Medicines as Common Commodities or Powerful Potions? What Makes Medicines Reusable in People’s Eyes
Abstract
1. Introduction
2. Materials and Methods
3. Results
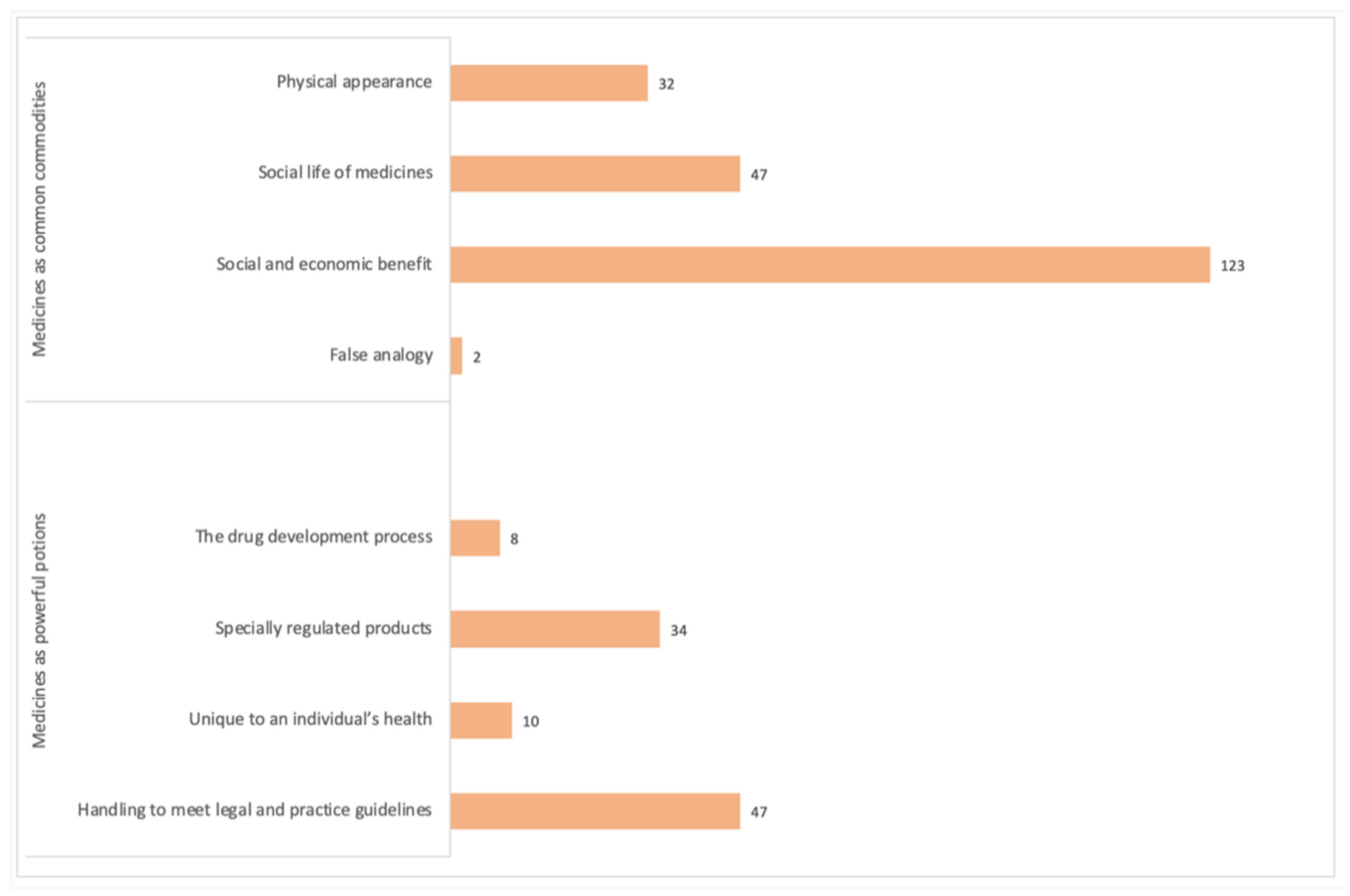
3.1. Medicines as Common Commodities
3.1.1. Physical Appearance
“When will the reuse of medication become legal? As long as it’s sealed, I would be happy.”(Participant 91).
“As long as medication is in sealed blister packs showing expiry date then it has to be a good thing.”(Participant 23).
“No reason at all not to re-use medication that is sealed and labelled.”(Participant 185).
3.1.2. Social Life of Medicines
“Please do it. I have had to return medication in the past just for it to be thrown away. It is wrong and wasteful when there is nothing wrong with it.”(Participant 112).
“I think it is a brilliant idea. I have returned medication to the pharmacy in the past and thought it wasteful to destroy.”(Participant 135).
“Having had to return medication from 2 people who died and had much surplus, it has always seemed to be to be such a waste.”(Participant 177).
3.1.3. Social and Economic Benefit
“I am entirely in favour of reusing medication. Far too much is wasted at great expense to the NHS and thus the taxpayer.”(Participant 10).
“Blisters go to landfill and cannot be recycled.”(Participant 40).
“I believe that unused, unopened pills should be reused, instead of being destroyed. Even given free to places where medications are too expensive for people who are living in poverty.”(Participant 191).
3.1.4. False Analogy
“(reuse) Applies to other things within NHS e.g., dressings, stoma products.”(Participant 21).
“As long as medication/dressing etc. has not been tampered with, use and not waste them.”(Participant 149).
3.2. Medicines as Powerful Potions
3.2.1. The Drug Development Process
“Need to see published trials.”(Participant 192).
“Only reuse quality medications not generics.”(Participant 88).
“Adhesive on morphine patches not of best quality.”(Participant 82).
3.2.2. Specially Regulated Products
“Conditions under which it may have been stored are unknown e.g., insulin in fridge.”(Participant 90).
“Many people will be afraid that re-using meds runs a risk of contamination.”(Participant 36).
“Even though the medication would appear to be sealed in its original packaging you don’t know how it has been stored, this could have an effect on it if stored in too hot or too cold temperatures.”(Participant 66).
“Proof of stability is a big concern.”(Participant 38).
3.2.3. Unique to an Individual’s Health
“I am type 1 diabetic and don’t feel that reusing medication is for any diabetic.”(Participant 203).
“I would reuse sealed medication only if my Dr said it was safe.”(Participant 5).
3.2.4. Handling to Meet Legal and Practice Guidelines
“I worry about fake medication.”(Participant 28).
“Providing everything has been checked out by professionals and have long use by date.”(Participant 96).
“There would need to be very strict guidelines in place to ensure patient safety.”(Participant 130).
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Department of Health [ARCHIVED CONTENT] Repeat Prescribing Systems: Department of Health—Publications. Available online: https://webarchive.nationalarchives.gov.uk/+/http://www.dh.gov.uk/en/Publicationsandstatistics/Publications/PublicationsPolicyAndGuidance/Browsable/DH_4892136 (accessed on 26 February 2021).
- House of Commons—Public Accounts—Minutes of Evidence Supplementary Memorandum from the Department of Health 2009. Available online: http://www.publications.parliament.uk/pa/cm200809/cmselect/cmpubacc/99/8121708 (accessed on 16 April 2021).
- Trueman, P.; Lowson, K.; Blighe, A.; Meszaros, A. Evaluation of the Scale, Causes and Costs of Waste Medicines Evaluation of the Scale, Causes and Costs of Waste Medicines; York Health Economics Consortium: York, UK, 2011; Volume 17. [Google Scholar]
- Council approves use of patient-returned and date-expired medicines in the event of pandemic flu. Pharm. J. 2008, 280, 645.
- Important Update—Extended Use of EpiPen® 300 mcg Adrenaline Auto-Injectors beyond Labelled Expiry Dates: PSNC Main Site. Available online: https://psnc.org.uk/our-news/important-update-extended-use-beyond-labelled-expiry-dates-for-select-lots-of-epipen-0-3mg-adrenaline-auto-injectors/ (accessed on 7 April 2021).
- Doyle, S. Canada lags behind United States in drug return, reuse and recycling programs. CMAJ. 2010, 182, E197–E198. [Google Scholar] [CrossRef] [PubMed]
- PRACTICE: Should Pharmacists Start Recycling Medicines? Available online: https://www.chemistanddruggist.co.uk/cpd-article/should-pharmacists-start-recycling-medicines (accessed on 10 April 2021).
- Neelam, S.; Vipula; Monica, K.; Mohini, K. Reusing Medicines-An Unexplored Concept in India. Indian J. Pharm. Pract. 2014, 7, 1–6. [Google Scholar]
- Nicoli, F.; Paudel, D.; Bresciani, G.; Rodi, D.; Siniscalchi, A. Donation programme of returned medicines: Role of donors and point of view of beneficiaries. Int. Health 2018, 10, 133–136. [Google Scholar] [CrossRef]
- Litchman, M.L.; Oser, T.K.; Wawrzynski, S.E.; Walker, H.R.; Oser, S. The Underground Exchange of Diabetes Medications and Supplies: Donating, Trading, and Borrowing, Oh My! J. Diabetes Sci. Technol. 2019, 14, 1000–1009. [Google Scholar] [CrossRef]
- Foroutan, B.; Foroutan, R. Household storage of medicines and self-medication practices in south-east Islamic Republic of Iran. East. Mediterr. Health J. 2014, 20, 547. [Google Scholar] [CrossRef]
- Patwary, M.A.; O’Hare, W.T.; Sarker, M.H. An illicit economy: Scavenging and recycling of medical waste. J. Environ. Manag. 2011, 92, 2900–2906. [Google Scholar] [CrossRef]
- GIVMED. Available online: https://givmed.org/en/ (accessed on 9 January 2020).
- Cauchi, R.; Berg, K. State Prescription Drug Return, Reuse and Recycling Laws. Available online: https://www.ncsl.org/research/health/state-prescription-drug-return-reuse-and-recycling.aspx (accessed on 9 January 2020).
- Nurolaini, K.; Sultana, S.; Wai See, W. Medication Wastage and its Disposal Amongst Patients at Suri Seri Begawan Hospital in Brunei Darussalam. Med. Health 2016, 11, 139–150. [Google Scholar] [CrossRef]
- Cuéllar, M.J.; Marco, J.L.; Pérez-Castelló, I.; Castelló Escrivá, A. Calidad en la conservación de los medicamentos termolábiles en el ámbito domiciliario. Rev. Calid. Asist. 2010, 25, 64–69. [Google Scholar] [CrossRef]
- Donyai, P.; McCrindle, R.; Sherratt, R.S.; Hui, T.K.L. COVID-19 Pandemic Is Our Chance to Learn How to Reuse Old Medicines. Available online: https://theconversation.com/covid-19-pandemic-is-our-chance-to-learn-how-to-reuse-old-medicines-137671 (accessed on 22 February 2020).
- Toh, M.R.; Chew, L. Turning waste medicines to cost savings: A pilot study on the feasibility of medication recycling as a solution to drug wastage. Palliat. Med. 2017, 31, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Kelly, F.; McMillan, S.; Spinks, J.; Bettington, E.; Wheeler, A.J. ‘You don’t throw these things out:’ an exploration of medicines retention and disposal practices in Australian homes. BMC Public Health 2018, 18, 1–12. [Google Scholar] [CrossRef]
- Alhamad, H.; Patel, N.; Donyai, P. How do people conceptualise the reuse of medicines? An interview study. Int. J. Pharm. Pract. 2018, 26, 232–241. [Google Scholar] [CrossRef]
- Bekker, C.L.; Gardarsdottir, H.; Egberts, T.C.G.; Bouvy, M.L.; van den Bemt, B.J.F. Redispensing of medicines unused by patients: A qualitative study among stakeholders. Int. J. Clin. Pharm. 2017, 39, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Alhamad, H.; Donyai, P. Intentions to “Reuse” Medication in the Future Modelled and Measured Using the Theory of Planned Behavior. Pharmacy 2020, 8, 213. [Google Scholar] [CrossRef]
- Braun, V.; Clarke, V. Using thematic analysis in psychology. Qual. Res. Psychol. 2006, 3, 77–101. [Google Scholar] [CrossRef]
- Hashimzade, N.; Myles, G.; Black, J. Commodity. In Oxford Reference; Oxford University Press: Oxford, UK, 2017; ISBN 9780191819940. [Google Scholar]
- van der Geest, S.; Hardon, A. Social and cultural efficacies of medicines: Complications for antiretroviral therapy. J. Ethnobiol. Ethnomed. 2006, 2, 48. [Google Scholar] [CrossRef][Green Version]
- De Boeck, F. On van der Geest and Whyte’s Article ‘The Charm of Medicines: Metaphors and Metonyms’. Med. Anthropol. Q. 1991, 5, 170–172. [Google Scholar] [CrossRef]
- Prosser, H. Marvelous medicines and dangerous drugs: The representation of prescription medicine in the UK newsprint media. Public Underst. Sci. 2010, 19, 52–69. [Google Scholar] [CrossRef]
- Gill, D.; Almutairi, S.; Donyai, P. “The Lesser of Two Evils” Versus “Medicines not Smarties”: Constructing Antipsychotics in Dementia. Gerontologist 2019, 59, 570–579. [Google Scholar] [CrossRef] [PubMed]
- Mackridge, A.J.; Marriott, J.F. Returned medicines: Waste or a wasted opportunity? J. Public Health 2007, 29, 258–262. [Google Scholar] [CrossRef] [PubMed]
- Bekker, C.L.; Gardarsdottir, H.; Egberts, A.C.G.; Molenaar, H.A.; Bouvy, M.L.; van den Bemt, B.J.F.; Hövels, A.M. What does it cost to redispense unused medications in the pharmacy? A micro-costing study. BMC Health Serv. Res. 2019, 19, 243. [Google Scholar] [CrossRef] [PubMed]
- Van Der Geest, S.; Whyte, S.R. The Charm of Medicines: Metaphors and Metonyms. Med Anthr. Q. 1989, 3, 345–367. [Google Scholar] [CrossRef]
- Hui, T.K.L.; Donyai, P.; McCrindle, R.; Sherratt, R.S. Enabling Medicine Reuse Using a Digital Time Temperature Humidity Sensor in an Internet of Pharmaceutical Things Concept. Sensors 2020, 20, 3080. [Google Scholar] [CrossRef]
- Hui, T.K.L.; Mohammed, B.; Donyai, P.; McCrindle, R.; Sherratt, R.S. Enhancing Pharmaceutical Packaging through a Technology Ecosystem to Facilitate the Reuse of Medicines and Reduce Medicinal Waste. Pharmacy 2020, 8, 58. [Google Scholar] [CrossRef] [PubMed]
| Medicines as Common Commodities | Medicines as Powerful Potions |
|---|---|
| This category encapsulates commonly held ideas about what makes medicines the same as any other commodity and therefore suitable for reuse. | This category describes what confers medicines their potency and special status distinct from ordinary commodities, thus cautioning against reuse. |
| Physical appearance | The drug development process |
| The external features and overall physical appearance of a commodity are adequate to indicate what is held within. Therefore, intact sealed packaging of medicines suggests an authentic product of good quality inside. | Drug discovery and development processes are time consuming, expensive and intricate. Numerous stages ensure stable and effective final formulations, making medicines complex compared to other commodities. |
| Social life of medicines | Specially regulated products |
| Medicines have metaphorical life stages, with a medicine’s death (when consumed) resulting in its afterlife (internal effects) to restore, improve or maintain health. Failure to reuse unused medication therefore makes its existence meaningless. | Medicines are strictly regulated by authorities to illustrate quality, safety and efficacy before and after authorization. This includes giving expiry dates and storage conditions to maintain the shelf life. |
| Social and economic benefit | Unique to an individual’s health |
| Here, medicines are standardized commercial goods with economic value, exchanged between manufacturers and consumers to meet their needs. Reusing medicines thus brings benefit by reducing medicines spending and waste. | Medicines are prescribed for specific individuals with the unique therapeutic effects dependent on the individual’s circumstances. Medicines must not be reshared as their outcome in others cannot be guaranteed. |
| False analogy | Handling to meet legal and practice guidelines |
| This fallacy assumes that if two things are alike in one aspect, then they will be similar in another aspect too. Thus, if devices and appliances used to diagnose and treat health conditions can be reused, then so can medicines. | The sale or exchange of medicines (over the counter or via prescription) must adhere to legal protocols and accuracy and clinical checks. As powerful substances, their casual handling could cause harm to patients. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chauhan, M.; Alhamad, H.; McCrindle, R.; Hui, T.K.L.; Sherratt, R.S.; Donyai, P. Medicines as Common Commodities or Powerful Potions? What Makes Medicines Reusable in People’s Eyes. Pharmacy 2021, 9, 88. https://doi.org/10.3390/pharmacy9020088
Chauhan M, Alhamad H, McCrindle R, Hui TKL, Sherratt RS, Donyai P. Medicines as Common Commodities or Powerful Potions? What Makes Medicines Reusable in People’s Eyes. Pharmacy. 2021; 9(2):88. https://doi.org/10.3390/pharmacy9020088
Chicago/Turabian StyleChauhan, Monica, Hamza Alhamad, Rachel McCrindle, Terence K. L. Hui, R. Simon Sherratt, and Parastou Donyai. 2021. "Medicines as Common Commodities or Powerful Potions? What Makes Medicines Reusable in People’s Eyes" Pharmacy 9, no. 2: 88. https://doi.org/10.3390/pharmacy9020088
APA StyleChauhan, M., Alhamad, H., McCrindle, R., Hui, T. K. L., Sherratt, R. S., & Donyai, P. (2021). Medicines as Common Commodities or Powerful Potions? What Makes Medicines Reusable in People’s Eyes. Pharmacy, 9(2), 88. https://doi.org/10.3390/pharmacy9020088








