Music Therapy Enhances Episodic Memory in Alzheimer’s and Mixed Dementia: A Double-Blind Randomized Controlled Trial
Abstract
1. Introduction
The Dual Retrieval Model of Episodic Memory
2. Materials and Methods
2.1. Design
2.2. Participants
2.3. Instruments
2.3.1. Tests for Participants’ Characterization
2.3.2. Primary Outcome Tests (Pre-Test and Post-Test)
2.3.3. Secondary Outcome Tests (Pre-Test and Post-Test)
2.4. Procedures
2.4.1. Music Therapy Intervention
- A.
- Intervention Theory
- B.
- Intervention Content
| Session | Theme | Activities | Objectives |
|---|---|---|---|
| 1 | Welcome |
|
|
| 2 | Introduction to patients |
|
|
| 3 | Identity |
|
|
| 4 | Musical memory |
|
|
| 5 | Musical memory |
|
|
| 6 | Musical memory |
|
|
| 7 | Music in everyday life |
|
|
| 8 | Music in everyday life |
|
|
| 9 | Training memory and attention |
|
|
| 10 | Playing instruments |
|
|
| 11 | Playing instruments |
|
|
| 12 | Farewell |
|
|
2.4.2. Data Analysis
3. Results
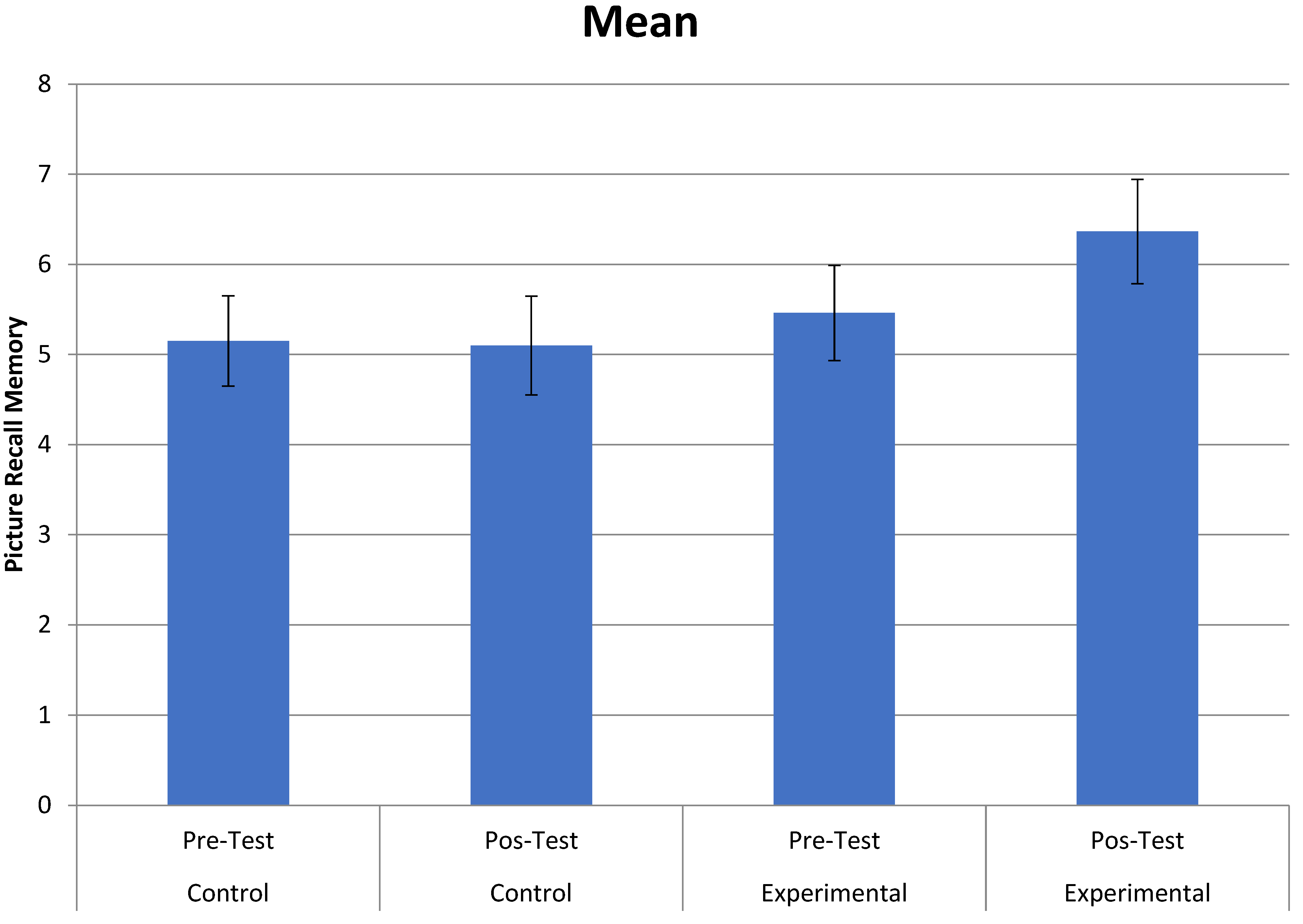
3.1. Primary Outcomes (Effects of Music Therapy on Memory)
3.2. Secondary Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Department of Economic and Social Affairs; United Nations. World Population Prospects 2019: Highlights. 2019. Available online: https://population.un.org/wpp/Publications/Files/WPP2019_10KeyFindings.pdf (accessed on 20 October 2022).
- Lam, H.L.; Li, W.T.V.; Laher, I.; Wong, R.Y. Effects of Music Therapy on Patients with Dementia—A Systematic Review. Geriatrics 2020, 5, 62. [Google Scholar] [CrossRef] [PubMed]
- van der Steen, J.T.; van Soest-Poortvliet, M.C.; van der Wouden, J.C.; Bruinsma, M.S.; Scholten, R.J.; Vink, A.C. Music-based therapeutic interventions for people with dementia. Cochrane Database Syst. Rev. 2017, 5, CD003477. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, C.E.L.; Nitrini, R.; Brucki, S.M.D. Indirect costs with dementia: A Brazilian study. Dement. Neuropsychol. 2015, 9, 42–50. [Google Scholar] [CrossRef]
- Arroyo-Anlló, E.M.; Díaz, J.P.; Gil, R. Familiar Music as an Enhancer of Self-Consciousness in Patients with Alzheimer’s Disease. Biomed. Res. Int. 2013, 2013, 752965. [Google Scholar] [CrossRef]
- Ballard, C.; Brown, R.; Fossey, J.; Douglas, S.; Bradley, P.; Hancock, J.; Howard, R. Brief psychosocial therapy for the treatment of agitation in Alzheimer disease (the CALM-AD trial). Am. J. Geriatr. Psychiatry 2009, 17, 726–733. [Google Scholar] [CrossRef]
- Delphin-Combe, F.; Rouch, I.; Martin-Gaujard, G.; Relland, S.; Krolak-Salmon, P. Effect of a non-pharmacological intervention, on behavior disturbances in Alzheimer disease and associated disorders. Gériatrie Psychol. Neuropsychiatr. Vieil. 2013, 11, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Fusar-Poli, L.; Bieleninik, L.; Brondino, N.; Chen, X.J.; Gold, C. The effect of music therapy on cognitive functions in patients with dementia: A systematic review and meta-analysis. Aging Ment. Health 2018, 22, 1097–1106. [Google Scholar] [CrossRef]
- Guétin, S.; Portet, F.; Picot, M.C.; Pommié, C.; Messaoudi, M.; Djabelkir, L.; Touchon, J. Effect of music therapy on anxiety and depression in patients with Alzheimer’s type dementia: Randomised, controlled study. Dement. Geriatr. Cogn. Disord. 2009, 28, 36–46. [Google Scholar] [CrossRef]
- Hailstone, J.C.; Rohani, O.; Jason, D.W. Relatively preserved knowledge of music in semantic dementia. J. Neurol. Neurosurg. Psychiatry 2009, 80, 808–809. [Google Scholar] [CrossRef] [PubMed]
- Holmes, C.; Knights, A.; Dean, C.; Hodkinson, S.; Hopkins, V. Keep music live: Music and the alleviation of apathy in dementia subjects. Int. Psychogeriatr. 2006, 18, 623–630. [Google Scholar] [CrossRef]
- Hsu, M.H.; Flowerdew, R.; Parker, M.; Fachner, J.; Odell-Miller, H. Individual music therapy for managing neuropsychiatric symptoms for people with dementia and their carers: A cluster randomised controlled feasibility study. BMC Geriatr. 2015, 15, 84. [Google Scholar] [CrossRef]
- Massimi, M.; Berry, E.; Browne, G.; Smyth, G.; Watson, P.; Baecker, R.M. An exploratory case study of the impact of ambient biographical displays on identity in a patient with Alzheimer’s disease. Neuropsychol. Rehabil. 2008, 18, 742–765. [Google Scholar] [CrossRef]
- Perrot, C.; Ligonnet, F.; Gire, P.; Auguste, N.; Fabre, F.; Gonthier, R. Traitement non médicamenteux de l’angoisse dans le cadre de syndromes démentiels: Aspects psychiques de l’utilisation de la musique dans une unité de neuropsychogériatrie./Non-drug treatment of anxiety in the context of dementiasyndromes: Psychological aspects of the use of music in a neuropsychogeriatric unit. Neurol. Psychiatr. Geriatr. 2014, 14, 334–342. [Google Scholar] [CrossRef]
- Hanser, S.B. The Effectiveness of Music-based Interventions for Dementia: An Umbrella Review. Music. Med. 2021, 13, 156–161. [Google Scholar] [CrossRef]
- Moreno-Morales, C.; Calero, R.; Moreno-Morales, P.; Pintado, C. Music Therapy in the Treatment of Dementia: A Systematic Review and Meta-Analysis. Front. Med. 2020, 7, 160. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Romero, M.; Jiménez-Palomares, M.; Rodríguez-Mansilla, J.; Flores-Nieto, A.; Garrido-Ardila, E.M.; González López-Arza, M.V. Benefits of music therapy on behaviour disorders in subjects diagnosed with dementia: A systematic review. Neurologia 2017, 32, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Benz, S.; Sellaro, R.; Hommel, B.; Colzato, L.S. Music Makes the World Go Round: The Impact of Musical Training on Non-musical Cognitive Functions. Front. Psychol. 2016, 6, 20–23. [Google Scholar] [CrossRef] [PubMed]
- Deason, R.G.; Strong, J.V.; Tat, M.J.; Simmons-Stern, N.R.; Budson, A.E. Explicit and implicit memory for music in healthy older adults and patients with mild Alzheimer’s disease. J. Clin. Exp. Neuropsychol. 2019, 41, 158–169. [Google Scholar] [CrossRef]
- Gómez, G.M.; Gómez, G.J. Music therapy and Alzheimer’s disease: Cognitive, psychological, and behavioural effects. Neurologia 2017, 32, 300–308. [Google Scholar] [CrossRef]
- Goudour, A.; Samson, S.; Bakchine, S.; Ehrlé, N. Semantic memory training in Alzheimer’s disease. Geriatr. Psychol. Neuropsychiatr. Vieil. 2011, 9, 237–247. [Google Scholar] [CrossRef]
- Ménard, M.C.; Belleville, S. Musical and verbal memory in Alzheimer’s disease: A study of long-term and short-term memory. Brain Cogn. 2009, 71, 38–45. [Google Scholar] [CrossRef]
- Moreira, S.V.; Justi, F.R.d.R.; Moreira, M. Can musical intervention improve memory in Alzheimer’s patients? Evidence from a systematic review. Dement. Neuropsychol. 2018, 12, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Ragot, R.; Ferrandez, A.M.; Pouthas, V. Time, Music, and Aging. Psychomusicology 2002, 18, 18–45. [Google Scholar] [CrossRef]
- Simmons-Stern, N.R.; Deason, R.G.; Brandler, B.J.; Frustace, B.S.; O’Connor, M.K.; Ally, B.A.; Budson, A.E. Music-based memory enhancement in Alzheimer’s disease: Promise and limitations. Neuropsychologia 2012, 50, 3295–3303. [Google Scholar] [CrossRef]
- Slattery, C.F.; Agustus, J.L.; Paterson, R.W.; McCallion, O.; Foulkes, A.J.; Macpherson, K.; Mummery, C.J. The functional neuroanatomy of musical memory in Alzheimer’s disease. Cortex 2019, 115, 357–370. [Google Scholar] [CrossRef]
- Vanstone, A.D.; Cuddy, L.L. Musical Memory in Alzheimer Disease. Aging Neuropsychol. Cogn. 2009, 17, 108–128. [Google Scholar] [CrossRef]
- Han, J.W.; Lee, H.; Hong, J.W.; Kim, K.; Kim, T.; Byun, H.J.; Kim, K.W. Multimodal cognitive enhancement therapy for patients with mild cognitive impairment and mild dementia: A multi-center, randomized, controlled, double-blind, crossover trial. J. Alzheimer’s Dis. 2017, 55, 787–796. [Google Scholar] [CrossRef]
- Baird, A.; Samson, S. Music and dementia. Prog. Brain Res. 2015, 217, 207–235. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, J.H.; Stelzer, J.; Fritz, T.H.; Chételat, G.; La Joie, R.; Turner, R. Why musical memory can be preserved in advanced Alzheimer’s disease. Brain 2015, 138 Pt 8, 2438–2450. [Google Scholar] [CrossRef] [PubMed]
- Thaut, M.H.; Hoemberg, V. Handbook of Neurologic Music Therapy; Oxford University Press: New York, NY, USA, 2014. [Google Scholar]
- Peck, K.J.; Girard, T.A.; Russo, F.A.; Fiocco, A.J. Music and Memory in Alzheimer’s Disease and The Potential Underlying Mechanisms. J. Alzheimer’s Dis. 2016, 51, 949–959. [Google Scholar] [CrossRef] [PubMed]
- Lord, T.R.; Garner, J.E. Effects of music on Alzheimer patients. Percept. Mot. Skills 1993, 76, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Särkämö, T.; Tervaniemi, M.; Laitinen, S.; Numminen, A.; Kurki, M.; Johnson, J.K.; Rantanen, P. Cognitive, Emotional, and Social Benefits of Regular Musical Activities in Early Dementia: Randomized Controlled Study. Gerontologist 2014, 54, 634–650. [Google Scholar] [CrossRef]
- Prickett, C.A.; Moore, R.S. The Use of Music to Aid Memory of Alzheimer’s Patients. J. Music Ther. 1991, 28, 101–110. [Google Scholar] [CrossRef]
- Mercadall-Brotons, M.; Tomaino, C.M.; Silva, T.R.A.; Moreira, S.V. Music therapy & music based interventions in dementia: Recommendations for clinical guidelines Part II. Music. Med. 2021, 13, 169–173. [Google Scholar] [CrossRef]
- Simmons-Stern, N.R.; Budson, A.E.; Ally, B.A. Music as a memory enhancer in patients with Alzheimer’s disease. Neuropsychologia 2010, 48, 3164–3167. [Google Scholar] [CrossRef]
- Brainerd, C.J.; Reyna, V.F.; Howe, M.L. Trichotomous processes in early memory development, aging, and neurocognitive impairment: A unified theory. Psychol. Rev. 2009, 116, 783–832. [Google Scholar] [CrossRef]
- Kristensen, C.H.; de Azevedo Gomes, C.F.; Justo, A.R.; Vieira, K. Normas brasileiras para o Affective Norms for English Words. Trends Psychiatry Psychother. 2011, 33, 135–146. [Google Scholar] [CrossRef]
- Janczura, G.A.; Castilho, G.M.; Rocha, N.O.; van Erven, T.J.C.; Huang, T.P. Normas de concretude para 909 palavras da língua portuguesa. Psicol. Teor. Pesqui. 2007, 23, 195–204. [Google Scholar] [CrossRef]
- Brainerd, C.J.; Reyna, V.F.; Gomes, C.F.A.; Kenney, A.E.; Gross, C.J.; Spreng, R.N. Dual-retrieval models and neurocognitive impairment. J. Exp. Psychol. Learn. Mem. Cogn. 2014, 40, 41–65. [Google Scholar] [CrossRef]
- Jacoby, L. A processes dissociation process: Separating automatic from intentional uses of memory. J. Mem. Lang. 1991, 30, 513–541. [Google Scholar] [CrossRef]
- Mandler, G. Familiarity Breeds Attempts: A Critical Review of Dual-Process Theories of Recognition. Perspect. Psychol. Sci. 2008, 3, 390–399. [Google Scholar] [CrossRef] [PubMed]
- Rummel, J. Current Issues in Memory; Routledge: London, UK, 2021. [Google Scholar]
- Yonelinas, A.P. The Nature of Recollection and Familiarity: A Review of 30 Years of Research. J. Mem. Lang. 2002, 46, 441–517. [Google Scholar] [CrossRef]
- Brainerd, C.J.; Reyna, V.F. Fuzzy-trace theory and memory development. Dev. Rev. 2004, 24, 396–439. [Google Scholar] [CrossRef]
- Gomes, C.F.A.; Brainerd, C.J.; Nakamura, K.; Reyna, V.F. Markovian interpretations of dual retrieval processes. J. Math. Psychol. 2014, 59, 50–64. [Google Scholar] [CrossRef] [PubMed]
- Jacoby, L.L.; Dallas, M. On the relationship between autobiographical memory and perceptual learning. J. Exp. Psychol. Gen. 1981, 110, 306–340. [Google Scholar] [CrossRef]
- Chaves, M.L.F.; Godinho, C.C.; Porto, C.S.; Mansur, L.; Carthery-Goulart, M.T.; Yassuda, M.S.; Beato, R. Doença de Alzheimer. Avaliação cognitiva, comportamental e funcional. Dement. Neuropsychol. 2011, 5 (Suppl. S1), 21–33. [Google Scholar]
- Justi, F.R.R.; Justi, C.N.G. As estatísticas de vizinhança ortográfica do português e do inglês são diferentes? Psicol. Pesqui. 2008, 2, 61–73. [Google Scholar]
- Pergher, G.K.; Stein, L.M. Recuperando memórias autobiográficas: Avaliação da versão brasileira do Teste de Memória Autobiográfica. Psico 2008, 39, 299–307. [Google Scholar]
- de Melo, D.M.; Barbosa, A.J.G. O uso do Miniexame do Estado Mental em pesquisas com idosos no Brasil: Uma revisão sistemática. Ciênc. Saúde Coletiva 2015, 20, 3865–3876. [Google Scholar] [CrossRef]
- Atalaia-Silva, K.C.; Lourenço, R.A. Tradução, adaptação e validação de construto do Teste do Relógio aplicado entre idosos no Brasil. Rev. Saude Publica 2008, 42, 930–937. [Google Scholar] [CrossRef]
- Fuzikawa, C.S.; Uchoa, E.; Lima-Costa, M.F. Teste do relógio: Uma revisão da literatura sobre este teste para rastreamento de déficit cognitivo. J. Bras. Psiquiatr. 2003, 52, 223–235. [Google Scholar]
- Paradela, E.M.P.; Lourenço, R.A.; Veras, R.P. Validação da escala de depressão geriátrica em um ambulatório geral. Rev. Saude Publica 2005, 39, 918–923. [Google Scholar] [CrossRef]
- Monteiro, E.A.; Mazin, S.C.; Dantas, R.A.S. Questionário de avaliação da sobrecarga do cuidador informal: Validação para o Brasil. Rev. Bras. Enferm. 2015, 68, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Robb, S.L.; Carpenter, J.S.; Burns, D.S. Reporting guidelines for music-based interventions. J. Health Psychol. 2011, 16, 342–352. [Google Scholar] [CrossRef]
- Brainerd, C.J.; Aydin, C.; Reyna, V.F. Development of dual-retrieval processes in recall: Learning, forgetting, and reminiscence. J. Mem. Lang. 2012, 66, 763–788. [Google Scholar] [CrossRef] [PubMed]
- Gomes, C.F.A.; Brainerd, C.J.; Stein, L.M. Effects of emotional valence and arousal on recollective and nonrecollective recall. J. Exp. Psychol. Learn. Mem. Cogn. 2013, 39, 663–677. [Google Scholar] [CrossRef]
- Fichman-Charchat, H.; Miranda, C.V.; Fernandes, C.S.; Mograbi, D.; Oliveira, R.M.; Novaes, R.; Aguiar, D. Brief Cognitive Screening Battery (BCSB) is a very useful tool for diagnosis of probable mild Alzheimer’s disease in a geriatric clinic. Arq. Neuropsiquiatr. 2016, 74, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Nitrini, R.; Caramelli, P.; Herrera, E., Jr.; Porto, C.S.; Charchat-Fichman, H.; Carthery, M.T.; Takada, L.T.; Lima, E.P. Performance of illiterate and literate nondemented elderly subjects in two tests of long-term memory. J. Int. Neuropsychol. Soc. 2004, 10, 634–638. [Google Scholar] [CrossRef]
- Palisson, J.; Roussel-Baclet, C.; Maillet, D.; Belin, C.; Ankri, J.; Narme, P. Music enhances verbal episodic memory in Alzheimer’s disease. J. Clin. Exp. Neuropsychol. 2015, 37, 503–517. [Google Scholar] [CrossRef] [PubMed]
- Basaglia-Pappas, S.; Laterza, M.; Borg, C.; Richard-Mornas, A.; Favre, E.; Thomas-Antérion, C. Exploration of verbal and non-verbal semantic knowledge and autobiographical memories starting from popular songs in Alzheimer’s disease. Int. Psychogeriatr. 2013, 25, 785–795. [Google Scholar] [CrossRef]
- Melby-Lervåg, M.; Redick, T.S.; Hulme, C. Working Memory Training Does Not Improve Performance on Measures of Intelligence or Other Measures of “Far Transfer”: Evidence from a Meta-Analytic Review. Perspect. Psychol. Sci. 2016, 11, 512–534. [Google Scholar] [CrossRef] [PubMed]
- Livingston, G.; Sommerlad, A.; Orgeta, V.; Costafred, S.G.; Huntley, J.; Ame, D.S.; Kales, H.C. Dementia prevention, intervention, and care. Lancet 2017, 390, 2673–2734. [Google Scholar] [CrossRef] [PubMed]
- Arvanitakis, Z.; Shah, R.C.; Bennett, D.A. Diagnosis and Management of Dementia: Review. JAMA 2019, 322, 15–89. [Google Scholar] [CrossRef] [PubMed]
- Shadish, W.; Cook, T.D.; Campbell, D.T. Experimental and Quasi-Experimental Designs for Generalized Causal Inference; Houghton Mifflin: Boston, MA, USA, 2002. [Google Scholar]


| Assessment | Tests |
|---|---|
| Questionnaire | Music-based therapy protocol (applied on the first session of music therapy, only in the experimental group) |
| Rating | CDR (provided by the attending physician) |
| Global Cognitive Assessment | MMSE |
| Functional | Katz Functional Assessment Scale (applied on family caregivers, only in the clinical group) |
| Bayer Scale (applied on family caregivers, only in the clinical group) | |
| Humor | Geriatric Depression Scale |
| Specific cognitive areas | Memory |
| Figure Memory Test of the BCSB | |
| Autobiographical Memory Test | |
| Musical Autobiographical Memory Test | |
| SASMET | |
| Attention and Executive Function | |
| Five Digits Test | |
| Corsi block-tapping test | |
| Wechsler Adult Intelligence Scale III battery digits subtest | |
| Visual Perception | |
| Shulman Clock Drawing Test |
| Sung Words | Spoken Words | |||
|---|---|---|---|---|
| Group | Diagnosis | Time | Mean G2 | Mean G2 |
| Control | Healthy | 1 | 0.690 | 0.317 |
| 2 | 0.989 | 1.452 | ||
| not healthy | 1 | 0.381 | 0.740 | |
| 2 | 0.400 | 0.680 | ||
| Experimental | Healthy | 1 | 0.643 | 1.079 |
| 2 | 0.473 | 0.512 | ||
| not healthy | 1 | 0.107 | 0.433 | |
| 2 | 0.668 | 1.406 |
| Sung Words | Spoken Words | |||
|---|---|---|---|---|
| Group | Diagnosis | Time | Mean G2 | Mean G2 |
| Control | Healthy | 1 | 0.734 | 1.147 |
| 2 | 1.376 | 1.425 | ||
| not healthy | 1 | 0.358 | 0.435 | |
| 2 | 0.478 | 0.122 | ||
| Experimental | Healthy | 1 | 1.235 | 1.731 |
| 2 | 0.948 | 1.556 | ||
| not healthy | 1 | 0.686 | 1.210 | |
| 2 | 0.706 | 0.396 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moreira, S.V.; Justi, F.R.d.R.; Gomes, C.F.d.A.; Moreira, M. Music Therapy Enhances Episodic Memory in Alzheimer’s and Mixed Dementia: A Double-Blind Randomized Controlled Trial. Healthcare 2023, 11, 2912. https://doi.org/10.3390/healthcare11222912
Moreira SV, Justi FRdR, Gomes CFdA, Moreira M. Music Therapy Enhances Episodic Memory in Alzheimer’s and Mixed Dementia: A Double-Blind Randomized Controlled Trial. Healthcare. 2023; 11(22):2912. https://doi.org/10.3390/healthcare11222912
Chicago/Turabian StyleMoreira, Shirlene Vianna, Francis Ricardo dos Reis Justi, Carlos Falcão de Azevedo Gomes, and Marcos Moreira. 2023. "Music Therapy Enhances Episodic Memory in Alzheimer’s and Mixed Dementia: A Double-Blind Randomized Controlled Trial" Healthcare 11, no. 22: 2912. https://doi.org/10.3390/healthcare11222912
APA StyleMoreira, S. V., Justi, F. R. d. R., Gomes, C. F. d. A., & Moreira, M. (2023). Music Therapy Enhances Episodic Memory in Alzheimer’s and Mixed Dementia: A Double-Blind Randomized Controlled Trial. Healthcare, 11(22), 2912. https://doi.org/10.3390/healthcare11222912






