Enumeration of Pentahexagonal Annuli in the Plane
Abstract
1. Introduction
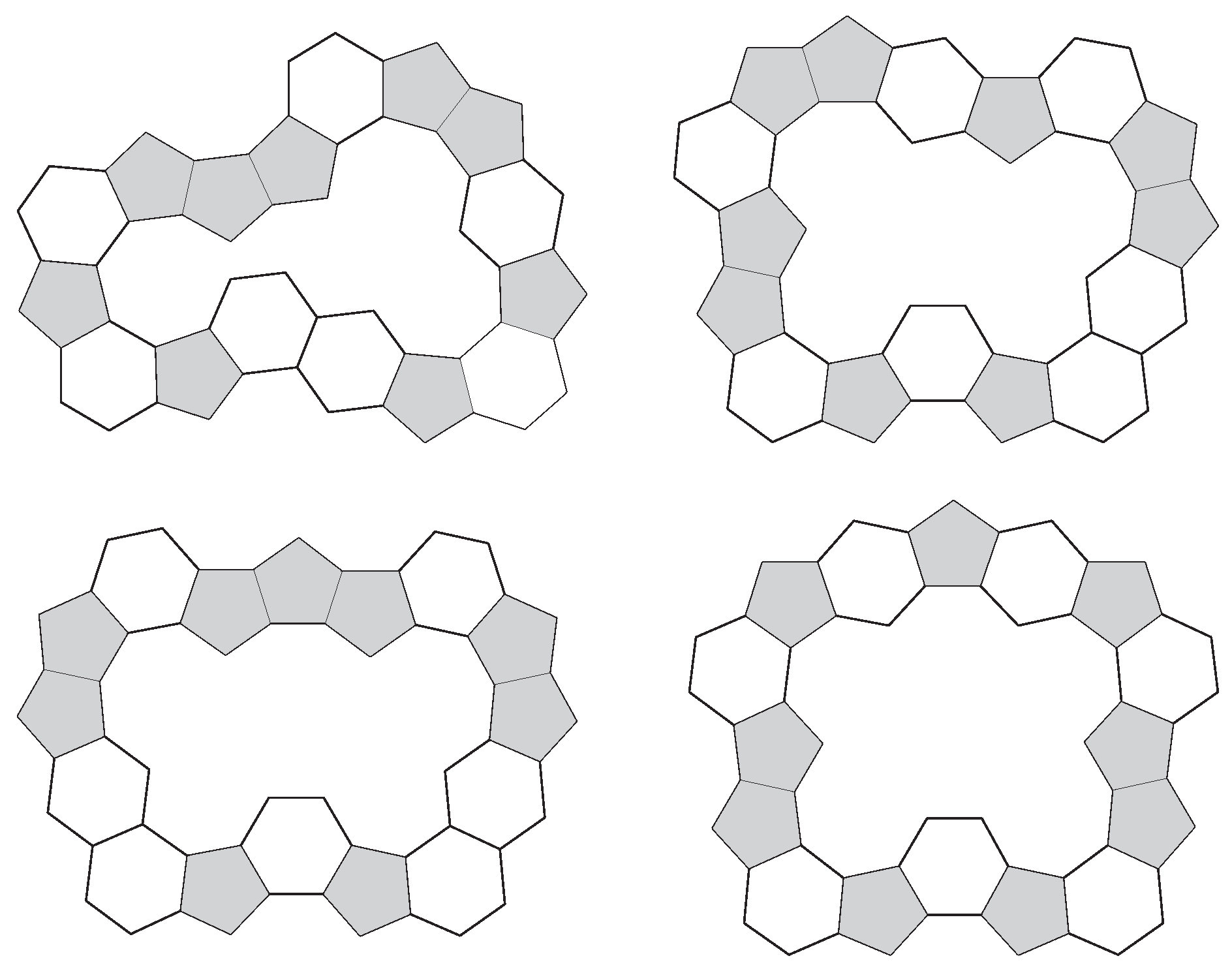
2. Classes of Annuli
3. Structural Properties of Annuli
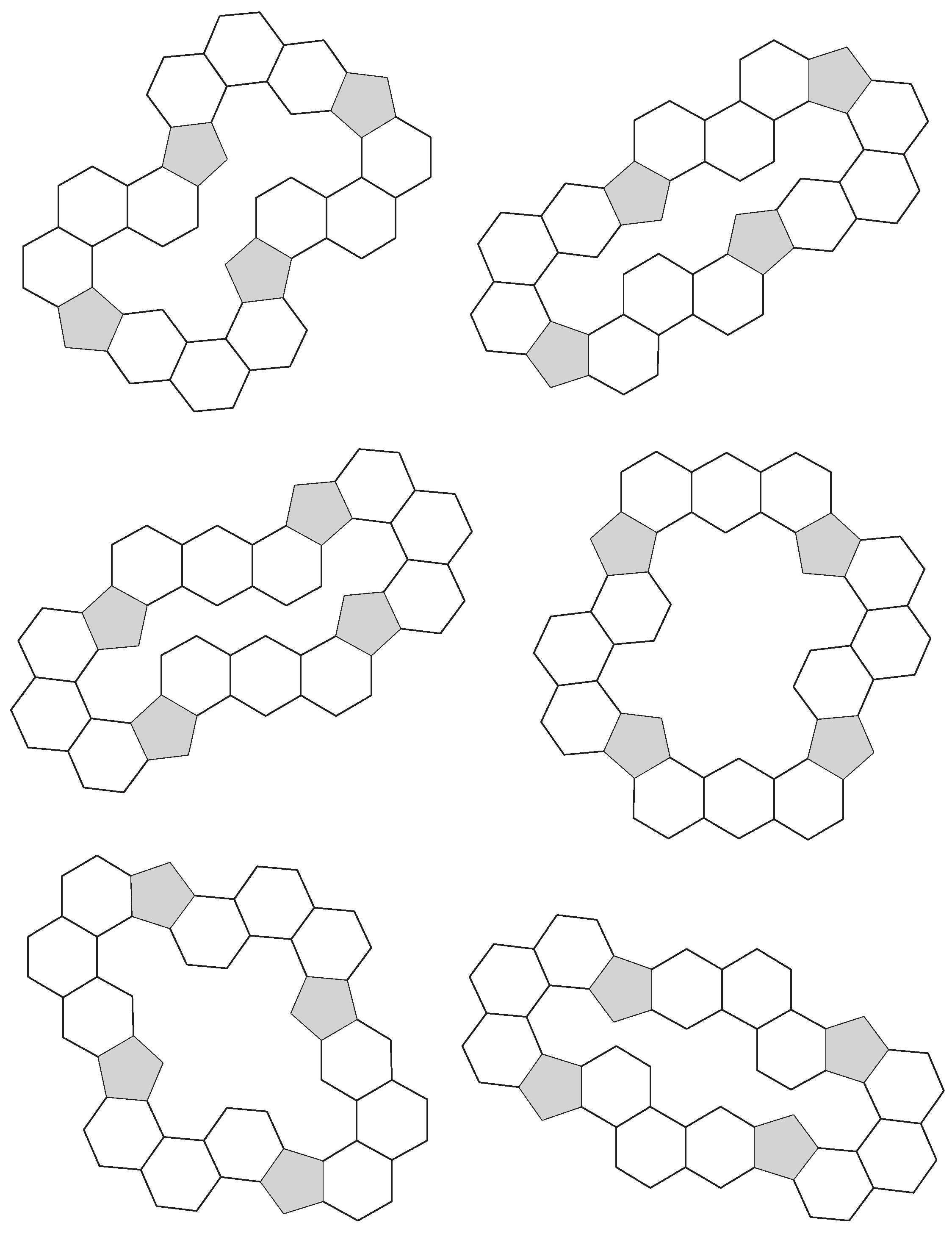
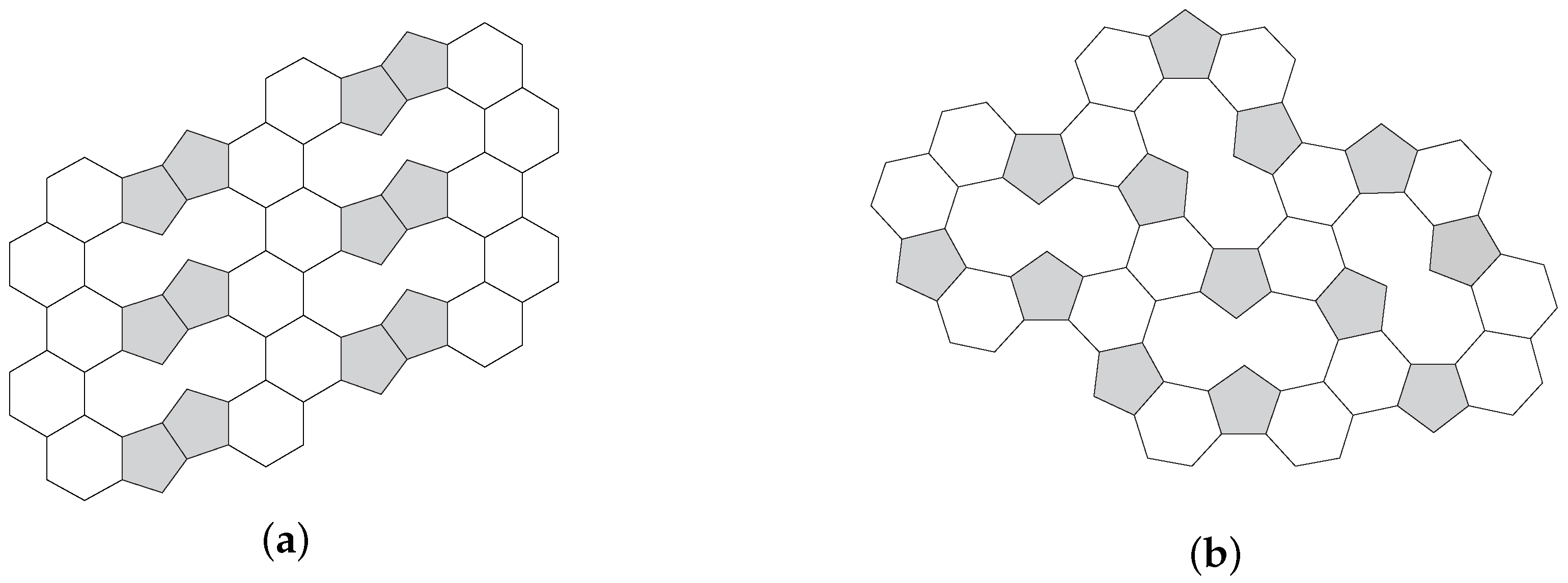
3.1. Pentagon Separation
3.2. Pentagon Distribution and Thickness
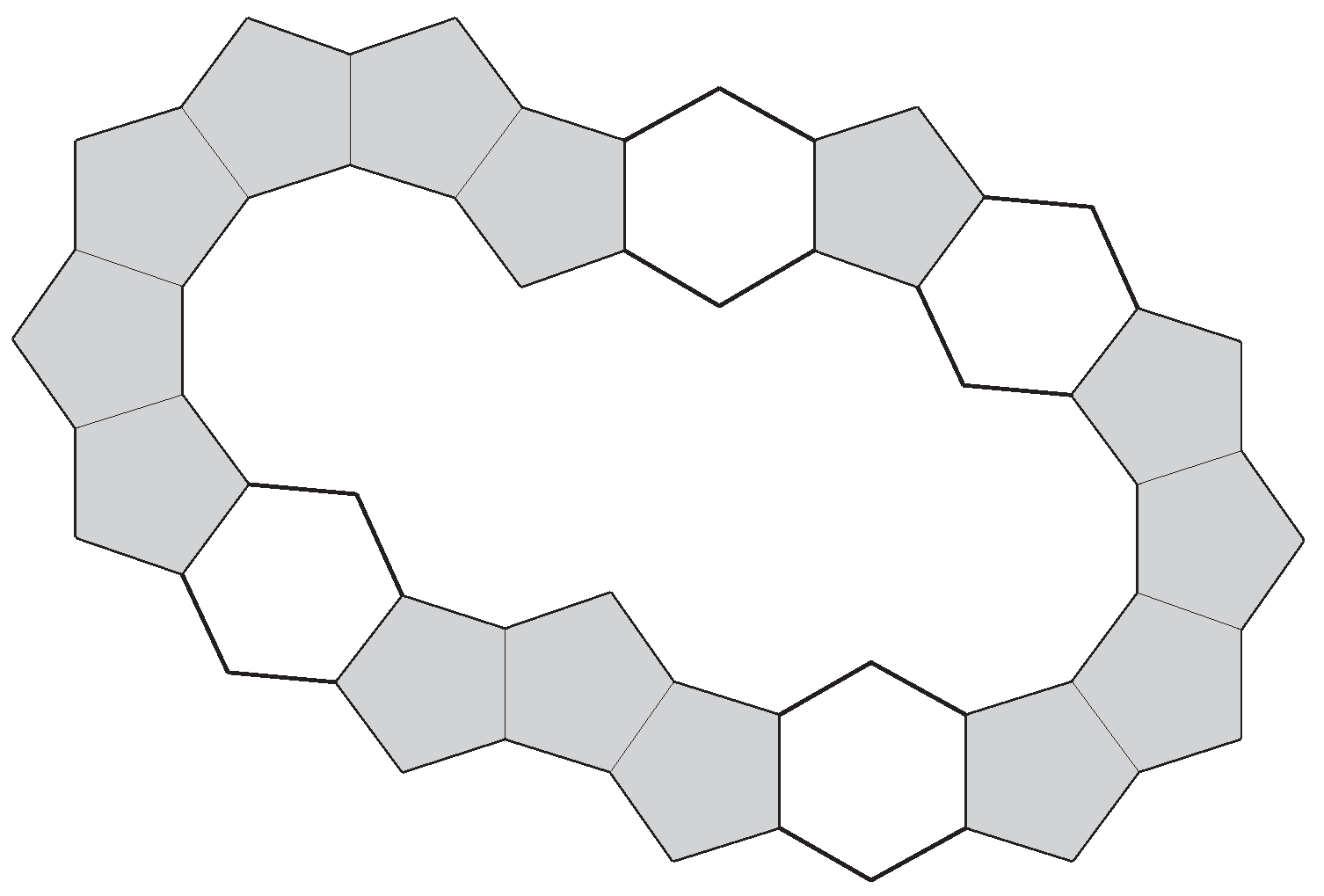
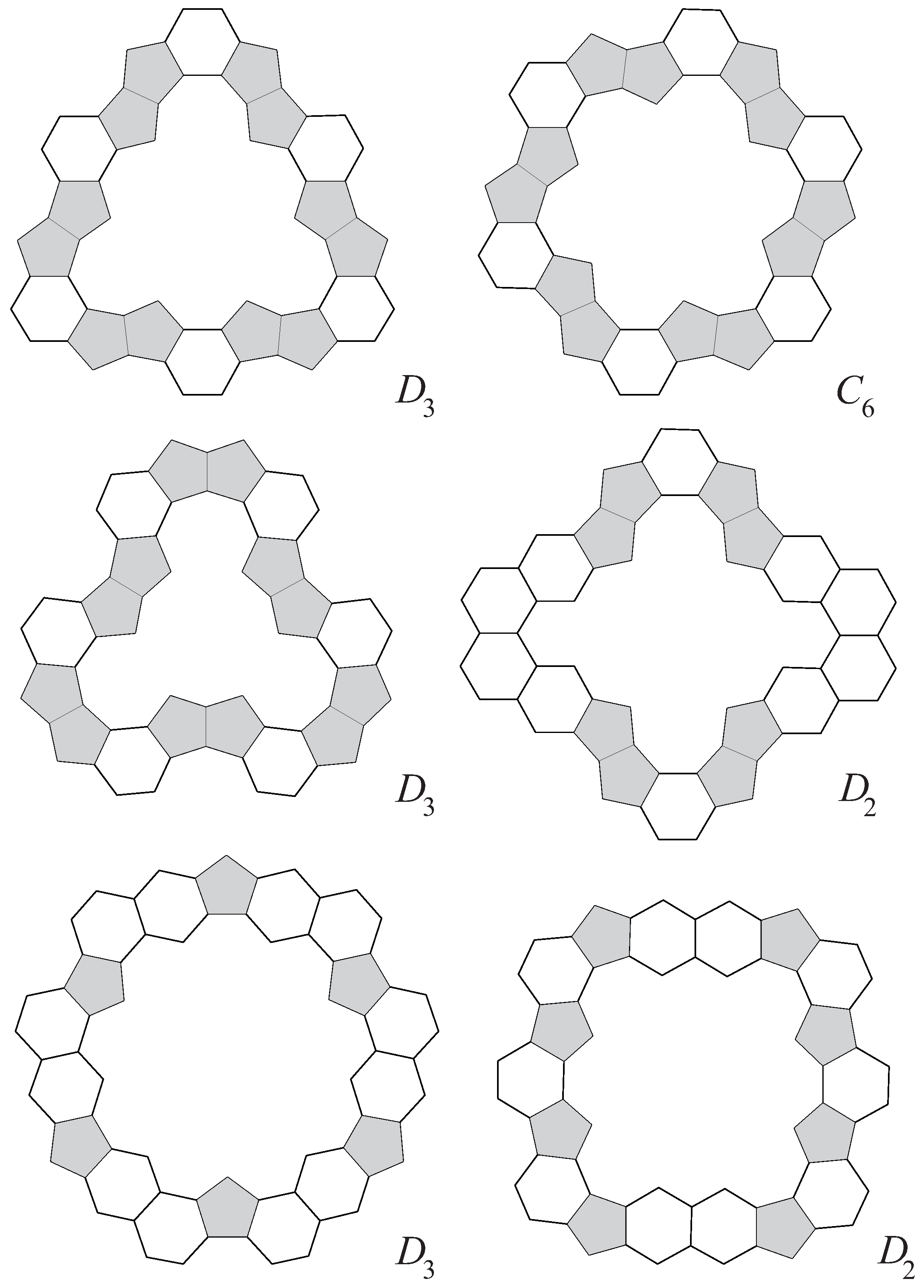
3.3. Symmetry of Annuli
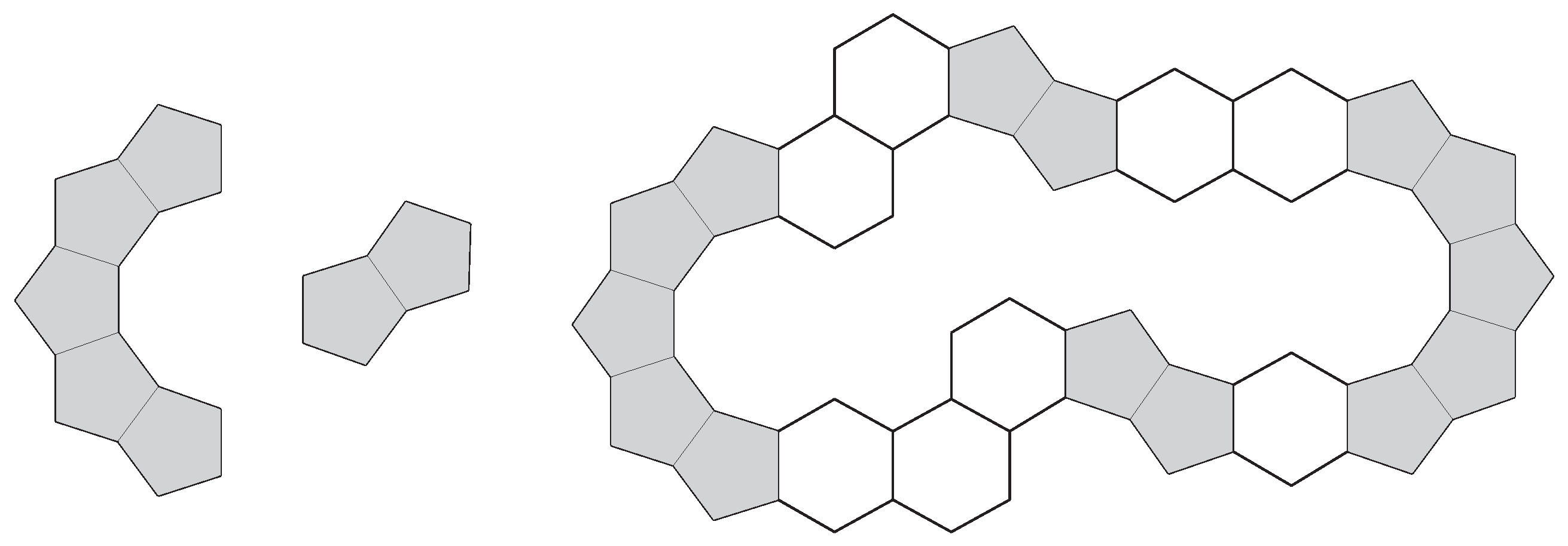
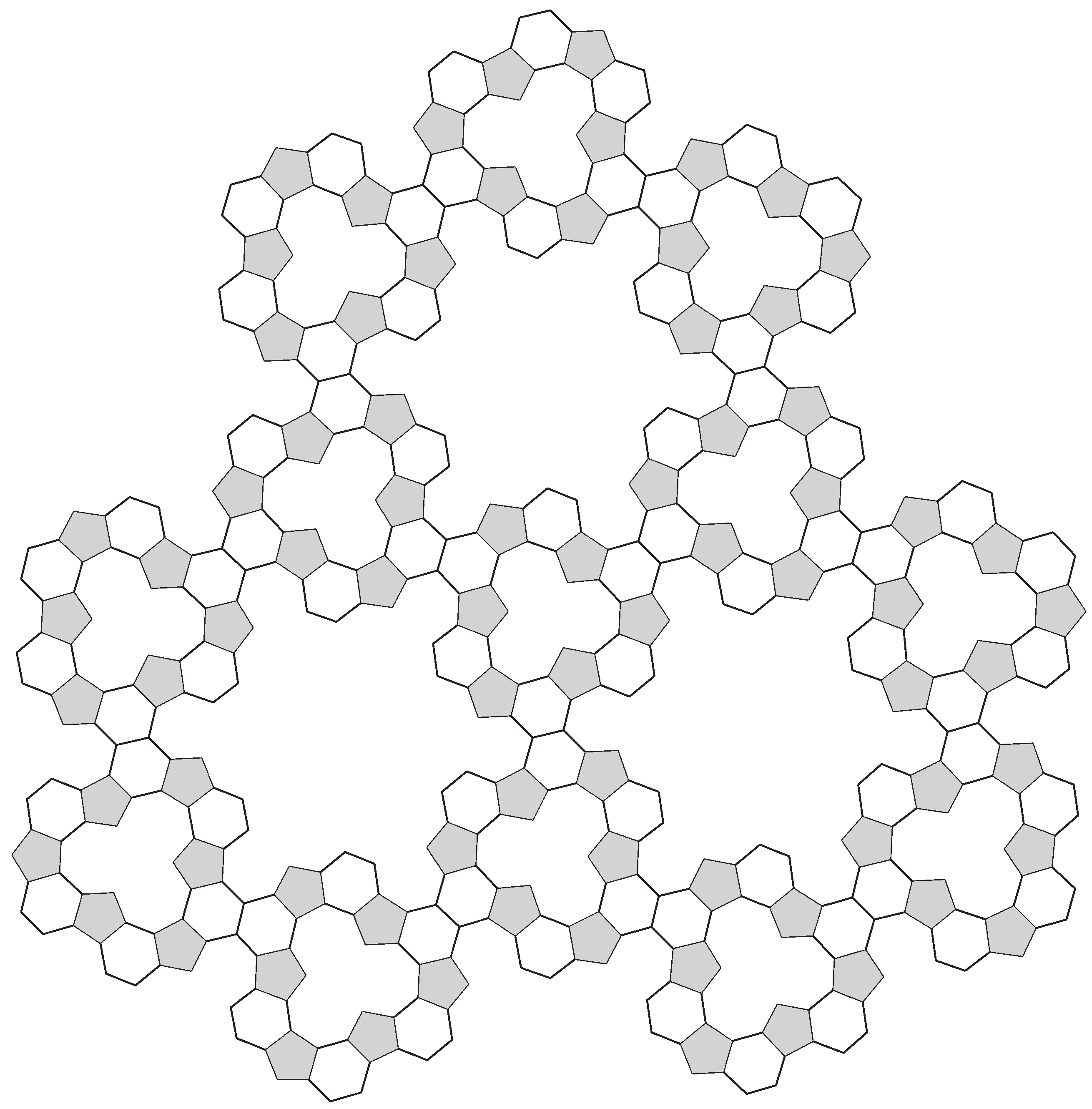
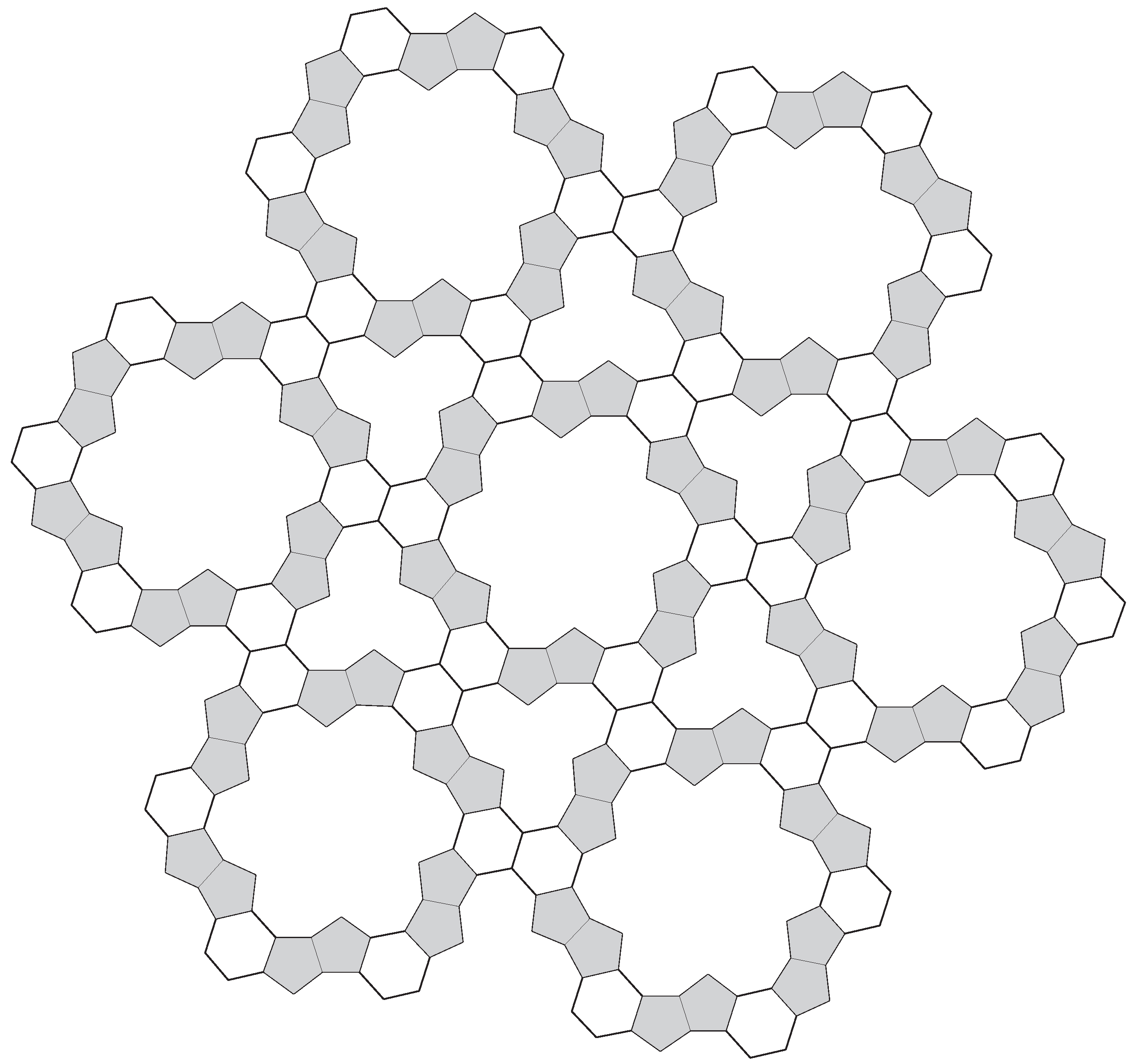
4. Complex Constructions
5. Conclusions
Supplementary Materials
Supplementary File 1Author Contributions
Funding
Conflicts of Interest
References
- Orchin, M.; Macomber, R.S.; Pinhas, A.R.; Wilson, R.M. The Vocabulary and Concepts of Organic Chemistry, 2nd ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005. [Google Scholar]
- Allis, D.G.; Spencer, J.T. Handbook of Nanoscience, Engineering, and Technology; Chapter Nanostructural Architectures from Molecular Building Blocks; CRC Press: Boca Raton, FL, USA, 2003; pp. 1601–1667. [Google Scholar]
- Lehn, J.M. Supamolecular Chemistry—Concepts and Perspectives; VCH Verlagsgesellschaft: Weinheim, NY, USA, 1995. [Google Scholar]
- Balaban, A.T. Challenging problems involving benzenoid polycycles and related systems. Pure Appl. Chem. 1982, 54, 1075–1096. [Google Scholar] [CrossRef][Green Version]
- Gutman, I.; Cyvin, S.J. Introduction to the Theory of Benzenoid Hydrocarbons; Springer: Berlin, Germany, 1989. [Google Scholar]
- Gutman, I.; Cyvin, S.J. (Eds.) Advances in the Theory of Benzenoid Hydrocarbons; Springer: Berlin, Germany, 1990; Volume 153. [Google Scholar]
- Gutman, I. (Ed.) Advances in the Theory of Benzenoid Hydrocarbons II; Springer: Berlin, Germany, 1992; Volume 162. [Google Scholar]
- Deza, M.; Fowler, P.W.; Shtogrin, M.; Vietze, K. Pentaheptite modifications of the graphite sheet. J. Chem. Inf. Comput. Sci. 2000, 40, 1325–1332. [Google Scholar] [CrossRef] [PubMed]
- Pop, R.; Medeleanu, M.; Diudea, M.V.; Szefler, B.; Cioslowski, J. Fullerenes patched by flowers with octagonal core. Cent. Eur. J. Chem. 2014, 12, 90–97. [Google Scholar] [CrossRef][Green Version]
- Sundholm, D.; Wirz, L.N.; Schwerdtfeger, P. Novel hollow all-carbon structures. Nanoscale 2015, 7, 15886–15894. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zhou, J.; Wang, Q.; Chen, X.; Kawazoe, Y.; Jena, P. Penta-graphene: A new carbon allotrope. Proc. Natl. Acad. Sci. USA 2015, 112, 2372–2377. [Google Scholar] [CrossRef]
- Grünbaum, B.; Shephard, G.C. Tilings by regular polygons. Patterns in the plane from Kepler to the present, including recent results and unsolved problems. Math. Mag. 1977, 50, 227–247. [Google Scholar] [CrossRef]
- Kuperberg, D.; Kuperberg, W. Double-lattice packings of convex bodies in the plane. Discret. Comput. Geom. 1990, 5, 389–397. [Google Scholar] [CrossRef]
- Bagina, O. Tiling the plane with congruent equilateral convex pentagons. J. Comb. Theory Ser. A 2004, 105, 221–232. [Google Scholar] [CrossRef]
- Zhu, H.; Balaban, A.T.; Klein, D.J.; Živković, T.P. Conjugated-circuit computations on two-dimensional carbon networks. J. Chem. Phys. 1994, 101, 5281–5292. [Google Scholar] [CrossRef]
- Fetter, H.L.; Arredondo Ruíz, J.H. On the overlappings in the unfoldings of the dodecahedron. J. Math. Sci. Adv. Appl. 2011, 9, 1–20. [Google Scholar]
- Pandey, S.; Ewing, M.; Kunas, A.; Nguyen, N.; Gracias, D.H.; Menon, G. Algorithmic design of self-folding polyhedra. Proc. Natl. Acad. Sci. USA 2011, 108, 19885–19890. [Google Scholar] [CrossRef] [PubMed]
- Fowler, P.W.; Manolopoulos, D.E. An Atlas of Fullerenes; Dover Publications Inc.: New York, NY, USA, 2006. [Google Scholar]
- Lord, E.A.; Mackay, A.L.; Ranganathan, S. New Geometries for New Materials; Cambridge University Press: Cambridge, UK, 2006. [Google Scholar]
- Sadoc, J.F.; Mosseri, R. Geometrical Frustration; Cambridge University Press: Cambridge, UK, 1999. [Google Scholar]
- Jenny, W.; Peter, R. Coronaphene, eine neue Gruppe cyclischer Kohlenwasserstoffe. Angew. Chem. 1965, 77, 1027. [Google Scholar] [CrossRef]
- Barth, W.E.; Lawton, R.G. Dibenzo[ghi,mno]fluoranthene. J. Am. Chem. Soc. 1966, 88, 380–381. [Google Scholar] [CrossRef]
- Barth, W.E.; Lawton, R.G. The synthesis of corannulene. J. Am. Chem. Soc. 1971, 93, 1730–1745. [Google Scholar]
- Cyvin, S.J.; Brunvoll, J.; Chen, R.S.; Cyvin, B.N.; Zhang, F.J. Theory of Coronoid Hydrocarbons II; Number 62 in Lecture Notes in Chem.; Springer: Berlin, Germany, 1994. [Google Scholar]
- Liu, C.; Sandoval-Salinas, M.E.; Hong, Y.; Gopalakrishna, T.Y.; Phan, H.; Aratani, N.; Herng, T.S.; Ding, J.; Yamada, H.; Kim, D.; et al. Macrocyclic polyradicaloids with unusual super-ring structure and global aromaticity. Chem 2018, 4, 1586–1595. [Google Scholar] [CrossRef]
- Stępień, M. An aromatic riddle: Decoupling annulene conjugation in coronoid cacrocycles. Proc. Natl. Acad. Sci. USA 2018, 4, 1481–1483. [Google Scholar] [CrossRef]
- Heddle, J.G. Protein cages, rings and tubes: useful components of future nanodevices? Nanotechnol. Sci. Appl. 2008, 1, 67–78. [Google Scholar] [CrossRef]
- Rosenfeld, V.R. The fractal nature of folds and the Walsh copolymers. J. Math. Chem. 2016, 54, 559–571. [Google Scholar] [CrossRef]
- Ke, Y.; Douglas, S.M.; Liu, M.; Sharma, J.; Cheng, A.; Leung, A.; Liu, Y.; Shih, W.M.; Yan, H. Multilayer DNA origami packed on a square lattice. J. Am. Chem. Soc. 2009, 131, 15903–15908. [Google Scholar] [CrossRef]
- Chandrasekhar, S.; Sadashiva, B.K.; Suresh, K.A. Liquid crystals of disc-like molecules. Pramana 1977, 9, 471–480. [Google Scholar] [CrossRef]
- Laschat, S.; Baro, A.; Steinke, N.; Giesselmann, F.; Hagele, C.; Scalia, G.; Judele, R.; Kapatsina, E.; Sauer, S.; Schreivogel, A.; et al. Discotic liquid crystals: from tailor-made synthesis to plastic electronics. Angew. Chem. Int. Ed. 2007, 46, 4832–4887. [Google Scholar] [CrossRef] [PubMed]
- Lin, L. Bowlic liquid crytals. Mol. Cryst. Liq. Cryst. 1987, 146, 41–54. [Google Scholar]
- Dobrynin, A.A. Generation of graphs of unbranched pentagonal and pentahexagonal systems. Vychisl. Sist. 1991, 140, 143–206. (In Russian) [Google Scholar]
- Cyvin, S.J.; Brunvoll, J.; Cyvin, B.N. Theory of Coronoid Hydrocarbons; Number 54 in Lecture Notes in Chem.; Springer: Berlin, Germany, 1991. [Google Scholar]
- Bašić, N.; Fowler, P.W.; Pisanski, T. Coronoids, patches and generalized altans. J. Math. Chem. 2016, 54, 977–1009. [Google Scholar] [CrossRef]
- Dobrynin, A.A. Enumeration of some subclasses of graphs of unbranched hexagonal systems. Vychisl. Sist. 1990, 136, 16–34. (In Russian) [Google Scholar]
- Zhang, F.; Brunvoll, J.; Cyvin, S.J.; Cyvin, B.N.; Guo, X.; Dobrynin, A.A. Theory of helicenic hydrocarbons. Part 5: irreducible helicenes and their enumeration. Struct. Chem. 1996, 7, 263–271. [Google Scholar]
- Balaban, A.T.; Harary, F. Chemical graphs-V: Enumeration and proposed nomenclature of benzenoid cata-condensed polycyclic aromatic hydrocarbons. Tetrahedron 1968, 24, 2505–2516. [Google Scholar] [CrossRef]
- He, W.; He, W. Generation and enumeration of planar polycyclic aromatic hydrocarbons. Tetrahedron 1986, 42, 5291–5299. [Google Scholar]
- Knop, J.V.; Müller, W.R.; Szymanski, K.; Trinajstić, N. Computer Generation of Certain Classes of Molecules; SKTH/Kemija u Industriji: Zagreb, Croatia, 1985. [Google Scholar]
- Ratko, T.; Kovačevič, M. Generating and counting unbranched catacondensed benzenoids. J. Chem. Inf. Comput. Sci. 1988, 1, 29–31. [Google Scholar]
- Trinajstić, N.; Nikolić, S.; Knop, J.V.; Müller, W.R.; Szymanski, K. Computational Chemical Graph Theory: Characterization, Enumeration, and Generation of Chemical Structures by Computer Methods; Ellis Horwood: New York, NY, USA, 1991. [Google Scholar]
- Vöge, M.; Guttmann, A.J.; Jensen, I. On the number of benzenoid hydrocarbons. J. Chem. Inf. Comput. Sci. 2002, 42, 456–466. [Google Scholar] [CrossRef]
- Brinkmann, G.; Caporossi, G.; Hansen, P. A survey and new results on computer enumeration of polyhex and fusene hydrocarbons. J. Chem. Inf. Comput. Sci. 2003, 43, 842–851. [Google Scholar] [CrossRef] [PubMed]
- Brinkmann, G.; Dobrynin, A.A.; Krause, A. Fast generation of polycyclic chains with arbitrary ring sizes. MATCH Commun. Math. Comput. Chem. 2000, 41, 137–144. [Google Scholar]
- Dobrynin, A.A. Generation of graphs of unbranched pentahexagonal catacondensed systems. Croat. Chem. Acta 1993, 66, 91–100. [Google Scholar]
- Rosenfeld, V.R.; Dobrynin, A.A.; Oliva, J.M.; Rue, J. Pentagonal chains and annuli as models for designing nanostructures from cages. J. Math. Chem. 2016, 54, 765–776. [Google Scholar] [CrossRef]
- Qian, J.; Zhang, F. Counting the cyclocized polyphenacenes. J. Comput. Chem. 2010, 31, 2577–2584. [Google Scholar] [CrossRef] [PubMed]
- Deng, K.; Zhang, X. Enumeration of cyclic polyazulenoids. J. Math. Chem. 2016, 54, 416–427. [Google Scholar] [CrossRef]
- Zhang, X.; Deng, K. Enumeration of Möbius type cyclic polyazulenoids. J. Math. Chem. 2017, 55, 132–141. [Google Scholar] [CrossRef]
- Albertazzi, E.; Domene, C.; Fowler, P.W.; Theine, T.; Sievert, G.; Van Alieno, C.; Terzetto, F. Pentagon adjacency as a determinant of fullerene stability. Phys. Chem. Chem. Phys. 1999, 1, 2913–2918. [Google Scholar] [CrossRef]
- Schmalz, T.G.; Seitz, W.A.; Klein, D.J.; Hite, G.E. Elemental carbon cages. J. Am. Chem. Soc. 1988, 110, 1113–1127. [Google Scholar] [CrossRef]
- Goedgebeur, J.; McKay, B.D. Fullerenes with distant pentagons. MATCH Commun. Math. Comput. Chem. 2015, 74, 659–672. [Google Scholar]
- Raghavachari, K. Ground state of C84: Two almost isoenergetic isomers. Chem. Phys. Lett. 1992, 190, 397–400. [Google Scholar] [CrossRef]
- Rodriguez-Fortea, A.; Alegret, N.; Balch, A.L.; Poblet, J.M. The maximum pentagon separation rule provides a guideline for the structures of endohedral metallofullerenes. Nat. Chem. 2010, 2, 955–961. [Google Scholar] [CrossRef] [PubMed]
- Schein, S.; Friedrich, T. A geometric constraint, the head-to-tail exclusion rule, may be the basis for the isolated-pentagon rule in fullerenes with more than 60 vertices. Proc. Natl. Acad. Sci. USA 2008, 105, 19142–19147. [Google Scholar] [CrossRef] [PubMed]
- Bašić, N.; Brinkmann, G.; Fowler, P.W.; Pisanski, T.; Van Cleemput, N. Sizes of pentagonal clusters in fullerenes. J. Math. Chem. 2017, 55, 1669–1682. [Google Scholar] [CrossRef]
- Babić, D.; Klein, D.J.; Sah, C.H. Symmetry of fullerenes. Chem. Phys. Lett. 1993, 211, 235–241. [Google Scholar] [CrossRef]
- Knor, M.; Škrekovski, R.; Tepeh, A. Mathematical aspects of Wiener index. ARS Math. Contemp. 2016, 11, 327–352. [Google Scholar] [CrossRef]
- Kovič, J.; Pisanski, T.; Balaban, A.T.; Fowler, P.W. On symmetries of benzenoid systems. MATCH Commun. Math. Comput. Chem. 2014, 72, 3–26. [Google Scholar]
- Mandal, B.; Banerjee, M.; Mukherjee, A.K. Construction of planar graphs for IPR fullerenes using 5- and 6-fold rotational symmetry: Some eigenspectral analysis. Phys. Chem. Chem. Phys. 2004, 6, 2040–2043. [Google Scholar] [CrossRef]
- Schwerdtfeger, P.; Wirz, L.N.; Avery, J. The topology of fullerenes. WIREs Comput. Mol. Sci. 2015, 5, 96–145. [Google Scholar] [CrossRef]
- Whitesides, G.M.; Boncheva, M. Beyond molecules: self-assembly of mesoscopic and macroscopic components. Proc. Natl. Acad. Sci. USA 2002, 99, 4769–4774. [Google Scholar] [CrossRef]

| r | p | Total | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 4 | 6 | 8 | 9 | 10 | 11 | 12 | 14 | 15 | 18 | ||
| 10 | 2 | - | - | - | 1 | 3 | |||||
| 12 | 11 | 2 | - | - | 1 | - | - | 14 | |||
| 13 | 3 | - | - | - | - | - | - | 3 | |||
| 14 | 61 | 3 | 18 | - | 6 | - | - | 1 | 89 | ||
| 15 | 49 | 8 | - | - | 1 | - | - | - | - | 58 | |
| 16 | 358 | 32 | 124 | 4 | 33 | - | - | 5 | - | 556 | |
| 17 | 515 | 60 | 42 | 1 | 3 | - | - | - | 621 | ||
| 18 | 2322 | 309 | 1137 | 1 | 199 | 9 | 104 | 52 | 1 | 5 | 4139 |
| r | Total | ||||
|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | ||
| 10 | 2 | 1 | - | - | 3 |
| 12 | 7 | 5 | 2 | - | 14 |
| 13 | 2 | 1 | - | - | 3 |
| 14 | 55 | 20 | 14 | - | 89 |
| 15 | 36 | 18 | 4 | - | 58 |
| 16 | 348 | 123 | 62 | 23 | 556 |
| 17 | 388 | 172 | 47 | 14 | 621 |
| 18 | 2758 | 846 | 322 | 213 | 4139 |
| r | p | Total | ||
|---|---|---|---|---|
| 4 | 6 | 8 | ||
| 10 | 1 | - | - | 1 |
| 12 | 2 | 1 | - | 3 |
| 13 | - | - | - | 0 |
| 14 | 14 | 1 | - | 15 |
| 15 | - | 3 | - | 3 |
| 16 | 23 | 4 | 4 | 31 |
| 17 | 14 | 6 | - | 20 |
| 18 | 183 | 7 | 54 | 244 |
| r | Total | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 14 | 18 | ||
| 10 | 1 | 1 | - | - | - | - | - | - | 1 | 3 | |||
| 12 | 7 | 6 | - | - | 1 | - | - | - | - | 14 | |||
| 13 | 1 | 2 | - | - | - | - | - | - | - | 3 | |||
| 14 | 34 | 38 | 8 | 6 | 2 | - | - | - | - | 1 | 89 | ||
| 15 | 22 | 36 | - | - | - | - | - | - | - | - | 58 | ||
| 16 | 208 | 248 | 60 | 30 | 5 | - | 4 | 1 | - | - | 556 | ||
| 17 | 233 | 363 | 10 | 13 | 2 | - | - | - | - | - | 621 | ||
| 18 | 1381 | 1930 | 469 | 265 | 48 | 21 | 15 | 2 | 3 | - | - | 5 | 4139 |
| r | p | Total | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 4 | 6 | 8 | 9 | 10 | 11 | 12 | 14 | 15 | 18 | ||
| 10 | 2 | - | - | - | 1 | 3 | |||||
| 12 | 10 | 2 | - | - | 1 | - | - | 13 | |||
| 13 | - | - | - | - | - | - | - | 0 | |||
| 14 | 39 | - | 18 | - | 6 | - | - | 1 | 64 | ||
| 15 | - | 6 | - | - | 1 | - | - | - | - | 7 | |
| 16 | 142 | 3 | 97 | 2 | 26 | - | - | 4 | - | 274 | |
| 17 | - | - | 10 | - | 3 | - | - | - | - | 13 | |
| 18 | 499 | 26 | 574 | - | 118 | 1 | 93 | 40 | 1 | 5 | 1357 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dobrynin, A.A.; Rosenfeld, V.R. Enumeration of Pentahexagonal Annuli in the Plane. Mathematics 2019, 7, 1156. https://doi.org/10.3390/math7121156
Dobrynin AA, Rosenfeld VR. Enumeration of Pentahexagonal Annuli in the Plane. Mathematics. 2019; 7(12):1156. https://doi.org/10.3390/math7121156
Chicago/Turabian StyleDobrynin, Andrey A., and Vladimir R. Rosenfeld. 2019. "Enumeration of Pentahexagonal Annuli in the Plane" Mathematics 7, no. 12: 1156. https://doi.org/10.3390/math7121156
APA StyleDobrynin, A. A., & Rosenfeld, V. R. (2019). Enumeration of Pentahexagonal Annuli in the Plane. Mathematics, 7(12), 1156. https://doi.org/10.3390/math7121156





