Systematic Predictive Analysis of Personalized Life Expectancy Using Smart Devices
Abstract
1. Introduction
2. Analysis of Existing Works
3. Review Conclusions
- The overwhelming number of variables that could be considered in predicting LE;
- Lack of accumulated data in one storage location for data processing and analysis to generate meaningful data;
- Difficulty of centralizing heterogeneous networks from different countries and regions;
- Unpredictable and fast changing lifestyle of humans with the increase of sophisticated technologies;
- Limited methods of health data collection such as data only from patients in a healthcare environment (hospitals, insurance companies), which may exclude the general population.
4. Work in Progress
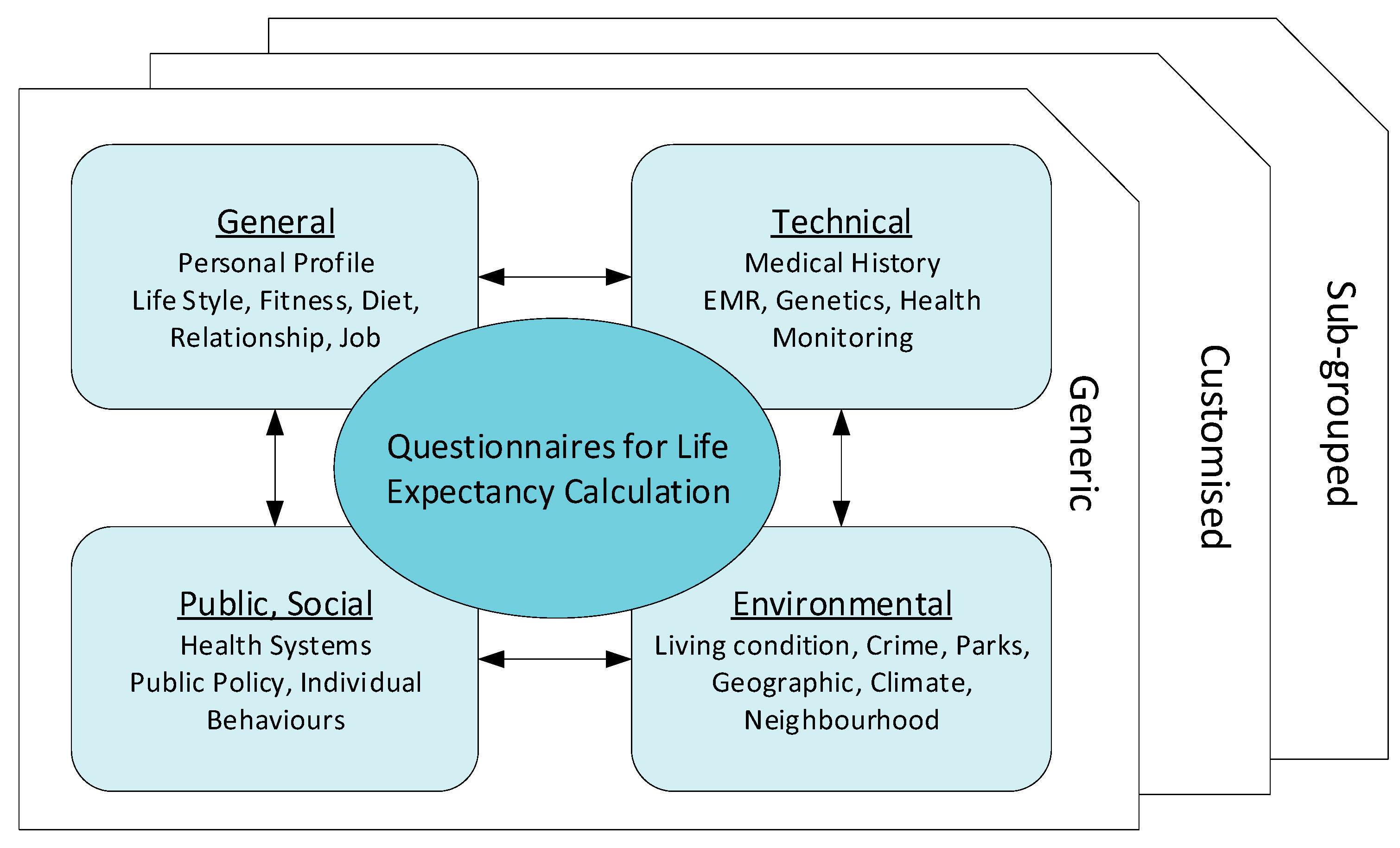
4.1. Raw Data Collection
4.1.1. General Questionnaire
4.1.2. Technical Questionnaire
- Individual lifestyle behaviors e.g., spending patterns, exercise, diet;
- Physical and social environments e.g., living density, pollution levels;
- Socioeconomic factors e.g., education level, financial status;
- Health outcomes e.g., illnesses;
- Health systems e.g., health insurance status.
4.1.3. Data Source Selection
4.1.4. Other Factors
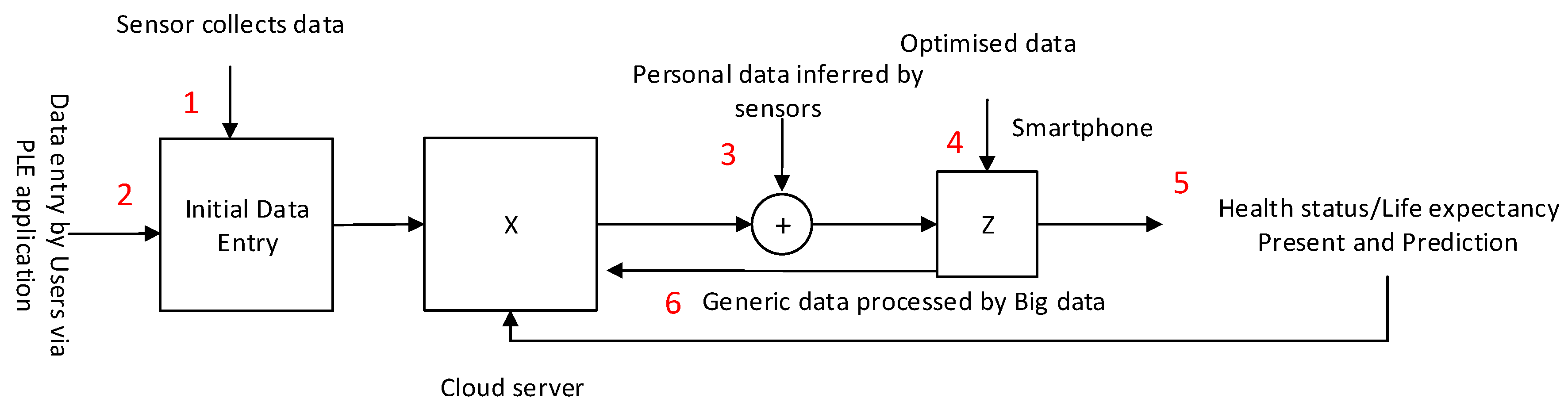
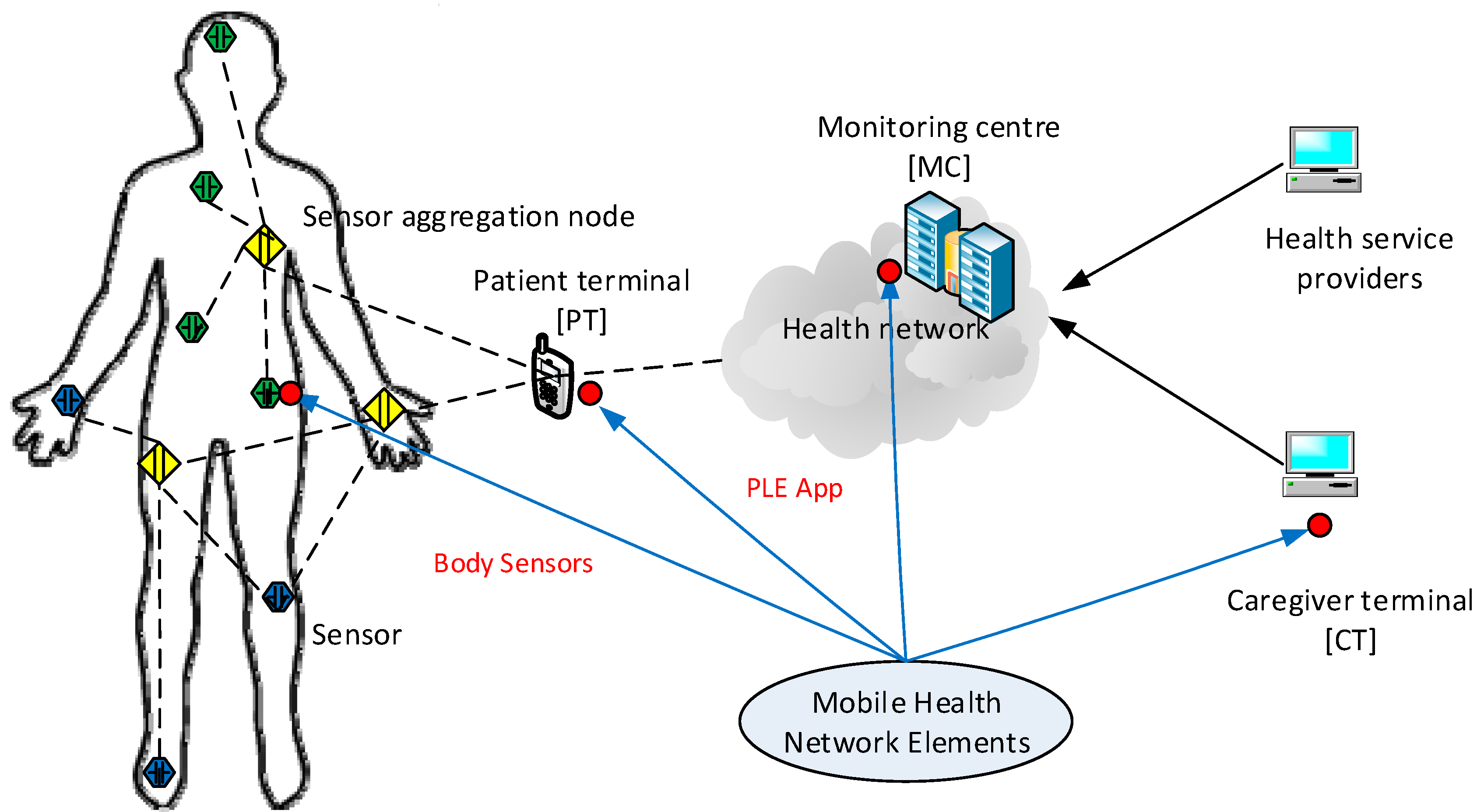
4.2. Information Flow of Data Creation
- A user installs a PLE app on their smartphone and adds personal information and health data as requested by the app. The user also gives a consent to the service provider so that they can access and retrieve their health data from the user’s health record located in a central server.
- The app communicates with the central server which initiates user registration and basic provisioning processes. It also pairs up (if needed) with the user’s body sensors to be ready for data transfer.
- The user’s body sensors collect and transfer the sensed data to the smartphone and follow instructions of when and how to collect and transmit data.
- When data have been collected, the smartphone app transmits them to the central server that will prepare and generate the initial results, and send them back to the app for optimization. The app may collect further data from the sensors as programmed prior to displaying the outcome of PLE for the user.
- The central server continuously updates and improves data quality to input to the generic group data as classified and grouped for the user. Each element fine tunes the data by inference algorithms before transmission, and discern how to handle the priority; e.g., QoS.
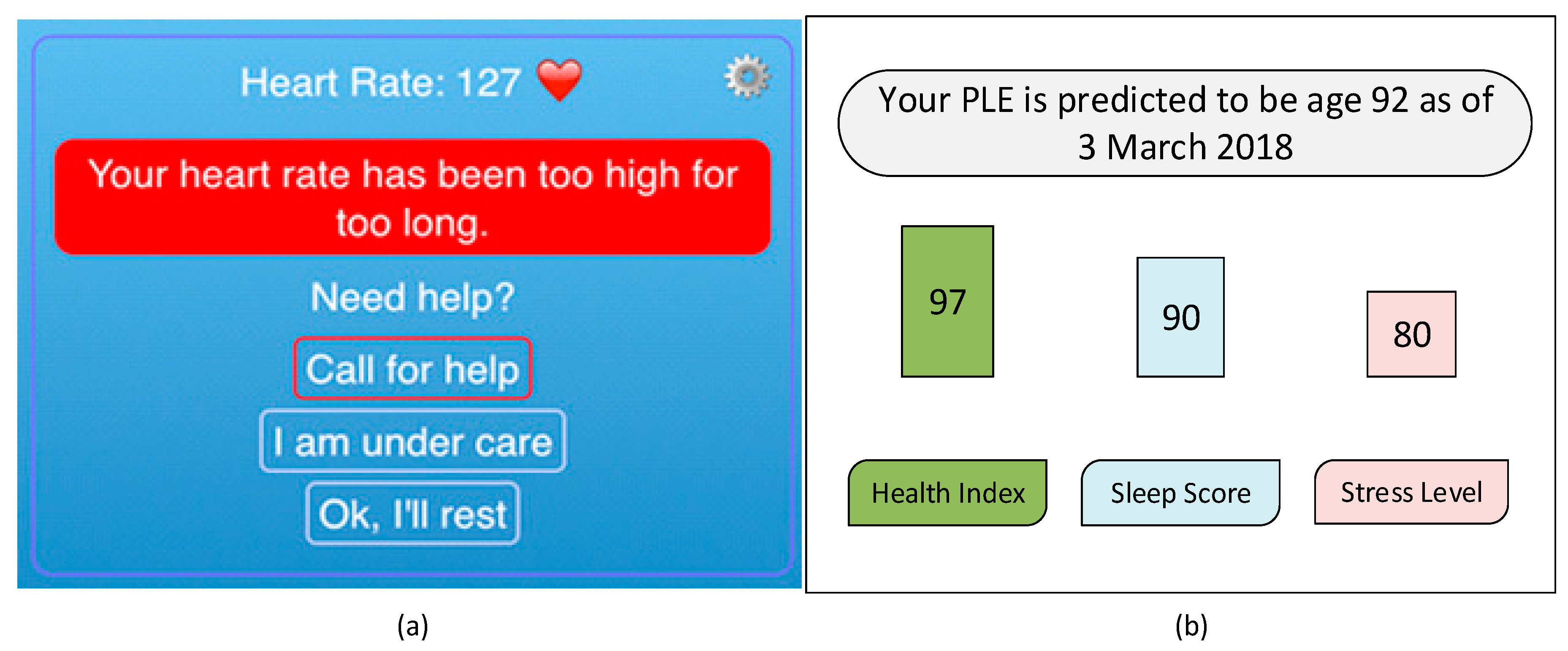
4.3. App Design
5. Applications
6. Conclusions and Future Works
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Katsis, Y.; Balac, N.; Chapman, D.; Kapoor, M.; Block, J.; Griswold, W.G.; Huang, J.; Koulouris, N.; Menarini, M.; Nandigam, V. Big Data Techniques for Public Health: A Case Study. In Proceedings of the 2017 IEEE/ACM International Conference on Connected Health: Applications, Systems and Engineering Technologies (CHASE), Philadelphia, PA, USA, 17–19 July 2017; pp. 222–231. [Google Scholar]
- Digiovanni, S.L.; Guaragnella, C.; Rizzi, M.; Falagario, M. Healthcare system: A digital green filter for smart health early cervical cancer diagnosis. In Proceedings of the 2016 IEEE 2nd International Forum on Research and Technologies for Society and Industry Leveraging a Better Tomorrow (RTSI), Bologna, Italy, 7–9 September 2016; pp. 1–6. [Google Scholar]
- Saha, H.N.; Raun, N.F.; Saha, M. Monitoring patient’s health with smart ambulance system using Internet of Things (IOTs). In Proceedings of the 2017 8th Annual Industrial Automation and Electromechanical Engineering Conference (IEMECON), Bangkok, Thailand, 16–18 August 2017; pp. 91–95. [Google Scholar]
- Bakar, N.H.B.A.; Abdullah, K.; Islam, M.R. Wireless Smart Health Monitoring System via Mobile Phone. In Proceedings of the 2016 International Conference on Computer and Communication Engineering (ICCCE), Kuala Lumpur, Malaysia, 26–27 July 2016; pp. 213–218. [Google Scholar]
- Ali, Z.; Muhammad, G.; Alhamid, M.F. An automatic health monitoring system for patients suffering from voice complications in smart cities. IEEE Access 2017, 5, 3900–3908. [Google Scholar] [CrossRef]
- Banaee, H.; Ahmed, M.U.; Loutfi, A. Data mining for wearable sensors in health monitoring systems: A review of recent trends and challenges. Sensors 2013, 13, 17472–17500. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.J.; Larkin, H. Inference of personal sensors in the internet of things. Int. J. Inf. Commun. Technol. Appl. 2016, 2, 1–23. [Google Scholar] [CrossRef]
- Kang, J.J.W. An Inference System Framework for Personal Sensor Devices in Mobile Health and Internet of Things Networks; Deakin University: Victoria, Australia, 2017. [Google Scholar]
- de Miguel-Bilbao, S.; Aguirre, E.; Lopez Iturri, P.; Azpilicueta, L.; Roldán, J.; Falcone, F.; Ramos, V. Evaluation of Electromagnetic Interference and Exposure Assessment from s-Health Solutions Based on Wi-Fi Devices. BioMed Res. Int. 2015, 2015, 9. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Adibi, S. A Review of Security Protocols in mHealth Wireless Body Area Networks (WBAN). In Proceedings of the Future Network Systems and Security, Paris, France, 11–13 June 2015; pp. 61–83. [Google Scholar]
- Blobel, B. Comparing approaches for advanced e-health security infrastructures. Int. J. Med. Inform. 2007, 76, 454–459. [Google Scholar] [CrossRef] [PubMed]
- Diamandis, P. What Silicon Valley Is Doing to Make Humans Live Longer. Available online: https://www.businessinsider.in/what-silicon-valley-is-doing-to-make-humans-live-longer/articleshow/63140991.cms (accessed on 7 March 2018).
- Łaszkiewicz, A.; Szymczak, S.; Cebrat, S. Prediction of the human life expectancy. Theory Biosci. 2003, 122, 313–320. [Google Scholar] [CrossRef]
- Sardi, B. Yuzurihara, the village of long life, reveals its secrets. Knowl. Health 2006, 10, 1–4. [Google Scholar]
- Bhosale, A.; Sundaram, K. Life prediction equation for human beings. In Proceedings of the 2010 International Conference on Bioinformatics and Biomedical Technology (ICBBT), Chengdu, China, 16–18 April 2010; pp. 266–268. [Google Scholar]
- Jafelice, R.; Barros, L. Fuzzy set-based model to compute the life expectancy of HIV infected populations. In Proceedings of the IEEE Annual Meeting of the Fuzzy Information, Banff, AL, Canada, 27–30 June 2004; pp. 314–318. [Google Scholar]
- Agrawal, A.; Mathias, J.; Baker, D.; Choudhary, A. Five year life expectancy calculator for older adults. In Proceedings of the 2016 IEEE 16th International Conference on Data Mining Workshops (ICDMW), Barcelona, Spain, 12–15 December 2016; pp. 1280–1283. [Google Scholar]
- Agrawal, J.S.M.A.; Baker, D.W.; Choudhary, A. Five Year Life Expectancy Calculator for Older Adults. 2016. Available online: http://info.eecs.northwestern.edu/FiveYearLifeExpectancyCalculator (accessed on 10 August 2018).
- Gil-Herrera, E.; Yalcin, A.; Tsalatsanis, A.; Barnes, L.E.; Djulbegovic, B. Rough set theory based prognostication of life expectancy for terminally ill patients. In Proceedings of the 2011 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, EMBC, Boston, MA, USA, 30 August–3 September 2011; pp. 6438–6441. [Google Scholar]
- Kerdprasop, N.K.A.K. Association of economic and environmental factors to life expectancy of people in the Mekong basin. In Proceedings of the 2017 12th IEEE Conference on Industrial Electronics and Applications (ICIEA), Siem Reap, Cambodia, 18–20 June 2017. [Google Scholar]
- Pascariu, M.D.; Canudas-Romo, V.; Vaupel, J.W. The double-gap life expectancy forecasting model. Insur. Math. Econ. 2018, 78, 339–350. [Google Scholar] [CrossRef]
- Lee, R.D.; Carter, L.R. Modeling and forecasting US mortality. J. Am. Stat. Assoc. 1992, 87, 659–671. [Google Scholar]
- Cairns, A.J.; Blake, D.; Dowd, K. A two-factor model for stochastic mortality with parameter uncertainty: Theory and calibration. J. Risk Insur. 2006, 73, 687–718. [Google Scholar] [CrossRef]
- Sammon, J.D.; Abdollah, F.; D’Amico, A.; Gettman, M.; Haese, A.; Suardi, N.; Vickers, A.; Trinh, Q.-D. Predicting Life Expectancy in Men Diagnosed with Prostate Cancer. Eur. Urol. 2015, 68, 756–765. [Google Scholar] [CrossRef] [PubMed]
- Lêng, C.H.; Chou, M.H.; Lin, S.-H.; Yang, Y.K.; Wang, J.-D. Estimation of life expectancy, loss-of-life expectancy, and lifetime healthcare expenditures for schizophrenia in Taiwan. Schizophr. Res. 2016, 171, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Zięba, M.; Tomczak, J.M.; Lubicz, M.; Świątek, J. Boosted SVM for extracting rules from imbalanced data in application to prediction of the post-operative life expectancy in the lung cancer patients. Appl. Soft Comput. 2014, 14, 99–108. [Google Scholar] [CrossRef]
- Pruksakorn, D.; Phanphaisarn, A.; Settakorn, J.; Arpornchayanon, U.; Tantraworasin, A.; Chaiyawat, P.; klangjorhor, J.; Teeyakasem, P. Prognostic score for life expectancy evaluation of lung cancer patients after bone metastasis. J. Bone Oncol. 2018, 10, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.; Pieri, A.; Cain, H. A systematic review of generic and breast cancer specific life expectancy models in the elderly. Eur. J. Surg. Oncol. 2017, 43, 1816–1827. [Google Scholar] [CrossRef] [PubMed]
- Ou, H.-T.; Yang, C.-Y.; Wang, J.-D.; Hwang, J.-S.; Wu, J.-S. Life Expectancy and Lifetime Health Care Expenditures for Type 1 Diabetes: A Nationwide Longitudinal Cohort of Incident Cases Followed for 14 Years. Value Health 2016, 19, 976–984. [Google Scholar] [CrossRef] [PubMed]
- Soga, Y.; Iida, O.; Takahaera, M.; Hirano, K.; Suzuki, K.; Kawasaki, D.; Miyashita, Y.; Tsuchiya, T. Two-Year Life Expectancy in Patients with Critical Limb Ischemia. JACC Cardiovasc. Interv. 2014, 7, 1444–1449. [Google Scholar] [CrossRef] [PubMed]
- Hastings, M.C.; Bursac, Z.; Julian, B.A.; Villa Baca, E.; Featherston, J.; Woodford, S.Y.; Bailey, L.; Wyatt, R.J. Life Expectancy for Patients From the Southeastern United States with IgA Nephropathy. Kidney Int. Rep. 2018, 3, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Yamada, S.-I.; Kurita, H.; Tomioka, T.; Ohta, R.; Yoshimura, N.; Nishimaki, F.; Koyama, Y.; Kondo, E.; Kamata, T. Healthy life expectancy of oral squamous cell carcinoma patients aged 75 years and older. Oral Oncol. 2017, 64, 22–26. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.-I.; Aas, E.; Howell, D.; Roman, E.; Patmore, R.; Jack, A.; Smith, A. Long-Term Medical Costs and Life Expectancy of Acute Myeloid Leukemia: A Probabilistic Decision Model. Value Health 2014, 17, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Hannerz, H.; Borgå, P.; Borritz, M. Life expectancies for individuals with psychiatric diagnoses. Public Health 2001, 115, 328–337. [Google Scholar] [CrossRef]
- Guzman-Castillo, M.; Ahmadi-Abhari, S.; Bandosz, P.; Capewell, S.; Steptoe, A.; Singh-Manoux, A.; Kivimaki, M.; Shipley, M.J.; Brunner, E.J.; O’Flaherty, M. Forecasted trends in disability and life expectancy in England and Wales up to 2025: A modelling study. Lancet Public Health 2017, 2, e307–e313. [Google Scholar] [CrossRef]
- Love-Koh, J.; Asaria, M.; Cookson, R.; Griffin, S. The Social Distribution of Health: Estimating Quality-Adjusted Life Expectancy in England. Value Health 2015, 18, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S. Active Life Expectancy of Elderly Koreans, 1994–2011. Asian Popul. Stud. 2015, 11, 312–327. [Google Scholar] [CrossRef]
- Hauck, K.; Martin, S.; Smith, P.C. Priorities for action on the social determinants of health: Empirical evidence on the strongest associations with life expectancy in 54 low-income countries, 1990–2012. Soc. Sci. Med. 2016, 167, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Kunze, L. Life expectancy and economic growth. J. Macroecon. 2014, 39, 54–65. [Google Scholar] [CrossRef]
- Bergh, A.; Nilsson, T. Good for Living? On the Relationship between Globalization and Life Expectancy. World Dev. 2010, 38, 1191–1203. [Google Scholar] [CrossRef]
- Sullivan, D.F. A single index of mortality and morbidity. HSMHA Health Rep. 1971, 86, 347. [Google Scholar] [CrossRef] [PubMed]
- Soomro, A.; Cavalcanti, D. Opportunities and challenges in using WPAN and WLAN technologies in medical environments [Accepted from Open Call]. IEEE Commun. Mag. 2007, 45, 114–122. [Google Scholar] [CrossRef]
- Barbiroli, M.; Carciofi, C.; Guiducci, D. Assessment of Population and Occupational Exposure to Wi-Fi Systems: Measurements and Simulations. IEEE Trans. Electromagn. Compat. 2011, 53, 219–228. [Google Scholar] [CrossRef]
- On the Minimum Health and Safety Requirements Regarding the Exposure of Workers to the Risks Arising from Physical Agents (Electromagnetic Fields) (20th Individual Directive within the Meaning of Article 16(1) of Directive 89/391/EEC) and Repealing Directive 2004/40/EC. Available online: https://osha.europa.eu/en/legislation/directives/directive-2013-35-eu-electromagnetic-fields (accessed on 10 August 2018).
- de Miguel-Bilbao, S.; Martín, M.A.; Del Pozo, A.; Febles, V.; Hernández, J.A.; de Aldecoa, J.C.F.; Ramos, V. Analysis of exposure to electromagnetic fields in a healthcare environment: Simulation and experimental study. Health Phys. 2013, 105, S209–S222. [Google Scholar] [CrossRef] [PubMed]
- Miguel-Bilbao, S.D.; Roldán, J.; García, J.; Ramos, V.; Fernández, J.; Suárez, Ó.J. Assessment of exposure from Wi-Fi devices. In Proceedings of the 2014 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Lisbon, Portugal, 11–12 June 2014; pp. 1–4. [Google Scholar]
- Aguirre Gallego, E.; Arpón Díaz-Aldagalán, J.; Azpilicueta Fernández de las Heras, L.; Ramos González, V.; Falcone Lanas, F.J. Evaluation of electromagnetic dosimetry of wireless systems in complex indoor scenarios with human body interaction. Prog. Electromagn. Res. B 2012, 43, 189–209. [Google Scholar] [CrossRef]
- Iturri, P.L.; Nazábal, J.A.; Azpilicueta, L.; Rodriguez, P.; Beruete, M.; Fernández-Valdivielso, C.; Falcone, F. Impact of high power interference sources in planning and deployment of wireless sensor networks and devices in the 2.4 GHz frequency band in heterogeneous environments. Sensors 2012, 12, 15689–15708. [Google Scholar] [CrossRef] [PubMed]
- Savola, R.M.; Abie, H. Metrics-driven security objective decomposition for an e-health application with adaptive security management. In Proceedings of the Proceedings of the International Workshop on Adaptive Security, Zurich, Switzerland, 8–12 September 2013; p. 6. [Google Scholar]
- Smith, E.; Eloff, J.H. Security in health-care information systems—Current trends. Int. J. Med. Inform. 1999, 54, 39–54. [Google Scholar] [CrossRef]
- Zhang, R.; Liu, L. Security Models and Requirements for Healthcare Application Clouds. In Proceedings of the 2010 IEEE 3rd International Conference on Cloud Computing, Miami, FL, USA, 5–10 July 2010; pp. 268–275. [Google Scholar]
- Martí, R.; Delgado, J.; Perramon, X. Network and application security in mobile e-health applications. In Proceedings of the International Conference on Information Networking, Busan, Korea, 18–20 February 2004; pp. 995–1004. [Google Scholar]
- Martí, R.; Delgado, J.; Perramon, X. Security specification and implementation for mobile e-health services. In Proceedings of the 2004 IEEE International Conference on e-Technology, e-Commerce and e-Service, Taipei, Taiwan, 28–31 March 2004; pp. 241–248. [Google Scholar]
- IEEE. Health Informatics–Personal Health Device Communication—Part 20601: Application Profile-Optimized Exchange Protocol; IEEE Std 11073-20601-2014 (Revision of ISO/IEEE 11073-20601:2010); IEEE: Piscataway, NJ, USA, 2014; pp. 1–253. [Google Scholar]
- Fieschi, M. Health Data Processing—Systemic Approaches, 1st ed.; ISTE: Washington, DC, USA, 2018. [Google Scholar]
- Al-Ali, Y.A.; Wafeeq, J.A. Systems and Methods for Monitoring a Patient Health Network. Available online: https://patents.justia.com/patent/20180225960 (accessed on 10 August 2018).
- Fries, J.F. Aging, natural death, and the compression of morbidity. Bull. World Health Organ. 2002, 80, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Woolf, S.H.; Aron, L.Y. The US health disadvantage relative to other high-income countries: Findings from a National Research Council/Institute of Medicine report. JAMA 2013, 309, 771–772. [Google Scholar] [CrossRef] [PubMed]
- Wearable. 50 Wearable Tech Predictions for 2018. Available online: https://www.wareable.com/wearable-tech/best-wearable-tech-2018-506 (accessed on 3 August 2018).
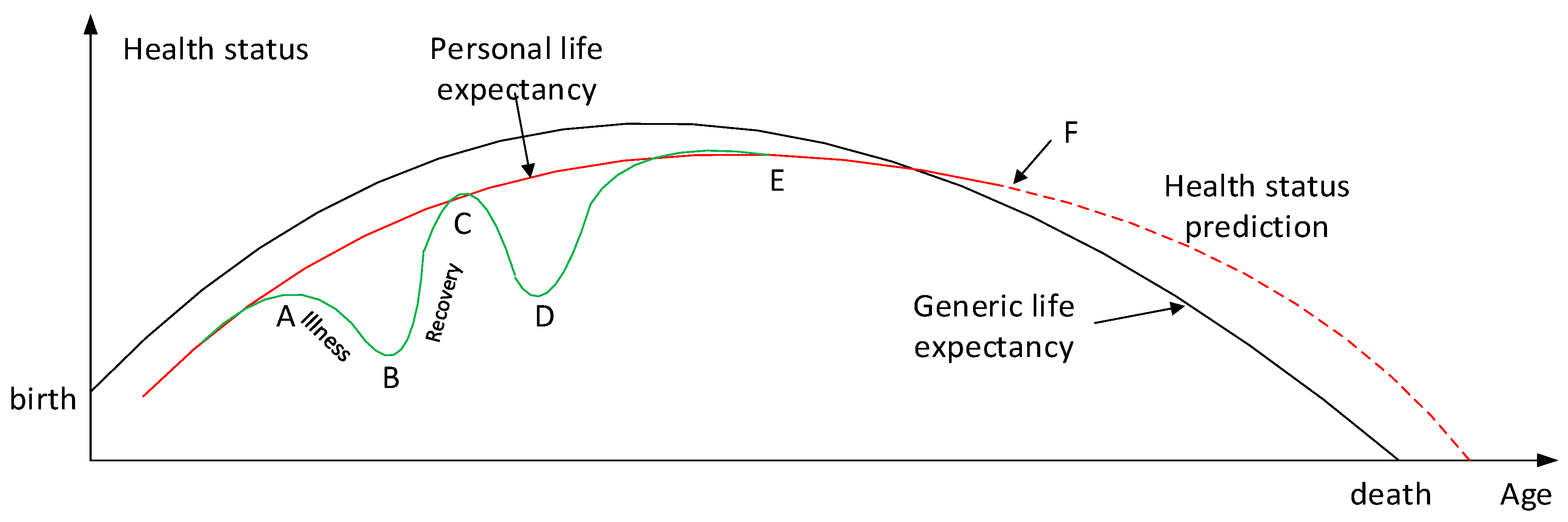
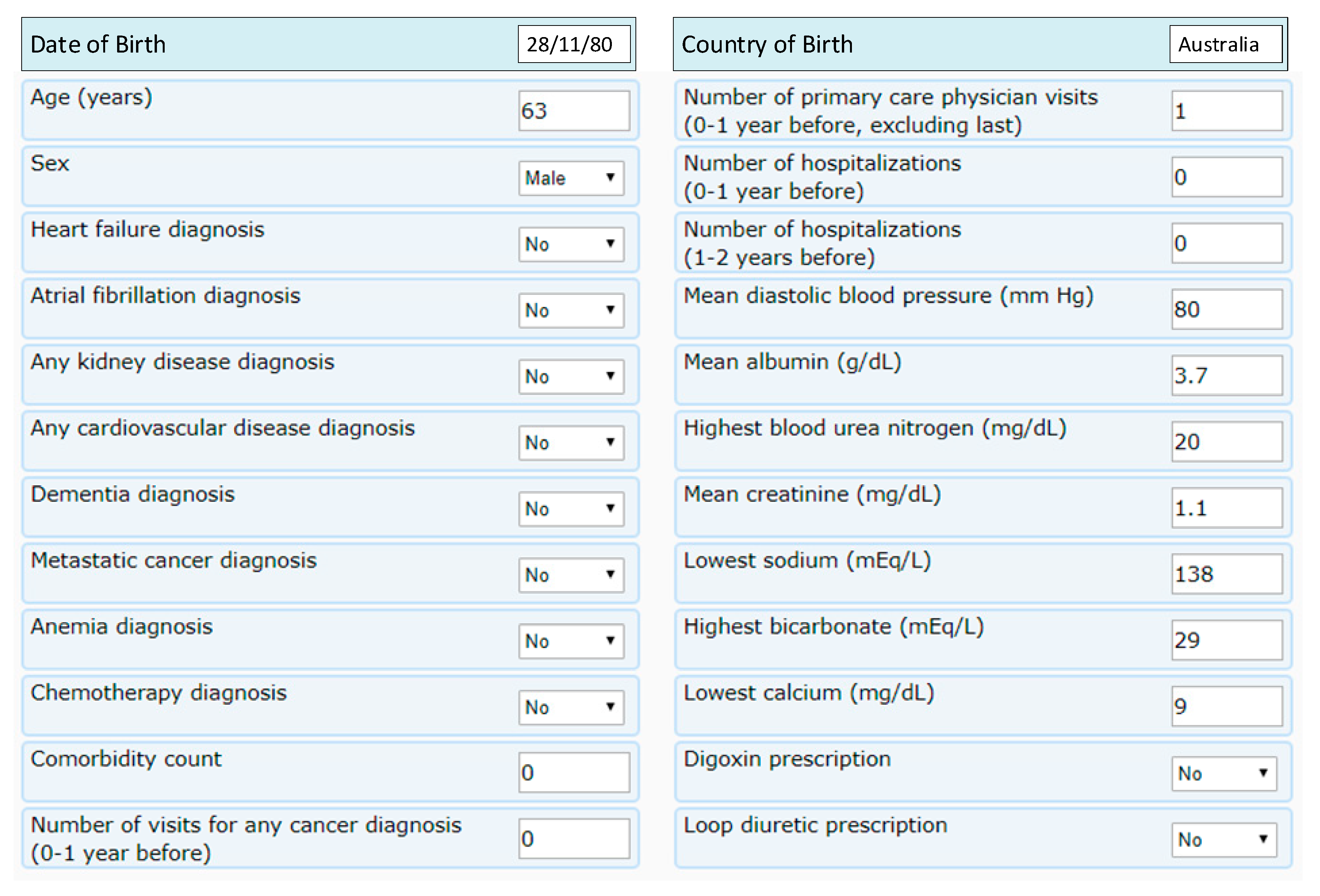
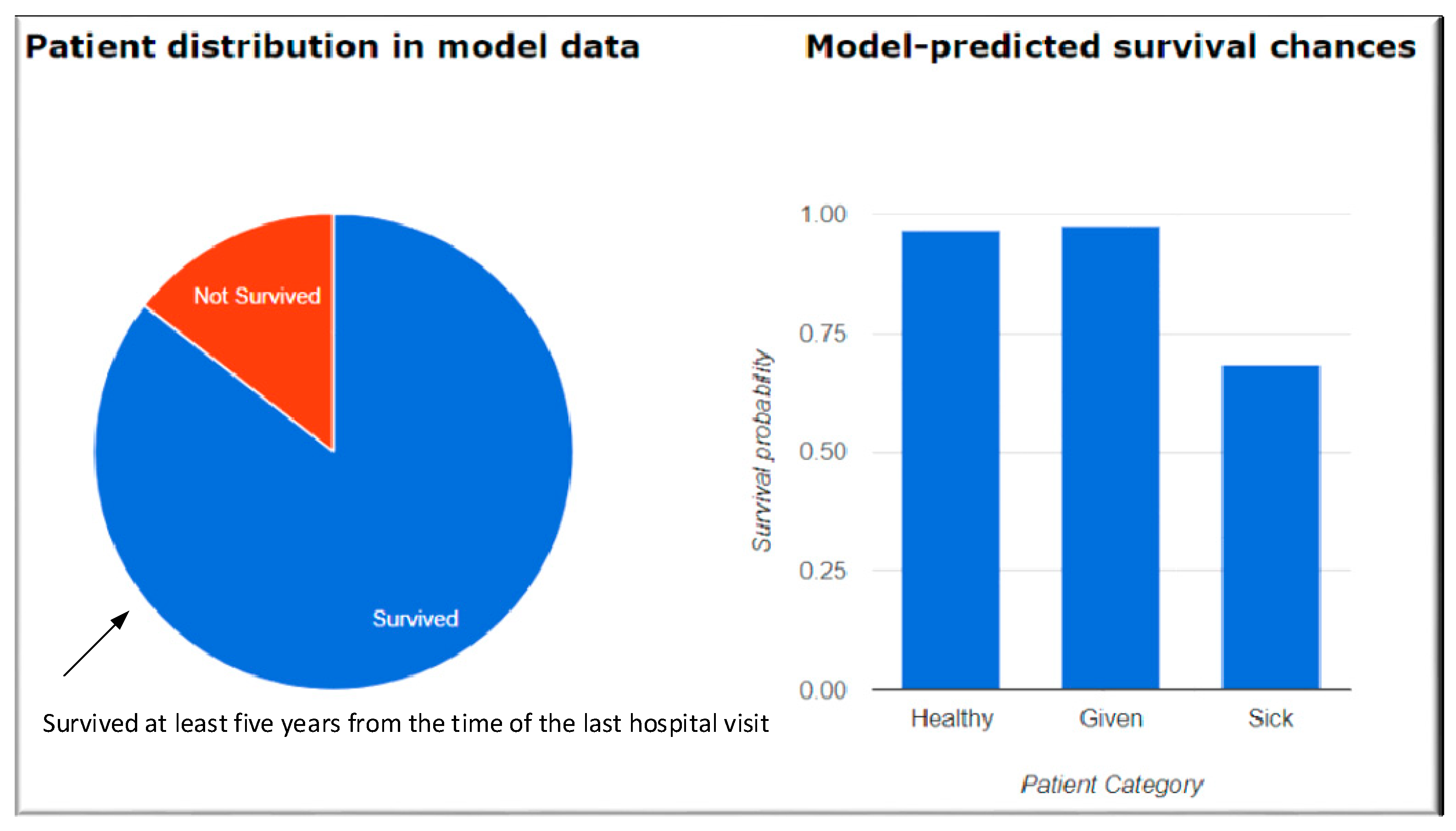
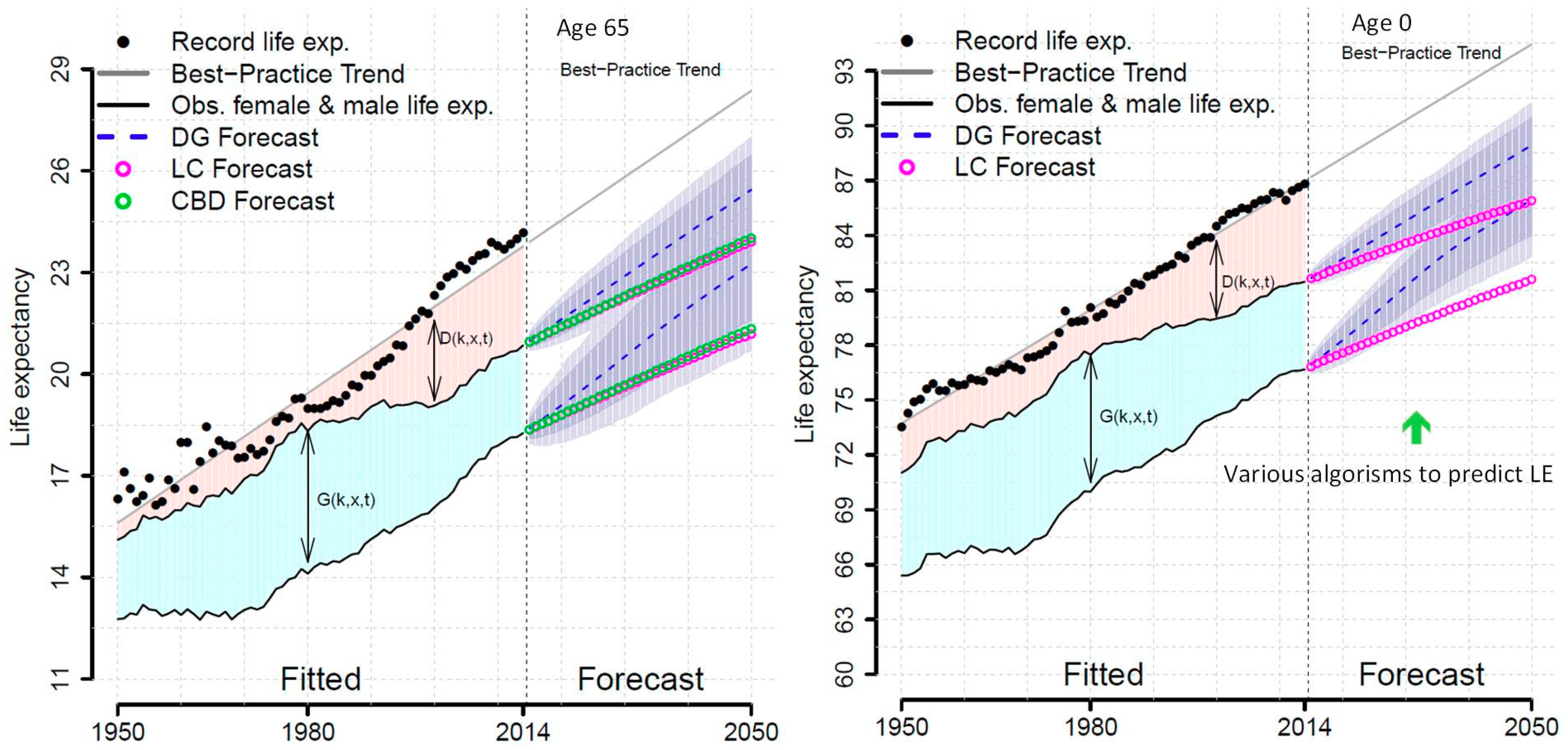
| Diseases Based | Geographic Areas | Relationships | Modelling Techniques |
|---|---|---|---|
| Cancer [24], schizophrenia [25], lung cancer [26,27], breast cancer [28], diabetes[29], limb ischemia [30], IgA nephropathy [31], oral squamous cell carcinoma [32], acute myeloid leukemia [33] and psychiatric disease [34] | America, Europe, and Asia [35,36,37] | Income related [38], relationship with economic growth [39] or globalization [40] | Sullivan [41], Lee and Carter [22], Cairns et al. [23], Jafelice [16], Agawal et al. [17] and Pascaliu et al. [21] |
| Modelling Techniques | Authors | Variables | Contribution |
|---|---|---|---|
| [41] | Sullivan | Skin color, sex, at birth and age of 65 | Used a life table model to calculate LE based with/without disability for mortality and morbidity. |
| [22] | Lee and Carter | Age groups, period-specific intensity index, direct time series methods | Used singular value decomposition model to account for almost all the variance over time in age-specific death rates as a group. |
| [23] | Cairns et al. | Mortality-rate dynamics at all ages to simulate the distribution of a survivor index over various time horizons | Proposed calculating the market risk-adjusted price of a longevity bond using two-factor model stochastic for the development of longevity through time. |
| [16] | Jafelice and Barros | Viral load and CD4+ level | Developed Fuzzy set-based model to compute the life expectancy of HIV infected populations to determine the average number of individuals and the life expectancy for specific population groups with no anti-retroviral therapy |
| [21] | Pascariu et al. | Male or Female at the age of 0 and 65 | LE forecasts based on the double-gap life expectancy forecasting model and compared with the Lee and Carter approach and the Cairns et al. strategy. The Double-Gap model approached the use of combination of models for the most promising forecasting tools. |
| [17] | Agrawal et al. | Electronic health records (EHR), Age, sex, health data including vitals | Used predictive models for five-year life expectancy of patients, built on electronic health records (EHR) of nearly 7500 patients aged 50 and above with more than 75 modeling configurations. They developed an online tool which takes a non-redundant subset of 24 patient attributes as the input and generates a patient-specific prediction of 5-year survival. |
| Classification | Attributes |
|---|---|
| Personal information | Gender, age race, country of birth, location |
| Body physique | Weight, height, Body Mass Index (BMI) |
| Life events | Education, marital status, retired status, residential type (urban/rural), living conditions (e.g., alone, with partner), pets, driving habits, relationship with family & friends |
| Work | Occupation, income, physical/office work, night shifts, full/part-time employment, working hours |
| Fitness | Exercise, general health, frequency, and amounts |
| Health conditions & diseases | Diabetes, high blood pressure, cholesterol levels, cardiovascular conditions, cancer, allergies |
| Diet | Vegan, vegetarian, meat intake, seafood, gluten, and other diet restrictions |
| Family history | Age of death of grandparents and parents, known family history of genetic conditions |
| Lifestyle | Alcohol consumption, smoking, hobbies, sleeping habits |
| Comorbidity count | Mean creatinine (mg/dL) |
| Number of visits for primary care, hospitalization in the past | Lowest sodium (mEq/L) |
| Mean diastolic blood pressure (mm Hg) | Lowest calcium (mg/dL) |
| Mean albumin (g/dL) | Digoxin prescription |
| Highest blood urea nitrogen (mg/dL) | Loop diuretic prescription |
| Sub-Regional Area (SRA)-Level Dataset (First Dataset) | Census-Tract Level Dataset (Second Dataset) |
|---|---|
| Health and human service agency (HHSA) Behavioral Health Data (Hospitalizations & Emergency Department visits for behavioral health conditions) | Census-tract level dataset |
| HHSA Demographics (Demographics) | American Community Survey 2012 (5-Year Estimates) (Census demographics) |
| Environmental Systems Research Institute (ESRI) Market Potential Data (Consumer buying patterns and behaviors) | CalEnviroScreen 2.0 (Pollution data) |
| San Diego Association of Governments (SANDAG) Healthy Communities Atlas (Data on physical and built environment) | Loop diuretic prescription |
| Average violent crime rate | Composite score: pedestrian traffic safety |
| Total park acreage | Percentage of block groups that interest with parks |
| Fast food density per square mile | Percentage of sidewalk coverage |
| Block groups with minority areas | Count of grocery stores locations |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, J.J.; Adibi, S. Systematic Predictive Analysis of Personalized Life Expectancy Using Smart Devices. Technologies 2018, 6, 74. https://doi.org/10.3390/technologies6030074
Kang JJ, Adibi S. Systematic Predictive Analysis of Personalized Life Expectancy Using Smart Devices. Technologies. 2018; 6(3):74. https://doi.org/10.3390/technologies6030074
Chicago/Turabian StyleKang, James Jin, and Sasan Adibi. 2018. "Systematic Predictive Analysis of Personalized Life Expectancy Using Smart Devices" Technologies 6, no. 3: 74. https://doi.org/10.3390/technologies6030074
APA StyleKang, J. J., & Adibi, S. (2018). Systematic Predictive Analysis of Personalized Life Expectancy Using Smart Devices. Technologies, 6(3), 74. https://doi.org/10.3390/technologies6030074






