Bioequivalence, Drugs with Narrow Therapeutic Index and the Phenomenon of Biocreep: A Critical Analysis of the System for Generic Substitution
Abstract
1. Introduction
2. Requirements for Approval of Generics and Criteria for Drug Substitution
3. Drugs with Narrow Therapeutic Index: Bioequivalence versus Therapeutic Equivalence
- Category 1, including phenytoin, carbamazepine, phenobarbital and primidone; switching between versions of these antiepileptics is not recommended;
- Category 2, including sodium valproate, lamotrigine, perampanel, retigabine, rufinamide, clobazam, clonazepam, oxcarbazepine, eslicarbazepine, topiramate and zonisamide; the decision about prescription is based on clinical status (such as seizure frequency and treatment history) and patient/caregiver point of view;
- Category 3, including levetiracetam, lacosamide, tiagabine, gabapentin, pregabalin, ethosuximide, brivaracetam and vigabatrin; in this case, patients can be freely switched among different versions of antiepileptics.
4. The Italian Regulation for Prescription of Generic Drugs: Strengths and Limits
- If the prescription only indicates the active substance, the pharmacist informs the patient and will deliver the medicine with the lowest price fully covered by the NHS [86]; if the patient expressly requests a product with a higher price, the price differential between the medicine requested and the one with the lowest price will be paid out-of-pocket;
- If the prescription indicates a specific medication without the non-replaceability clause, the pharmacist can provide the prescribed medicine only if no cheaper equivalent is available; otherwise, the pharmacist must supply the medicine with the lowest price, unless the patient requests and pays for the brand-name product;
- Finally, if the prescription indicates the non-replaceability condition, the pharmacist should ask the patient to pay the difference between any higher price of the prescribed medicine.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Borgheini, G. The bioequivalence and therapeutic efficacy of generic versus brand-name psychoactive drugs. Clin. Ther. 2003, 25, 1578–1592. [Google Scholar] [CrossRef]
- Sisko, A.M.; Truffer, C.J.; Keehan, S.P.; Poisal, J.A.; Clemens, M.K.; Madison, A.J. National Health Spending Projections: The Estimated Impact of Reform Through 2019. Health Aff. 2010, 29, 1933–1941. [Google Scholar] [CrossRef] [PubMed]
- Barros, P.P. Pharmaceutical policies in European countries. Adv. Health Econ. Health Serv. Res. 2010, 22, 3–27. [Google Scholar] [CrossRef] [PubMed]
- Karalis, V.; Bialer, M.; Macheras, P. Quantitative assessment of the switchability of generic products. Eur. J. Pharm. Sci. 2013, 50, 476–483. [Google Scholar] [CrossRef]
- Seoane-Vazquez, E.; Rodriguez-Monguio, R.; Hansen, R. Interchangeability, Safety and Efficacy of Modified-Release Drug Formulations in the USA: The Case of Opioid and Other Nervous System Drugs. Clin. Drug Investig. 2016, 36, 281–292. [Google Scholar] [CrossRef]
- Directive 2001/83/EC of the European Parliament and of the Council of 6 November 2001 on the Community Code Relating to Medicinal Products for Human Use. OJ L 311, 28 November 2001. pp. 67–128. Available online: https://www.ema.europa.eu/en/documents/regulatory-procedural-guideline/directive-2001/83/ec-european-parliament-council-6-november-2001-community-code-relating-medicinal-products-human-use_en.pdf (accessed on 15 January 2022).
- GUIDELINE ON THE INVESTIGATION OF BIOEQUIVALENCE. London, 20 January 2010. Available online: https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-investigation-bioequivalence-rev1_en.pdf (accessed on 15 January 2022).
- WHO. WHO Technical report series 937, 2006. In Proceedings of the 40th Report of the WHO Expert Committee on Specifications for Pharmaceutical Preparations, Geneva, Switzerland, 24–28 October 2005; Available online: http://apps.who.int/iris/bitstream/handle/10665/43443/WHO_TRS_937_eng.pdf;jsessionid=17413E84D7E24B06437B5D2C4C6C6913?sequence=1 (accessed on 15 January 2022).
- Carpenter, D.; Tobbell, D.A. Bioequivalence: The Regulatory Career of a Pharmaceutical Concept. Bull. Hist. Med. 2011, 85, 93–131. [Google Scholar] [CrossRef]
- Desai, R.J.; Sarpatwari, A.; Dejene, S.; Khan, N.F.; Lii, J.; Rogers, J.R.; Dutcher, S.K.; Raofi, S.; Bohn, J.; Connolly, J.G.; et al. Comparative effectiveness of generic and brand-name medication use: A database study of US health insurance claims. PLoS Med. 2019, 16, e1002763. [Google Scholar] [CrossRef]
- Corrao, G.; Soranna, D.; Merlino, L.; Mancia, G. Similarity between generic and brand-name antihypertensive drugs for primary prevention of cardiovascular disease: Evidence from a large population-based study. Eur. J. Clin. Investig. 2014, 44, 933–939. [Google Scholar] [CrossRef]
- Kesselheim, A.S.; Misono, A.S.; Lee, J.L.; Stedman, M.R.; Brookhart, M.A.; Choudhry, N.K.; Shrank, W.H. Clinical equivalence of generic and brand-name drugs used in cardiovascular disease: A systematic review and meta-analysis. JAMA 2008, 300, 2514–2526. [Google Scholar] [CrossRef]
- Corrao, G.; Soranna, D.; Arfè, A.; Casula, M.; Tragni, E.; Merlino, L.; Mancia, G.; Catapano, A.L. Are generic and brand-name statins clinically equivalent? Evidence from a real data-base. Eur. J. Intern. Med. 2014, 25, 745–750. [Google Scholar] [CrossRef]
- Manzoli, L.; Flacco, M.E.; Boccia, S.; D’Andrea, E.; Panic, N.; Marzuillo, C.; Siliquini, R.; Ricciardi, W.; Villari, P.; Ioannidis, J.P.A. Generic versus brand-name drugs used in cardiovascular diseases. Eur. J. Epidemiol. 2015, 31, 351–368. [Google Scholar] [CrossRef]
- Gagne, J.J.; Choudhry, N.K.; Kesselheim, A.S.; Polinski, J.M.; Hutchins, D.; Matlin, O.S.; Brennan, T.A.; Avorn, J.; Shrank, W.H. Comparative effectiveness of generic and brand-name statins on patient outcomes: A cohort study. Ann. Intern. Med. 2014, 161, 400–407. [Google Scholar] [CrossRef]
- Puglia, C.; Santonocito, D.; Romeo, G.; Intagliata, S.; Romano, G.; Strettoi, E.; Novelli, E.; Ostacolo, C.; Campiglia, P.; Sommella, E.; et al. Lipid Nanoparticles Traverse Non-Corneal Path to Reach the Posterior Eye Segment: In Vivo Evidence. Molecules 2021, 26, 4673. [Google Scholar] [CrossRef]
- FDA. Guidance on Bioavailability and Bioequivalence Studies for Orally Administrated Drug Products–General Considerations; Center for Drug Evaluation and Research, the US Food and Drug Administration: Rockville, MD, USA, 2003.
- Meyer, M.C. United States Food and Drug Administration requirements for approval of generic drug products. J. Clin. Psychiatry 2001, 62 (Suppl. S5), 23–24. [Google Scholar]
- Blier, P. Generic medications: Another variable in the treatment of illnesses. J. Psychopharmacol. 2006, 21, 459–460. [Google Scholar] [CrossRef]
- FDA. FDA Fact Sheet: What’s Involved in Reviewing and Approving Generic Drug Applications? Food and Drug Administration: Rockville, MD, USA, 2017.
- Health Canada. The Safety and Effectiveness of Generic Drugs; Health Canada: Ottawa, ON, Canada, 2012.
- Conti, F.; Romano, G.L.; Eandi, C.M.; Toro, M.D.; Rejdak, R.; Di Benedetto, G.; Lazzara, F.; Bernardini, R.; Drago, F.; Cantarella, G.; et al. Brimonidine is Neuroprotective in Animal Paradigm of Retinal Ganglion Cell Damage. Front. Pharmacol. 2021, 12, 705405. [Google Scholar] [CrossRef]
- Kamińska, A.; Romano, G.; Rejdak, R.; Zweifel, S.; Fiedorowicz, M.; Rejdak, M.; Bajka, A.; Amato, R.; Bucolo, C.; Avitabile, T.; et al. Influence of Trace Elements on Neurodegenerative Diseases of The Eye—The Glaucoma Model. Int. J. Mol. Sci. 2021, 22, 4323. [Google Scholar] [CrossRef]
- FDA. Approved Drug Products with Therapeutic Equivalence Evaluations, 42nd ed. Available online: https://www.fda.gov/media/71474/download (accessed on 15 February 2022).
- Godman, B.; Shrank, W.; Wettermark, B.; Andersen, M.; Bishop, I.; Burkhardt, T.; Garuolienè, K.; Kalaba, M.; Laius, O.; Joppi, R.; et al. Use of Generics—A Critical Cost Containment Measure for All Healthcare Professionals in Europe? Pharmaceuticals 2010, 3, 2470–2494. [Google Scholar] [CrossRef]
- Godman, B.; Shrank, W.; Andersen, M.; Berg, C.; Bishop, I.; Burkhardt, T.; Garuoliene, K.; Herholz, H.; Joppi, R.; Kalaba, M.; et al. Policies to Enhance Prescribing Efficiency in Europe: Findings and Future Implications. Front. Pharmacol. 2011, 1, 141. [Google Scholar] [CrossRef]
- CGDS. Competition Bureau Canada; CGDS: Ottawa, ON, Canada, 2007. [Google Scholar]
- McGinn, D.; Godman, B.; Lonsdale, J.; Way, R.; Wettermark, B.; Haycox, A. Initiatives to enhance the quality and efficiency of statin and PPI prescribing in the UK: Impact and implications. Expert Rev. Pharm. Outcomes Res. 2010, 10, 73–85. [Google Scholar] [CrossRef]
- van Woerkom, M.; Piepenbrink, H.; Godman, B.; de Metz, J.; Campbell, S.; Bennie, M.; Eimers, M.; Gustafsson, L.L. Ongoing measures to enhance the efficiency of prescribing of proton pump inhibitors and statins in The Netherlands: Influence and future implications. J. Comp. Eff. Res. 2012, 1, 527–538. [Google Scholar] [CrossRef]
- MacBride-Stewart, S.; McTaggart, S.; Kurdi, A.; Sneddon, J.; McBurney, S.; do Nascimento, R.C.R.M.; Mueller, T.; Kwon, H.Y.; Morton, A.; Seaton, R.A.; et al. Initiatives and reforms across Scotland in recent years to improve prescribing; findings and global implications of drug prescriptions. Int. J. Clin. Exp. Med. 2021, 14, 2563–2586. [Google Scholar]
- Duerden, M.G.; Hughes, D.A. Generic and therapeutic substitutions in the UK: Are they a good thing? Br. J. Clin. Pharmacol. 2010, 70, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Ferner, R.E.; Lenney, W.; Marriott, J.F. Controversy over generic substitution. BMJ 2010, 340, c2548. [Google Scholar] [CrossRef]
- Chou, T.-H.; Musada, G.R.; Romano, G.L.; Bolton, E.; Porciatti, V. Anesthetic Preconditioning as Endogenous Neuroprotection in Glaucoma. Int. J. Mol. Sci. 2018, 19, 237. [Google Scholar] [CrossRef]
- Goodman, B.; Wilcock, M.; Martin, A.; Fraeyman, J. Generic pregabalin; current situation and implications for health authorities, generics and biosimilars manufacturers in the future. Generics Biosimilars Initiat. J. 2015, 4, 125–135. [Google Scholar] [CrossRef][Green Version]
- Blier, P.; Margolese, H.C.; Wilson, E.A.; Boucher, M. Switching medication products during the treatment of psychiatric illness. Int. J. Psychiatry Clin. Pract. 2019, 23, 2–13. [Google Scholar] [CrossRef]
- Ferguson, D.C. Chapter 3—Principles of Pharmacodynamics and Toxicodynamics. In Haschek and Rousseaux’s Handbook of Toxicologic Pathology, 3rd ed.; Haschek, W.M., Rousseaux, C.G., Wallig, M.A., Bolon, B., Ochoa, R., Eds.; Academic Press: Cambridge, MA, USA, 2013; pp. 61–76. ISBN 9780124157590. [Google Scholar] [CrossRef]
- Eaton, D.L.; Gilbert, S.G. Principles of Toxicology, Reference Module in Biomedical Sciences; Elsevier: Amsterdam, The Netherlands, 2014; ISBN 9780128012383. [Google Scholar] [CrossRef]
- Linsay McCallum, S.L.; Padmanabhan, S. Chapter 18—Pharmacodynamic Pharmacogenomics. In Handbook of Pharmacogenomics and Stratified Medicine; Padmanabhan, S., Ed.; Academic Press: Cambridge, MA, USA, 2014; pp. 365–383. ISBN 9780123868824. [Google Scholar] [CrossRef]
- Elizabeth, S.; Lowe, J.J.L.L. Chapter 20—Dose–Effect and Concentration–Effect Analysis. In Principles of Clinical Pharmacology, 3rd ed.; Huang, S.M., Lertora, J.J., Atkinson, A.J., Eds.; Academic Press: Cambridge, MA, USA, 2012; pp. 343–356. ISBN 9780123854711. [Google Scholar] [CrossRef]
- Toro, M.D.; Gozzo, L.; Tracia, L.; Cicciù, M.; Drago, F.; Bucolo, C.; Avitabile, T.; Rejdak, R.; Nowomiejska, K.; Zweifel, S.; et al. New Therapeutic Perspectives in the Treatment of Uveal Melanoma: A Systematic Review. Biomedicines 2021, 9, 1311. [Google Scholar] [CrossRef]
- Paixão, P.; Guerreiro, R.B.; Silva, N.; Blake, K.; Bonelli, M.; Morais, J.A.G.; Arieta, A.G.; Gouveia, L.F. A Proposed Approach for the Determination of the Bioequivalence Acceptance Range for Narrow Therapeutic Index Drugs in the European Union. Clin. Pharmacol. Ther. 2021, 111, 470–476. [Google Scholar] [CrossRef]
- Yu, L.; Jiang, W.; Zhang, X.; Lionberger, R.; Makhlouf, F.; Schuirmann, D.; Muldowney, L.; Chen, M.-L.; Davit, B.; Conner, D.; et al. Novel bioequivalence approach for narrow therapeutic index drugs. Clin. Pharmacol. Ther. 2014, 97, 286–291. [Google Scholar] [CrossRef]
- Tamargo, J.; Le Heuzey, J.-Y.; Mabo, P. Narrow therapeutic index drugs: A clinical pharmacological consideration to flecainide. Eur. J. Clin. Pharmacol. 2015, 71, 549–567. [Google Scholar] [CrossRef] [PubMed]
- Katzung, B.M.S.; Trevor, A. Basic and Clinical Pharmacology, 12th ed.; Companies, Inc.: New York, NY, USA, 2012. [Google Scholar]
- Jiang, W.; Makhlouf, F.; Schuirmann, D.J.; Zhang, X.; Zheng, N.; Conner, D.; Yu, L.X.; Lionberger, R. A Bioequivalence Approach for Generic Narrow Therapeutic Index Drugs: Evaluation of the Reference-Scaled Approach and Variability Comparison Criterion. AAPS J. 2015, 17, 891–901. [Google Scholar] [CrossRef] [PubMed]
- Odi, R.; Franco, V.; Perucca, E.; Bialer, M. Bioequivalence and switchability of generic antiseizure medications (ASMs): A re-appraisal based on analysis of generic ASM products approved in Europe. Epilepsia 2021, 62, 285–302. [Google Scholar] [CrossRef]
- Crawford, P.; Feely, M.; Guberman, A.; Kramer, G. Are there potential problems with generic substitution of antiepileptic drugs?: A review of issues. Seizure 2006, 15, 165–176. [Google Scholar] [CrossRef]
- Berg, M.J.; Gross, R.A.; Tomaszewski, K.J.; Zingaro, W.M.; Haskins, L.S. Generic substitution in the treatment of epilepsy: Case evidence of breakthrough seizures. Neurology 2008, 71, 525–530. [Google Scholar] [CrossRef]
- Kesselheim, A.S.; Stedman, M.R.; Bubrick, E.J.; Gagne, J.J.; Misono, A.S.; Lee, J.L.; Brookhart, M.A.; Avorn, J.; Shrank, W.H. Seizure outcomes following the use of generic versus brand-name antiepileptic drugs: A systematic review and meta-analysis. Drugs 2010, 70, 605–621. [Google Scholar] [CrossRef]
- Macdonald, J.T. Breakthrough seizure following substitution of Depakene capsules (Abbott) with a generic product. Neurology 1987, 37, 1885. [Google Scholar] [CrossRef]
- Patel, V.; Cordato, D.J.; Dias, M.; Beran, R.G. Changed constitution without change in brand name—The risk of generics in epilepsy. Epilepsy Res. 2011, 98, 269–272. [Google Scholar] [CrossRef][Green Version]
- Baumgärtel, C. Myths, questions, facts about generic drugs in the EU. Generics Biosimilars Initiat. J. 2012, 1, 34–38. [Google Scholar] [CrossRef]
- Heaney, D.C.; Sander, J.W. Antiepileptic drugs: Generic versus branded treatments. Lancet Neurol. 2007, 6, 465–468. [Google Scholar] [CrossRef]
- Brancati, S.; Gozzo, L.; Longo, L.; Vitale, D.C.; Russo, G.; Drago, F. Fertility Preservation in Female Pediatric Patients with Cancer: A Clinical and Regulatory Issue. Front. Oncol. 2021, 11, 641450. [Google Scholar] [CrossRef]
- Brancati, S.; Gozzo, L.; Longo, L.; Vitale, D.C.; Drago, F. Rituximab in Multiple Sclerosis: Are We Ready for Regulatory Approval? Front. Immunol. 2021, 12, 661882. [Google Scholar] [CrossRef]
- Lessing, C.; Ashton, T.; Davis, P. The Impact on Health Outcomes and Healthcare Utilisation of Switching to Generic Medicines Consequent to Reference Pricing: The Case of Lamotrigine in New Zealand. Appl. Health Econ. Health Policy 2014, 12, 537–546. [Google Scholar] [CrossRef]
- Gagne, J.J.; Kesselheim, A.S.; Choudhry, N.K.; Polinski, J.M.; Hutchins, D.; Matlin, O.S.; Brennan, T.A.; Avorn, J.; Shrank, W.H. Comparative effectiveness of generic versus brand-name antiepileptic medications. Epilepsy Behav. 2015, 52, 14–18. [Google Scholar] [CrossRef]
- Berg, M.; Welty, T.E.; Gidal, B.E.; Diaz, F.J.; Krebill, R.; Szaflarski, J.P.; Dworetzky, B.A.; Pollard, J.R.; Elder, E.J.; Jiang, W.; et al. Bioequivalence between Generic and Branded Lamotrigine in People with Epilepsy: The EQUIGEN Randomized Clinical Trial. JAMA Neurol. 2017, 74, 919–926. [Google Scholar] [CrossRef]
- Agency MaHpR. Drug Safety Update. Antiepileptic Drugs: Updated Advice on Switching between Different Manufacturers’ Products. Available online: https://www.gov.uk/drug-safety-update/antiepileptic-drugs-updated-advice-on-switching-between-different-manufacturers-products (accessed on 15 February 2022).
- Agency MaHpR. Drug Safety Update.Antiepileptic Drugs: New Advice on Switching between Different Manufacturers’ Products for a Particular Drug. Available online: https://www.gov.uk/drug-safety-update/antiepileptic-drugs-new-advice-on-switching-between-different-manufacturers-products-for-a-particular-drug (accessed on 15 January 2022).
- Van Paesschen, W.; Hauman, H.; Lagae, L. The use of generic medication in epilepsy: A review of potential issues and challenges. Eur. J. Paediatr. Neurol. 2009, 13, 87–92. [Google Scholar] [CrossRef]
- Godman, B.; Baumgartel, C. Are generic immunosuppressants safe and effective? BMJ 2015, 350, h3248. [Google Scholar] [CrossRef][Green Version]
- Overholser, B.R.; Foster, D.R. Opioid pharmacokinetic drug-drug interactions. Am. J. Manag. Care 2011, 17 (Suppl. S11), S276–S287. [Google Scholar]
- AIFA. Riassunto Delle Caratteristiche del Prodotto Morfina. Available online: https://farmaci.agenziafarmaco.gov.it/aifa/servlet/PdfDownloadServlet?pdfFileName=footer_000141_030677_RCP.pdf&sys=m0b1l3 (accessed on 15 December 2021).
- AIFA. Riassunto Delle Caratteristiche del Prodotto Fentanil. Available online: https://farmaci.agenziafarmaco.gov.it/aifa/servlet/PdfDownloadServlet?pdfFileName=footer_008043_037609_RCP.pdf&sys=m0b1l3 (accessed on 15 December 2021).
- Krcevski Skvarc, N.; Morlion, B.; Vowles, K.E.; Bannister, K.; Buchsner, E.; Casale, R.; Chenot, J.F.; Chumbley, G.; Drewes, A.M.; Dom, G.; et al. European clinical practice recommendations on opioids for chronic noncancer pain—Part 2: Special situations. Eur. J. Pain. 2021, 25, 969–985. [Google Scholar] [CrossRef]
- Caraci, F.; Merlo, S.; Drago, F.; Caruso, G.; Parenti, C.; Sortino, M.A. Rescue of Noradrenergic System as a Novel Pharmacological Strategy in the Treatment of Chronic Pain: Focus on Microglia Activation. Front. Pharmacol. 2019, 10, 1024. [Google Scholar] [CrossRef]
- Arendt-Nielsen, L.; Morlion, B.; Perrot, S.; Dahan, A.; Dickenson, A.; Kress, H.G.; Wells, C.; Bouhassira, D.; Drewes, A.M. Assessment and manifestation of central sensitisation across different chronic pain conditions. Eur. J. Pain 2018, 22, 216–241. [Google Scholar] [CrossRef] [PubMed]
- Pasternak, G.W. When it comes to opiates, just say NO. J. Clin. Investig. 2007, 117, 3185–3187. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, T.; Christrup, L.; Drewes, A.; Fallon, M.; Kress, H.; McQuay, H.; Mikus, G.; Morlion, B.; Perez-Cajaraville, J.; Pogatzki-Zahn, E.; et al. European Pain Federation position paper on appropriate opioid use in chronic pain management. Eur. J. Pain 2016, 21, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Drewes, A.; Jensen, R.D.; Nielsen, L.M.; Droney, J.; Christrup, L.; Arendt-Nielsen, L.; Riley, J.; Dahan, A. Differences between opioids: Pharmacological, experimental, clinical and economical perspectives. Br. J. Clin. Pharmacol. 2012, 75, 60–78. [Google Scholar] [CrossRef]
- Benyamin, R.; Trescot, A.M.; Datta, S.; Buenaventura, R.; Adlaka, R.; Sehgal, N.; Glaser, S.E.; Vallejo, R. Opioid complications and side effects. Pain Phys. 2008, 11, S105–S120. [Google Scholar] [CrossRef]
- Pergolizzi, J.V., Jr.; Raffa, R.B.; Rosenblatt, M.H. Opioid withdrawal symptoms, a consequence of chronic opioid use and opioid use disorder: Current understanding and approaches to management. J. Clin. Pharm. Ther. 2020, 45, 892–903. [Google Scholar] [CrossRef]
- Neuman, M.D.; Bateman, B.T.; Wunsch, H. Inappropriate opioid prescription after surgery. Lancet 2019, 393, 1547–1557. [Google Scholar] [CrossRef]
- Sadhasivam, S.; Chidambaran, V. Pharmacogenomics of opioids and perioperative pain management. Pharmacogenomics 2012, 13, 1719–1740. [Google Scholar] [CrossRef]
- Walter, C.; Felden, L.; Lotsch, J. Bioequivalence criteria for transdermal fentanyl generics: Do these need a relook? Clin. Pharm. 2009, 48, 625–633. [Google Scholar] [CrossRef]
- Smith, H.S.; Peppin, J.F. Toward a systematic approach to opioid rotation. J. Pain Res. 2014, 7, 589–608. [Google Scholar]
- Powell, V.D.; Rosenberg, J.M.; Yaganti, A.; Garpestad, C.; Lagisetty, P.; Shannon, C.; Silveira, M.J. Evaluation of Buprenorphine Rotation in Patients Receiving Long-term Opioids for Chronic Pain: A Systematic Review. JAMA Netw. Open 2021, 4, e2124152. [Google Scholar] [CrossRef]
- Pergolizzi, J.V., Jr.; Labhsetwar, S.A.; Puenpatom, R.A.; Joo, S.; Ben-Joseph, R.H.; Summers, K.H. Prevalence of Exposure to Potential CYP450 Pharmacokinetic Drug-Drug Interactions among Patients with Chronic Low Back Pain Taking Opioids. Pain Pract. 2010, 11, 230–239. [Google Scholar] [CrossRef]
- Ellis, J.J.; Sadosky, A.B.; Eyck, L.L.T.; Mudumby, P.; Cappelleri, J.C.; Ndehi, L.; Suehs, B.T.; Parsons, B. A retrospective, matched cohort study of potential drug-drug interaction prevalence and opioid utilization in a diabetic peripheral neuropathy population initiated on pregabalin or duloxetine. BMC Health Serv. Res. 2015, 15, 159. [Google Scholar] [CrossRef]
- Pergolizzi, J.V.; Ma, L.; Foster, D.R.; Overholser, B.R.; Sowinski, K.M.; Taylor, R., Jr.; Summers, K.H. The Prevalence of Opioid-Related Major Potential Drug-Drug Interactions and Their Impact on Health Care Costs in Chronic Pain Patients. J. Manag. Care Pharm. 2014, 20, 467–476. [Google Scholar] [CrossRef]
- Matos, A.; Bankes, D.L.; Bain, K.T.; Ballinghoff, T.; Turgeon, J. Opioids, Polypharmacy, and Drug Interactions: A Technological Paradigm Shift Is Needed to Ameliorate the Ongoing Opioid Epidemic. Pharmacy 2020, 8, 154. [Google Scholar] [CrossRef]
- BfArM. Bundesinstitut fur Arzneimittel und Medizinprodukte [BfArM]. Neues zur Aut-Idem Regelung: Ist die Aut-Idem-Regelung auch bei Schmerzpflastern Anwendbar? BfArM: Bonn, Germany, 2008. Available online: http://www.bfarm.de/cln_028/nn_683944/SharedDocs/FAQ/DE/BtmGrundstoffeAMVV/BtM/Rezepte/t2-verschr-ambul/ha-t2-BTMRezepte-faq8.html (accessed on 15 February 2022).
- AIFA. Health Card Project, Pharmaceutical Prescription-Update Pursuant to Art. 15, Paragraph 11-Bis of Legislative Decree 95/2012 and Subsequent Amendments. 2012. Available online: https://www.aifa.gov.it/documents/20142/241044/Documento_linee_guida_prescrizione_principio_25.10.2013.pdf (accessed on 15 January 2022).
- DECRETO-LEGGE 24 Gennaio 2012, n. 1. 2012. Available online: https://www.normattiva.it/uri-res/N2Ls?urn:nir:stato:decreto.legge:2012-01-24;1 (accessed on 15 January 2022).
- Decree-Law, No. 347/2001, Converted with Modifications by Law No. 405/2001. 2001. Available online: https://www.parlamento.it/parlam/leggi/decreti/01347d.htm (accessed on 15 January 2022).
- AIFA. Secondo Position Paper AIFA sui Farmaci Biosimilari. 2019. Available online: https://www.aifa.gov.it/documents/20142/808717/2_Position-Paper-AIFA-Farmaci-Biosimilari.pdf (accessed on 15 December 2021).
- Olimpieri, P.; Di Lenarda, A.; Mammarella, F.; Gozzo, L.; Cirilli, A.; Cuomo, M.; Gulizia, M.; Colivicchi, F.; Murri, G.; Gabrielli, D.; et al. Non-vitamin K antagonist oral anticoagulation agents in patients with atrial fibrillation: Insights from Italian monitoring registries. IJC Heart Vasc. 2020, 26, 100465. [Google Scholar] [CrossRef]
- Breccia, M.; Olimpieri, P.P.; Olimpieri, O.; Pane, F.; Iurlo, A.; Foggi, P.; Cirilli, A.; Colatrella, A.; Cuomo, M.; Gozzo, L.; et al. How many chronic myeloid leukemia patients who started a frontline second-generation tyrosine kinase inhibitor have to switch to a second-line treatment? A retrospective analysis from the monitoring registries of the italian medicines agency (AIFA). Cancer Med. 2020, 9, 4160–4165. [Google Scholar] [CrossRef]
- Breccia, M.; Celant, S.; Olimpieri, P.P.; Olimpieri, O.M.; Pane, F.; Iurlo, A.; Cirilli, A.; Colatrella, A.; Gozzo, L.; on behalf of AIFA Monitoring Registries Group; et al. Mortality rate in patients with chronic myeloid leukemia in chronic phase treated with frontline second generation tyrosine kinase inhibitors: A retrospective analysis by the monitoring registries of the Italian Medicines Agency (AIFA). Ann. Hematol. 2021, 100, 481–485. [Google Scholar] [CrossRef]
- Gozzo, L.; Di Lenarda, A.; Mammarella, F.; Olimpieri, P.P.; Cirilli, A.; Cuomo, M.; Gulizia, M.M.; Colivicchi, F.; Murri, G.; Kunutsor, S.K.; et al. Starting dose and dose adjustment of non-vitamin K antagonist oral anticoagulation agents in a nationwide cohort of patients with atrial fibrillation. Sci. Rep. 2021, 11, 20689. [Google Scholar] [CrossRef]
- Law No. 178 of 8 August 2002 Art. 9, Paragraph. 2002. Available online: https://www.parlamento.it/parlam/leggi/02178l.htm (accessed on 15 January 2022).
- Gazzetta Ufficiale-Serie Generale n. 39 la Determinazione Direttoriale AIFA n. 166 del 10 Febbraio 2021, Avente ad Oggetto la “Definizione dei Criteri per L’inserimento in Lista di Trasparenza dei Medicinali ai Sensi Dell’art. 7 del D.L. 18.9.2001, n. 347 (convertito in Legge 16.11.2001, n. 405). 2021. Available online: https://www.aifa.gov.it/documents/20142/1134592/Determina_DG-166-2021.pdf (accessed on 15 January 2022).
- Gozzo, L.; Longo, L.; Vitale, D.C.; Drago, F. Dexamethasone Treatment for COVID-19, a Curious Precedent Highlighting a Regulatory Gap. Front. Pharmacol. 2020, 11, 621934. [Google Scholar] [CrossRef]
- Gozzo, L.; Longo, L.; Vitale, D.C.; Drago, F. The Regulatory Challenges for Drug Repurposing During the COVID-19 Pandemic: The Italian Experience. Front. Pharmacol. 2020, 11, 588132. [Google Scholar] [CrossRef] [PubMed]
- Gozzo, L.; Romano, G.L.; Romano, F.; Brancati, S.; Longo, L.; Vitale, D.C.; Drago, F. Health Technology Assessment of Advanced Therapy Medicinal Products: Comparison Among 3 European Countries. Front. Pharmacol. 2021, 12, 755052. [Google Scholar] [CrossRef] [PubMed]
- Boulkrane, M.S.; Ilina, V.; Melchakov, R.; Arisov, M.; Fedotova, J.; Gozzo, L.; Drago, F.; Lucia, G.; Sarapultsev, A.; Tseilikman, V.; et al. SARS-Cov-2 Damage on the Nervous System and Mental Health. Curr. Neuropharmacol. 2022, 20, 412–431. [Google Scholar] [CrossRef] [PubMed]
- AIFA. AIFA Communication on Medicinal Products Containing Levetiracetam and Topiramate (17 September 2012); AIFA: Seoul, Korea, 2012.
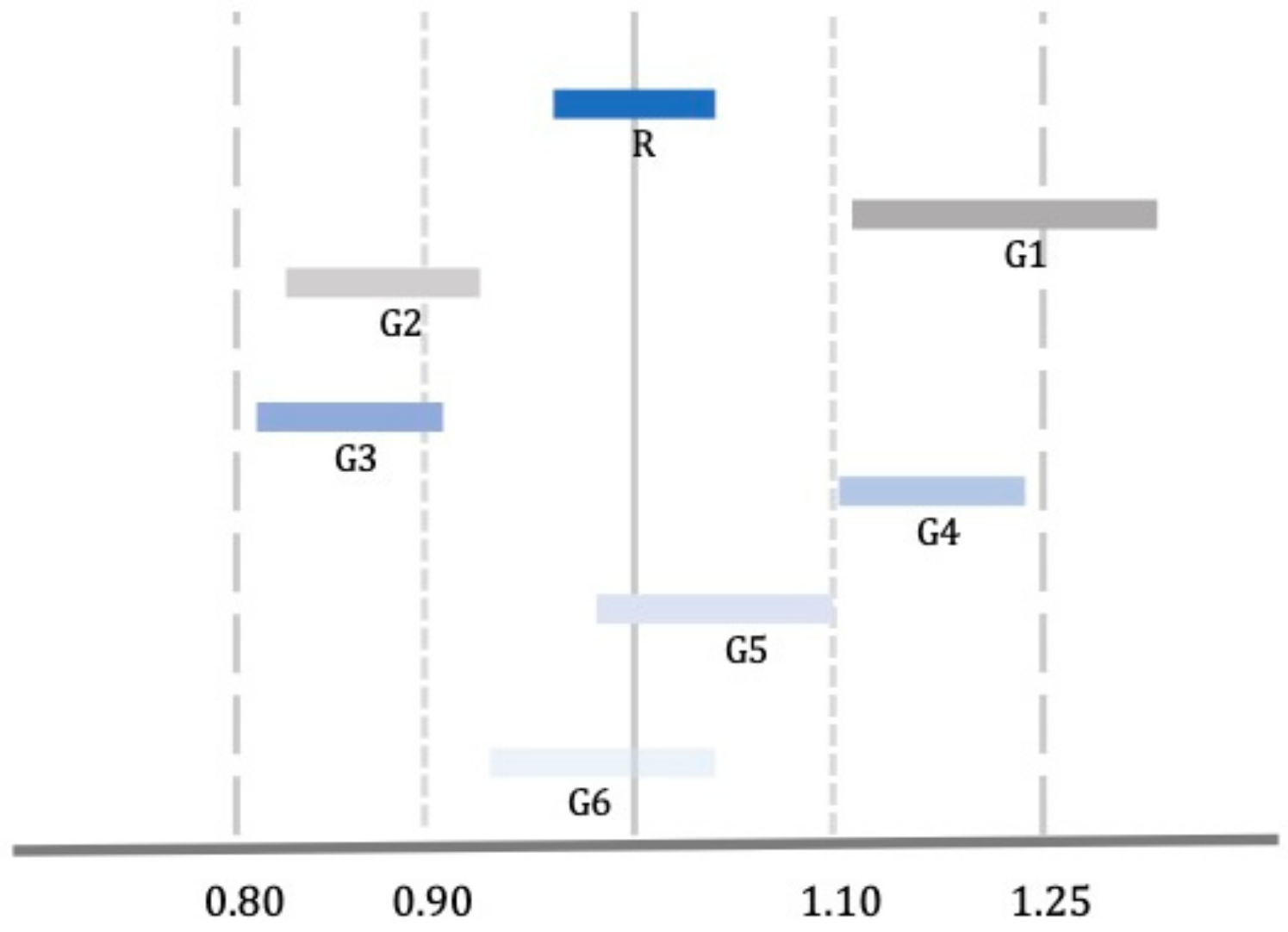
| Agency | Bioequivalence Criteria | Bioequivalence Criteria for Narrow Therapeutic Index Drugs |
|---|---|---|
| European Medicines Agency (EMA) | 80.00–125.00% | 90.00–111.11% |
| U.S. Food and Drug Administration (FDA) | 80.00–125.00% | 90.00–111.11% |
| Japanese Institute of Health Sciences (National Institute of Health Sciences, NIHS) | 80.00–125.00% | 90.00–111.11% |
| Health Protection and Food Branch (HPFB) of Canada | 80.00–125.00% | 90.00–112.00% |
| Category | Drugs |
|---|---|
| Anticoagulants | Vitamin K antagonists, Heparin |
| Antiepileptic drugs | Valproic acid, Phenobarbital, Phenytoin, Carbamazepine |
| Aminoglycosides | Streptomycin, Kanamycin, Netilmicin, Tobramycin, Neomycin |
| Immunosuppressant | Cyclosporine, Sirolimus, Mycophenolic acid |
| Glycoside | Digoxin, Digitoxin |
| Mood-stabilizing agent | Lithium carbonate |
| Central analgesics | Opioids |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gozzo, L.; Caraci, F.; Drago, F. Bioequivalence, Drugs with Narrow Therapeutic Index and the Phenomenon of Biocreep: A Critical Analysis of the System for Generic Substitution. Healthcare 2022, 10, 1392. https://doi.org/10.3390/healthcare10081392
Gozzo L, Caraci F, Drago F. Bioequivalence, Drugs with Narrow Therapeutic Index and the Phenomenon of Biocreep: A Critical Analysis of the System for Generic Substitution. Healthcare. 2022; 10(8):1392. https://doi.org/10.3390/healthcare10081392
Chicago/Turabian StyleGozzo, Lucia, Filippo Caraci, and Filippo Drago. 2022. "Bioequivalence, Drugs with Narrow Therapeutic Index and the Phenomenon of Biocreep: A Critical Analysis of the System for Generic Substitution" Healthcare 10, no. 8: 1392. https://doi.org/10.3390/healthcare10081392
APA StyleGozzo, L., Caraci, F., & Drago, F. (2022). Bioequivalence, Drugs with Narrow Therapeutic Index and the Phenomenon of Biocreep: A Critical Analysis of the System for Generic Substitution. Healthcare, 10(8), 1392. https://doi.org/10.3390/healthcare10081392








