. [Google Scholar] [CrossRef] [PubMed]
- Kadoglou, N.P.; Fotiadis, G.; Kapelouzou, A.; Kostakis, A.; Liapis, C.D.; Vrabas, I.S. The differential anti-inflammatory effects of exercise modalities and their association with early carotid atherosclerosis progression in patients with type 2 diabetes. Diabet. Med. 2013, 30, e41–e50. [Google Scholar] [CrossRef]
- Karavirta, L.; Häkkinen, A.; Sillanpää, E.; García-López, D.; Kauhanen, A.; Haapasaari, A.; Alen, M.; Pakarinen, A.; Kraemer, W.J.; Izquierdo, M.; et al. Effects of combined endurance and strength training on muscle strength, power and hypertrophy in 40-67-year-old men. Scand. J. Med. Sci. Sports 2011, 21, 402–411. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Long, J.; Dan, S.; Johannsen, N.M.; Talamoa, R.; Raghuram, S.; Chung, S.; Kent, K.; Basina, M.; Lamendola, C. Strength training is more effective than aerobic exercise for improving glycaemic control and body composition in people with normal-weight type 2 diabetes: A randomised controlled trial. Diabetologia 2023, 66, 1897–1907. [Google Scholar] [PubMed]
- Piralaiy, E.; Siahkuhian, M.; Nikookheslat, S.D.; Pescatello, L.S.; Sheikhalizadeh, M.; Khani, M. Cardiac Autonomic Modulation in Response to Three Types of Exercise in Patients with Type 2 Diabetic Neuropathy. J. Diabetes Metab. Disord. 2021, 20, 1469–1478. [Google Scholar] [CrossRef]
- Ruangthai, R.; Phoemsapthawee, J. Combined exercise training improves blood pressure and antioxidant capacity in elderly individuals with hypertension. J. Exerc. Sci. Fit. 2019, 17, 67–76. [Google Scholar] [CrossRef]
- Schroeder, E.C.; Franke, W.D.; Sharp, R.L.; Lee, D.C. Comparative effectiveness of aerobic, resistance, and combined training on cardiovascular disease risk factors: A randomized controlled trial. PLoS ONE 2019, 14, e0210292. [Google Scholar] [CrossRef]
- Scott, J.M.; Thomas, S.M.; Herndon, J.E., II; Douglas, P.S.; Yu, A.F.; Rusch, V.; Huang, J.; Capaci, C.; Harrison, J.N.; Stoeckel, K.J.; et al. Effects and tolerability of exercise therapy modality on cardiorespiratory fitness in lung cancer: A randomized controlled trial. J. Cachexia Sarcopenia Muscle 2021, 12, 1456–1465. [Google Scholar] [CrossRef]
- Al-Mhanna, S.B.; Batrakoulis, A.; Mohamed, M.; Alkhamees, N.H.; Sheeha, B.B.; Ibrahim, Z.M.; Aldayel, A.; Muhamad, A.S.; Rahman, S.A.; Afolabi, H.A.; et al. Home-based circuit training improves blood lipid profile, liver function, musculoskeletal fitness, and health-related quality of life in overweight/obese older adult patients with knee osteoarthritis and type 2 diabetes: A randomized controlled trial during the COVID-19 pandemic. BMC Sports Sci. Med. Rehabil. 2024, 16, 125. [Google Scholar] [CrossRef]
- Khalafi, M.; Sakhaei, M.H.; Rosenkranz, S.K.; Symonds, M.E. Impact of concurrent training versus aerobic or resistance training on cardiorespiratory fitness and muscular strength in middle-aged to older adults: A systematic review and meta-analysis. Physiol. Behav. 2022, 254, 113888. [Google Scholar] [PubMed]
- Hickson, R.C. Interference of strength development by simultaneously training for strength and endurance. Eur. J. Appl. Physiol. Occup. Physiol. 1980, 45, 255–263. [Google Scholar] [PubMed]
- Fyfe, J.J.; Bishop, D.J.; Stepto, N.K. Interference between concurrent resistance and endurance exercise: Molecular bases and the role of individual training variables. Sports Med. 2014, 44, 743–762. [Google Scholar] [PubMed]
- Coffey, V.G.; Hawley, J.A. Concurrent exercise training: Do opposites distract? J. Physiol. 2017, 595, 2883–2896. [Google Scholar]
- Thomson, D.M.; Fick, C.A.; Gordon, S.E. AMPK activation attenuates S6K1, 4E-BP1, and eEF2 signaling responses to high-frequency electrically stimulated skeletal muscle contractions. J. Appl. Physiol. 2008, 104, 625–632. [Google Scholar]
- Bolster, D.R.; Crozier, S.J.; Kimball, S.R.; Jefferson, L.S. AMP-activated protein kinase suppresses protein synthesis in rat skeletal muscle through down-regulated mammalian target of rapamycin (mTOR) signaling. J. Biol. Chem. 2002, 277, 23977–23980. [Google Scholar]
- Murach, K.A.; Bagley, J.R. Skeletal muscle hypertrophy with concurrent exercise training: Contrary evidence for an interference effect. Sports Med. 2016, 46, 1029–1039. [Google Scholar]
- Petré, H.; Hemmingsson, E.; Rosdahl, H.; Psilander, N. Development of Maximal Dynamic Strength During Concurrent Resistance and Endurance Training in Untrained, Moderately Trained, and Trained Individuals: A Systematic Review and Meta-analysis. Sports Med. 2021, 51, 991–1010. [Google Scholar] [CrossRef]
- Schumann, M.; Feuerbacher, J.F.; Sünkeler, M.; Freitag, N.; Rønnestad, B.R.; Doma, K.; Lundberg, T.R. Compatibility of Concurrent Aerobic and Strength Training for Skeletal Muscle Size and Function: An Updated Systematic Review and Meta-Analysis. Sports Med. 2022, 52, 601–612. [Google Scholar] [CrossRef]
- Wan, X.; Wang, W.; Liu, J.; Tong, T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med. Res. Methodol. 2014, 14, 135. [Google Scholar] [CrossRef] [PubMed]
- Hozo, S.P.; Djulbegovic, B.; Hozo, I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med. Res. Methodol. 2005, 5, 13. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions; Wiley-Blackwell: Oxford, UK, 2008. [Google Scholar]
- Duval, S.; Tweedie, R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics 2000, 56, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Balducci, S.; Zanuso, S.; Nicolucci, A.; Fernando, F.; Cavallo, S.; Cardelli, P.; Fallucca, S.; Alessi, E.; Letizia, C.; Jimenez, A.; et al. Anti-inflammatory effect of exercise training in subjects with type 2 diabetes and the metabolic syndrome is dependent on exercise modalities and independent of weight loss. Nutr. Metab. Cardiovasc. Dis. 2010, 20, 608–617. [Google Scholar] [CrossRef]
- Bernard, S.; Whittom, F.; Leblanc, P.; Jobin, J.; Belleau, R.; Bérubé, C.; Carrier, G.; Maltais, F. Aerobic and strength training in patients with chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 1999, 159, 896–901. [Google Scholar] [CrossRef]
- Bouchla, A.; Karatzanos, E.; Dimopoulos, S.; Tasoulis, A.; Agapitou, V.; Diakos, N.; Tseliou, E.; Terrovitis, J.; Nanas, S. The addition of strength training to aerobic interval training: Effects on muscle strength and body composition in CHF patients. J. Cardiopulm. Rehabil. Prev. 2011, 31, 47–51. [Google Scholar] [CrossRef]
- Boulé, N.G.; Kenny, G.P.; Larose, J.; Khandwala, F.; Kuzik, N.; Sigal, R.J. Does metformin modify the effect on glycaemic control of aerobic exercise, resistance exercise or both? Diabetologia 2013, 56, 2378–2382. [Google Scholar] [CrossRef]
- Burich, R.; Teljigović, S.; Boyle, E.; Sjøgaard, G. Aerobic training alone or combined with strength training affects fitness in elderly: Randomized trial. Eur. J. Sport. Sci. 2015, 15, 773–783. [Google Scholar] [CrossRef]
- Chang, Y.-H.; Shiow-Ching, S.; Min-Hsin, C.; Chang, Y.-F. Feasibility of Different Exercise Modalities for Community-Dwelling Residents With Physical Inactivity: A Randomized Controlled Trial. J. Nurs. Res. 2023, 31, e301. [Google Scholar] [CrossRef]
- Choi, M.; Ahn, N.; Park, J.; Kim, K. 12-Week Exercise Training of Knee Joint and Squat Movement Improves Gait Ability in Older Women. Int. J. Environ. Res. Public Health 2021, 18, 1515. [Google Scholar] [CrossRef]
- Cortez-Cooper, M.Y.; Anton, M.M.; Devan, A.E.; Neidre, D.B.; Cook, J.N.; Tanaka, H. The effects of strength training on central arterial compliance in middle-aged and older adults. Eur. J. Cardiovasc. Prev. Rehabil. 2008, 15, 149–155. [Google Scholar] [CrossRef]
- Cuff, D.J.; Meneilly, G.S.; Martin, A.; Ignaszewski, A.; Tildesley, H.D.; Frohlich, J.J. Effective exercise modality to reduce insulin resistance in women with type 2 diabetes. Diabetes Care 2003, 26, 2977–2982. [Google Scholar] [CrossRef] [PubMed]
- Delaney, C.L.; Miller, M.D.; Chataway, T.K.; Spark, J.I. A randomised controlled trial of supervised exercise regimens and their impact on walking performance, skeletal muscle mass and calpain activity in patients with intermittent claudication. Eur. J. Vasc. Endovasc. Surg. 2014, 47, 304–310. [Google Scholar] [CrossRef] [PubMed]
- Delecluse, C.; Colman, V.; Roelants, M.; Verschueren, S.; Derave, W.; Ceux, T.; Eijnde, B.O.; Seghers, J.; Pardaens, K.; Brumagne, S.; et al. Exercise programs for older men: Mode and intensity to induce the highest possible health-related benefits. Prev. Med. 2004, 39, 823–833. [Google Scholar] [CrossRef] [PubMed]
- do Amaral, V.T.; Fernandes, B.; Ngomane, A.Y.; Marcal, I.R.; Zanini, G.D.; Ciolac, E.G. Short-term community-based exercise programs in low-income older women: Does exercise intensity and modality matters? Exp. Gerontol. 2021, 156, 111591. [Google Scholar] [CrossRef]
- Feiereisen, P.; Delagardelle, C.; Vaillant, M.; Lasar, Y.; Beissel, J. Is strength training the more efficient training modality in chronic heart failure? Med. Sci. Sports Exerc. 2007, 39, 1910–1917. [Google Scholar]
- Gayda, M.; Choquet, D.; Ahmaidi, S. Effects of exercise training modality on skeletal muscle fatigue in men with coronary heart disease. J. Electromyogr. Kinesiol. 2009, 19, e32–e39. [Google Scholar] [CrossRef]
- Gonzalo-Encabo, P.; Valadés, D.; García-Honduvilla, N.; de Cos Blanco, A.; Friedenreich, C.M.; Pérez-López, A. Exercise type and fat mass loss regulate breast cancer-related sex hormones in obese and overweight postmenopausal women. Eur. J. Appl. Physiol. 2020, 120, 1277–1287. [Google Scholar] [CrossRef]
- Hansen, D.; Eijnde, B.O.; Roelants, M.; Broekmans, T.; Rummens, J.L.; Hensen, K.; Daniels, A.; Van Erum, M.; Bonné, K.; Reyckers, I.; et al. Clinical benefits of the addition of lower extremity low-intensity resistance muscle training to early aerobic endurance training intervention in patients with coronary artery disease: A randomized controlled trial. J. Rehabil. Med. 2011, 43, 800–807. [Google Scholar] [CrossRef]
- Jorge, M.L.M.P.; De Oliveira, V.N.; Resende, N.M.; Paraiso, L.F.; Calixto, A.; Diniz, A.L.D.; Resende, E.S.; Ropelle, E.R.; Carvalheira, J.B.; Espindola, F.S.; et al. The effects of aerobic, resistance, and combined exercise on metabolic control, inflammatory markers, adipocytokines, and muscle insulin signaling in patients with type 2 diabetes mellitus. Metab. Clin. Exp. 2011, 60, 1244–1252. [Google Scholar] [CrossRef]
- Kim, D.I.; Lee, D.H.; Hong, S.; Jo, S.W.; Won, Y.S.; Jeon, J.Y. Six weeks of combined aerobic and resistance exercise using outdoor exercise machines improves fitness, insulin resistance, and chemerin in the Korean elderly: A pilot randomized controlled trial. Arch. Gerontol. Geriatr. 2018, 75, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Kim, C.G.; Seo, T.B.; Kim, H.G.; Yoon, S.J. Effects of 8-week combined training on body composition, isokinetic strength, and cardiovascular disease risk factors in older women. Aging Clin. Exp. Res. 2015, 27, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Lima, L.G.; Bonardi, J.T.M.; Campos, G.O.; Bertani, R.F.; Scher, L.M.L.; Moriguti, J.C.; Ferriolli, E.; Lima, N.K.C. Combined aerobic and resistance training: Are there additional benefits for older hypertensive adults? Clinics 2017, 72, 363–369. [Google Scholar] [CrossRef]
- Marzolini, S.; Brooks, D.; Oh, P.; Jagroop, D.; MacIntosh, B.J.; Anderson, N.D.; Alter, D.; Corbett, D. Aerobic With Resistance Training or Aerobic Training Alone Poststroke: A Secondary Analysis From a Randomized Clinical Trial. Neurorehabil Neural Repair. 2018, 32, 209–222. [Google Scholar] [CrossRef] [PubMed]
- Marzolini, S.; Oh, P.I.; Thomas, S.G.; Goodman, J.M. Aerobic and resistance training in coronary disease: Single versus multiple sets. Med. Sci. Sports Exerc. 2008, 40, 1557–1564. [Google Scholar] [CrossRef]
- Moreno-Cabañas, A.; Ortega, J.F.; Morales-Palomo, F.; Ramirez-Jimenez, M.; Alvarez-Jimenez, L.; Mora-Rodriguez, R. Concurrent endurance and resistance training enhances muscular adaptations in individuals with metabolic syndrome. Scand. J. Med. Sci. Sports 2021, 31, 1440–1449. [Google Scholar] [CrossRef]
- Pedralli, M.L.; Marschner, R.A.; Kollet, D.P.; Neto, S.G.; Eibel, B.; Tanaka, H.; Lehnen, A.M. Different exercise training modalities produce similar endothelial function improvements in individuals with prehypertension or hypertension: A randomized clinical trial Exercise, endothelium and blood pressure. Sci. Rep. 2020, 10, 7628. [Google Scholar] [CrossRef]
- Rossi, F.E.; Fortaleza, A.C.; Neves, L.M.; Buonani, C.; Picolo, M.R.; Diniz, T.A.; Kalva-Filho, C.A.; Papoti, M.; Lira, F.S.; Freitas Junior, I.F. Combined Training (Aerobic Plus Strength) Potentiates a Reduction in Body Fat but Demonstrates No Difference on the Lipid Profile in Postmenopausal Women When Compared With Aerobic Training With a Similar Training Load. J. Strength Cond. Res. 2016, 30, 226–234. [Google Scholar] [CrossRef]
- Rossi, F.E.; Diniz, T.A.; Fortaleza, A.C.S.; Neves, L.M.; Picolo, M.R.; Monteiro, P.A.; Buonani, C.; Lira, F.S.; Freitas, I.F., Jr. Concurrent Training Promoted Sustained Anti-atherogenic Benefits in the Fasting Plasma Triacylglycerolemia of Postmenopausal Women at 1-Year Follow-up. J. Strength Cond. Res. 2018, 32, 3564–3573. [Google Scholar] [CrossRef]
- Sénéchal, M.; Swift, D.L.; Johannsen, N.M.; Blair, S.N.; Earnest, C.P.; Lavie, C.J.; Church, T.S. Changes in body fat distribution and fitness are associated with changes in hemoglobin A1c after 9 months of exercise training: Results from the HART-D study. Diabetes Care 2013, 36, 2843–2849. [Google Scholar] [CrossRef][Green Version]
- Seo, D.I.; Jun, T.W.; Park, K.S.; Chang, H.; So, W.Y.; Song, W. 12 weeks of combined exercise is better than aerobic exercise for increasing growth hormone in middle-aged women. Int. J. Sport. Nutr. Exerc. Metab. 2010, 20, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Sigal, R.J.; Kenny, G.P.; Boulé, N.G.; Wells, G.A.; Prud’homme, D.; Fortier, M.; Reid, R.D.; Tulloch, H.; Coyle, D.; Phillips, P.; et al. Effects of aerobic training, resistance training, or both on glycemic control in type 2 diabetes: A randomized trial. Ann. Intern. Med. 2007, 147, 357–369. [Google Scholar] [CrossRef] [PubMed]
- Sillanpää, E.; Häkkinen, A.; Laaksonen, D.E.; Karavirta, L.; Kraemer, W.J.; Häkkinen, K. Serum basal hormone concentrations, nutrition and physical fitness during strength and/or endurance training in 39-64-year-old women. Int. J. Sports Med. 2010, 31, 110–117. [Google Scholar] [CrossRef]
- Sillanpää, E.; Häkkinen, A.; Nyman, K.; Mattila, M.; Cheng, S.; Karavirta, L.; Laaksonen, D.E.; Huuhka, N.; Kraemer, W.J.; Häkkinen, K. Body composition and fitness during strength and/or endurance training in older men. Med. Sci. Sports Exerc. 2008, 40, 950–958. [Google Scholar] [CrossRef] [PubMed]
- Sparks, L.M.; Johannsen, N.M.; Church, T.S.; Earnest, C.P.; Moonen-Kornips, E.; Moro, C.; Hesselink, M.K.; Smith, S.R.; Schrauwen, P. Nine months of combined training improves ex vivo skeletal muscle metabolism in individuals with type 2 diabetes. J. Clin. Endocrinol. Metab. 2013, 98, 1694–1702. [Google Scholar] [CrossRef]
- Swift, D.L.; Johannsen, N.M.; Earnest, C.P.; Blair, S.N.; Church, T.S. Effect of exercise training modality on C-reactive protein in type 2 diabetes. Med. Sci. Sports Exerc. 2012, 44, 1028–1034. [Google Scholar] [CrossRef]
- Theodorou, A.A.; Panayiotou, G.; Volaklis, K.A.; Douda, H.T.; Paschalis, V.; Nikolaidis, M.G.; Smilios, I.; Toubekis, A.; Kyprianou, D.; Papadopoulos, I.; et al. Aerobic, resistance and combined training and detraining on body composition, muscle strength, lipid profile and inflammation in coronary artery disease patients. Res. Sports Med. 2016, 24, 171–184. [Google Scholar] [CrossRef]
- Timmons, J.F.; Minnock, D.; Hone, M.; Cogan, K.E.; Murphy, J.C.; Egan, B. Comparison of time-matched aerobic, resistance, or concurrent exercise training in older adults. Scand. J. Med. Sci. Sports 2018, 28, 2272–2283. [Google Scholar] [CrossRef]
- Zhou, Y.; Wu, W.L.; Zou, Y.Q.; Huang, W.T.; Lin, S.S.; Ye, J.S.; Lan, Y.T. Benefits of different combinations of aerobic and resistance exercise for improving plasma glucose and lipid metabolism and sleep quality among elderly patients with metabolic syndrome: A randomized controlled trial. Endocr. J. 2022, 69, 819–830. [Google Scholar] [CrossRef]
- Newsome, A.N.M.; Batrakoulis, A.; Camhi, S.M.; McAvoy, C.; Sansone, J.; Reed, R.; Contributors. 2025 ACSM Worldwide Fitness Trends: Future Directions of the Health and Fitness Industry. ACSM’s Health Fit. J. 2024, 28, 11–25. [Google Scholar] [CrossRef]
- Decaria, J.; Sharp, C.; Petrella, R. Scoping review report: Obesity in older adults. Int. J. Obes. 2012, 36, 1141–1150. [Google Scholar]
- Houston, D.K.; Nicklas, B.J.; Zizza, C.A. Weighty concerns: The growing prevalence of obesity among older adults. J. Am. Diet. Assoc. 2009, 109, 1886–1895. [Google Scholar]
- Yarizadeh, H.; Eftekhar, R.; Anjom-Shoae, J.; Speakman, J.R.; Djafarian, K. The effect of aerobic and resistance training and combined exercise modalities on subcutaneous abdominal fat: A systematic review and meta-analysis of randomized clinical trials. Adv. Nutr. 2021, 12, 179–196. [Google Scholar]
- Morze, J.; Rücker, G.; Danielewicz, A.; Przybyłowicz, K.; Neuenschwander, M.; Schlesinger, S.; Schwingshackl, L. Impact of different training modalities on anthropometric outcomes in patients with obesity: A systematic review and network meta-analysis. Obes. Rev. 2021, 22, e13218. [Google Scholar] [PubMed]
- Donnelly, J.E.; Smith, B.; Jacobsen, D.J.; Kirk, E.; DuBose, K.; Hyder, M.; Bailey, B.; Washburn, R. The role of exercise for weight loss and maintenance. Best Pract. Res. Clin. Gastroenterol. 2004, 18, 1009–1029. [Google Scholar]
- Tremblay, A.; Després, J.-P.; Bouchard, C. The effects of exercise-training on energy balance and adipose tissue morphology and metabolism. Sports Med. 1985, 2, 223–233. [Google Scholar]
- Donnelly, J.E.; Blair, S.N.; Jakicic, J.M.; Manore, M.M.; Rankin, J.W.; Smith, B.K. American College of Sports Medicine Position Stand. Appropriate physical activity intervention strategies for weight loss and prevention of weight regain for adults. Med. Sci. Sports Exerc. 2009, 41, 459–471. [Google Scholar] [PubMed]
- Hughes, D.C.; Ellefsen, S.; Baar, K. Adaptations to endurance and strength training. Cold Spring Harb. Perspect. Med. 2018, 8, a029769. [Google Scholar] [PubMed]
- Thomas, D.R. Loss of skeletal muscle mass in aging: Examining the relationship of starvation, sarcopenia and cachexia. Clin. Nutr. 2007, 26, 389–399. [Google Scholar]
- Nilwik, R.; Snijders, T.; Leenders, M.; Groen, B.B.; van Kranenburg, J.; Verdijk, L.B.; van Loon, L.J. The decline in skeletal muscle mass with aging is mainly attributed to a reduction in type II muscle fiber size. Exp. Gerontol. 2013, 48, 492–498. [Google Scholar]
- Csapo, R.; Alegre, L. Effects of resistance training with moderate vs heavy loads on muscle mass and strength in the elderly: A meta-analysis. Scand. J. Med. Sci. Sports 2016, 26, 995–1006. [Google Scholar] [CrossRef]
- Wilson, J.M.; Marin, P.J.; Rhea, M.R.; Wilson, S.M.; Loenneke, J.P.; Anderson, J.C. Concurrent training: A meta-analysis examining interference of aerobic and resistance exercises. J. Strength Cond. Res. 2012, 26, 2293–2307. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, T.R.; Feuerbacher, J.F.; Sünkeler, M.; Schumann, M. The effects of concurrent aerobic and strength training on muscle fiber hypertrophy: A systematic review and meta-analysis. Sports Med. 2022, 52, 2391–2403. [Google Scholar] [CrossRef]
- Beasley, J.M.; Shikany, J.M.; Thomson, C.A. The role of dietary protein intake in the prevention of sarcopenia of aging. Nutr. Clin. Pract. 2013, 28, 684–690. [Google Scholar] [CrossRef] [PubMed]
- van der Burgh, A.C.; Rizopoulos, D.; Ikram, M.A.; Hoorn, E.J.; Chaker, L. Determinants of the Evolution of Kidney Function With Age. Kidney Int. Rep. 2021, 6, 3054–3063. [Google Scholar] [CrossRef] [PubMed]
- Guppy, M.; Thomas, E.T.; Glasziou, P.; Clark, J.; Jones, M.; O’Hara, D.V.; Doust, J. Rate of decline in kidney function with age: A systematic review. BMJ Open 2024, 14, e089783. [Google Scholar] [CrossRef]
- Sirago, G.; Picca, A.; Calvani, R.; Coelho-Júnior, H.J.; Marzetti, E. Mammalian Target of Rapamycin (mTOR) Signaling at the Crossroad of Muscle Fiber Fate in Sarcopenia. Int. J. Mol. Sci. 2022, 23, 13823. [Google Scholar] [CrossRef]
- Levine, M.E.; Suarez, J.A.; Brandhorst, S.; Balasubramanian, P.; Cheng, C.W.; Madia, F.; Fontana, L.; Mirisola, M.G.; Guevara-Aguirre, J.; Wan, J.; et al. Low protein intake is associated with a major reduction in IGF-1, cancer, and overall mortality in the 65 and younger but not older population. Cell Metab. 2014, 19, 407–417. [Google Scholar] [CrossRef]
- Sabag, A.; Najafi, A.; Michael, S.; Esgin, T.; Halaki, M.; Hackett, D. The compatibility of concurrent high intensity interval training and resistance training for muscular strength and hypertrophy: A systematic review and meta-analysis. J. Sports Sci. 2018, 36, 2472–2483. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]



| First Author, Year | Sample Size (Sex) | Health Status | Age (Years) | BMI (kg/m2) | Intervention Groups | Outcomes |
|---|---|---|---|---|---|---|
| Ahtiainen et al., 2009 [34] | 26 (M) | Healthy | CT: 64 ± 3 AT: 58 ± 8 RT: 61 ± 5 | CT: >28 AT: >28 RT: >28 | CT AT RT | Weight, FP, LBM, CSA |
| Aminilari et al., 2017 [35] | 37 (F) | Overweight and T2D | CT: 45–60 AT: 45–60 RT: 45–60 | CT: 29.01 ± 2.57 AT: 30.03 ± 5.48 RT: 29.11 ± 1.92 | CT AT RT | Weight, BMI, FP, MM |
| Balducci et al., 2010 [65] | 42 (26 M, 16 F) | T2D and MetS | CT: 60.60 ± 9.30 AT: 64.30 ± 8.10 | CT: 30.50 ± 0.90 AT: 29.40 ± 1.10 | CT AT | Weight, BMI, WC, FP, |
| Bernard et al., 1999 [66] | 36 (28 M, 8 F) | COPD | CT: 64.00 ± 7.00 AT: 67.00 ± 9.00 | CT: 27.00 ± 5.00 AT: 25.00 ± 4.00 | CT AT | CSA |
| Bouchla et al., 2011 [67] | 20 (16 M, 4 F) | CHF | CT: 56.7 ± 7.2 AT: 50.5 ± 11.4 | CT: 28.6 ± 4.4 AT: 28.1 ± 2.7 | CT AT | LBM, FM |
| Boulé et al., 2013 [68] | 62 (31 M, 31 F) | T2D | 53.1 ± 6.9 | 33.3 ± 6.4 | CT AT RT | Weight, WC |
| Burich et al., 2015 [69] | 33 (6 M, 27 F) | Healthy | CT: 62.70 ± 5.10 AT: 63.60 ± 4.40 | ND | CT AT | Weight, BF |
| Cadore et al., 2010 [36] | 23 (M) | Healthy | CT: 66.80 ± 4.80 AT: 64.40 ± 3.50 RT: 64.00 ± 3.50 | ND | CT AT RT | Weight, FM, BF |
| Campos et al., 2013 [37] | 19 (F) | Healthy | CT1: 62.00 ± 2.50 CT2: 66.00 ± 3.50 AT: 63.60 ± 2.51 RT: 70.00 ± 6.27 | CT1: 28.58 ± 7.63 CT2: 26.24 ± 4.90 AT: 27.40 ± 6.62 RT: 25.32 ± 4.24 | CT1 CT2 AT RT | Weight, WC, FM, FP, MM |
| Chang et al., 2023 [70] | 28 (3 M, 25 F) | Healthy and chronic disease (hypertension, T2D, cancer and MetS) | CT: 58.19 ± 8.16 AT: 59.59 ± 7.03 | ND | CT AT | Weight, BMI, WC, FP, VF |
| Chen et al., 2017 [38] | 45 (8 M, 37 F) | Sarcopenic obesity | CT: 68.50 ± 2.70 AT: 69.30 ± 3.00 RT: 68.90 ± 4.40 | CT: 27.20 ± 2.90 AT: 26.80 ± 3.80 RT: 28.30 ± 4.40 | CT AT RT | Weight, BMI, VF, MM, FP, FM |
| Choi et al., 2021 [71] | 36 (F) | Overweight | CT: 73.19 ± 2.17 AT: 73.11 ± 2.11 | CT: 25.11 ± 3.65 AT: 25.12 ± 2.11 | CT AT | Weight, BMI, FP, LBM |
| Church et al., 2010 [39] | 208 (78 M, 130 F) | T2D | CT: 55.40 ± 8.30 AT: 53.70 ± 9.10 RT: 56.90 ± 8.70 | CT: 35.80 ± 6.20 AT: 34.70 ± 6.10 RT: 34.10 ± 5.40 | CT AT RT | Weight, WC, FM, LBM |
| Cortez-Cooper et al., 2007 [72] | 25 (6 M, 19 F) | Overweight | CT: 51.00 ± 1.00 RT: 52.00 ± 2.00 | CT: 27.00 ± 1.10 RT: 26.80 ± 1.10 | CT RT | Weight, BMI, FP, LBM |
| Cuff et al., 2003 [73] | 19 (F) | T2D | CT: 63.40 ± 6.95 AT: 59.40 ± 5.70 | CT: 33.30 ± 4.74 AT: 32.50 ± 4.20 | CT AT | Weight, VF, CSA |
| Davidson et al., 2009 [40] | 93 (37 M, 56 F) | Obesity | CT1: 67.10 ± 4.50 CT2: 66.50 ± 5.30 AT1: 68.80 ± 6.00 AT2: 69.10 ± 6.50 RT1: 67.40 ± 5.70 RT2: 67.60 ± 4.20 | CT1: 31.10 ± 3.10 CT2: 29.50 ± 3.00 AT1: 29.90 ± 3.00 AT2: 29.20 ± 3.70 RT1: 30.10 ± 2.60 RT2: 30.00 ± 3.40 | CT AT RT | Weight, BMI, WC, VF, MM, FM |
| Delaney et al., 2014 [74] | 35 (26 M, 9 F) | Patients with intermittent claudication (overweight) | CT: 69.40 ± 9.60 AT: 73.40 ± 9.10 | CT: 29.00 ± 5.60 AT: 27.00 ± 4.30 | CT AT | MM |
| Delecluse et al., 2004 [75] | 62 (M) | Overweight | CT1: 63.80 ± 4.80 CT2: 63.70 ± 6.00 AT: 64.50 ± 5.30 | CT1: 27.10 ± 3.40 CT2: 26.80 ± 3.80 AT: 26.60 ± 2.80 | CT1 CT2 AT | WC, FFM, FP |
| Do Amaral et al., 2021 [76] | 28 (F) | Obesity | CT: 67.50 ± 3.40 RT: 66.10 ± 16.00 | CT: 31.00 ± 7.90 RT: 30.00 ± 6.10 | CT RT | Weight, BMI, WC |
| Feiereisen et al., 2007 [77] | 45 (38 M, 7 F) | CHF | CT: 60.60 ± 5.60 AT: 59.40 ± 6.50 RT: 57.90 ± 5.80 | ND | CT AT RT | MV |
| Gayda et al., 2009 [78] | 16 (M) | CHD | 55.00 ± 8.00 | ND | CT AT | Weight, FP, LBM, MV |
| Gonzalo-Encabo et al., 2020 [79] | 23 (F) | Overweight and obese postmenopausal | CT: 58 [56.5, 61.0] AT: 56.5 [55.0, 58.5] | CT: 32.5 [28.5, 37.5] AT: 32.3 [29.6, 35.5] | CT AT | Weight, FM, LBM |
| Hansen et al., 2011 [80] | 47 (44 M, 3 F) | CAD | CT: 60.40 ± 8.90 AT: 58.90 ± 7.20 | CT: 26.60 ± 3.10 AT: 27.00 ± 3.20 | CT AT | FM, LBM |
| Ho et al., 2012 [41] | 48 (6 M, 42 F) | Overweight obese | CT: 53.00 ± 1.30 AT: 55.00 ± 1.20 RT: 52.00 ± 1.10 | CT: 33.30 ± 1.20 AT: 32.70 ± 1.30 RT: 33.00 ± 1.30 | CT AT RT | Weight, BMI, WC, FM, FP |
| Irving et al., 2015 [42] | 30 (15 M, 15 F) | Healthy | CT: 71.00 ± 6.32 AT: 70.00 ± 3.31 RT: 70.00 ± 3.31 | CT: 28.00 ± 3.16 AT: 28.00 ± 3.31 RT: 27.00 ± 3.31 | CT AT RT | FP, FM, LBM, CSA |
| Izquterdo et al., 2004 [43] | 31 (M) | Healthy | CT: 66.40 ± 4.50 AT: 68.20 ± 1.70 RT: 64.80 ± 2.60 | CT: 25.20 ± 8.70 AT: 27.60 ± 2.70 RT: 29.60 ± 4.10 | CT AT RT | Weight, BMI, FP, FFM, CSA |
| Jorge et al., 2011 [81] | 36 (14 M, 22 F) | T2D | CT: 57.90 ± 8.06 AT: 52.09 ± 8.71 RT: 54.10 ± 8.93 | CT: 31.23 ± 3.88 AT: 29.29 ± 2.19 RT: 30.89 ± 4.09 | CT AT RT | BMI |
| Kadoglou et al., 2013 [44] | 66 (18 M & 48 F) | T2D | CT: 57.90 ± 6.50 AT: 58.30 ± 5.40 RT: 56.10 ± 5.30 | CT: 31.91 ± 2.93 AT: 31.55 ± 3.11 RT: 32.89 ± 3.26 | CT AT RT | BMI, FP |
| Karavirta et al., 2011 [45] | 30 (M) | Healthy | CT: 56.00 ± 7.00 AT: 54.00 ± 8.00 RT: 56.00 ± 6.00 | CT: 26.20 ± 3.20 AT: 25.50 ± 3.40 RT: 26.40 ± 2.90 | CT AT RT | CSA |
| Kim et al., 2018 [82] | 25 (3 M, 22 F) | Healthy | 73.20 ± 4.90 | CT: 24.94 ± 1.63 RT: 25.75 ± 2.60 | CT RT | Weight, BMI, LBM, FP, WC |
| Kobayashi et al., 2023 [46] | 186 (112 M, 74 F) | T2D | CT: 59 (52–68) AT: 59 (54–65) RT: 60 (53–66) | CT: 23.80 (22.9–24.7) AT: 23.60 (22.2–24.7) RT: 23.50 (22.6–24.4) | CT AT RT | LBM, FM |
| Lee et al., 2015 [83] | 19 (F) | Healthy | CT: 68.38 ± 2.93 AT: 67.64 ± 2.82 | CT: 24.23 ± 1.33 AT: 24.09 ± 1.28 | CT AT | Weight, BMI, FM, LBM, FP |
| Lima et al., 2017 [84] | 30 (5 M, 25 F) | Hypertension | CT: 67.80 ± 5.20 AT: 67.80 ± 4.30 | CT: 28.00 ± 3.20 AT: 28.90 ± 3.50 | CT AT | BMI, LBM, FM, WC |
| Marzolini et al., 2018 [85] | 68 (44 M, 24 F) | Chronic stroke with motor impairments | CT: 61.70 ± 10.00 AT: 65.60 ± 13.20 | CT: 27.78 ± 4.30 AT: 25.32 ± 5.10 | CT AT | BMI, LBM, FP |
| Marzolini et al., 2008 [86] | 53 (47 M, 6 F) | CAD | CT1: 62.70 ± 11.45 CT2: 60.90 ± 10.02 AT: 57.90 ± 10.40 | CT1: 25.70 ± 3.01 CT2: 27.60 ± 3.61 AT: 26.80 ± 3.48 | CT AT | LBM, FP |
| Moreno-Cabañas et al., 2021 [87] | 66 (48 M, 18 F) | MetS | 56.00 ± 7.00 | CT: 32.70 ± 5.20 AT: 32.00 ± 4.10 | CT AT | Weight, BMI, FM, FFM, WC |
| Pedralli et al., 2020 [88] | 37 (20 M, 17 F) | Prehypertension or hypertension | CT: 53.80 ± 9.10 AT: 50.90 ± 14.20 RT: 55.10 ± 6.90 | CT: 27.90 ± 5.50 AT: 29.80 ± 4.10 RT: 28.50 ± 6.01 | CT AT RT | Weight, BMI, WC |
| Piralaiy et al., 2021 [47] | 33 (M) | T2D neuropathy | 55.24 ± 8.11 | CT: 30.36 ± 3.02 AT: 30.39 ± 3.36 RT: 28.79 ± 2.59 | CT AT RT | Weight, BMI, FP |
| Rossi et al., 2016 [89] | 52 (F) | Hyper-triacylglycerolemic postmenopause | 61.0 ± 6.3 | CT: 28.3 ± 2.7 AT: 28.4 ± 2.9 | CT AT | Weight, BMI, FM, FP, FFM |
| Rossi et al., 2018 [90] | 29 (F) | Postmenopause | CT: 61.40 ± 5.00 AT: 61.80 ± 7.9 | CT: 30.50 ± 4.30 AT: 28.40 ± 2.90 | CT AT | FM, LBM |
| Ruangthai et al., 2019 [48] | 42 (6 M, 36 F) | Hypertension | CT: 67.30 ± 5.90 AT: 65.60 ± 4.50 RT: 68.00 ± 7.40 | CT: 24.10 ± 2.00 AT: 23.80 ± 2.40 RT: 22.60 ± 2.60 | CT AT RT | Weight, BMI, FM, FP, MM |
| Schroeder et al., 2019 [49] | 52 (21 M, 31 F) | Hypertension, overweight/obesity | CT: 58.00 ± 7.00 AT: 58.00 ± 7.00 RT: 57.00 ± 9.00 | CT: 31.90 ± 5.50 AT: 32.50 ± 5.90 RT: 33.10 ± 5.90 | CT AT RT | Weight, BMI, WC, LBM, FM (kg), FP |
| Scott et al., 2021 [50] | 67 (24 M, 43 F) | Lung cancer survivors with poor cardiorespiratory fitness | CT: 63.00 ± 11.00 AT: 64.00 ± 9.00 RT: 64.00 ± 9.00 | CT: 27.00 ± 4.00 AT: 26.00 ± 6.00 RT: 27.00 ± 6.00 | CT AT RT | Weight, LBM, FP |
| Sénéchal et al., 2013 [91] | 171 (62 M, 109 F) | T2D | CT: 55.90 ± 7.20 AT: 55.10 ± 8.30 RT: 58.10 ± 8.30 | CT: 34.70 ± 5.90 AT: 34.00 ± 5.60 RT: 33.90 ± 5.40 | CT AT RT | Weight, BMI, WC, FP, FM, LBM |
| Seo et al., 2010 [92] | 15 (F) | Healthy postmenopausal | CT: 54.00 ± 3.60 AT: 55.00 ± 4.80 | CT: 24.00 ± 1.90 AT: 27.40 ± 3.40 | CT AT | Weight, BMI, WC, FP |
| Sigal et al., 2007 [93] | 188 (119 M, 69 F) | T2D | CT: 53.50 ± 7.30 AT: 53.90 ± 6.60 RT: 54.70 ± 7.50 | CT: 35.00 ± 9.60 AT: 35.60 ± 10.10 RT: 34.10 ± 9.60 | CT AT RT | Weight, BMI, WC, LBM, FM, FP, VF, CSA |
| Sillanpää et al., 2010 [94] | 70 (F) | Healthy | CT: 51.00 ± 7.00 AT: 53.00 ± 8.00 RT: 52.00 ± 8.00 | CT: 25.00 ± 3.10 AT: 25.10 ± 2.60 RT: 24.70 ± 3.20 | CT AT RT | BMI, FP, LBM |
| Sillanpää et al., 2008 [95] | 42 (M) | Healthy | CT: 56.30 ± 6.80 AT: 54.10 ± 7.70 RT: 54.60 ± 6.10 | CT: 24.70 ± 3.00 AT: 23.70 ± 2.00 RT: 25.30 ± 1.90 | CT AT RT | Weight, BMI, WC, LBM, FP |
| Sparks et al., 2013 [96] | 42 (21 M, 21 F) | T2D | CT: 54.10 ± 6.20 AT: 54.20 ± 6.00 RT: 60.40 ± 7.30 | CT: 37.10 ± 6.80 AT: 33.40 ± 5.80 RT: 33.90 ± 5.20 | CT AT RT | BMI, FP, FFM |
| Swift et al., 2012 [97] | 167 (69 M, 98 F) | T2D | CT: 56.70 ± 7.80 AT: 55.80 ± 7.90 RT: 58.70 ± 8.00 | CT: 35.00 ± 6.20 AT: 33.90 ± 5.70 RT: 34.10 ± 5.40 | CT AT RT | Weight, WC, FP, FM, LBM |
| Theodorou et al., 2016 [98] | 41 (M) | CAD | CT: 64.00 ± 6.00 AT: 61.00 ± 7.00 RT: 62.00 ± 8.00 | CT: 29.80 ± 0.90 AT: 31.10 ± 0.70 RT: 31.60 ± 1.40 | CT AT RT | Weight, BMI, FP |
| Timmons et al., 2018 [99] | 63 (37 M, 26 F) | Healthy | CT: 69.20 ± 2.70 AT: 69.20 ± 3.10 RT: 69.60 ± 4.90 | CT: 27.50 ± 3.70 AT: 24.90 ± 4.0 RT: 26.90 ± 3.60 | CT AT RT | Weight, FP, FM, LBM |
| Zhou et al., 2022 [100] | 66 (21 M, 45 F) | MetS | CT1: 82.82 ± 3.81 CT2: 78.41 ± 6.12 AT: 79.46 ± 4.82 RT: 78.88 ± 5.40 | CT1: 24.54 ± 3.97 CT2: 24.82 ± 2.92 AT: 24.05 ± 3.00 RT: 26.09 ± 3.05 | CT1 CT2 AT RT | BMI, WC |
| First Author, Year | Concurrent Training (CT) | Same Session or Separate | Aerobic Training (AT) | Resistance Training (RT) | Supervised /Non-Supervised | Frequency (Times /Week) | Intervention Duration |
|---|---|---|---|---|---|---|---|
| Ahtiainen et al., 2009 [34] | AT: 30–90 min, under the level of aerobic threshold to a steady pace under the aerobic threshold RT: 3 sets, 15–30 reps, 40–90% 1RM, 1–3 min rests between sets, including leg press, knee extension and flexion, bench press, triceps pushdown, lateral pull-down, sit-up, and elbow flexion | Separate | 30–90 min, under the level of aerobic threshold to a steady pace under the aerobic threshold, cycling | 3 sets, 15–30 reps, 40–90% 1RM, 1–3 min rests between sets, including leg press, knee extension and flexion, bench press, triceps pushdown, lateral pull-down, sit-up, elbow flexion | Supervised | CT: 4 AT: 2 RT: 2 | 21 weeks |
| Aminilari et al., 2017 [35] | AT: 25 min, 50–55% HRmax RT: 3 sets, 8 reps, 50–55% 1RM, including leg extension, prone leg curl, abdominal crunch, biceps, triceps, and seated calf | Same | 25 min, 50–55% HRmax, cycling | 3 sets, 8 reps, 50–55% 1RM, including leg extension, prone leg curl, abdominal crunch, biceps, triceps, and seated calf | ND | 3 | 12 weeks |
| Balducci et al., 2010 [65] | AT: 40 min, 70–80% VO2max RT: 20 min, 80% 1RM, including chest press, lateral pull-down, leg press, and trunk flexion | Same | 60 min, 70–80% VO2max, treadmill/cycling | – | Supervised | 2 | 12 months |
| Bernard et al., 1999 [66] | AT: 30 min, 80% work rate achieved during the baseline incremental exercise test RT: 2–3 sets, 8–10 reps, 60–80% 1RM, including seated press, elbow flexion, shoulder adduction, leg press, and bilateral knee extension | Same | 30 min, 80% work rate achieved during the baseline incremental exercise test, cycling | – | Supervised | 3 | 12 weeks |
| Bouchla et al., 2011 [67] | AT: 20 min, interval training (30-s exercise, 60-s rest), 50% of the workload peak RT: 20 min, 3 sets, 10–12 reps, 30-s rest between the sets; intensity for quadriceps was 55–65% of 2RM, intensity for the hamstrings was that of quadriceps minus 0.5–1 kg, intensity for the upper extremities exercises was set at 10RM including leg extension, leg curls, arm curls, and lateral arm abduction | Same | 40 min, interval (30 s exercise, 60 s rest), 50% of the workload peak, cycling | 40 min, 3 sets, 10–12 reps, 30-s rest between the sets; intensity for quadriceps was 55–65% of 2RM, intensity for the hamstrings was that of quadriceps minus 0.5–1 kg, intensity for the upper extremities exercises was set at 10RM including leg extension, leg curls, arm curls, and lateral arm abduction | Supervised | 3 | 12 weeks |
| Boulé et al., 2013 [68] | AT: 45 min, 75% HRmax RT: 2–3 sets, 7–9 reps, maximum weight including 7 exercises on weight machines | Same | 45 min, 75% HRmax, aerobic training progressed | 2–3 sets, 7–9 reps, maximum weight, including 7 exercises on weight machines | Supervised | 3 | 22 weeks |
| Burich et al., 2015 [69] | AT: 20 min, 65% HRR RT: 3 sets, 10–15 reps, including leg press, calf extension, and knee extension | Same | 40 min, 65% HRR, cycling | – | Supervised | 3 | 12 weeks |
| Cadore et al., 2010 [36] | AT: 20–30 min, 80–100% heart rate at VT2 RT: 2–3 sets, 6–20 reps, until failure including leg press, knee extension, leg curl, bench press, lat pull-down, seated row, triceps curl, biceps curl, and abdominal exercises | Same | 20–30 min, 80–100% heart rate at VT2, cycling | 2–3 sets, 6–20 reps, until failure including leg press, knee extension, leg curl, bench press, lat pull-down, seated row, triceps curl, biceps curl, and abdominal exercises | Supervised | 3 | 12 weeks |
| Campos et al., 2013 [37] | AT: 20–30 min, 65–85% MHR, treadmill RT: 3 sets, 4–20 reps, including chest press, behind the neck lat pull-down, knee extension, knee flexion, biceps pulley, triceps pulley, and leg press 3 sets, 30 reps, curl abdominal on the ground | Same | 20–30 min at 65–85% MHR, treadmill | 3 sets, 4–20 reps, including chest press, behind the neck lat pull-down, knee extension, knee flexion, biceps pulley, triceps pulley, and leg press 3 sets, 30 reps, curl abdominal on the ground | ND | 3 | 12 weeks |
| Chang et al., 2023 [70] | AT: 30 min, implemented core muscle training including side swings, rocking horses, marches, and jumping jacks RT: 3–5 sets, 8–12 reps, 30 s rest between sets, 10 min including free weights or a 25-pound theraband including leg press, chest press, lateral pull-downs, shoulder press, arm curls, and triceps extensions | Same | AT: 30 min, implemented core muscle training including side swings, rocking horses, marches, and jumping jacks | – | Supervised and non-supervised | 3 | 12 weeks |
| Chen et al., 2017 [38] | AT: 40–45 min, combination of dance steps including dance steps such as stepping on the spot, knee lifts, high knee running, rowing arm swings, arm swings, twist steps, arm raises, squats, V steps, mambo steps, diamond steps, and point step jumps RT: 3 sets, 8–12 reps, 60–70% 1RM including shoulder presses, bicep curls, triceps curls, bench presses, deadlifts, leg swings, squats, standing rows, unilateral rows, and split front squats | Separate | 40–45 min, combination dance steps including stepping on the spot, knee lifts, high knee running, rowing arm swings, arm swings, twist steps, arm raises, squats, V steps, mambo steps, diamond steps, and point step jumps | 3 sets, 8–12 reps, 60–70% 1RM, including shoulder presses, bicep curls, triceps curls, bench presses, deadlifts, leg swings, squats, standing rows, unilateral rows, and split front squats | ND | 2 | 8 weeks |
| Choi et al., 2021 [71] | AT: 20 min, 60–70% HRmax/peak RT: 3 sets, 15 reps, 60–70% 1RM including squat, wide squat, tubing band squat, tubing band wide squat, single leg-link exercise, double leg-link exercise, and tubing band leg-link exercise | Same | 50 min, 60–70% HRmax/peak, walking/jogging | – | ND | 3 | 12 weeks |
| Church et al., 2010 [39] | AT: 110 min per week, 50–80% VO2max/peak RT: 1 set, 10–12 reps, including bench press, seated row, shoulder press, pull-down, leg press, extension, flexion, abdominal crunches, and back extensions | ND | 140 min per week, 50–80% VO2max/peak, treadmill | 2–3 sets, 10–12 reps, including bench press, seated row, shoulder press, pull-down, leg press, extension, flexion, abdominal crunches, and back extensions; one set with | Supervised | 3 | 9 months |
| Cortez-Cooper et al., 2007 [72] | AT: 30–45 min, 60–75% HRR, 2 days/week, walking or cycling RT: 1 set, 8–12 reps, 70% 1RM, including seated chest press, horizontal leg press, shoulder press, abdominal crunches, seated hamstring curls, seated row, seated calf raises, low back extension, triceps curls, and bicep dumbbell curls, 2 days/week | Separate | – | 1 set, 8–12 reps, 70% 1RM, including seated chest press, horizontal leg press, shoulder press, abdominal crunches, seated hamstring curls, seated row, seated calf raises, low back extension, triceps curls, and bicep dumbbell curls | Supervised and non-supervised | CT: 4 RT: 3 | 13 weeks |
| Cuff et al., 2003 [73] | AT: 65–75% HRR, treadmill, bicycle, stepper, elliptical trainers, and rowing machines RT: 2 sets, 12 reps, began with light loads and there after progressed including leg press, leg curl, hip extension, chest press, and latissimus pull-down | Same | 75 min, 65–75% HRR, treadmill, bicycle, stepper, elliptical trainer, and rowing machines | – | ND | 3 | 16 weeks |
| Davidson et al., 2009 [40] | AT: 30 min, 60–75% VO2max/peak RT: 1 set with reps to volitional fatigue including chest press, shoulder raise, shoulder flexion, leg extension, leg flexion, triceps extension, biceps curl, abdominal crunches, and modified push-ups | Same | 30 min, 60–75% VO2max/peak, walking | 1 set with reps to volitional fatigue including chest press, shoulder raise, shoulder flexion, leg extension, leg flexion, triceps extension, biceps curl, abdominal crunches, and modified push-ups | Supervised | CT: 3 AT: 5 RT: 3 | 6 months |
| Delaney et al., 2014 [74] | AT: 60 min, walk until claudication pain became unbearable, then rested and repeated the cycle Initial treadmill speed was determined by distance covered in the baseline 6 min walking test If the participant did not experience symptoms within 10 min of walking, the pace or gradient of walking was increased by 10% RT: 3 sets, 8–12 reps, including hamstring curls, seated calf press, knee extension, and hip abduction/adduction | Same | 60 min, walk until claudication pain became unbearable, then rested and repeated the cycle Initial treadmill speed was determined by distance covered in the baseline 6-min walking test; if the participant did not experience symptoms within 10 min of walking, the pace or gradient of walking was increased by 10% | – | Supervised | 2 | 12 weeks |
| Delecluse et al., 2004 [75] | AT: 12–20 min, 60–80% HRR RT1: 2 sets, 8–20 reps, moderate to high resistance load, including leg press, leg extension, leg curl, adductor, abductor, vertical row, chest press, arm curl, shoulder press, and abdomen RT2: 2 sets, 30 reps, low resistance load during including leg press, leg extension, leg curl, adductor, abductor, vertical row, chest press, arm curl, shoulder press, and abdomen | Same | 36–70 min, 60–80% HRR, cycling/walking | – | Supervised | 2–3 | 20 weeks |
| Do Amaral et al., 2021 [76] | AT: 15–30 min, walking at 11–13 RPE RT:1–2 sets, 10–20 reps, 15–17 RPE including bodyweight squat, push-up on the wall or counter, deadlift with a stich or rubber band, shoulder press or shoulder abduction with rubber band, and abdominal crunch on the floor or chair | Same | – | 2–4 sets, 10–20 reps, 15–17 RPE, 20–50 min, including bodyweight squat, push-up on the wall or counter, deadlift with a stich or rubber band, shoulder press or shoulder abduction with rubber band, and abdominal crunch on the floor or chair | Supervised | 2 | 12 weeks |
| Feiereisen et al., 2007 [77] | AT: 20 min, 60–75% VO2max/peak RT: 4 sets, 10 reps, 60–70% 1RM, including pull-down, reverse butterfly, rowing, knee extension, and knee flexion | Same | 40 min, 60–75% VO2max/peak, cycling and treadmill | 4 sets, 10 reps, 60–70% 1RM, including pull-down, reverse butterfly, rowing, arm abduction, knee extension, knee flexion, leg press, calf raises, and trunk flexion and trunk extension | ND | 3 | 3 months |
| Gayda et al., 2009 [78] | AT: 60 min, ventilatory threshold power RT: 3 sets, 10 reps, 40% MVC including quadriceps leg extension and lateral pull-down | Separate | 60 min, ventilatory threshold power, cycling/calisthenics movements, stretching, and respiratory exercises | – | ND | CT: 6 AT: 3 | 7 weeks |
| Gonzalo-Encabo et al., 2020 [79] | AT: 10–20 min, 55–75% of HRR, treadmill, cycle ergometer, and elliptical machine RT: 2–3 sets of 8–12 reps, 65% 1RM, 60–90 s rest, 40 min/session, including chest press, seated row, leg press, leg extension, biceps curl, and triceps extension | Same | 10–60 min, 55–75% of HRR, treadmill, cycle ergometer, and elliptical machine | – | Supervised | 3 | 12 weeks |
| Hansen et al., 2011 [80] | AT: 40 min, 65% VO2max/peak RT: 2 sets, 12–20 reps, 65% 1RM, including leg extension leg press | Same | 40 min, 65% VO2max/peak, cycling/walking/arm cranking | – | Supervised | 3 | 6 weeks |
| Ho et al., 2012 [41] | AT: 15 min, treadmill walking RT: 2 sets, 15 min, including leg press, leg curl, leg extension, bench press, and rear deltoid row, with each set completed in ~30 s with 1 min rest | Same | 30 min, 60% HRR, treadmill walking | 4 sets, 8–12 reps, 10RM, 30 min, including leg press, leg curl, leg extension, bench press, and rear deltoid row, with each set completed in ~30 s with 1 min rest | Supervised and non-supervised | 5 | 12 weeks |
| Irving et al., 2015 [42] | AT: 30 min, 65% VO2max/peak RT: 4 sets, 8–10 reps, multiple muscle groups | Same | 60 min, 65% VO2max/peak, cycling | 4 sets, 8–10 reps, multiple muscle groups | Supervised | CT: 5 AT: 5 RT: 4 | 8 weeks |
| Izquterdo et al., 2004 [43] | AT: 30–40 min, 70–90% HRmax, cycling at a constant rate of 60 rmp RT: Week 1–8: 3–4 sets, 10–15 reps, 50–70% 1RM, 45–60 min Week 8–16: 3–5 sets, 5–6 reps, 70–80% 1RM, 45–60 min Week 1–16: exercises included bilateral leg press and bilateral knee extension exercises, the bench press, chest press, lateral pull-down, and/or shoulder press, abdominal crunch and/or rotary torso and/or another exercise for the trunk extensors, and the standing leg curl and/or adductor–abductor exercises Week 8–6: 3–4 sets, 6–8 reps including a part (20%) of the leg extensor and bench press sets with the loads ranging from 30% to 50% and 30% to 40% of the maximum, respectively | Separate | 30–40 min, 70–90% HRmax, cycling at a constant rate of 60 RPM | Week 1–8: 3–4 sets, 10–15 reps, 50–70% 1RM, 45–60 min Week 8–16: 3–5 sets, 5–6 reps, 70–80% 1RM, 45–60 min Week 1–16: exercises included bilateral leg press and bilateral knee extension exercises, the bench press, chest press, lateral pull-down, and/or shoulder press, abdominal crunch and/or rotary torso and/or another exercises for the trunk extensors, and the standing leg curl and/or adductor-abductor exercises Week 8–6: 3–4 sets, 6–8 reps, including a part (20%) of the leg extensor and bench press sets with the loads ranging from 30% to 50% and 30% to 40% of the maximum, respectively | Supervised | CT: 1 AT: 1 RT: 2 | 16 weeks |
| Jorge et al., 2011 [81] | AT: 30 min, cycling at the heart rate corresponding to the lactate threshold RT: 30 min, including leg press, bench press, lat pull-down, seated rowing, shoulder press, abdominal curls, and knee curls | Same | 60 min, cycling at the heart rate corresponding to the lactate threshold | 60 min, including leg press, bench press, lat pull-down, seated rowing, shoulder press, abdominal curls, and knee curls | Supervised | 3 | 12 weeks |
| Kadoglou et al., 2013 [44] | Half the volume of AT: 60 min, 60–75% HRmax/peak, walking or running, and cycling or calisthenics RT: 2–3 sets, 8–10 reps, 60–80% 1RM, including seated leg press, knee extension, knee flexion, chest press, lat pull-down, overhead press, biceps curl, and triceps extension) | Same and Separate | 60 min, 60–75% HRmax/peak, walking or running, cycling or calisthenics | 2–3 sets, 8–10 reps, 60–80% 1RM, including seated leg press, knee extension, knee flexion, chest press, lat pull-down, overhead press, biceps curl, and triceps extension | Supervised | 4 | 6 months |
| Karavirta et al., 2011 [45] | AT: Weeks 1–7: 30 min below the level of the aerobic threshold Weeks 5–7: intensity was above the aerobic threshold by a 10 min interval in the middle of the sessions Weeks 8–14: 45 min included a 10 min interval between the aerobic-anaerobic thresholds and a 5 min interval above the anaerobic threshold The other weekly training session involved 60 min of cycling below the aerobic threshold Weeks 15–21: one of weekly sessions lasted for 60 min, included 2 10 min intervals between the aerobic-anaerobic thresholds, 2 5 min intervals above the anaerobic threshold and 30 min below the aerobic threshold; the other weekly session included 90 min of cycling at a steady pace below the aerobic threshold RT:1st cycle: 3 sets, 12–20 reps, 40–60% 1RM RT 2nd cycle: 2–4 sets, 5–12 reps, 60–80% 1RM RT 3rd cycle: 2–4 sets, 5–8 reps, 70–85% 1RM Including leg press, knee extension, leg curl, seated calf raise, hip abduction or adduction, bench press, biceps curl, triceps push-down, lateral pull-down, abdominal crunch, and seated back extension. In addition, ~20% of the leg press, knee extension and bench press exercises were performed with light loads of 40–50% of 1RM and 5–8 reps | Separate | Weeks 1–7: 30 min, below the level of the aerobic threshold. Weeks 5–7: intensity was above the aerobic threshold by a 10 min interval in the middle of the sessions Weeks 8–14: 45 min included a 10 min interval between the aerobic-anaerobic thresholds and a 5 min interval above the anaerobic threshold; the other weekly training session involved 60 min of cycling below the aerobic threshold Weeks 15–21: one of weekly sessions lasted for 60 min, included two 10 min intervals between the aerobic-anaerobic thresholds, two 5 min intervals above the anaerobic threshold and 30 min below the aerobic threshold; the other weekly session included 90 min of cycling at a steady pace below the aerobic threshold | 1st cycle: 3 sets, 12–20 reps, 40–60% 1RM 2nd cycle: 2–4 sets, 5–12 reps, 60–80% 1RM 3rd cycle: 2–4 sets, 5–8 reps, 70–85% 1RM Including leg press, knee extension, leg curl, seated calf raise, hip abduction or adduction, bench press, biceps curl, triceps push-down, lateral pull-down, abdominal crunch, and seated back extension In addition, 5–8 reps, 40–50% 1RM, ~20% of the leg press, knee extension and bench press exercises were performed with light loads | Supervised | AT and RT: 2 CT: 4 | 21 weeks |
| Kim et al., 2018 [82] | AT: 2 sets, 6–8 RPE, 20 min, including sky-walk and cross-country RT: 1–3 sets, 12–15 reps, 6–8 RPE, 50 min, including pull weight, chair pull, leg extension | Same | – | 1–3 sets, 12–15 reps, 6–8 RPE, 70 min, including pull weight, chair pull, leg extension | Supervised | 3 | 6 weeks |
| Kobayashi et al., 2023 [46] | AT: 50–80% peak MET, AT was standardized to body weight at 41.8 kJ (kcal) kg body weight−1 week−1 on a treadmill, an elliptical, or a stationary bike RT: 1 set including bench press, seated row, shoulder press, pull-down, leg press, extension and flexion, and abdominal crunches, and back extensions, 8–12 reps | Same | 50–80% peak MET, AT was standardized to body weight at 50.2 kJ (kcal) kg body weight−1 week−1 on a treadmill, an elliptical or a stationary bike | 2 sets (including bench press, seated row, shoulder press, and pull-down), 3 sets (including leg press, extension and flexion), and 2 sets (including abdominal crunches and back extensions), 8–12 reps | Supervised | 3 | 9 months |
| Lee et al., 2015 [83] | AT: 40 min, 40–70% HRR RT: 2 sets, 15–20 reps, 10–13 RPE including ankle dorsiflexion, ankle plantar flexion, ankle eversion, ankle inversion, leg press, knee extension, knee flexion, hip extension, hip flexion, hip abduction, hip adduction, crunches, and hyperextensions | Separate | 40 min, 40–70% HRR, treadmill | – | ND | 5 | 8 weeks |
| Lima et al., 2017 [84] | AT: 20–30 min, treadmill ergometer, intensity was based on the physical conditioning of each participant RT: 1–2 sets, 15 reps upper limbs, 20 reps trunk and lower limbs, 50–60% 1RM, including leg press 45°, bench press, extensor bench, handle front, flexor bench-seated, upright row, plantar flexion, seated row, and abdominals | Same | 20–30 min, treadmill ergometer, intensity was based on the physical conditioning of each participant | – | Supervised | 3 | 10 weeks |
| Marzolini et al., 2018 [85] | AT: 20–60 min, 60–80% HRR RT: 1–2 sets, 10–15 reps, 50–70% 1RM including lunge, squat, abdominal curl-up, heel raise, bicep curl, supine triceps extension, affected-side hip flexion/extension, affected-side ankle dorsiflexion, and single-limb knee extension and flexion | Separate | 20–60 min, 60–80% HRR, cycling/walking | – | Supervised and non- supervised | 5 | 6 months |
| Marzolini et al., 2008 [86] | AT: 30–60 min, 60–80% VO2max/peak RT: 1–3 set, 10–15 reps, 60–75% 1RM, including bent over dumbbell row, half squat, alternating right and left arm bicep curl, heel raises, standing lateral raise, leg curl, supine lateral raise, curl up abdominal exercise, tricep extension, and four-point alternate arm and leg lift | Separate | 30–60 min, 60–80% VO2max/peak, cycling/jogging | – | Supervised and non- supervised | 5 | 24 weeks |
| Moreno-Cabañas et al., 2021 [87] | AT: 4 × 4 min, 90% HRmax/peak, 3 min recovery 70% HRmax/peak RT: 3 sets, 10 reps, 20–30% 1RM, including squat, lunge, and dead lift | Same | 5 × 4 min, 90% HRmax/peak; 3 min recovery 70% HRmax/peak, cycling | – | Supervised | 3 | 16 weeks |
| Pedralli et al., 2020 [88] | AT: 20 min, 50–75% HRR RT: 2 sets, 12 reps, 20 min, 60–80% 1RM, including leg press, bench press, knee extension, biceps direct threading, knee flexion, and low row | Same | 40 min, 50–70% HRR or 11–14 RPE, including cycle ergometer | 4 sets, 8–12 reps, ~40 min with 60–80% 1RM including leg press, bench press, knee extension, biceps direct threading, knee flexion, and low row | ND | 2 | 8 weeks |
| Piralaiy et al., 2021 [47] | AT: 25–45 min, 70% MHR RT: 1 set, each exercise at the maximum weight that could be lifted 8–12 times including bench press, seated row, shoulder press, pull-down, leg press/extension/flexion, abdominal crunches, and back extensions | Separate | 25–45 min, 70% MHR, full aerobic-training program | 2–3 sets, each exercise at the maximum weight that could be lifted 8–12 times including bench press, seated row, shoulder press, pull-down, leg press/extension/flexion, abdominal crunches, and back extensions | Supervised | CT: 5 AT: 3 RT: 3 | 12 weeks |
| Rossi et al., 2016 [89] | AT: 30 min, 100% critical velocity RT: 3 sets, 8–15 reps, 60 s between sets, 30 min including 45° leg press, leg extension, leg curl, bench press, seated row, arm curl, triceps extension, side elevation with dumbbells, and abdominal exercises | Same | 50 min, 100% critical velocity | – | ND | ND | 16 weeks |
| Rossi et al., 2018 [90] | AT: 30 min, 100% of critical velocity RT: 3–4 sets, 8–15 reps at ~65–80% of maximum repetitions, 60–90 s between sets, 27 min including 45° leg press, leg extension, leg curl, bench press, seated row, arm curl, triceps extension, side elevation with dumbbells, and abdominal exercises | Same | 52 min, 100% of critical velocity | – | ND | ND | 16 weeks |
| Ruangthai et al., 2019 [48] | AT: 20 min, 50–70% HRmax, including walk with arms up, heel hit behind, tiptoe, arms adduction/abduction, and knee lift RT: 2 sets, 8–12 reps, 50–80% 1RM, 60 min including squat, legs raise, knee extension, unilateral knee flexion, leg adduction/abduction, leg kick back, shoulder press, bench press, bicep curl, triceps dip, lateral flexion, sit-up, and back extension | Same | 60 min, 50–70% HRmax, including walk with arms up, heel hit behind, tiptoe, arms adduction/abduction, and knee lift | 3 sets, 10–15 reps, 50–80% 1RM, 60 min, including squat, legs raise, knee extension, unilateral knee flexion, leg adduction/abduction, leg kick back, shoulder press, bench press, bicep curl, triceps dip, lateral flexion, sit-up, and back extension | Supervised and non-supervised | 3 | 24 weeks |
| Schroeder et al., 2019 [49] | AT: 30 min, 40–80% HRR RT: 2 sets, 10–20 reps, maximal load including chest press, pull-down, lower-back extension, abdominal crunch, torso rotation, leg press, leg curl, and hip abduction | Same | 60 min, 40–80% HRR, treadmill/cycling | 2–3 sets, 10–20 reps, maximal load including chest press, shoulder press, pull-down, lower-back extension, abdominal crunch, torso rotation, biceps curl, triceps extension, leg press, quadriceps extension, leg curl, and hip abduction | Supervised | 3 | 8 weeks |
| Scott et al., 2021 [50] | Weeks 1–8: 3 AT plus RT sessions per week Weeks 9–16: 2 AT plus RT and 1 interval AT sessions per week CT followed AT and RT dosing schedules for a total of 30–90 min per session; within each session, AT was performed first followed by RT | Same | 20–60 min, 55->95% of VO2peak, interval sessions consisted of 60–120 s at VO2peak followed by 120–180 s of active recovery for 4–10 intervals per session consisted of stationary cycle ergometry | 2–3 sets, 6–18 reps, 50–85% including chest press, seated row, lateral pull-down, pec deck, bicep curl, triceps extension, push-up, leg press, leg curl, leg extension, hip abduction, hip adduction, step up, and sit-to-stand | Supervised | 3 | 16 weeks |
| Sénéchal et al., 2013 [91] | AT: an aerobic exercise dose of 10 kcal/kg body weight per week RT:1 set of 10–12 reps including bench press, seated row, shoulder press, lat pull-down, leg press, leg extension, and leg curl, abdominal crunches, and back extensions | Separate | 50–80% VO2peak | 2 sets including bench press, seated row, shoulder press, and lat pull-down, 3 sets including leg press, leg extension, and leg curl, and 2 sets of abdominal crunches and back extensions, 10–12 reps | Supervised | AT: 3–5 RT: 3 | 9 months |
| Seo et al., 2010 [92] | AT: 60–80% HRR, walked and aerobic exercise RT: 3 sets at 50–70% of 1RM, 30–40 min including seated chest press, lat pull-down, seated leg extension, leg curl, biceps curl, seated triceps extension, shoulder press, and sit-up | ND | 60–80% HRR, walked and aerobic exercise | – | Supervised | 3 | 12 weeks |
| Sigal et al., 2007 [93] | AT: 15–45 min, 60–75% HRmax treadmill/bicycle ergometers RT: 2–3 sets, the maximum weight that could be lifted 7–9 times | Same | 15–45 min, 60–75% HRmax treadmill/bicycle ergometers | 2–3 sets, the maximum weight that could be lifted 7–9 times | Supervised and non-supervised | 3 | 6 months |
| Sillanpää et al., 2010 [94] | AT plus RT | ND | Week 1–5: intensity was under the level of aerobic threshold and lasted 30 min Weeks 5–7: intensity was between the aerobic and anaerobic thresholds to accustom to higher intensities Weeks 8–14: intensity was under the aerobic threshold (60 min) and every second session included 45 min cycling with intensities varying from under the aerobic threshold to over the anaerobic threshold; during the last training weeks, every other training session included 75–90 min of cycling at a steady pace under the aerobic threshold and every other session 50–60 min of cycling with intensities varying from under the aerobic threshold to over the anaerobic threshold | 3–4 sets, 40–90% 1RM, 1–3 min rest between sets and exercises, 6–20 reps averaged length 60–90 min, including leg press, knee extension, bilateral or unilateral knee flexion, bench press, triceps pushdown, lateral pull-down, sit-up exercises for the trunk flexors or another exercise for the trunk extensors, and bilateral/unilateral elbow flexion exercise or leg adduction/abduction exercise | Supervised | 2 | 21 weeks |
| Sillanpää et al., 2008 [95] | AT:1st cycle: 30 min under and above their aerobic threshold 2nd cycle: 45 min (15 min under the level of aerobic threshold, 10 min between the aerobic-anaerobic thresholds, 5 min above anaerobic threshold, and 15 min again under the aerobic threshold) 3rd cycle: 60 min (30 min under the level of aerobic threshold, 2 × 10 min between the aerobic-anaerobic thresholds, 2 × 5 min above anaerobic threshold); every other training session included 90 min of cycling at a steady pace under the aerobic threshold including bicycle ergometer RT:40–90% 1RM, 60–90 min including leg press, knee extension, bench press, triceps pushdown, or lateral pull-down, sit-up, bilateral/unilateral elbow flexion, or leg adduction/abduction | Separate | 1st cycle: 30 min under and above their aerobic threshold 2nd cycle: 45 min (15 min under the level of aerobic threshold, 10 min between the aerobic and anaerobic thresholds, 5 min above the anaerobic threshold, and 15 min again under the aerobic threshold) 3rd cycle: 60 min (30 min under the level of aerobic threshold, 2 × 10 min between the aerobic and anaerobic thresholds, 2 × 5 min above the anaerobic threshold); every other training session include 90 min of cycling at a steady pace under the aerobic threshold including bicycle ergometer | 40–90% 1RM, 60–90 min including leg press, knee extension, bench press, triceps pushdown, or lateral pull-down, sit-up, and bilateral/unilateral elbow flexion or leg adduction/abduction | Supervised | CT: 4 AT: 2 RT: 2 | 21 weeks |
| Sparks et al., 2013 [96] | AT: (10 kcal/kg body weight/week) at 50–80% VO2peak RT: 1 set including bench press, seated row, shoulder press, lat pull-down, abdominal crunches, back extensions, leg press, extension, and flexion, 10–12 reps, 45–50 min | Separate | 150 min/week moderate intensity exercise (12 kcal/kg body weight/week) at 50–80% VO2peak | 2 sets (bench press, seated row, shoulder press, lat pull-down, abdominal crunches and back extensions) 3 sets (leg press, extension, and flexion), 10–12 reps, 45–50 min | Supervised | 3 | 9 months |
| Swift et al., 2012 [97] | AT: (10 kcal/kg body weight/week), 50–80% VO2max RT: 1 set, 10–12 reps, including bench press, seated row, shoulder press, lat pull-down, abdominal crunches, back extensions, leg press, extension, and flexion, | Separate | (12 kcal/kg body weight/week), 50–80% VO2max | 2 sets, 10–12 reps (bench press, seated row, shoulder press, lat pull-down, abdominal crunches and back extensions), 3 sets (leg press, extension, and flexion), | Supervised | 3 | 9 months |
| Theodorou et al., 2016 [98] | AT: 2 sets, (10 min each), 60–85% HRmax/peak running treadmill or cycling ergometer RT: 1 set, 12–15 reps, 60% 1RM, including abdominal, rotary torso, shoulder press, leg extension, leg curl, pulley, chest press, and leg press | Same | 40 min, 60–85% HRmax/peak. treadmill/cycling | 2 sets, 12–15 reps, 60% 1RM, including abdominal, rotary torso, shoulder press, leg extension, leg curl, pulley, chest press, and leg press | Supervised | 3 | 8 months |
| Timmons et al., 2018 [99] | AT: 3 × 4 min, 80% HRmax/peak, 1 min recovery RT: 2 sets, 15 reps, 60% 1RM, including leg press, seated row, chest press, lat pull-down, leg extension, and tricep dips | Same | 6 × 4 min, 80% HRmax/peak, 1 min recovery, cross trainer or cycle ergometer | 4 sets, 15 reps, 60% 1RM, including leg press, seated row, chest press, lat pull-down, leg extension, and tricep dips | Supervised | 3 | 12 weeks |
| Zhou et al., 2022 [100] | AT: 50 min, 40–60% MHR treadmill/cycling/elliptical RT: 3 sets, 10–13 reps, 45–65% 1RM, 2 min rest between sets, 50 min, including chest press, back extension, shoulder press, leg press, leg extension/leg flexion, bicep curl/triceps extension, abdominal crunch, lat pull-down, and seated overhead press using dumbbells | Separate | 50 min, 40–60% MHR treadmill/cycling/elliptical | 3 sets, 10–13 reps, 45–65% 1RM, 2 min rest between sets, 50 min, including chest press, back extension, shoulder press, leg press, leg extension/leg flexion, bicep curl/triceps extension, abdominal crunch, lat pull-down, and seated overhead press using dumbbells | Supervised | 3 | 12 weeks |
| Outcome | Trials | WMD or SMD (95% CI) | p-Value | |
|---|---|---|---|---|
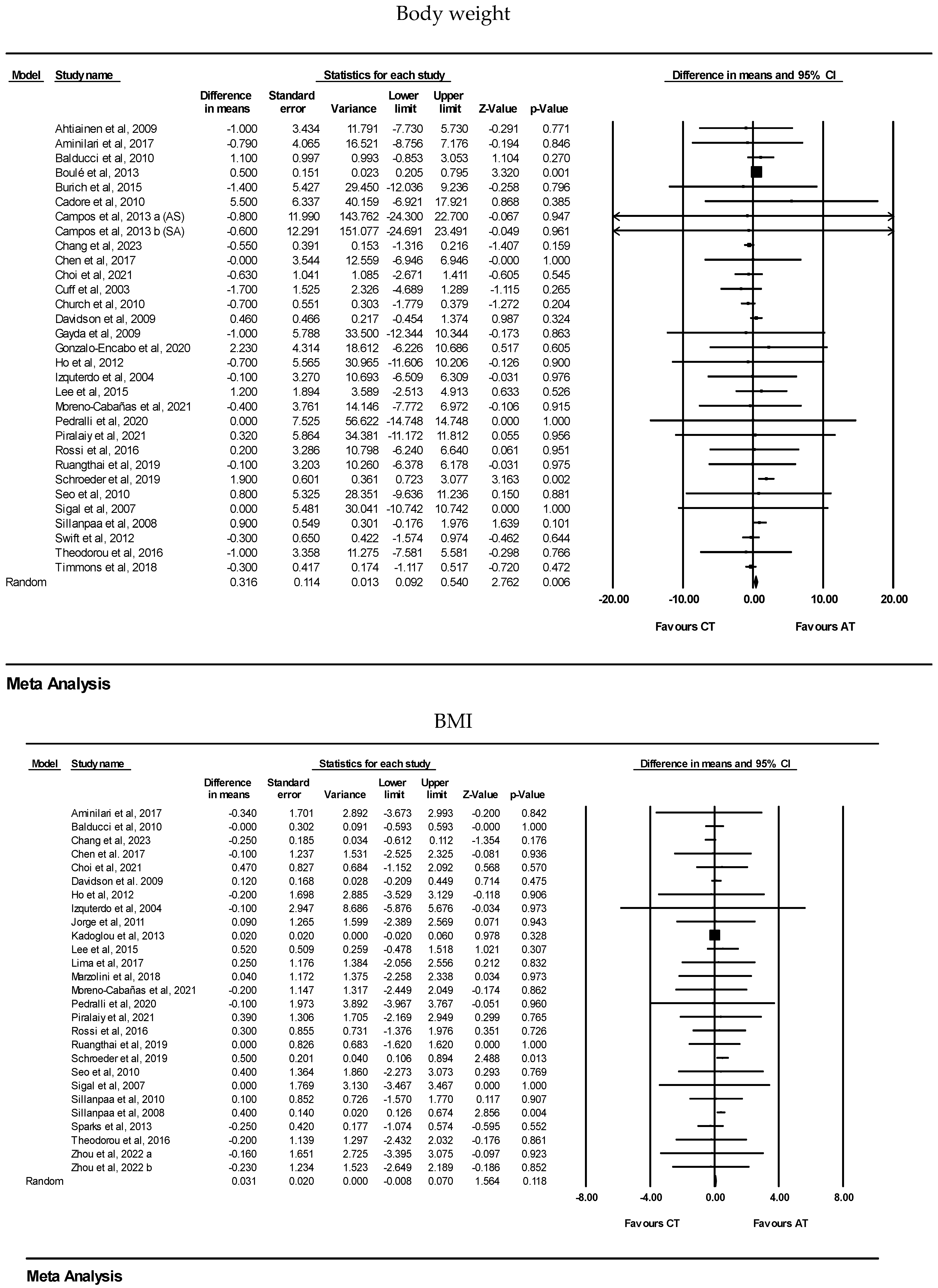
| CT vs. AT | Body weight | 31 | WMD: 0.31 kg (95% CI: 0.09 to 0.54) | 0.006 |
| BMI | 27 | WMD: 0.03 kg·m2 (95% CI: −0.00 to 0.07) | 0.11 | |
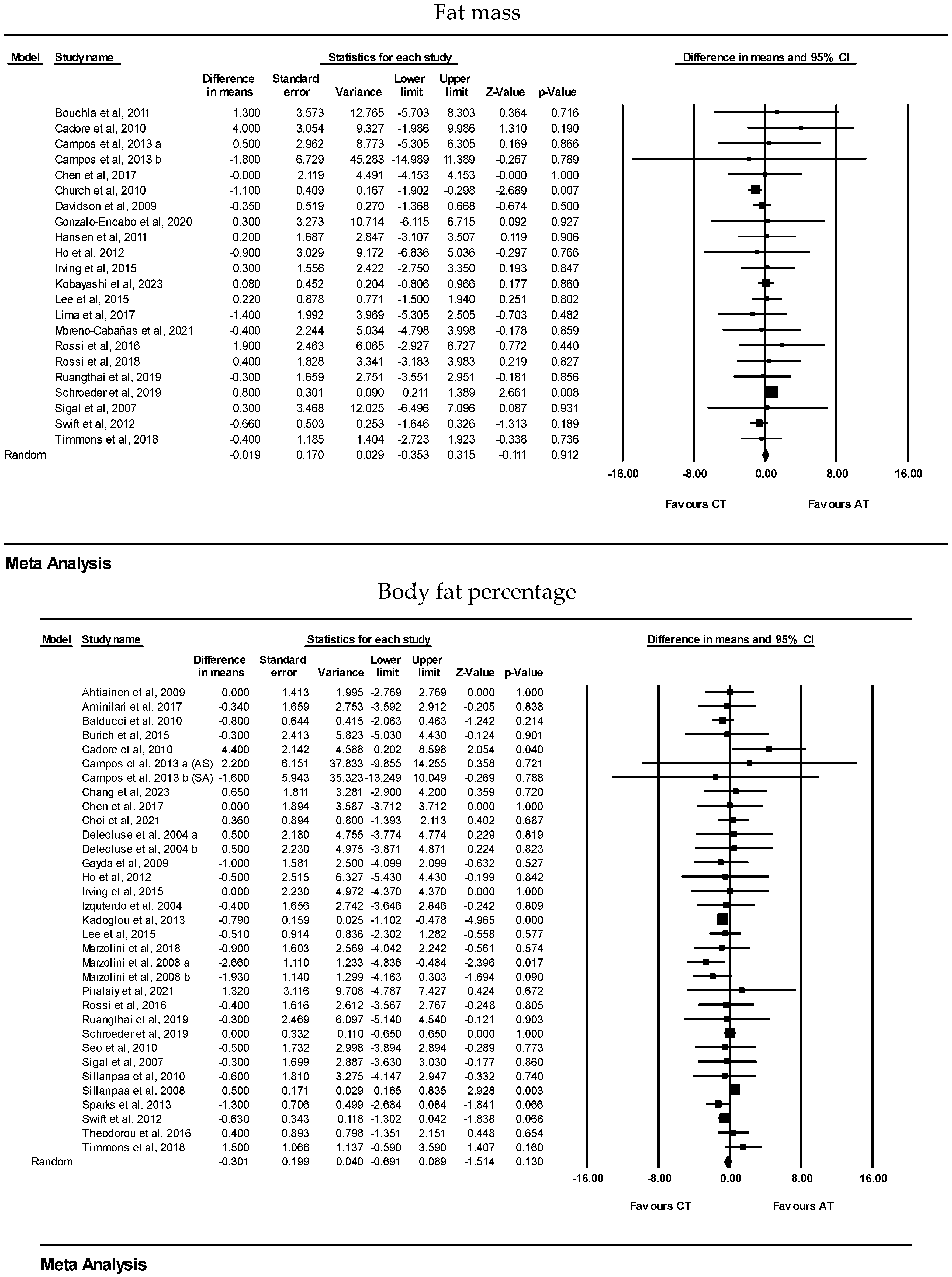
| Body fat% | 33 | WMD: −0.30% (95% CI: −0.69 to 0.08) | 0.13 | |
| Fat mass | 22 | WMD: −0.01 kg (95% CI: −0.35 to 0.31) | 0.91 | |
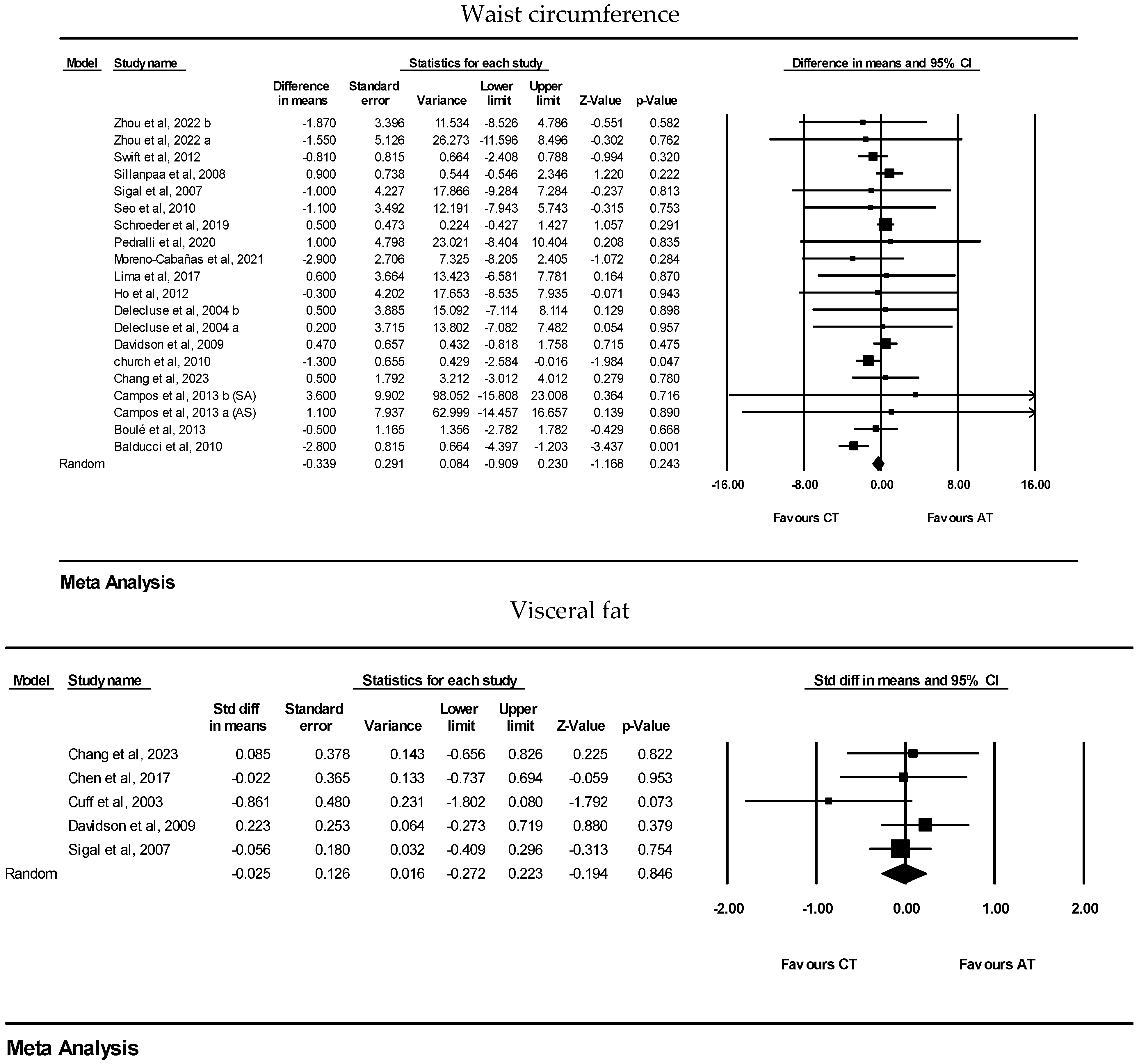
| Waist circumference | 20 | WMD: −0.33 cm (95% CI: −0.90 to 0.23) | 0.24 | |
| Visceral fat | 5 | SMD: −0.02 (95% CI: −0.27 to 0.22) | 0.84 | |
| Lean body mass | 27 | WMD: 0.51 kg (95% CI: 0.29 to 0.73) | 0.001 | |
| Muscle mass/volume | 10 | SMD: 0.22 (95% CI: −0.01 to 0.45) | 0.06 | |
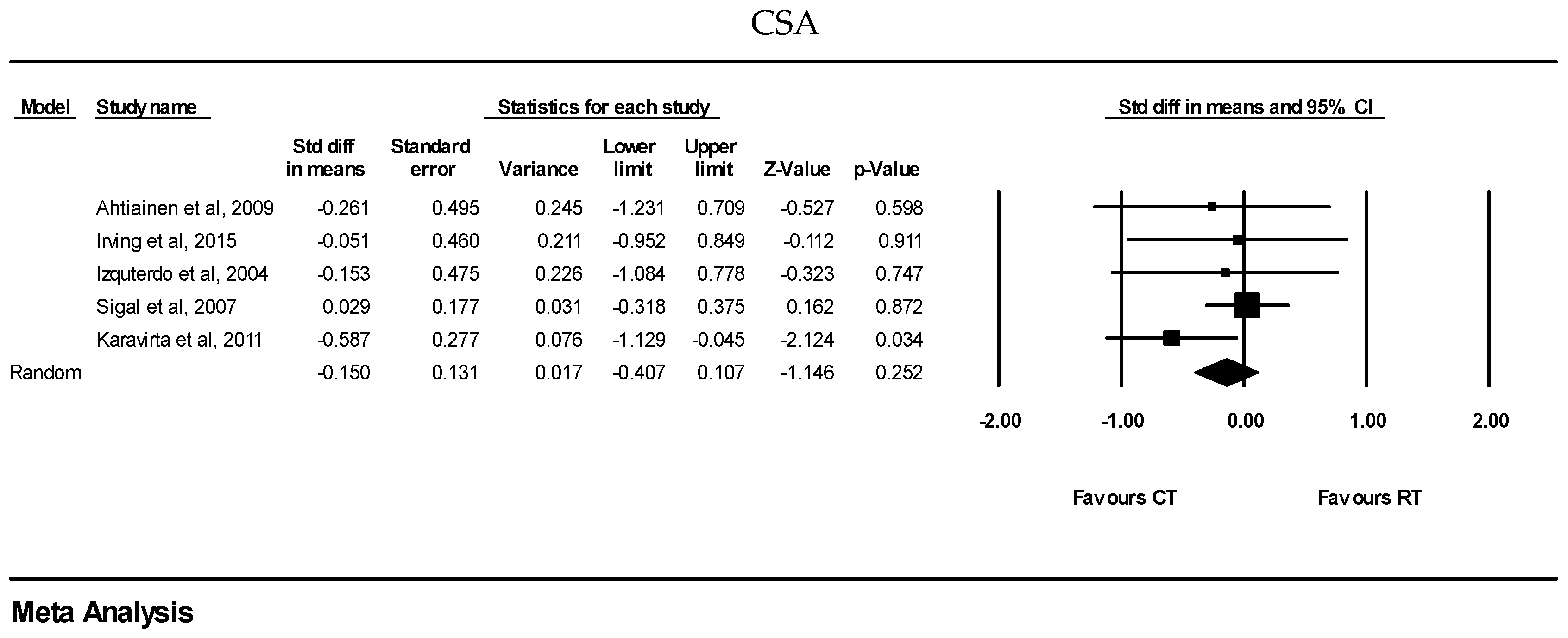
| CSA | 7 | SMD: 0.21 (95% CI: −0.02 to 0.44) | 0.07 | |
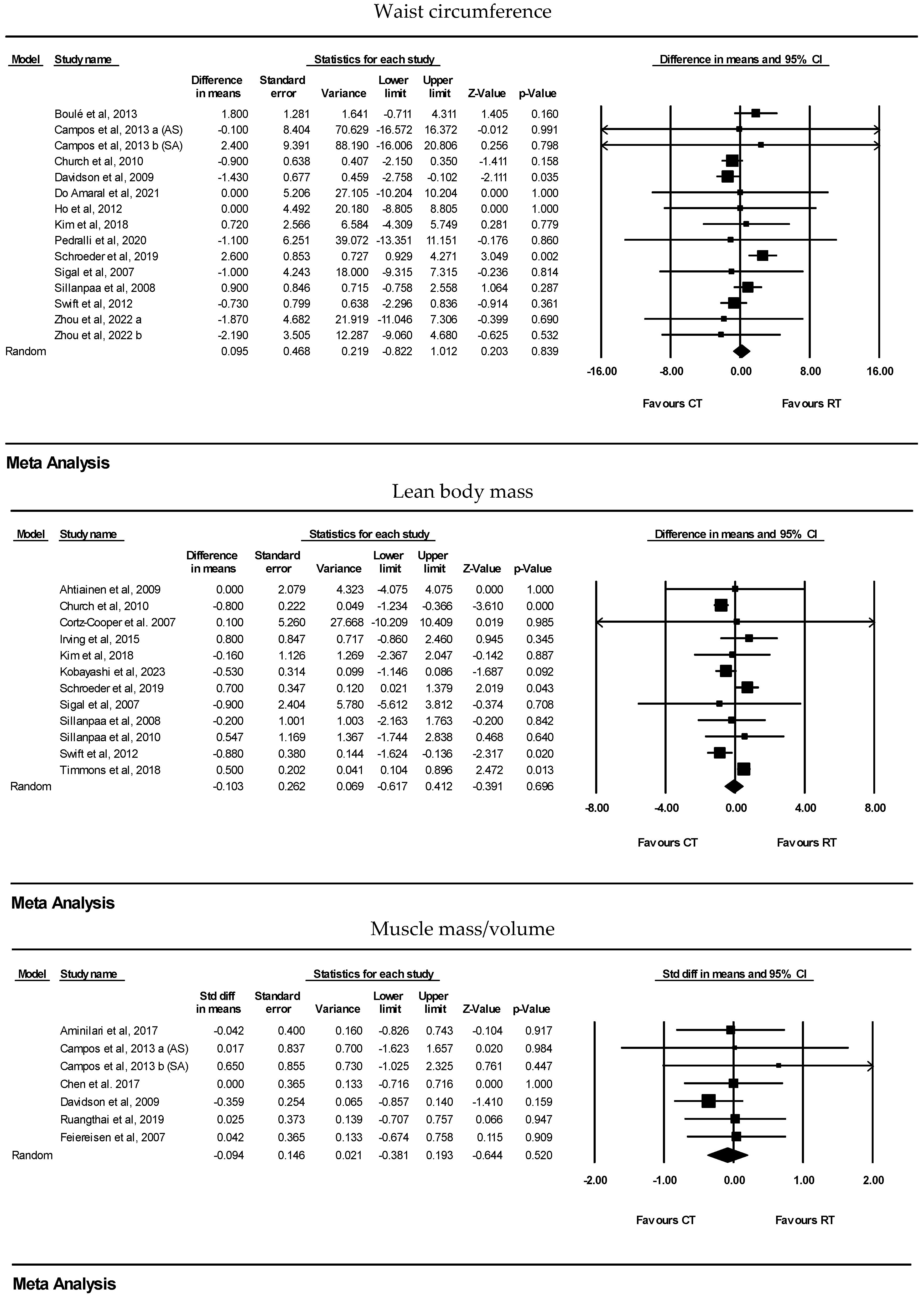
| CT vs. RT | Body weight | 23 | WMD: −0.39 kg (95% CI: −0.96 to 0.16) | 0.16 |
| BMI | 21 | WMD: −0.19 kg·m2 (95% CI: −0.43 to 0.04) | 0.10 | |
| Body fat% | 21 | WMD: −0.25% (95% CI: −0.74 to 0.23) | 0.36 | |
| Fat mass | 13 | WMD: −0.56 kg (95% CI: −1.23 to 0.10) | 0.09 | |
| Waist circumference | 15 | WMD: 0.09 cm (95% CI: −0.82 to 1.01) | 0.83 | |
| Lean body mass | 12 | WMD: −0.10 kg (95% CI: −0.61 to 0.41) | 0.69 | |
| Muscle mass/volume | 7 | SMD: −0.09 (95% CI: −0.38 to 0.19) | 0.52 | |
| CSA | 5 | SMD: −0.15 (95% CI: −0.40 to 0.10) | 0.25 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khalafi, M.; Kheradmand, S.; Habibi Maleki, A.; Symonds, M.E.; Rosenkranz, S.K.; Batrakoulis, A. The Effects of Concurrent Training Versus Aerobic or Resistance Training Alone on Body Composition in Middle-Aged and Older Adults: A Systematic Review and Meta-Analysis. Healthcare 2025, 13, 776. https://doi.org/10.3390/healthcare13070776
Khalafi M, Kheradmand S, Habibi Maleki A, Symonds ME, Rosenkranz SK, Batrakoulis A. The Effects of Concurrent Training Versus Aerobic or Resistance Training Alone on Body Composition in Middle-Aged and Older Adults: A Systematic Review and Meta-Analysis. Healthcare. 2025; 13(7):776. https://doi.org/10.3390/healthcare13070776
Chicago/Turabian StyleKhalafi, Mousa, Shokoufeh Kheradmand, Aref Habibi Maleki, Michael E. Symonds, Sara K. Rosenkranz, and Alexios Batrakoulis. 2025. "The Effects of Concurrent Training Versus Aerobic or Resistance Training Alone on Body Composition in Middle-Aged and Older Adults: A Systematic Review and Meta-Analysis" Healthcare 13, no. 7: 776. https://doi.org/10.3390/healthcare13070776
APA StyleKhalafi, M., Kheradmand, S., Habibi Maleki, A., Symonds, M. E., Rosenkranz, S. K., & Batrakoulis, A. (2025). The Effects of Concurrent Training Versus Aerobic or Resistance Training Alone on Body Composition in Middle-Aged and Older Adults: A Systematic Review and Meta-Analysis. Healthcare, 13(7), 776. https://doi.org/10.3390/healthcare13070776










