Osteopathic Manipulative Treatment Regulates Autonomic Markers in Preterm Infants: A Randomized Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Trial Design
2.2. Participants
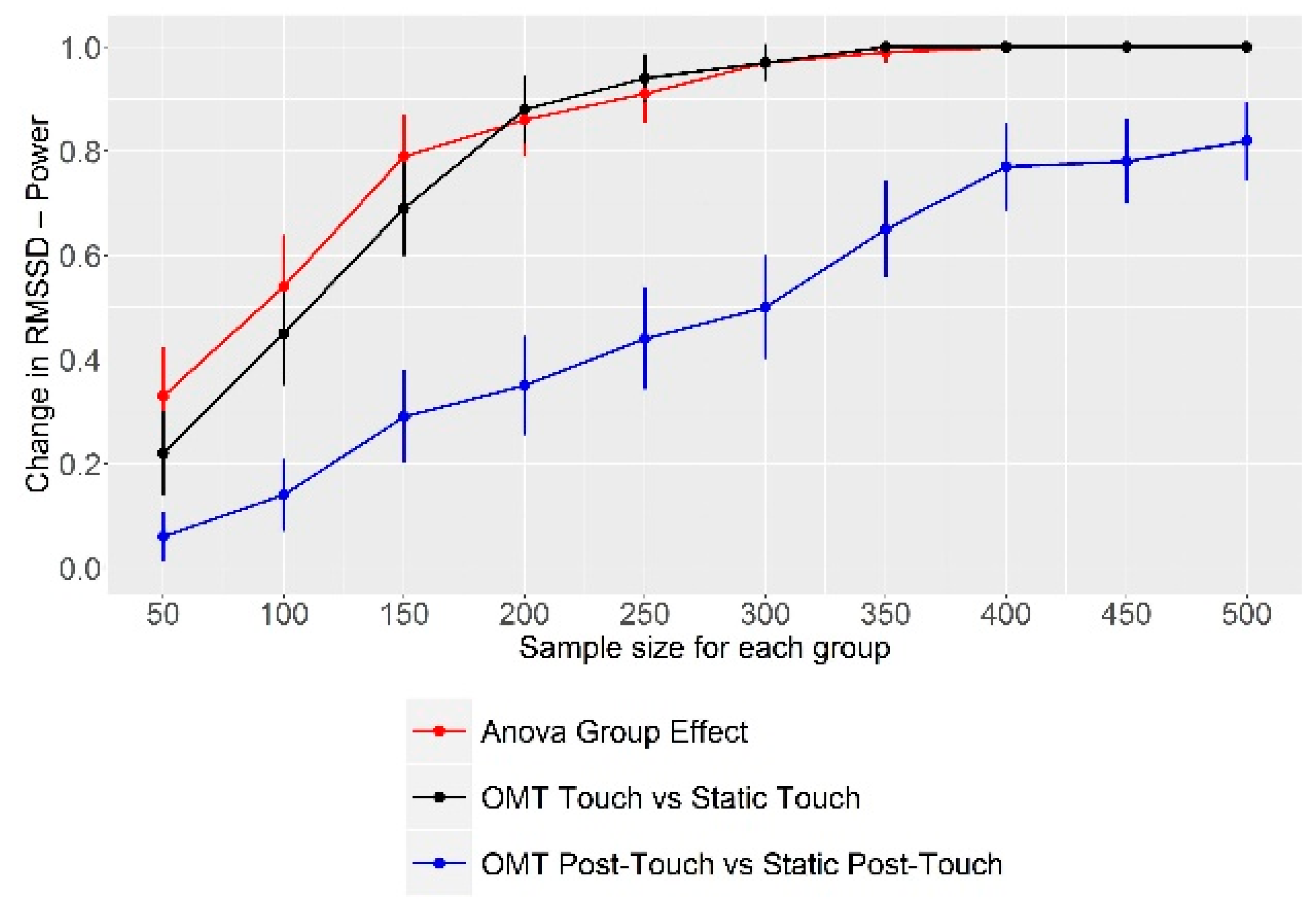
2.3. Sample Size
2.4. Randomization
2.5. Allocation Concealment
2.6. Physiological Monitoring and Data Collection
2.7. Interventions
2.8. Data Preprocessing and Extraction
2.9. Primary and Secondary Outcomes
2.10. Statistical Analysis
3. Results
3.1. General Characteristics
3.2. HRV Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liu, L.; Johnson, H.L.; Cousens, S.; Perin, J.; Scott, S.; Lawn, J.E.; Rudan, I.; Campbell, H.; Cibulskis, R.; Li, M.; et al. Global, regional, and national causes of child mortality: An updated systematic analysis for 2010 with time trends since 2000. Lancet 2012, 379, 2151–2161. [Google Scholar] [CrossRef]
- World Health Organization. World Health Statistics 2016: Monitoring Health for the SDGs, Sustainable Development Goals; World Health Organization: Geneva, Switzerland, 2016; ISBN 978-92-4-156526-4. [Google Scholar]
- Zeiner, V.; Storm, H.; Doheny, K.K. Preterm infants’ behaviors and skin conductance responses to nurse handling in the NICU. J. Matern.-Fetal Neonatal Med. 2016, 29, 2531–2536. [Google Scholar] [CrossRef] [PubMed]
- Weber, A.; Harrison, T.M. Reducing toxic stress in the neonatal intensive care unit to improve infant outcomes. Nurs. Outlook 2019, 67, 169–189. [Google Scholar] [CrossRef] [PubMed]
- Vinall, J.; Grunau, R.E.; Brant, R.; Chau, V.; Poskitt, K.J.; Synnes, A.R.; Miller, S.P. Slower postnatal growth is associated with delayed cerebral cortical maturation in preterm newborns. Sci. Transl. Med. 2013, 5, 168ra8. [Google Scholar] [CrossRef]
- McAnulty, G.B.; Butler, S.C.; Bernstein, J.H.; Als, H.; Duffy, F.H.; Zurakowski, D. Effects of the newborn individualized developmental care and assessment program (NIDCAP) at age 8 years: Preliminary data. Clin. Pediatr. 2010, 49, 258–270. [Google Scholar] [CrossRef]
- Manzotti, A.; Cerritelli, F.; Lombardi, E.; la Rocca, S.; Chiera, M.; Galli, M.; Lista, G. Effects of osteopathic treatment versus static touch on heart rate and oxygen saturation in premature babies: A randomized controlled trial. Complement. Ther. Clin. Pract. 2020, 39, 101116. [Google Scholar] [CrossRef]
- Morton, S.U.; Brodsky, D. Fetal physiology and the transition to extrauterine life. Clin. Perinatol. 2016, 43, 395–407. [Google Scholar] [CrossRef]
- Mulkey, S.B.; du Plessis, A. The critical role of the central autonomic nervous system in fetal-neonatal transition. Semin. Pediatr. Neurol. 2018, 28, 29–37. [Google Scholar] [CrossRef]
- Garzoni, L.; Faure, C.; Frasch, M.G. Fetal cholinergic anti-inflammatory pathway and necrotizing enterocolitis: The brain-gut connection begins in utero. Front. Integr. Neurosci. 2013, 7, 57. [Google Scholar] [CrossRef]
- Javorka, K.; Lehotska, Z.; Kozar, M.; Uhrikova, Z.; Kolarovszki, B.; Javorka, M.; Zibolen, M. Heart rate variability in newborns. Physiol. Res. 2017, 66, S203–S214. [Google Scholar] [CrossRef]
- Chiera, M.; Cerritelli, F.; Casini, A.; Barsotti, N.; Boschiero, D.; Cavigioli, F.; Corti, C.G.; Manzotti, A. Heart rate variability in the perinatal period: A critical and conceptual review. Front. Neurosci. 2020, 14, 561186. [Google Scholar] [CrossRef]
- Cerritelli, F.; Frasch, M.G.; Antonelli, M.C.; Viglione, C.; Vecchi, S.; Chiera, M.; Manzotti, A. A review on the vagus nerve and autonomic nervous system during fetal development: Searching for critical windows. Front. Neurosci. 2021, 15, 721605. [Google Scholar] [CrossRef]
- Selig, F.A.; Tonolli, E.R.; da Silva, É.V.C.M.; de Godoy, M.F. Variabilidadedafrequênciacardíacaemneonatosprematuros e de termo. Arq. Bras. Cardiol. 2011, 96, 443–449. [Google Scholar] [CrossRef]
- Patural, H.; Pichot, V.; Flori, S.; Giraud, A.; Franco, P.; Pladys, P.; Beuchée, A.; Roche, F.; Barthelemy, J.-C. Autonomic maturation from birth to 2 years: Normative values. Heliyon 2019, 5, e01300. [Google Scholar] [CrossRef]
- Thayer, J.F.; Sternberg, E. Beyond heart rate variability: Vagal regulation of allostatic systems. Ann. N. Y. Acad. Sci. 2006, 1088, 361–372. [Google Scholar] [CrossRef]
- Mulkey, S.B.; Hitchings, L.; Persaud, R.; Kota, S.; Maxwell, G.L.; Baker, R.; du Plessis, A.; Govindan, R. Cerebral cortical autonomic connectivity in low-risk term newborns. Clin. Auton. Res. 2021, 31, 415–424. [Google Scholar] [CrossRef]
- Frasch, M.G. Heart rate variability code: Does it exist and can we hack it? arXiv 2020, arXiv:200108264. Available online: https://arxiv.org/ftp/arxiv/papers/2001/2001.08264.pdf (accessed on 26 April 2022).
- Suga, A.; Uraguchi, M.; Tange, A.; Ishikawa, H.; Ohira, H. Cardiac interaction between mother and infant: Enhancement of heart rate variability. Sci. Rep. 2019, 9, 20019. [Google Scholar] [CrossRef]
- Fairchild, K.D.; O’Shea, T.M. Heart rate characteristics: Physiomarkers for detection of late-onset neonatal sepsis. Clin. Perinatol. 2010, 37, 581–598. [Google Scholar] [CrossRef]
- Mulkey, S.B.; du Plessis, A.J. Autonomic nervous system development and its impact on neuropsychiatric outcome. Pediatr. Res. 2019, 85, 120–126. [Google Scholar] [CrossRef]
- Hofer, M.A. Early relationships as regulators of infant physiology and behavior. Acta Paediatr. 1994, 83, 9–18. [Google Scholar] [CrossRef]
- Carozza, S.; Leong, V. The role of affectionate caregiver touch in early neurodevelopment and parent–infant interactional synchrony. Front. Neurosci. 2021, 14, 613378. [Google Scholar] [CrossRef]
- McGlone, F.; Wessberg, J.; Olausson, H. Discriminative and affective touch: Sensing and feeling. Neuron 2014, 82, 737–755. [Google Scholar] [CrossRef]
- Van Puyvelde, M.; Gorissen, A.-S.; Pattyn, N.; McGlone, F. Does touch matter? The impact of stroking versus non-stroking maternal touch on cardio-respiratory processes in mothers and infants. Physiol. Behav. 2019, 207, 55–63. [Google Scholar] [CrossRef]
- Pawling, R.; Cannon, P.R.; McGlone, F.P.; Walker, S.C. C-tactile afferent stimulating touch carries a positive affective value. PLoS ONE 2017, 12, e0173457. [Google Scholar] [CrossRef]
- Norholt, H. Revisiting the roots of attachment: A review of the biological and psychological effects of maternal skin-to-skin contact and carrying of full-term infants. Infant Behav. Dev. 2020, 60, 101441. [Google Scholar] [CrossRef]
- Crucianelli, L.; Wheatley, L.; Filippetti, M.L.; Jenkinson, P.M.; Kirk, E.; Fotopoulou, A.K. The mindedness of maternal touch: An investigation of maternal mind-mindedness and mother-infant touch interactions. Dev. Cogn. Neurosci. 2019, 35, 47–56. [Google Scholar] [CrossRef]
- Montirosso, R.; McGlone, F. The body comes first. Embodied reparation and the co-creation of infant bodily-self. Neurosci. Biobehav. Rev. 2020, 113, 77–87. [Google Scholar] [CrossRef]
- Mariani Wigley, I.L.C.; Mascheroni, E.; Fontana, C.; Giorda, R.; Morandi, F.; Bonichini, S.; McGlone, F.; Fumagalli, M.; Montirosso, R. The role of maternal touch in the association between SLC6A4 methylation and stress response in very preterm infants. Dev. Psychobiol. 2021, 63, e22218. [Google Scholar] [CrossRef]
- Provenzi, L.; Fumagalli, M.; Giorda, R.; Morandi, F.; Sirgiovanni, I.; Pozzoli, U.; Mosca, F.; Borgatti, R.; Montirosso, R. Maternal sensitivity buffers the association between SLC6A4 methylation and socio-emotional stress response in 3-month-old full term, but not very preterm infants. Front. Psychiatry 2017, 8, 171. [Google Scholar] [CrossRef]
- Bystrova, K. Novel mechanism of human fetal growth regulation: A potential role of lanugo, vernix caseosa and a second tactile system of unmyelinated low-threshold c-afferents. Med. Hypotheses 2009, 72, 143–146. [Google Scholar] [CrossRef] [PubMed]
- Boundy, E.O.; Dastjerdi, R.; Spiegelman, D.; Fawzi, W.W.; Missmer, S.A.; Lieberman, E.; Kajeepeta, S.; Wall, S.; Chan, G.J. Kangaroo mother care and neonatal outcomes: A meta-analysis. Pediatrics 2016, 137, e20152238. [Google Scholar] [CrossRef] [PubMed]
- Croy, I.; Luong, A.; Triscoli, C.; Hofmann, E.; Olausson, H.; Sailer, U. Interpersonal stroking touch is targeted to C tactile afferent activation. Behav. Brain Res. 2016, 297, 37–40. [Google Scholar] [CrossRef] [PubMed]
- Maastrup, R.; Greisen, G. Extremely preterm infants tolerate skin-to-skin contact during the first weeks of life: Skin-to-skin contact in extremely preterm infants. Acta Paediatr. 2010, 99, 1145–1149. [Google Scholar] [CrossRef]
- McGlone, F.; Cerritelli, F.; Walker, S.; Esteves, J. The role of gentle touch in perinatal osteopathic manual therapy. Neurosci. Biobehav. Rev. 2017, 72, 1–9. [Google Scholar] [CrossRef]
- Lanaro, D.; Ruffini, N.; Manzotti, A.; Lista, G. Osteopathic manipulative treatment showed reduction of length of stay and costs in preterm infants: A systematic review and meta-analysis. Medicine 2017, 96, e6408. [Google Scholar] [CrossRef]
- Cerritelli, F.; Cardone, D.; Pirino, A.; Merla, A.; Scoppa, F. Does osteopathic manipulative treatment induce autonomic changes in healthy participants? A thermal imaging study. Front. Neurosci. 2020, 14, 887. [Google Scholar] [CrossRef]
- Ruffini, N.; D’Alessandro, G.; Mariani, N.; Pollastrelli, A.; Cardinali, L.; Cerritelli, F. Variations of high frequency parameter of heart rate variability following osteopathic manipulative treatment in healthy subjects compared to control group and sham therapy: Randomized controlled trial. Front. Neurosci. 2015, 9, 272. [Google Scholar] [CrossRef]
- Carnevali, L.; Lombardi, L.; Fornari, M.; Sgoifo, A. Exploring the effects of osteopathic manipulative treatment on autonomic function through the lens of heart rate variability. Front. Neurosci. 2020, 14, 1075. [Google Scholar] [CrossRef]
- Fornari, M.; Carnevali, L.; Sgoifo, A. Single osteopathic manipulative therapy session dampens acute autonomic and neuroendocrine responses to mental stress in healthy male participants. J. Osteopath. Med. 2017, 117, 559–567. [Google Scholar] [CrossRef]
- Sgoifo, A.; Braglia, F.; Costoli, T.; Musso, E.; Meerlo, P.; Ceresini, G.; Troisi, A. Cardiac autonomic reactivity and salivary cortisol in men and women exposed to social stressors: Relationship with individual ethological profile. Neurosci. Biobehav. Rev. 2003, 27, 179–188. [Google Scholar] [CrossRef]
- Cerritelli, F.; Chiacchiaretta, P.; Gambi, F.; Saggini, R.; Perrucci, M.G.; Ferretti, A. Osteopathy modulates brain–heart interaction in chronic pain patients: An ASL study. Sci. Rep. 2021, 11, 4556. [Google Scholar] [CrossRef]
- Carnevali, L.; Cerritelli, F.; Guolo, F.; Sgoifo, A. Osteopathic manipulative treatment and cardiovascular autonomic parameters in rugby players: A randomized, sham-controlled trial. J. Manip. Physiol. Ther. 2021, 44, 319–329. [Google Scholar] [CrossRef]
- Yiallourou, S.R.; Sands, S.A.; Walker, A.M.; Horne, R.S.C. Maturation of heart rate and blood pressure variability during sleep in term-born infants. Sleep 2012, 35, 177–186. [Google Scholar] [CrossRef]
- Schlatterer, S.D.; Govindan, R.B.; Barnett, S.D.; Al-Shargabi, T.; Reich, D.A.; Iyer, S.; Hitchings, L.; Larry Maxwell, G.; Baker, R.; du Plessis, A.J.; et al. Autonomic development in preterm infants is associated with morbidity of prematurity. Pediatr. Res. 2022, 91, 171–177. [Google Scholar] [CrossRef]
- Manzotti, A.; Cerritelli, F.; Chiera, M.; Lombardi, E.; la Rocca, S.; Biasi, P.; Galli, M.; Esteves, J.; Lista, G. Neonatal assessment manual score: Is there a role of a novel, structured touch-based evaluation in neonatal intensive care unit? Front. Pediatr. 2020, 8, 432. [Google Scholar] [CrossRef]
- Cerritelli, F.; Martelli, M.; Renzetti, C.; Pizzolorusso, G.; Cozzolino, V.; Barlafante, G. Introducing an osteopathic approach into neonatology ward: The NE-O model. Chiropr. Man. Ther. 2014, 22, 18. [Google Scholar] [CrossRef]
- Pizzolorusso, G.; Cerritelli, F.; Accorsi, A.; Lucci, C.; Tubaldi, L.; Lancellotti, J.; Barlafante, G.; Renzetti, C.; D’Incecco, C.; Perri, F.P. The effect of optimally timed osteopathic manipulative treatment on length of hospital stay in moderate and late preterm infants: Results from a RCT. Evid.-Based Complement. Altern. Med. 2014, 2014, 243539. [Google Scholar] [CrossRef]
- Preprocessing of HRV Data. Available online: https://www.kubios.com/hrv-preprocessing/ (accessed on 18 January 2022).
- Shaffer, F.; Ginsberg, J.P. An overview of heart rate variability metrics and norms. Front. Public Health 2017, 5, 258. [Google Scholar] [CrossRef]
- Cardoso, S.; Silva, M.J.; Guimarães, H. Autonomic nervous system in newborns: A review based on heart rate variability. Child’s Nerv. Syst. 2017, 33, 1053–1063. [Google Scholar] [CrossRef]
- Dimitrijević, L.; Bjelaković, B.; Čolović, H.; Mikov, A.; Živković, V.; Kocić, M.; Lukić, S. Assessment of general movements and heart rate variability in prediction of neurodevelopmental outcome in preterm infants. Early Hum. Dev. 2016, 99, 7–12. [Google Scholar] [CrossRef]
- Marsillio, L.E.; Manghi, T.; Carroll, M.S.; Balmert, L.C.; Wainwright, M.S. Heart rate variability as a marker of recovery from critical illness in children. PLoS ONE 2019, 14, e0215930. [Google Scholar] [CrossRef]
- Ferrario, M.; Signorini, M.G.; Magenes, G.; Cerutti, S. Comparison of entropy-based regularity estimators: Application to the fetal heart rate signal for the identification of fetal distress. IEEE Trans. Biomed. Eng. 2006, 53, 119–125. [Google Scholar] [CrossRef]
- Butruille, L.; de Jonckheere, J.; Marcilly, R.; Boog, C.; Bras da Costa, S.; Rakza, T.; Storme, L.; Logier, R. Development of a pain monitoring device focused on newborn infant applications: The NeoDoloris project. IRBM 2015, 36, 80–85. [Google Scholar] [CrossRef]
- Weissman, A.; Zimmer, E.Z.; Aranovitch, M.; Blazer, S. Heart rate dynamics during acute pain in newborns. Pflüg. Arch.-Eur. J. Physiol. 2012, 464, 593–599. [Google Scholar] [CrossRef]
- Kumar, N.; Akangire, G.; Sullivan, B.; Fairchild, K.; Sampath, V. Continuous vital sign analysis for predicting and preventing neonatal diseases in the twenty-first century: Big data to the forefront. Pediatr. Res. 2020, 87, 210–220. [Google Scholar] [CrossRef]
- Joshi, R.; Kommers, D.; Oosterwijk, L.; Feijs, L.; van Pul, C.; Andriessen, P. Predicting neonatal sepsis using features of heart rate variability, respiratory characteristics, and ECG-derived estimates of infant motion. IEEE J. Biomed. Health Inform. 2020, 24, 681–692. [Google Scholar] [CrossRef]
- Hardstone, R.; Poil, S.-S.; Schiavone, G.; Jansen, R.; Nikulin, V.V.; Mansvelder, H.D.; Linkenkaer-Hansen, K. Detrended fluctuation analysis: A scale-free view on neuronal oscillations. Front. Physiol. 2012, 3, 450. [Google Scholar] [CrossRef] [PubMed]
- Herry, C.L.; Burns, P.; Desrochers, A.; Fecteau, G.; Durosier, L.D.; Cao, M.; Seely, A.J.E.; Frasch, M.G. Vagal contributions to fetal heart rate variability: An omics approach. Physiol. Meas. 2019, 40, 065004. [Google Scholar] [CrossRef] [PubMed]
- HRV in Evaluating ANS Function. Available online: https://www.kubios.com/hrv-ans-function/ (accessed on 18 January 2022).
- Hayano, J.; Yuda, E. Pitfalls of assessment of autonomic function by heart rate variability. J. Physiol. Anthropol. 2019, 38, 3. [Google Scholar] [CrossRef] [PubMed]
- Andersen, M.; Andelius, T.C.K.; Pedersen, M.V.; Kyng, K.J.; Henriksen, T.B. Severity of hypoxic ischemic encephalopathy and heart rate variability in neonates: A systematic review. BMC Pediatr. 2019, 19, 242. [Google Scholar] [CrossRef]
- Marvin, M.; Gardner, F.; Sarsfield, K.; Travagli, R.; Doheny, K. Increased frequency of skin-to-skin contact is associated with enhanced vagal tone and improved health outcomes in preterm neonates. Am. J. Perinatol. 2019, 36, 505–510. [Google Scholar] [CrossRef]
- Kommers, D.R.; Joshi, R.; van Pul, C.; Feijs, L.; Bambang Oetomo, S.; Andriessen, P. Changes in autonomic regulation due to kangaroo care remain unaffected by using a swaddling device. Acta Paediatr. 2019, 108, 258–265. [Google Scholar] [CrossRef]
- Cong, X.; Ludington-Hoe, S.M.; McCain, G.; Fu, P. Kangaroo care modifies preterm infant heart rate variability in response to heel stick pain: Pilot study. Early Hum. Dev. 2009, 85, 561–567. [Google Scholar] [CrossRef]
- Cong, X.; Cusson, R.M.; Walsh, S.; Hussain, N.; Ludington-Hoe, S.M.; Zhang, D. Effects of skin-to-skin contact on autonomic pain responses in preterm infants. J. Pain 2012, 13, 636–645. [Google Scholar] [CrossRef]
- Butruille, L.; Blouin, A.; de Jonckheere, J.; Mur, S.; Margez, T.; Rakza, T.; Storme, L. Impact of skin-to-skin contact on the autonomic nervous system in the preterm infant and his mother. Infant Behav. Dev. 2017, 49, 83–86. [Google Scholar] [CrossRef]
- Vogl, J.L.; Dunne, E.C.; Liu, C.; Bradley, A.; Rwei, A.; Lonergan, E.K.; Hopkins, B.S.; Kwak, S.S.; Simon, C.D.; Rand, C.M.; et al. Kangaroo father care: A pilot feasibility study of physiologic, biologic, and psychosocial measures to capture the effects of father–infant and mother–infant skin-to-skin contact in the neonatal intensive care unit. Dev. Psychobiol. 2021, 63, 1521–1533. [Google Scholar] [CrossRef]
- Özdemir, R.; Olukman, Ö.; Karadeniz, C.; Çelik, K.; Katipoğlu, N.; Muhtar Yılmazer, M.; Çalkavur, Ş.; Meşe, T.; Arslanoğlu, S. Effect of unconjugated hyperbilirubinemia on neonatal autonomic functions: Evaluation by heart rate variability. J. Matern. Fetal Neonatal Med. 2018, 31, 2763–2769. [Google Scholar] [CrossRef]
- Hoyer, D.; Schmidt, A.; Gustafson, K.M.; Lobmaier, S.M.; Lakhno, I.; van Leeuwen, P.; Cysarz, D.; Preisl, H.; Schneider, U. Heart rate variability categories of fluctuation amplitude and complexity: Diagnostic markers of fetal development and its disturbances. Physiol. Meas. 2019, 40, 064002. [Google Scholar] [CrossRef]
- Metzler, M.; Govindan, R.; Al-Shargabi, T.; Vezina, G.; Andescavage, N.; Wang, Y.; du Plessis, A.; Massaro, A.N. Pattern of brain injury and depressed heart rate variability in newborns with hypoxic ischemic encephalopathy. Pediatr. Res. 2017, 82, 438–443. [Google Scholar] [CrossRef]
- Ceunen, E.; Vlaeyen, J.W.S.; van Diest, I. On the origin of interoception. Front. Psychol. 2016, 7, 743. [Google Scholar] [CrossRef]
- D’Alessandro, G.; Cerritelli, F.; Cortelli, P. Sensitization and interoception as key neurological concepts in osteopathy and other manual medicines. Front. Neurosci. 2016, 10, 100. [Google Scholar] [CrossRef]
- Cerritelli, F.; Chiacchiaretta, P.; Gambi, F.; Ferretti, A. Effect of continuous touch on brain functional connectivity is modified by the operator’s tactile attention. Front. Hum. Neurosci. 2017, 11, 368. [Google Scholar] [CrossRef]
- Zahr, L.K.; Balian, S. Responses of premature infants to routine nursing interventions and noise in the NICU. Nurs. Res. 1995, 44, 179–185. [Google Scholar] [CrossRef]
- Frasch, M.G.; Szynkaruk, M.; Prout, A.P.; Nygard, K.; Cao, M.; Veldhuizen, R.; Hammond, R.; Richardson, B.S. Decreased neuroinflammation correlates to higher vagus nerve activity fluctuations in near-term ovine fetuses: A case for the afferent cholinergic anti-inflammatory pathway? J. Neuroinflamm. 2016, 13, 103. [Google Scholar] [CrossRef]
- Bonaz, B.; Sinniger, V.; Pellissier, S. Therapeutic potential of vagus nerve stimulation for inflammatory bowel diseases. Front. Neurosci. 2021, 15, 650971. [Google Scholar] [CrossRef]
- Bravi, A.; Longtin, A.; Seely, A.J. Review and classification of variability analysis techniques with clinical applications. Biomed. Eng. Online 2011, 10, 90. [Google Scholar] [CrossRef]
| Parameter | Unit | Definition |
|---|---|---|
| HR | bpm | Number of heart beats per minute |
| RRI | n. | RR intervals obtained through the formula 60,000/HR |
| Time-domain | ||
| SDNN | ms | Standard deviation of NN intervals |
| RMSSD | ms | Root mean square of consecutive RR interval differences |
| Frequency-domain | ||
| LF Power | % | Relative power of the low-frequency band (0.04–0.2 Hz for newborns and 0.04–0.15 Hz for infants), obtained through the division of the absolute LF power by the summed absolute power of the LF and HF bands |
| HF Power | % | Relative power of the high-frequency band (0.20–2.00 Hz for newborns and 0.20–1.40 Hz for infants), obtained through the division of the absolute HF power by the summed absolute power of the LF and HF bands |
| Non-linear | ||
| ApEn | Approximate entropy, which measures the regularity and complexity of a time series (mathematically speaking, ApEn is the negative natural logarithm of the conditional probability that a dataset of length N, having repeated itself for m samples within a tolerance r, will repeat itself again for one extra sample) | |
| SampEn | Sample entropy, which measures the regularity and complexity of a time series (mathematically speaking, SampEN is obtained in the same way as ApEn, but excludes the counts where a vector is compared with itself) | |
| DFA1 | Detrended fluctuation analysis, which extracts the correlations between successive RR intervals over different time scales. Specifically DFA1 describes short-term fluctuations | |
| Composite | ||
| PNS index | An index regarding the parasympathetic nervous system modulation computed by the software Kubios through the analysis of the following metrics: mean RRI (longer RRI is tied to higher vagal modulation), RMSSD (it reflects vagal modulation on HR), and Poincaré plot index SD1 in normalized units (a non-linear metric tied to RMSSD) | |
| SNS index | An index regarding the sympathetic nervous system modulation computed by the software Kubios through the analysis of the following metrics: mean RRI (shorter RRI is tied to higher sympathetic modulation), Baevsky’s stress index (a geometric parameter reflecting cardiovascular stress), and Poincaré plot index SD2 in normalized units (a non-linear metric tied to SDNN) | |
| Characteristic | OMT (N = 50) | Static Touch (N = 46) | p-Value |
|---|---|---|---|
| Gestational age (weeks) | 32.9 ± 4.4 | 33.9 ± 4.2 | 0.27 |
| Birthweight (grams) | 1967 ± 910 | 2173 ± 948 | 0.28 |
| Sex | 20 (40) | 21 (46) | 0.72 |
| Heart rate | 139.8 ± 16.3 | 145.6 ± 12.4 | <0.001 |
| Group Comparison | RMSSD Difference (95% CI) | p-Value |
|---|---|---|
| Static T0–OMT T0 | −0.813 (−2.033, 0.407) | 0.394 |
| Static T1–OMT T1 | −0.724 (−1.944, 0.496) | 0.529 |
| Static T2–OMT T2 | −0.433 (−1.658, 0.791) | 0.911 |
| Static T1–Static T0 | 0.313 (−0.547, 1.172) | 0.902 |
| Static T2–Static T1 | −0.297 (−1.163, 0.569) | 0.922 |
| Static T2–Static T0 | 0.016 (−0.850, 0.882) | 1.000 |
| OMT T1–OMT T0 | 0.223 (−0.601, 1.048) | 0.971 |
| OMT T2–OMT T1 | −0.587 (−1.412, 0.237) | 0.318 |
| OMT T2–OMT T0 | −0.364 (−1.188, 0.461) | 0.801 |
| Group Comparison | SDNN Difference (95% CI) | p-Value |
|---|---|---|
| Static T0–OMT T0 | −1.746 (−5.310, 1.818) | 0.722 |
| Static T1–OMT T1 | −0.636 (−4.201, 2.928) | 0.996 |
| Static T2–OMT T2 | 0.577 (−3.001, 4.154) | 0.997 |
| Static T1–Static T0 | 1.471 (−1.083, 4.026) | 0.563 |
| Static T2–Static T1 | −0.465 (−3.038, 2.109) | 0.995 |
| Static T2–Static T0 | 1.007 (−1.567, 3.581) | 0.871 |
| OMT T1–OMT T0 | 0.362 (−2.089, 2.812) | 0.998 |
| OMT T2–OMT T1 | −1.677 (−4.128, 0.773) | 0.365 |
| OMT T2–OMT T0 | −1.316 (−3.766, 1.135) | 0.637 |
| Group Comparison | PNS Index Difference (95% CI) | p-Value |
|---|---|---|
| Static T0–OMT T0 | −0.199 (−0.422, 0.024) | 0.110 |
| Static T1–OMT T1 | −0.864 (−1.088, −0.641) | <0.001 |
| Static T2–OMT T2 | −0.894 (−1.118, −0.670) | <0.001 |
| Static T1–Static T0 | −0.310 (−0.439, −0.181) | <0.001 |
| Static T2–Static T1 | −0.007 (−0.137, 0.124) | 1.000 |
| Static T2–Static T0 | −0.317 (−0.447, −0.186) | <0.001 |
| OMT T1–OMT T0 | 0.355 (0.231, 0.479) | <0.001 |
| OMT T2–OMT T1 | 0.023 (−0.101, 0.147) | 0.995 |
| OMT T2–OMT T0 | 0.378 (0.254, 0.502) | <0.001 |
| Group Comparison | SNS Index Difference (95% CI) | p-Value |
|---|---|---|
| Static T0–OMT T0 | 0.487 (−1.814, 2.789) | 0.990 |
| Static T1–OMT T1 | 5.250 (2.948, 7.551) | <0.001 |
| Static T2–OMT T2 | 6.675 (4.364, 8.986) | <0.001 |
| Static T1–Static T0 | 0.952 (−0.766, 2.670) | 0.606 |
| Static T2–Static T1 | 2.796 (1.066, 4.526) | <0.001 |
| Static T2–Static T0 | 3.748 (2.017, 5.478) | <0.001 |
| OMT T1–OMT T0 | −3.810 (−5.458, −2.163) | <0.001 |
| OMT T2–OMT T1 | 1.371 (−0.277, 3.018) | 0.165 |
| OMT T2–OMT T0 | −2.440 (−4.087, −0.792) | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manzotti, A.; Cerritelli, F.; Lombardi, E.; Monzani, E.; Savioli, L.; Esteves, J.E.; Galli, M.; La Rocca, S.; Biasi, P.; Chiera, M.; et al. Osteopathic Manipulative Treatment Regulates Autonomic Markers in Preterm Infants: A Randomized Clinical Trial. Healthcare 2022, 10, 813. https://doi.org/10.3390/healthcare10050813
Manzotti A, Cerritelli F, Lombardi E, Monzani E, Savioli L, Esteves JE, Galli M, La Rocca S, Biasi P, Chiera M, et al. Osteopathic Manipulative Treatment Regulates Autonomic Markers in Preterm Infants: A Randomized Clinical Trial. Healthcare. 2022; 10(5):813. https://doi.org/10.3390/healthcare10050813
Chicago/Turabian StyleManzotti, Andrea, Francesco Cerritelli, Erica Lombardi, Elena Monzani, Luca Savioli, Jorge E. Esteves, Matteo Galli, Simona La Rocca, Pamela Biasi, Marco Chiera, and et al. 2022. "Osteopathic Manipulative Treatment Regulates Autonomic Markers in Preterm Infants: A Randomized Clinical Trial" Healthcare 10, no. 5: 813. https://doi.org/10.3390/healthcare10050813
APA StyleManzotti, A., Cerritelli, F., Lombardi, E., Monzani, E., Savioli, L., Esteves, J. E., Galli, M., La Rocca, S., Biasi, P., Chiera, M., & Lista, G. (2022). Osteopathic Manipulative Treatment Regulates Autonomic Markers in Preterm Infants: A Randomized Clinical Trial. Healthcare, 10(5), 813. https://doi.org/10.3390/healthcare10050813








