Can Exercise Affect the Pain Characteristics in Patients with Fibromyalgia? A Randomized Controlled Trial
Abstract
1. Introduction
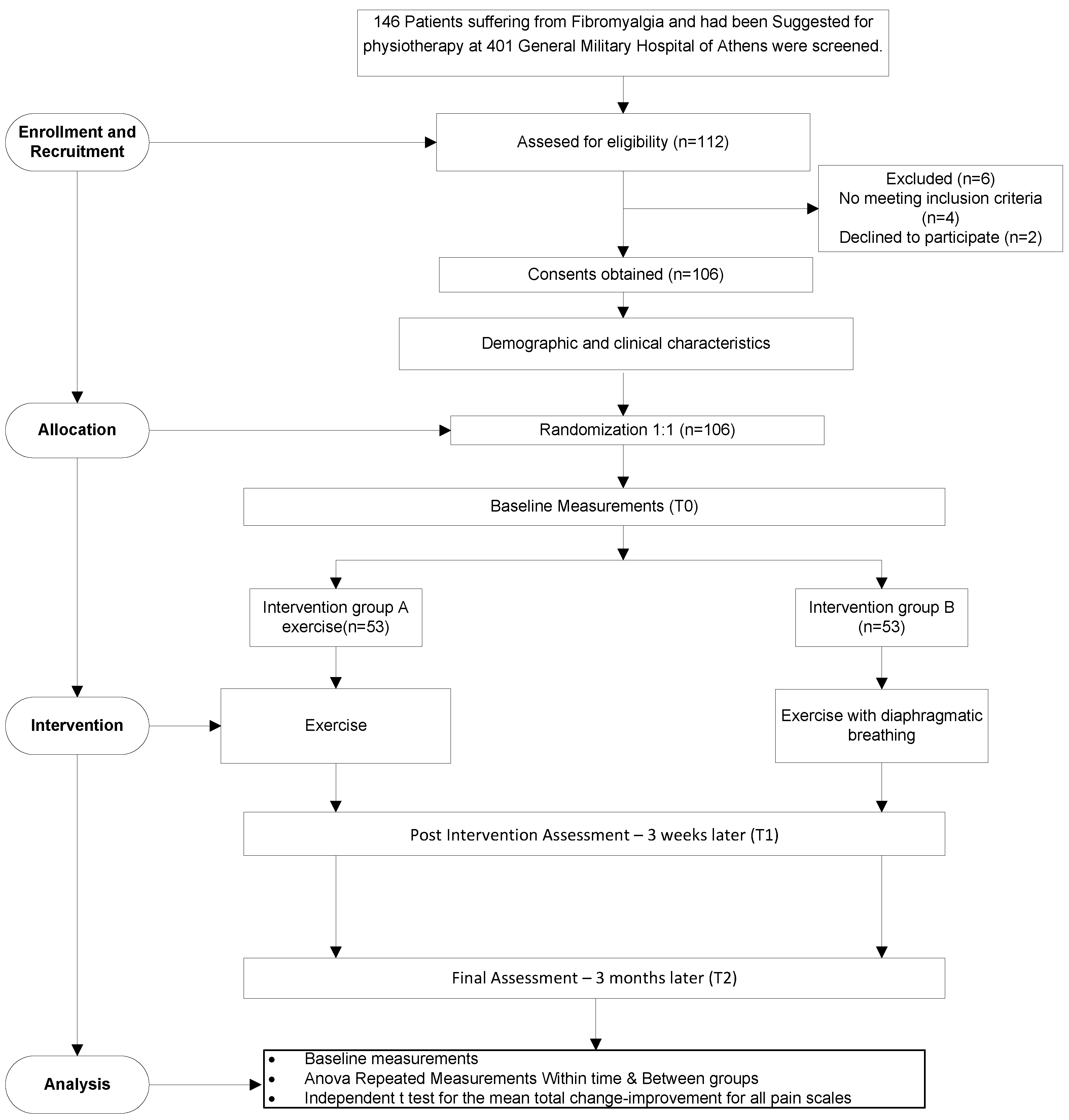
2. Materials and Methods
2.1. Procedure
2.2. Interventions
2.3. Statistical Analysis
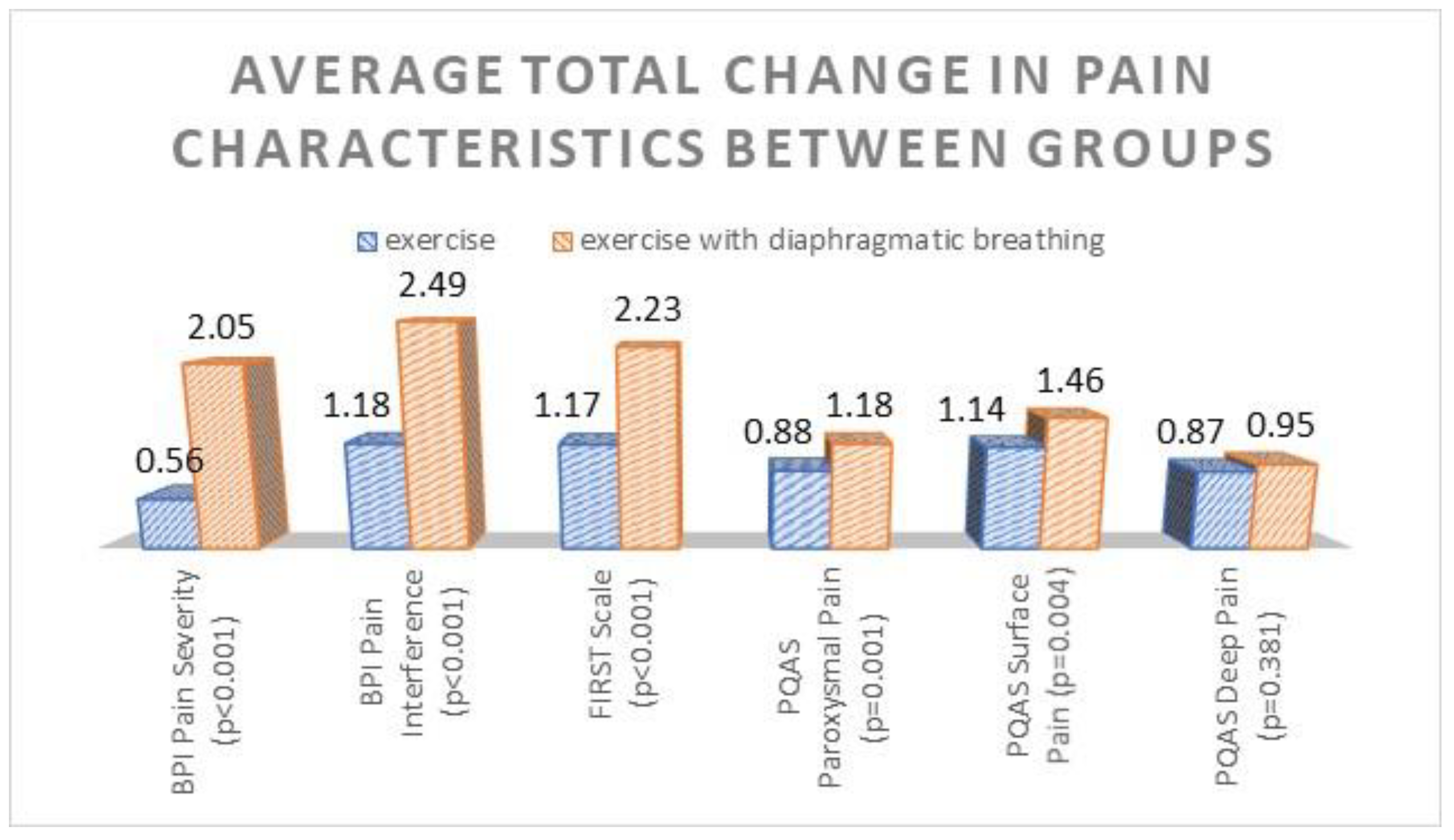
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Talotta, R.; Bazzichi, L.; Di Franco, M.; Casale, R.; Batticciotto, A.; Gerardi, M.C.; Sarzi-Puttini, P. One year in review 2017: Fibromyalgia. Clin. Exp. Rheumatol. 2017, 35 (Suppl. S105), 6–12. [Google Scholar]
- Theoharides, T.C.; Tsilioni, I.; Arbetman, L.; Panagiotidou, S.; Stewart, J.M.; Gleason, R.M.; Russell, I.J. Fibromyalgia Syndrome in Need of Effective Treatments. J. Pharmacol. Exp. Ther. 2015, 355, 255–263. [Google Scholar] [CrossRef]
- Sarzi-Puttini, P.; Giorgi, V.; Marotto, D.; Atzeni, F. Fibromyalgia: An update on clinical characteristics, aetiopathogenesis and treatment. Nat. Rev. Rheumatol. 2020, 16, 645–660. [Google Scholar] [CrossRef]
- Kim, S.Y.; Busch, A.J.; Overend, T.J.; Schachter, C.L.; Van Der Spuy, I.; Boden, C.; Góes, S.M.; Foulds, H.J.; Bidonde, J. Flexibility exercise training for adults with fibromyalgia. Cochrane Database Syst. Rev. 2019, 9, CD013419. [Google Scholar] [CrossRef]
- Macfarlane, G.J.; Kronisch, C.; Dean, L.E.; Atzeni, F.; Häuser, W.; Fluß, E.; Choy, E.; Kosek, E.; Amris, K.; Branco, J.; et al. EULAR revised recommendations for the management of fibromyalgia. Ann. Rheum. Dis. 2017, 76, 318–328. [Google Scholar] [CrossRef]
- Perrot, S.; Bouhassira, D.; Fermanian, J.; CEDR (Cercle d’Etude de la Douleur en Rhumatologie). Development and validation of the Fibromyalgia Rapid Screening Tool (FiRST). Pain 2010, 150, 250–256. [Google Scholar] [CrossRef]
- Zis, P.; Brozou, V.; Stavropoulou, E.; Argyra, E.; Siafaka, I.; Kararizou, E.; Bouhassira, D.; Perrot, S.; Zis, V.; Vadalouca, A. Validation of the Greek Version of the Fibromyalgia Rapid Screening Tool. Pain Pract. 2017, 17, 925–929. [Google Scholar] [CrossRef]
- Mystakidou, K.; Mendoza, T.; Tsilika, E.; Befon, S.; Parpa, E.; Bellos, G.; Vlahos, L.; Cleeland, C. Greek Brief Pain Inventory: Validation and Utility in Cancer Pain. Oncology 2000, 60, 35–42. [Google Scholar] [CrossRef]
- Jensen, M.P.; Gammaitoni, A.R.; Olaleye, D.O.; Oleka, N.; Nalamachu, S.R.; Galer, B.S. The Pain Quality Assessment Scale: Assessment of Pain Quality in Carpal Tunnel Syndrome. J. Pain 2006, 7, 823–832. [Google Scholar] [CrossRef]
- Ferguson, B. ACSM’s Guidelines for Exercise Testing and Prescription 9th Ed. 2014. J. Can. Chiropr. Assoc. 2014, 58, 328. [Google Scholar]
- Kokkonen, J.; Nelson, A.G.; Eldredge, C.; Winchester, J.B. Chronic Static Stretching Improves Exercise Performance. Med. Sci. Sports Exerc. 2007, 39, 1825–1831. [Google Scholar] [CrossRef]
- Couto, N.; Monteiro, D.; Cid, L.; Bento, T. Effect of different types of exercise in adult subjects with fibromyalgia: A systematic review and meta-analysis of randomised clinical trials. Sci. Rep. 2022, 12, 10391. [Google Scholar] [CrossRef]
- Rodríguez-Mansilla, J.; Mejías-Gil, A.; Garrido-Ardila, E.M.; Jiménez-Palomares, M.; Montanero-Fernández, J.; González-López-Arza, M.V. Effects of Non-Pharmacological Treatment on Pain, Flexibility, Balance and Quality of Life in Women with Fibromyalgia: A Randomised Clinical Trial. J. Clin. Med. 2021, 10, 3826. [Google Scholar] [CrossRef]
- Bidonde, J.; Busch, A.J.; Schachter, C.L.; Webber, S.; Musselman, K.; Overend, T.J.; Góes, S.M.; Bello-Haas, V.D.; Boden, C. Mixed exercise training for adults with fibromyalgia. Cochrane Database Syst. Rev. 2019, 5, CD013340. [Google Scholar] [CrossRef]
- Sarmento, C.V.M.; Moon, S.; Pfeifer, T.; Steinbacher, M.; Smirnova, I.V.; Colgrove, Y.; Lai, S.M.; Maz, M.; Liu, W. Inflammatory response to a bout of high-intensity exercise in females with fibromyalgia. Sport Sci. Health 2022, 18, 1051–1059. [Google Scholar] [CrossRef]
- Bidonde, J.; Busch, A.J.; Schachter, C.L.; Overend, T.J.; Kim, S.Y.; Góes, S.M.; Boden, C.; Foulds, H.J. Aerobic exercise training for adults with fibromyalgia. Cochrane Database Syst. Rev. 2017, 6, CD012700. [Google Scholar] [CrossRef]
- Bhargava, J.; Hurley, J.A. Fibromyalgia; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Ablin, J.N.; Wolfe, F. A Comparative Evaluation of the 2011 and 2016 Criteria for Fibromyalgia. J. Rheumatol. 2017, 44, 1271–1276. [Google Scholar] [CrossRef]
- Blickenstaff, C.; Pearson, N. Reconciling movement and exercise with pain neuroscience education: A case for consistent education. Physiother. Theory Pract. 2016, 32, 396–407. [Google Scholar] [CrossRef]
- Tabor, A.; Thacker, M.A.; Moseley, L.; Körding, K.P. Pain: A Statistical Account. PLoS Comput. Biol. 2017, 13, e1005142. [Google Scholar] [CrossRef]
- Louw, A.; Zimney, K.; Puentedura, E.J.; Diener, I. The efficacy of pain neuroscience education on musculoskeletal pain: A systematic review of the literature. Physiother. Theory Pract. 2016, 32, 332–355. [Google Scholar] [CrossRef]
- Busch, V.; Magerl, W.; Kern, U.; Haas, J.; Hajak, G.; Eichhammer, P. The Effect of Deep and Slow Breathing on Pain Perception, Autonomic Activity, and Mood Processing—An Experimental Study. Pain Med. 2012, 13, 215–228. [Google Scholar] [CrossRef]
- Jafari, H.; Courtois, I.; Bergh, O.V.D.; Vlaeyen, J.W.; Van Diest, I. Pain and respiration: A systematic review. Pain 2017, 158, 995–1006. [Google Scholar] [CrossRef]
- McNaughton, D.; Beath, A.; Hush, J.; Jones, M. Perceptual sensory attenuation in chronic pain subjects and healthy controls. Sci. Rep. 2022, 12, 8958. [Google Scholar] [CrossRef]
- Joseph, A.E.; Moman, R.N.; Barman, R.A.; Kleppel, D.J.; Eberhart, N.D.; Gerberi, D.J.; Murad, M.H.; Hooten, W.M. Effects of Slow Deep Breathing on Acute Clinical Pain in Adults: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Evid.-Based Integr. Med. 2022, 27. [Google Scholar] [CrossRef]
- Hamasaki, H. Effects of Diaphragmatic Breathing on Health: A Narrative Review. Medicines 2020, 7, 65. [Google Scholar] [CrossRef]
| Age | 46.83 ± 5.991 | (35–57) |
| Sex b (male/female) | 48 (45.3)/58 (54.7) | |
| Areas feeling Pain b (Axial region/back/back & leg/shoulder) | 17 (16)/71 (67)/6 (5.7)/12 (11.3) | |
| Treatment or medication b (yes/no) | 58 (54.7)/48 (45.3) | |
| FiRST total score b (<5/≥5) | 43 (40.6)/63 (59.4) | |
| BPI Pain Severity Score | 6.09 ± 0.769 | (4.5–7.75) |
| BPI Pain Interference Score | 8.06 ± 0.812 | (6–10) |
| PQAS Paroxysmal pain | 6.08 ± 0.491 | (4.6–7.2) |
| PQAS Surface pain | 6.30 ± 0.582 | (4.8–7.4) |
| PQAS Deep pain | 5.71 ± 0.507 | (4–7.2) |
| PQAS (deep/superficial) pain b | 82 (77.4)/24 (22.6) | |
| PQAS different time qualities of Pain b (intermittent/stable/variable) | 11 (10.4)/70 (66)/25 (23.6) |
| Exercise (n = 53) | Exercise with Diaphragmatic Breathing (n = 53) | p-Value | |
|---|---|---|---|
| Age | 47.04 ± 6.28 | 46.62 ± 5.73 | 0.72 |
| Sex b (male/female) | 21 (39.6)/32 (60.4) | 27 (50.9)/26 (49.1) | 0.24 |
| Treatment or medication b (y/n) | 34 (64.2)/19 (35.8) | 24 (45.3)/29 (54.7) | 0.05 |
| BPI Pain Severity Score | 5.99 ± 0.83 | 6.18 ± 0.69 | 0.21 |
| BPI Pain Interference Score | 7.98 ± 0.90 | 8.13 ± 0.72 | 0.34 |
| PQAS Paroxysmal pain | 6.08 ± 0.50 | 6.07 ± 0.48 | 0.97 |
| PQAS Surface pain | 6.31 ± 0.57 | 6.28 ± 0.60 | 0.79 |
| PQAS Deep pain | 5.71 ± 0.55 | 5.70 ± 0.47 | 0.97 |
| PQAS (deep/superficial) pain b | 42 (79.2)/11 (20.8) | 40 (75.5)/13 (24.5) | 0.64 |
| PQAS diff. time qualities of pain b (intermittent/stable/variable) | 6 (11.3)/34 (64.2)/13 (24.5) | 5 (9.4)/36 (67.9)/12 (22.6) | 0.91 |
| Pain Scale | Group | Baseline | 3 Weeks | 3 Months | F | p-Value |
|---|---|---|---|---|---|---|
| BPI Severity | Exercise | 6.00 ± 0.11 | 5.48 ± 0.10 | 5.43 ± 0.10 | 373.6 | <0.001 |
| Exercise + diaphragmatic breathing | 6.18 ± 0.11 | 4.15 ± 0.10 | 4.13 ± 0.10 | |||
| BPI Interference | Exercise | 7.98 ± 0.11 | 6.85 ± 0.10 | 6.80 ± 0.10 | 853.7 | <0.001 |
| Exercise + diaphragmatic breathing | 8.13 ± 0.11 | 5.63 ± 0.10 | 5.64 ± 0.10 | |||
| FiRST | Exercise | 4.77 ± 0.12 | 3.87 ± 0.14 | 3.60 ± 0.14 | 240,52 | <0.001 |
| Exercise with diaphragmatic breathing | 4.55 ± 0.11 | 2.43 ± 0.10 | 2.32 ± 0.10 | |||
| PQAS Paroxysmal | Exercise | 6.08 ± 0.07 | 5.28 ± 0.06 | 5.21 ± 0.06 | 498.76 | <0.001 |
| Exercise with diaphragmatic breathing | 6.08 ± 0.07 | 4.93 ± 0.06 | 4.90 ± 0.06 | |||
| PQAS Surface | Exercise | 6.32 ± 0.08 | 5.23 ± 0.05 | 5.18 ± 0.05 | 536.49 | <0.001 |
| Exercise with diaphragmatic breathing | 6.29 ± 0.08 | 4.83 ± 0.05 | 4.82 ± 0.05 | |||
| PQAS Deep | Exercise | 5.71 ± 0.07 | 4.89 ± 0.08 | 4.84 ± 0.07 | 342.37 | <0.001 |
| Exercise with diaphragmatic breathing | 5.71 ± 0.07 | 4.77 ± 0.08 | 4.75 ± 0.07 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vrouva, S.; Sopidou, V.; Koutsioumpa, E.; Chanopoulos, K.; Nikolopoulou, A.; Papatsimpas, V.; Koumantakis, G.A. Can Exercise Affect the Pain Characteristics in Patients with Fibromyalgia? A Randomized Controlled Trial. Healthcare 2022, 10, 2426. https://doi.org/10.3390/healthcare10122426
Vrouva S, Sopidou V, Koutsioumpa E, Chanopoulos K, Nikolopoulou A, Papatsimpas V, Koumantakis GA. Can Exercise Affect the Pain Characteristics in Patients with Fibromyalgia? A Randomized Controlled Trial. Healthcare. 2022; 10(12):2426. https://doi.org/10.3390/healthcare10122426
Chicago/Turabian StyleVrouva, Sotiria, Varvara Sopidou, Evangelia Koutsioumpa, Konstantinos Chanopoulos, Alexandra Nikolopoulou, Vasileios Papatsimpas, and George A. Koumantakis. 2022. "Can Exercise Affect the Pain Characteristics in Patients with Fibromyalgia? A Randomized Controlled Trial" Healthcare 10, no. 12: 2426. https://doi.org/10.3390/healthcare10122426
APA StyleVrouva, S., Sopidou, V., Koutsioumpa, E., Chanopoulos, K., Nikolopoulou, A., Papatsimpas, V., & Koumantakis, G. A. (2022). Can Exercise Affect the Pain Characteristics in Patients with Fibromyalgia? A Randomized Controlled Trial. Healthcare, 10(12), 2426. https://doi.org/10.3390/healthcare10122426






