Spatial–Temporal Trends of Cancer Among Women in Central Serbia, 1999–2021: Implications for Disaster and Public Health Preparedness
Abstract
1. Introduction
2. Materials and Methods
Demographic Characteristics
3. Results
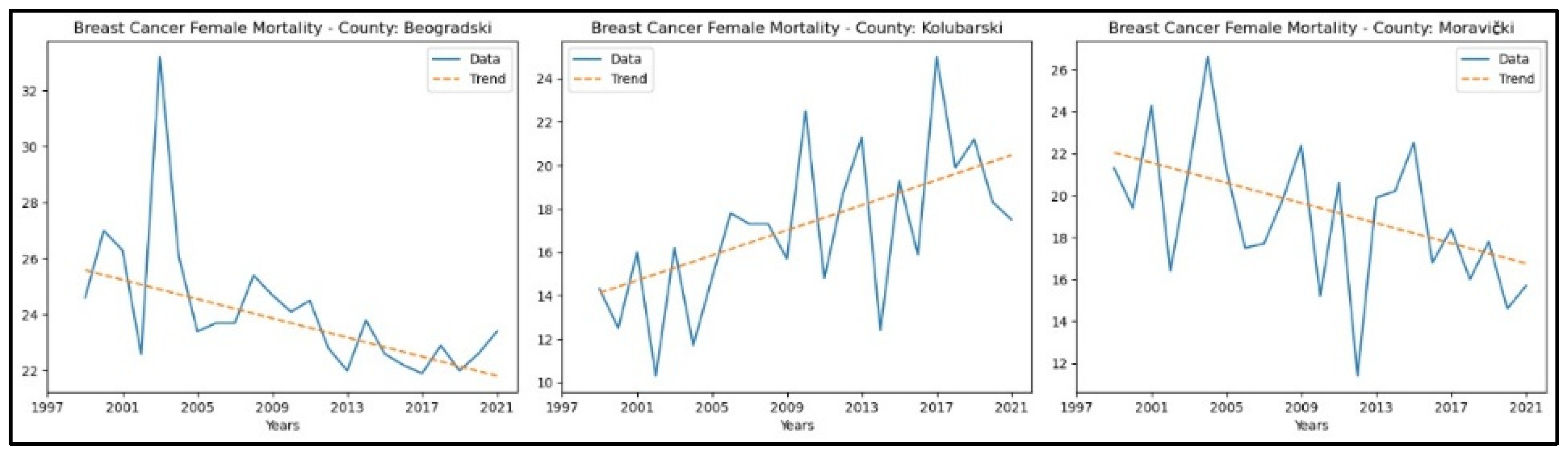
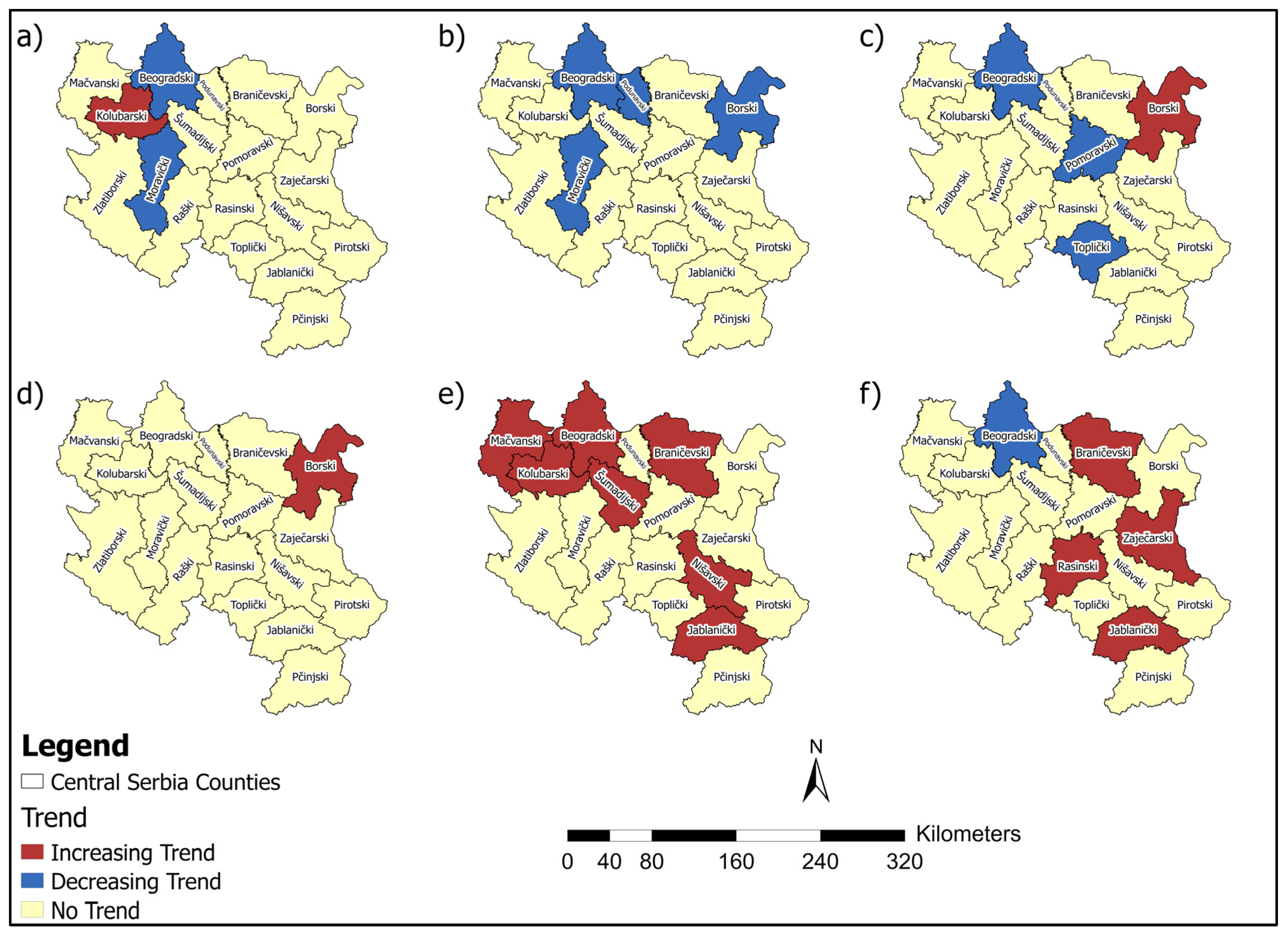
3.1. Breast Cancer
3.2. Cervical Cancer
3.3. Lung and Bronchus Cancer
3.4. Ovarian Cancer
3.5. Uterine Cancer
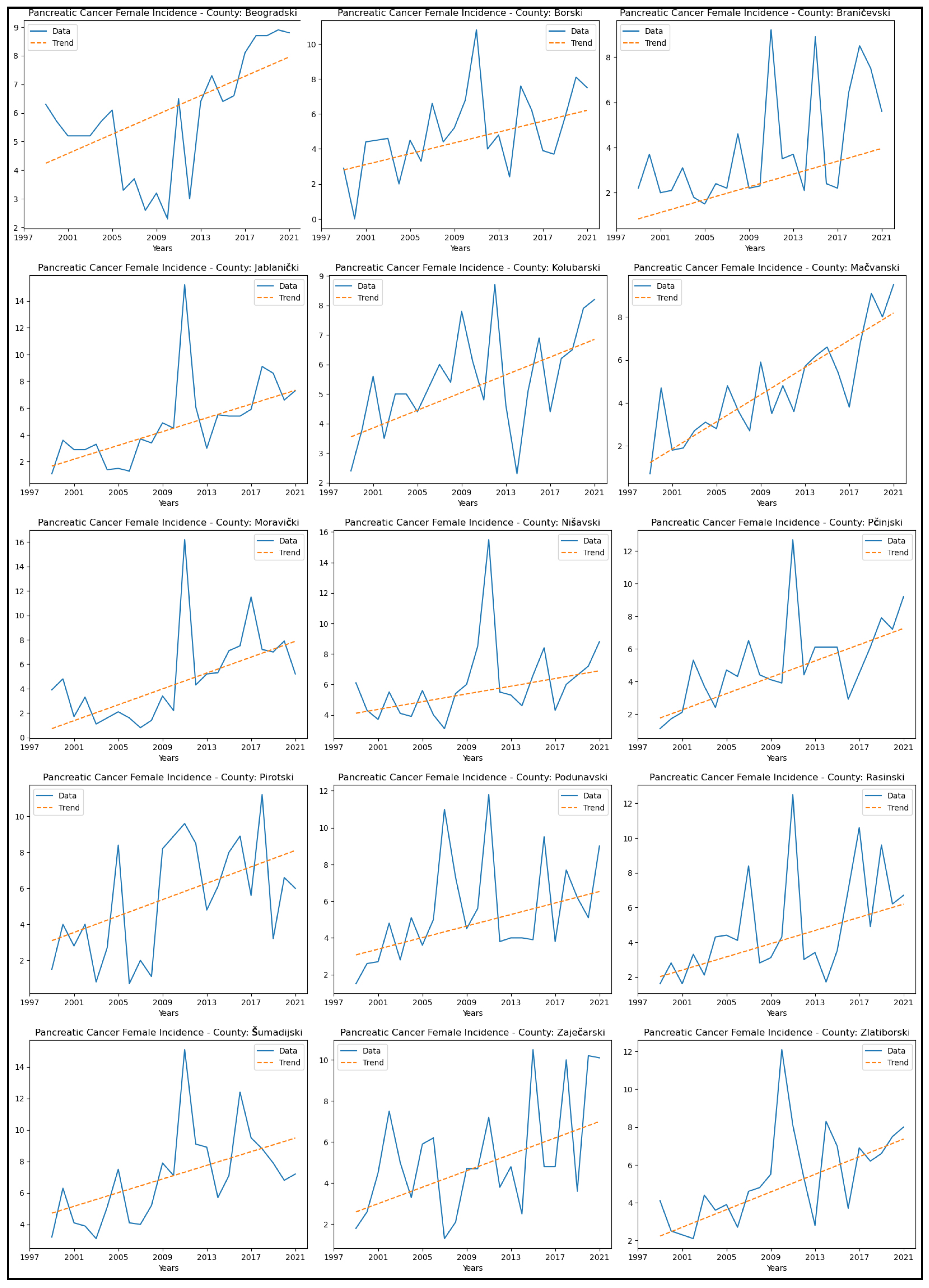
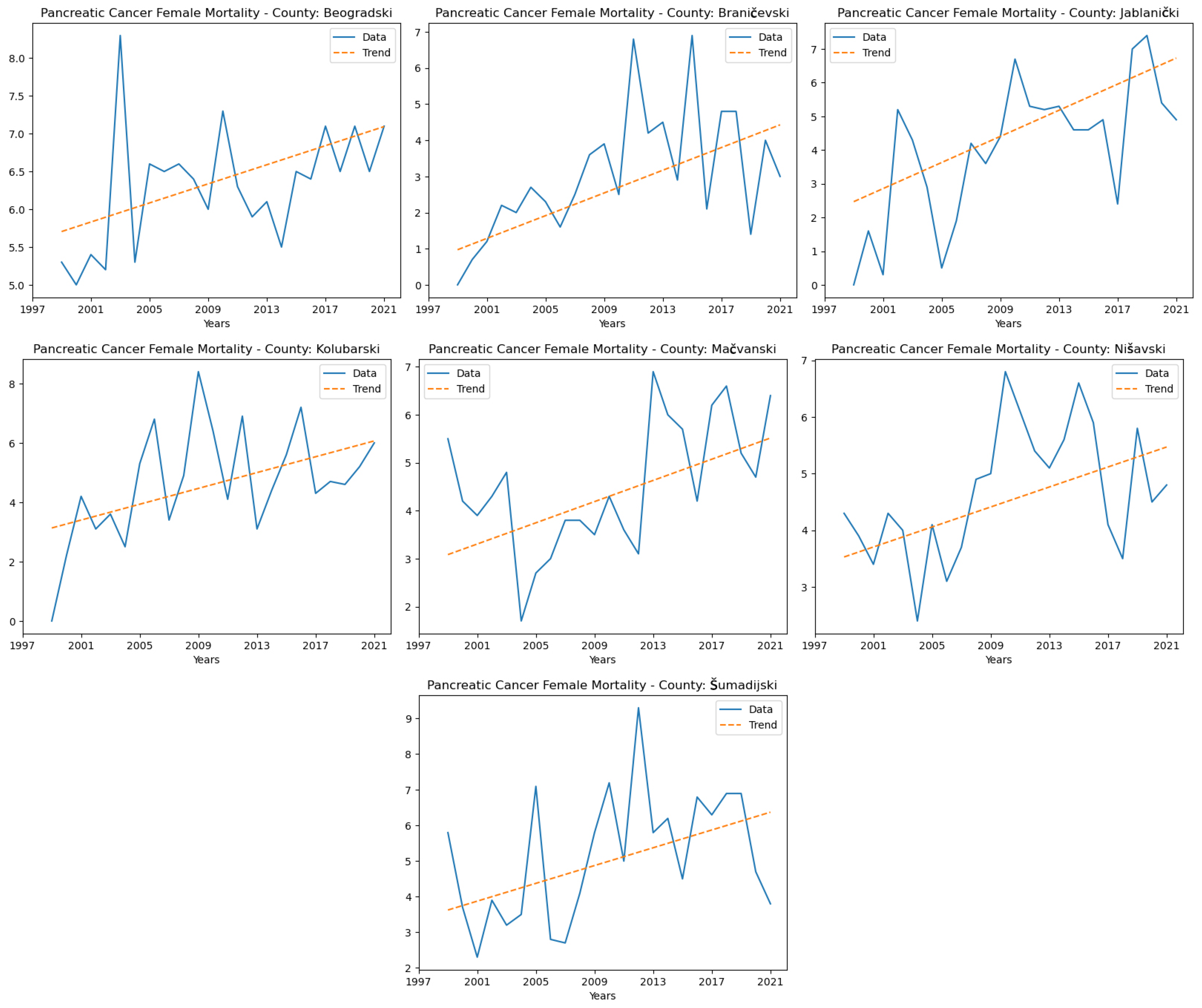
3.6. Pancreatic Cancer
3.7. Bladder Cancer
3.8. Colorectal Cancer
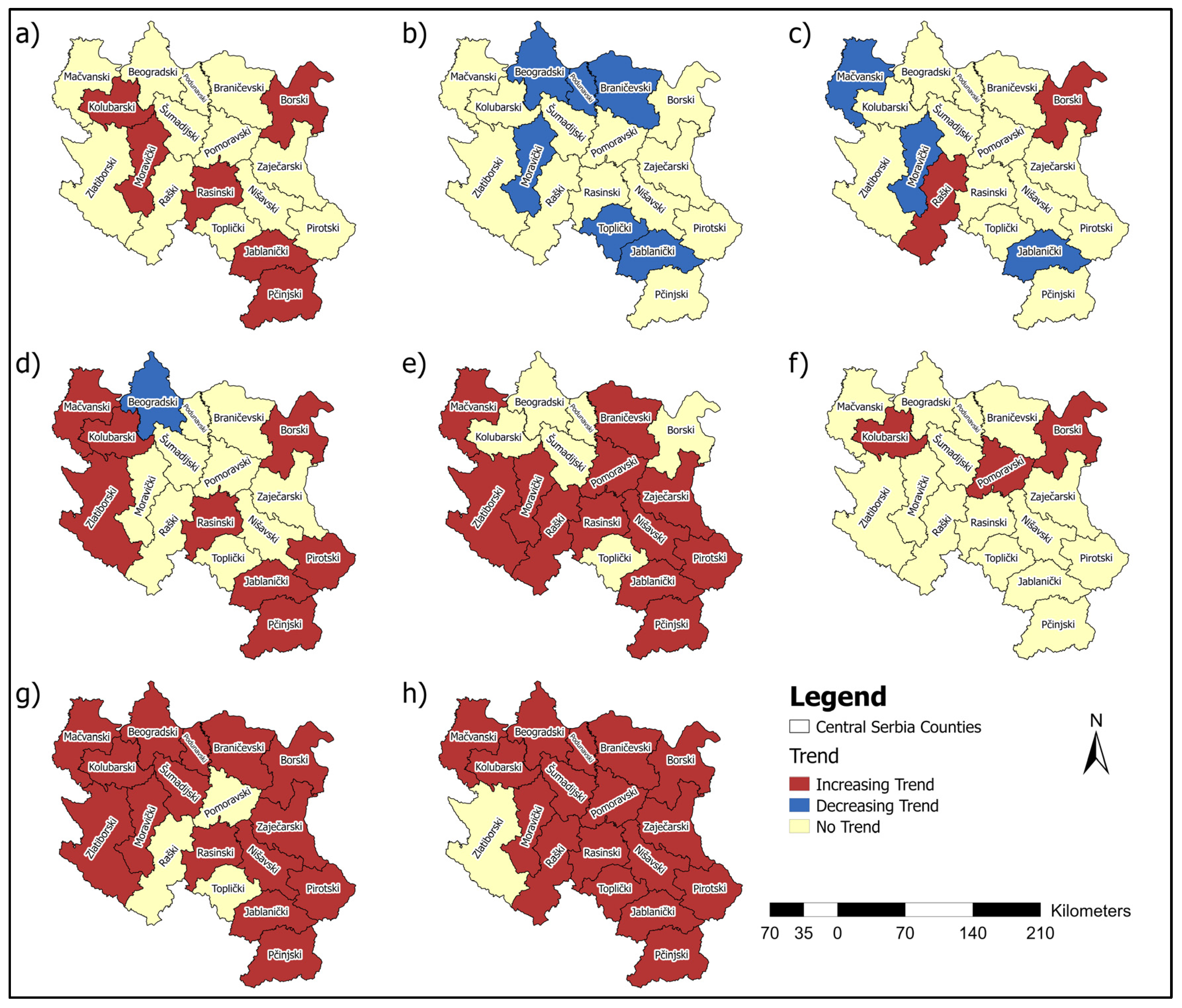
3.9. Overview of MK Analysis of Cancer Incidence and Mortality Rates by County
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| County | Breast Cancer | |||||||
| Incidence | Mortality | |||||||
| p | Z | s | b | p | Z | s | b | |
| Beogradski | 1.00 | 0 | 0 | 74.50 | 1.03 × 10−3 | −3.28 | −0.17 | 25.59 |
| Borski | 0.01 | 2.59 | 1.25 | 43.55 | 0.07 | −1.80 | −0.21 | 20.94 |
| Braničevski | 0.65 | −0.45 | −0.13 | 44.88 | 0.26 | 1.14 | 0.05 | 15.25 |
| Jablanički | 2.18 × 10−4 | 3.70 | 1.50 | 30.90 | 0.09 | 1.72 | 0.20 | 15.10 |
| Kolubarski | 1.95 × 10−4 | 3.73 | 1.13 | 41.70 | 0.01 | 2.80 | 0.29 | 14.12 |
| Mačvanski | 0.09 | 1.72 | 0.51 | 44.36 | 0.71 | 0.37 | 0.04 | 17.29 |
| Moravički | 9.56 × 10−8 | 5.33 | 1.40 | 47.60 | 0.02 | −2.25 | −0.24 | 22.04 |
| Nišavski | 0.12 | −1.56 | −0.31 | 69.77 | 0.98 | −0.03 | −0.02 | 20.27 |
| Pčinjski | 1.04 × 10−3 | 3.28 | 1.41 | 27.12 | 0.65 | 0.45 | 0.05 | 15.11 |
| Pirotski | 0.22 | 1.21 | 0.40 | 53.80 | 0.40 | −0.85 | −0.08 | 19.18 |
| Podunavski | 0.87 | 0.16 | 0.06 | 54.47 | 0.69 | −0.40 | −0.04 | 20.88 |
| Pomoravski | 0.09 | 1.72 | 0.76 | 43.86 | 0.24 | −1.16 | −0.12 | 18.55 |
| Rasinski | 0.03 | 2.22 | 1.10 | 60.90 | 0.60 | −0.53 | −0.05 | 20.70 |
| Raški | 0.05 | 1.93 | 0.55 | 50.21 | 0.23 | 1.19 | 0.10 | 19.00 |
| Šumadijski | 0.05 | 1.93 | 0.34 | 68.16 | 0.83 | −0.21 | −0.02 | 19.18 |
| Toplički | 0.79 | 0.26 | 0.08 | 47.48 | 1.00 | 0 | 0 | 14.30 |
| Zaječarski | 0.37 | 0.90 | 0.19 | 44.29 | 0.27 | −1.11 | −0.12 | 18.67 |
| Zlatiborski | 0.69 | −0.40 | −0.09 | 52.69 | 0.58 | 0.56 | 0.06 | 16.44 |
Appendix B
| County | Cervical Cancer | |||||||
| Incidence | Mortality | |||||||
| p | Z | s | b | p | Z | s | b | |
| Beogradski | 2.65 × 10−4 | −3.65 | −0.62 | 28.62 | 0.02 | −2.39 | −0.06 | 6.83 |
| Borski | 0.96 | −0.05 | −0.02 | 34.78 | 0.01 | −2.59 | −0.29 | 13.74 |
| Braničevski | 0.02 | −2.32 | −0.89 | 29.19 | 0.13 | −1.51 | −0.11 | 9.61 |
| Jablanički | 0.01 | −2.48 | −0.41 | 26.18 | 0.58 | −0.56 | −0.03 | 6.25 |
| Kolubarski | 0.18 | 1.35 | 0.24 | 18.79 | 0.94 | −0.08 | 0 | 6.60 |
| Mačvanski | 0.19 | −1.30 | −0.11 | 17.78 | 0.56 | 0.58 | 0.03 | 6.73 |
| Moravički | 0.01 | −2.80 | −0.59 | 32.53 | 0.01 | −2.75 | −0.22 | 10.42 |
| Nišavski | 0.62 | −0.50 | −0.06 | 23.01 | 0.94 | −0.08 | 0 | 7.00 |
| Pčinjski | 0.75 | −0.32 | −0.12 | 24.08 | 0.56 | 0.58 | 0.05 | 6.25 |
| Pirotski | 0.06 | 1.90 | 0.43 | 15.43 | 0.63 | 0.48 | 0.05 | 3.71 |
| Podunavski | 0.01 | −2.48 | −0.36 | 24.03 | 0.01 | −2.59 | −0.32 | 11.89 |
| Pomoravski | 0.34 | −0.95 | −0.15 | 19.85 | 0.87 | −0.16 | −0.03 | 8.29 |
| Rasinski | 0.12 | −1.56 | −0.22 | 20.68 | 0.41 | 0.82 | 0.04 | 5.12 |
| Raški | 0.07 | −1.80 | −0.38 | 23.95 | 0.56 | −0.58 | −0.03 | 8.08 |
| Šumadijski | 0.09 | −1.72 | −0.34 | 28.84 | 0.34 | −0.95 | −0.08 | 7.66 |
| Toplički | 0.04 | −2.06 | −0.42 | 25.77 | 0.27 | 1.11 | 0.11 | 5.74 |
| Zaječarski | 0.37 | −0.90 | −0.23 | 29.37 | 0.71 | −0.37 | −0.07 | 9.33 |
| Zlatiborski | 0.30 | −1.03 | −0.17 | 19.79 | 0.81 | −0.24 | −0.01 | 6.96 |
Appendix C
| County | Lung and Bronchus Cancer | |||||||
| Incidence | Mortality | |||||||
| p | Z | s | b | p | Z | s | b | |
| Beogradski | 0.02 | 2.43 | 0.44 | 19.96 | 4.64 × 10−5 | 4.07 | 0.35 | 17.55 |
| Borski | 4.17 × 10−5 | 4.10 | 0.70 | 9.00 | 1.39 × 10−3 | 3.20 | 0.53 | 6.13 |
| Braničevski | 7.09 × 10−4 | 3.39 | 0.50 | 10.50 | 3.98 × 10−4 | 3.54 | 0.40 | 8.90 |
| Jablanički | 1.26 × 10−3 | 3.22 | 0.58 | 6.41 | 3.62 × 10−4 | 3.57 | 0.42 | 6.00 |
| Kolubarski | 1.42 × 10−5 | 4.34 | 0.59 | 10.71 | 0.03 | 2.17 | 0.26 | 10.15 |
| Mačvanski | 1.74 × 10−6 | 4.78 | 0.72 | 9.58 | 1.96 × 10−6 | 4.76 | 0.44 | 10.25 |
| Moravički | 4.25 × 10−6 | 4.60 | 0.36 | 14.04 | 0.03 | 2.17 | 0.22 | 9.82 |
| Nišavski | 2.40 × 10−4 | 3.67 | 0.46 | 10.29 | 9.36 × 10−8 | 5.34 | 0.51 | 6.73 |
| Pčinjski | 7.25 × 10−5 | 3.97 | 0.98 | 3.12 | 0.13 | 1.53 | 0.24 | 9.73 |
| Pirotski | 0.02 | 2.30 | 0.27 | 8.47 | 0.75 | 0.32 | 0.03 | 8.76 |
| Podunavski | 1.15 × 10−4 | 3.86 | 0.94 | 12.53 | 2.95 × 10−4 | 3.62 | 0.57 | 14.41 |
| Pomoravski | 0.01 | 2.77 | 0.59 | 6.40 | 0.04 | 2.01 | 0.20 | 13.10 |
| Rasinski | 0.01 | 2.67 | 0.53 | 12.53 | 1.75 × 10−4 | 3.75 | 0.33 | 7.09 |
| Raški | 3.36 × 10−3 | 2.93 | 0.41 | 9.64 | 2.17 × 10−3 | 3.07 | 0.34 | 7.80 |
| Šumadijski | 3.95 × 10−3 | 2.88 | 0.29 | 17.26 | 4.22 × 10−5 | 4.10 | 0.50 | 9.70 |
| Toplički | 0.01 | 2.64 | 0.42 | 10.48 | 0.01 | 2.67 | 0.39 | 6.26 |
| Zaječarski | 1.02 × 10−5 | 4.41 | 0.78 | 6.78 | 1.05 × 10−3 | 3.28 | 0.44 | 5.25 |
| Zlatiborski | 0.17 | 1.37 | 0.23 | 11.53 | 0.01 | 2.78 | 0.28 | 8.08 |
Appendix D
| County | Ovarian Cancer | |||||||
| Incidence | Mortality | |||||||
| p | Z | s | b | p | Z | s | b | |
| Beogradski | 0.07 | −1.80 | −0.10 | 12.50 | 0.83 | 0.21 | 0.01 | 5.80 |
| Borski | 2.38 × 10−3 | 3.04 | 0.44 | 6.51 | 2.38 × 10−3 | 3.04 | 0.23 | 2.95 |
| Braničevski | 0.41 | 0.82 | 0.16 | 6.59 | 0.44 | 0.77 | 0.07 | 3.03 |
| Jablanički | 0.09 | 1.69 | 0.21 | 6.94 | 0.19 | 1.32 | 0.08 | 3.88 |
| Kolubarski | 2.17 × 10−3 | 3.07 | 0.35 | 7.75 | 0.18 | 1.35 | 0.07 | 3.77 |
| Mačvanski | 0.18 | 1.35 | 0.07 | 7.73 | 0.65 | 0.45 | 0.02 | 3.86 |
| Moravički | 0.18 | 1.35 | 0.13 | 8.39 | 0.21 | −1.24 | −0.10 | 6.65 |
| Nišavski | 0.96 | −0.05 | −0.01 | 12.98 | 0.23 | 1.19 | 0.04 | 4.73 |
| Pčinjski | 0.07 | 1.80 | 0.23 | 5.93 | 0.83 | 0.21 | 0.02 | 3.80 |
| Pirotski | 0.10 | 1.64 | 0.20 | 8.80 | 0.92 | 0.11 | 0.01 | 3.39 |
| Podunavski | 0.71 | 0.37 | 0.05 | 9.52 | 0.19 | −1.30 | −0.05 | 5.21 |
| Pomoravski | 3.95 × 10−3 | 2.88 | 0.30 | 5.20 | 0.29 | 1.06 | 0.06 | 4.19 |
| Rasinski | 0.98 | −0.03 | 0.00 | 9.70 | 0.09 | 1.72 | 0.10 | 3.30 |
| Raški | 0.58 | 0.55 | 0.06 | 9.11 | 0.81 | −0.24 | −0.02 | 5.08 |
| Šumadijski | 0.15 | 1.45 | 0.15 | 10.85 | 0.12 | −1.54 | −0.06 | 5.78 |
| Toplički | 0.96 | 0.05 | 0.02 | 11.60 | 0.53 | −0.63 | −0.03 | 3.37 |
| Zaječarski | 0.62 | 0.50 | 0.05 | 9.25 | 0.49 | −0.69 | −0.05 | 6.45 |
| Zlatiborski | 0.20 | 1.27 | 0.16 | 8.59 | 0.10 | 1.67 | 0.07 | 3.90 |
Appendix E
| County | Uterine Cancer | |||||||
| Incidence | Mortality | |||||||
| p | Z | s | b | p | Z | s | b | |
| Beogradski | 0.65 | −0.45 | −0.07 | 14.65 | N/A | N/A | N/A | N/A |
| Borski | 3.36 × 10−5 | 4.15 | 0.50 | 6.30 | N/A | N/A | N/A | N/A |
| Braničevski | 0.40 | −0.85 | −0.12 | 12.15 | N/A | N/A | N/A | N/A |
| Jablanički | 0.01 | −2.56 | −0.47 | 23.23 | N/A | N/A | N/A | N/A |
| Kolubarski | 0.33 | 0.98 | 0.10 | 10.30 | N/A | N/A | N/A | N/A |
| Mačvanski | 0.02 | −2.41 | −0.18 | 11.18 | N/A | N/A | N/A | N/A |
| Moravički | 0.02 | −2.38 | −0.26 | 13.31 | N/A | N/A | N/A | N/A |
| Nišavski | 0.51 | 0.66 | 0.08 | 16.58 | N/A | N/A | N/A | N/A |
| Pčinjski | 0.41 | 0.82 | 0.10 | 14.80 | N/A | N/A | N/A | N/A |
| Pirotski | 0.81 | 0.24 | 0.01 | 12.32 | N/A | N/A | N/A | N/A |
| Podunavski | 0.27 | −1.11 | −0.08 | 10.87 | N/A | N/A | N/A | N/A |
| Pomoravski | 0.75 | 0.32 | 0.03 | 8.99 | N/A | N/A | N/A | N/A |
| Rasinski | 0.85 | −0.19 | −0.02 | 17.40 | N/A | N/A | N/A | N/A |
| Raški | 1.82 × 10−3 | 3.12 | 0.33 | 9.13 | N/A | N/A | N/A | N/A |
| Šumadijski | 0.62 | −0.50 | −0.05 | 13.15 | N/A | N/A | N/A | N/A |
| Toplički | 0.25 | 1.16 | 0.14 | 9.76 | N/A | N/A | N/A | N/A |
| Zaječarski | 0.21 | 1.24 | 0.18 | 9.48 | N/A | N/A | N/A | N/A |
| Zlatiborski | 0.20 | −1.27 | −0.11 | 9.54 | N/A | N/A | N/A | N/A |
Appendix F
| County | Pancreatic Cancer | |||||||
| Incidence | Mortality | |||||||
| p | Z | s | b | p | Z | s | b | |
| Beogradski | 3.59 × 10−3 | 2.91 | 0.17 | 4.24 | 0.02 | 2.28 | 0.06 | 5.71 |
| Borski | 0.02 | 2.27 | 0.16 | 2.80 | 0.28 | −1.08 | −0.06 | 5.05 |
| Braničevski | 0.01 | 2.47 | 0.14 | 0.84 | 1.52 × 10−3 | 3.17 | 0.16 | 0.97 |
| Jablanički | 4.70 × 10−5 | 4.07 | 0.26 | 1.67 | 7.11 × 10−4 | 3.39 | 0.19 | 2.47 |
| Kolubarski | 0.01 | 2.70 | 0.15 | 3.55 | 0.01 | 2.46 | 0.13 | 3.13 |
| Mačvanski | 2.88 × 10−6 | 4.68 | 0.32 | 1.22 | 0.04 | 2.09 | 0.11 | 3.08 |
| Moravički | 1.82 × 10−3 | 3.12 | 0.33 | 0.73 | 0.21 | 1.24 | 0.07 | 3.77 |
| Nišavski | 0.02 | 2.38 | 0.13 | 4.11 | 0.04 | 2.06 | 0.09 | 3.53 |
| Pčinjski | 4.24 × 10−4 | 3.52 | 0.25 | 1.75 | 0.65 | 0.45 | 0.01 | 3.74 |
| Pirotski | 0.03 | 2.11 | 0.23 | 3.09 | 0.46 | 0.74 | 0.07 | 4.21 |
| Podunavski | 0.01 | 2.46 | 0.16 | 3.07 | 0.58 | 0.56 | 0.03 | 4.13 |
| Pomoravski | 0.07 | 1.82 | 0.13 | 1.93 | 0.62 | −0.50 | −0.03 | 5.59 |
| Rasinski | 3.34 × 10−3 | 2.93 | 0.19 | 2.01 | 0.13 | −1.51 | −0.07 | 5.69 |
| Raški | 0.60 | 0.53 | 0.05 | 5.05 | 0.43 | 0.79 | 0.07 | 4.65 |
| Šumadijski | 3.34 × 10−3 | 2.93 | 0.22 | 4.72 | 0.04 | 2.06 | 0.13 | 3.63 |
| Toplički | 0.77 | 0.29 | 0.03 | 4.13 | 0.40 | −0.85 | −0.11 | 5.41 |
| Zaječarski | 0.04 | 2.06 | 0.20 | 2.60 | 0.06 | 1.85 | 0.12 | 3.20 |
| Zlatiborski | 0.00 | 3.33 | 0.23 | 2.23 | 0.15 | 1.45 | 0.07 | 3.53 |
Appendix G
| County | Bladder Cancer | |||||||
| Incidence | Mortality | |||||||
| p | Z | s | b | p | Z | s | b | |
| Beogradski | 0.73 | 0.34 | 0.03 | 4.83 | N/A | N/A | N/A | N/A |
| Borski | 0.83 | −0.21 | −0.03 | 5.33 | N/A | N/A | N/A | N/A |
| Braničevski | 0.01 | 2.67 | 0.24 | 1.14 | N/A | N/A | N/A | N/A |
| Jablanički | 1.51 × 10−3 | 3.17 | 0.19 | 1.77 | N/A | N/A | N/A | N/A |
| Kolubarski | 0.20 | 1.27 | 0.10 | 3.30 | N/A | N/A | N/A | N/A |
| Mačvanski | 0.01 | 2.48 | 0.13 | 2.73 | N/A | N/A | N/A | N/A |
| Moravički | 1.56 × 10−4 | 3.78 | 0.41 | 1.13 | N/A | N/A | N/A | N/A |
| Nišavski | 0.01 | 2.62 | 0.20 | 2.70 | N/A | N/A | N/A | N/A |
| Pčinjski | 0.02 | 2.33 | 0.21 | 1.27 | N/A | N/A | N/A | N/A |
| Pirotski | 0.02 | 2.33 | 0.20 | 2.50 | N/A | N/A | N/A | N/A |
| Podunavski | 0.07 | 1.82 | 0.16 | 3.91 | N/A | N/A | N/A | N/A |
| Pomoravski | 1.49 × 10−3 | 3.18 | 0.31 | 0.78 | N/A | N/A | N/A | N/A |
| Rasinski | 0.01 | 2.75 | 0.27 | 3.33 | N/A | N/A | N/A | N/A |
| Raški | 0.02 | 2.35 | 0.16 | 2.75 | N/A | N/A | N/A | N/A |
| Šumadijski | 0.53 | 0.63 | 0.04 | 6.66 | N/A | N/A | N/A | N/A |
| Toplički | 0.08 | 1.78 | 0.10 | 1.20 | N/A | N/A | N/A | N/A |
| Zaječarski | 0.02 | 2.41 | 0.13 | 2.93 | N/A | N/A | N/A | N/A |
| Zlatiborski | 0.01 | 2.75 | 0.24 | 1.66 | N/A | N/A | N/A | N/A |
Appendix H
| County | Colorectal Cancer | |||||||
| Incidence | Mortality | |||||||
| p | Z | s | b | p | Z | s | b | |
| Beogradski | 0.01 | −2.46 | −0.40 | 26.10 | 0.03 | −2.14 | −0.10 | 12.95 |
| Borski | 8.99 × 10−6 | 4.44 | 0.77 | 9.27 | 0.01 | 2.49 | 0.18 | 7.22 |
| Braničevski | 0.11 | 1.58 | 0.20 | 14.40 | 0.41 | 0.82 | 0.08 | 7.84 |
| Jablanički | 3.36 × 10−3 | 2.93 | 0.52 | 10.68 | 0.15 | −1.43 | −0.10 | 9.60 |
| Kolubarski | 0.01 | 2.77 | 0.41 | 19.44 | 0.94 | −0.08 | −0.01 | 11.18 |
| Mačvanski | 0.02 | 2.35 | 0.40 | 17.80 | 0.62 | −0.50 | −0.04 | 11.69 |
| Moravički | 0.27 | 1.11 | 0.15 | 18.29 | 0.44 | −0.77 | −0.07 | 8.03 |
| Nišavski | 0.21 | 1.24 | 0.15 | 18.05 | 0.79 | 0.26 | 0.02 | 8.93 |
| Pčinjski | 0.04 | 2.04 | 0.45 | 11.25 | 0.08 | −1.75 | −0.10 | 9.40 |
| Pirotski | 0.02 | 2.35 | 0.68 | 16.42 | 0.07 | 1.80 | 0.15 | 6.90 |
| Podunavski | 1.00 | 0 | −0.02 | 20.08 | 0.38 | −0.87 | −0.08 | 11.62 |
| Pomoravski | 0.14 | 1.48 | 0.25 | 14.25 | 0.04 | −2.09 | −0.22 | 11.88 |
| Rasinski | 3.25 × 10−4 | 3.59 | 0.65 | 13.78 | 0.85 | 0.18 | 0.02 | 10.59 |
| Raški | 0.15 | 1.45 | 0.20 | 16.60 | 0.60 | −0.53 | −0.03 | 8.70 |
| Šumadijski | 0.22 | 1.22 | 0.18 | 21.77 | 0.96 | −0.05 | 0 | 9.30 |
| Toplički | 0.79 | −0.26 | −0.05 | 17.65 | 0.02 | −2.27 | −0.15 | 9.20 |
| Zaječarski | 0.10 | 1.64 | 0.28 | 18.38 | 0.07 | −1.82 | −0.15 | 13.15 |
| Zlatiborski | 0.01 | 2.80 | 0.36 | 13.14 | 0.44 | 0.77 | 0.07 | 8.73 |
References
- Cvetković, V.M.; Nikolić, N.; Ocal, A.; Martinović, J.; Dragašević, A. A Predictive Model of Pandemic Disaster Fear Caused by Coronavirus (COVID-19): Implications for Decision-Makers. Int. J. Environ. Res. Public Health 2022, 19, 652. [Google Scholar] [CrossRef] [PubMed]
- Cvetković, V.; Tanasić, J.; Ocal, A.; Živković-Šulović, M.; Ćurić, N.; Milojević, S.; Knežević, S. The Assessment of Public Health Capacities at Local Self-Governments in Serbia. Lex Localis J. Local Self-Gov. 2023, 21, 1201–1234. [Google Scholar] [CrossRef] [PubMed]
- Cvetković, V.M.; Nikolić, N.; Nenadić, U.R.; Öcal, A.; Noji, E.K.; Zečević, M. Preparedness and Preventive Behaviors for a Pandemic Disaster Caused by COVID-19 in Serbia. Int. J. Environ. Res. Public Health 2020, 17, 4124. [Google Scholar] [CrossRef]
- Cvetković, V.M.; Tanasić, J.; Ocal, A.; Kešetović, Ž.; Nikolić, N.; Dragašević, A. Capacity Development of Local Self-Governments for Disaster Risk Management. Int. J. Environ. Res. Public Health 2021, 18, 10406. [Google Scholar] [CrossRef]
- Cvetković, V.M.; Tanasić, J.; Renner, R.; Rokvić, V.; Beriša, H. Comprehensive Risk Analysis of Emergency Medical Response Systems in Serbian Healthcare: Assessing Systemic Vulnerabilities in Disaster Preparedness and Response. Healthcare 2024, 12, 1962. [Google Scholar] [CrossRef]
- Zhang, J.; Sun, Z.; Chen, L.; Li, J.; Wang, Q.; Li, Y.; Su, G.; Dou, Z. Rapid assessment of the vulnerability of densely populated urban communities under major epidemics. Int. J. Disaster Risk Reduct. 2024, 116, 105082. [Google Scholar] [CrossRef]
- Li, A.; Toll, M.; Bentley, R. Mapping social vulnerability indicators to understand the health impacts of climate change: A scoping review. Lancet Planet. Health 2023, 7, e925–e937. [Google Scholar] [CrossRef]
- Mah, J.C.; Penwarden, J.L.; Pott, H.; Theou, O.; Andrew, M.K. Social vulnerability indices: A scoping review. BMC Public Health 2023, 23, 1253. [Google Scholar] [CrossRef]
- Akerlof, K.L.; Delamater, P.L.; Boules, C.R.; Upperman, C.R.; Mitchell, C.S. Vulnerable Populations Perceive Their Health as at Risk from Climate Change. Int. J. Environ. Res. Public Health 2015, 12, 15419–15433. [Google Scholar] [CrossRef] [PubMed]
- Rupa, F.H.; Hossian, M. Addressing Public Health Risks: Strategies to Combat Infectious Diseases After the August 2024 Floods in Bangladesh. J. Prev. Med. Public Health 2024, 57, 600–603. [Google Scholar] [CrossRef]
- Morgan, R.; Tan, H.-L.; Oveisi, N.; Memmott, C.; Korzuchowski, A.; Hawkins, K.; Smith, J. Women healthcare workers’ experiences during COVID-19 and other crises: A scoping review. Int. J. Nurs. Stud. Adv. 2022, 4, 100066. [Google Scholar] [CrossRef] [PubMed]
- Karanikolos, M.; Heino, P.; McKee, M.; Stuckler, D.; Legido-Quigley, H. Effects of the Global Financial Crisis on Health in High-Income Oecd Countries. Int. J. Health Serv. 2016, 46, 208–240. [Google Scholar] [CrossRef] [PubMed]
- Meadows, J.; Montano, M.; Alfar, A.J.K.; Başkan, Ö.Y.; De Brún, C.; Hill, J.; McClatchey, R.; Kallfa, N.; Fernandes, G.S. The impact of the cost-of-living crisis on population health in the UK: Rapid evidence review. BMC Public Health 2024, 24, 561. [Google Scholar] [CrossRef]
- Jaradat, J.H.; Alabdallat, Y.J.; Al-Qaqa, O.; Baker, M.B. Struggling hearts: Cardiovascular health in a war-torn Gaza. Avicenna 2024, 2024, 4. [Google Scholar] [CrossRef]
- Aday, L.A. Health status of vulnerable populations. Annu. Rev. Public Health 1994, 15, 487–509. [Google Scholar] [CrossRef]
- Gonzalez, T.N.; Paludo, C.d.S.; Meucci, R.D. Vulnerability among rural older adults in southern Brazil: Population-based study. Rural. Remote Health 2023, 23, 7714. [Google Scholar] [CrossRef]
- Sun, P.; Yu, C.; Yin, L.; Chen, Y.; Sun, Z.; Zhang, T.; Shuai, P.; Zeng, K.; Yao, X.; Chen, J.; et al. Global, regional, and national burden of female cancers in women of child-bearing age, 1990–2021: Analysis of data from the global burden of disease study 2021. EClinicalMedicine 2024, 74, 102713. [Google Scholar] [CrossRef]
- Cai, Y.; Dai, F.; Ye, Y.; Qian, J. The global burden of breast cancer among women of reproductive age: A comprehensive analysis. Sci. Rep. 2025, 15, 9347. [Google Scholar] [CrossRef]
- Torre, L.A.; Islami, F.; Siegel, R.L.; Ward, E.M.; Jemal, A. Global Cancer in Women: Burden and Trends. Cancer Epidemiol. Biomark. Prev. 2017, 26, 444–457. [Google Scholar] [CrossRef]
- Yi, M.; Li, T.; Niu, M.; Luo, S.; Chu, Q.; Wu, K. Epidemiological trends of women’s cancers from 1990 to 2019 at the global, regional, and national levels: A population-based study. Biomark. Res. 2021, 9, 55. [Google Scholar] [CrossRef] [PubMed]
- Ning, W.; Liu, J.; Lu, Y.; Zhu, B.; Zhang, W.-H.; Mao, Y. Trends in the Disease Burden and Risk Factors of Women’s Cancers in China From 1990 to 2019. Int. J. Public Health 2024, 69, 1607245. [Google Scholar] [CrossRef] [PubMed]
- Fund, W.C.R. Global Cancer Data by Country: Cancer Incidence in Women 2022. Available online: https://www.wcrf.org/cancer-trends/global-cancer-data-by-country (accessed on 21 December 2024).
- Sedeta, E.T.; Jobre, B.; Avezbakiyev, B. Breast cancer: Global patterns of incidence, mortality, and trends. J. Clin. Oncol. 2023, 41, 10528. [Google Scholar] [CrossRef]
- Li, T.; Zhang, H.; Lian, M.; He, Q.; Lv, M.; Zhai, L.; Zhou, J.; Wu, K.; Yi, M. Global status and attributable risk factors of breast, cervical, ovarian, and uterine cancers from 1990 to 2021. J. Hematol. Oncol. 2025, 18, 5. [Google Scholar] [CrossRef]
- Jiang, D.; Niu, Z.; Tan, X.; He, H.; Ren, L.; Shen, J.; Zhu, X.; Zhao, P.; Liu, M.; Chen, H.; et al. The mortalities of female-specific cancers in China and other countries with distinct socioeconomic statuses: A longitudinal study. J. Adv. Res. 2022, 49, 127–139. [Google Scholar] [CrossRef]
- Cotangco, K.R.; Liao, C.-I.; Eakin, C.M.; Chan, A.; Cohen, J.; Kapp, D.S.; Chan, J.K. Trends in Incidence of Cancers Associated With Obesity and Other Modifiable Risk Factors Among Women, 2001–2018. Prev. Chronic Dis. 2023, 20, 220211. [Google Scholar] [CrossRef]
- Keyvani, V.; Kheradmand, N.; Navaei, Z.N.; Mollazadeh, S.; Esmaeili, S.-A. Epidemiological trends and risk factors of gynecological cancers: An update. Med. Oncol. 2023, 40, 1–11. [Google Scholar] [CrossRef]
- Jin, Q.; Shi, N.; Lee, D.H.; Rexrode, K.M.; Manson, J.E.; Balasubramanian, R.; Zhang, X.; Neuhouser, M.L.; Lopez-Pentecost, M.; Thomson, C.A.; et al. Hyperinsulinemic and Pro-Inflammatory Dietary Patterns and Metabolomic Profiles Are Associated with Increased Risk of Total and Site-Specific Cancers among Postmenopausal Women. Cancers 2023, 15, 1756. [Google Scholar] [CrossRef]
- Lortet-Tieulent, J.; Ferlay, J.; Bray, F.; Jemal, A. International Patterns and Trends in Endometrial Cancer Incidence, 1978–2013. J. Natl. Cancer Inst. 2018, 110, 354–361. [Google Scholar] [CrossRef] [PubMed]
- He, R.; Zhu, B.; Liu, J.; Zhang, N.; Zhang, W.-H.; Mao, Y. Women’s cancers in China: A spatio-temporal epidemiology analysis. BMC Women’s Health 2021, 21, 116. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, A.D.; Neupane, D.; Vedsted, P.; Kallestrup, P. Cervical cancer prevalence, incidence and mortality in low and middle income countries: A systematic review. Asian Pac. J. Cancer Prev. APJCP 2018, 19, 319–324. [Google Scholar]
- Olson, B.; Gribble, B.; Dias, J.; Curryer, C.; Vo, K.; Kowal, P.; Byles, J. Cervical cancer screening programs and guidelines in low-and middle-income countries. Int. J. Gynecol. Obstet. 2016, 134, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wang, H.; Lyu, W.; Xu, R. The Mann-Kendall-Sneyers test to identify the change points of COVID-19 time series in the United States. BMC Med. Res. Methodol. 2022, 22, 233. [Google Scholar] [CrossRef]
- Rizzi, D.; Ciuffo, G.; El Gour, F.; Erradi, J.; Barone, L.; Ionio, C. Perspectives on early insights: Pediatric cancer caregiving amidst natural calamities—A call for future preparedness. Front. Public Health 2024, 11, 1319850. [Google Scholar] [CrossRef]
- Gudenkauf, L.M.; Hathaway, C.A.; Carroll, J.E.; Small, B.J.; Li, X.; Hoogland, A.I.; Castro, E.; Armaiz-Pena, G.N.; Oswald, L.B.; Jim, H.S.; et al. Inequities in the Impacts of Hurricanes and Other Extreme Weather Events for Cancer Survivors. Cancer Epidemiol. Biomark. Prev. 2024, 33, 771–778. [Google Scholar] [CrossRef]
- Yasui, A.; Kinoshita, S.; Endo, M.; Kaneda, Y.; Ikeguchi, R.; Yamamoto, C.; Abe, T.; Zhao, T.; Sawano, T.; Gonda, K.; et al. Disaster response and older adult cancer care in super-aged societies: Insights from the 2024 Noto Peninsula Earthquake in Oku-Noto, Japan. Front. Public Health 2024, 12, 1427987. [Google Scholar] [CrossRef] [PubMed]
- Prohaska, T.; Peters, K. Impact of Natural Disasters on Health Outcomes and Cancer Among Older Adults. Gerontologist 2019, 59 (Suppl. S1), S50–S56. [Google Scholar] [CrossRef]
- Yang, F.; Sun, D.; Ding, C.; Xia, C.; Li, H.; Cao, M.; Yan, X.; He, S.; Zhang, S.; Chen, W. Global patterns of cancer transitions: A modelling study. Int. J. Cancer 2023, 153, 1612–1622. [Google Scholar] [CrossRef]
- Gorji, H.A.; Jafari, H.; Heidari, M.; Seifi, B. Cancer Patients During and after Natural and Man-Made Disasters: A Systematic Review. Asian Pac. J. Cancer Prev. APJCP 2018, 19, 2695–2700. [Google Scholar] [CrossRef] [PubMed]
- Ginex, P.; Dickman, E.; Elia, M.R.; Burbage, D.; Wilson, R.; Koos, J.A.; Sivakumaran, K.; Morgan, R.L. Climate disasters and oncology care: A systematic review of effects on patients, healthcare professionals, and health systems. Support. Care Cancer 2023, 31, 1–10. [Google Scholar] [CrossRef]
- Espinel, Z.; Shultz, J.M.; Aubry, V.P.; Abraham, O.M.; Fan, Q.; Crane, T.E.; Sahar, L.; Nogueira, L.M. Protecting Vulnerable Patient Populations from Climate Hazards: The Role of the Nations’ Cancer Centers. J. Natl. Cancer Inst. 2023, 115, 1252–1261. [Google Scholar] [CrossRef]
- Espinel, Z.; Shultz, J.; Aubry, V.P.; Abraham, O.M.; Fan, Q.; Sahar, L.; Nogueira, L.M. Cancer centers as potential knowledge hubs for emergency preparedness efforts. J. Clin. Oncol. 2023, 41, e18729. [Google Scholar] [CrossRef]
- Yang, X.; Gao, L.; Zhang, S. Comparative pan-cancer DNA methylation analysis reveals cancer common and specific patterns. Brief. Bioinform. 2016, 18, 761. [Google Scholar] [CrossRef] [PubMed]
- Ilic, M.; Vlajinac, H.; Markinovic, J.; Blazic, Z. Mortality from cancer of the lung in Serbia. JBUON 2013, 18, 723–727. [Google Scholar]
- Ilic, M.; Ilic, I. Cancer mortality in Serbia, 1991–2015: An age-period-cohort and joinpoint regression analysis. Cancer Commun. 2018, 38, 1–16. [Google Scholar] [CrossRef]
- Markovic-Denic, L.; Cirkovic, A.; Zivkovic, S.; Stanic, D.; Skodric-Trifunovic, V. Cancer mortality in central Serbia. JBUON 2014, 19, 273–277. [Google Scholar]
- Kričković, E.; Lukić, T.; Kričković, Z.; Stojšić-Milosavljević, A.; Živanović, M.; Srejić, T. Spatiotemporal and trend analysis of common cancers in men in Central Serbia (1999–2021). Open Geosci. 2025, 17, 20250802. [Google Scholar] [CrossRef]
- Micić, T. Geospatial analysis of incidence and mortality caused by lung cancer in the male population in the territory of central Serbia (1999–2013). In Proceedings of the GIS Day—2016, Belgrade, Serbia, 16 November 2016. [Google Scholar]
- Antonijevic, A.; Rancic, N.; Ilic, M.; Tiodorovic, B.; Stojanovic, M.; Stevanovic, J. Incidence and mortality trends of ovarian cancer in central Serbia. JBUON 2017, 22, 508–512. [Google Scholar] [PubMed]
- Šipetić-Grujičić, S.; Nikolić, A.; Pislar, A.; Pavlović, A.; Banašević, M.; Maksimović, J.; Krivokapić, Z. Trends in mortality rates of colorectal cancer in central Serbia during the period 1999–2014: A joinpoint regression analysis. Zdr. Zaštita 2019, 48, 33–42. [Google Scholar] [CrossRef]
- Ignjatović, A.; Stojanović, M.; Milošević, Z.; Anđelković Apostolović, M.; Filipović, T.; Rančić, N.; Marković, R.; Topalović, M.; Stojanović, D.; Otašević, S. Cancer of unknown primary-incidence, mortality trend, and mortality-to-incidence ratio is associated with human development index in Central Serbia, 1999–2018: Evidence from the national cancer registry. Eur. J. Cancer Care 2022, 31, e13526. [Google Scholar] [CrossRef]
- Stojanovic, M.M.; Rancic, N.K.; Apostolović, M.R.A.; Ignjatović, A.M.; Stojanovic, D.R.; Lakusic, V.R.M.; Ilic, M.V.J.M. Temporal Changes in Incidence Rates of the Most Common Gynecological Cancers in the Female Population in Central Serbia. Medicina 2022, 58, 306. [Google Scholar] [CrossRef] [PubMed]
- Nikolić, A.; Mitrašinović, P.; Mićanović, D.; Grujičić, S. Kretanje obolevanja i umiranja od kolorektalnog karcinoma kod muškaraca i žena centralne srbije za period 1999–2020. GODINE. Zdr. Zaštita 2023, 52, 46–63. [Google Scholar] [CrossRef]
- Mihalj, I.; Šušak, S.; Palanački-Malešević, T.; Važić, T.; Jurca, T.; Pavić, D.; Simeunović, J.; Vulin, A.; Meriluoto, J.; Svirčev, Z. Particulate air pollution in Central Serbia and some proposed measures for the restoration of degraded and disturbed mining areas. Geogr. Pannonica 2024, 28, 205–220. [Google Scholar] [CrossRef]
- Sipetic-Grujicic, S.; Murtezani, Z.; Ratkov, I.; Grgurevic, A.; Marinkovic, J.; Bjekic, M.; Miljus, D. Comparison of male and female breast cancer incidence and mortality trends in Central Serbia. Asian Pac. J. Cancer Prev. 2013, 14, 5681–5685. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mihajlović, J.; Pechlivanoglou, P.; Miladinov-Mikov, M.; Živković, S.; Postma, M.J. Cancer incidence and mortality in Serbia 1999–2009. BMC Cancer 2013, 13, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Perišić, Ž.; Plešinac-Karapandžić, V.; Džinić, M.; Zamurović, M.; Perišić, N. Cervical cancer screening in Serbia. Vojnosanit. Pregl. 2013, 70, 86–89. [Google Scholar] [CrossRef] [PubMed]
- Naumovic, T.; Miljus, D.; Djoric, M.; Zivkovic, S.; Perisic, Z. Mortality from cervical cancer in Serbia in the period 1991–2011. JBUON 2015, 20, 231–234. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Malignant Cancers for the Republic of Serbia 2021; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2023. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Malignant Cancers for the Republic of Serbia 2020; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2022. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Malignant Cancers for the Republic of Serbia 2019; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2021. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Malignant Cancers for the Republic of Serbia 2018; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2020. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Malignant Cancers for the Republic of Serbia 2017; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2020. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Malignant Cancers for the Republic of Serbia 2016; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2020. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Cancer Incidence and Mortality in Central Serbia 2015; Report No. 17; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2017. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Cancer Incidence and Mortality in Central Serbia 2014; Report No. 16; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2016. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Cancer Incidence and Mortality in Central Serbia 2013; Report No. 15; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2015. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Cancer Incidence and Mortality in Central Serbia 2012; Report No. 14; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2014. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Cancer Incidence and Mortality in Central Serbia 2011; Report No. 13; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2013. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Cancer Incidence and Mortality in Central Serbia 2010; Report No. 12; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2012. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Cancer Incidence and Mortality in Central Serbia 2009; Report No. 11; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2011. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Cancer Incidence and Mortality in Central Serbia 2008; Report No. 10; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2010. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Cancer Incidence and Mortality in Central Serbia 2006; Report No. 8; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2009. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Cancer Incidence and Mortality in Central Serbia 2005; Report No. 7; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2008. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Cancer Incidence and Mortality in Central Serbia 2004; Report No. 6; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2007. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Cancer Incidence and Mortality in Central Serbia 2003; Report No. 5; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2006. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Cancer Incidence and Mortality in Central Serbia 2002; Report No. 4; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2005. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Cancer Incidence and Mortality in Central Serbia 2001; Report No. 3; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2005. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Cancer Incidence and Mortality in Central Serbia 2000; Report No. 2; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2004. [Google Scholar]
- Institute of Public Health of Serbia “Dr Milan Jovanović Batut”. Cancer Incidence and Mortality in Central Serbia 1999; Report No. 1; Institute of Public Health of Serbia “Dr Milan Jovanović Batut”: Belgrade, Serbia, 2002. [Google Scholar]
- Salmi, T.M.A.; Anttila, P.; Ruoho-Airola, T.; Amnell, T. Detecting trends of annual values of atmospheric pollutants by the Mann-Kendall test and Sen’s slope estimates—The Excel template application MAKESENS. In Publications on Air Quality; No. 31.; Finnish Meteorological Institute: Helsinki, Finland, 2002; p. 35. [Google Scholar]
- Mann, H.B. Nonparametric tests against trend. Econom. J. Econom. Soc. 1945, 13, 245–259. [Google Scholar] [CrossRef]
- Kendall, M.G. Rank Correlation Methods, 4th ed.; Charles Griffin: London, UK, 1975. [Google Scholar]
- Gilbert, R.O. Statistical Methods for Environmental Pollution Monitoring; Van Nostrand Reinhold: New York, NY, USA, 1987; Volume 320. [Google Scholar]
- Bačević, N.R.; Pavlović, M.; Rasljanin, I. Trend assessing using Mann-Kendall’s test for Priština meteorological station temperature and precipitation data, Kosovo and Metohija, Serbia. Bull. Nat. Sci. Res. 2018, 8. [Google Scholar] [CrossRef]
- Milentijević, N.; Bačević, N.; Ristić, D.; Valjarević, A.; Pantelić, M.; Kićović, D. Application of Mann-Kendal (MK) test in trend analysis of air temperature and precipitation: Case of Mačva district (Serbia). Bull. Nat. Sci. Res. 2020, 10, 37–43. [Google Scholar] [CrossRef]
- Trbić, G.; Popov, T.; Gnjato, S. Analysis of air temperature trends in Bosnia and Herzegovina. Geogr. Pannonica 2017, 21, 68–84. [Google Scholar] [CrossRef]
- Kričković, E.; Lukić, T.; Srejić, T.; Stojšić-Milosavljević, A.; Stojanović, V.; Kričković, Z. Spatial-temporal and trend analysis of traffic accidents in AP Vojvodina (North Serbia). Open Geosci. 2024, 16, 20220630. [Google Scholar] [CrossRef]
- Kričković, E.; Lukić, T.; Jovanović-Popović, D. Geographic medical overview of noncommunicable diseases (cardiovascular diseases and diabetes) in the territory of the AP Vojvodina (Northern Serbia). Healthcare 2022, 11, 48. [Google Scholar] [CrossRef]
- Gavrilov, M.B.; Marković, S.B.; Jarad, A.; Korać, V.M. The analysis of temperature trends in Vojvodina (Serbia) from 1949 to 2006. Therm. Sci. 2015, 19, 339–350. [Google Scholar] [CrossRef]
- Hussain, M.; Mahmud, I. pyMannKendall: A python package for non parametric Mann Kendall family of trend tests. J. Open Source Softw. 2019, 4, 1556. [Google Scholar] [CrossRef]
- Drobnjaković, M.; Čikić, J. The Socio-Spatial Aspect of Rurality—A Case Study of Rural Settlements in Central Serbia. Mitt. Österreich. Geogr. Ges. 2020, 162, 469–494. [Google Scholar] [CrossRef]
- Statistical Office of the Republic of Serbia. Demographic Yearbook 2022; Statistical Office of the Republic of Serbia: Belgrade, Serbia, 2023.
- Marinković, I. Census of Population 2011. In Basic Population Structures of Serbia, Demographic Review; Rašević, M., Ed.; Ministry of Labour, Employment, and Social Policy in Cooperation with the Centre for Demographic Research of the Institute of Social Sciences and the Serbian Demographers’ Society: Belgrade, Serbia, 2013; Year XIII, No. 47. Available online: https://www.minrzs.gov.rs/sites/default/files/201811/47%20Popis%20stanovnistva%202011Osnovne%20strukture%20populacije%20Srbije%2C.pdf (accessed on 13 August 2025).
- Marković-Denić, L.J.; Živković, S.; Sipetić, S.; Vlajinać, H.; Kočev, N.; Marinković, J. Time trends in cancer mortality in central Serbia. Soz.-Präventivmed. 2006, 51, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Fu, Z.; Lu, Z.; Chu, J.; Xu, A.; Guo, X.; Ma, J. Cancer survival analysis and spatial distribution during 2014–2016 in Shandong Province, China. Sci. Rep. 2023, 13, 10324. [Google Scholar] [CrossRef] [PubMed]
- Aldrich, N.; Benson, W.F. Disaster Preparedness and the Chronic Disease Needs of Vulnerable Older Adults. Prev. Chronic Dis. 2007, 5, A27. [Google Scholar] [PubMed]
- Bethel, J.W.; Foreman, A.N.; Burke, S.C. Disaster preparedness among medically vulnerable populations. Am. J. Prev. Med. 2011, 40, 139–143. [Google Scholar] [CrossRef]
- Hassan, S.; Nguyen, M.; Buchanan, M.; Grimshaw, A.; Adams, O.P.; Hassell, T.; Ragster, L.; Nunez-Smith, M. Management Of Chronic Noncommunicable Diseases After Natural Disasters In The Caribbean: A Scoping Review. Health Aff. 2020, 39, 2136–2143. [Google Scholar] [CrossRef] [PubMed]
- Kang, K. Disaster preparedness among vulnerable older adults with chronic diseases: Results from a cross-sectional study in Incheon, Korea. Nurs. Health Sci. 2014, 16, 46–51. [Google Scholar] [CrossRef]
- Ko, J.Y.; Strine, T.W.; Allweiss, P. Chronic Conditions and Household Preparedness for Public Health Emergencies: Behavioral Risk Factor Surveillance System, 2006–2010. Prehospital Disaster Med. 2013, 29, 13–20. [Google Scholar] [CrossRef] [PubMed]
- IQAir. Serbia Air Quality Index (AQI) and Air Pollution Information. 2024. Available online: https://www.iqair.com/serbia (accessed on 13 August 2025).
- World Health Organization. Health Impact of Ambient Air Pollution in Serbia. 2019. Available online: https://serbia.un.org/sites/default/files/2019-10/Health-impact-pollution-Serbia_0.pdf (accessed on 13 August 2025).
- Stojanović, M.M.; Rančić, N.K.; Apostolović, M.R.A.; Ignjatović, A.M.; Ilić, M.V. Trends of Stomach Cancer in Central Serbia. Medicina 2021, 57, 665. [Google Scholar] [CrossRef] [PubMed]
- Wroblewski, L.E.; Peek, R.M., Jr.; Wilson, K.T. Helicobacter pylori and Gastric Cancer: Factors That Modulate Disease Risk. Clin. Microbiol. Rev. 2010, 23, 713–739. [Google Scholar] [CrossRef]
- Singh, S.; Jha, H.C. Status of Epstein-Barr Virus Coinfection with Helicobacter pylori in Gastric Cancer. J. Oncol. 2017, 2017, 3456264. [Google Scholar] [CrossRef]
- Sasco, A.J.; Secretan, M.B.; Straif, K. Tobacco Smoking and Cancer: A Brief Review of Recent Epidemiological Evidence. Lung Cancer 2004, 45 (Suppl. S2), S3–S9. [Google Scholar] [CrossRef]
- La Rosa, F.; Iannacci, F.; Trotta, F. Fumo di Tabacco e Tumori Maligni. Analisi per Coorti di Nascita dal 1875 al 1935. Tobacco Smoke and Malignant Tumors. Analysis by Birth Cohorts from 1875 to 1935. Ann. Ig. 1989, 1, 1549–1571. (In Italian) [Google Scholar] [PubMed]
- O’Rourke, M.; Djukic, J. Welcome to Serbia: Feel Free to Smoke. Tob. Control. 2008, 17, 428–430. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer (IARC) Working Group. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans: Alcohol Consumption; IARC: Lyon, France, 2010; Volume 96. [Google Scholar]
- Statistical Office of the Republic of Serbia; Institute for Public Health of Serbia “Dr. Milan Jovanović Batut”; Milić, N.; Stanisavljević, D.; Krstić, M. (Eds.) Survey of the Health of the Population of Serbia in 2019; Statistical Office of the Republic of Serbia: Belgrade, Serbia, 2021.
- Kruk, J.; Aboul-Enein, B.H.; Bernstein, J.; Gronostaj, M. Psychological Stress and Cellular Aging in Cancer: A Meta-Analysis. Oxidative Med. Cell. Longev. 2019, 2019, 1270397. [Google Scholar] [CrossRef] [PubMed]
- Massicotte, V.; Ivers, H.; Savard, J. COVID-19 Pandemic Stressors and Psychological Symptoms in Breast Cancer Patients. Curr. Oncol. 2021, 28, 294–300. [Google Scholar] [CrossRef]
- Yang, T.; Qiao, Y.; Xiang, S.; Li, W.; Gan, Y.; Chen, Y. Work stress and the risk of cancer: A meta-analysis of observational studies. Int. J. Cancer 2018, 144, 2390–2400. [Google Scholar] [CrossRef]
- Dai, S.; Mo, Y.; Wang, Y.; Xiang, B.; Liao, Q.; Zhou, M.; Li, X.; Li, Y.; Xiong, W.; Li, G.; et al. Chronic Stress Promotes Cancer Development. Front. Oncol. 2020, 10, 1492. [Google Scholar] [CrossRef]
- Oh, H.-M.; Son, C.-G. The Risk of Psychological Stress on Cancer Recurrence: A Systematic Review. Cancers 2021, 13, 5816. [Google Scholar] [CrossRef]
- Gallaway, M.S.; Idaikkadar, N.; Tai, E.; Momin, B.; Rohan, E.A.; Townsend, J.; Puckett, M.; Stewart, S.L. Emergency department visits among people with cancer: Frequency, symptoms, and characteristics. J. Am. Coll. Emerg. Physicians Open 2021, 2, e12438. [Google Scholar] [CrossRef]
- McFerran, E.; Cairnduff, V.; Elder, R.; Gavin, A.; Lawler, M. Cost consequences of unscheduled emergency admissions in cancer patients in the last year of life. Support. Care Cancer 2023, 31, 201. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.; Grudzen, C.; Kyriacou, D.N.; Obermeyer, Z.; Quest, T.; Rivera, D.; Stone, S.; Wright, J.; Shelburne, N. The Emergency Care of Patients With Cancer: Setting the Research Agenda. Ann. Emerg. Med. 2016, 68, 706–711. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, S.; Aryal, K.; King, J.; Bischof, J.J.; Hong, A.S.; Wood, N.; Rothberg, B.E.G.; Hudson, M.F.; Heinert, S.W.; Wattana, M.K.; et al. Understanding oncologic emergencies and related emergency department visits and hospitalizations: A systematic review. BMC Emerg. Med. 2025, 25, 40. [Google Scholar] [CrossRef]
- Mavrodieva, A.V.; Budiarti, D.S.; Yu, Z.; Pasha, F.A.; Shaw, R. Governmental Incentivization for SMEs’ Engagement in Disaster Resilience in Southeast Asia. Int. J. Disaster Risk Manag. 2019, 1, 32–50. [Google Scholar] [CrossRef]
- Al-Ramlawi, A.H.; El-Mougher, M.M.; Al-Agha, M.R. The Role of Al-Shifa Medical Complex Administration in Evacuation & Sheltering Planning. Int. J. Disaster Risk Manag. 2020, 2, 19–36. [Google Scholar] [CrossRef]
- De Pinto, G.; Mignozzi, S.; La Vecchia, C.; Levi, F.; Negri, E.; Santucci, C. Global trends in cutaneous malignant melanoma incidence and mortality. Melanoma Res. 2024, 34, 265–275. [Google Scholar] [CrossRef]
- Huang, J.; Deng, Y.; Tin, M.S.; Lok, V.; Ngai, C.H.; Zhang, L.; Lucero-Prisno, D.E.; Xu, W.; Zheng, Z.-J.; Elcarte, E.; et al. Distribution, Risk Factors, and Temporal Trends for Lung Cancer Incidence and Mortality: A global analysis. Chest 2022, 161, 1101–1111. [Google Scholar] [CrossRef]
- Santucci, C.; Carioli, G.; Bertuccio, P.; Malvezzi, M.; Pastorino, U.; Boffetta, P.; Negri, E.; Bosetti, C.; La Vecchia, C. Progress in cancer mortality, incidence, and survival: A global overview. Eur. J. Cancer Prev. 2020, 29, 367–381. [Google Scholar] [CrossRef]
- Torre, L.A.; Siegel, R.L.; Ward, E.M.; Jemal, A. Global Cancer Incidence and Mortality Rates and Trends—An Update. Cancer Epidemiol. Biomark. Prev. 2016, 25, 16–27. [Google Scholar] [CrossRef]
- Yang, J.; Lou, S.; Yao, T. Trends in primary malignant bone cancer incidence and mortality in the United States, 2000–2017: A population-based study. J. Bone Oncol. 2024, 46, 100607. [Google Scholar] [CrossRef]
- Abdin, A.F.; Fang, Y.-P.; Caunhye, A.; Alem, D.; Barros, A.; Zio, E. An optimization model for planning testing and control strategies to limit the spread of a pandemic—The case of COVID-19. Eur. J. Oper. Res. 2023, 304, 308–324. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Chong, W.F.; Feng, R.; Zhang, L. Pandemic risk management: Resources contingency planning and allocation. Insur. Math. Econ. 2021, 101, 359–383. [Google Scholar] [CrossRef]
- Gail, M.H.; Pee, D. Robustness of risk-based allocation of resources for disease prevention. Stat. Methods Med. Res. 2020, 29, 3511–3524. [Google Scholar] [CrossRef]
- Sheng, M.; Wang, H.; Ma, M.; Sun, Y.; Zhou, R. Risk Assessment Edge Contract for Efficient Resource Allocation. Mathematics 2024, 12, 983. [Google Scholar] [CrossRef]
- Wei, Z.; Zhuang, J. Modeling defensive resource allocation in multilayered systems under probabilistic and strategic risks. Risk Anal. 2024, 45, 177–193. [Google Scholar] [CrossRef]
- Johnson, M.; Albizri, A.; Simsek, S. Artificial intelligence in healthcare operations to enhance treatment outcomes: A framework to predict lung cancer prognosis. Ann. Oper. Res. 2020, 308, 275–305. [Google Scholar] [CrossRef]
- Hommes, S.; Clouth, F.; Vromans, R.; Geleijnse, G.; Pauws, S.; Vermunt, J.; van de Poll-Franse, L.V.; Krahmer, E. Data-driven shared decision-making on cancer treatment. In Proceedings of the Big Data, Amersfoort, The Netherlands, 2 October 2019. [Google Scholar]
- Liu, K.; Chen, Z.; Wu, J.; Tan, Y.; Wang, L.; Yan, Y.; Zhang, H.; Long, J. Big Medical Data Decision-Making Intelligent System Exploiting Fuzzy Inference Logic for Prostate Cancer in Developing Countries. IEEE Access 2019, 7, 2348–2363. [Google Scholar] [CrossRef]
- Lewis, L.; Wagland, R.; Patel, H.; Bridges, J.; Farrington, N.; Hunt, K. 2816 Decision-making experiences with older adults following a cancer diagnosis: A systematic review. Age Ageing 2025, 54, I29–I30. [Google Scholar] [CrossRef]












| County/Region/Area | Population Number | Percentage in Women | Average Age | ||||
|---|---|---|---|---|---|---|---|
| Total | Men | Women | Total | Men | Women | ||
| Beogradski | 1,681,405 | 794,413 | 886,992 | 52.8% | 42.7 | 41.0 | 44.3 |
| Borski | 101,100 | 49,453 | 51,647 | 51.1% | 47.5 | 46.0 | 49.0 |
| Braničevski | 156,367 | 76,881 | 79,486 | 50.8% | 46.5 | 44.9 | 48.1 |
| Jablanički | 184,502 | 92,220 | 92,282 | 50.0% | 44.4 | 43.5 | 45.3 |
| Kolubarski | 154,497 | 76,563 | 77,934 | 50.4% | 45.0 | 43.8 | 46.1 |
| Mačvanski | 265,377 | 131,188 | 134,189 | 50.6% | 44.4 | 43.3 | 45.5 |
| Moravički | 189,281 | 92,817 | 96,464 | 51.0% | 45.1 | 43.7 | 46.4 |
| Nišavski | 343,950 | 168,367 | 175,583 | 51.0% | 44.3 | 43.2 | 45.4 |
| Pčinjski | 193,802 | 97,273 | 96,529 | 49.8% | 41.7 | 41.0 | 42.5 |
| Pirotski | 76,700 | 38,762 | 37,938 | 49.5% | 47.2 | 46.4 | 47.9 |
| Podunavski | 175,573 | 86,811 | 88,762 | 50.6% | 44.0 | 42.5 | 45.5 |
| Pomoravski | 182,047 | 88,333 | 93,714 | 51.5% | 46.2 | 44.8 | 47.6 |
| Rasinski | 207,197 | 101,873 | 105,324 | 50.8% | 46.0 | 44.6 | 47.2 |
| Raški | 296,532 | 147,411 | 149,121 | 50.3% | 40.7 | 39.6 | 41.7 |
| Šumadijski | 269,728 | 131,388 | 138,340 | 51.3% | 44.2 | 42.9 | 45.3 |
| Toplički | 77,341 | 39,031 | 38,310 | 49.5% | 44.3 | 43.4 | 45.2 |
| Zaječarski | 96,715 | 47,188 | 49,527 | 51.2% | 48.6 | 47.0 | 50.2 |
| Zlatiborski | 254,659 | 126,267 | 128,392 | 50.4% | 44.9 | 43.8 | 46.0 |
| Serbia—South | 3,225,368 | 1,591,826 | 1,633,542 | 50.6% | 44.6 | 43.3 | 45.7 |
| Region of Šumadija and Western Serbia | 1,819,318 | 895,840 | 923,478 | 50.8% | 44.3 | 43.1 | 45.5 |
| Region of Southern and Eastern Serbia | 1,406,050 | 695,986 | 710,064 | 50.5% | 44.9 | 43.7 | 46.0 |
| Serbia | 6,647,003 | 3,231,978 | 3,415,025 | 51.4% | 43.9 | 42.4 | 45.2 |
| Research Focus Area | Detected Trends (1999–2021) | Recommended Actions |
|---|---|---|
| Breast cancer incidence rates (6 counties) | Increasing trends in Borski, Jablanički, Kolubarski, Moravički, Pčinjski, Rasinski | Strengthen local screening programmes (mammography), deploy mobile units; educate health workers and the public; integrate oncology protocols into crisis plans. |
| Breast cancer mortality rates | Increase in Kolubarski; decreases in Beogradski and Moravički | Evaluate treatment accessibility and transportation in Kolubarski; share best practices from regions with declining mortality; implement rapid response teams for treatment continuity. |
| Cervical cancer incidence and mortality rates | Declines in Beogradski, Braničevski, Jablanički, Moravički, Podunavski, Toplički (incidence and/or mortality) | Expand HPV vaccination; update and broaden screening coverage; monitor areas where decline is not observed. |
| Lung/bronchus cancer incidence and mortality rates | Incidence up in 17 counties; mortality up in 16 | Broaden anti-smoking initiatives; adopt early diagnostic protocols; integrate oncology scenarios into disaster and pollution response plans. |
| Ovarian cancer incidence and mortality rates | Incidence up in Borski, Kolubarski, Pomoravski; mortality up in Borski | Launch reproductive health education programmes; support gynecologic oncology clinics in affected regions; include protocols in emergency preparedness plans. |
| Uterine cancer incidence rates | Increase in Borski and Raški; decrease in Jablanički and Moravički | Investigate causes of regional variation; apply targeted public health strategies (education, early testing). |
| Pancreatic cancer incidence and mortality rates | Incidence up in 15 counties; mortality up in 7 | Develop early diagnostic approaches (biomarkers, imaging); train medical staff in symptom recognition; include oncology in mobile crisis health units. |
| Bladder cancer incidence rates | Increase in 12 counties | Monitor exposure factors (pollution, occupational risks); promote healthy diets and risk reduction; include as long-term health risks in crisis scenarios. |
| Colorectal cancer incidence and mortality rates | Increased incidence in 8 counties; only Borski saw increased mortality rates | Promote dietary interventions and regular screenings (e.g., colonoscopy); analyse regional health trends; prioritise outpatient clinics in emergency planning. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kričković, E.; Cvetković, V.M.; Kričković, Z.; Lukić, T. Spatial–Temporal Trends of Cancer Among Women in Central Serbia, 1999–2021: Implications for Disaster and Public Health Preparedness. Healthcare 2025, 13, 2169. https://doi.org/10.3390/healthcare13172169
Kričković E, Cvetković VM, Kričković Z, Lukić T. Spatial–Temporal Trends of Cancer Among Women in Central Serbia, 1999–2021: Implications for Disaster and Public Health Preparedness. Healthcare. 2025; 13(17):2169. https://doi.org/10.3390/healthcare13172169
Chicago/Turabian StyleKričković, Emina, Vladimir M. Cvetković, Zoran Kričković, and Tin Lukić. 2025. "Spatial–Temporal Trends of Cancer Among Women in Central Serbia, 1999–2021: Implications for Disaster and Public Health Preparedness" Healthcare 13, no. 17: 2169. https://doi.org/10.3390/healthcare13172169
APA StyleKričković, E., Cvetković, V. M., Kričković, Z., & Lukić, T. (2025). Spatial–Temporal Trends of Cancer Among Women in Central Serbia, 1999–2021: Implications for Disaster and Public Health Preparedness. Healthcare, 13(17), 2169. https://doi.org/10.3390/healthcare13172169








