Abstract
Human immunodeficiency virus (HIV) infections and less-than-optimal care of people living with HIV (PLHIV) continue to challenge public health and clinical care organizations in the communities that are most impacted by HIV. In the era of evidence-based public health, it is imperative to monitor viral load (VL) in PLHIV according to global and national guidelines and assess the factors associated with variation in VL levels. Purpose: This study had two objectives—(a) to describe the levels of HIV VL in persons on antiretroviral therapy (ART), and (b) to analyze the significance of variation in VL by patients’ demographic and clinical characteristics, outcomes of HIV care, and geographic characteristics of HIV care facilities. Methods: The study population for this quantitative study was 49,460 PLHIV in the Democratic Republic of Congo (DRC) receiving ART from 241 CDC-funded HIV/AIDS clinics in the Haut-Katanga and Kinshasa provinces of the DRC. Analysis of variance (ANOVA) was performed, including Tamhane’s T2 test for pairwise comparisons using de-identified data on all patients enrolled in the system by the time the data were extracted for this study by the HIV programs in May 2019. Results: The VL was undetectable (<40 copies/mL) for 56.4% of the patients and 24.7% had VL between 40 copies/mL and less than 1000 copies per mL, indicating that overall, 81% had VL < 1000 and were virologically suppressed. The remaining 19% had a VL of 1000 copies/mL or higher. The mean VL was significantly (p < 0.001) higher for males than for females (32,446 copies/mL vs. 20,786, respectively), persons <15 years of age compared to persons of ages ≥ 15 years at the time of starting ART (45,753 vs. 21,457, respectively), patients who died (125,086 vs. 22,090), those who were lost to follow-up (LTFU) (69,882 vs. 20,018), those with tuberculosis (TB) co-infection (64,383 vs. 24,090), and those who received care from urban clinics (mean VL = 25,236) compared to rural (mean VL = 3291) or semi-rural (mean VL = 26,180) clinics compared to urban. WHO clinical stages and duration on ART were not statistically significant at p ≤ 0.05 in this cohort. Conclusions: The VL was >1000 copies/mL for 19% of PLHIV receiving ART, indicating that these CDC-funded clinics and programs in the Haut-Katanga and Kinshasa provinces of DRC have more work to do. Strategically designed innovations in services are desirable, with customized approaches targeting PLHIV who are younger, male, those LTFU, with HIV/TB co-infection, and those receiving care from urban clinics.
1. Introduction
With the advancement of technology and science, HIV has transitioned from a life-threatening terminal illness to a manageable chronic disease. In order for patients to have an overall good prognosis, HIV prevention programs have to assess and address the severity of infection by relying on regular measurements of viral load (VL), which is described as the number of copies of HIV RNA in a milliliter of blood [1]. VL suppression is the most important milestone on the continuum of HIV care in the era of undetectable equals untransmittable (U = U) [2]. According to the Centers for Disease Control and Prevention (CDC) data, an HIV-positive patient is considered to be virally suppressed and at “effectively no risk” of transmission to an HIV-negative person when the VL is considered “durably undetectable” and remains undetectable for at least six months after the first undetectable result [3,4]. Both the CDC and the World Health Organization (WHO) recognize and promote the benefits of ART for VL suppression in PLHIV [5,6]. The WHO also recommends that VL monitoring be regularly conducted after PLHIV are placed on ART, with first the VL test at six months after ART initiation and annual testing thereafter [7].
To eradicate HIV/AIDS by 2030, the Joint United Nations Program on HIV and AIDS (UNAIDS) developed goals that focus on diagnosis, therapy, and viral suppression. UNAIDS has set 95–95–95 targets to be met by the year 2030 [8]. Globally in 2019, data show that only 31% of PLHIV that are receiving ART are virally suppressed. In the western and central regions of Africa, the viral suppression rate is 45% and varies greatly by country [9]. According to the 2020 UNAIDS Report, the Democratic Republic of Congo (DRC) had achieved 54% of the first 95 (percentage of PLHIV who know their status), and 98% of the second 95 (percentage of PLHIV that know their status who are on ART). However, the data regarding PLHIV who are on ART and have suppressed VL (third 95) were not available [9].
As HIV programs continue to prioritize their efforts in increasing the number of PLHIV that are receiving ART and subsequently have VL suppression, there is mounting clinical evidence that increases the recognition that there are many genetic and behavioral factors that can affect how a person may respond to therapy. Some of these factors include the age and gender of the patient, whether the patient continues to seek care, disease progression before ART is initiated, and adherence to the therapy protocol. According to the WHO and the CDC, the primary determining factor of VL suppression and risk of transmission is the patient’s adherence to ART. Unfortunately, long-term adherence in low- and middle-income countries (such as the DRC) is met with numerous patient- and program-related challenges, such as forgetting to take medication, low health literacy, substance abuse, long wait times at clinics, distance to clinics, and costs of care [7,10]. Although adherence to ART directly influences viral suppression, some patients with long-term adherence to ART are also experiencing viral rebound due to either suboptimal adherence after a period of good adherence or ART-resistant mutations [11,12]. ART interruption is also known as interruption in treatment (IIT). IIT is another critical issue for HIV prevention and management programs because it not only increases the risk of HIV spread, but PLHIV experiencing IIT also tend to have poorer outcomes, such as a higher risk of mortality compared to those that have not experienced IIT [13].
Previous studies have shown that there are differences in VL based on gender; women have been shown to have significantly lower VL than men, and the differences have been attributable to hormone levels and plasma HIV RNA thresholds [14,15,16,17]. Additional studies have also shown that children (18 months to 18 years of age) were more likely to reach viral suppression than adults, which can be attributed to children being less likely to experience IIT because they often visit vaccination clinics, their growth may be monitored regularly, and parents may continue to bring children to clinics for treatment [18,19]. The studies also reported that older adults (65 years and older) were more likely to achieve VL suppression than younger adults [2,18,20]. However, the gender and age differences diminish at later stages of the disease [15,16,17,21,22]. Thus, researchers have recommended that providers ensure that ART is being administered appropriately based on gender, age, and CD4 counts [14,15,16].
Efficient program planning and strategic priority-setting in HIV clinical care programs require systematic assessment and evaluation of the outcomes in the populations served by the programs. An important objective of such data-driven assessments is to generate evidence of change in HIV patient outcomes and to assess the positive impact of clinical services over time. To that end, this study had two objectives—to examine the levels of HIV VL in persons on ART, and to analyze factors associated with variation in VL, including patients’ demographic and clinical characteristics, outcomes of HIV care such as death and LTFU, and rurality–urbanicity of HIV care clinics. In the DRC, the HIV programs are mainly funded by the U.S. President’s Emergency Plan for AIDS Relief (PEPFAR) and the Global Fund. The national HIV/AIDS program coordinates HIV service activities.
2. Materials and Methods
2.1. Data
This study utilized secondary data received from the National HIV/AIDS Program (PNLS). The PNLS managed the HIV clinics with the support of CDC/PEPFAR implementing partners. The study population for this quantitative study was 49,460 PLHIV receiving ART from 241 CDC-funded HIV/AIDS clinics in the Haut-Katanga and Kinshasa provinces of the DRC. The de-identified dataset comprised all patients enrolled in these 241 HIV programs at the time of data extraction in May 2019, with ART initiation dates from 2014 to 2019. The clinical database of HIV counseling, testing, and service delivery is maintained for these clinics using a national electronic records system for patient management, TIER.Net (accessed on 21 June 2021). The original data were collected under local IRB approval No. ESP/CE/229/2019 by FHI 360. Georgia Southern University (protocol number HI 9260) exempted this research from a full IRB review.
2.2. Dependent Variable
The dependent variable viral load (VL) was recorded in the database as the number of copies of HIV RNA in a milliliter of blood at the time of the last VL test. This continuous variable was used as the primary outcome measure for its univariate associations with independent variables. In addition, for the graphical depiction of the VL in ranges of interest to HIV programs, the following ranges were computed (as displayed in Figure 1): <40 copies/mL, 40 to <50 copies, 50 to <200 copies, 200 to <400 copies, 400 to <1000 copies, and 1000 or more copies/mL of blood [9,12].
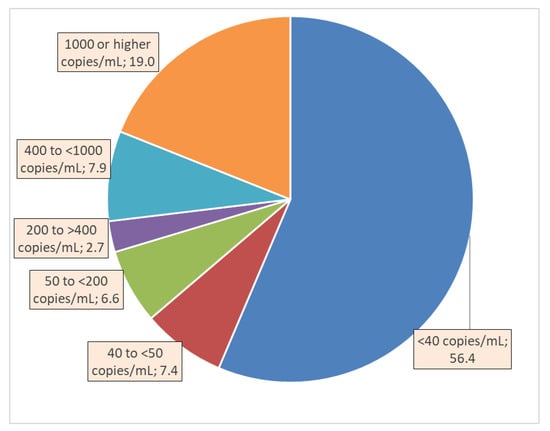
Figure 1.
Percent distribution of persons on antiretroviral therapy by viral load levels in HIV/AIDS clinics of Haut-Katanga and Kinshasa provinces, Democratic Republic of Congo, 2014–2019.
2.3. Independent Variables
The dichotomous variable TB/HIV co-infection was coded as “TB present” and “TB not present,” whereas records with codes 0 and 1 were excluded from our analysis. The variable patient outcome death was coded as 1 “died” and 2 “in care, transferred out, or LTFU.” The variable WHO Stages had four attributes: stage 1 (asymptomatic), stage 2 (mildly symptomatic), stage 3 (moderately symptomatic), and stage 4 (severely symptomatic). The variable ART initiation mode had two categories: “new patient” and “transferred in.” The variable duration on ART was computed by computing the time difference between “date of the last visit for ART” and “ART start date.” For our analysis, the variable duration on ART was recoded into quartiles, with the following categories: “<3.23 months,” “3.23 to 14.52 months,” “14.53 to 40.37 months,” and “>40.37 months.” Two demographic variables were patient sex, with the categories “male” and “female,” and age at the time of the start of ART, with the attributes shown in Table 1. Province of health facility location (Haut-Katanga and Kinshasa) and rurality/urbanicity status of the health zone were the two geographic variables. The variable rurality/urbanicity consisted of three categories based on the health zone—“rural,” “semi-rural,” and “urban,” as identified by the population density in cities or towns within the zone.

Table 1.
Descriptive statistics for characteristics of patients in HIV/AIDS clinics of Haut-Katanga and Kinshasa provinces, DRC, 2014–2019.
2.4. Analytical Methods
We computed descriptive statistics—frequencies and percentages—for the dependent variable and all independent variables. To assess the associations of categorical independent variables with the VL as the continuous dependent variable, we performed analysis of variance (ANOVA). We also performed multiple comparison tests for independent variables with more than two categories if the p-value for ANOVA was ≤0.05, indicating that at least one pair of categories for an independent variable differed significantly in mean VL. The pairwise multiple comparisons were included in the analysis to identify which pairs of categories of each independent variable had significantly different mean VL. Because the assumption of equal variance was violated during the ANOVA test, the pairwise comparisons were based on the statistics of Tamhane’s T2 [23]. The outcome variable was found to be normally distributed, and equality of variance was assumed based upon the results of Leven’s test. All analyses for this study were performed using IBM SPSS Statistics version 25.0 (IBM Corporation, Armonk, NY, USA) [24].
3. Results
The VL in 56.4% of the patients was undetectable (i.e., <40 copies/mL), whereas 19.0% had the heaviest VL of 1000 copies or higher count per mL (Figure 1). The VL was 40 to <50 copies/mL for 7.4%, 50 to <200 copies/mL for 6.6%, 200 to <400 copies/mL for 2.7%, and 400 to <1000 copies/mL for 7.9% of the patients.
Descriptive statistics for patients receiving ART at HIV/AIDS clinics (Table 1) show that 69.0% were female, and 89.7% were 15 years or older. Death was the outcome for 5.8% of patients, as opposed to 94.2% who remained in care, were transferred out, or had therapy interruption (LTFU). After excluding those who died, 16.6% were LTFU, as opposed to 83.4% who remained in care or were transferred out. TB/HIV co-infection existed in 3.6% of the patients. The highest proportion of patients, 41.9%, were in the WHO’s stage 1 of disease progression, whereas 22.9%, 30.7%, and 4.4% were in stages 2, 3, and 4, respectively. Most of the patients initiating ART therapy (95.2%) were new patients. Other trends in patient characteristics are displayed in Table 1.
The results of the one-way ANOVA test performed to compare the mean VL by patient characteristic (Table 2) show that mean VL was significantly higher for males than for females (32,446 copies/mL vs. 20,786; p < 0.001, respectively). The mean VL was significantly higher in patients less than 15 years of age (45,753 vs. 21,457; p < 0.001). The age–sex interaction variable had a significant association with VL (p < 0.001); specific multiple comparisons for this and other non-dichotomous variables are discussed in the next paragraph. Patients who died had a much higher VL than those still alive (125,086 vs. 22,090; p < 0.001, respectively). The mean VL for patients who were LTFU was significantly higher than for those in care or transferred out (69,882 vs. 20,018, p < 0.001, respectively). Patients with TB co-infection also had a significantly higher mean VL of 64,383 copies per mL compared to 24,090 in those without TB co-infection (p < 0.001). A statistically significantly higher (p < 0.001) mean VL existed in patients from Kinshasa compared to those from Haut-Katanga (31,541 vs. 8021, respectively). Mean VL significantly differed by rurality levels of the health zones, with a mean VL of 3291 for rural health zones and 25,236 for urban (p < 0.001). WHO clinical stages and ART were not statistically significant in this cohort.

Table 2.
Analysis of variance (ANOVA) results for the association of characteristics of patients with viral load in HIV/AIDS clinics of Haut-Katanga and Kinshasa provinces, DRC, 2014–2019.
Multiple comparison analysis for independent variables with more than two categories (Table 3) tested the significance of mean VL differences across multiple attributes of those independent variables. Post-hoc comparison using Tamhane indicated that the mean VL for <15-year-old males was significantly higher than for males 15 years or older (mean difference (MD), 22,080 copies per mL) and females 15 years or older (MD, 31,636 copies per mL). The mean VL for <15-year-old females was also significantly higher than females 15 years or older (MD, 22,593 copies per mL). Although urban vs. semi-rural facilities did not differ significantly, urban vs. rural (MD, 21,945) and semi-rural vs. rural (MD, 22,890) location of clinics showed statistically significant differences in mean VL (p < 0.001).

Table 3.
Multiple comparisons of mean viral loads from ANOVA for independent variables with >2 categories in HIV/AIDS clinics of Haut-Katanga and Kinshasa provinces, DRC, 2014–2019.
4. Discussion
This research aimed to assess the HIV VL and factors associated with variation in VL among PLHIV using data from the DRC’s National HIV/AIDS Program (PNLS). Our study findings regarding VL distribution will assist HIV prevention and therapy programs in the DRC to predict prognosis, classify disease progression, assess therapy success, and evaluate the risk of HIV transmission to others. In our study population, the last tested VL was undetectable (<40 copies/mL) for the majority (56.4%) of the patients, whereas roughly one-quarter (24.7%) had a VL between 40 copies/mL and less than 1000 copied per mL. Since 19% had a VL of 1000 or higher, this indicates that cumulatively, 81% had a VL < 1000 and were virologically suppressed. Other recent studies in Sub-Saharan Africa countries, such as Rwanda and Malawi, showed that up to 88% to 91% of persons on ART had a VL < 1000 [25,26]. In our study, a relatively lower proportion of PLHIV with a suppressed VL may indicate therapy gaps, which would call for evaluations by the HIV programs in the DRC to strategically revisit their therapy and coordinated services. Yet, the differences shown could be attributable to a different mix of study samples with respect to factors that can affect VL.
Our study findings on the distribution of patients by the WHO clinical stages showed that 30.7% of patients were in stage 3 and 4.4% were in stage 4. Because PLHIV at these moderately to severely symptomatic stages are at much higher risk of poor outcomes, our findings of variation in VL by specific WHO stages may inform any therapy customization effort for specific disease progression stages.
Our findings of significantly higher VL for patients who were younger than 15 years of age at the start of ART may mean that onset and diagnosis of the infection at an early age may result in greater difficulties in managing the disease. A more in-depth future study may provide specific research evidence. Our study also showed that mean VL was significantly higher for males than for females, which is consistent with the existing body of literature, and is thought to be due to differences in hormone levels, plasma and HIV RNA thresholds, and lower likelihood to access health services, meaning that males may be diagnosed later in their disease progression [14,15,16,17]. However, it may also be because, in the DRC, women have more points of entry to care and are more likely to seek care. Thus, their familiarity with more service points providing care may also improve their chances of early referrals to HIV prevention and management programs. We also noted that males who were on ART before age 15 had much higher VL compared to males or females in other age groups, potentially pointing to a synergetic effect of early-onset/diagnosis and being a male. Our study also showed that there was no association between the duration of ART and VL, perhaps due to the complexity of this relationship. Although adherence to ART is known to improve VL, the risk of therapy interruptions increases with longer duration on ART, particularly in resource-challenged environments, which can be associated with patient- and/or program-related challenges [7,10]. Our analysis of mean VL by the interaction of age at the start of ART and duration in ART showed that the worst VL levels existed in patients who started ART at ages <15 years and had been on ART for >40.37 months, a difference of 32,762 copies/mL compared with those who had started ART when they were 15 years old or older and the ART duration was 3.23 to 14.52 months. Unfortunately, patients who began ART some time ago may have started with a less effective regimen, thus allowing their disease to progress to more advanced stages before the availability of new, more effective regimens.
Our results show that therapy interruption (LTFU) has severely negative consequences for HIV VL management, calling for special attention from HIV programs to aim to improve their patient retention performance. Our findings indicate that the arithmetic mean VL for those in care or transferred out to another care facility was 20,018 copies per mL, a stark contrast to the mean VL for those with therapy interruption indicated by LTFU (69,882 copies per mL). Therefore, the strategies to reduce the IIT must be proactively and collaboratively devised by HIV prevention and management programs in order to prevent poor health outcomes such as death and/or the risk of HIV transmission [13,27]. One strategy to reduce the risk of IIT could be maintaining interoperable patient information systems, ongoing data sharing across health facilities, and ongoing data linkage to detect the patients who are LTFU at a facility but seek treatment at another facility. Another strategy is to implement decentralized drug distribution (DDD) models where ART patients can receive their medication refills in a community setting instead of at the health clinic. Multi-month dispensing may also increase the chances of retention in care, because compliant patients do not need to return often to the facility for medication pick-up. Data linkage with other social services databases may also reduce the risk of IIT. In a recent cohort study, interventions that included longer periods between ART refills (i.e., multi-month dispensation), home delivery of ART refills, linkage to social service support programs after diagnosis, and “silent” transfer of care between clinics for patients relocating (e.g., due to change in employment) were all possible successful strategies for increasing retention in care and preventing the potential for therapy interruption [28].
We found that the mean VL was significantly lower for patients seeking care at rural clinics than semi-rural or urban locations. Urban patients also have more options and may move more frequently to other clinics, interrupting their care and being listed as lost to follow-up in the absence of a unique identifier in clinical HIV care systems. Lower levels of VL in rural areas may be primarily due to the patterns of care-seeking by the severity of illness, with the sickest rural residents seeking care in urban areas. Even in semi-rural areas, the trend is for the sickest patients to move to urban areas with better-equipped and better-staffed facilities. Rural communities are often more closely knit, so the social stigma often associated with HIV may motivate the sickest rural residents to seek HIV care from urban centers away from their communities. On the other hand, in rural locations, the patients tend to have closer relationships with their healthcare providers, and patients may therefore have better adherence to healthcare providers’ instructions.
This quantitative study has some limitations; therefore, our findings should be interpreted within the context of these limitations. First, the secondary data did not contain repeated measures of the VL. In the absence of these data, VL from the last virological test was used. Were all measures of VL available for the period of study, it would have been beneficial to analyze patterns of VL changes and rebounds. Secondly, our choice of variables was limited to a small number of variables captured in the program data and shared with the research team. There are many socio-economic and lifestyle measures not captured in the dataset, which may explain the variation in VL. Finally, our data did not include information concerning the history of a patient having received enhanced adherence counseling, which may have influenced therapy adherence and, in turn, VL levels. In addition, data for other comorbidities were also not available, meriting a suggestion that future studies should also focus on the comorbidity profile of the patients. Regardless of these limitations, our findings are generalizable to HIV services in the DRC.
5. Conclusions
Evidence-based continuous quality improvement is increasingly deemed essential for public health services, including HIV prevention, as is management services being efficient in the face of increasing demands and shrinking resources [29]. The current study produced practice-relevant research evidence showing that although 81% of the PLHIV who were on ART had their VL suppressed below <1000 copies/mL, the clinics and programs offering these services in the Haut-Katanga and Kinshasa provinces of the DRC have more work to do. Strategically designed innovations in services, with better collaborations with community partners for referrals, are desirable to come closer to the target of 95% of all people receiving ART to have viral suppression and working towards the elimination of the AIDS epidemic. Our research findings concerning risk for un-suppressed VL suggest that innovation in the efficient delivery of services to all PLHIV is even more essential during the COVID-19 pandemic and in the post-pandemic era, because some public health and healthcare resources have been reallocated to address the emergent COVID-19 care needs, and in some settings, layering of chronic non-communicable services is becoming necessary to deal with resource shortages [29].
Author Contributions
All authors made substantial contributions to this manuscript, with the following areas of specific contributions: conceptualization, G.H.S., G.D.E., L.M., R.B., K.C.W., E.E., A.M. and O.I.; methodology, G.H.S., G.D.E. and L.M.; software, G.H.S.; validation, G.H.S. and G.D.E.; formal analysis, G.H.S. and K.C.W.; investigation, G.H.S.; resources, G.H.S. and G.D.E..; data curation, E.E. and A.M.; writing—original draft preparation, G.H.S., L.M., G.D.E. and K.C.W.; writing—review and editing, G.H.S., G.D.E., L.M., K.C.W., E.E., O.I., R.B. and A.M.; visualization, G.H.S.; supervision, G.H.S.; project administration, G.H.S. and L.M.; funding acquisition, G.H.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the President’s Emergency Plan for AIDS Relief (PEPFAR) through the Centers for Disease Control and Prevention (CDC) under the terms of grant number 5 NU2GGH002033-02-00. The findings and conclusions in this journal article are those of the authors and do not necessarily represent the official position of the funding agencies.
Institutional Review Board Statement
Georgia Southern University’s Institutional Review Board approved the study under project protocol number H l9260, exempting it from a full IRB review.
Informed Consent Statement
Patient consent was waived because the secondary data were completely de-identified.
Data Availability Statement
The program-implementing partners required that data be destroyed after publication. The authors do have data until the publication of the article. The authors can facilitate data access if requested with proper permission from the DRC Ministry of Health.
Acknowledgments
The authors acknowledge the following organizations and individuals for sharing their data for this research and/or providing review and feedback: Peter Fonjungo, Minlan Minlangu, Francois Kitenge, and Galaxy Ngalamulume from the Centers for Disease Control and Prevention (CDC), DRC; Aime Mboyo, Bijou Makianding, and Gaetan Nsiku from PNLS; Faustin Malele, Tania Tchissambou, and Yves Ilunga from ICAP; Denis Matshifi, Leonard Yabadile, and David Mukeba from SANRU; Elizabeth Ayangunnah from Georgia Southern University; and Etienne Mpoyi from the WHO.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kranzer, K.; Lawn, S.D.; Johnson, L.F.; Bekker, L.G.; Wood, R. Community viral load and CD4 count distribution among people living with HIV in a South African Township: Implications for treatment as prevention. J. Acquir. Immune Defic. Syndr. 2013, 63, 498–505. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Okoli, C.; Van de Velde, N.; Richman, B.; Allan, B.; Castellanos, E.; Young, B.; Brough, G.; Eremin, A.; Corbelli, G.M.; Mc Britton, M.; et al. Undetectable equals untransmittable (U = U): Awareness and associations with health outcomes among people living with HIV in 25 countries. Sex. Transm. Infect. 2021, 97, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Mermin, J.; McCray, E. Dear Colleague Letters. NCHHSTP. 2017. Available online: https://www.cdc.gov/nchhstp/dear_colleague/2017/dcl-092717-National-Gay-Mens-HIV-AIDS-Awareness-Day.html (accessed on 18 June 2020).
- Undetectable Viral Load Definition. AIDSinfo. 2020. Available online: https://aidsinfo.nih.gov/understanding-hiv-aids/glossary/876/undetectable-viral-load (accessed on 21 June 2021).
- Centers for Disease Control and Prevention. HIV Treatment as Prevention. In Cdc.gov.; 2020. Available online: https://www.cdc.gov/hiv/risk/art/index.html (accessed on 18 June 2021).
- World Health Organization. Viral Suppression for HIV Treatment Success and Prevention of Sexual Transmission of HIV. WHO News. 2018. Available online: https://www.who.int/hiv/mediacentre/news/viral-supression-hiv-transmission/en/ (accessed on 19 June 2021).
- World Health Organization. Guideline on When to Start Antiretroviral Therapy and on Pre-Exposure Prophylaxis for HIV; World Health Organization Press: Geneva, Switzerland, 2015. [Google Scholar]
- UNAIDS. Understanding Fast-Track Accelerating Action to End the Aids Epidemic by 2030; Joint United Nations Programme on HIV/AIDS: Geneva, Switzerland, 2015; Available online: https://www.unaids.org/sites/default/files/media_asset/201506_JC2743_Understanding_FastTrack_en.pdf (accessed on 8 September 2020).
- UNAIDS. UNAIDS Data 2020; Joint United Nations Programme on HIV/AIDS: Geneva, Switzerland, 2020; Available online: https://www.unaids.org/sites/default/files/media_asset/2019-UNAIDS-data_en.pdf (accessed on 18 June 2020).
- Centers for Disease Control and Prevention; Health Resources and Services Administration; National Institutes of Health; American Academy of HIV Medicine; Association of Nurses in AIDS Care; International Association of Providers of AIDS Care; National Minority AIDS Council; Urban Coalition for HIV/AIDS Prevention Services. Recommendations for HIV Prevention with Adults and Adolescents with HIV in the United States, 2014: Summary for Clinical Providers; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2014.
- Maina, E.K.; Mureithi, H.; Adan, A.A.; Muriuki, J.; Lwembe, R.M.; Bukusi, E.A. Incidences and factors associated with viral suppression or rebound among HIV patients on combination antiretroviral therapy from three counties in Kenya. Int. J. Infect. Dis. 2020, 97, 151–158. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, J.; Smith, C.; Lampe, F.C.; Johnson, M.A.; Chadwick, D.R.; Nelson, M.; Dunn, D.; Winston, A.; Post, F.A.; Sabin, C.; et al. Durability of viral suppression with first-line antiretroviral therapy in patients with HIV in the UK: An observational cohort study. Lancet HIV 2017, 4, e295–e302. [Google Scholar] [CrossRef] [Green Version]
- Hill, T.; Bansi, L.; Sabin, C.; Phillips, A.; Dunn, D.; Anderson, J.; Easterbrook, P.; Fisher, M.; Gazzard, B.; Gilson, R.; et al. Data linkage reduces loss to follow-up in an observational HIV cohort study. J. Clin. Epidemiol. 2010, 63, 1101–1109. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, C.A.; Bartley, L.M.; Ghani, A.C.; Le Fevre, A.M.; Kwong, G.P.; Cowling, B.J.; van Sighem, A.I.; de Wolf, F.; Rode, R.A.; Anderson, R.M. Gender difference in HIV-1 RNA viral loads. HIV Med. 2005, 6, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Sterling, T.R.; Lyles, C.M.; Vlahov, D.; Astemborski, J.; Margolick, J.B.; Quinn, T.C. Sex differences in longitudinal human immunodeficiency virus type 1 RNA levels among seroconverters. J. Infect. Dis. 1999, 180, 666–672. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anastos, K.; Gange, S.J.; Lau, B.; Weiser, B.; Detels, R.; Giorgi, J.V.; Margolick, J.B.; Cohen, M.; Phair, J.; Melnick, S.; et al. Association of race and gender with HIV-1 RNA levels and immunologic progression. J. Acquir. Immune Defic. Syndr. 2000, 24, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Rezza, G.; Lepri, A.C.; d’Arminio Monforte, A.; Pezzotti, P.; Castelli, F.; Dianzani, F.; Lazzarin, A.; De Luca, A.; Arlotti, M.; Leoncini, F.; et al. Plasma viral load concentrations in women and men from different exposure categories and with known duration of hiv infection. J. Acquir. Immune Defic. Synd. 2000, 25, 56–62. [Google Scholar] [CrossRef]
- Jiamsakul, A.; Kariminia, A.; Althoff, K.N.; Cesar, C.; Cortes, C.P.; Davies, M.A.; Do, V.C.; Eley, B.; Gill, J.; Kumarasamy, N.; et al. HIV viral load suppression in adults and children receiving antiretroviral therapy—Results from the ieDEA collaboration. J Acquir. Immune Defic. Syndr. 2017, 76, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Shearer, W.T.; Quinn, T.C.; LaRussa, P.; Lew, J.F.; Mofenson, L.; Almy, S.; Rich, K.; Handelsman, E.; Diaz, C.; Pagano, M.; et al. Viral Load and Disease Progression in Infants Infected with Human Immunodeficiency Virus Type 1. N. Engl. J. Med. 1997, 336, 1337–1342. [Google Scholar] [CrossRef] [PubMed]
- Shet, A.; Neogi, U.; Kumarasamy, N.; DeCosta, A.; Shastri, S.; Rewari, B. Virological efficacy with first-line antiretroviral treatment in India: Predictors of viral failure and evidence of viral resuppression. Trop. Med. Int. Health 2015, 20, 1462–1472. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, P.; Tan, J.; Ma, W.; Zheng, H.; Lu, Y.; Wang, N.; Peng, Z.; Yu, R. Outcomes of antiretroviral treatment in HIV-infected adults: A dynamic and observational cohort study in Shenzhen, China, 2003–2014. BMJ Open 2015, 5, e007508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trichavaroj, R.; de Souza, M.S.; Buapunth, P.; Markowitz, L.; Sukwit, S.; Nitayaphan, S.; Brown, A.E. HIV viral load in Thai men and women with subtype E infections. J. Acquir. Immune Defic. Syndr. 2001, 26, 345–347. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Lee, D.K. What is the proper way to apply the multiple comparison test? Korean J. Anesthesiol. 2018, 71, 353–360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- IBM Corporation. IBM SPSS Statistics for Windows; Version 25.0; IBM Corp.: Armonk, NY, USA, 2017. [Google Scholar]
- Singano, V.; van Oosterhout, J.J.; Gondwe, A.; Nkhoma, P.; Cataldo, F.; Singogo, E.; Theu, J.; Ching’ani, W.; Hosseinpour, M.C.; Amberbir, A. Leveraging routine viral load testing to integrate diabetes screening among patients on antiretroviral therapy in Malawi. Int. Health 2021, 13, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Ross, J.; Ribakare, M.; Remera, E.; Murenzi, G.; Munyaneza, A.; Hoover, D.R.; Shi, Q.; Nsanzimana, S.; Yotebieng, M.; Nash, D.; et al. High levels of viral load monitoring and viral suppression under Treat All in Rwanda—A cross-sectional study. J. Int. AIDS Soc. 2020, 23, e25543. [Google Scholar] [CrossRef] [PubMed]
- Cornell, M.; Lessells, R.; Fox, M.P.; Garone, D.B.; Giddy, J.; Fenner, L.; Myer, L.; Boulle, A. Mortality among adults transferred and lost to follow-up from antiretroviral therapy programmes in South Africa. J. Acquir. Immune Defic. Syndr. 2014, 67, e67–e75. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalinjuma, A.V.; Glass, T.R.; Weisser, M.; Myeya, S.J.; Kasuga, B.; Kisung’a, Y.; Sikalengo, G.; Katende, A.; Battegay, M.; Vanobberghen, F. Prospective assessment of loss to follow-up: Incidence and associated factors in a cohort of HIV-positive adults in rural Tanzania. J. Int. AIDS Soc. 2020, 23, e25460. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Osetinsky, B.; Mwangi, A.; Pastakia, S.; Wilson-Barthes, M.; Kimetto, J.; Rono, K.; Laktabai, J.; Galárraga, O. Layering and scaling up chronic non-communicable disease care on existing HIV care systems and acute care settings in Kenya: A cost and budget impact analysis. J. Int. AIDS Soc. 2020, 23, e25496. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).