Cold-Water Immersion and Sports Massage Can Improve Pain Sensation but Not Functionality in Athletes with Delayed Onset Muscle Soreness
Abstract
1. Introduction
2. Methods
2.1. Participants
2.2. Outcome Measurements
2.3. Delayed-Onset Muscle Soreness Induction Exercise
2.4. Interventions
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Newham, D.; Mills, K.R.; Quigley, B.M.; Edwards, R.H.T. Pain and Fatigue after Concentric and Eccentric Muscle Contractions. Clin. Sci. 1983, 64, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, P.M.; Byrnes, W.C.; McCormick, K.M.; Turcotte, L.P.; White, J.S. Muscle Soreness and Serum Creatine Kinase Activity Following Isometric, Eccentric, and Concentric Exercise. Int. J. Sports Med. 1986, 07, 152–155. [Google Scholar] [CrossRef] [PubMed]
- Cheung, K.H.; Hume, P.A.; Maxwell, L. Delayed onset muscle soreness: Treatment strategies and performance factors. Sports Med. 2003, 33, 145–164. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, R.B. Mechanisms of exercise-induced delayed onset muscular soreness: A brief review. Med. Sci. Sports Exerc. 1984, 16, 529–538. [Google Scholar] [CrossRef]
- Paddon-Jones, D.J.; Quigley, B.M. Effect of cryotherapy on muscle soreness and strength following eccentric exercise. Int. J. Sports Med. 1997, 18, 588–590. [Google Scholar] [CrossRef]
- Proske, U.; Morgan, D.L. Muscle damage from eccentric exercise: Mechanism, mechanical signs, adaptation and clinical applications. J. Physiol. 2001, 537, 333–345. [Google Scholar] [CrossRef]
- Clarkson, P.M.; Sayers, S.P. Etiology of exercise-induced muscle damage. Can. J. Appl. Physiol. 1999, 24, 234–248. [Google Scholar] [CrossRef]
- Clarkson, P.M.; Ebbeling, C. Investigation of serum creatine kinase variability after muscle-damaging exercise. Clin. Sci. 1988, 75, 257–261. [Google Scholar] [CrossRef]
- Fridén, J.; Lieber, R.L. Structural and mechanical basis of exercise-induced muscle injury. Med. Sci. Sports Exerc. 1992, 24, 521–530. [Google Scholar] [CrossRef]
- Gulick, D.T.; Kimura, I.F. Delayed Onset Muscle Soreness: What Is It and How Do We Treat It? J. Sport Rehabil. 1996, 5, 234–243. [Google Scholar] [CrossRef]
- Bobbert, M.F.; Hollander, A.P.; Huijing, P.A. Factors in delayed onset muscular soreness of man. Med. Sci. Sports Exerc. 1986, 18, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Crameri, R.M.; Aagaard, P.; Qvortrup, K.; Langberg, H.; Olesen, J.; Kjær, M. Myofibre damage in human skeletal muscle: Effects of electrical stimulation versus voluntary contraction. J. Physiol. 2007, 583, 365–380. [Google Scholar] [CrossRef]
- Smith, L.L. Acute inflammation: The underlying mechanism in delayed onset muscle soreness? Med. Sci. Sports Exerc. 1991, 23, 542–551. [Google Scholar] [CrossRef]
- MacIntyre, D.L.; Sorichter, S.; Mair, J.; McKenzie, D.C.; Berg, A. Markers of inflammation and myofibrillar proteins following eccentric exercise in humans. Eur. J. Appl. Physiol. 2001, 84, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Nie, H.; Madeleine, P.; Arendt-Nielsen, L.; Graven-Nielsen, T. Temporal summation of pressure pain during muscle hyperalgesia evoked by nerve growth factor and eccentric contractions. Eur. J. Pain 2009, 13, 704–710. [Google Scholar] [CrossRef] [PubMed]
- O’Reilly, K.P.; Warhol, M.J.; Fielding, R.A.; Frontera, W.R.; Meredith, C.N.; Evans, W.J. Eccentric exercise-induced muscle damage impairs muscle glycogen repletion. J. Appl. Physiol. 1987, 63, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Eston, R.G.; Lemmey, A.B.; McHugh, P.; Byrne, C.; Walsh, S.E. Effect of stride length on symptoms of exercise-induced muscle damage during a repeated bout of downhill running. Scand. J. Med. Sci. Sports 2000, 10, 199–204. [Google Scholar] [CrossRef]
- Kraemer, W.J.; Bush, J.A.; Wickham, R.B.; Denegar, C.R.; Gómez, A.L.; Gotshalk, L.A.; Duncan, N.D.; Volek, J.S.; Putukian, M.; Sebastianelli, W.J. Influence of Compression Therapy on Symptoms Following Soft Tissue Injury from Maximal Eccentric Exercise. J. Orthop. Sports Phys. Ther. 2001, 31, 282–290. [Google Scholar] [CrossRef]
- Torres, R.; Appell, H.-J.; Duarte, J.A. Acute Effects of Stretching on Muscle Stiffness After a Bout of Exhaustive Eccentric Exercise. Endoscopy 2007, 28, 590–594. [Google Scholar] [CrossRef]
- Law, L.A.F.; Evans, S.; Knudtson, J.; Nus, S.; Scholl, K.; Sluka, K. Massage Reduces Pain Perception and Hyperalgesia in Experimental Muscle Pain: A Randomized, Controlled Trial. J. Pain 2008, 9, 714–721. [Google Scholar] [CrossRef]
- Itoh, K.; Ochi, H.; Kitakoji, H. Effects of tender point acupuncture on delayed onset muscle soreness (DOMS)—A pragmatic trial. Chin. Med. 2008, 3, 14. [Google Scholar] [CrossRef] [PubMed]
- Ingram, J.; Dawson, B.; Goodman, C.; Wallman, K.; Beilby, J. Effect of water immersion methods on post-exercise recovery from simulated team sport exercise. J. Sci. Med. Sport 2009, 12, 417–421. [Google Scholar] [CrossRef] [PubMed]
- Pinkrah, H. The Effect of Self-Myofascial Release on Recovery and Performance in Resistance Training. Ph.D. Thesis, University of Wales Institute Cardiff, Cardiff, UK, 2012. [Google Scholar]
- Bae, S.-H.; Lee, Y.-S.; Kim, G.-D.; Kim, K.-Y. The Effects of Kinesio-taping Applied to Delayed Onset Muscle Soreness on Changes in Pain. Int. J. Bio-Sci. Bio-Technol. 2014, 6, 133–142. [Google Scholar] [CrossRef]
- Han, J.-H.; Kim, M.-J.; Yang, H.-J.; Lee, Y.-J.; Sung, Y.-H. Effects of therapeutic massage on gait and pain after delayed onset muscle soreness. J. Exerc. Rehabilit. 2014, 10, 136–140. [Google Scholar] [CrossRef]
- Kim, S.K.; Kim, M.C. The Affect on Delayed Onset Muscle Soreness Recovery for Ultrasound with Bee Venom. J. Phys. Ther. Sci. 2014, 26, 1419–1421. [Google Scholar] [CrossRef]
- Best, T.M.; Hunter, R.; Wilcox, A.; Haq, F. Effectiveness of Sports Massage for Recovery of Skeletal Muscle From Strenuous Exercise. Clin. J. Sport Med. 2008, 18, 446–460. [Google Scholar] [CrossRef] [PubMed]
- Ascensão, A.; Leite, M.; Rebelo, A.; Magalhäes, S.; Magalhães, J. Effects of cold water immersion on the recovery of physical performance and muscle damage following a one-off soccer match. J. Sports Sci. 2011, 29, 217–225. [Google Scholar] [CrossRef]
- Klich, S.; Krymski, I.; Michalik, K.; Kawczyński, A. Effect of short-term cold-water immersion on muscle pain sensitivity in elite track cyclists. Phys. Ther. Sport 2018, 32, 42–47. [Google Scholar] [CrossRef]
- Sellwood, K.L.; Brukner, P.; Williams, D.; Nicol, A.; Hinman, R. Ice-water immersion and delayed-onset muscle soreness: A randomised controlled trial. Br. J. Sports Med. 2007, 41, 392–397. [Google Scholar] [CrossRef]
- Chen, M.J.; Fan, X.; Moe, S.T. Criterion-related validity of the Borg ratings of perceived exertion scale in healthy individuals: A meta-analysis. J. Sports Sci. 2002, 20, 873–899. [Google Scholar] [CrossRef]
- Miyama, M.; Nosaka, K. Influence of surface on muscle damage and soreness induced by consecutive drop jumps. Journal of Strength and Conditioning research. J. Strength Cond. Res. 2004, 18, 206–211. [Google Scholar] [PubMed]
- Jakeman, J.R.; Byrne, C.; Eston, R.G. Lower limb compression garment improves recovery from exercise-induced muscle damage in young, active females. Eur. J. Appl. Physiol. 2011, 109, 1137–1144. [Google Scholar] [CrossRef] [PubMed]
- Kirby, T.J.; Triplett, N.T.; Haines, T.L.; Skinner, J.W.; Fairbrother, K.R.; McBride, J.M. Effect of leucine supplementation on indices of muscle damage following drop jumps and resistance exercise. Amino Acids 2011, 42, 1987–1996. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Buchner, A.; Lang, A.G. Statistical power analyses using G*Power 3.1: Tests for correlation and re-gression analyses. Behav. Res. Methods 2009, 41, 149–1160. [Google Scholar] [CrossRef]
- Gill, N.D.; Beaven, C.M.; Cook, C. Effectiveness of post-match recovery strategies in rugby players. Br. J. Sports Med. 2006, 40, 260–263. [Google Scholar] [CrossRef] [PubMed]
- Wigernæs, I.; Høstmark, A.T.; Kierulf, P.; Strømme, S.B. Active Recovery Reduces the Decrease in Circulating White Blood Cells after Exercise. Endoscopy 2000, 21, 608–612. [Google Scholar] [CrossRef] [PubMed]
- Davis, H.L.; Alabed, S.; Chico, T.J.A. Effect of sports massage on performance and recovery: A systematic review and meta-analysis. BMJ Open Sport Exerc. Med. 2020, 6, e000614. [Google Scholar] [CrossRef] [PubMed]
- Hohenauer, E.; Taeymans, J.; Baeyens, J.-P.; Clarys, P.; Clijsen, R. The Effect of Post-Exercise Cryotherapy on Recovery Characteristics: A Systematic Review and Meta-Analysis. PLoS ONE 2015, 10, e0139028. [Google Scholar] [CrossRef]
| Cryotherapy (n = 15) | Sports Massage (n = 15) | Sports Massage-Cryotherapy (n = 15) | Control (n = 15) | ||
|---|---|---|---|---|---|
| Variables | Measurements | Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD |
| BORG | Day-1, pre | 7.4 ± 1.0 | 7.4 ± 1.8 | 7.3 ± 1.2 | 7.7 ± 1.6 |
| Day-1, post | 13.1 ± 2.1 | 11.9 ± 3.1 | 12.1 ± 2.3 | 13.9 ± 2.8 | |
| Day-2 | 11.7 ± 3.6 | 9.6 ± 3.2 | 10.7 ± 1.8 | 10.9 ± 2.5 | |
| Day-3 | 10.4 ± 2.4 | 8.5 ± 2.1 | 9.9 ± 1.9 | 10.9 ± 3.2 | |
| Day-4 | 8.1 ± 1.5 | 7.4 ± 1.6 | 8.3 ± 1.7 | 9.3 ± 3.0 | |
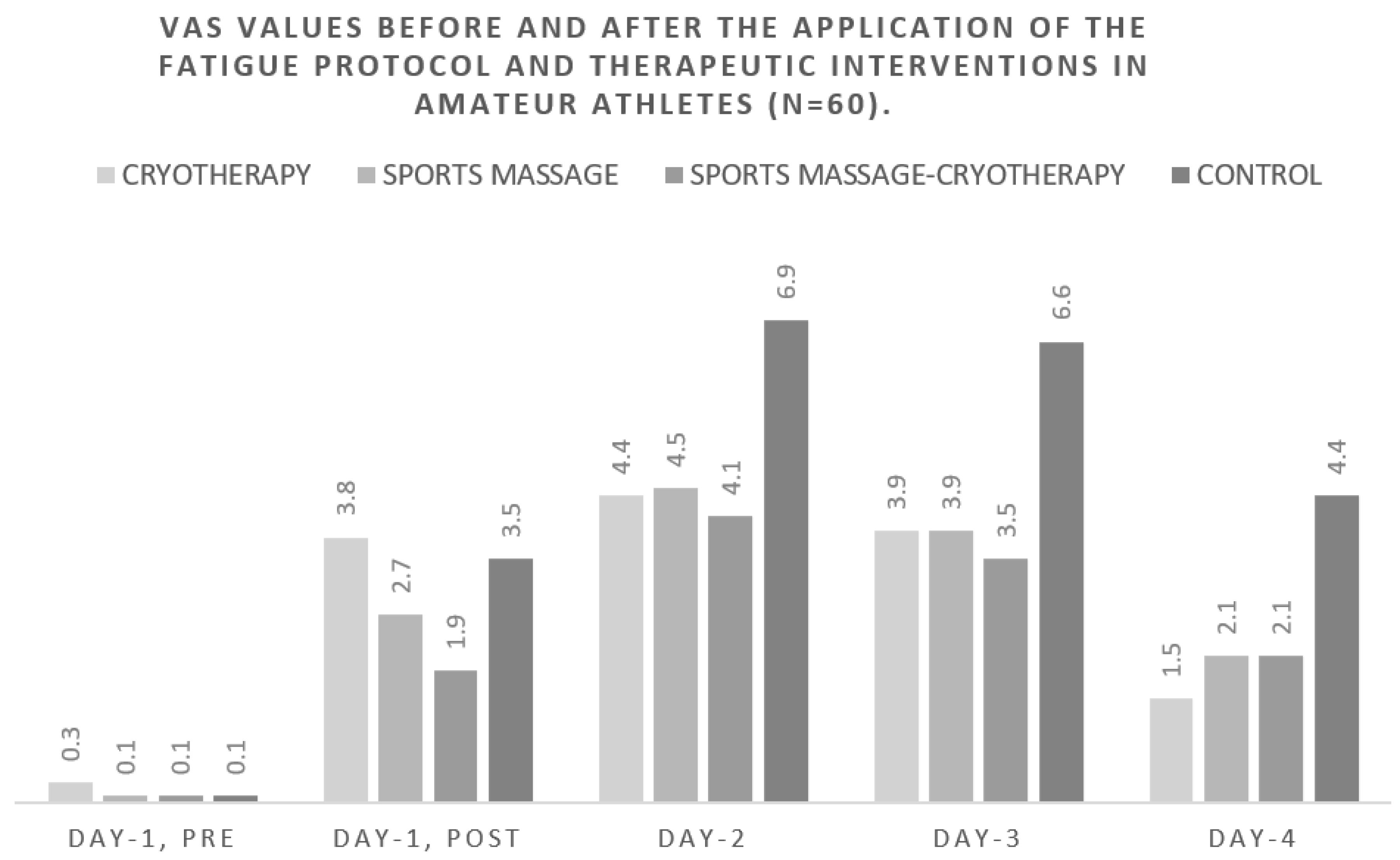
| VAS | Day-1, pre | 0.3 ±0.5 | 0.1 ± 0.4 | 0.1 ± 0.4 | 0.1 ± 0.3 |
| Day-1, post | 3.8 ± 2.1 | 2.7 ± 1.3 | 1.9 ± 1.1 | 3.5 ± 1.8 | |
| Day-2 | 4.4 ± 2.3 | 4.5 ± 1.9 | 4.1 ± 1.7 | 6.9 ± 1.5 | |
| Day-3 | 3.9 ± 1.7 | 3.9 ± 2.1 | 3.5 ± 1.7 | 6.6 ± 1.8 | |
| Day-4 | 1.5 ± 1.0 | 2.1 ± 1.7 | 2.1 ± 1.5 | 4.4 ± 2.0 | |
| ROM | Day-1, pre | 144.3 ± 8.9 | 153.1 ± 5.0 | 151.7 ± 5.1 | 154.2 ± 4.8 |
| Day-1, post | 141.7 ± 9.5 | 152.0 ± 5.1 | 150.2 ± 5.5 | 153.5 ± 5.0 | |
| Day-2 | 140.3 ± 9.4 | 150.9 ± 6.0 | 149.4 ± 5.8 | 152.1± 4.6 | |
| Day-3 | 141.3 ± 9.1 | 152.0 ± 5.2 | 150.4 ± 5.7 | 152.2 ± 5.2 | |
| Day-4 | 143.7 ± 7.8 | 153.1 ± 4.6 | 151.0 ± 5.4 | 153.3 ± 4.8 | |
| STRENGTH | Day-1, pre | 289.4 ± 60.4 | 291.5 ± 58.2 | 283.7± 57.0 | 289.0 ± 67.1 |
| Day-1, post | 260.0 ± 52.8 | 236.1 ± 44.3 | 238.2 ± 57.7 | 235.5 ± 70.6 | |
| Day-2 | 275.7 ± 53.3 | 256.1 ± 43.3 | 252.5 ± 67.3 | 242.5 ± 74.8 | |
| Day-3 | 293.1 ± 41.9 | 268.0 ± 41.8 | 279.0 ± 67.1 | 257.8 ± 81.1 | |
| Day-4 | 321.3 ± 51.9 | 289.6 ± 50.8 | 287.2 ± 70.0 | 274.4 ± 83.0 | |
| Quadriceps circumference (relaxed) | Day-1, pre | 24,819.8 ± 4883.2 | 25,858.3 ± 4740.6 | 27,640.6 ± 5159.1 | 25,783.7 ± 4062.5 |
| Day-1, post | 26,042.2 ± 4588.4 | 27,825.6 ± 5209.7 | 29,171.0 ± 5932.2 | 27,637.8 ± 3469.5 | |
| Day-2 | 25,412.6 ± 4459.5 | 26,735.4 ± 5033.8 | 27,878.7 ± 5215.5 | 27,323.0 ± 3413.2 | |
| Day-3 | 25,578.3 ± 5274.2 | 26,626.8 ± 5859.5 | 27,583.9 ± 5576.0 | 27,181.3 ± 3589.0 | |
| Day-4 | 25,193.4 ± 5640.1 | 26,658.3 ± 6263.2 | 27,067.2 ± 5312.6 | 26,681.0 ± 3864.2 | |
| Quadriceps circumference (contracted) | Day-1, pre | 24,595.7 ± 6955.5 | 23,860.2 ± 7769.0 | 24,377.9 ± 6487.1 | 23,137.5 ± 6101.3 |
| Day-1, post | 26,132.0 ± 6224.0 | 25,350.2 ± 7347.0 | 26,388.2 ± 6814.5 | 24,287.7 ± 5618.8 | |
| Day-2 | 25,104.3 ± 6128.0 | 24,838.2 ± 7609.9 | 24,928.6 ± 5890.2 | 24,048.0 ± 5318.5 | |
| Day-3 | 24,502.2 ± 7244.3 | 24,038.7 ± 7693.8 | 24,637.2 ± 6193.5 | 23,857.7 ± 5586.5 | |
| Day-4 | 24,034.3 ± 6575.3 | 23,600.0 ± 7880.7 | 24,007.9 ± 5816.8 | 23,434.9 ± 5641.0 | |
| CPK | Day-1, pre | 638.4 ± 1546.3 | 263.4 ± 376.9 | 179.5 ± 71.1 | 178.9 ± 61.1 |
| Day-1, post | 1713.1 ± 1928.3 | 1160.6 ± 658.4 | 1236.2 ± 859.5 | 874.9 ± 439.4 | |
| Day-2 | 864.9 ± 756.0 | 599.8 ± 319.2 | 683.7 ± 485.2 | 500.9 ± 346.3 | |
| Day-3 | 472.1 ± 306.4 | 447.5 ± 200.1 | 420.8 ± 292.3 | 342.5 ± 234.1 | |
| Variables | Day-1pre–Day-1post Measurement (for All Participants) | Day-1pre–Day-1post Measurement (Comparison between Groups) | Day-1pre–Day-2 Measurement (for All Participants | Day-1pre–Day-3 Measurement (for All Participants-Time Effect) | Day-1pre–Day-3 Measurement (Comparison between Groups) | 1st–4th Measurement (Comparison between Groups) |
|---|---|---|---|---|---|---|
| BORG (6–20 scale) | F (1, 56) = 31.113, p < 0.001, ηp2 = 0.357 (95% CI: 0.162, 0.511) | F (3, 56) = 1.011, p = 0.395 | F (1, 56) = 69.540, p < 0.001, ηp2 = 0.554 (90% CI: 0.369, 0.668) | F (3, 56) = 0.567, p = 0.639 | F (1, 56) = 179.332, p < 0.001, ηp2 = 0.762 (90% CI: 0.643, 0.825) | F (3, 56) = 0.534, p = 0.661 |
| VAS (0–10 scale) | F (1, 56) = 71.388, p < 0.001, ηp2 = 0.560 (95% CI: 0.377, 0.672) | F (3, 56) = 5.724, p = 0.002, ηp2 = 0.235 (95% CI: 0.042, 0.378) | F (1, 56) = 32.444, p < 0.001, ηp2 = 0.367 (95% CI: 0.170, 0.518) | F (3, 56) = 5.662, p = 0.002, ηp2 = 0.233 (95% CI: 0.041, 0.376) | F (1, 56) = 3.651, p = 0.061, ηp2 = 0.061 (95% CI: 0.000, 0.207) | F (3, 56) = 7.688, p < 0.001, ηp2 = 0.292 (95% CI: 0.082, 0.433) |
| Knee flexion ROM (deg) | F (1, 56) = 10.624, p = 0.002, ηp2 = 0.159 (95% CI: 0.024, 0.326) | F (3, 56) = 0.151, p = 0.929 | F (1, 56) = 1.129, p = 0.293 | F (3, 56) = 0.972, p = 0.413 | F (1, 56) = 9.378, p = 0.003, ηp2 = 0.143 (95% CI: 0.017, 0.309) | F (3, 56) = 2.070, p = 0.114 |
| Knee extension isometric strength (Nm) | F (1, 56) = 14.524, p < 0.001, ηp2 = 0.206 (95% CI: 0.048, 0.373) | F (3, 56) = 0.514, p = 0.674 | F (1, 56) = 54.028, p < 0.001, ηp2 = 0.491 (95% CI: 0.296, 0.619) | F (3, 56) = 0.759, p = 0.522 | F (1, 56) = 119.372, p < 0.001, ηp2 = 0.681 (95% CI: 0.530, 0.764) | F (3, 56) = 1.016 p = 0.393 |
| Quadriceps circumference-relaxed (cm) | F (1, 56) = 21.009, p < 0.001, ηp2 = 0.273 (95% CI: 0.092, 0.436) | F (3, 56) = 1.486, p = 0.228 | F (1, 56) = 15.091, p < 0.001, ηp2 = 0.212 (95% CI: 0.052, 380) | F (3, 56) = 1.385, p = 0.257 | F (1, 56) = 21.704, p < 0.001, ηp2 = 0.279 (95% CI: 0.097, 0.442) | F (3, 56) = 1.102, p = 0.356 |
| Quadriceps circumference -contracted (cm) | F (1, 56) = 13.416, p < 0.001, ηp2 = 0.193 (95% CI: 0.041, 0.361) | F (3, 56) = 1.5060, p = 0.223 | F (1, 56) = 20.205, p < 0.001, ηp2 = 0.265 (95% CI: 0.087, 0.429) | F (3, 56) = 1.096, p = 0.358 | F (1, 56) = 48.002, p < 0.001, ηp2 = 0.462 (95% CI: 0.264, 0.596) | F (3, 56) = 1.687, p = 0.180 |
| CPK(U/L) | F (1, 56) = 30.610, p < 0.001, ηp2 = 0.353 (95% CI: 0.158, 0.507) | F (3, 56) = 0.273, p = 0.845 | F (1, 56) = 8.629, p = 0.005 | F (3, 56) = 0.238, p = 0.869 | F (1, 56) = 0.964, p = 0.331 | F (3, 56) = 0.732, p = 0.537 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Angelopoulos, P.; Diakoronas, A.; Panagiotopoulos, D.; Tsekoura, M.; Xaplanteri, P.; Koumoundourou, D.; Saki, F.; Billis, E.; Tsepis, E.; Fousekis, K. Cold-Water Immersion and Sports Massage Can Improve Pain Sensation but Not Functionality in Athletes with Delayed Onset Muscle Soreness. Healthcare 2022, 10, 2449. https://doi.org/10.3390/healthcare10122449
Angelopoulos P, Diakoronas A, Panagiotopoulos D, Tsekoura M, Xaplanteri P, Koumoundourou D, Saki F, Billis E, Tsepis E, Fousekis K. Cold-Water Immersion and Sports Massage Can Improve Pain Sensation but Not Functionality in Athletes with Delayed Onset Muscle Soreness. Healthcare. 2022; 10(12):2449. https://doi.org/10.3390/healthcare10122449
Chicago/Turabian StyleAngelopoulos, Pavlos, Anastasios Diakoronas, Dimitrios Panagiotopoulos, Maria Tsekoura, Panagiota Xaplanteri, Dimitra Koumoundourou, Farzaneh Saki, Evdokia Billis, Elias Tsepis, and Konstantinos Fousekis. 2022. "Cold-Water Immersion and Sports Massage Can Improve Pain Sensation but Not Functionality in Athletes with Delayed Onset Muscle Soreness" Healthcare 10, no. 12: 2449. https://doi.org/10.3390/healthcare10122449
APA StyleAngelopoulos, P., Diakoronas, A., Panagiotopoulos, D., Tsekoura, M., Xaplanteri, P., Koumoundourou, D., Saki, F., Billis, E., Tsepis, E., & Fousekis, K. (2022). Cold-Water Immersion and Sports Massage Can Improve Pain Sensation but Not Functionality in Athletes with Delayed Onset Muscle Soreness. Healthcare, 10(12), 2449. https://doi.org/10.3390/healthcare10122449







