Association between Quality of Life and Physical Functioning in a Gardening Intervention for Cancer Survivors
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Gardening Intervention
2.3. Outcomes and Measures:
Sociodemographic and Health Data
2.4. Data Analysis
3. Results
4. Discussion
COVID-19 and Cancer Survivorship
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- American Cancer Society. Cancer-Treatment-and-Survivorship-Facts-and-Figures-2019–2021. Available online: https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/cancer-treatment-and-survivorship-facts-and-figures/cancer-treatment-and-survivorship-facts-and-figures-2019-2021.pdf (accessed on 18 February 2022).
- Holmes, H.M.; Nguyen, H.T.; Nayak, P.; Oh, J.H.; Escalante, C.P.; Elting, L.S. Chronic conditions and health status in older cancer survivors. Eur. J. Intern. Med. 2014, 25, 374–378. [Google Scholar] [CrossRef] [PubMed]
- Mohile, S.G.; Fan, L.; Reeve, E.; Jean-Pierre, P.; Mustian, K.; Peppone, L.; Janelsins, M.; Morrow, G.; Hall, W.; Dale, W. Association of Cancer With Geriatric Syndromes in Older Medicare Beneficiaries. J. Clin. Oncol. 2011, 29, 1458–1464. [Google Scholar] [CrossRef] [PubMed]
- Stafford, R.S.; Cyr, P.L. The impact of cancer on the physical function of the elderly and their utilization of health care. Cancer 1997, 80, 1973–1980. [Google Scholar] [CrossRef]
- Chou, C.-H.; Hwang, C.-L.; Wu, Y.-T. Effect of Exercise on Physical Function, Daily Living Activities, and Quality of Life in the Frail Older Adults: A Meta-Analysis. Arch. Phys. Med. Rehabil. 2012, 93, 237–244. [Google Scholar] [CrossRef]
- Stopeck, A.T.; Brown-Glaberman, U. Impact of denosumab on bone mass in cancer patients. Clin. Pharmacol. Adv. Appl. 2013, 5, 117–129. [Google Scholar] [CrossRef][Green Version]
- Ha, D.; Ries, A.L.; Lippman, S.M.; Fuster, M.M. Effects of curative-intent lung cancer therapy on functional exercise capacity and patient-reported outcomes. Support. Care Cancer 2020, 28, 4707–4720. [Google Scholar] [CrossRef]
- Silbermann, R.; Roodman, G.D. Bone effects of cancer therapies. Curr. Opin. Support. Palliat. Care 2011, 5, 251–257. [Google Scholar] [CrossRef]
- Blanchard, C.M.; Courneya, K.S.; Stein, K. Cancer Survivors’ Adherence to Lifestyle Behavior Recommendations and Associations With Health-Related Quality of Life: Results From the American Cancer Society′s SCS-II. J. Clin. Oncol. 2008, 26, 2198–2204. [Google Scholar] [CrossRef]
- Zhang, F.F.; Liu, S.; John, E.M.; Must, A.; Demark-Wahnefried, W. Diet quality of cancer survivors and noncancer individuals: Results from a national survey. Cancer 2015, 121, 4212–4221. [Google Scholar] [CrossRef]
- Chalmin-Pui, L.S.; Griffiths, A.; Roe, J.; Heaton, T.; Cameron, R. Why garden?—Attitudes and the perceived health benefits of home gardening. Cities 2021, 112, 103118. [Google Scholar] [CrossRef]
- Blair, C.; Madan-Swain, A.; Locher, J.L.; Desmond, R.A.; Santos, J.D.L.; Affuso, O.; Glover, T.; Smith, K.; Carley, J.; Lipsitz, M.; et al. Harvest for health gardening intervention feasibility study in cancer survivors. Acta Oncol. 2012, 52, 1110–1118. [Google Scholar] [CrossRef]
- Park, S.-A.; Shoemaker, C.; Haub, M. Can Older Gardeners Meet the Physical Activity Recommendation through Gardening? HortTechnology 2008, 18, 639–643. [Google Scholar] [CrossRef]
- Sommerfeld, A.J.; McFarland, A.L.; Waliczek, T.M.; Zajicek, J.M. Growing Minds: Evaluating the Relationship between Gardening and Fruit and Vegetable Consumption in Older Adults. HortTechnology 2010, 20, 711–717. [Google Scholar] [CrossRef]
- Zick, C.D.; Smith, K.R.; Kowaleski-Jones, L.; Uno, C.; Merrill, B.J. Harvesting More Than Vegetables: The Potential Weight Control Benefits of Community Gardening. Am. J. Public Health 2013, 103, 1110–1115. [Google Scholar] [CrossRef]
- Ribeiro, A.I.; Triguero-Mas, M.; Santos, C.J.; Gómez-Nieto, A.; Cole, H.; Anguelovski, I.; Silva, F.M.; Baró, F. Exposure to nature and mental health outcomes during COVID-19 lockdown. A comparison between Portugal and Spain. Environ. Int. 2021, 154, 106664. [Google Scholar] [CrossRef]
- Scott, T.L.; Masser, B.M.; Pachana, N.A. Exploring the health and wellbeing benefits of gardening for older adults. Ageing Soc. 2014, 35, 2176–2200. [Google Scholar] [CrossRef]
- Bail, J.R.; Frugé, A.D.; Cases, M.G.; Santos, J.F.D.L.; Locher, J.L.; Ms, K.P.S.; Cantor, A.B.; Cohen, H.J.; Demark-Wahnefried, W. A home-based mentored vegetable gardening intervention demonstrates feasibility and improvements in physical activity and performance among breast cancer survivors. Cancer 2018, 124, 3427–3435. [Google Scholar] [CrossRef]
- Demark-Wahnefried, W.; Cases, M.G.; Cantor, A.B.; Frugé, A.D.; Smith, K.P.; Locher, J.; Cohen, H.J.; Tsuruta, Y.; Daniel, M.; Kala, R.; et al. Pilot Randomized Controlled Trial of a Home Vegetable Gardening Intervention among Older Cancer Survivors Shows Feasibility, Satisfaction, and Promise in Improving Vegetable and Fruit Consumption, Reassurance of Worth, and the Trajectory of Central Adiposity. J. Acad. Nutr. Diet. 2018, 118, 689–704. [Google Scholar] [CrossRef]
- Cases, M.G.; Frugé, A.D.; Santos, J.F.D.L.; Locher, J.L.; Cantor, A.B.; Smith, K.P.; Glover, T.A.; Cohen, H.J.; Daniel, M.; Morrow, C.D.; et al. Detailed methods of two home-based vegetable gardening intervention trials to improve diet, physical activity, and quality of life in two different populations of cancer survivors. Contemp. Clin. Trials 2016, 50, 201–212. [Google Scholar] [CrossRef]
- Blair, C.; Adsul, P.; Guest, D.; Sussman, A.; Cook, L.; Harding, E.; Rodman, J.; Duff, D.; Burgess, E.; Quezada, K.; et al. Southwest Harvest for Health: An Adapted Mentored Vegetable Gardening Intervention for Cancer Survivors. Nutrients 2021, 13, 2319. [Google Scholar] [CrossRef]
- Blair, C.K.; Harding, E.M.; Adsul, P.; Moran, S.; Guest, D.; Clough, K.; Sussman, A.L.; Duff, D.; Cook, L.S.; Rodman, J.; et al. Southwest Harvest for Health: Adapting a mentored vegetable gardening intervention for cancer survivors in the southwest. Contemp. Clin. Trials Commun. 2021, 21, 100741. [Google Scholar] [CrossRef] [PubMed]
- National Institute of Food and Agriculture. Extension. Available online: https://nifa.usda.gov/about-nifa/how-we-work/extension (accessed on 18 February 2022).
- Extension Master Gardener National Committee. Extension Master Gardener. Available online: https://mastergardener.extension.org/ (accessed on 18 February 2022).
- Health Measures. Intro to PROMIS. Available online: https://www.healthmeasures.net/explore-measurement-systems/promis/intro-to-promis (accessed on 27 March 2022).
- Cella, D.; Choi, S.W.; Condon, D.M.; Schalet, B.; Hays, R.D.; Rothrock, N.E.; Yount, S.; Cook, K.F.; Gershon, R.C.; Amtmann, D.; et al. PROMIS® Adult Health Profiles: Efficient Short-Form Measures of Seven Health Domains. Value Health 2019, 22, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Cook, K.F.; Jensen, S.E.; Schalet, B.D.; Beaumont, J.L.; Amtmann, D.; Czajkowski, S.; Dewalt, D.A.; Fries, J.F.; Pilkonis, P.A.; Reeve, B.B.; et al. PROMIS measures of pain, fatigue, negative affect, physical function, and social function demonstrated clinical validity across a range of chronic conditions. J. Clin. Epidemiol. 2016, 73, 89–102. [Google Scholar] [CrossRef] [PubMed]
- Schalet, B.D.; Hays, R.D.; Jensen, S.E.; Beaumont, J.L.; Fries, J.F.; Cella, D. Validity of PROMIS physical function measured in diverse clinical samples. J. Clin. Epidemiol. 2016, 73, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Schalet, B.D.; Pilkonis, P.A.; Yu, L.; Dodds, N.; Johnston, K.L.; Yount, S.; Riley, W.; Cella, D. Clinical validity of PROMIS Depression, Anxiety, and Anger across diverse clinical samples. J. Clin. Epidemiol. 2016, 73, 119–127. [Google Scholar] [CrossRef]
- Chastin, S.; Granat, M. Methods for objective measure, quantification and analysis of sedentary behaviour and inactivity. Gait Posture 2010, 31, 82–86. [Google Scholar] [CrossRef]
- Grant, P.M.; Ryan, C.G.; Tigbe, W.; Granat, M.H. The validation of a novel activity monitor in the measurement of posture and motion during everyday activities. Br. J. Sports Med. 2006, 40, 992–997. [Google Scholar] [CrossRef]
- Keadle, S.; Libertine, A.; Lyden, K.; Staudenmayer, J.; Freedson, P.S. Validation of Wearable Monitors for Assessing Sedentary Behavior. Med. Sci. Sports Exerc. 2011, 43, 1561–1567. [Google Scholar] [CrossRef]
- Sellers, C.; Dall, P.; Grant, M.; Stansfield, B. Validity and reliability of the activPAL3 for measuring posture and stepping in adults and young people. Gait Posture 2016, 43, 42–47. [Google Scholar] [CrossRef]
- Amireault, S.; Godin, G. The Godin-Shephard Leisure-Time Physical Activity Questionnaire: Validity Evidence Supporting its Use for Classifying Healthy Adults into Active and Insufficiently Active Categories. Percept. Mot. Ski. 2015, 120, 604–622. [Google Scholar] [CrossRef]
- Amireault, S.; Godin, G.; Lacombe, J.; Sabiston, C.M. Validation of the Godin-Shephard Leisure-Time Physical Activity Questionnaire classification coding system using accelerometer assessment among breast cancer survivors. J. Cancer Surviv. 2015, 9, 532–540. [Google Scholar] [CrossRef]
- Shaw, J.D.; McEntarfer, R.; Ferrel, J.; Greene, N.; Presson, A.P.; Zhang, C.; Lawrence, B.D.; Spiker, W.R.; Spina, N.; Brodke, D.S. What Does Your PROMIS Score Mean? Improving the Utility of Patient-Reported Outcomes at the Point of Care. Glob. Spine J. 2020, 12, 588–597. [Google Scholar] [CrossRef]
- Van den Beuken-van Everdingen, M.H.J.; Hochstenbach, L.M.J.; Joosten, E.A.J.; Tjan-Heijnen, V.C.G.; Janssen, D.J.A. Update on Prevalence of Pain in Patients With Cancer: Systematic Review and Meta-Analysis. J. Pain Symptom Manag. 2016, 51, 1070–1090.e9. [Google Scholar] [CrossRef]
- Muhandiramge, J.; Orchard, S.G.; Warner, E.T.; van Londen, G.J.; Zalcberg, J.R. Functional Decline in the Cancer Patient: A Review. Cancers 2022, 14, 1368. [Google Scholar] [CrossRef]
- Carroll, J.E.; Bower, J.E.; Ganz, P.A. Cancer-related accelerated ageing and biobehavioural modifiers: A framework for research and clinical care. Nat. Rev. Clin. Oncol. 2021, 19, 173–187. [Google Scholar] [CrossRef]
- Schmitz, K.H.; Cappola, A.R.; Stricker, C.T.; Sweeney, C.; Norman, S.A. The Intersection of Cancer and Aging: Establishing the Need for Breast Cancer Rehabilitation. Cancer Epidemiol. Biomark. Prev. 2007, 16, 866–872. [Google Scholar] [CrossRef]
- Schmidt, A.L.; Bakouny, Z.; Bhalla, S.; Steinharter, J.A.; Tremblay, D.A.; Awad, M.M.; Kessler, A.J.; Haddad, R.I.; Evans, M.; Busser, F.; et al. Cancer Care Disparities during the COVID-19 Pandemic: COVID-19 and Cancer Outcomes Study. Cancer Cell 2020, 38, 769–770. [Google Scholar] [CrossRef]
- Fiorentino, L.; Rissling, M.; Liu, L.; Ancoli-Israel, S. The symptom cluster of sleep, fatigue and depressive symptoms in breast cancer patients: Severity of the problem and treatment options. Drug Discov. Today Dis. Models 2012, 8, 167–173. [Google Scholar] [CrossRef]
- Loh, K.P.; Zittel, J.; Kadambi, S.; Pandya, C.; Xu, H.; Flannery, M.; Magnuson, A.; Bautista, J.; McHugh, C.; Mustian, K.; et al. Elucidating the associations between sleep disturbance and depression, fatigue, and pain in older adults with cancer. J. Geriatr. Oncol. 2018, 9, 464–468. [Google Scholar] [CrossRef]
- Theobald, D.E. Cancer pain, fatigue, distress, and insomnia in cancer patients. Clin. Cornerstone 2004, 6, S15–S21. [Google Scholar] [CrossRef]
- Mock, V.; Atkinson, A.; Barsevick, A.; Cella, D.; Cimprich, B.; Cleeland, C.; Donnelly, J.; A Eisenberger, M.; Escalante, C.; Hinds, P.; et al. NCCN Practice Guidelines for Cancer-Related Fatigue. Oncology 2000, 14, 151–161. [Google Scholar]
- Mustian, K.M.; Sprod, L.K.; Janelsins, M.; Peppone, L.J.; Mohile, S. Exercise Recommendations for Cancer-Related Fatigue, Cognitive Impairment, Sleep problems, Depression, Pain, Anxiety, and Physical Dysfunction: A Review. Oncol. Hematol. Rev. 2012, 8, 81–88. [Google Scholar] [CrossRef]
- Savard, J.; Morin, C.M. Insomnia in the Context of Cancer: A Review of a Neglected Problem. J. Clin. Oncol. 2001, 19, 895–908. [Google Scholar] [CrossRef]
- Pirl, W.; Roth, A.J. Diagnosis and treatment of depression in cancer patients. Oncology 1999, 13, 1293–1301. [Google Scholar]
- Weinberger, M.I.; Roth, A.J.; Nelson, C.J. Untangling the Complexities of Depression Diagnosis in Older Cancer Patients. Oncologist 2009, 14, 60–66. [Google Scholar] [CrossRef]
- Otte, J.L.; Carpenter, J.S.; Russell, K.M.; Bigatti, S.; Champion, V.L. Prevalence, Severity, and Correlates of Sleep-Wake Disturbances in Long-Term Breast Cancer Survivors. J. Pain Symptom Manag. 2010, 39, 535–547. [Google Scholar] [CrossRef]
- Loh, K.P.; Pandya, C.; Zittel, J.; Kadambi, S.; Flannery, M.; Reizine, N.; Magnuson, A.; Braganza, G.; Mustian, K.; Dale, W.; et al. Associations of sleep disturbance with physical function and cognition in older adults with cancer. Support. Care Cancer 2017, 25, 3161–3169. [Google Scholar] [CrossRef] [PubMed]
- Hahn, E.A.; Beaumont, J.; Pilkonis, P.A.; Garcia, S.F.; Magasi, S.; DeWalt, D.A.; Cella, D. The PROMIS satisfaction with social participation measures demonstrated responsiveness in diverse clinical populations. J. Clin. Epidemiol. 2016, 73, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Physical Activity Guidelines Advisory Committee. 2018 Physical Activity Guidelines Advisory Committee Report; U.S. Department of Health and Human Services: Washington, DC, USA, 2018; pp. G6-1–G6-31.
- Blair, C.; Morey, M.C.; Desmond, R.A.; Cohen, H.J.; Sloane, R.; Snyder, D.C.; Demark-Wahnefried, W. Light-Intensity Activity Attenuates Functional Decline in Older Cancer Survivors. Med. Sci. Sports Exerc. 2014, 46, 1375–1383. [Google Scholar] [CrossRef] [PubMed]
- Reid, N.; Daly, R.M.; Winkler, E.A.H.; Gardiner, P.A.; Eakin, E.G.; Owen, N.; Dunstan, D.W.; Healy, G.N. Associations of Monitor-Assessed Activity with Performance-Based Physical Function. PLoS ONE 2016, 11, e0153398. [Google Scholar] [CrossRef] [PubMed]
- Varma, V.R.; Tan, E.J.; Wang, T.; Xue, Q.-L.; Fried, L.P.; Seplaki, C.; King, A.C.; Seeman, T.E.; Rebok, G.W.; Carlson, M.C. Low-Intensity Walking Activity Is Associated with Better Health. J. Appl. Gerontol. 2013, 33, 870–887. [Google Scholar] [CrossRef]
- Thraen-Borowski, K.M.; Trentham-Dietz, A.; Edwards, D.F.; Koltyn, K.F.; Colbert, L.H. Dose–response relationships between physical activity, social participation, and health-related quality of life in colorectal cancer survivors. J. Cancer Surviv. 2013, 7, 369–378. [Google Scholar] [CrossRef]
- Vusirikala, A.; Ben-Shlomo, Y.; Kuh, D.; Stafford, M.; Cooper, R.; Morgan, G.S. Mid-life social participation and physical performance at age 60–64: Evidence from the 1946 British Birth Cohort Study. Eur. J. Public Health 2019, 29, 986–992. [Google Scholar] [CrossRef]
- He, Q.; Cui, Y.; Liang, L.; Zhong, Q.; Li, J.; Li, Y.; Lv, X.; Huang, F. Social participation, willingness and quality of life: A population-based study among older adults in rural areas of China. Geriatr. Gerontol. Int. 2016, 17, 1593–1602. [Google Scholar] [CrossRef]
- Corbett, D.B.; Rejeski, W.J.; Tudor-Locke, C.; Glynn, N.W.; Kritchevsky, S.B.; McDermott, M.M.; Church, T.S.; A Fielding, R.; Gill, T.; King, A.C.; et al. Social Participation Modifies the Effect of a Structured Physical Activity Program on Major Mobility Disability Among Older Adults: Results From the LIFE Study. J. Gerontol. Ser. B Psychol. Sci. Soc. Sci. 2017, 73, 1501–1513. [Google Scholar] [CrossRef]
- Kikuchi, H.; Inoue, S.; Fukushima, N.; Takamiya, T.; Odagiri, Y.; Ohya, Y.; Amagasa, S.; Oka, K.; Owen, N. Social participation among older adults not engaged in full- or part-time work is associated with more physical activity and less sedentary time. Geriatr. Gerontol. Int. 2017, 17, 1921–1927. [Google Scholar] [CrossRef]
- Soga, M.; Cox, D.; Yamaura, Y.; Gaston, K.J.; Kurisu, K.; Hanaki, K. Health Benefits of Urban Allotment Gardening: Improved Physical and Psychological Well-Being and Social Integration. Int. J. Environ. Res. Public Health 2017, 14, 71. [Google Scholar] [CrossRef]
- Soga, M.; Gaston, K.J.; Yamaura, Y. Gardening is beneficial for health: A meta-analysis. Prev. Med. Rep. 2016, 5, 92–99. [Google Scholar] [CrossRef]
- Spees, C.K.; Joseph, A.; Darragh, A.; Lyons, F.; Wolf, K.N. Health Behaviors and Perceptions of Cancer Survivors Harvesting at an Urban Garden. Am. J. Health Behav. 2015, 39, 257–266. [Google Scholar] [CrossRef]
- Lampert, T.; Costa, J.; Santos, O.; Sousa, J.; Ribeiro, T.; Freire, E. Evidence on the contribution of community gardens to promote physical and mental health and well-being of non-institutionalized individuals: A systematic review. PLoS ONE 2021, 16, e0255621. [Google Scholar] [CrossRef]
- Chen, T.-Y.; Janke, M.C. Gardening as a potential activity to reduce falls in older adults. J. Aging Phys. Act. 2012, 20, 15–31. [Google Scholar] [CrossRef]
- Berg, A.E.V.D.; Van Winsum-Westra, M.; De Vries, S.; Van Dillen, S.M. Allotment gardening and health: A comparative survey among allotment gardeners and their neighbors without an allotment. Environ. Health 2010, 9. [Google Scholar] [CrossRef]
- Nicklett, E.J.; Anderson, L.A.; Yen, I.H. Gardening Activities and Physical Health Among Older Adults: A Review of the Evidence. J. Appl. Gerontol. 2014, 35, 678–690. [Google Scholar] [CrossRef]
- Hawkins, J.L.; Smith, A.; Backx, K.; Clayton, D.A. Exercise Intensities of Gardening Tasks Within Older Adult Allotment Gardeners in Wales. J. Aging Phys. Act. 2015, 23, 161–168. [Google Scholar] [CrossRef]
- Park, S.-A.; Shoemaker, C.A.; Haub, M.D. A Preliminary Investigation on Exercise Intensities of Gardening Tasks in Older Adults. Percept. Mot. Ski. 2008, 107, 974–980. [Google Scholar] [CrossRef]
- Mullins, L.; Charlebois, S.; Finch, E.; Music, J. Home Food Gardening in Canada in Response to the COVID-19 Pandemic. Sustainability 2021, 13, 3056. [Google Scholar] [CrossRef]
- Theodorou, A.; Panno, A.; Carrus, G.; Carbone, G.A.; Massullo, C.; Imperatori, C. Stay home, stay safe, stay green: The role of gardening activities on mental health during the Covid-19 home confinement. Urban For. Urban Green. 2021, 61, 127091. [Google Scholar] [CrossRef]
- Marwah, P.; Zhang, Y.Y.; Gu, M. Impacts of COVID-19 on the Green Industry. Horticulturae 2021, 7, 329. [Google Scholar] [CrossRef]
- Oregon State University. Extension Master Gardener Program Rises to Challenge during Pandemic. Available online: https://ourimpact.oregonstate.edu/story/extension-master-gardener-program-rises-challenge-during-pandemic (accessed on 18 February 2022).
- Kassianos, A.P.; Georgiou, A.; Kyprianidou, M.; Lamnisos, D.; Ļubenko, J.; Presti, G.; Squatrito, V.; Constantinou, M.; Nicolaou, C.; Papacostas, S.; et al. Mental Health and Adherence to COVID-19 Protective Behaviors among Cancer Patients during the COVID-19 Pandemic: An International, Multinational Cross-Sectional Study. Cancers 2021, 13, 6294. [Google Scholar] [CrossRef] [PubMed]
- Bethea, T.N.; Zhai, W.; Zhou, X.; Ahles, T.A.; Ahn, J.; Cohen, H.J.; Dilawari, A.A.; Graham, D.M.A.; Jim, H.S.L.; McDonald, B.C.; et al. Associations between longitudinal changes in sleep disturbance and depressive and anxiety symptoms during the COVID -19 virus pandemic among older women with and without breast cancer in the thinking and living with breast cancer study. Cancer Med. 2022. [Google Scholar] [CrossRef] [PubMed]
- Rentscher, K.E.; Zhou, X.; Small, B.J.; Cohen, H.J.; Dilawari, A.A.; Patel, S.K.; Bethea, T.N.; Van Dyk, K.M.; Nakamura, Z.M.; Ahn, J.; et al. Loneliness and mental health during the COVID-19 pandemic in older breast cancer survivors and noncancer controls. Cancer 2021, 127, 3671–3679. [Google Scholar] [CrossRef]
- Tabaczynski, A.; Bastas, D.; Whitehorn, A.; Trinh, L. Changes in physical activity and associations with quality of life among a global sample of cancer survivors during the COVID-19 pandemic. J Cancer Surviv. 2022. [Google Scholar] [CrossRef]
- Hoffman, G.J.; Malani, P.N.; Solway, E.; Kirch, M.; Singer, D.C.; Kullgren, J.T. Changes in activity levels, physical functioning, and fall risk during the COVID -19 pandemic. J. Am. Geriatr. Soc. 2021, 70, 49–59. [Google Scholar] [CrossRef]
- Faro, J.M.; Mattocks, K.M.; Nagawa, C.S.; Lemon, S.C.; Wang, B.; Cutrona, S.L.; Sadasivam, R.S. Physical Activity, Mental Health, and Technology Preferences to Support Cancer Survivors During the COVID-19 Pandemic: Cross-sectional Study. JMIR Cancer 2021, 7, e25317. [Google Scholar] [CrossRef]
- Gurgel, A.R.B.; Mingroni-Netto, P.; Farah, J.C.; de Brito, C.M.M.; Levin, A.S.; Brum, P.C. Determinants of Health and Physical Activity Levels Among Breast Cancer Survivors During the COVID-19 Pandemic: A Cross-Sectional Study. Front. Physiol. 2021, 12, 624169. [Google Scholar] [CrossRef]

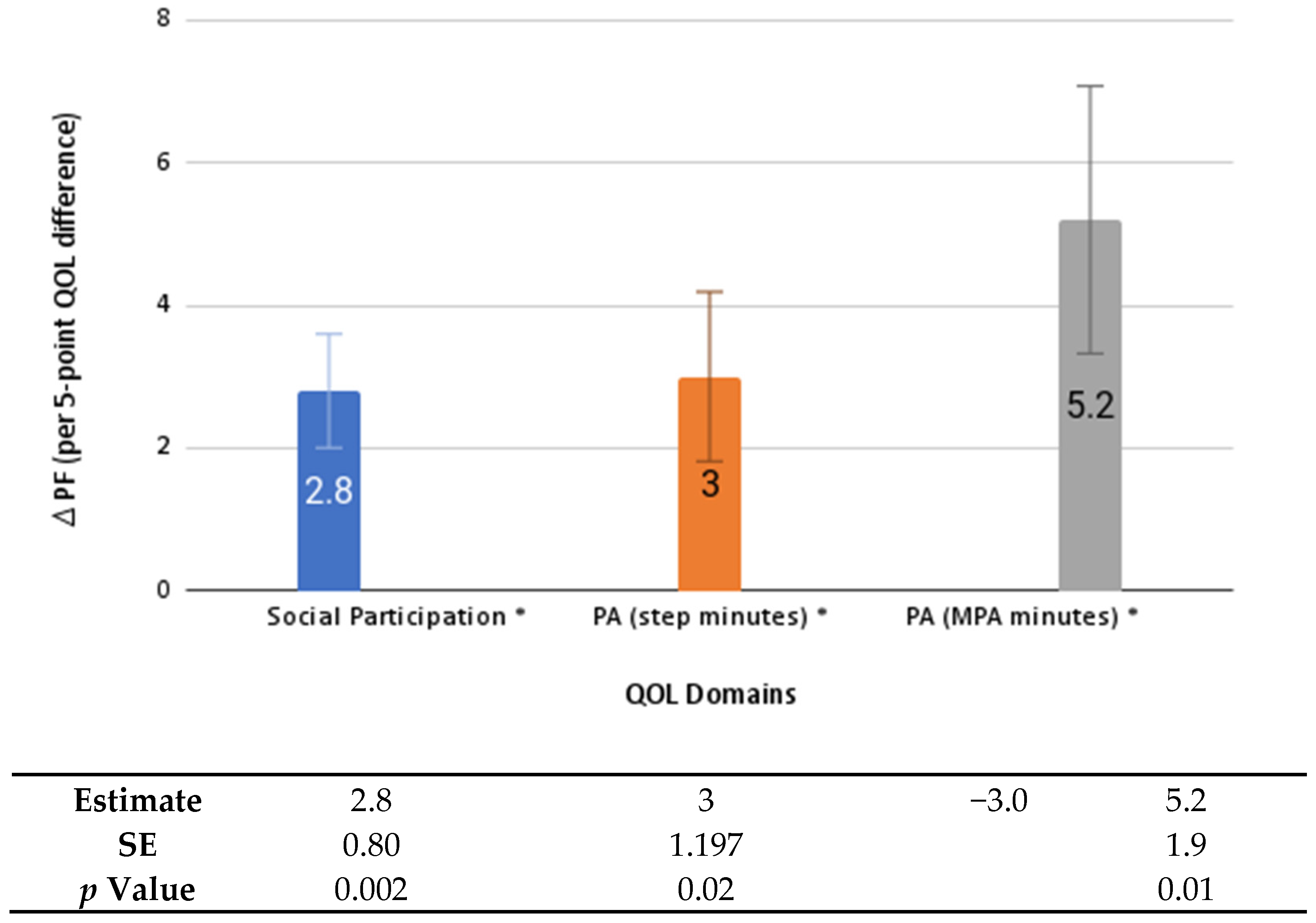
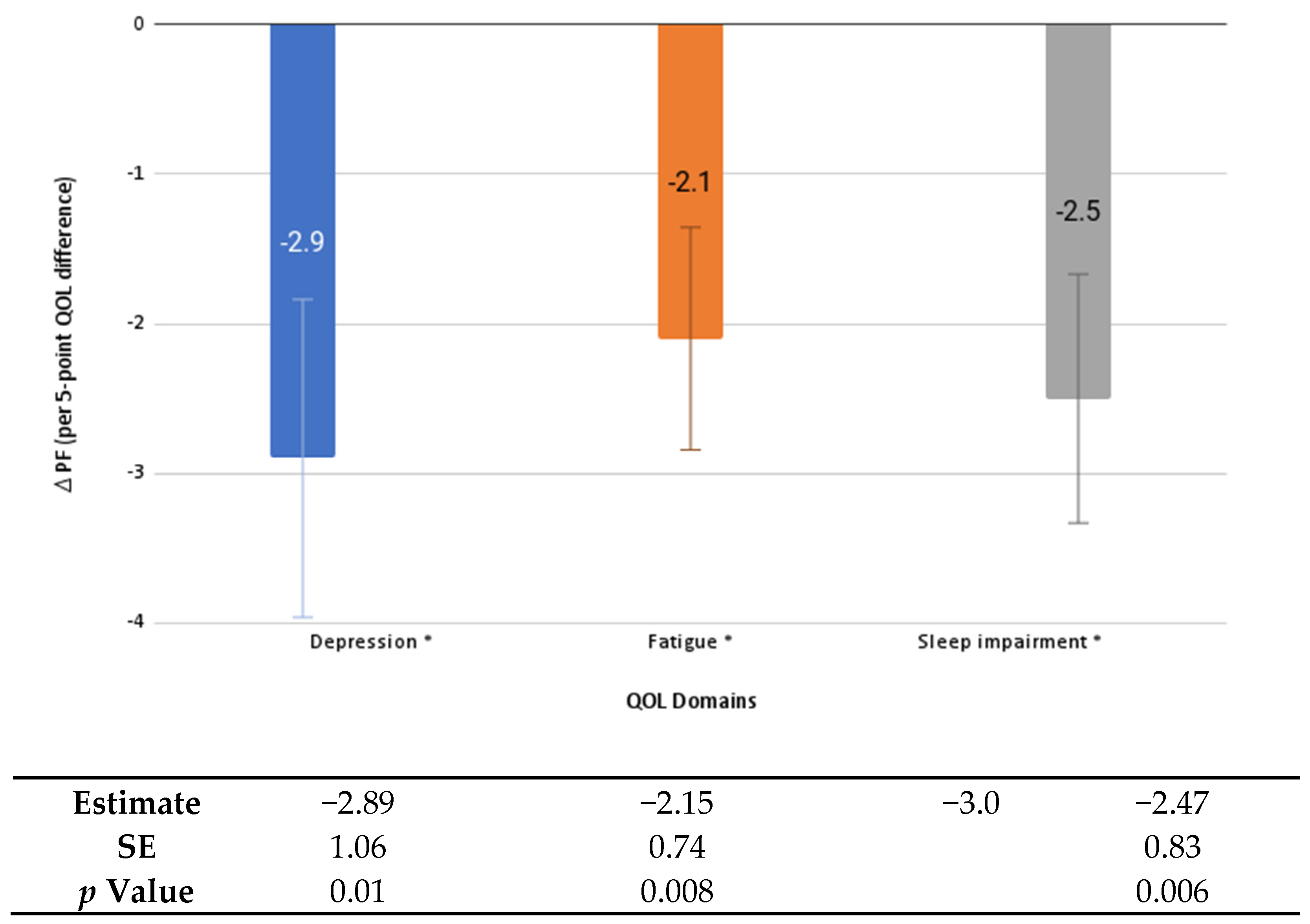
| Characteristics | Frequency (%) or Mean (SD) |
|---|---|
| Sociodemographics | |
| Age | |
| 50–64 | 9 (30%) |
| ≥65 | 21 (70%) |
| Sex | |
| Female | 21 (70%) |
| Male | 9 (30%) |
| Race–ethnicity | |
| Non-Hispanic White | 22 (73%) |
| Hispanic White | 6 (20%) |
| Other | 2 (7%) |
| Earned College Degree | |
| Yes | 17 (57%) |
| No | 13 (43%) |
| Income Group | |
| <USD 50,000 | 12 (40%) |
| ≥USD 50,000 | 15 (50%) |
| Declined to answer | 3 (10%) |
| Health Characteristics | |
| Self-reported general health | |
| Fair, poor | 5 (17%) |
| Good | 18 (60%) |
| Very good, excellent | 7 (23%) |
| Living Arrangement | |
| Alone | 13 (43%) |
| With Others | 17 (57%) |
| Number of comorbidities | |
| 0–1 | 5 (17%) |
| 2–3 | 15 (50%) |
| ≥4 | 10 (33%) |
| Type of cancer | |
| Breast | 11 (37%) |
| Prostate | 6 (20%) |
| Lung | 4 (13%) |
| Other | 9 (30%) |
| Physical Activity/exercise | |
| Objective physical activity (step minutes/day) 1 | 98.6 (34.8) |
| Self-reported exercise (minutes/week) 2 | 24.7 (39.5) |
| Mean (SD) range | |
| Quality of Life3 | |
| PF | 47.9 (8.2) (29.7–60.1) |
| Satisfaction with social roles/activities | 50.9 (9.1) (29.9–65.4) |
| Anxiety | 48.1 (9.4) (37.1–67.0) |
| Depression | 46.6 (8.4) (38.2–61.2) |
| Fatigue | 50.2 (9.5) (33.1–63.2) |
| Pain | 52.7 (8.4) (40.7–66.3) |
| Sleep disturbance | 50.1 (5.0) (34.1–58.7) |
| Sleep impairment | 48.7 (8.8) (35.9–60.6) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharma, H.; Pankratz, V.S.; Demark-Wahnefried, W.; Pestak, C.R.; Blair, C.K. Association between Quality of Life and Physical Functioning in a Gardening Intervention for Cancer Survivors. Healthcare 2022, 10, 1421. https://doi.org/10.3390/healthcare10081421
Sharma H, Pankratz VS, Demark-Wahnefried W, Pestak CR, Blair CK. Association between Quality of Life and Physical Functioning in a Gardening Intervention for Cancer Survivors. Healthcare. 2022; 10(8):1421. https://doi.org/10.3390/healthcare10081421
Chicago/Turabian StyleSharma, Harsh, Vernon S. Pankratz, Wendy Demark-Wahnefried, Claire R. Pestak, and Cindy K. Blair. 2022. "Association between Quality of Life and Physical Functioning in a Gardening Intervention for Cancer Survivors" Healthcare 10, no. 8: 1421. https://doi.org/10.3390/healthcare10081421
APA StyleSharma, H., Pankratz, V. S., Demark-Wahnefried, W., Pestak, C. R., & Blair, C. K. (2022). Association between Quality of Life and Physical Functioning in a Gardening Intervention for Cancer Survivors. Healthcare, 10(8), 1421. https://doi.org/10.3390/healthcare10081421







