Does Lower-Limb Tendon Structure Influence Walking Gait?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Procedures
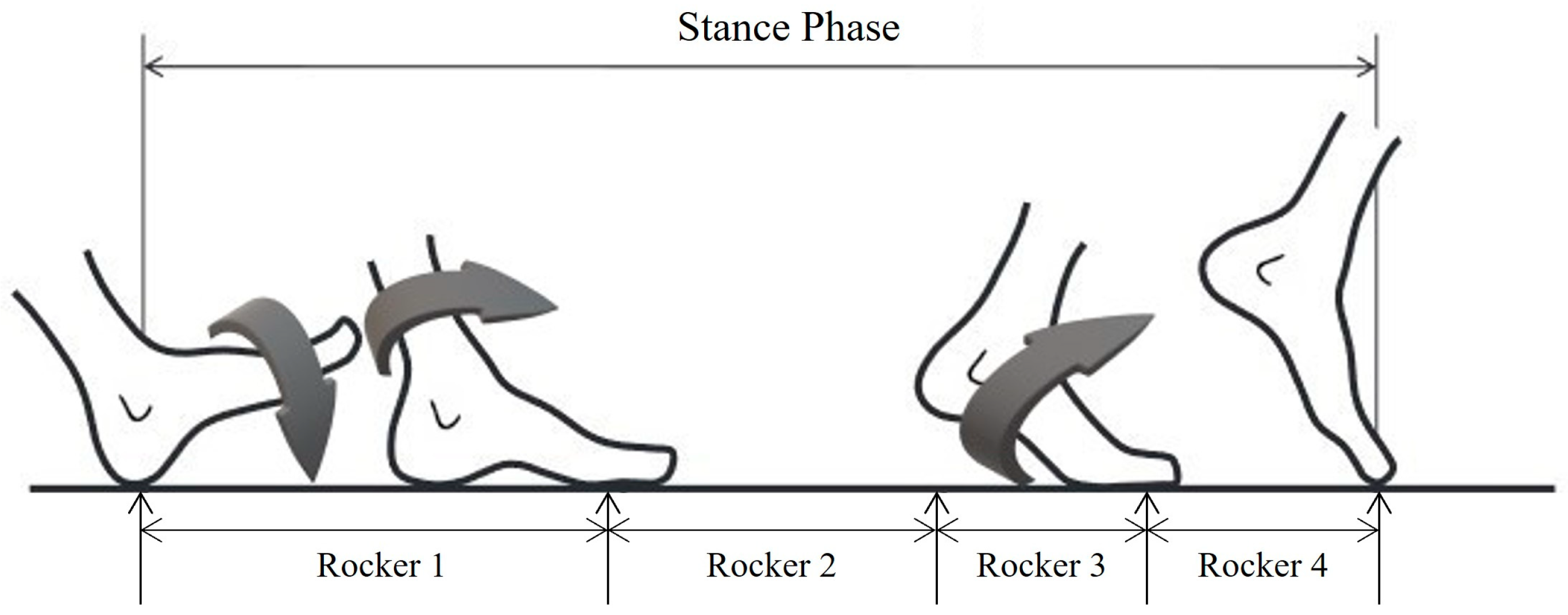
2.3. Material and Testing
2.4. Tendon Morphology Characteristics
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kannus, P. Structure of the tendon connective tissue. Scand. J. Med. Sci. Sports 2000, 10, 312–320. [Google Scholar] [CrossRef] [PubMed]
- McMahon, T.A.; Cheng, G.C. The mechanics of running: How does stiffness couple with speed? J. Biomech. 1990, 23 (Suppl. S1), 65–78. [Google Scholar] [CrossRef] [PubMed]
- Gruber, M.; Kramer, A.; Mulder, E.; Rittweger, J. The Importance of Impact Loading and the Stretch Shortening Cycle for Spaceflight Countermeasures. Front. Physiol. 2019, 10, 311. [Google Scholar] [CrossRef]
- Wang, J.H.; Guo, Q.; Li, B. Tendon biomechanics and mechanobiology—A minireview of basic concepts and recent advancements. J. Hand Ther. Off. J. Am. Soc. Hand Ther. 2012, 25, 133–140; quiz 141. [Google Scholar] [CrossRef]
- Shaw, H.M.; Vazquez, O.T.; McGonagle, D.; Bydder, G.; Santer, R.M.; Benjamin, M. Development of the human Achilles tendon enthesis organ. J. Anat. 2008, 213, 718–724. [Google Scholar] [CrossRef] [PubMed]
- Kubo, K.; Miyazaki, D.; Ikebukuro, T.; Yata, H.; Okada, M.; Tsunoda, N. Active muscle and tendon stiffness of plantar flexors in sprinters. J. Sports Sci. 2017, 35, 742–748. [Google Scholar] [CrossRef] [PubMed]
- Del Baño-Aledo, M.E.; Martínez-Payá, J.J.; Ríos-Díaz, J.; Mejías-Suárez, S.; Serrano-Carmona, S.; de Groot-Ferrando, A. Ultrasound measures of tendon thickness: Intra-rater, Inter-rater and Inter-machine reliability. Muscles Ligaments Tendons J. 2017, 7, 192. [Google Scholar] [CrossRef]
- Whittle, M.W. Gait Analysis: An Introduction; Butterworth-Heinemann: Oxford, UK, 2014. [Google Scholar]
- Perry, J.; Burnfield, J.M. Gait Analysis. Normal and Pathological Function, 2nd ed.; SLACK: San Francisco, CA, USA, 2010. [Google Scholar]
- Shah, K.; Solan, M.; Dawe, E. The gait cycle and its variations with disease and injury. Orthop. Trauma 2020, 34, 153–160. [Google Scholar] [CrossRef]
- Szczepanowska-Wolowiec, B.; Sztandera, P.; Kotela, I.; Zak, M. Assessment of the Foot’s Longitudinal Arch by Different Indicators and Their Correlation with the Foot Loading Paradigm in School-Aged Children: A Cross Sectional Study. Int. J. Environ. Res. Public Health 2021, 18, 5196. [Google Scholar] [CrossRef]
- Szczepanowska-Wolowiec, B.; Sztandera, P.; Kotela, I.; Zak, M. Feet deformities and their close association with postural stability deficits in children aged 10–15 years. BMC Musculoskelet. Disord. 2019, 20, 537. [Google Scholar] [CrossRef]
- Forczek, W.; Maslon, A.; Fraczek, B.; Curylo, M.; Salamaga, M.; Suder, A. Does the first trimester of pregnancy induce alterations in the walking pattern? PLoS ONE 2019, 14, e0209766. [Google Scholar] [CrossRef]
- Wrobel, J.S.; Najafi, B. Diabetic foot biomechanics and gait dysfunction. J. Diabetes Sci. Technol. 2010, 4, 833–845. [Google Scholar] [CrossRef]
- Giacomozzi, C.; D’Ambrogi, E.; Uccioli, L.; Macellari, V. Does the thickening of Achilles tendon and plantar fascia contribute to the alteration of diabetic foot loading? Clin. Biomech. 2005, 20, 532–539. [Google Scholar] [CrossRef]
- Cen, X.; Song, Y.; Yu, P.; Sun, D.; Simon, J.; Biro, I.; Gu, Y. Effects of plantar fascia stiffness on the internal mechanics of idiopathic pes cavus by finite element analysis: Implications for metatarsalgia. Comput. Methods Biomech. Biomed. Eng. 2023, 1–9. [Google Scholar] [CrossRef]
- Sun, D.; Song, Y.; Cen, X.; Wang, M.; Baker, J.S.; Gu, Y. Workflow assessing the effect of Achilles tendon rupture on gait function and metatarsal stress: Combined musculoskeletal modeling and finite element analysis. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2022, 236, 676–685. [Google Scholar] [CrossRef]
- Gomez Bernal, A.; Becerro-de-Bengoa-Vallejo, R.; Losa-Iglesias, M.E. Reliability of the OptoGait portable photoelectric cell system for the quantification of spatial-temporal parameters of gait in young adults. Gait Posture 2016, 50, 196–200. [Google Scholar] [CrossRef]
- Núñez-Trull, A.; Alvarez-Medina, J.; Jaén-Carrillo, D.; Roche-Seruendo, L.E.; Gómez-Trullén, E. Absolute agreement and consistency of the OptoGait system and Freemed platform for measuring walking gait. Med. Eng. Phys. 2022, 110, 103912. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.M.; Zifchock, R.A.; Hillstrom, H.J. The effects of limb dominance and fatigue on running biomechanics. Gait Posture 2014, 39, 915–919. [Google Scholar] [CrossRef] [PubMed]
- Mc Auliffe, S.; Mc Creesh, K.; Purtill, H.; O’Sullivan, K. A systematic review of the reliability of diagnostic ultrasound imaging in measuring tendon size: Is the error clinically acceptable? Phys. Ther. Sport Off. J. Assoc. Chart. Physiother. Sports Med. 2017, 26, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Peirotén, A.; Garcia-Pinillos, F.; Jaen-Carrillo, D.; Carton-Llorente, A.; Roche-Seruendo, L.E. Is There a Relationship between the Morphology of Connective Tissue and Reactivity during a Drop Jump? Influence of Sex and Athletic Performance Level. Int. J. Environ. Res. Public Health 2021, 18, 1969. [Google Scholar] [CrossRef] [PubMed]
- Kernozek, T.W.; Knaus, A.; Rademaker, T.; Almonroeder, T.G. The effects of habitual foot strike patterns on Achilles tendon loading in female runners. Gait Posture 2018, 66, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Routledge: London, UK, 2013. [Google Scholar]
- Mayich, D.J.; Novak, A.; Vena, D.; Daniels, T.R.; Brodsky, J.W. Gait analysis in orthopedic foot and ankle surgery—Topical review, part 1: Principles and uses of gait analysis. Foot Ankle Int. 2014, 35, 80–90. [Google Scholar] [CrossRef] [PubMed]
- Komi, P.V. Stretch-shortening cycle: A powerful model to study normal and fatigued muscle. J. Biomech. 2000, 33, 1197–1206. [Google Scholar] [CrossRef] [PubMed]
- Petrofsky, J.; Lee, S.; Bweir, S. Gait characteristics in people with type 2 diabetes mellitus. Eur. J. Appl. Physiol. 2005, 93, 640–647. [Google Scholar] [CrossRef]
- Rubio-Peirotén, A.G.-P.F.; Cartón-Llorente, A.; Jaén-Carrillo, D.; Abat, F.; Roche-Seruendo, L.E. Lower-Limb Connective Tissue Morphologic Characteristics in Runners. How Do They Relate with Running Biomechanics and Tendon Pathology? A Systematic Review. Muscles Ligaments Tendons J. 2022, 12, 331–351. [Google Scholar] [CrossRef]
| Variable | |
|---|---|
| Age (years) | 28.97 (7.7) |
| Height (m) | 176.40 (6.3) |
| Body mass (kg) | 71.66 (7.8) |
| BMI (Kg·m−2) | 23.02 |
| PT-Thickness (mm) PT-CSA (mm2) | 3.73 (0.7) 94.99 (22.8) |
| AT-Thickness (mm) AT-CSA (mm2) | 5.19 (0.6) 53.68 (8.6) |
| PF-Thickness (mm) | 2.66 (0.4) |
| HG (n = 22) | LG (n = 17) | p-Value ^ | ES (d) | |
|---|---|---|---|---|
| R1 (ms) | 190.77 ± 51.94 | 156.82 ± 42.01 | 0.035 * | 0.718 |
| %R1 (% gait cycle) | 31.13 ± 9.11 | 25.54 ± 6.59 | 0.040 * | 0.703 |
| R2 (ms) | 107.5 ± 51.75 | 110.59 ± 50.45 | 0.853 | 0.060 |
| %R2 (% gait cycle) | 17.44 ± 8.41 | 17.87 ± 8.12 | 0.875 | 0.052 |
| R3 (ms) | 300.82 ± 64.14 | 321.76 ± 50.04 | 0.274 | 0.364 |
| %R3 (% gait cycle) | 48.50 ± 8.68 | 52.56 ± 8.8 | 0.158 | 0.464 |
| R4 (ms) | 18.55 ± 16.63 | 24.76 ± 14.34 | 0.227 | 0.135 |
| %R4 (% gait cycle) | 2.93 ± 2.6 | 2.6 ± 2.3 | 0.179 | 0.134 |
| HG (n = 20) | LG (n = 19) | p-Value ^ | ES (d) | |
|---|---|---|---|---|
| R1 (ms) | 190.40 ± 52.46 | 160.79 ± 44.16 | 0.065 | 0.610 |
| %R1 (% gait cycle) | 30.98 ± 9 | 26.29 ± 7.39 | 0.084 | 0.569 |
| R2 (ms) | 105.55 ± 35.28 | 112.32 ± 63.67 | 0.682 | 0.131 |
| %R2 (% gait cycle) | 16.9 ± 5.65 | 18.3 ± 10.32 | 0.626 | 0.168 |
| R3 (ms) | 301.45 ± 58.62 | 318.89 ± 58.88 | 0.360 | 0.296 |
| %R3 (% gait cycle) | 48.63 ± 8.60 | 52 ± 9 | 0.239 | 0.382 |
| R4 (ms) | 21.65 ± 17.92 | 20.84 ± 13.65 | 0.876 | 0.050 |
| %R4 (% gait cycle) | 3.4 ± 2.7 | 3.4 ± 2.3 | 0.984 | 0.000 |
| HG (n = 18) | LG (n = 21) | p-Value ^ | ES (d) | |
|---|---|---|---|---|
| R1 (ms) | 169.72 ± 45.75 | 181.33 ± 54.33 | 0.479 | 0.231 |
| %R1 (% gait cycle) | 27.44 ± 8.1 | 29.77 ± 8.89 | 0.401 | 0.273 |
| R2 (ms) | 121.5 ± 49.45 | 98 ± 50.1 | 0.150 | 0.472 |
| %R2 (% gait cycle) | 19.54 ± 7.92 | 15.99 ± 8.22 | 0.180 | 0.439 |
| R3 (ms) | 312.72 ± 59.37 | 307.57 ± 59.36 | 0.789 | 0.086 |
| %R3 (% gait cycle) | 50.06 ± 7.86 | 50.46 ± 9.8 | 0.891 | 0.045 |
| R4 (ms) | 18.72 ± 14.16 | 23.43 ± 16.76 | 0.360 | 0.303 |
| %R4 (% gait cycle) | 2.96 ± 2.29 | 3.78 ± 2.67 | 0.312 | 0.329 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Núñez-Trull, A.; Álvarez-Medina, J.; Jaén-Carrillo, D.; Rubio-Peirotén, A.; Abat, F.; Roche-Seruendo, L.E.; Gómez-Trullén, E.M. Does Lower-Limb Tendon Structure Influence Walking Gait? Healthcare 2023, 11, 3142. https://doi.org/10.3390/healthcare11243142
Núñez-Trull A, Álvarez-Medina J, Jaén-Carrillo D, Rubio-Peirotén A, Abat F, Roche-Seruendo LE, Gómez-Trullén EM. Does Lower-Limb Tendon Structure Influence Walking Gait? Healthcare. 2023; 11(24):3142. https://doi.org/10.3390/healthcare11243142
Chicago/Turabian StyleNúñez-Trull, Alejandro, Javier Álvarez-Medina, Diego Jaén-Carrillo, Alberto Rubio-Peirotén, Ferrán Abat, Luis E. Roche-Seruendo, and Eva M. Gómez-Trullén. 2023. "Does Lower-Limb Tendon Structure Influence Walking Gait?" Healthcare 11, no. 24: 3142. https://doi.org/10.3390/healthcare11243142
APA StyleNúñez-Trull, A., Álvarez-Medina, J., Jaén-Carrillo, D., Rubio-Peirotén, A., Abat, F., Roche-Seruendo, L. E., & Gómez-Trullén, E. M. (2023). Does Lower-Limb Tendon Structure Influence Walking Gait? Healthcare, 11(24), 3142. https://doi.org/10.3390/healthcare11243142











