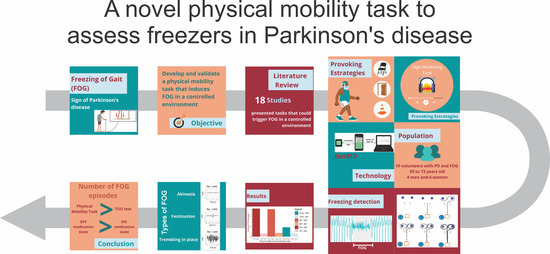
A Novel Physical Mobility Task to Assess Freezers in Parkinson’s Disease
Abstract
1. Introduction
2. Materials and Methods
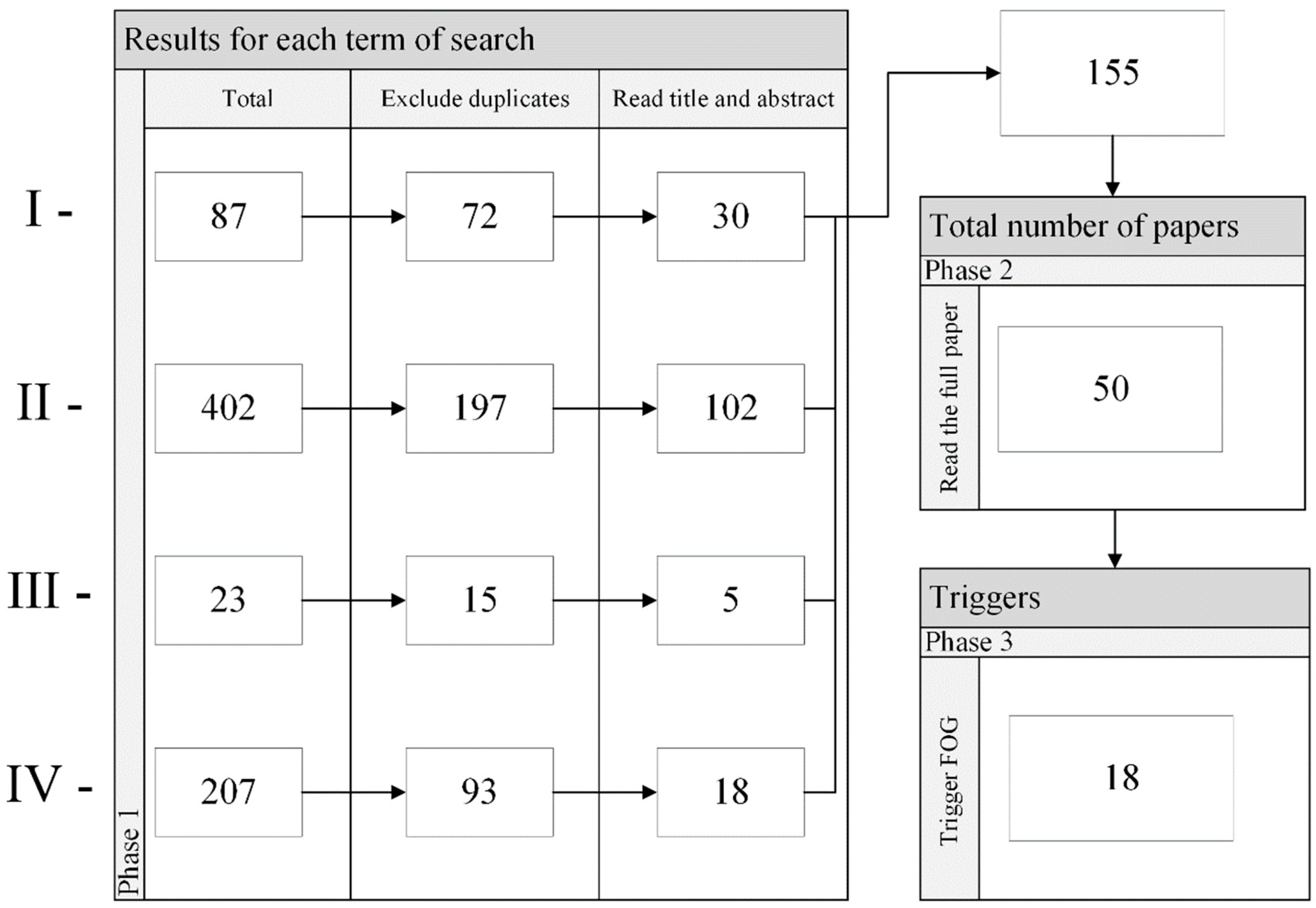
2.1. Literature Review
Result of the Literature Review
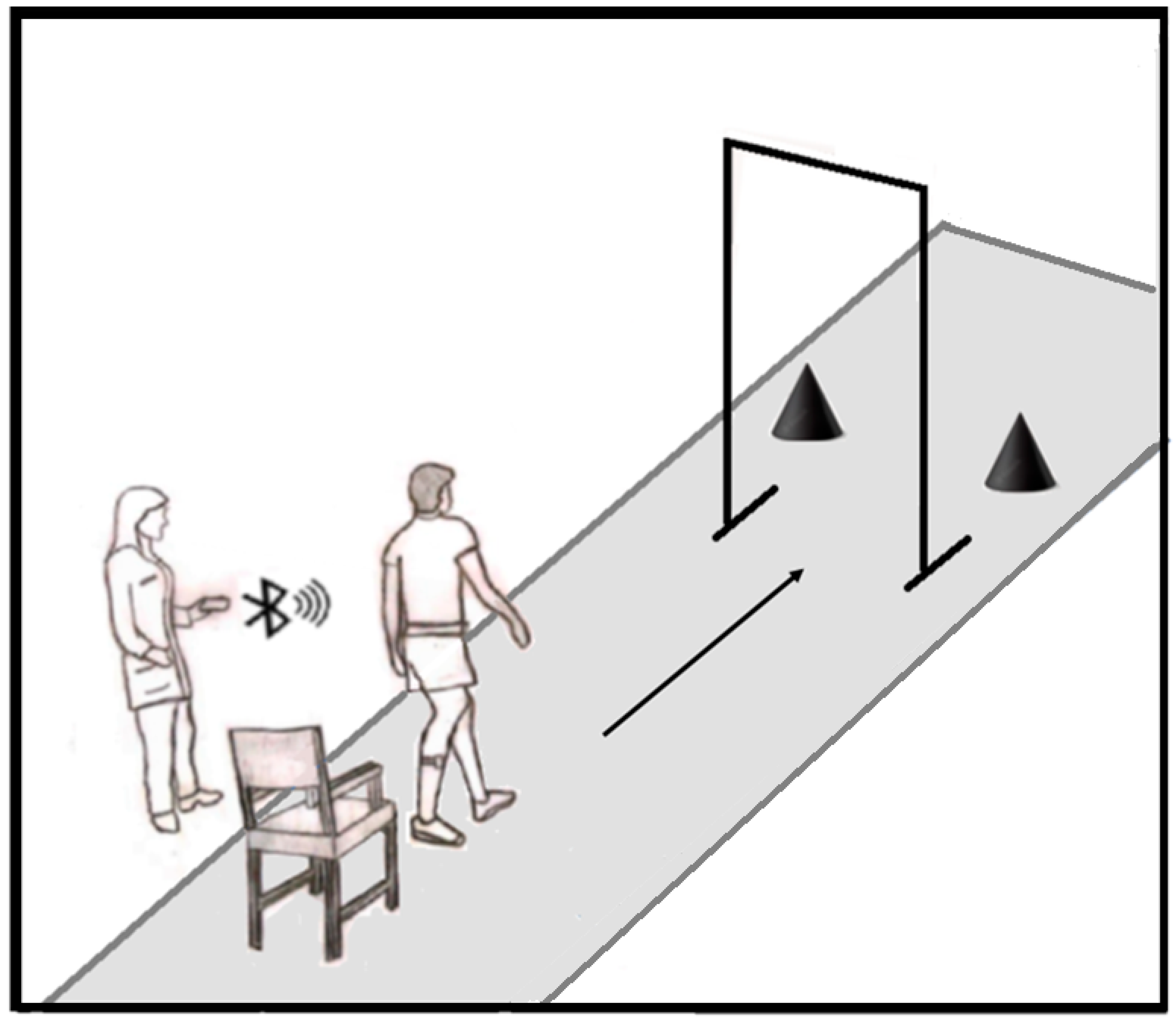
2.2. Creating the Physical Mobility Task to Trigger FOG
2.2.1. Motor Task
- Initially, the subject performs a 360-degree turn to avoid the obstacle located on the same side as the subject’s most affected body part, as determined during the clinical evaluation by the physical therapist;
- The volunteer then moves toward the second obstacle and performs a complete 360-degree turn to avoid it;
- Finally, they move in the direction of the first obstacle and execute a second 360-degree turn to avoid it;
- Then they return to the direction of the 67.5 cm-wide opening.The task is complete once the subject is seated in the chair.
2.2.2. Cognitive-Motor Dual Task
2.3. Testing the Physical Mobility Motor Task
2.3.1. Subjects
2.3.2. Data Analysis
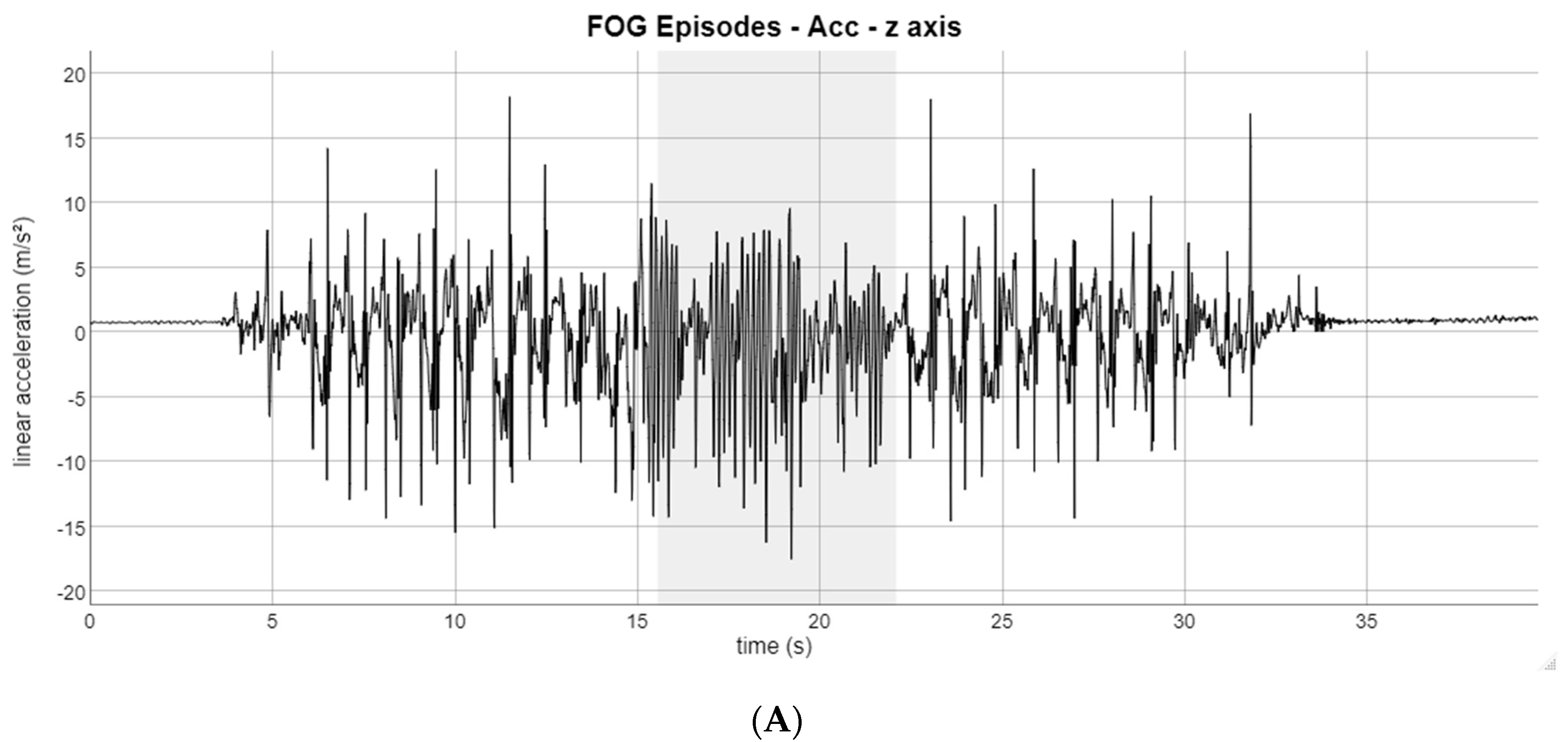
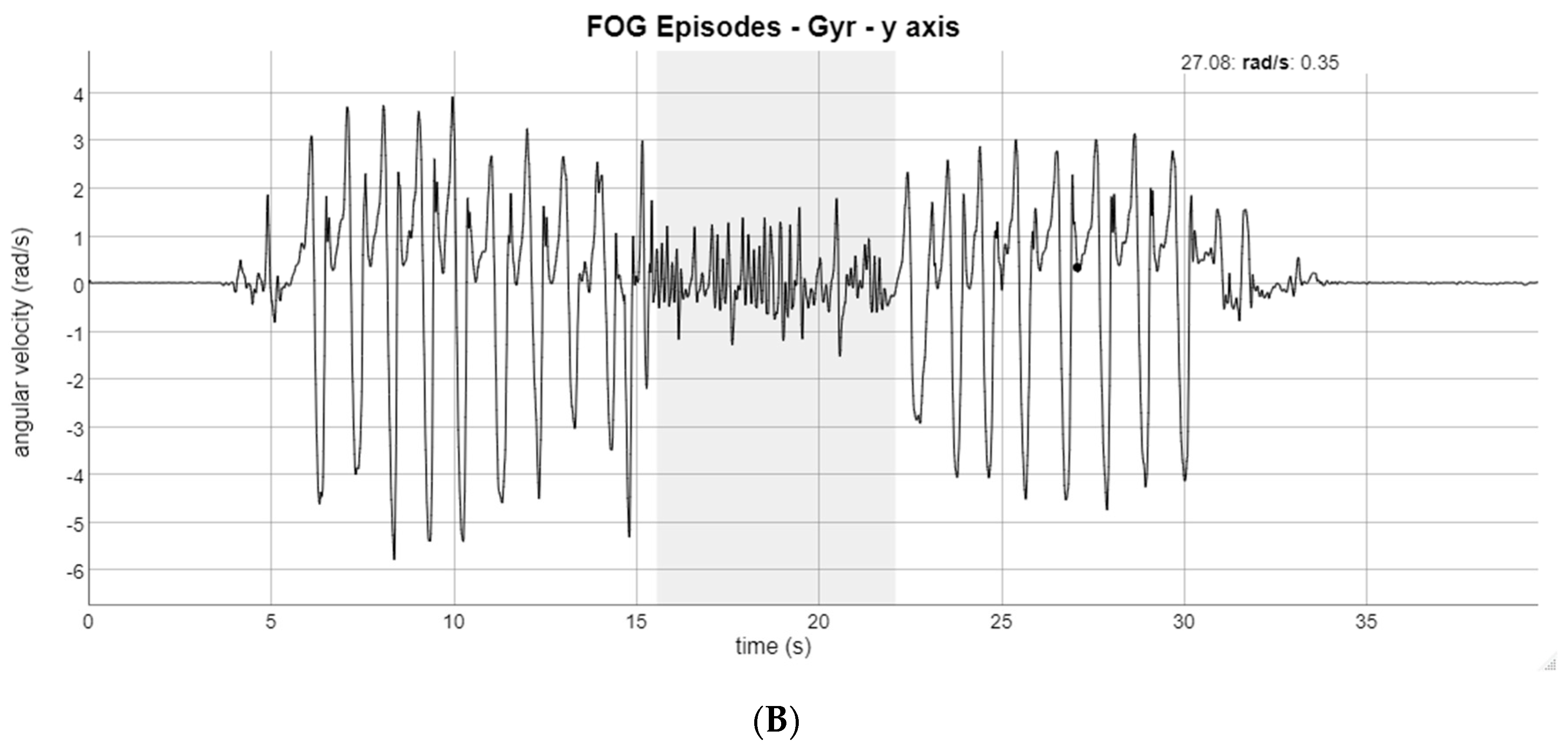
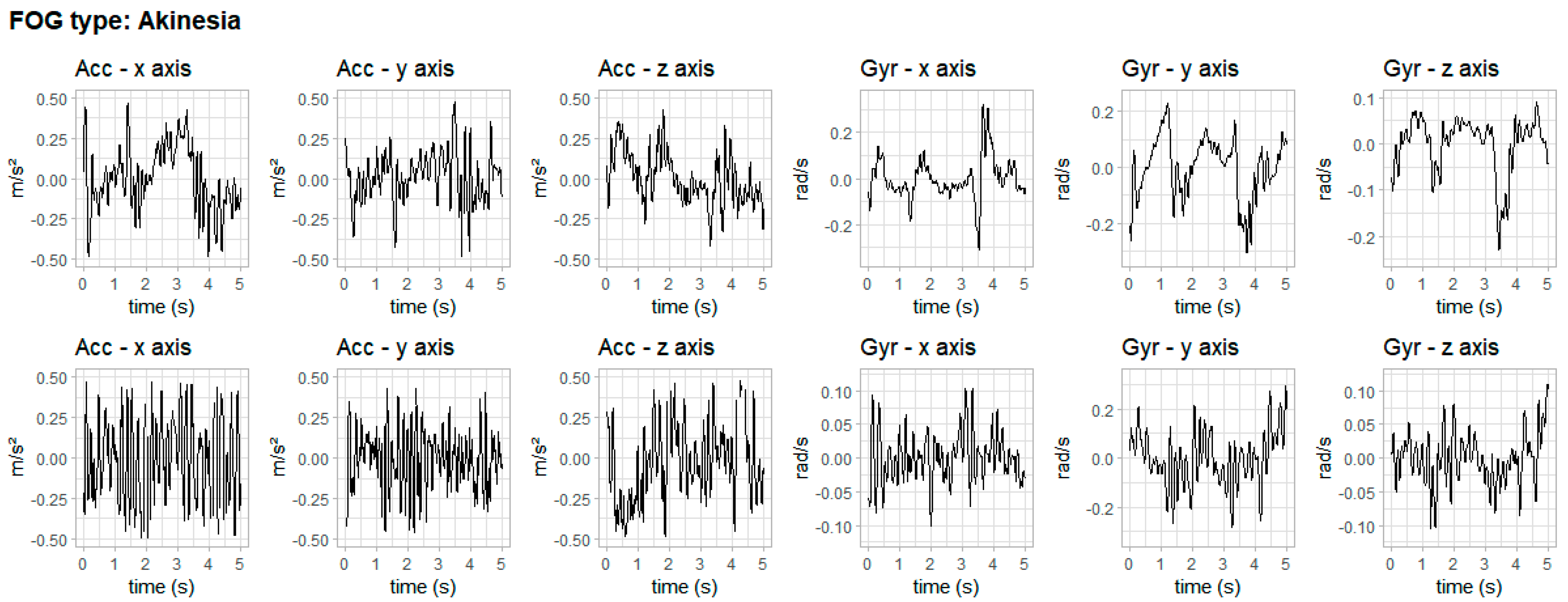
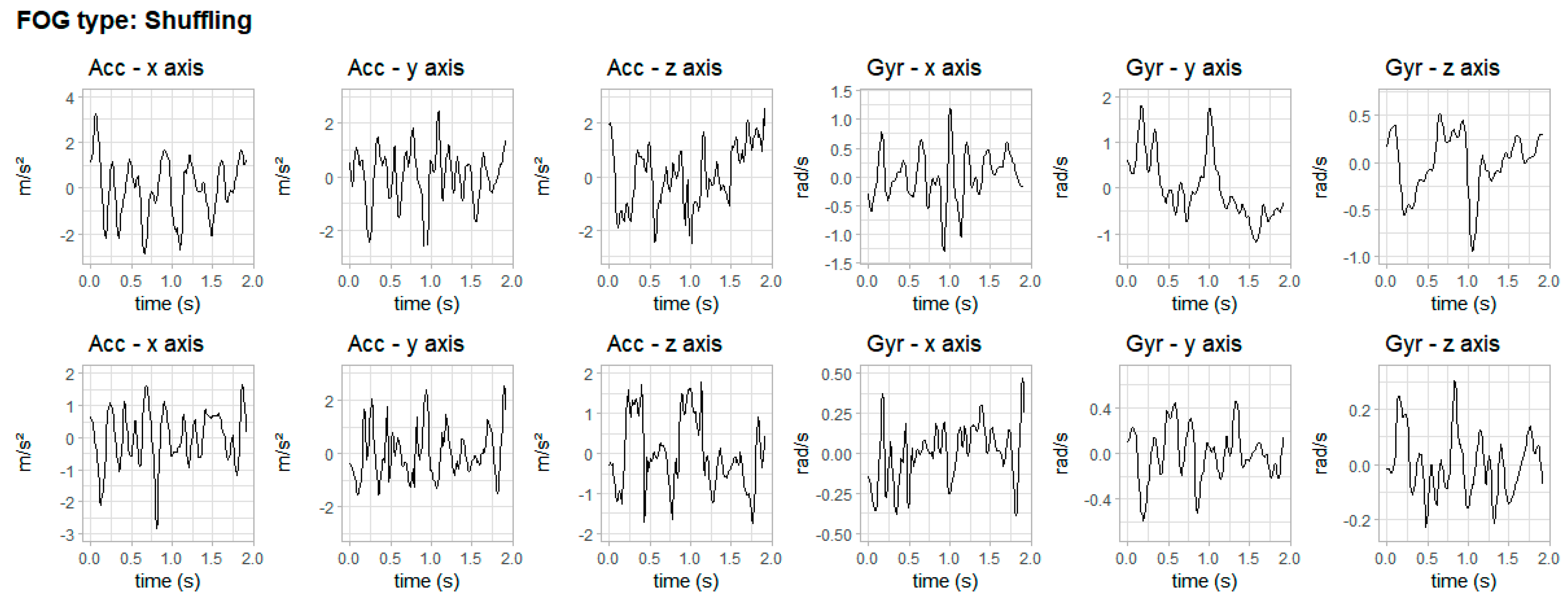
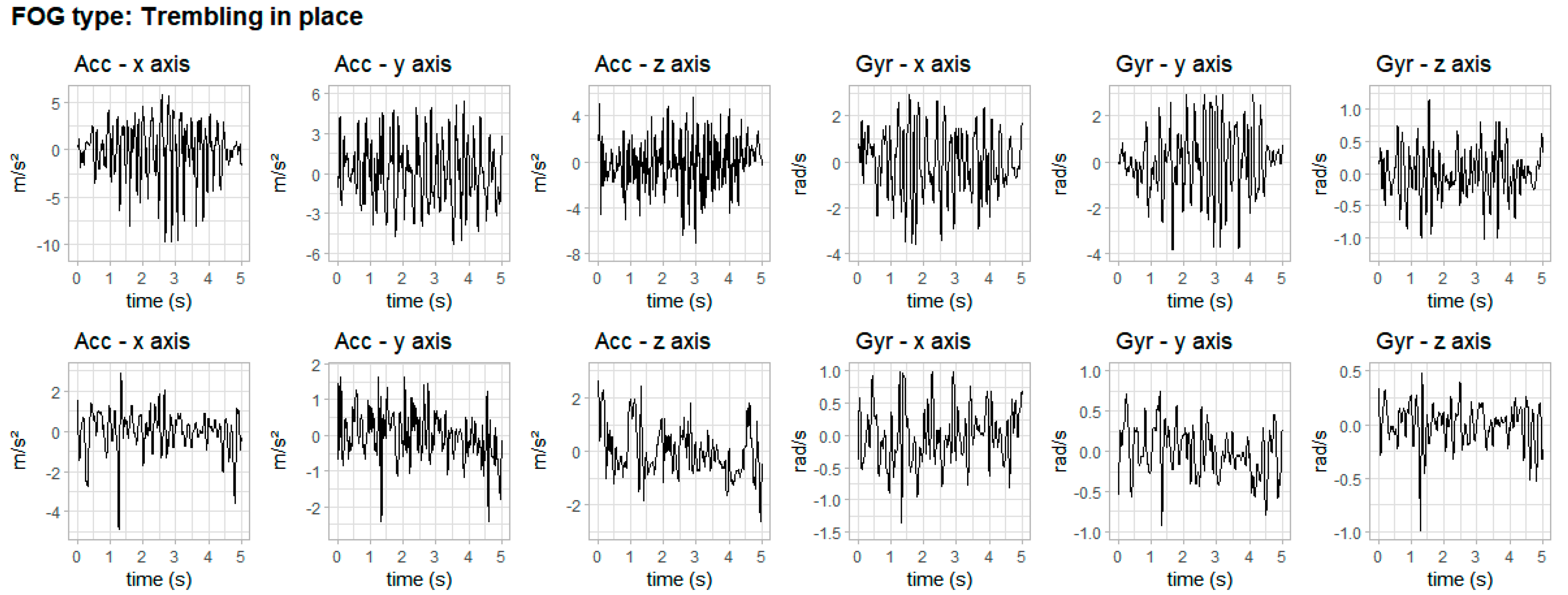
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bertoli, M.; Cereatti, A.; Della Croce, U.; Mancini, M. An Objective Assessment to Investigate the Impact of Turning Angle on Freezing of Gait in Parkinson’s Disease. In Proceedings of the 2017 IEEE Biomedical Circuits and Systems Conference (BioCAS), Turin, Italy, 19–21 October 2017; pp. 1–4. [Google Scholar]
- Mancini, M.; Smulders, K.; Cohen, R.G.; Horak, F.B.; Giladi, N.; Nutt, J.G. The Clinical Significance of Freezing While Turning in Parkinson’s Disease. Neuroscience 2017, 343, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Okuma, Y. Practical Approach to Freezing of Gait in Parkinson’s Disease. Pract. Neurol. 2014, 14, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Bartels, A.L.; Balash, Y.; Gurevich, T.; Schaafsma, J.D.; Hausdorff, J.M.; Giladi, N. Relationship between Freezing of Gait (FOG) and Other Features of Parkinson’s: FOG Is Not Correlated with Bradykinesia. J. Clin. Neurosci. 2003, 10, 584–588. [Google Scholar] [CrossRef] [PubMed]
- Rawson, K.S.; Creel, P.; Templin, L.; Horin, A.P.; Duncan, R.P.; Earhart, G.M. Freezing of Gait Boot Camp: Feasibility, Safety and Preliminary Efficacy of a Community-Based Group Intervention. Neurodegener. Dis. Manag. 2018, 8, 307–314. [Google Scholar] [CrossRef]
- Saad, A.; Guerin, F.; Zaarour, I.; Ayache, M.; Lefebvre, D. Sensoring and Features Extraction for the Detection of Freeze of Gait in Parkinson Disease. In Proceedings of the 2014 IEEE 11th International Multi-Conference on Systems, Signals & Devices (SSD14), Barcelona, Spain, 11–14 February 2014; IEEE: Piscataway, NJ, USA; Laboratoire GREAH-Groupe de Recherche en Electrotechnique et Automatique du Havre, Université du Havre: Le Havre, France, 2014; pp. 1–6. [Google Scholar]
- Morris, M.E.; Iansek, R.; Galna, B. Gait Festination and Freezing in Parkinson’s Disease: Pathogenesis and Rehabilitation. Mov. Disord. 2008, 23, S451–S460. [Google Scholar] [CrossRef] [PubMed]
- Popovic, M.B.; Djuric-Jovicic, M.; Radovanovic, S.; Petrovic, I.; Kostic, V. A Simple Method to Assess Freezing of Gait in Parkinson’s Disease Patients. Braz. J. Med. Biol. Res. 2010, 43, 883–889. [Google Scholar] [CrossRef] [PubMed]
- Jovanov, E.; Wang, E.; Verhagen, L.; Fredrickson, M.; Fratangelo, R. DeFOG—A Real Time System for Detection and Unfreezing of Gait of Parkinson’s Patients. In Proceedings of the 2009 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Minneapolis, MN, USA, 3–6 September 2009; IEEE: Piscataway, NJ, USA, 2009; pp. 5151–5154. [Google Scholar]
- Okuma, Y. Freezing of Gait and Falls in Parkinson’s Disease. J. Parkinsons Dis. 2014, 4, 255–260. [Google Scholar] [CrossRef]
- Moore, S.T.; MacDougall, H.G.; Ondo, W.G. Ambulatory Monitoring of Freezing of Gait in Parkinson’s Disease. J. Neurosci. Methods 2008, 167, 340–348. [Google Scholar] [CrossRef]
- Schaafsma, J.D.; Balash, Y.; Gurevich, T.; Bartels, A.L.; Hausdorff, J.M.; Giladi, N. Characterization of Freezing of Gait Subtypes and the Response of Each to Levodopa in Parkinson’s Disease. Eur. J. Neurol. 2003, 10, 391–398. [Google Scholar] [CrossRef]
- Spildooren, J.; Vercruysse, S.; Desloovere, K.; Vandenberghe, W.; Kerckhofs, E.; Nieuwboer, A. Freezing of Gait in Parkinson’s Disease: The Impact of Dual-Tasking and Turning. Mov. Disord. 2010, 25, 2563–2570. [Google Scholar] [CrossRef]
- Shine, J.M.; Matar, E.; Ward, P.B.; Frank, M.J.; Moustafa, A.A.; Pearson, M.; Naismith, S.L.; Lewis, S.J.G. Freezing of Gait in Parkinson’s Disease Is Associated with Functional Decoupling between the Cognitive Control Network and the Basal Ganglia. Brain 2013, 136, 3671–3681. [Google Scholar] [CrossRef] [PubMed]
- Beck, E.N.; Ehgoetz Martens, K.A.; Almeida, Q.J. Freezing of Gait in Parkinson’s Disease: An Overload Problem? PLoS ONE 2015, 10, e0144986. [Google Scholar] [CrossRef] [PubMed]
- Goetz, C.G.; Tilley, B.C.; Shaftman, S.R.; Stebbins, G.T.; Fahn, S.; Martinez-Martin, P.; Poewe, W.; Sampaio, C.; Stern, M.B.; Dodel, R.; et al. Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): Scale Presentation and Clinimetric Testing Results. Mov. Disord. 2008, 23, 2129–2170. [Google Scholar] [CrossRef] [PubMed]
- Huerta, M.; Barzallo, B.; Punin, C.; Garcia-Cedeño, A.; Clotet, R. Review of Active Extracorporeal Medical Devices to Counteract Freezing of Gait in Patients with Parkinson Disease. Healthcare 2022, 10, 976. [Google Scholar] [CrossRef]
- Podsiadlo, D.; Richardson, S. The Timed Up and Go: A Test of Basic Functional Mobility for Frail Elderly Persons. J. Am. Geriatr. Soc. 1991, 39, 142–148. [Google Scholar] [CrossRef]
- Muralidharan, V.; Balasubramani, P.P.; Chakravarthy, V.S.; Gilat, M.; Lewis, S.J.G.; Moustafa, A.A. A Neurocomputational Model of the Effect of Cognitive Load on Freezing of Gait in Parkinson’s Disease. Front. Hum. Neurosci. 2017, 10, 649. [Google Scholar] [CrossRef]
- Shine, J.M.; Moustafa, A.A.; Matar, E.; Frank, M.J.; Lewis, S.J.G. The Role of Frontostriatal Impairment in Freezing of Gait in Parkinson’s Disease. Front. Syst. Neurosci. 2013, 7, 61. [Google Scholar] [CrossRef]
- Killane, I.; Fearon, C.; Newman, L.; McDonnell, C.; Waechter, S.M.; Sons, K.; Lynch, T.; Reilly, R.B. Dual Motor-Cognitive Virtual Reality Training Impacts Dual-Task Performance in Freezing of Gait. IEEE J. Biomed. Health Inform. 2015, 19, 1855–1861. [Google Scholar] [CrossRef]
- Waechter, S.M.; Fearon, C.; McDonnell, C.; Gallego, J.; Quinlivan, B.; Killane, I.; Butler, J.S.; Lynch, T.; Reilly, R.B. The Impact of Dual Tasking on Cognitive Performance in a Parkinson’s Disease Cohort with and without Freezing of Gait: An EEG and Behavioral Based Approach. In Proceedings of the 2015 7th International IEEE/EMBS Conference on Neural Engineering (NER), Montpellier, France, 22–24 April 2015; IEEE: Piscataway, NJ, USA, 2015; pp. 1072–1075. [Google Scholar]
- Tard, C.; Dujardin, K.; Bourriez, J.; Molaee-ardekani, B. Attention Modulation during Motor Preparation in Parkinsonian Freezers: A Time-Frequency EEG Study. Clin. Neurophysiol. 2016, 127, 3506–3515. [Google Scholar] [CrossRef]
- Velik, R. Effect of On-Demand Cueing on Freezing of Gait in Parkinson’ s Patients. Int. J. Med. Pharm. Sci. Eng. 2012, 6, 10–15. [Google Scholar]
- Alvarez, F.; Popa, M.; Solachidis, V.; Hernandez-Penaloza, G.; Belmonte-Hernandez, A.; Asteriadis, S.; Vretos, N.; Quintana, M.; Theodoridis, T.; Dotti, D.; et al. Behavior Analysis through Multimodal Sensing for Care of Parkinson’s and Alzheimer’s Patients. IEEE Multimed. 2018, 25, 14–25. [Google Scholar] [CrossRef]
- Zirek, E.; Ersoz Huseyinsinoglu, B.; Tufekcioglu, Z.; Bilgic, B.; Hanagasi, H. Which Cognitive Dual-Task Walking Causes Most Interference on the Timed Up and Go Test in Parkinson’s Disease: A Controlled Study. Neurol. Sci. 2018, 39, 2151–2157. [Google Scholar] [CrossRef] [PubMed]
- Handojoseno, A.M.A.; Shine, J.M.; Nguyen, T.N.; Tran, Y.; Lewis, S.J.G.; Nguyen, H.T. Analysis and Prediction of the Freezing of Gait Using EEG Brain Dynamics. IEEE Trans. Neural Syst. Rehabil. Eng. 2015, 23, 887–896. [Google Scholar] [CrossRef]
- Wang, Y.; Beuving, F.; Nonnekes, J.; Cohen, M.X.; Long, X.; Aarts, R.M.; Van Wezel, R. Freezing of Gait Detection in Parkinson’s Disease via Multimodal Analysis of EEG and Accelerometer Signals. Proc. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. EMBS 2020, 2020, 847–850. [Google Scholar] [CrossRef]
- Almeida, Q.J.; Lebold, C.A. Freezing of Gait in Parkinson’s Disease: A Perceptual Cause for a Motor Impairment? J. Neurol. Neurosurg. Psychiatry 2010, 81, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Nieuwboer, A.; Dom, R.; De Weerdt, W.; Desloovere, K.; Fieuws, S.; Broens-Kaucsik, E. Abnormalities of the Spatiotemporal Characteristics of Gait at the Onset of Freezing in Parkinson’s Disease. Mov. Disord. 2001, 16, 1066–1075. [Google Scholar] [CrossRef]
- Nieuwboer, A.; Rochester, L.; Herman, T.; Vandenberghe, W.; Emil, G.E.; Thomaes, T.; Giladi, N. Reliability of the New Freezing of Gait Questionnaire: Agreement between Patients with Parkinson’s Disease and Their Carers. Gait Posture 2009, 30, 459–463. [Google Scholar] [CrossRef]
- Vertesi, A.; Lever, J.A.; Molloy, D.W.; Sanderson, B.; Tuttle, I.; Pokoradi, L.; Principi, E. Standardized Mini-Mental State Examination. Use and Interpretation. Can. Fam. Physician 2001, 47, 2018–2023. [Google Scholar]
- Cabral, A.M.; Andrade, A.O. Cheat Sheet: Sistema de Monitoramento de Distúrbios Do Movimento. Available online: http://doi.org/10.5281/zenodo.3735485 (accessed on 28 September 2021).
- López-Blanco, R.; Velasco, M.A.; Méndez-Guerrero, A.; Romero, J.P.; del Castillo, M.D.; Serrano, J.I.; Benito-León, J.; Bermejo-Pareja, F.; Rocon, E. Essential Tremor Quantification Based on the Combined Use of a Smartphone and a Smartwatch: The NetMD Study. J. Neurosci. Methods 2018, 303, 95–102. [Google Scholar] [CrossRef]
- Mancini, M.; Curtze, C.; Stuart, S.; El-Gohary, M.; McNames, J.; Nutt, J.G.; Horak, F.B. The Impact Of Freezing Of Gait On Balance Perception And Mobility In Community-Living With Parkinson’S Disease. In Proceedings of the 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Honolulu, HI, USA, 18–21 July 2018; IEEE: Piscataway, NJ, USA, 2018; pp. 3040–3043. [Google Scholar]
- Bachlin, M.; Plotnik, M.; Roggen, D.; Maidan, I.; Hausdorff, J.M.; Giladi, N.; Troster, G. Wearable Assistant for Parkinson’s Disease Patients With the Freezing of Gait Symptom. IEEE Trans. Inf. Technol. Biomed. 2010, 14, 436–446. [Google Scholar] [CrossRef]
- Vilas-Boas, M.D.C.; Cunha, J.P.S. Movement Quantification in Neurological Diseases: Methods and Applications. IEEE Rev. Biomed. Eng. 2016, 9, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Andrade, A.O.; Ferreira, L.C.V.; Rabelo, A.G.; Vieira, M.F.; Campos, A.R.; Gonçalves, B.F.; Pereira, A.A. Pelvic Movement Variability of Healthy and Unilateral Hip Joint Involvement Individuals. Biomed. Signal Process. Control 2017, 32, 10–19. [Google Scholar] [CrossRef]
- Mileti, I.; Germanotta, M.; Alcaro, S.; Pacilli, A.; Imbimbo, I.; Petracca, M.; Erra, C.; Di Sipio, E.; Aprile, I.; Rossi, S.; et al. Gait Partitioning Methods in Parkinson’s Disease Patients with Motor Fluctuations: A Comparative Analysis. In Proceedings of the 2017 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Rochester, MN, USA, 7–10 May 2017; IEEE: Piscataway, NJ, USA, 2017; pp. 402–407. [Google Scholar]
- Cando, O.A.; Hidalgo, K.R.; Palacios, B.C. A Low-Cost Vibratory Stimulus System to Mitigate Freezing of Gait in Parkinson’s Disease. In Proceedings of the 2016 IEEE ANDESCON, Arequipa, Peru, 19–21 October 2016; IEEE: Piscataway, NJ, USA, 2016; pp. 1–4. [Google Scholar]
- Meudt, S.; Bigalke, L.; Schwenker, F. Atlas—Annotation Tool Using Partially Supervised Learning and Multi-View Co-Learning in Human-Interaction Scenarios. In Proceedings of the 11th International Conference on Information Sciencees, Special Sessions, Chengdu, China, 21–23 May 2021; pp. 1309–1312. [Google Scholar]
- Rocha, P.A.; Porfírio, G.M.; Ferraz, H.B.; Trevisani, V.F.M. Effects of External Cues on Gait Parameters of Parkinson’s Disease Patients: A Systematic Review. Clin. Neurol. Neurosurg. 2014, 124, 127–134. [Google Scholar] [CrossRef] [PubMed]
| Author and Year of Publication | Sit and Stand from a Chair | Narrow Doorway | Reach a Destination | 180-Degree Turn | 360-Degree Turn | Dual Task |
|---|---|---|---|---|---|---|
| SCHAAFSMA (2003) [12] | X | X | X | X | X | |
| BARTELS (2003) [4] | X | X | X | |||
| JOVANOV (2009) [9] | X | X | ||||
| SPILDOOREN (2010) [13] | X | X | X | |||
| POPOVIC (2010) [8] | X | X | X | |||
| VELIK (2012) [19] | X | X | X | X | X | X |
| SHINE (2013) [14] | X | |||||
| BECK (2015) [15] | X | X | ||||
| HANDOJOSENO (2015) [20] | X | X | ||||
| KILLANE (2015) [21] | X | X | ||||
| WAECHTER (2015) [22] | X | X | ||||
| CANDO (2016) [23] | X | X | X | X | ||
| TARD (2016) [24] | X | |||||
| BERTOLI (2017) [1] | X | X | ||||
| MURALIDHARAN (2017) [25] | X | X | ||||
| ALVAREZ (2018) [26] | X | X | X | X | X | |
| WANG (2020) [27] | X | |||||
| PRESENT STUDY | X | X | X | X | X | X |
| V | Sex | Age (Years) | Time of PD Diagnosis (Years) | New FOG-Q | MMSE | Time OFF | Part II MDS-UPDRS | Item 2.13 of the MDS-UPDRS (Gait Freezing) | Part III MDS-UPDRS | Total MDS-UPDRS Score | TUG Score | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OFF | ON | OFF | ON | OFF | ON | |||||||||
| 1 | M | 50 | 15 | 14 | 27 | 12 h | 18 | 2 | 62 | 50 | 80 | 68 | 2 | 2 |
| 2 | F | 51 | 7 | 23 | 28 | 13 h | 16 | 2 | 49 | 39 | 65 | 55 | 2 | 2 |
| 3 | M | 57 | 15 | 19 | 15 | 13 h | 21 | 1 | 51 | 26 | 72 | 47 | 2 | 1.33 |
| 4 | F | 59 | 12 | 21 | 27 | 13 h | 10 | 1 | 20 | 7 | 30 | 17 | 2 | 2 |
| 5 | F | 63 | 10 | 23 | 26 | 13 h | 12 | 1 | 53 | 32 | 65 | 44 | 3.33 | 2 |
| 6 | F | 65 | 14 | 16 | 28 | 10 h | 6 | 1 | 35 | 37 | 41 | 43 | 2 | 2 |
| 7 | F | 66 | 25 | 15 | 24 | 13 h | 22 | 2 | 94 | 57 | 116 | 79 | 3.66 | 2 |
| 8 | M | 68 | 6 | 23 | 28 | 13 h | 9 | 1 | 49 | 37 | 58 | 46 | 2.66 | 1 |
| 9 | M | 73 | 6 | 26 | 27 | 12 h 30 | 44 | 3 | 115 | 100 | 159 | 144 | 4 | 4 |
| 10 | F | 56 | 15 | 12 | 25 | 13 h 30 | 13 | 1 | 47 | 32 | 60 | 45 | 2 | 2 |
| v | TUG | |||||||
|---|---|---|---|---|---|---|---|---|
| OFF | ON | |||||||
| TUG1 | TUG2 | TUG3 | TOTAL | TUG1 | TUG2 | TUG3 | TOTAL | |
| 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 4 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 5 | 2 | 1 | 0 | 3 | 0 | 0 | 1 | 1 |
| 6 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 7 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 0 |
| 8 | 0 | 3 | 4 | 7 | 0 | 0 | 0 | 0 |
| 9 | 2 | 3 | 2 | 7 | 1 | 2 | 2 | 5 |
| 10 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Total number of FOG—TUG OFF | 18 | Total number of FOG—TUG ON | 6 | |||||
| v | MT | |||||||
|---|---|---|---|---|---|---|---|---|
| OFF | ON | |||||||
| MT1 | MT2 | MT3 | TOTAL | MT1 | MT2 | MT3 | TOTAL | |
| 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 3 | 7 | 9 | 7 | 23 | 0 | 0 | 0 | 0 |
| 4 | 2 | 3 | 3 | 8 | 0 | 0 | 0 | 0 |
| 5 | 2 | 3 | 1 | 6 | 1 | 0 | 0 | 1 |
| 6 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 0 |
| 7 | 1 | 2 | 0 | 3 | 0 | 0 | 0 | 0 |
| 8 | 2 | 1 | 1 | 4 | 0 | 0 | 0 | 0 |
| 9 | 1 | 5 | 1 | 7 | 2 | 2 | 2 | 6 |
| 10 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 0 |
| Total number of FOG—MT OFF | 53 | Total number of FOG—MT ON | 7 | |||||
| v | DT | |||||||
|---|---|---|---|---|---|---|---|---|
| OFF | ON | |||||||
| DT1 | DT2 | DT3 | TOTAL | DT1 | DT2 | DT3 | TOTAL | |
| 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 2 | 2 | 0 | 1 | 3 | 0 | 0 | 0 | 0 |
| 3 | 3 | 6 | 7 | 16 | 0 | 0 | 0 | 0 |
| 4 | 5 | 5 | 3 | 13 | 0 | 0 | 0 | 0 |
| 5 | 2 | 1 | 0 | 3 | 0 | 0 | 2 | 2 |
| 6 | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 0 |
| 7 | 0 | 2 | 0 | 2 | 0 | 0 | 0 | 0 |
| 8 | 4 | 4 | 2 | 10 | 0 | 0 | 0 | 0 |
| 9 | 1 | 2 | 3 | 6 | 1 | 2 | 1 | 4 |
| 10 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Total number of FOG—DT OFF | 54 | Total number of FOG—DT ON | 6 | |||||
| V | NFOG-Q | OFF | ON | ||
|---|---|---|---|---|---|
| Number of FOG | TF (s) | Number of FOG | TF (s) | ||
| 1 | 14 | 0 | 0 | 0 | 0 |
| 2 | 23 | 3 | 4.498 | 0 | 0 |
| 3 | 19 | 39 | 179.735 | 0 | 0 |
| 4 | 21 | 21 | 691.521 | 0 | 0 |
| 5 | 23 | 9 | 27.448 | 3 | 4.922 |
| 6 | 16 | 2 | 6.273 | 0 | 0 |
| 7 | 15 | 5 | 12.918 | 0 | 0 |
| 8 | 23 | 14 | 571.886 | 0 | 0 |
| 9 | 26 | 13 | 88.480 | 10 | 64.821 |
| 10 | 12 | 1 | 1.769 | 0 | 0 |
| Total | 107 | 1584.528 | 13 | 69.743 | |
| Author | N | Number of Freezers | Number of FOG Events | Recording Time | Number of Trials |
|---|---|---|---|---|---|
| Bartels 2003 [4] | 19 | --- | 237 | --- | 57 |
| Popovic 2010 [8] | 9 | --- | 24 | --- | 18 |
| Beck 2015 [15] | 20 | 4 | 23 | --- | 240 |
| Handojoseno 2015 [20] | 16 | --- | 404 | 5 h 30 min | --- |
| Cando 2016 [23] | 5 | 4 | 11 | --- | 10 |
| Alvarez 2018 [26] | 18 | --- | 200 | 8 h 20 min | 700 |
| Wang 2020 [27] | 15 | 15 | 700 | 22 h 30 min | 225 |
| Present study | 10 | 9 | 107 | --- | 60 |
| Patient | Age | Date | Walking Aid | New FOG-Q | State | TFOG | Number of FOG Events |
|---|---|---|---|---|---|---|---|
| 1 | 57 | 1 March | --- | 12 | OFF | 6 s | 2 |
| ON | 0 s | 0 | |||||
| 1 May | --- | 12 | OFF | 30 s | 6 | ||
| ON | 0 s | 0 | |||||
| 1 July | --- | 13 | OFF | 8 s | 3 | ||
| ON | 0 s | 0 | |||||
| 2 | 63 | 10 April | cane | 19 | OFF | 27 s | 4 |
| ON | 2 s | 1 | |||||
| 10 July | cane | 20 | OFF | 32 s | 6 | ||
| ON | 0 s | 0 | |||||
| 10 October | cane | 20 | OFF | 30 s | 8 | ||
| ON | 0 s | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nóbrega, L.R.; Rocon, E.; Pereira, A.A.; Andrade, A.d.O. A Novel Physical Mobility Task to Assess Freezers in Parkinson’s Disease. Healthcare 2023, 11, 409. https://doi.org/10.3390/healthcare11030409
Nóbrega LR, Rocon E, Pereira AA, Andrade AdO. A Novel Physical Mobility Task to Assess Freezers in Parkinson’s Disease. Healthcare. 2023; 11(3):409. https://doi.org/10.3390/healthcare11030409
Chicago/Turabian StyleNóbrega, Lígia Reis, Eduardo Rocon, Adriano Alves Pereira, and Adriano de Oliveira Andrade. 2023. "A Novel Physical Mobility Task to Assess Freezers in Parkinson’s Disease" Healthcare 11, no. 3: 409. https://doi.org/10.3390/healthcare11030409
APA StyleNóbrega, L. R., Rocon, E., Pereira, A. A., & Andrade, A. d. O. (2023). A Novel Physical Mobility Task to Assess Freezers in Parkinson’s Disease. Healthcare, 11(3), 409. https://doi.org/10.3390/healthcare11030409