Prevalence of Spontaneous Bacterial Peritonitis (SBP) in Hepatitis B (HBV), and Hepatitis C (HCV) Liver Cirrhosis: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Study Design and Protocol
2.2. Literature Review Search Strategy
2.3. Inclusion and Exclusion Criteria
2.4. Data Extraction
2.5. Quality Assessment
2.6. Data Analysis
2.7. Heterogeneity Analysis and Publication Bias Test
3. Results
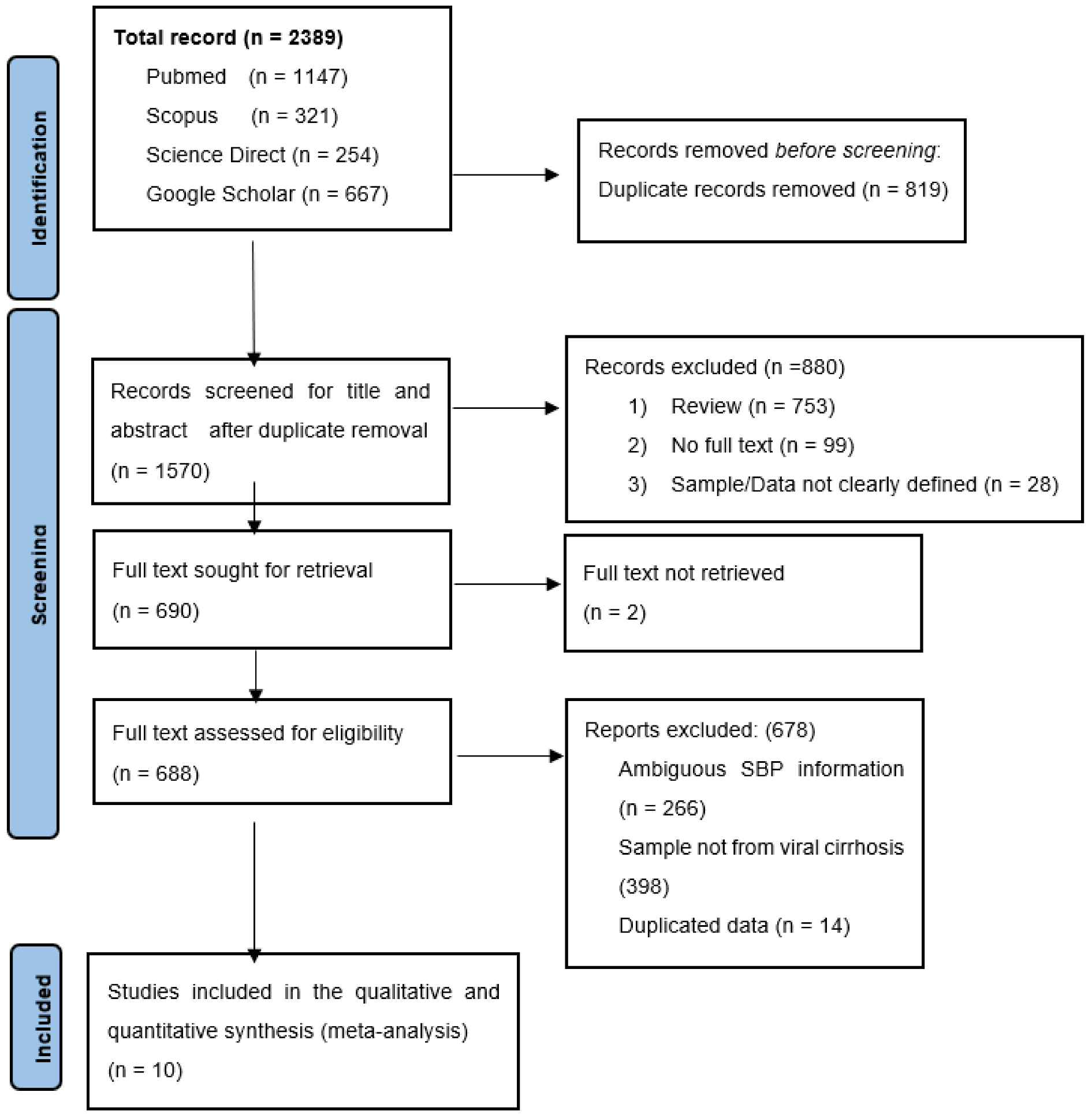
3.1. Search Results and Eligible Studies
3.2. Characteristics of the Eligible Studies
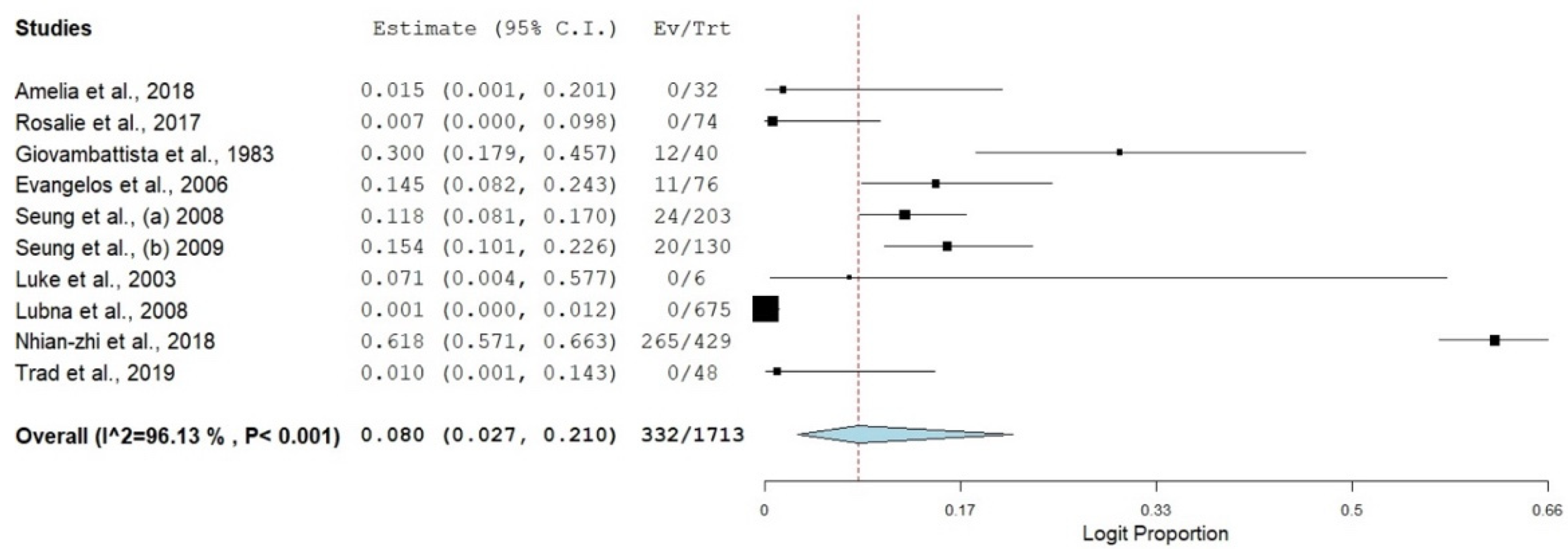
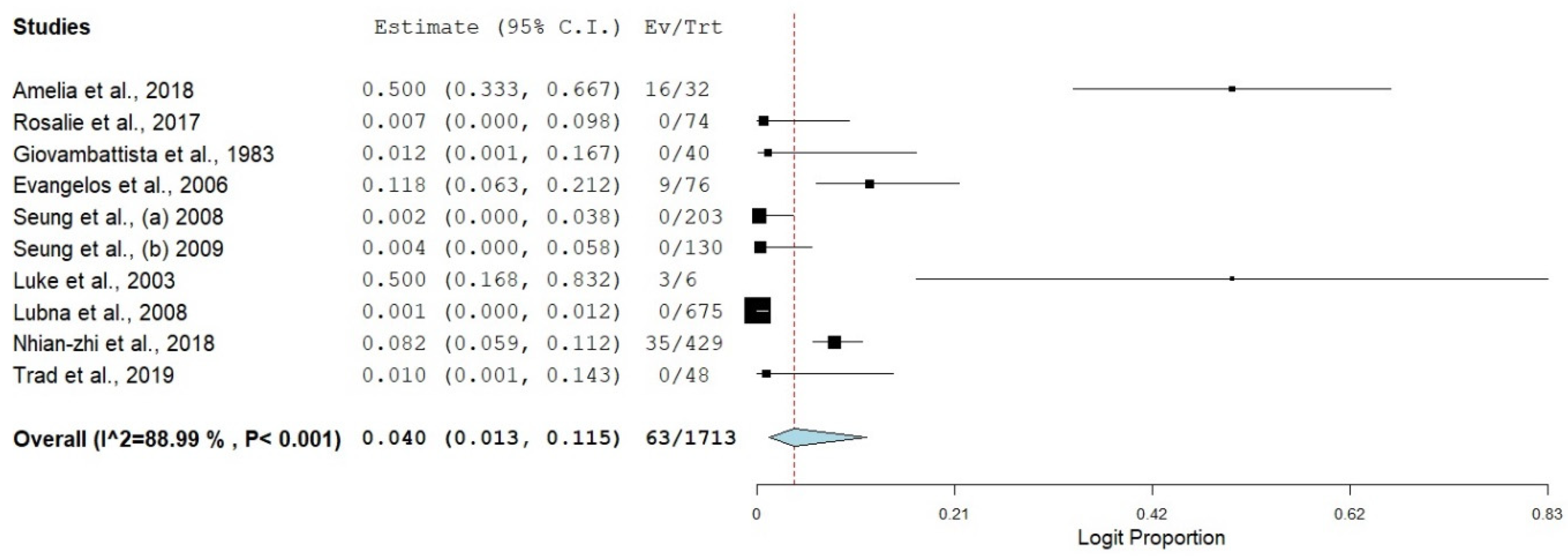
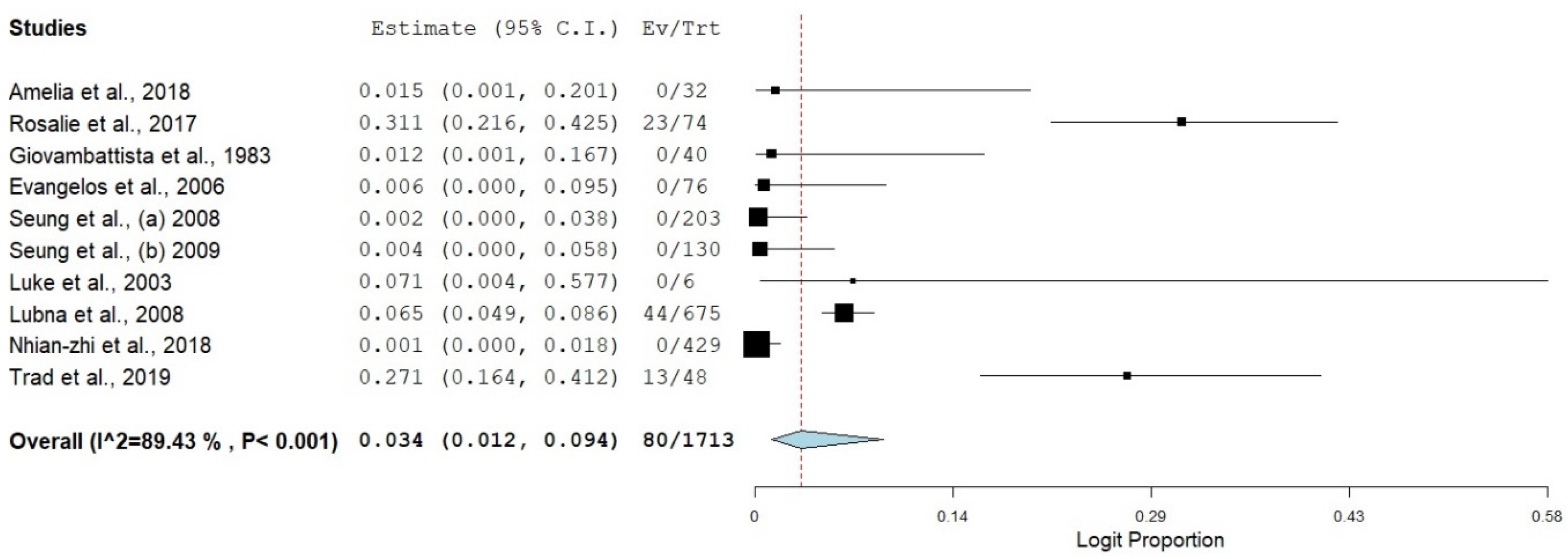
3.3. Prevalence of SBP in Viral Hepatitis-Related Cirrhosis
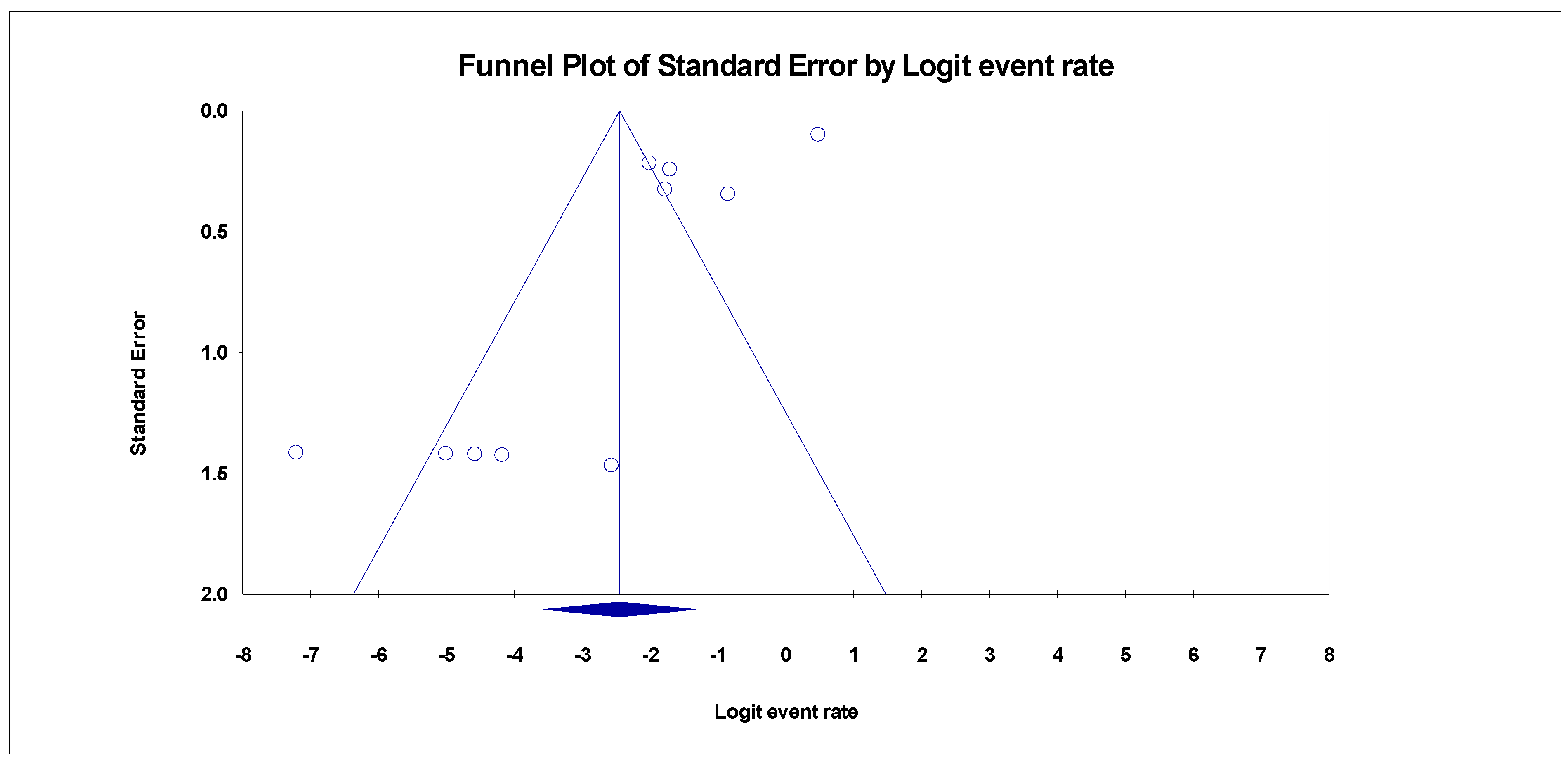
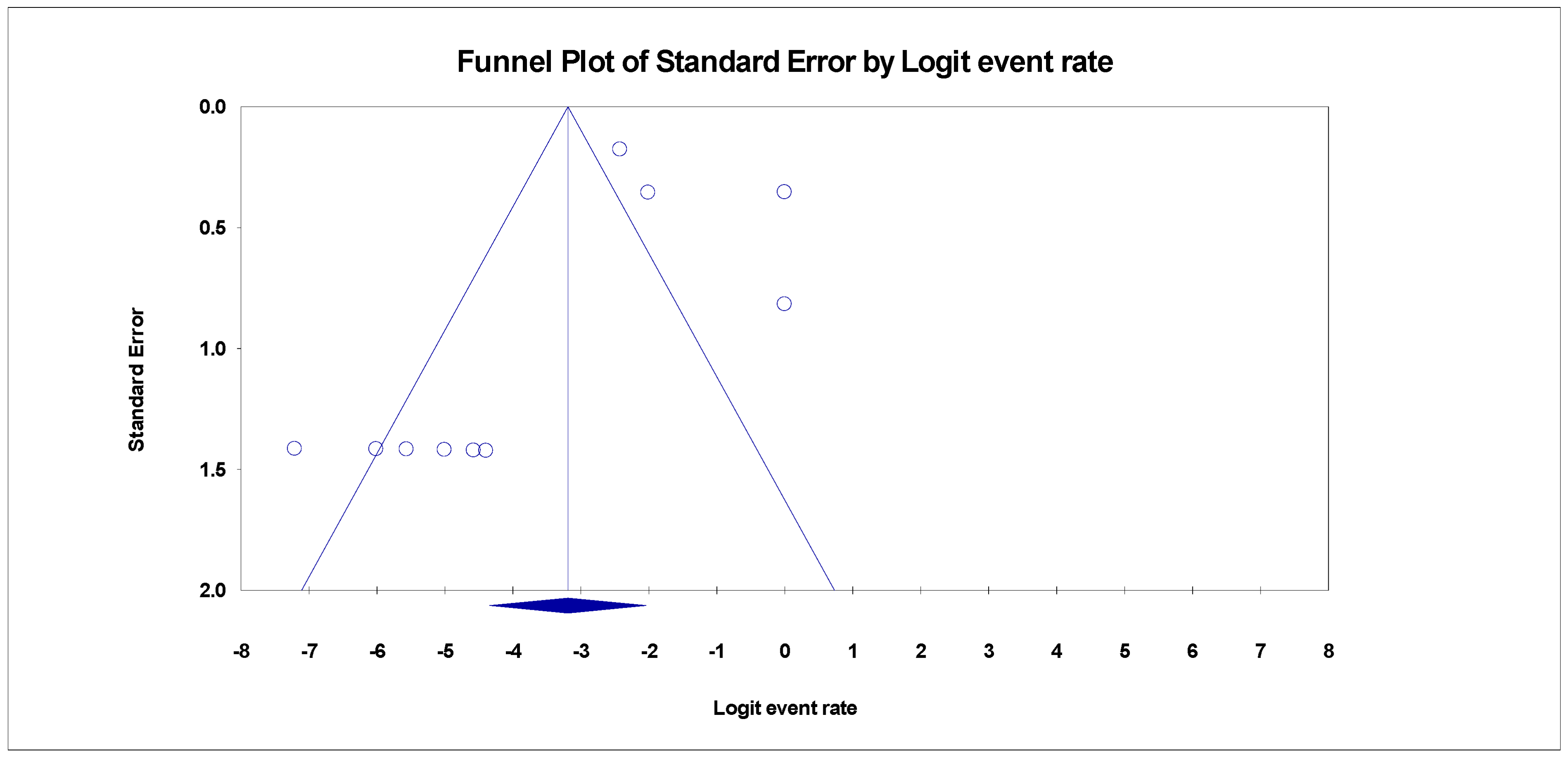
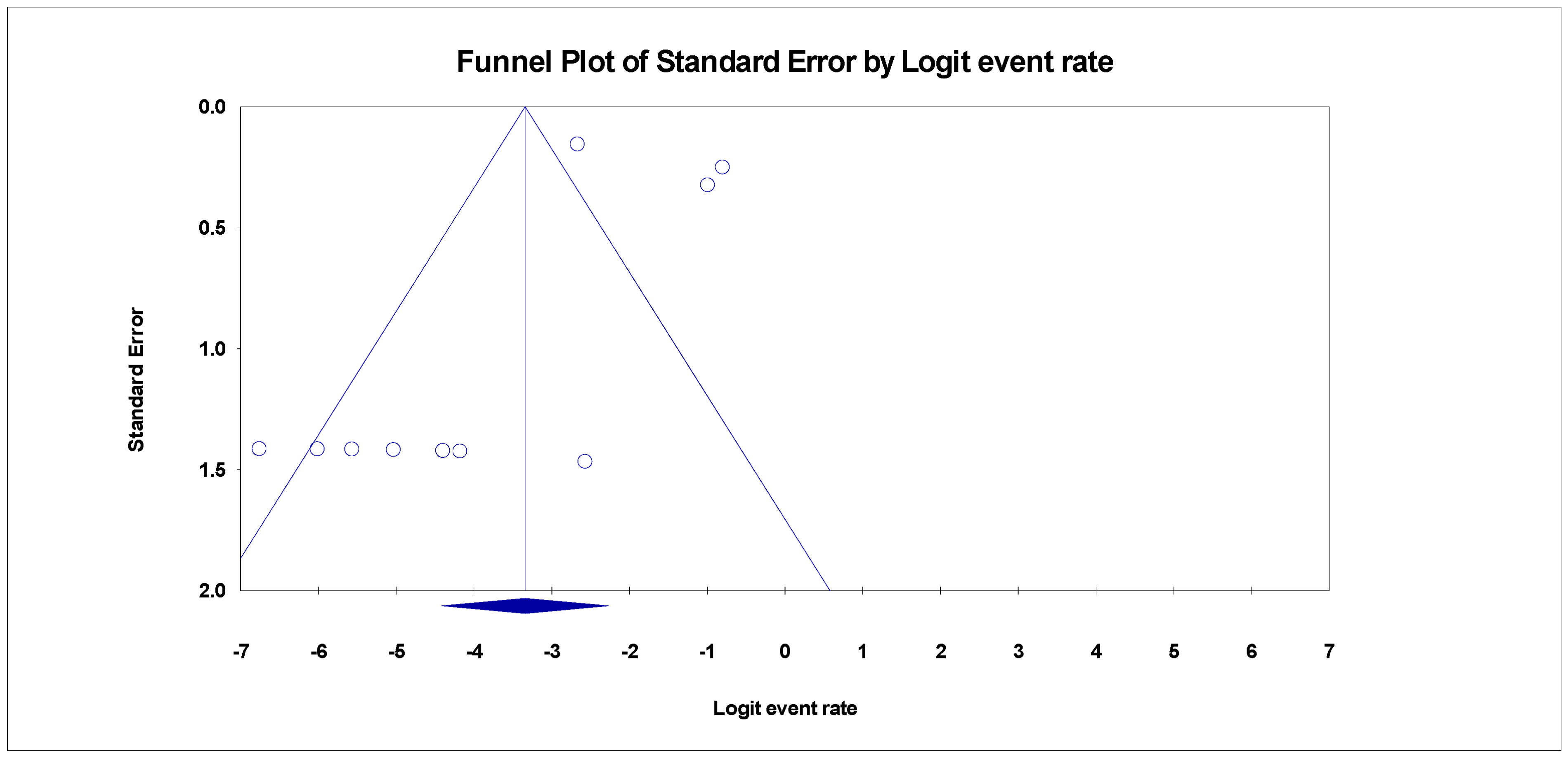
3.4. Publication Bias
3.5. Subgroup Meta-Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rostkowska, K.A.; Szymanek-Pasternak, A.; Simon, K.A. Spontaneous bacterial peritonitis—therapeutic challenges in the era of increasing drug resistance of bacteria. Clin. Exp. Hepatol. 2018, 4, 224–231, Termedia Publishing House Ltd (Warsaw, Poland). [Google Scholar] [CrossRef]
- Heo, J.; Seo, Y.S.; Yim, H.J.; Hahn, T.; Park, S.H.; Ahn, S.H.; Park, J.Y.; Park, J.Y.; Kim, M.Y.; Park, S.K.; et al. Clinical Features and Prognosis of Spontaneous Bacterial Peritonitis in Korean Patients with Liver Cirrhosis: A Multicenter Retrospective Study. 2009. Available online: http://www.unos.org (accessed on 23 August 2022).
- Piroth, L.; Pechinot, A.; Minello, A.; Jaulhac, B.; Patry, I.; Hadou, T.; Hansmann, Y.; Rabaud, C.; Chavanet, P.; Neuwirth, C. Bacterial epidemiology and antimicrobial resistance in ascitic fluid: A 2-year retrospective study. Scand. J. Infect. Dis. 2009, 41, 847–851. [Google Scholar] [CrossRef]
- Ding, X.; Yu, Y.; Chen, M.; Wang, C.; Kang, Y.; Lou, J. Causative agents and outcome of spontaneous bacterial peritonitis in cirrhotic patients: Community-acquired versus nosocomial infections. BMC Infect. Dis. 2019, 19, 463. [Google Scholar] [CrossRef]
- Williams, E.J. Liver cirrhosis. Postgrad. Med. J. 1998, 74, 193–202, BMJ Publishing Group: London, UK. [Google Scholar] [CrossRef]
- Stickel, F.; Datz, C.; Hampe, J.; Bataller, R. Pathophysiology and management of alcoholic liver disease: Update 2016. Gut Liver 2017, 11, 173–188. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Zhang, C.; Zhan, Y.T. Nonalcoholic Fatty Liver Disease Cirrhosis: A Review of Its Epidemiology, Risk Factors, Clinical Presentation, Diagnosis, Management, and Prognosis. Can. J. Gastroenterol. Hepatol. 2018, 2018, 2784537, Hindawi Limited: London, UK, 2018. [Google Scholar] [CrossRef]
- Li, Y.T.; Huang, J.R.; Peng, M.L. Current Status and Prospects of Spontaneous Peritonitis in Patients with Cirrhosis. BioMed Res. Int. 2020, 2020, 3743962. [Google Scholar] [CrossRef]
- Ekpanyapong, S.; Reddy, K.R. Infections in Cirrhosis. Curr. Treat. Options Gastroenterol. 2019, 17, 254–270. [Google Scholar] [CrossRef]
- Irekeola, A.A.; Malek, N.A.; Wada, Y.; Mustaffa, N.; Muhamad, N.I.; Shueb, R.H. Prevalence of HCV genotypes and subtypes in Southeast Asia: A systematic review and meta-analysis. PLoS ONE 2021, 16, e0251673. [Google Scholar] [CrossRef]
- Tay, P.W.L.; Xiao, J.; Tan, D.J.H.; Ng, C.; Lye, Y.N.; Lim, W.H.; Teo, V.X.Y.; Heng, R.R.Y.; Yeow, M.W.X.; Lum, L.H.W.; et al. An Epidemiological Meta-Analysis on the Worldwide Prevalence, Resistance, and Outcomes of Spontaneous Bacterial Peritonitis in Cirrhosis. Front. Med. 2021, 8, 1254. [Google Scholar] [CrossRef]
- Shamseer, L.; Moher, D.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (Prisma-p) 2015: Elaboration and explanation. BMJ 2015, 349, g7647, BMJ Publishing Group: London, UK. [Google Scholar] [CrossRef]
- Munn, Z.; MClinSc, S.M.; Lisy, K.; Riitano, D.; Tufanaru, C. Methodological guidance for systematic reviews of observational epidemiological studies reporting prevalence and cumulative incidence data. Int. J. Evid. -Based Healthc. 2015, 13, 147–153. [Google Scholar] [CrossRef]
- Bax, L.; Yu, L.M.; Ikeda, N.; Moons, K.G.M. A systematic comparison of software dedicated to a meta-analysis of causal studies. BMC Med. Res. Methodol. 2007, 7, 40. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef]
- George, B.J.; Aban, I.B. An application of meta-analysis based on DerSimonian and Laird method. J. Nucl. Cardiol. 2016, 23, 690–692, Springer New York LLC: New York, NY, USA. [Google Scholar] [CrossRef]
- Sofjan, A.K.; Musgrove, R.J.; Beyda, N.D.; Russo, H.P.; Lasco, T.M.; Yau, R.; Restrepo, A.; Garey, K.W. Prevalence and predictors of spontaneous bacterial peritonitis due to ceftriaxone-resistant organisms at a large tertiary centre in the USA. J. Glob. Antimicrob. Resist. 2018, 15, 41–47. [Google Scholar] [CrossRef]
- Oey, R.C.; de Man, R.A.; Erler, N.S.; Verbon, A.; van Buuren, H.R. Microbiology and antibiotic susceptibility patterns in spontaneous bacterial peritonitis: A study of two Dutch cohorts at a 10-year interval. United Eur. Gastroenterol. J. 2018, 6, 614. [Google Scholar] [CrossRef]
- Simonetti, R.G.; Craxí, A.; Piazza, S.D.; Spanó, C.; Pagliaro, L. Spontaneous bacterial peritonitis: A prospective investigation in predominantly nonalcoholic cirrhotic patients. Hepatology 1983, 3, 545–549. [Google Scholar] [CrossRef]
- Cholongitas, E.; Papatheodoridis, G.; Manesis, E.K.; Burroughs, A.K.; Archimandritis, A.J. Spontaneous bacterial peritonitis in cirrhotic patients: Is prophylactic propranolol therapy beneficial? J. Gastroenterol. Hepatol. 2006, 21, 581–587. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.U.; Han, K.H.; Nam, C.M.; Park, J.Y.; Kim, D.Y.; Chon, C.Y.; Ahn, S.H. Natural history of hepatitis B virus-related cirrhotic patients hospitalized to control ascites. J. Gastroenterol. Hepatol. 2008, 23, 1722–1727. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.U.; Kim, D.Y.; Lee, C.K.; Park, J.Y.; Kim, S.H.; Kim, H.M.; Choi, E.H.; Kim, S.; Han, K.H.; Chon, C.Y.; et al. Ascitic fluid infection in patients with hepatitis B virus-related liver cirrhosis: Culture-negative neutrocytic ascites versus spontaneous bacterial peritonitis. J. Gastroenterol. Hepatol. 2010, 25, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Evans, L.T.; Kim, W.R.; Poterucha, J.J.; Kamath, P.S. Spontaneous bacterial peritonitis in asymptomatic outpatients with cirrhotic ascites. Hepatology 2003, 37, 897–901. [Google Scholar] [CrossRef] [PubMed]
- Kamani, L.; Mumtaz, K.; Ahmed, U.S.; Ali, A.W.; Jafri, W. Outcomes in culture positive and culture negative ascitic fluid infection in patients with viral cirrhosis: Cohort study. BMC Gastroenterol. 2008, 8, 59. [Google Scholar] [CrossRef] [PubMed]
- Ning, N.Z.; Li, T.; Zhang, J.L.; Qu, F.; Huang, J.; Liu, X.; Li, Z.; Geng, W.; Fu, J.L.; Huan, W.; et al. Clinical and bacteriological features and prognosis of ascitic fluid infection in Chinese patients with cirrhosis. BMC Infect. Dis. 2018, 18, 253. [Google Scholar] [CrossRef]
- Trad, D. Cirrhotic Patients: Predictive Factors of Recurrence and Survival. Ann. Clin. Hepatol. 2019, 3, 1013. [Google Scholar]
- El Khouri, M.; Santos, V.A.D. Hepatitis B: Epidemiological, immunological, and serological considerations emphasizing mutation. Rev. Hosp. Clin. Fac. Med. 2004, 59, 216–224. [Google Scholar] [CrossRef]
- Smith, J.M.; Uvin, A.Z.; Macmadu, A.; Rich, J.D. Epidemiology and Treatment of Hepatitis B in Prisoners. Curr. Hepatol. Rep. 2017, 16, 178–183. [Google Scholar] [CrossRef]
- Rehermann, B.; Nascimbeni, M. Immunology of hepatitis B virus and hepatitis C virus infection. Nat. Rev. Immunol. 2005, 5, 215–229. [Google Scholar] [CrossRef]
- Thimme, R.; Oldach, D.; Chang, K.M.; Steiger, C.; Ray, S.C.; Chisari, F. Determinants of Viral Clearance and Persistence during Acute Hepatitis C Virus Infection. J. Exp. Med. 2001, 194, 1395–1406. [Google Scholar] [CrossRef]
- Gou, Y.Z.; Liu, B.; Pan, L.; Yu, H.T.; Wang, J.P.; Wang, D.C. Pathogens of spontaneous bacterial peritonitis change in northern China. Saudi Med. J. 2010, 31, 1152–1156. Available online: https://europepmc.org/article/med/20953533 (accessed on 20 October 2022).
- Shi, L.; Wu, D.; Wei, L.; Liu, S.; Zhao, P.; Tu, B.; Xie, Y.; Liu, Y.; Wang, X.; Liu, L.; et al. Nosocomial and Community-Acquired Spontaneous Bacterial Peritonitis in patients with liver cirrhosis in China: Comparative Microbiology and Therapeutic Implications. Sci. Rep. 2017, 7, 46025. [Google Scholar] [CrossRef] [PubMed]
- Piano, S.; Fasolato, S.; Salinas, F.; Romano, A.; Tonon, M.; Morando, F.; Cavallin, M.; Gola, E.; Sticca, A.; Loregian, A.; et al. The empirical antibiotic treatment of nosocomial spontaneous bacterial peritonitis: Results of a randomized, controlled clinical trial. Hepatology 2016, 63, 1299–1309. [Google Scholar] [CrossRef] [PubMed]
- Wasley, A.; Kruszon-Moran, D.; Kuhnert, W.; Simard, E.P.; Finelli, L.; McQuillan, G.; Bell, B. The Prevalence of Hepatitis B Virus Infection in the United States in the Era of Vaccination. J. Infect. Dis. 2010, 202, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Aziz, A.; Ashraf, S.; Tahir Hussain Talpur, M.; Aamer, N.; Ahmed Solangi, S.; Ullah Shabir, K.; Ahmed, M.; Abbas, K. Spontaneous bacterial peritonitis in asymptomatic cirrhotic patients with ascites in a tertiary care hospital: A cross-sectional study. Pak. Armed Forces Med. J. 2020, 70, 1408–1420. [Google Scholar]
- Ennaifer, R.; Cheikh, M.; Hefaiedh, R.; Romdhane, H.; Nejma, H.; Hadj, N.B. Glucose Metabolism Disorders in Cirrhosis: Frequency and Risk Factors in Tunisian Population. Results of a Cross-Sectional Study. Open J. Gastroenterol. 2014, 2014, 289–294. [Google Scholar] [CrossRef]
- Kwiatkowski, C.F.; Corsi, K.F.; Booth, R.E. The association between knowledge of hepatitis C virus status and risk behaviors in injection drug users. Addiction 2002, 97, 1289–1294. [Google Scholar] [CrossRef]
- Ryerson, A.B.; Schillie, S.; Barker, L.K.; Kupronis, B.A.; Wester, C. Vital Signs: Newly Reported Acute and Chronic Hepatitis C Cases—United States, 2009–2018. Morb. Mortal. Wkly. Rep. 2020, 69, 399. [Google Scholar] [CrossRef]
- Hofmeister, M.G.; Rosenthal, E.M.; Barker, L.K.; Rosenberg, E.S.; Barranco, M.A.; Hall, E.W.; Edlin, B.R.; Mermin, J.; Ward, J.W.; Ryerson, A.B. Estimating Prevalence of Hepatitis C Virus Infection in the United States, 2013–2016. Hepatology 2019, 69, 1020–1031. [Google Scholar] [CrossRef]
- Pimpin, L.; Cortez-Pinto, H.; Negro, F.; Corbould, E.; Lazarus, J.V.; Webber, L.; Sheron, N.; EASL HEPAHEALTH Steering Committee. Burden of liver disease in Europe: Epidemiology and analysis of risk factors to identify prevention policies. J. Hepatol. 2018, 69, 718–735. [Google Scholar] [CrossRef]
- Popoiag, R.E.; Panaitescu, E.; Suceveanu, A.I.; Suceveanu, A.P.; Micu, S.I.; Mazilu, L.; Parepa, I.; Voinea, F.; Costea, D.O.; Enache, F.; et al. Spontaneous bacterial peritonitis mortality trends of cirrhotic patients in the last decade in Constanta County. Exp. Ther. Med. 2021, 22, 732. [Google Scholar] [CrossRef]
- Liu, Z.; Yang, Q.; Shi, O.; Ye, W.; Chen, X.; Zhang, T. The epidemiology of hepatitis B and hepatitis C infections in China from 2004 to 2014: An observational population-based study. J. Viral. Hepat. 2018, 25, 1543–1554. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liang, W.; Jing, W.; Liu, M. Countdown to 2030: Eliminating hepatitis B disease, China. Bull. World Health Organ. 2019, 97, 230. [Google Scholar] [CrossRef]
- Lazarus, J.V.; Picchio, C.A.; Guy, D.; Aleman, S.; James, C.; Nava, F.A.; Øvrehus, A.; Turnes, J.; Ward, J.W.; Ustianowski, A. Hepatitis C standards of care: A review of good practices since the advent of direct-acting antiviral therapy. Clin. Res. Hepatol. Gastroenterol. 2021, 45, 101564. [Google Scholar] [CrossRef]
- Cox, A.L. Challenges and Promise of a Hepatitis C Virus Vaccine. Cold Spring Harb. Perspect. Med. 2020, 10, a036947. [Google Scholar] [CrossRef]
- Matsui, C.; Deng, L.; Minami, N.; Abe, T.; Koike, K.; Shoji, I. Hepatitis C Virus NS5A Protein Promotes the Lysosomal Degradation of Hepatocyte Nuclear Factor 1α via Chaperone-Mediated Autophagy. J. Virol. 2018, 92, e00639-18. [Google Scholar] [CrossRef]
- Das, S.; Ramakrishnan, K.; Behera, S.K.; Ganesapandian, M.; Xavier, A.S.; Selvarajan, S. Hepatitis B Vaccine and Immunoglobulin: Key Concepts. J. Clin. Transl. Hepatol. 2019, 7, 165. [Google Scholar] [CrossRef]
- Kim, M.J.; Song, K.H.; Kim, N.H.; Choe, P.G.; Park, W.B.; Bang, J.H.; Kim, E.S.; Park, S.W.; Kim, H.B.; Lee, H.S.; et al. Clinical outcomes of spontaneous bacterial peritonitis due to extended-spectrum beta-lactamase-producing Escherichia coli or Klebsiella pneumoniae: A retrospective cohort study. Hepatol. Int. 2014, 8, 582–587. [Google Scholar] [CrossRef]
| Author, Year | Year | Country | Sampling Period | HBV Sampled | SBP in HBV | HCV Sampled | SBP in HCV | HBV + HCV Sampled | SBP in HBV + HCV | Study Design | Method of Detection |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Amelia et al. [17] | 2018 | USA | November 2011–March 2016 | 0 | 0 | 32 | 16 | 0 | 0 | Case-control | Cell count and culture |
| Rosalie et al. [18] | 2017 | Holland | January 2003–December 2005 | 0 | 0 | 0 | 0 | 74 | 23 | Retrospective | Culture |
| Giovambattista et al. [19] | 1983 | Italy | December 1976–December 1978 | 40 | 12 | 0 | 0 | 0 | 0 | Retrospective | Culture |
| Evangelos et al. [20] | 2006 | Greece | June 1999–June 2001 | 49 | 11 | 27 | 9 | 0 | 0 | Retrospective | Culture |
| Seung et al. (a) [21] | 2008 | South Korea | January 1996–December 2015 | 203 | 24 | 0 | 0 | 0 | 0 | Retrospective | Cell count and culture |
| Seung et al. (b) [22] | 2009 | South Korea | January 1998–December 2007 | 130 | 20 | 0 | 0 | 0 | 0 | Retrospective | Culture |
| Luke et al. [23] | 2003 | USA | July 1994–December 2002 | 0 | 0 | 6 | 3 | 0 | 0 | Prospective | Culture |
| Lubna et al. [24] | 2008 | Pakistan | Nov 2005–December 2007 | 0 | 0 | 0 | 0 | 675 | 44 | Retrospective | Culture |
| Nhian-Zhi et al., [25] | 2018 | China | January 2012–December 2015 | 370 | 265 | 59 | 35 | 0 | 0 | Retrospective | Culture |
| Trad et al. [26] | 2019 | Tunisia | January 2003–December 2017 | 0 | 0 | 0 | 0 | 48 | 13 | Retrospective | Cell count and culture |
| Random Effect Model | |||||
|---|---|---|---|---|---|
| Kendal Tau’s p | |||||
| Studies Trimmed | Point Estimates | Q Value | Without Continuity Correction | With Continuity Correction | |
| SBP in HBV cirrhosis | |||||
| Adjusted | 4 | 1.299 | 253.55 | 0.180 | 0.211 |
| Observed | - | 2.448 | 232.86 | ||
| SBP in HCV cirrhosis | |||||
| Adjusted | 4 | 1.916 | 119.08 | 0.788 | 0.858 |
| Observed | - | 3.189 | 81.73 | ||
| SBP in HBV + HCV cirrhosis | |||||
| Adjusted | 4 | 2.213 | 112.00 | 0.180 | 0.211 |
| Observed | - | 3.342 | 85.12 | ||
| Country | Number of Studies | Prevalence (%) | 95% CI | I2 (%) | Q | Heterogeneity Test | |
|---|---|---|---|---|---|---|---|
| DF | P | ||||||
| SBP in both HBV and HCV cirrhosis | |||||||
| USA | 2 | 3.3 | 0.5–19.9 | 0.00 | 0.619 | 1 | 0.431 |
| Holland | 1 | 31.1 | 21.6–42.5 | - | - | - | - |
| Italy | 1 | 1.2 | 0.1–16.7 | - | - | - | - |
| Greece | 1 | 0.6 | 0.0–9.5 | - | - | - | - |
| South Korea | 2 | 0.3 | 0.0–2.1 | 0.00 | 0.049 | 1 | 0.824 |
| Pakistan | 1 | 0.1 | 4.9–8.6 | - | - | - | - |
| China | 1 | 0.1 | 0.0–1.8 | - | - | - | - |
| Tunisia | 1 | 27.1 | 16.4–32.5 | - | - | - | - |
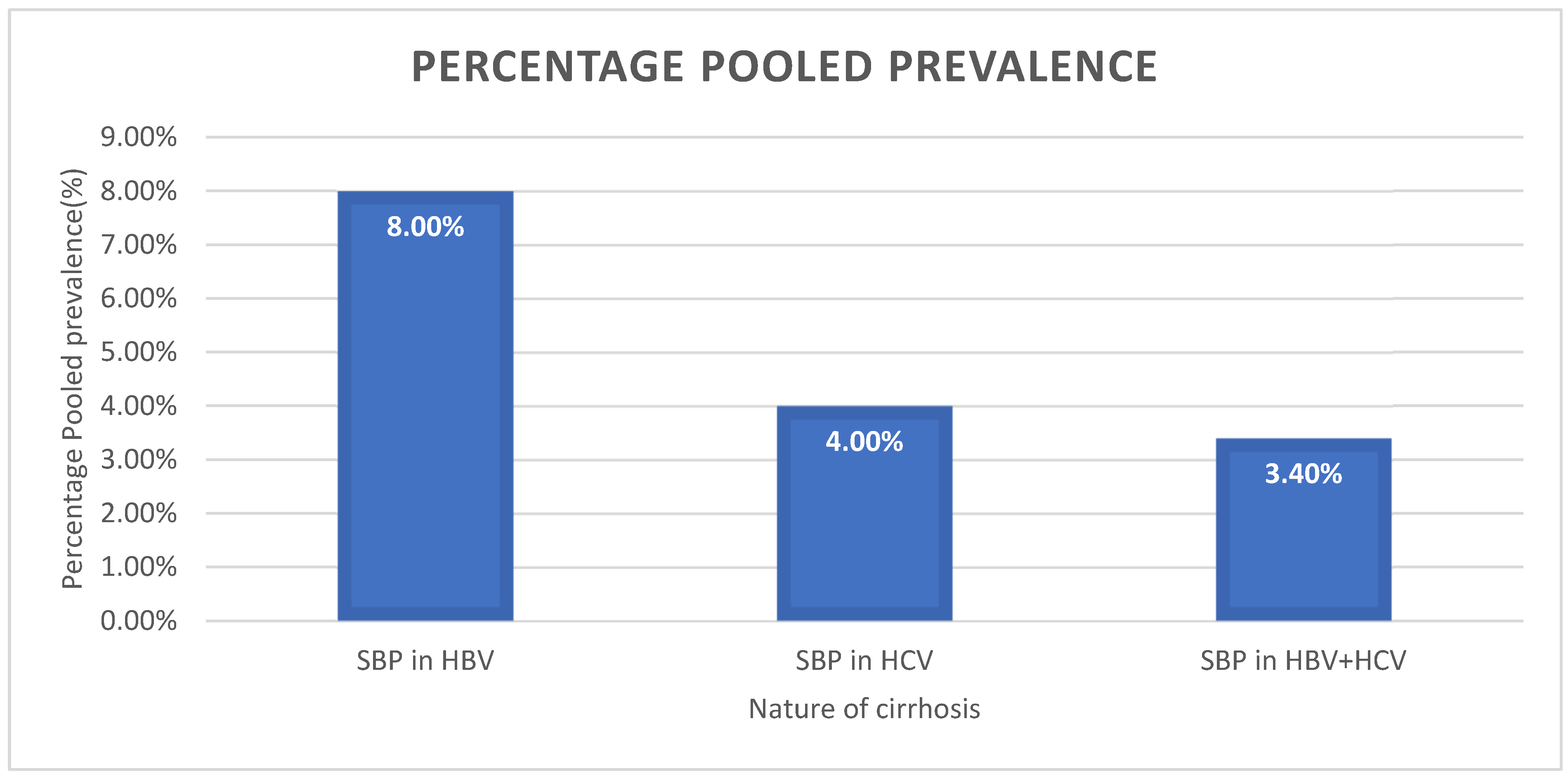
| Overall | 10 | 3.4 | 1.2–9.4 | 89.43 | 65.12 | 9 | <0.001 |
| SBP in HCV cirrhosis | |||||||
| USA | 2 | 50.0 | 34.6–65.4 | 0.00 | 0.00 | 1 | 1.000 |
| Holland | 1 | 0.7 | 0.0–9.8 | - | - | - | - |
| Italy | 1 | 1.2 | 0.1–16.7 | - | - | - | - |
| Greece | 1 | 11.8 | 6.3–21.2 | - | - | - | - |
| South Korea | 2 | 0.3 | 0.0–2.1 | 0.00 | 0.049 | 1 | 0.824 |
| Pakistan | 1 | 0.1 | 0.0–1.2 | - | - | - | - |
| China | 1 | 8.2 | 5.9–11.2 | - | - | - | - |
| Tunisia | 1 | 1.0 | 0.1–14.3 | - | - | - | - |
| Overall | 10 | 4.0 | 1.3–11.5 | 88.99 | 81.73 | 9 | <0.001 |
| SBP in HBV cirrhosis | |||||||
| USA | 2 | 3.3 | 0.5–19.9 | 0.00 | 0.619 | 1 | 0.431 |
| Holland | 1 | 0.7 | 0.0–9.8 | - | - | - | - |
| Italy | 1 | 30.0 | 17.9–45.7 | - | - | - | - |
| Greece | 1 | 14.5 | 8.2–24.3 | - | - | - | - |
| South Korea | 2 | 13.3 | 10.1–17.4 | 0.00 | 0.872 | 1 | 0.350 |
| Pakistan | 1 | 0.1 | 0.0–1.2 | - | - | - | - |
| China | 1 | 61.8 | 57.1–66.3 | - | - | - | - |
| Tunisia | 1 | 1.0 | 0.1–14.3 | - | - | - | - |
| Overall | 10 | 8.0 | 2.7–21.0 | 96.13 | 232.86 | 9 | <0.001 |
| Sampling Period | Number of Studies | Prevalence (%) | 95% CI | I2 (%) | Q | Heterogeneity Test | |
|---|---|---|---|---|---|---|---|
| DF | P | ||||||
| SBP in HBV cirrhosis | |||||||
| >2016 | 2 | 1.2 | 0.2–8.3 | 0.00 | 0.040 | 1 | 0.842 |
| 2006–2015 | 4 | 11.0 | 2.1–41.9 | 98.32 | 178.95 | 3 | <0.001 |
| <2006 | 4 | 12.9 | 4.6–31.7 | 72.48 | 10.90 | 3 | 0.012 |
| Overall | 10 | 8.0 | 2.7–21.0 | 96.13 | 232.86 | 9 | <0.001 |
| SBP in HCV cirrhosis | |||||||
| >2016 | 2 | 11.1 | 0.1–91.6 | 89.75 | 9.75 | 1 | 0.002 |
| 2006–2015 | 4 | 0.6 | 0.0–8.7 | 86.25 | 21.81 | 3 | <0.001 |
| <2006 | 4 | 7.6 | 1.3–34.1 | 77.46 | 13.31 | 3 | 0.004 |
| Overall | 10 | 4.0 | 11.5–58.8 | 88.99 | 81.73 | 9 | <0.001 |
| SBP in both HBV and HCV cirrhosis | |||||||
| >2016 | 2 | 9.3 | 0.5–68.6 | 78.93 | 4.75 | 1 | 0.029 |
| 2006–2015 | 4 | 0.7 | 0.1–7.3 | 82.87 | 17.52 | 3 | <0.001 |
| <2006 | 4 | 4.9 | 0.5–36.0 | 80.61 | 15.48 | 3 | 0.001 |
| Overall | 10 | 3.4 | 1.2–9.4 | 89.43 | 85.12 | 9 | <0.001 |
| Study Design | Number of Studies | Prevalence (%) | 95% CI | I2 (%) | Q | Heterogeneity Test | |
|---|---|---|---|---|---|---|---|
| DF | P | ||||||
| SBP in HBV cirrhosis | |||||||
| Case-control | 1 | 1.5 | 0.1–20.1 | - | - | - | - |
| Retrospective | 8 | 9.2 | 3.0–25.3 | 96.87 | 223.41 | 7 | <0.001 |
| Prospective | 1 | 7.1 | 0.4–57.7 | - | - | - | - |
| Overall | 10 | 8.0 | 2.7–21.0 | 96.13 | 232.86 | 9 | <0.001 |
| SBP in HCV cirrhosis | |||||||
| Case-control | 1 | 50.0 | 33.3–66.7 | - | - | - | - |
| Retrospective | 8 | 1.7 | 0.6–5.0 | 77.35 | 30.91 | 7 | <0.001 |
| Prospective | 1 | 50.0 | 16.8–83.2 | - | - | - | - |
| Overall | 10 | 4.0 | 1.3–11.5 | 88.99 | 81.73 | 9 | <0.001 |
| SBP in both HBV and HCV cirrhosis | |||||||
| Case-control | 1 | 1.5 | 0.1–20.1 | - | - | - | - |
| Retrospective | 8 | 3.4 | 1.1–10.2 | 91.56 | 82.94 | 7 | <0.001 |
| Prospective | 1 | 7.1 | 0.4–57.7 | - | - | - | - |
| Overall | 10 | 3.4 | 1.2–9.4 | 89.43 | 85.12 | 9 | <0.001 |
| Method of Detection | Number of Studies | Prevalence (%) | 95% CI | I2 (%) | Q | Heterogeneity Test | |
|---|---|---|---|---|---|---|---|
| DF | P | ||||||
| SBP in HBV cirrhosis | |||||||
| Cell count and culture | 3 | 4.1 | 0.6–21.7 | 62.4 | 5.32 | 2 | 0.007 |
| Only culture | 7 | 10.7 | 3.2–30.3 | 95.97 | 148.75 | 6 | <0.001 |
| Overall | 8.0 | 2.7 | 2.7–57.2 | 96.13 | 232.86 | 9 | <0.001 |
| SBP in HCV cirrhosis | |||||||
| Cell count and culture | 3 | 3.3 | 0.0–70.8 | 92.09 | 25.29 | 2 | <0.001 |
| Only culture | 7 | 4.1 | 1.4–11.6 | 81.1 | 31.75 | 6 | <0.001 |
| Overall | 8.0 | 4.0 | 1.3–11.5 | 88.99 | 81.73 | 9 | <0.001 |
| SBP in both HBV and HCV cirrhosis | |||||||
| Cell count and culture | 3 | 2.9 | 0.1–46.4 | 87.48 | 15.97 | 2 | <0.001 |
| Only culture | 7 | 2.9 | 0.8–10.2 | 90.37 | 62.28 | 6 | <0.001 |
| Overall | 8.0 | 3.4 | 1.2–9.4 | 89.43 | 85.12 | 9 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bello, K.E.; Irekeola, A.A.; Al-Mhanna, S.B.; Oseni, O.M.-L.; Omebije, A.P.; Shueb, R.H.; Mustaffa, N. Prevalence of Spontaneous Bacterial Peritonitis (SBP) in Hepatitis B (HBV), and Hepatitis C (HCV) Liver Cirrhosis: A Systematic Review and Meta-Analysis. Healthcare 2023, 11, 275. https://doi.org/10.3390/healthcare11020275
Bello KE, Irekeola AA, Al-Mhanna SB, Oseni OM-L, Omebije AP, Shueb RH, Mustaffa N. Prevalence of Spontaneous Bacterial Peritonitis (SBP) in Hepatitis B (HBV), and Hepatitis C (HCV) Liver Cirrhosis: A Systematic Review and Meta-Analysis. Healthcare. 2023; 11(2):275. https://doi.org/10.3390/healthcare11020275
Chicago/Turabian StyleBello, Kizito Eneye, Ahmad Adebayo Irekeola, Sameer Badri Al-Mhanna, Okolo Martin-Luther Oseni, Adejo Patience Omebije, Rafidah Hanim Shueb, and Nazri Mustaffa. 2023. "Prevalence of Spontaneous Bacterial Peritonitis (SBP) in Hepatitis B (HBV), and Hepatitis C (HCV) Liver Cirrhosis: A Systematic Review and Meta-Analysis" Healthcare 11, no. 2: 275. https://doi.org/10.3390/healthcare11020275
APA StyleBello, K. E., Irekeola, A. A., Al-Mhanna, S. B., Oseni, O. M.-L., Omebije, A. P., Shueb, R. H., & Mustaffa, N. (2023). Prevalence of Spontaneous Bacterial Peritonitis (SBP) in Hepatitis B (HBV), and Hepatitis C (HCV) Liver Cirrhosis: A Systematic Review and Meta-Analysis. Healthcare, 11(2), 275. https://doi.org/10.3390/healthcare11020275