Sensitivity Analysis of In Silico Fluid Simulations to Predict Thrombus Formation after Left Atrial Appendage Occlusion
Abstract
:1. Introduction
2. Materials and Methods
2.1. Database
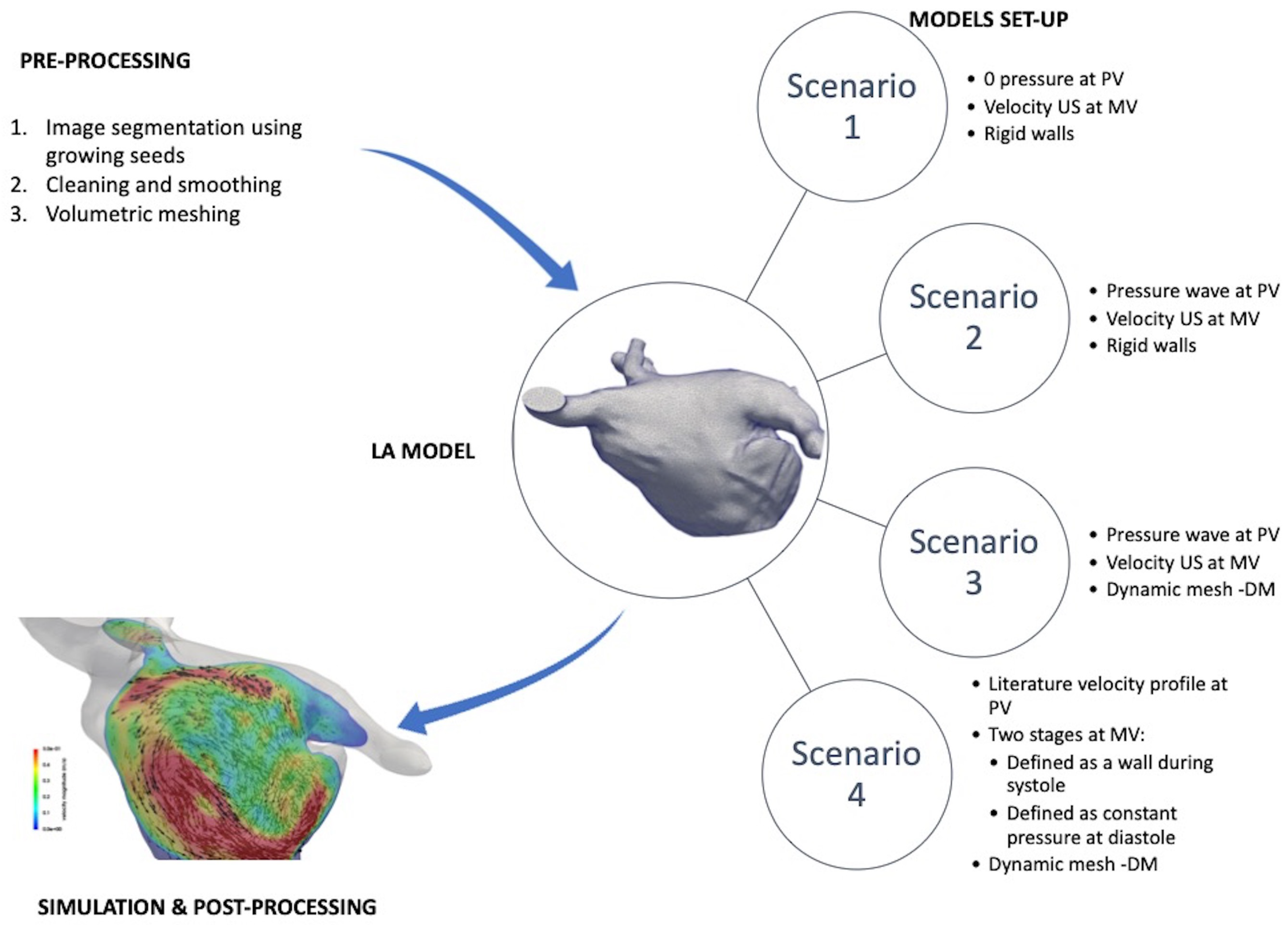
2.2. Methodological Steps
2.2.1. 3D Model Construction
2.2.2. Simulation Setup
2.3. Boundary Conditions Scenarios
2.3.1. Scenario 1: Constant (Null) Inlet Pressure, Imaging-Based Personalised Outlet Velocities, and Rigid Wall
2.3.2. Scenario 2: Generic Patient Pressure Wave as Inlet, Imaging-Based Personalised Outlet Velocities, and Rigid Wall
2.3.3. Scenario 3: Generic Patient Pressure Wave as Inlet, Imaging-Based Personalised Outlet Velocities, and Dynamic Mesh Wall Deformation
2.3.4. Scenario 4: Literature Velocity Profile as Inlet, Mitral Valve as Wall (Systole) or Constant Pressure (Diastole), and Dynamic Mesh Wall Deformation
2.4. In Silico Haemodynamic Indices
3. Results
3.1. Inlet/Outlet and Wall Behaviour Scenarios
3.1.1. Scenario 1: Constant (Null) Inlet Pressure, Imaging-Based Outlet Velocities, and Rigid Wall
3.1.2. Scenario 2: Patient Pressure Wave as Inlet, Imaging-Based Outlet Velocities, and Rigid Wall
3.1.3. Scenario 3: Patient Pressure Wave as Inlet, Imaging-Based Outlet Velocities, and Dynamic Mesh Wall Deformation
3.1.4. Scenario 4: Literature Velocity Profile as Inlet, Mitral Valve as Wall (Systole) or Constant Pressure (Diastole), and Dynamic Mesh Wall Deformation
3.2. Mesh and Cardiac Cycle Convergence
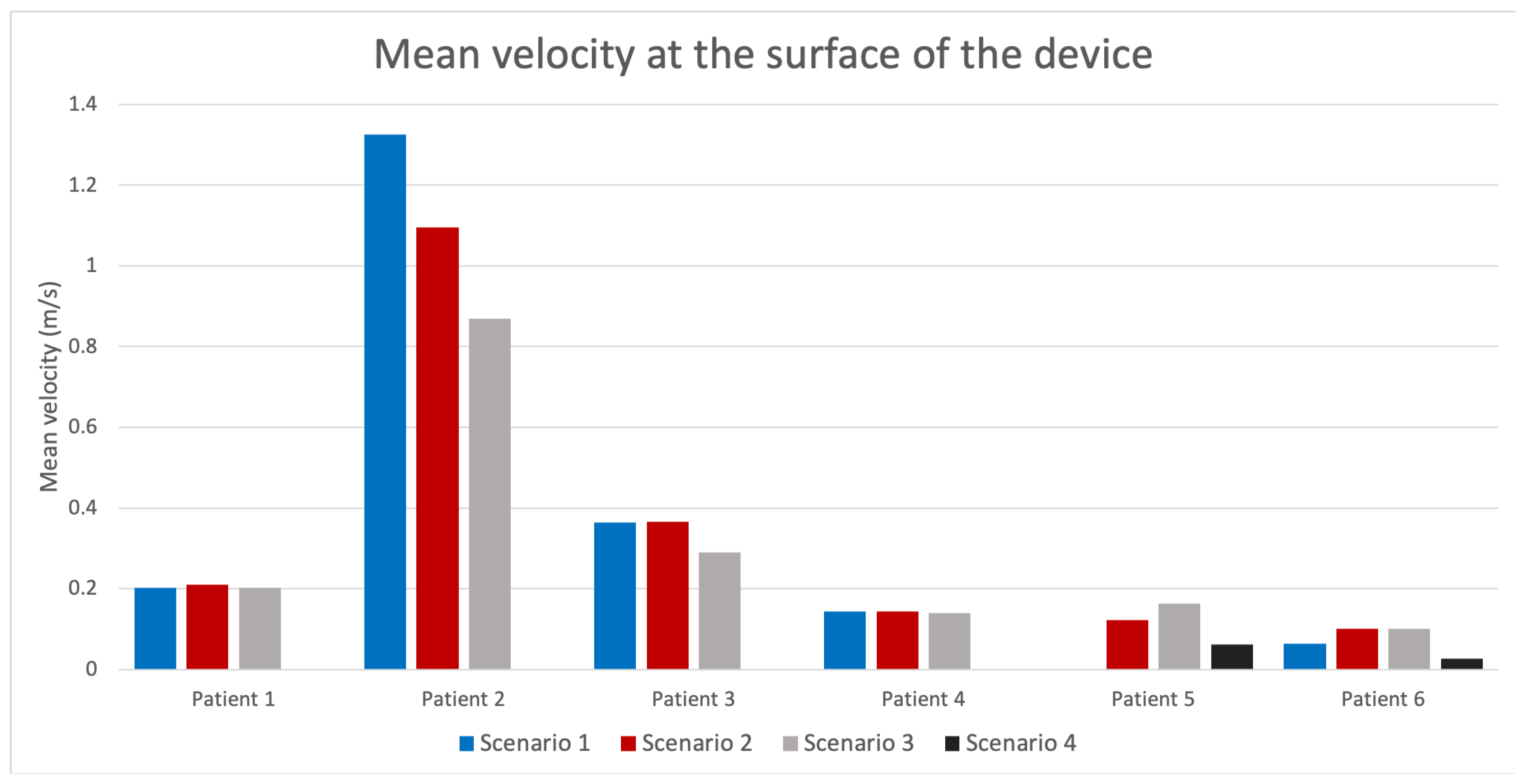
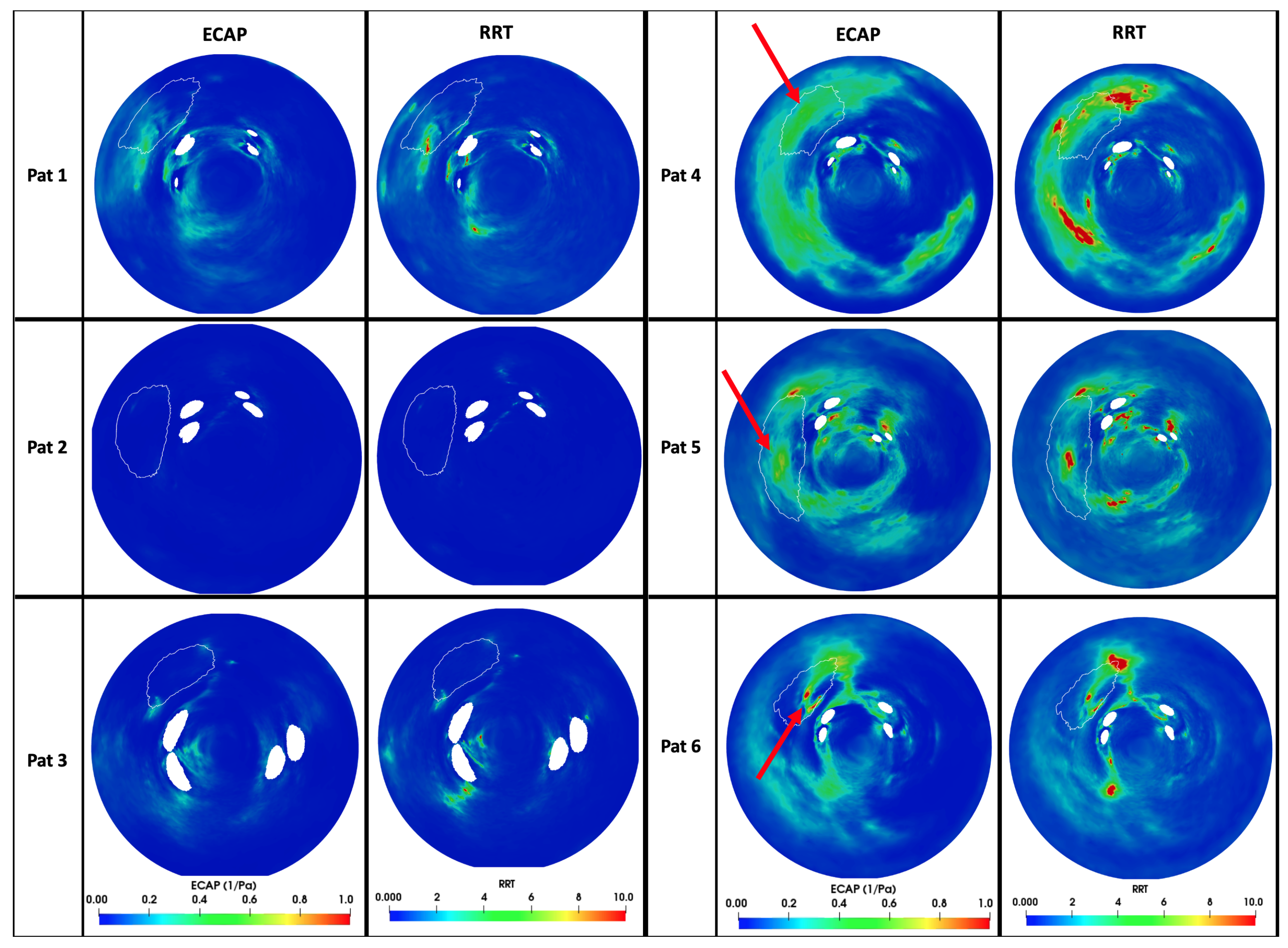
3.3. In Silico Prediction of Device-Related Thrombosis
4. Discussion
4.1. Boundary Conditions Scenarios
4.2. In Silico Prediction of Device-Related Thrombosis Risk
4.3. Mesh and Cardiac Cycle Convergence Analysis
4.4. Limitations and Future Work
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Abbreviations
| AF | Atrial Fibrillation |
| BC | Boundary Conditions |
| CFD | Computational Fluid Dynamics |
| CT | Computed Tomography |
| dCT | Dynamic CT |
| DM | Dynamic Mesh |
| DRT | Device-related Thrombus |
| ECAP | Endothelial Cell Activation Potential |
| ECG | Electrocardiogram |
| FSI | Fluid–Structure Interaction |
| LA | Left Atrium |
| LAA | Left Atrial Appendage |
| LAAO | Left Atrial Appendage Occlusion |
| MV | Mitral Valve |
| MRI | Magnetic Resonance Imaging |
| OSI | Oscillatory Shear Index |
| PS | Patient Specific |
| PV | Pulmonary Veins |
| Re | Reynolds Number |
| RSPV | Right Superior Pulmonary Vein |
| RRT | Residence Time |
| LSPV | Left Superiror Pulmonary Vein |
| TTE | Transthoracic Echocardiography |
| TAWSS | Time Average Wall Shear Stress |
| WSS | Wall Shear Stress |
References
- Rahman, F.; Kwan, G.F.; Benjamin, E.J. Global epidemiology of atrial fibrillation. Nat. Rev. Cardiol. 2014, 11, 639–654. [Google Scholar] [CrossRef]
- Cresti, A.; García-Fernández, M.A.; Sievert, H.; Mazzone, P.; Baratta, P.; Solari, M.; Geyer, A.; De Sensi, F.; Limbruno, U. Prevalence of extra-appendage thrombosis in non-valvular atrial fibrillation and atrial flutter in patients undergoing cardioversion: A large transoesophageal echo study. Eurointervention 2019, 15, e225–e230. [Google Scholar] [CrossRef] [Green Version]
- Benjamin, E.J.; Wolf, P.A.; D’Agostino, R.B.; Silbershatz, H.; Kannel, W.B.; Levy, D. Impact of atrial fibrillation on the risk of death: The Framingham Heart Study. Circulation 1998, 98, 946–952. [Google Scholar] [CrossRef] [Green Version]
- Takada, T.; Yasaka, M.; Nagatsuka, K.; Minematsu, K.; Yamaguchi, T. Blood flow in the left atrial appendage and embolic stroke in nonvalvular atrial fibrillation. Eur. Neurol. 2001, 46, 148–152. [Google Scholar] [CrossRef] [PubMed]
- García-Fernández, M.A.; Torrecilla, E.G.; Román, D.S.; Azevedo, J.; Bueno, H.; Moreno, M.M.; Delcán, J.L. Left atrial appendage doppler flow patterns: Implications on thrombus formation. Am. Heart J. 1992, 124, 955–961. [Google Scholar] [CrossRef]
- Watson, T.; Shantsila, E.; Lip, G.Y. Mechanisms of thrombogenesis in atrial fibrillation: Virchow’s triad revisited. Lancet 2009, 373, 155–166. [Google Scholar] [CrossRef]
- Sanchez, J.M.; Lee, A.; Bartus, K.; Lee, R.J. Percutaneous epicardial approach for LAA ligation. J. Interv. Card. Electrophysiol. 2020. [Google Scholar] [CrossRef]
- Bedeir, K.; Warriner, S.; Kofsky, E.; Gullett, C.; Ramlawi, B. Left Atrial Appendage Epicardial Clip (AtriClip): Essentials and Post-Procedure Management. J. Atr. Fibrillation 2019, 11, 2087. [Google Scholar] [CrossRef] [PubMed]
- Reddy, V.Y.; Doshi, S.K.; Kar, S.; Gibson, D.N.; Price, M.J.; Huber, K.; Horton, R.P.; Buchbinder, M.; Neuzil, P.; Gordon, N.T.; et al. 5-Year Outcomes After Left Atrial Appendage Closure: From the PREVAIL and PROTECT AF Trials. J. Am. Coll. Cardiol. 2017, 70, 2964–2975. [Google Scholar] [CrossRef] [PubMed]
- Aminian, A.; Schmidt, B.; Mazzone, P.; Berti, S.; Fischer, S.; Montorfano, M.; Lam, S.C.C.; Lund, J.; Asch, F.M.; Gage, R.; et al. Incidence, Characterization, and Clinical Impact of Device-Related Thrombus Following Left Atrial Appendage Occlusion in the Prospective Global AMPLATZER Amulet Observational Study. JACC Cardiovasc. Interv. 2019, 12, 1003–1014. [Google Scholar] [CrossRef]
- Fauchier, L.; Cinaud, A.; Brigadeau, F.; Lepillier, A.; Pierre, B.; Abbey, S.; Fatemi, M.; Franceschi, F.; Guedeney, P.; Jacon, P.; et al. Device-Related Thrombosis After Percutaneous Left Atrial Appendage Occlusion for Atrial Fibrillation. J. Am. Coll. Cardiol. 2018, 71, 1528–1536. [Google Scholar] [CrossRef] [PubMed]
- López-Mínguez, J.R.; Eldoayen-Gragera, J.; González-Fernández, R.; Fernández-Vegas, C.; Fuentes-Cañamero, M.E.; Millán-Nuñez, V.; Nogales-Asensio, J.M.; Martínez-Naharro, A.; Sánchez-Giralt, S.; Doblado-Calatrava, M.; et al. Immediate and One-year Results in 35 Consecutive Patients After Closure of Left Atrial Appendage With the Amplatzer Cardiac Plug. Rev. Esp. Cardiol. (Engl. Ed.) 2013, 66, 90–97. [Google Scholar] [CrossRef]
- Glikson, M.; Wolff, R.; Hindricks, G.; Mandrola, J.; Camm, A.J.; Lip, G.Y.H.; Fauchier, L.; Betts, T.R.; Lewalter, T.; Saw, J.; et al. EHRA/EAPCI expert consensus statement on catheter-based left atrial appendage occlusion—An update. EuroIntervention 2020, 15, 1133–1180. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.T.; Gay, M. Characterizing left atrial appendage functions in sinus rhythm and atrial fibrillation using computational models. J. Biomech. 2008, 41, 2515–2523. [Google Scholar] [CrossRef] [PubMed]
- Dahl, S.K.; Thomassen, E.; Hellevik, L.R.; Skallerud, B. Impact of Pulmonary Venous Locations on the Intra-Atrial Flow and the Mitral Valve Plane Velocity Profile. Cardiovasc. Eng. Technol. 2012, 3, 269–281. [Google Scholar] [CrossRef] [Green Version]
- Koizumi, R.; Funamoto, K.; Hayase, T.; Kanke, Y.; Shibata, M.; Shiraishi, Y.; Yambe, T. Numerical analysis of hemodynamic changes in the left atrium due to atrial fibrillation. J. Biomech. 2015, 48, 472–478. [Google Scholar] [CrossRef]
- Otani, T.; Al-Issa, A.; Pourmorteza, A.; McVeigh, E.R.; Wada, S.; Ashikaga, H. A Computational Framework for Personalized Blood Flow Analysis in the Human Left Atrium. Ann. Biomed. Eng. 2016, 44, 3284–3294. [Google Scholar] [CrossRef] [PubMed]
- Bosi, G.M.; Cook, A.; Rai, R.; Menezes, L.J.; Schievano, S.; Torii, R.; Burriesci, G. Computational Fluid Dynamic Analysis of the Left Atrial Appendage to Predict Thrombosis Risk. Front. Cardiovasc. Med. 2018, 5, 34. [Google Scholar] [CrossRef] [Green Version]
- García-Isla, G.; Olivares, A.L.; Silva, E.; Nuñez-Garcia, M.; Butakoff, C.; Sanchez-Quintana, D.; Morales, H.G.; Freixa, X.; Noailly, J.; De Potter, T.; et al. Sensitivity analysis of geometrical parameters to study haemodynamics and thrombus formation in the left atrial appendage. Int. J. Numer. Methods Biomed. Eng. 2018, 34, 1–14. [Google Scholar] [CrossRef]
- Dillon-Murphy, D.; Marlevi, D.; Ruijsink, B.; Qureshi, A.; Chubb, H.; Kerfoot, E.; O’Neill, M.; Nordsletten, D.; Aslanidi, O.; De Vecchi, A. Modeling left atrial flow, energy, blood heating distribution in response to catheter ablation therapy. Front. Physiol. 2018, 9, 1757. [Google Scholar] [CrossRef] [Green Version]
- Masci, A.; Barone, L.; Dedè, L.; Fedele, M.; Tomasi, C.; Quarteroni, A.; Corsi, C. The impact of left atrium appendage morphology on stroke risk assessment in atrial fibrillation: A computational fluid dynamics study. Front. Physiol. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Aguado, A.M.; Olivares, A.L.; Yagüe, C.; Silva, E.; Nuñez-García, M.; Fernandez-Quilez, A.; Mill, J.; Genua, I.; Arzamendi, D.; De Potter, T.; et al. In silico Optimization of Left Atrial Appendage Occluder Implantation Using Interactive and Modeling Tools. Front. Physiol. 2019, 10, 237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jia, D.; Jeon, B.; Park, H.B.; Chang, H.J.; Zhang, L.T. Image-Based Flow Simulations of Pre- and Post-left Atrial Appendage Closure in the Left Atrium. Cardiovasc. Eng. Technol. 2019, 10, 225–241. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Gao, H.; Griffith, B.; Niederer, S.; Luo, X. Analysis of a coupled fluid-structure interaction model of the left atrium and mitral valve. Int. J. Numer. Methods Biomed. Eng. 2019, 35, e3254. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Masci, A.; Alessandrini, M.; Forti, D.; Menghini, F.; Dedé, L.; Tomasi, C.; Quarteroni, A.; Corsi, C. A Proof of concept for computational fluid dynamic analysis of the left atrium in atrial fibrillation on a patient-specific basis. J. Biomech. Eng. 2020, 142, 011002. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Qiao, Y.; Mao, Y.; Jiang, C.; Fan, J.; Luo, K. Numerical prediction of thrombosis risk in left atrium under atrial fibrillation. Math. Biosci. Eng. 2020, 17, 2348–2360. [Google Scholar]
- Mill, J.; Olivares, A.L.; Arzamendi, D.; Agudelo, V.; Regueiro, A.; Camara, O.; Freixa, X. Impact of flow-dynamics on device related thrombosis after left atrial appendage occlusion. Can. J. Cardiol. 2020, 36, 968.e13–968.e14. [Google Scholar] [CrossRef]
- D’Alessandro, N.; Masci, A.; Andalo, A.; Dede, L.; Tomasi, C.; Quarteroni, A.; Corsi, C. Simulation of the Hemodynamic Effects of the Left Atrial Appendage Occlusion in Atrial Fibrillation: Preliminary Results. In Proceedings of the Computing in Cardiology, Rimini, Italy, 13–16 September 2020. [Google Scholar]
- Qureshi, A.; Darwish, O.; Dillon-Murphy, D.; Chubb, H.; Williams, S.; Nechipurenko, D.; Ataullakhanov, F.; Nordsletten, D.; Aslanidi, O.; De Vecchi, A. Modelling Left Atrial Flow and Blood Coagulation for Risk of Thrombus Formation in Atrial Fibrillation. In Proceedings of the Computing in Cardiology, Rimini, Italy, 13–16 September 2020. [Google Scholar]
- García-Villalba, M.; Rossini, L.; Gonzalo, A.; Vigneault, D.; Martinez-Legazpi, P.; Durán, E.; Flores, O.; Bermejo, J.; McVeigh, E.; Kahn, A.M.; et al. Demonstration of Patient-Specific Simulations to Assess Left Atrial Appendage Thrombogenesis Risk. Front. Physiol. 2021, 12, 596596. [Google Scholar] [CrossRef]
- Fang, R.; Li, Y.; Zhang, Y.; Chen, Q.; Liu, Q.; Li, Z. Impact of left atrial appendage location on risk of thrombus formation in patients with atrial fibrillation. Biomech. Model. Mechanobiol. 2021, 20, 1431–1443. [Google Scholar] [CrossRef]
- Sanatkhani, S.; Nedios, S.; Menon, P.G.; Bollmann, A.; Hindricks, G.; Shroff, S.G. Subject-Specific Calculation of Left Atrial Appendage Blood-Borne Particle Residence Time Distribution in Atrial Fibrillation. Front. Physiol. 2021, 12, 633135. [Google Scholar] [CrossRef]
- Mill, J.; Harrison, J.; Legghe, B.; Olivares, A.L.; Morales, X.; Noailly, J.; Iriart, X.; Cochet, H.; Sermesant, M.; Camara, O. In-Silico Analysis of the Influence of Pulmonary Vein Configuration on Left Atrial Haemodynamics and Thrombus Formation in a Large Cohort. Lect. Notes Comput. Sci. 2021, 605–616. [Google Scholar] [CrossRef]
- Morales Ferez, X.; Mill, J.; Juhl, K.A.; Acebes, C.; Iriart, X.; Legghe, B.; Cochet, H.; Backer, O.D.; Paulsen, R.R.; Camara, O. Deep Learning Framework for Real-Time Estimation of In-silico Thrombotic Risk Indices in the Left Atrial Appendage. Front. Physiol. 2021, 12, 694945. [Google Scholar] [CrossRef]
- Taubin, G. A Signal Processing Approach to Fair Surface Design. In Proceedings of the 22nd Annual Conference on Computer Graphics and Interactive Techniques, Los Angeles, CA, USA, 6–11 August 1995; pp. 351–358. [Google Scholar]
- Berger, S.A.; Jou, L.D. Flows in Stenotic Vessels. Annu. Rev. Fluid Mech. 2000, 32, 347–382. [Google Scholar] [CrossRef]
- Veronesi, F.; Corsi, C.; Sugeng, L.; Caiani, E.G.; Weinert, L.; Mor-Avi, V.; Cerutti, S.; Lamberti, C.; Lang, R.M. Quantification of Mitral Apparatus Dynamics in Functional and Ischemic Mitral Regurgitation Using Real-time 3-Dimensional Echocardiography. J. Am. Soc. Echocardiogr. 2008, 21, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Emilsson, K.; Wandt, B. The relation between ejection fraction and mitral annulus motion before and after direct-current electrical cardioversion. Clin. Physiol. 2000, 20, 218–224. [Google Scholar] [CrossRef]
- Fernandez-Perez, G.C.; Duarte, R.; Corral de la Calle, M.; Calatayud, J.; Sanchez Gonzalez, J. Analysis of left ventricular diastolic function using magnetic resonance imaging. Radiologia 2012, 54, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Di Achille, P.; Tellides, G.; Figueroa, C.A.; Humphrey, J.D. A haemodynamic predictor of intraluminal thrombus formation in abdominal aortic aneurysms. Proc. R. Soc. Math. Phys. Eng. Sci. 2014, 470, 20140163. [Google Scholar] [CrossRef] [Green Version]
- Himburg, H.A.; Grzybowski, D.M.; Hazel, A.L.; LaMack, J.A.; Li, X.M.; Friedman, M.H. Spatial comparison between wall shear stress measures and porcine arterial endothelial permeability. Am. J. Physiol. Heart Circ. Physiol. 2004, 286, H1916–H1922. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nunez-Garcia, M.; Bernardino, G.; Alarcon, F.; Caixal, G.; Mont, L.; Camara, O.; Butakoff, C. Fast Quasi-Conformal Regional Flattening of the Left Atrium. IEEE Trans. Vis. Comput. Graph. 2020, 26, 2591–2602. [Google Scholar] [CrossRef] [Green Version]
- Viceconti, M.; Pappalardo, F.; Rodriguez, B.; Horner, M.; Bischoff, J.; Musuamba Tshinanu, F. In silico trials: Verification, validation and uncertainty quantification of predictive models used in the regulatory evaluation of biomedical products. Methods 2021, 185, 120–127. [Google Scholar] [CrossRef]
- Cresti, A.; Galli, C.A.; Alimento, M.L.; De Sensi, F.; Baratta, P.; D’Aiello, I.; Limbruno, U.; Pepi, M.; Fusini, L.; Maltagliati, A.C. Does mitral regurgitation reduce the risks of thrombosis in atrial fibrillation and flutter? J. Cardiovasc. Med. 2019, 20, 660–666. [Google Scholar] [CrossRef] [PubMed]
- Freixa, X.; Cepas-Guillen, P.; Flores-Umanzor, E.; Regueiro, A.; Sanchis, L.; Fernandez-Valledor, A.; Brugaletta, S.; Carretero, M.J.; Vidal, B.; Masotti, M.; et al. Pulmonary ridge coverage and device-related thrombosis after left atrial appendage occlusion. EuroIntervention 2021, 16, E1288–E1294. [Google Scholar] [CrossRef]
- Dueñas-Pamplona, J.; Sierra-Pallares, J.; García, J.; Castro, F.; Munoz-Paniagua, J. Boundary-Condition Analysis of an Idealized Left Atrium Model. Ann. Biomed. Eng. 2021, 49, 1507–1520. [Google Scholar] [CrossRef]
- Markl, M.; Lee, D.C.; Carr, M.L.; Foucar, C.; Ng, J.; Schnell, S.; Carr, J.C.; Goldberger, J.J. Assessment of left atrial and left atrial appendage flow and stasis in atrial fibrillation. J. Cardiovasc. Magn. Reson. 2015, 17, M3. [Google Scholar] [CrossRef] [Green Version]
- Morales, X.; Mill, J.; Delso, G.; Loncaric, F.; Doltra, A.; Freixa, X.; Sitges, M.; Bijnens, B.; Camara, O. 4D Flow Magnetic Resonance Imaging for Left Atrial Haemodynamic Characterization and Model Calibration. Lect. Notes Comput. Sci. 2021, 12592, 156–165. [Google Scholar]



| Study | PV Inlet | MV Outlet LV Systole/Diastole | Wall Behaviour | Cardiac Cycles | Mesh Elements () | Geoms |
|---|---|---|---|---|---|---|
| Zhang [14] | Lit. vels. | Wall/0 mmHg | FSI | 3 | 2 | 1 |
| Dahl [15] | Patient flow | Added mass flux | Rigid | 1 | 21 | 1 |
| Koizumi [16] | 10 mmHg | Wall/8 mmHg | dMRI | 5 | 1.5 | 1 |
| Otani [17] | dCT vels. | Wall/dCT flow | dCT | 5 | 3.6–5.5 | 2 |
| Bosi [18] | 0 mmHg | Lit. vels. | Rigid | 4 | 22–30 | 4 |
| García-Isla [19] | Lit. vels. | Wall/8 mmHg | Rigid | 1 | 3.5–5.0 | 36 * |
| Dillon-Murphy [20] | 0 mmHg | dMRI flow | dMRI | 10 | 12 ** | 2 |
| Masci-a [21] | Flow balance | Lit. flow | Sinusoidal | 5 | 17–19 | 5 |
| Aguado [22] | Lit. vels. | Wall/8 mmHg | Rigid | 1 | 2–9.6 | 2 |
| Jia [23] | Synthetic vels. | Wall/0 mmHg | Rigid | 10 | 0.4 | 1 |
| Feng [24] | Lit. press. | Lit. press. | FSI | 2 | 1 | 1 |
| Masci-b [25] | Flow balance | Lit. flow | Sinusoidal | 7 | 8–10 | 2 |
| Wang [26] | 10 mmHg | Lit. flow | Rigid | 20 | 24 | 1 |
| Mill-a [27] | Lit vels. | Wall/8 mmHg | Diff. DM | 2 | 5 | 2 |
| D’Alessandro [28] | Flow balance | Lit. vels. | Sinusoidal | 5 | 17–19 | 2 |
| Qureshi [29] | Synthetic vels. | Unknown | dMRI | 15 | 4 | 2 |
| García-Villalba [30] | Flow balance | Wall/Open | Rigid/dCT | 20 | 5–9 ** | 6 |
| Fang [31] | AF vels. | Wall/0 mmHg | FSI | 4 | Unknown | 1 * |
| Sanatkhani [32] | Lit. vels. | Open/0 mmHg | Rigid | 25 | 3.5–5 | 16 |
| Mill-b [33] | AF press. | AF vels. | Diff. DM | 3 | 8–9 | 52 |
| Morales [34] | Lit. vels. | Wall/8 mmHg | Diff. DM | 3 | 3.5–9 | 370 *** |
| This work | AF press. | Personalised vels. | Rigid/Diff. DM | 1–2 | 1–5 | 6 |
| Patient ID | Number of PV | LA Volume (mL) | Device Size (In mm) | AF Type | DRT |
|---|---|---|---|---|---|
| Patient 1 | 5 | 201 | 28 | Permanent | No |
| Patient 2 | 5 | 261 | 28 | Paroxysmal | No |
| Patient 3 | 6 | 215 | 22 | Paroxysmal | No |
| Patient 4 | 5 | 143 | 22 | Permanent | Yes |
| Patient 5 | 6 | 281 | 31 | Permanent | Yes |
| Patient 6 | 4 | 176 | 28 | Permanent | Yes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mill, J.; Agudelo, V.; Olivares, A.L.; Pons, M.I.; Silva, E.; Nuñez-Garcia, M.; Morales, X.; Arzamendi, D.; Freixa, X.; Noailly, J.; et al. Sensitivity Analysis of In Silico Fluid Simulations to Predict Thrombus Formation after Left Atrial Appendage Occlusion. Mathematics 2021, 9, 2304. https://doi.org/10.3390/math9182304
Mill J, Agudelo V, Olivares AL, Pons MI, Silva E, Nuñez-Garcia M, Morales X, Arzamendi D, Freixa X, Noailly J, et al. Sensitivity Analysis of In Silico Fluid Simulations to Predict Thrombus Formation after Left Atrial Appendage Occlusion. Mathematics. 2021; 9(18):2304. https://doi.org/10.3390/math9182304
Chicago/Turabian StyleMill, Jordi, Victor Agudelo, Andy L. Olivares, Maria Isabel Pons, Etelvino Silva, Marta Nuñez-Garcia, Xabier Morales, Dabit Arzamendi, Xavier Freixa, Jérôme Noailly, and et al. 2021. "Sensitivity Analysis of In Silico Fluid Simulations to Predict Thrombus Formation after Left Atrial Appendage Occlusion" Mathematics 9, no. 18: 2304. https://doi.org/10.3390/math9182304
APA StyleMill, J., Agudelo, V., Olivares, A. L., Pons, M. I., Silva, E., Nuñez-Garcia, M., Morales, X., Arzamendi, D., Freixa, X., Noailly, J., & Camara, O. (2021). Sensitivity Analysis of In Silico Fluid Simulations to Predict Thrombus Formation after Left Atrial Appendage Occlusion. Mathematics, 9(18), 2304. https://doi.org/10.3390/math9182304







