Influence of Probiotics on the Development of Clostridioides difficile Infection in Patients Receiving Fluoroquinolones
Abstract
:1. Introduction
2. Materials and Methods
2.1. Setting and Study Design
2.2. Demographics and Patient Characteristics
2.3. Statistical Analysis
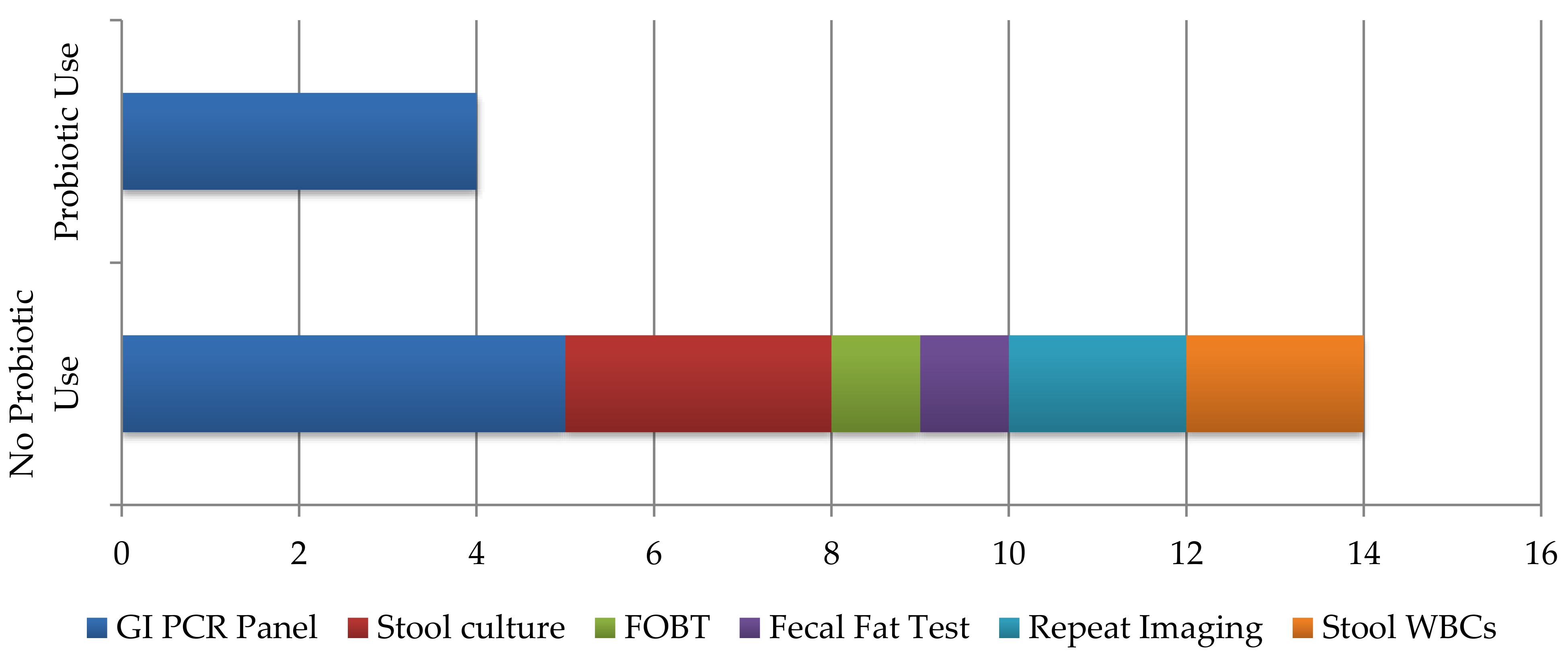
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- DePestel, D.D.; Aronoff, D.M. Epidemiology of clostridium difficile infection. J. Pharm Pract. 2013, 26, 464–475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Magill, S.S.; Edwards, J.R.; Bamberg, W.; Beldavs, Z.G.; Dumyati, G.; Kainer, M.A.; Lynfield, R.; Maloney, M.; McAllister-Hollod, L.; Nadle, J.; et al. Multistate point-prevalence survey of health care-associated infections. N. Engl. J. Med. 2014, 370, 1198–1208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tabak, Y.P.; Zilberberg, M.D.; Johannes, R.S.; Sun, X.; McDonald, L.C. Attributable burden of hospital-onset clostridium difficile infection: A propensity score matching study. Infect. Control. Hosp. Epidemiol. 2013, 34, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Pakyz, A.L.; Jawahar, R.; Wang, Q.; Harpe, S.E. Medication risk factors associated with healthcare-associated clostridium difficile infection: A multilevel model case-control study among 64 US academic medical centres. J. Antimicrob. Chemother. 2014, 69, 1127–1131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- The Pew Charitable Trusts. Health Experts Establish Targets to Improve Hospital Antibiotic Prescribing. Published 19 March 2021. Available online: https://www.pewtrusts.org/en/research-and-analysis/reports/2021/03/health-experts-establish-targets-to-improve-hospital-antibiotic-prescribing (accessed on 4 June 2021).
- Hudson, S.L.; Arnoczy, G.; Gibson, H. Probiotic use as prophylaxis for Clostridium difficile-associated diarrhea in a community hospital. Am. J. Infect. Control. 2019, 47, 1028–1029. [Google Scholar] [CrossRef] [PubMed]
- McFarland, L.V.; Ship, N.; Auclair, J.; Millette, M. Primary prevention of clostridium difficile infections with a specific probiotic combining lactobacillus acidophilus, l. casei, and l. rhamnosus strains: Assessing the evidence. J. Hosp. Infect. 2018, 99, 443–452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shen, N.T.; Maw, A.; Tmanova, L.L.; Pino, A.; Ancy, K.; Crawford, C.V.; Simon, M.S.; Evans, A.T. Timely use of probiotics in hospitalized adults prevents clostridium difficile infection: A systematic review with meta-regression analysis. Gastroenterology 2017, 152, 1889–1900. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, S.; Maziade, P.J.; McFarland, L.V.; Trick, W.; Donskey, C.; Currie, B.; Low, D.E.; Goldstein, E.J.C. Is primary prevention of clostridium difficile infection possible with specific probiotics? Int. J. Infect. Dis. 2012, 16, e786–e792. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goldenberg, J.Z.; Yap, C.; Lytvyn, L.; Lo, C.K.F.; Beardsley, J.; Mertz, D.; Johnston, B.C. Probiotics for the prevention of Clostridium difficile-associated diarrhea in adults and children. Cochrane Database Syst. Rev. 2017, 12, CD006095. [Google Scholar] [CrossRef] [PubMed]
- McDonald, L.C.; Gerding, D.N.; Johnson, S.; Bakken, J.S.; Carroll, K.C.; Coffin, S.E.; Dubberke, E.R.; Garey, K.W.; Gould, C.V.; Kelly, C.; et al. Clinical practice guidelines for Clostridium difficile infection in adults and children: 2017 update by the Infectious Diseases Society of America (IDSA) and Society for Healthcare Epidemiology of America (SHEA). Clin. Infect. Dis. 2018, 66, e1–e48. [Google Scholar] [CrossRef] [PubMed]
- Schooneveld, T.V.; Bergman, S.; Rupp, M. Clostridium difficile Infection (CDI) Management Guideline. Nebraska Medicine Antimicrobial Stewardship Program. 2021, pp. 1–6. Available online: https://www.unmc.edu/intmed/divisions/id/asp/docs/cdi-2021-final.pdf (accessed on 22 June 2021).
- Kazakova, S.V.; Baggs, J.; McDonald, L.C.; Yi, S.H.; Hatfield, K.M.; Guh, A.; Reddy, S.C.; Jernigan, J.A. Association between antibiotic use and hospital-onset clostridioides difficile infection in US acute care hospitals, 2006–2012: An ecologic analysis. Clin. Infect. Dis. 2020, 70, 11–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marra, A.R.; Perencevich, E.N.; Nelson, R.E.; Samore, M.; Khader, K.; Chiang, H.Y.; Chorazy, M.L.; Herwaldt, L.A.; Diekema, D.J.; Kuxhausen, M.F.; et al. Incidence and outcomes associated with clostridium difficile infections: A systematic review and meta-analysis. JAMA Netw. Open 2020, 3, e1917597. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schultz, K.; Sickbert-Bennett, E.; Marx, A.; Weber, D.J.; DiBiase, L.M.; Campbell-Bright, S.; Bode, L.E.; Baker, M.; Belhorn, T.; Buchanan, M.; et al. Preventable patient harm: A multidisciplinary, bundled approach to reducing Clostridium difficile infections while using a glutamate dehydrogenase/toxin immunochromatographic assay/nucleic acid amplification test diagnostic algorithm. J. Clin. Microbiol. 2018, 56, e00625. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gareau, M.G.; Sherman, P.M.; Walker, W.A. Probiotics and the gut microbiota in intestinal health and disease. Nat. Rev. Gastroenterol. Hepatol. 2010, 7, 503–514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaisawat, M.B.; MacPherson, C.W.; Tremblay, J.; Piano, A.; Iskandar, M.M.; Tompkins, T.A.; Kubow, S. Probiotic supplementation in a Clostridium difficile-infected gastrointestinal model is associated with restoring metabolic function of microbiota. Microorganisms 2019, 8, 60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allen, S.J.; Wareham, K.; Wang, D.; Bradley, C.; Hutchings, H.; Harris, W.; Dhar, A.; Brown, H.; Foden, A.; Gravenor, M.B.; et al. Lactobacilli and bifidobacteria in the prevention of antibiotic-associated diarrhoea and clostridium difficile diarrhoea in older inpatients (PLACIDE): A randomised, double-blind, placebo-controlled, multicentre trial. Lancet 2013, 382, 1249–1257. [Google Scholar] [CrossRef] [Green Version]
- Heil, E.L.; Harris, A.D.; Brown, C.H.; Seung, H.; Thom, K.A.; Rosenvinge, E.V.; Sorongon, S.; Pineles, L.; Goodman, K.E.; Leekha, S. A multi-center evaluation of probiotic use for the primary prevention of Clostridioides difficile infection. Clin. Infect. Dis. 2021, ciab417. [Google Scholar] [CrossRef] [PubMed]
| Characteristic † | Probiotic Use (n = 100) | No Probiotic Use (n = 100) | p-Value |
|---|---|---|---|
| Age, years | 68 (57–78) | 64 (55–74.75) | 0.120 |
| Male, no. (%) | 41 (41) | 35 (35) | 0.382 |
| Race, no. (%) | |||
| Caucasian | 72 (72) | 57 (57) | 0.027 |
| Black | 28 (28) | 41 (41) | 0.053 |
| Hispanic | 0 (0) | 1 (1) | 1.000 |
| American Indian/Alaskan Native | 0 (0) | 1 (1) | 1.000 |
| Charlson comorbidity index | 4 (2–5) | 4 (2–5) | 0.652 |
| Definitive monotherapy, no. (%) | 0.670 | ||
| Levofloxacin | 53 (53) | 56 (56) | |
| Ciprofloxacin | 47 (47) | 44 (44) | |
| Fluoroquinolone duration, days | 7 (5–10) | 7 (5–9) | 0.277 |
| PPI use, no. (%) | 41 (41) | 61 (61) | 0.005 |
| H2RA use, no. (%) | 26 (26) | 25 (25) | 0.871 |
| Prior antibiotic use, no. (%) | 51 (51) | 9 (9) | <0.001 |
| Probiotic use, no. (%) * | |||
| Lactobacillus | 76 (76) | - | |
| Saccharomyces | 22 (22) | - | |
| Probiotic frequency, no. (%) | |||
| Daily | 30 (30) | - | |
| BID | 24 (24) | - | |
| TID | 44 (44) | - | |
| Other | 2 (2) | - | |
| Duration of probiotics, days | 6 (4–9) | - | |
| Time from the start of FQ to first probiotic dose, days | 0 (0,1) | - |
| Symptoms | Probiotic Use (n = 100) | No Probiotic Use (n = 100) | p-Value |
|---|---|---|---|
| No Symptoms | 70 (70) | 65 (65) | 0.450 |
| Nausea | 11 (11) | 13 (13) | 0.663 |
| Vomiting | 2 (2) | 9 (9) | 0.030 |
| Bloating | 3 (3) | 4 (4) | 1.000 |
| Gas | 6 (6) | 10 (10) | 0.297 |
| Non-CDI diarrhea | 17 (17) | 20 (20) | 0.585 |
| Other | 0 (0) | 2 (2) † | 0.497 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sheffield, M.E.; Jones, B.M.; Terrell, B.; Wagner, J.L.; Bland, C.M. Influence of Probiotics on the Development of Clostridioides difficile Infection in Patients Receiving Fluoroquinolones. Pharmacy 2021, 9, 141. https://doi.org/10.3390/pharmacy9030141
Sheffield ME, Jones BM, Terrell B, Wagner JL, Bland CM. Influence of Probiotics on the Development of Clostridioides difficile Infection in Patients Receiving Fluoroquinolones. Pharmacy. 2021; 9(3):141. https://doi.org/10.3390/pharmacy9030141
Chicago/Turabian StyleSheffield, Mary E., Bruce M. Jones, Blake Terrell, Jamie L. Wagner, and Christopher M. Bland. 2021. "Influence of Probiotics on the Development of Clostridioides difficile Infection in Patients Receiving Fluoroquinolones" Pharmacy 9, no. 3: 141. https://doi.org/10.3390/pharmacy9030141
APA StyleSheffield, M. E., Jones, B. M., Terrell, B., Wagner, J. L., & Bland, C. M. (2021). Influence of Probiotics on the Development of Clostridioides difficile Infection in Patients Receiving Fluoroquinolones. Pharmacy, 9(3), 141. https://doi.org/10.3390/pharmacy9030141








