CADUCEO: A Platform to Support Federated Healthcare Facilities through Artificial Intelligence
Abstract
1. Introduction
- The need for quality and representative data;
- The difficulty of interpreting and explaining the results given as output by ANNs, i.e., explaining their inner workings;
- The legal and ethical implications arising from the use of intelligent systems in clinical practice.
2. Materials and Methods
2.1. Data Augmentation
2.2. Federated Learning
2.3. Explainable AI
- Intrinsically Interpretable Methods: These methods use inherently interpretable models, such as Decision Trees and Linear Regression.
- Sample-Based Methods: Specific samples from the dataset are used to describe the model’s behavior. An example is the K-Nearest Neighbors method.
- Model-Agnostic Methods: The explanation method is separate from the model being explained. Examples include Global Surrogate and Local Surrogate.
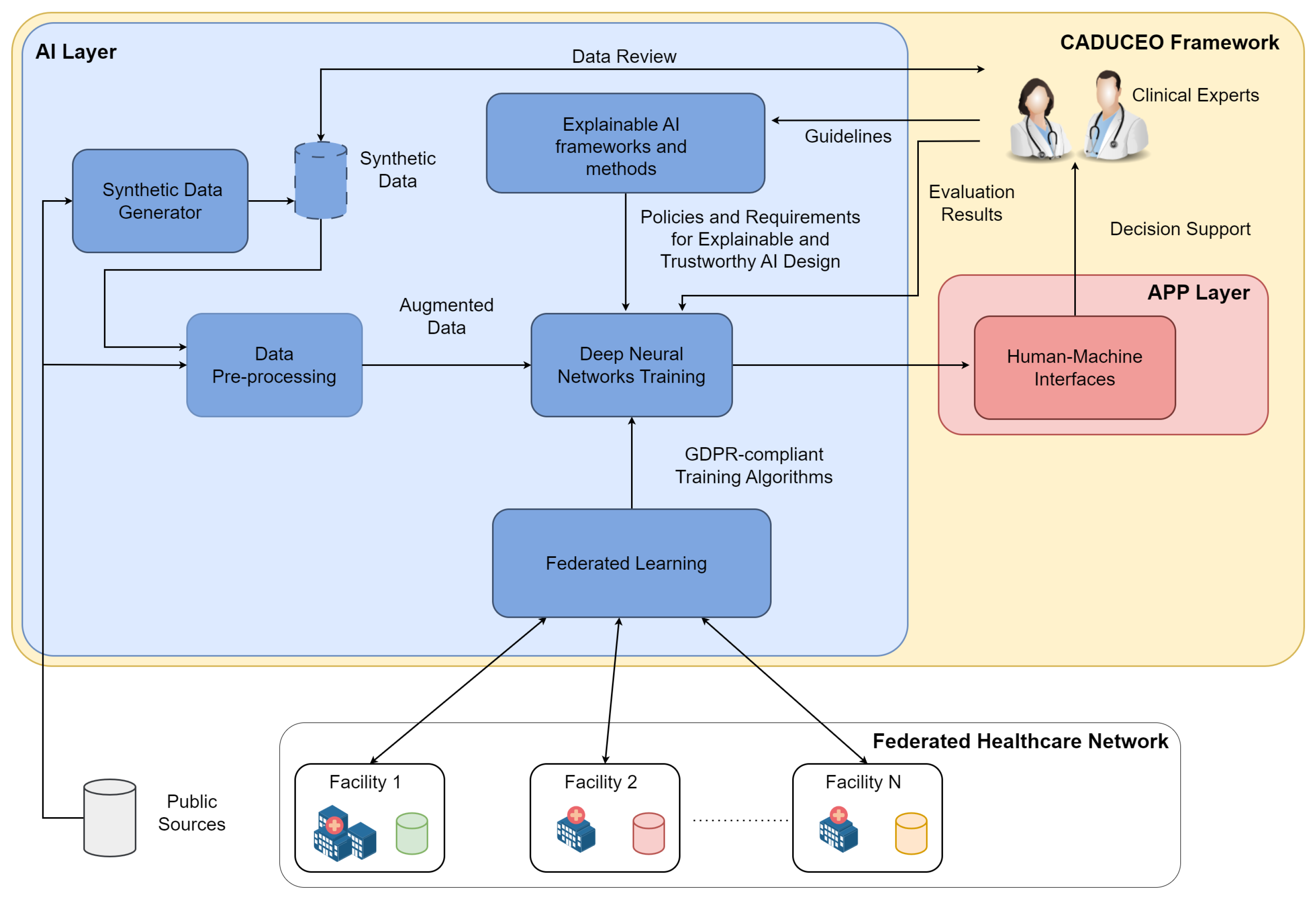
3. Results
- The data pre-processing system is a functional block that contains all the software solutions for data and clinical image enhancement. These solutions may employ both classical computer vision techniques, such as de-noising [51] and feature selection [52], and deep learning techniques, such as deep image inpainting [53], depending on the nature and characteristics of the data being processed. Data transformation and curation is one of the fundamental steps behind the design and implementation of any data-driven system and has to be designed in such a way that the integrity and correctness of the data to be analyzed are preserved.
- The synthetic data generator block gathers the functionalities related to the generation of synthetic, or artificial, data, whose function is to support and improve the training process of the AI functionalities. The role of synthetic data is of central importance in all applications where the availability (and quality) of real data is limited, as is common in the medical domain. Starting from publicly available data, as well as anonymized data samples coming from clinical facilities and deep learning solutions, including generative adversarial networks [54] and deep reinforcement learning [55], appropriately specialized on the specific clinical use cases, synthetic data will be generated. And it will undergo a review process by clinical experts to assess its quality to increase the trustworthiness of the overall trained system.
- Federated learning (FL) has been identified as an enabling technology for the distributed and decentralized training of the ML models underlying the platform’s AI capabilities. As the solutions of the CADUCEO framework are developed for clinical use cases, it is crucial to ensure their full compatibility with the GDPR and to observe in their design all appropriate precautions related to the protection of patient privacy. In this respect, FL makes it possible to ensure collaboration between different institutions, including prospective customers interested in improving certain functionalities, without the need for any exchange of clinical data. Integrating an FL system into the AI Layer thus ensures its maximum compatibility in terms of adoption and further developments.
- The explainable AI framework and methods functional block contains all the software features and guidelines to direct the design and development of AI solutions from the perspective of the explainability and interpretability of their results. Indeed, it is critical to the goals of the CADUCEO framework to offer clinical experts not only a set of recommendations and suggestions but to supplement them with adequate information critical to their correct interpretation.
- The training of machine learning models block, representing from a functional point of view the whole design process of the machine learning models (such as deep learning solutions), deals with the process of training them on the basis of available data (from project sources, public sources, and synthetic ones). The training of the models will take place under the constant supervision of clinical experts so that aspects related to the interpretability of the results and the quality of the proposed suggestions can be improved.
4. Discussion
4.1. Data Pre-Processing
- Missing data. It is often the case that, during data acquisition, some features could not be recorded for several records. In this case, we speak of missing data, which therefore cannot be used as is for data mining. There are several approaches to deal with this problem, first of all eliminating all instances that are stumped [59]. However, this method results in a substantial thinning of the dataset itself, resulting in worse performance when (for example) training a neural network. Other more refined methods, such as the ones employed in this work, make use of statistical procedures such as maximum likelihood to model each feature, as if it were a random variable, and then include an estimate of the missing numerical values. In this case, the drawback lies in the “goodness-of-fit” of the random model, which may even deviate greatly from the true value of the stumped data [60].
- Noise in the data. Even when there are no missing data, those that are present are often characterized by imperfections and inaccuracies, mainly due to measurement errors committed by hardware instruments or, especially in the clinical setting, by human operators. In these cases, it is possible to intervene by constructing noise filtering or data polishing algorithms [61], with the purpose of automatic noise detection and the consequent choice of intervention, which may reside in the modification of the data or, in extreme cases, its elimination.
- Feature selection. In every dataset, each record is characterized by countless features, which often turn out to be useless for the purpose and/or redundant. Feature selection algorithms allow features with high correlation to be eliminated, an aspect that results in lowering the risk of overfitting and speeding up data mining processes [62].
- Record selection or generation. Shifting the discussion from features to records, high correlation can also occur between data [63]. In the medical domain, for example, it may be the case that two different patients have very similar medical records, and as a result, for some machine learning or data mining applications, knowledge of only one clinical status may suffice. Conversely, it may be the case that a data acquisition campaign has collected too few or too little variety, so that manual augmentation of the dataset itself is necessary. Such augmentation techniques will be discussed at length in the next section.
- Class imbalance. In many supervised learning applications whose goal is classification, there is a significant difference among the absolute number of records of different classes in a classification problem. This situation is known as class imbalance [64]. The presence of class imbalance leads to a net imbalance toward the majority class and, as a result, there is a higher rate of misclassification for all other classes, which are in the minority. In these cases, pre-processing techniques can be used to resample the data to balance the class distribution. Within resampling, two main groups can be distinguished [65]: (i) undersampling, through which majority instances are removed to create a balance, and (ii) oversampling, which creates a superset of the original dataset by replicating some instances or creating new instances from existing ones.The application of data pre-processing techniques in the medical context improves clinical interpretation, exempts healthcare personnel from manual data cleaning, reduces computational costs, and improves the performance of predictors [66]. The study conducted in [67] found that the use of pre-processing is most common in critical disciplines of medicine, such as cardiology, endocrinology, and oncology. Regardless of the relevant clinical field, the most commonly used techniques are data reduction due to noise and/or the absence of data, and feature selection.
4.2. Data Augmentation
4.3. Federated Learning
- Non-IID (not independent and identically distributed), i.e., not representative of the statistical properties of the population identified for the problem under consideration;
- Unbalanced, from the point of view of numerosity, because the users do not behave in the same way;
- Highly distributed, because the number of devices participating in the training process may be greater than the total number of data available;
- Communication-limited, because they may not be accessible during training.
4.4. Use Cases
4.4.1. Eosinophilic Esophagitis (EoE)
4.4.2. Inflammatory Bowel Disease (IBD)
- Ulcerative Rectocolitis (UCR), characterized by an inflammatory process localized exclusively at the level of the colon, which starts in the rectum and may extend continuously to the cecum;
- Crohn’s disease (MC), characterized by inflammation that can affect the entire intestinal tract and involve all layers of the intestinal wall, leaving several healthy sections between the inflamed areas;
- Unclassified chronic inflammatory bowel disease, a term reserved for those cases in which anatomopathological features common to both UC and MC are present.
4.4.3. Portal Hypertension (PH)
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pantanowitz, L.; Quiroga-Garza, G.M.; Bien, L.; Heled, R.; Laifenfeld, D.; Linhart, C.; Sandbank, J.; Shach, A.A.; Shalev, V.; Vecsler, M.; et al. An artificial intelligence algorithm for prostate cancer diagnosis in whole slide images of core needle biopsies: A blinded clinical validation and deployment study. Lancet Digit. Health 2020, 2, e407–e416. [Google Scholar] [CrossRef]
- Smalley, E. AI-powered drug discovery captures pharma interest. Nat. Biotechnol. 2017, 35, 604–606. [Google Scholar] [PubMed]
- Gunduz, H. Deep learning-based Parkinson’s disease classification using vocal feature sets. IEEE Access 2019, 7, 115540–115551. [Google Scholar]
- Wen, Z.; Wang, S.; Yang, D.M.; Xie, Y.; Chen, M.; Bishop, J.; Xiao, G. Deep learning in digital pathology for personalized treatment plans of cancer patients. Semin. Diagn. Pathol. 2023, 40, 109–119. [Google Scholar]
- Wang, Y.; Nazir, S.; Shafiq, M. An overview on analyzing deep learning and transfer learning approaches for health monitoring. Comput. Math. Methods Med. 2021, 2021, 5552743. [Google Scholar]
- Budd, S.; Robinson, E.C.; Kainz, B. A survey on active learning and human-in-the-loop deep learning for medical image analysis. Med. Image Anal. 2021, 71, 102062. [Google Scholar] [CrossRef]
- European Parliament; Council of the European Union. Regulation (EU) 2016/679 of the European Parliament and of the Council. Regulation 2016, 679, 2016. [Google Scholar]
- McMahan, B.; Moore, E.; Ramage, D.; Hampson, S.; y Arcas, B.A. Communication-efficient learning of deep networks from decentralized data. Artif. Intell. Stat. 2017, 54, 1273–1282. [Google Scholar]
- Wang, L.; Zhang, X.; Su, H.; Zhu, J. A comprehensive survey of continual learning: Theory, method and application. arXiv 2023, arXiv:2302.00487. [Google Scholar]
- Xu, F.; Uszkoreit, H.; Du, Y.; Fan, W.; Zhao, D.; Zhu, J. Explainable AI: A brief survey on history, research areas, approaches and challenges. In Proceedings of the Natural Language Processing and Chinese Computing: 8th CCF International Conference, NLPCC 2019, Dunhuang, China, 9–14 October 2019; Proceedings, Part II 8. Springer: Berlin/Heidelberg, Germany, 2019; pp. 563–574. [Google Scholar]
- Selvaraju, R.R.; Das, A.; Vedantam, R.; Cogswell, M.; Parikh, D.; Batra, D. Grad-CAM: Why did you say that? arXiv 2016, arXiv:1611.07450. [Google Scholar]
- Ribeiro, M.T.; Singh, S.; Guestrin, C. Why Should I Trust You? In Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining, San Francisco, CA, USA, 13–17 August 2016; ACM: New York, NY, USA, 2016. [Google Scholar] [CrossRef]
- Agarwal, R.; Melnick, L.; Frosst, N.; Zhang, X.; Lengerich, B.; Caruana, R.; Hinton, G.E. Neural additive models: Interpretable machine learning with neural nets. Adv. Neural Inf. Process. Syst. 2021, 34, 4699–4711. [Google Scholar]
- Daniel, N.; Larey, A.; Aknin, E.; Osswald, G.A.; Caldwell, J.M.; Rochman, M.; Collins, M.H.; Yang, G.Y.; Arva, N.C.; Capocelli, K.E.; et al. A Deep Multi-Label Segmentation Network For Eosinophilic Esophagitis Whole Slide Biopsy Diagnostics. In Proceedings of the 2022 44th Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Glasgow, Scotland, UK, 11–15 July 2022. [Google Scholar] [CrossRef]
- Ghimire, K.; Lai, W.; Omar, Y.; Schwebke, T.; Vo, J. Machine Learning Approach to Distinguish Ulcerative Colitis and Crohn’s Disease Using SMOTE (Synthetic Minority Oversampling Technique) Methods. SMU Data Sci. Rev. 2021, 5, 9. [Google Scholar]
- Reiniš, J.; Petrenko, O.; Simbrunner, B.; Hofer, B.S.; Schepis, F.; Scoppettuolo, M.; Saltini, D.; Indulti, F.; Guasconi, T.; Albillos, A.; et al. Assessment of portal hypertension severity using machine learning models in patients with compensated cirrhosis. J. Hepatol. 2023, 78, 390–400. [Google Scholar] [CrossRef]
- Shorten, C.; Khoshgoftaar, T.M. A survey on image data augmentation for deep learning. J. Big Data 2019, 6, 1–48. [Google Scholar] [CrossRef]
- Bagherinezhad, H.; Horton, M.; Rastegari, M.; Farhadi, A. Label refinery: Improving imagenet classification through label progression. arXiv 2018, arXiv:1805.02641. [Google Scholar]
- Jurio, A.; Pagola, M.; Galar, M.; Lopez-Molina, C.; Paternain, D. A comparison study of different color spaces in clustering based image segmentation. In Proceedings of the International Conference on Information Processing and Management of Uncertainty in Knowledge-Based Systems, Dortmund, Germany, 28 June– 2 July 2010; Springer: Berlin/Heidelberg, Germany, 2010; pp. 532–541. [Google Scholar]
- Moreno-Barea, F.J.; Strazzera, F.; Jerez, J.M.; Urda, D.; Franco, L. Forward noise adjustment scheme for data augmentation. In Proceedings of the 2018 IEEE Symposium Series on Computational Intelligence (SSCI), Bangalore, India, 18–21 November 2018; pp. 728–734. [Google Scholar]
- Inoue, H. Data augmentation by pairing samples for images classification. arXiv 2018, arXiv:1801.02929. [Google Scholar]
- Zhong, Z.; Zheng, L.; Kang, G.; Li, S.; Yang, Y. Random erasing data augmentation. Proc. Aaai Conf. Artif. Intell. 2020, 34, 13001–13008. [Google Scholar]
- Abdollahi, B.; Tomita, N.; Hassanpour, S. Data augmentation in training deep learning models for medical image analysis. In Deep Learners and Deep Learner Descriptors for Medical Applications; Springer: Berlin/Heidelberg, Germany, 2020; pp. 167–180. [Google Scholar]
- Konno, T.; Iwazume, M. Icing on the cake: An easy and quick post-learnig method you can try after deep learning. arXiv 2018, arXiv:1807.06540. [Google Scholar]
- Zajac, M.; Zołna, K.; Rostamzadeh, N.; Pinheiro, P.O. Adversarial framing for image and video classification. Proc. Aaai Conf. Artif. Intell. 2019, 33, 10077–10078. [Google Scholar] [CrossRef]
- Goodfellow, I.; Pouget-Abadie, J.; Mirza, M.; Xu, B.; Warde-Farley, D.; Ozair, S.; Courville, A.; Bengio, Y. Generative adversarial networks. Commun. ACM 2020, 63, 139–144. [Google Scholar] [CrossRef]
- Lashgari, E.; Liang, D.; Maoz, U. Data augmentation for deep-learning-based electroencephalography. J. Neurosci. Methods 2020, 346, 108885. [Google Scholar]
- Nalepa, J.; Marcinkiewicz, M.; Kawulok, M. Data augmentation for brain-tumor segmentation: A review. Front. Comput. Neurosci. 2019, 13, 83. [Google Scholar] [CrossRef] [PubMed]
- Bissoto, A.; Valle, E.; Avila, S. Gan-based data augmentation and anonymization for skin-lesion analysis: A critical review. In Proceedings of the IEEE/CVF Conference on Computer Vision and Pattern Recognition, Nashville, TN, USA, 19–25 June 2021; pp. 1847–1856. [Google Scholar]
- Pezoulas, V.C.; Grigoriadis, G.I.; Gkois, G.; Tachos, N.S.; Smole, T.; Bosnić, Z.; Pičulin, M.; Olivotto, I.; Barlocco, F.; Robnik-Šikonja, M.; et al. A computational pipeline for data augmentation towards the improvement of disease classification and risk stratification models: A case study in two clinical domains. Comput. Biol. Med. 2021, 134, 104520. [Google Scholar]
- Hao, R.; Namdar, K.; Liu, L.; Haider, M.A.; Khalvati, F. A comprehensive study of data augmentation strategies for prostate cancer detection in diffusion-weighted MRI using convolutional neural networks. J. Digit. Imaging 2021, 34, 862–876. [Google Scholar] [CrossRef]
- Shijie, J.; Ping, W.; Peiyi, J.; Siping, H. Research on data augmentation for image classification based on convolution neural networks. In Proceedings of the 2017 Chinese Automation Congress (CAC), Jinan, China, 20–22 October 2017; pp. 4165–4170. [Google Scholar]
- Hospedales, T.; Antoniou, A.; Micaelli, P.; Storkey, A. Meta-learning in neural networks: A survey. IEEE Trans. Pattern Anal. Mach. Intell. 2021, 44, 5149–5169. [Google Scholar] [CrossRef] [PubMed]
- Zoph, B.; Le, Q.V. Neural architecture search with reinforcement learning. arXiv 2016, arXiv:1611.01578. [Google Scholar]
- Lemley, J.; Bazrafkan, S.; Corcoran, P. Smart augmentation learning an optimal data augmentation strategy. IEEE Access 2017, 5, 5858–5869. [Google Scholar] [CrossRef]
- Wang, J.; Perez, L. The effectiveness of data augmentation in image classification using deep learning. Convolutional Neural Netw. Vis. Recognit 2017, 11, 1–8. [Google Scholar]
- Nguyen, D.C.; Pham, Q.V.; Pathirana, P.N.; Ding, M.; Seneviratne, A.; Lin, Z.; Dobre, O.A.; Hwang, W.J. Federated learning for smart healthcare: A survey. ACM Comput. Surv. (CSUR) 2022, 55, 1–37. [Google Scholar]
- Gunter, T.D.; Terry, N.P. The emergence of national electronic health record architectures in the United States and Australia: Models, costs, and questions. J. Med. Internet Res. 2005, 7, e3. [Google Scholar] [CrossRef]
- Hao, M.; Li, H.; Xu, G.; Liu, Z.; Chen, Z. Privacy-aware and resource-saving collaborative learning for healthcare in cloud computing. In Proceedings of the IEEE International Conference on Communications (ICC), Dublin, Ireland, 7–11 June 2020; pp. 1–6. [Google Scholar]
- Vepakomma, P.; Gupta, O.; Swedish, T.; Raskar, R. Split learning for health: Distributed deep learning without sharing raw patient data. arXiv 2018, arXiv:1812.00564. [Google Scholar]
- Brisimi, T.S.; Chen, R.; Mela, T.; Olshevsky, A.; Paschalidis, I.C.; Shi, W. Federated learning of predictive models from federated electronic health records. Int. J. Med. Inform. 2018, 112, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, O.; Gkoulalas-Divanis, A.; Salonidis, T.; Sylla, I.; Park, Y.; Hsu, G.; Das, A. Differential privacy-enabled federated learning for sensitive health data. arXiv 2019, arXiv:1910.02578. [Google Scholar]
- Wei, K.; Li, J.; Ding, M.; Ma, C.; Yang, H.H.; Farhad, F.; Jin, S.; Quek, T.Q.S.; Poor, H.V. Federated learning with differential privacy: Algorithms and performance analysis. IEEE Trans. Inf. Forensics Secur. 2020, 15, 3454–3469. [Google Scholar]
- Liu, D.; Miller, T.; Sayeed, R.; Mandl, K.D. FADL: Federated-autonomous deep learning for distributed electronic health record. arXiv 2018, arXiv:1811.11400. [Google Scholar]
- Shao, R.; He, H.; Liu, H.; Liu, D. Stochastic channel-based federated learning for medical data privacy preserving. arXiv 2019, arXiv:1910.11160. [Google Scholar]
- Arrieta, A.B.; Díaz-Rodríguez, N.; Ser, J.D.; Bennetot, A.; Tabik, S.; Barbado, A.; Garcia, S.; Gil-Lopez, S.; Molina, D.; Benjamins, R.; et al. Explainable Artificial Intelligence (XAI): Concepts, taxonomies, opportunities and challenges toward responsible AI. Inf. Fusion 2020, 58, 82–115. [Google Scholar] [CrossRef]
- Zhang, Y.; Weng, Y.; Lund, J. Applications of Explainable Artificial Intelligence in Diagnosis and Surgery. Diagnostics 2022, 12, 237A. [Google Scholar]
- Adadi, A.; Berrada, M. Peeking Inside the Black-Box: A Survey on Explainable Artificial Intelligence (XAI). IEEE Access 2018, 6, 52138–52160. [Google Scholar] [CrossRef]
- Islam, S.R.; Eberle, W.; Ghafoor, S.K.; Ahmed, M. Explainable artificial intelligence approaches: A survey. arXiv 2021, arXiv:2101.09429. [Google Scholar]
- Tjoa, E.; Guan, C. A Survey on Explainable Artificial Intelligence (XAI): Toward Medical XAI. IEEE Trans. Neural Netw. Learn. Syst. 2020, 32, 4793–4813. [Google Scholar] [CrossRef] [PubMed]
- Kaur, R.; Kaur, R. Survey of de-noising methods using filters and fast wavelet transform. Int. J. Adv. Res. Comput. Sci. Softw. Eng 2013, 3, 133–136. [Google Scholar]
- Miao, J.; Niu, L. A survey on feature selection. Procedia Comput. Sci. 2016, 91, 919–926. [Google Scholar]
- Xiang, H.; Zou, Q.; Nawaz, M.A.; Huang, X.; Zhang, F.; Yu, H. Deep learning for image inpainting: A survey. Pattern Recognit. 2023, 134, 109046. [Google Scholar] [CrossRef]
- Goodfellow, I.; Pouget-Abadie, J.; Mirza, M.; Xu, B.; Warde-Farley, D.; Ozair, S.; Courville, A.; Bengio, Y. Generative adversarial nets. Adv. Neural Inf. Process. Syst. 2014, 27–32. [Google Scholar]
- Arulkumaran, K.; Deisenroth, M.P.; Brundage, M.; Bharath, A.A. A brief survey of deep reinforcement learning. arXiv 2017, arXiv:1708.05866. [Google Scholar] [CrossRef]
- Seifert, J.W. Data mining: An overview. Natl. Secur. 2004, 201–217. [Google Scholar]
- García, S.; Ramírez-Gallego, S.; Luengo, J.; Benítez, J.M.; Herrera, F. Big data preprocessing: Methods and prospects. Big Data Anal. 2016, 1, 9. [Google Scholar] [CrossRef]
- Pyle, D. Data Preparation for Data Mining; Morgan Kaufmann: Burlington, MA, USA, 1999. [Google Scholar]
- Little, R.J.; Rubin, D.B. Statistical Analysis with Missing Data; John Wiley and Sons: Hoboken, NJ, USA, 2019; Volume 793. [Google Scholar]
- Luengo, J.; García, S.; Herrera, F. On the choice of the best imputation methods for missing values considering three groups of classification methods. Knowl. Inf. Syst. 2011, 32, 77–108. [Google Scholar] [CrossRef]
- Zhu, X.; Wu, X. Class Noise vs. Attribute Noise: A Quantitative Study. Artif. Intell. Rev. 2004, 22, 177–210. [Google Scholar] [CrossRef]
- Hall, M.A. Correlation-Based Feature Selection for Machine Learning. Ph.D. Thesis, The University of Waikato, Hamilton, New Zealand, 1999. [Google Scholar]
- Liu, H.; Motoda, H. On issues of instance selection. Data Min. Knowl. Discov. 2002, 6, 115. [Google Scholar]
- López, V.; Fernández, A.; García, S.; Palade, V.; Herrera, F. An insight into classification with imbalanced data: Empirical results and current trends on using data intrinsic characteristics. Inf. Sci. 2013, 250, 113–141. [Google Scholar] [CrossRef]
- Mohammed, R.; Rawashdeh, J.; Abdullah, M. Machine Learning with Oversampling and Undersampling Techniques: Overview Study and Experimental Results. In Proceedings of the 2020 11th International Conference on Information and Communication Systems (ICICS), Irbid, Jordan, 7–9 April 2020. [Google Scholar] [CrossRef]
- Mendes, D.; Paredes, S.; Rocha, T.; Carvalho, P.; Henriques, J.; Cabiddu, R.; Morais, J. Assessment of cardiovascular risk based on a data-driven knowledge discovery approach. In Proceedings of the 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milan, Italy, 25–29 August 2015. [Google Scholar] [CrossRef]
- Idri, A.; Benhar, H.; Fernández-Alemán, J.; Kadi, I. A systematic map of medical data preprocessing in knowledge discovery. Comput. Methods Programs Biomed. 2018, 162, 69–85. [Google Scholar] [CrossRef] [PubMed]
- Litjens, G.; Kooi, T.; Bejnordi, B.E.; Setio, A.A.A.; Ciompi, F.; Ghafoorian, M.; van der Laak, J.A.; van Ginneken, B.; Sánchez, C.I. A survey on deep learning in medical image analysis. Med. Image Anal. 2017, 42, 60–88. [Google Scholar] [CrossRef]
- Ying, X. An Overview of Overfitting and its Solutions. J. Phys. Conf. Ser. 2019, 1168, 022022. [Google Scholar] [CrossRef]
- Chlap, P.; Min, H.; Vandenberg, N.; Dowling, J.; Holloway, L.; Haworth, A. A review of medical image data augmentation techniques for deep learning applications. J. Med. Imaging Radiat. Oncol. 2021, 65, 545–563. [Google Scholar] [CrossRef]
- Sutton, R.; Barto, A. Reinforcement Learning: An Introduction. IEEE Trans. Neural Netw. 1998, 9, 1054. [Google Scholar] [CrossRef]
- Cubuk, E.D.; Zoph, B.; Mane, D.; Vasudevan, V.; Le, Q.V. AutoAugment: Learning Augmentation Policies from Data. arXiv 2018, arXiv:1805.09501. [Google Scholar]
- Minh, T.N.; Sinn, M.; Lam, H.T.; Wistuba, M. Automated Image Data Preprocessing with Deep Reinforcement Learning. arXiv 2018, arXiv:1806.05886. [Google Scholar]
- Hu, M.; Zhang, J.; Matkovic, L.; Liu, T.; Yang, X. Reinforcement learning in medical image analysis: Concepts, applications, challenges, and future directions. J. Appl. Clin. Med. Phys. 2023, 24, e13898. [Google Scholar] [CrossRef]
- Hochreiter, S. The Vanishing Gradient Problem During Learning Recurrent Neural Nets and Problem Solutions. Int. J. Uncertain. Fuzziness Knowl.-Based Syst. 1998, 6, 107–116. [Google Scholar] [CrossRef]
- Zhou, S.K.; Le, H.N.; Luu, K.; Nguyen, H.V.; Ayache, N. Deep reinforcement learning in medical imaging: A literature review. Med. Image Anal. 2021, 73, 102193. [Google Scholar] [CrossRef]
- Hard, A.; Rao, K.; Mathews, R.; Ramaswamy, S.; Beaufays, F.; Augenstein, S.; Eichner, H.; Kiddon, C.; Ramage, D. Federated Learning for Mobile Keyboard Prediction. arXiv 2018, arXiv:1811.03604. [Google Scholar]
- Sheller, M.J.; Edwards, B.; Reina, G.A.; Martin, J.; Pati, S.; Kotrotsou, A.; Milchenko, M.; Xu, W.; Marcus, D.; Colen, R.R.; et al. Federated learning in medicine: Facilitating multi-institutional collaborations without sharing patient data. Sci. Rep. 2020, 10, 12598. [Google Scholar] [CrossRef]
- Viorescu, R. 2018 Reform Of Eu Data Protection Rules. Eur. J. Law Public Adm. 2017, 4, 27–39. [Google Scholar] [CrossRef]
- Huang, L.; Shea, A.L.; Qian, H.; Masurkar, A.; Deng, H.; Liu, D. Patient clustering improves efficiency of federated machine learning to predict mortality and hospital stay time using distributed electronic medical records. J. Biomed. Inform. 2019, 99, 103291. [Google Scholar] [CrossRef]
- Silva, S.; Gutman, B.A.; Romero, E.; Thompson, P.M.; Altmann, A.; Lorenzi, M. Federated Learning in Distributed Medical Databases: Meta-Analysis of Large-Scale Subcortical Brain Data. In Proceedings of the 2019 IEEE 16th International Symposium on Biomedical Imaging (ISBI 2019), Venice, Italy, 8–11 April 2019. [Google Scholar] [CrossRef]
- Barstugan, M.; Ozkaya, U.; Ozturk, S. Coronavirus (COVID-19) Classification using CT Images by Machine Learning Methods. arXiv 2020, arXiv:2003.09424. [Google Scholar]
- Gozes, O.; Frid-Adar, M.; Greenspan, H.; Browning, P.D.; Zhang, H.; Ji, W.; Bernheim, A.; Siegel, E. Rapid ai development cycle for the coronavirus (COVID-19) pandemic: Initial results for automated detection & patient monitoring using deep learning ct image analysis. arXiv 2020, arXiv:2003.05037. [Google Scholar]
- Wang, L.; Lin, Z.Q.; Wong, A. COVID-Net: A tailored deep convolutional neural network design for detection of COVID-19 cases from chest X-ray images. Sci. Rep. 2020, 10, 19549. [Google Scholar] [CrossRef]
- Li, T.; Sahu, A.K.; Zaheer, M.; Sanjabi, M.; Talwalkar, A.; Smith, V. Federated optimization in heterogeneous networks. Proc. Mach. Learn. Syst. 2020, 2, 429–450. [Google Scholar]
- Giuseppi, A.; Torre, L.D.; Menegatti, D.; Pietrabissa, A. AdaFed: Performance-based Adaptive Federated Learning. In Proceedings of the 2021 the 5th International Conference on Advances in Artificial Intelligence (ICAAI), Virtual, 20–22 November 2021; ACM: New York, NY, USA, 2021. [Google Scholar] [CrossRef]
- Chu, W.; Xie, C.; Wang, B.; Li, L.; Yin, L.; Zhao, H.; Li, B. FOCUS: Fairness via Agent-Awareness for Federated Learning on Heterogeneous Data. arXiv 2022, arXiv:2207.10265. [Google Scholar]
- Roth, A.E. Introduction to the Shapley value. In The Shapley Value; Cambridge University Press: Cambridge, UK, 1988; pp. 1–28. [Google Scholar] [CrossRef]
- Giuseppi, A.; Manfredi, S.; Menegatti, D.; Pietrabissa, A.; Poli, C. Decentralized Federated Learning for Nonintrusive Load Monitoring in Smart Energy Communities. In Proceedings of the 2022 30th Mediterranean Conference on Control and Automation (MED), Vouliagmeni, Greece, 28 June 2022–1 July 2022. [Google Scholar] [CrossRef]
- Savazzi, S.; Nicoli, M.; Rampa, V. Federated Learning with Cooperating Devices: A Consensus Approach for Massive IoT Networks. IEEE Internet Things J. 2020, 7, 4641–4654. [Google Scholar] [CrossRef]
- Giuseppi, A.; Manfredi, S.; Pietrabissa, A. A Weighted Average Consensus Approach for Decentralized Federated Learning. Mach. Intell. Res. 2022, 19, 319–330. [Google Scholar] [CrossRef]
- Ahuja, N.; Weedon, J.; Schwarz, S.M.; Sklar, R.; Rabinowitz, S.S. Applying the Eosinophilic Esophagitis Endoscopic Reference Scores (EREFS) to Different Aged Children. J. Pediatr. Gastroenterol. Nutr. 2020, 71, 328–332. [Google Scholar] [CrossRef] [PubMed]
- Jain, J.; Zhou, Y.; Yu, N.; Shi, H. Keys to Better Image Inpainting: Structure and Texture Go Hand in Hand. In Proceedings of the WACV, Waikoloa, HI, USA, 2–7 January 2023. [Google Scholar]
- Mirza, M.; Osindero, S. Conditional generative adversarial nets. arXiv 2014, arXiv:1411.1784. [Google Scholar]
- Redmon, J.; Divvala, S.; Girshick, R.; Farhadi, A. You only look once: Unified, real-time object detection. In Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition, Las Vegas, NV, USA, 27–30 June 2016; pp. 779–788. [Google Scholar]
- Lin, E.; Chen, Q.; Qi, X. Deep reinforcement learning for imbalanced classification. Appl. Intell. 2020, 50, 2488–2502. [Google Scholar] [CrossRef]
- Jauregui-Amezaga, A.; Geerits, A.; Das, Y.; Lemmens, B.; Sagaert, X.; Bessissow, T.; Lobatón, T.; Ferrante, M.; Assche, G.V.; Bisschops, R.; et al. A Simplified Geboes Score for Ulcerative Colitis. J. Crohn’s Colitis 2016, 11, 305–313. [Google Scholar] [CrossRef]
- Peyrin-Biroulet, L.; Arenson, E.; Rubin, D.T.; Siegel, C.A.; Lee, S.; Laroux, F.S.; Zhou, W.; Finney-Hayward, T.; Gonzalez, Y.S.; Shields, A.L. A Comparative Evaluation of the Measurement Properties of Three Histological Indices of Mucosal Healing in Ulcerative Colitis: Geboes Score, Robarts Histopathology Index and Nancy Index. J. Crohn’s Colitis 2023, 1, jjad087. [Google Scholar] [CrossRef]
- Naegeli, A.N.; Hunter, T.; Dong, Y.; Hoskin, B.; Middleton-Dalby, C.; Hetherington, J.; Stefani-Hunyady, D.; Canavan, J.B. Full, Partial, and Modified Permutations of the Mayo Score: Characterizing Clinical and Patient-Reported Outcomes in Ulcerative Colitis Patients. Crohn’s Colitis 360 2021, 3, otab007. [Google Scholar] [CrossRef]
- Ikeya, K.; Hanai, H.; Sugimoto, K.; Osawa, S.; Kawasaki, S.; Iida, T.; Maruyama, Y.; Watanabe, F. The Ulcerative Colitis Endoscopic Index of Severity More Accurately Reflects Clinical Outcomes and Long-term Prognosis than the Mayo Endoscopic Score. J. Crohn’s Colitis 2015, 10, 286–295. [Google Scholar] [CrossRef]
- Best, W.R. Predicting the Crohn’s disease activity index from the harvey-bradshaw index. Inflamm. Bowel Dis. 2006, 12, 304–310. [Google Scholar] [CrossRef]
- Koutroumpakis, E.; Katsanos, K. Implementation of the simple endoscopic activity score in crohn’s disease. Saudi J. Gastroenterol. 2016, 22, 183. [Google Scholar] [CrossRef]
- Karras, T.; Laine, S.; Aittala, M.; Hellsten, J.; Lehtinen, J.; Aila, T. Analyzing and improving the image quality of stylegan. In Proceedings of the IEEE/CVF Conference on Computer Vision and Pattern Recognition, Seattle, WA, USA, 13–19 June 2020; pp. 8110–8119. [Google Scholar]
- Lathuilière, S.; Mesejo, P.; Alameda-Pineda, X.; Horaud, R. A comprehensive analysis of deep regression. IEEE Trans. Pattern Anal. Mach. Intell. 2019, 42, 2065–2081. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Menegatti, D.; Giuseppi, A.; Delli Priscoli, F.; Pietrabissa, A.; Di Giorgio, A.; Baldisseri, F.; Mattioni, M.; Monaco, S.; Lanari, L.; Panfili, M.; et al. CADUCEO: A Platform to Support Federated Healthcare Facilities through Artificial Intelligence. Healthcare 2023, 11, 2199. https://doi.org/10.3390/healthcare11152199
Menegatti D, Giuseppi A, Delli Priscoli F, Pietrabissa A, Di Giorgio A, Baldisseri F, Mattioni M, Monaco S, Lanari L, Panfili M, et al. CADUCEO: A Platform to Support Federated Healthcare Facilities through Artificial Intelligence. Healthcare. 2023; 11(15):2199. https://doi.org/10.3390/healthcare11152199
Chicago/Turabian StyleMenegatti, Danilo, Alessandro Giuseppi, Francesco Delli Priscoli, Antonio Pietrabissa, Alessandro Di Giorgio, Federico Baldisseri, Mattia Mattioni, Salvatore Monaco, Leonardo Lanari, Martina Panfili, and et al. 2023. "CADUCEO: A Platform to Support Federated Healthcare Facilities through Artificial Intelligence" Healthcare 11, no. 15: 2199. https://doi.org/10.3390/healthcare11152199
APA StyleMenegatti, D., Giuseppi, A., Delli Priscoli, F., Pietrabissa, A., Di Giorgio, A., Baldisseri, F., Mattioni, M., Monaco, S., Lanari, L., Panfili, M., & Suraci, V. (2023). CADUCEO: A Platform to Support Federated Healthcare Facilities through Artificial Intelligence. Healthcare, 11(15), 2199. https://doi.org/10.3390/healthcare11152199







