The Use of a Decision Support System in Swedish Pharmacies to Identify Potential Drug-Related Problems—Effects of a National Intervention Focused on Reviewing Elderly Patients’ Prescriptions
Abstract
1. Introduction
- examine the impact of the national intervention on the weekly number of EES analyses;
- describe the proportion of patients aged 75 years or older getting an analysis with EES; the number of alerts for potential DRPs; and the proportion being resolved before, during, and after the intervention;
- describe what types of alerts for potential DRPs that were generated, which types were resolved, and what kinds of actions that were taken to resolve them during the week of the intervention; and
- describe pharmacists’ perceptions and experience with EES and the national intervention.
2. Materials and Methods
2.1. Setting
2.1.1. Electronic Expert Support System (EES)
2.1.2. National Intervention
2.2. Data and Statistics on The Use of EES
2.3. Questionnaire among Pharmacists
2.4. Data Analysis
2.5. Ethics Statement
3. Results
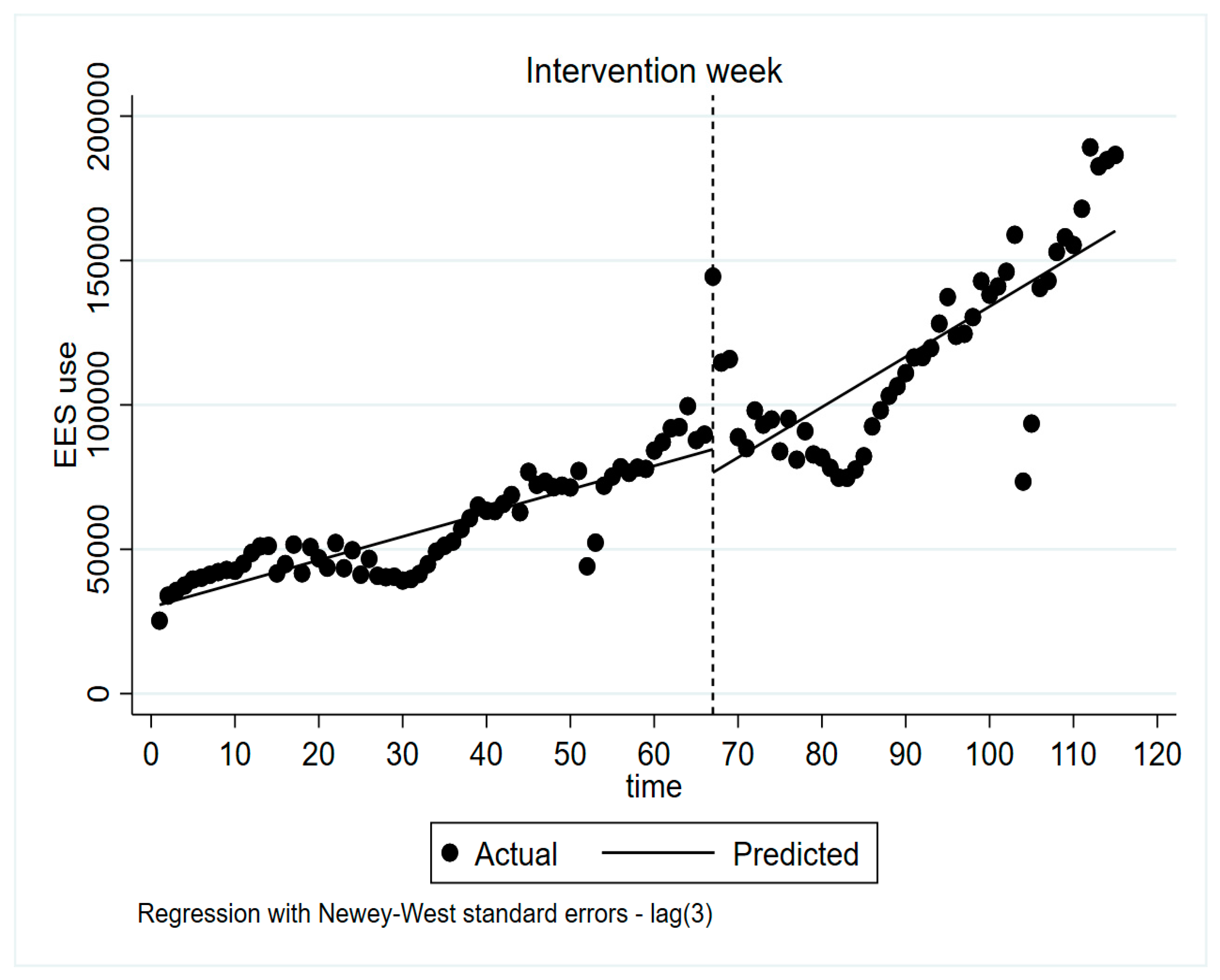
3.1. Effects of The National Intervention on The Use of EES
3.2. EES Analyses and The Numbers of Alerts Generated and Resolved before and after The Intervention
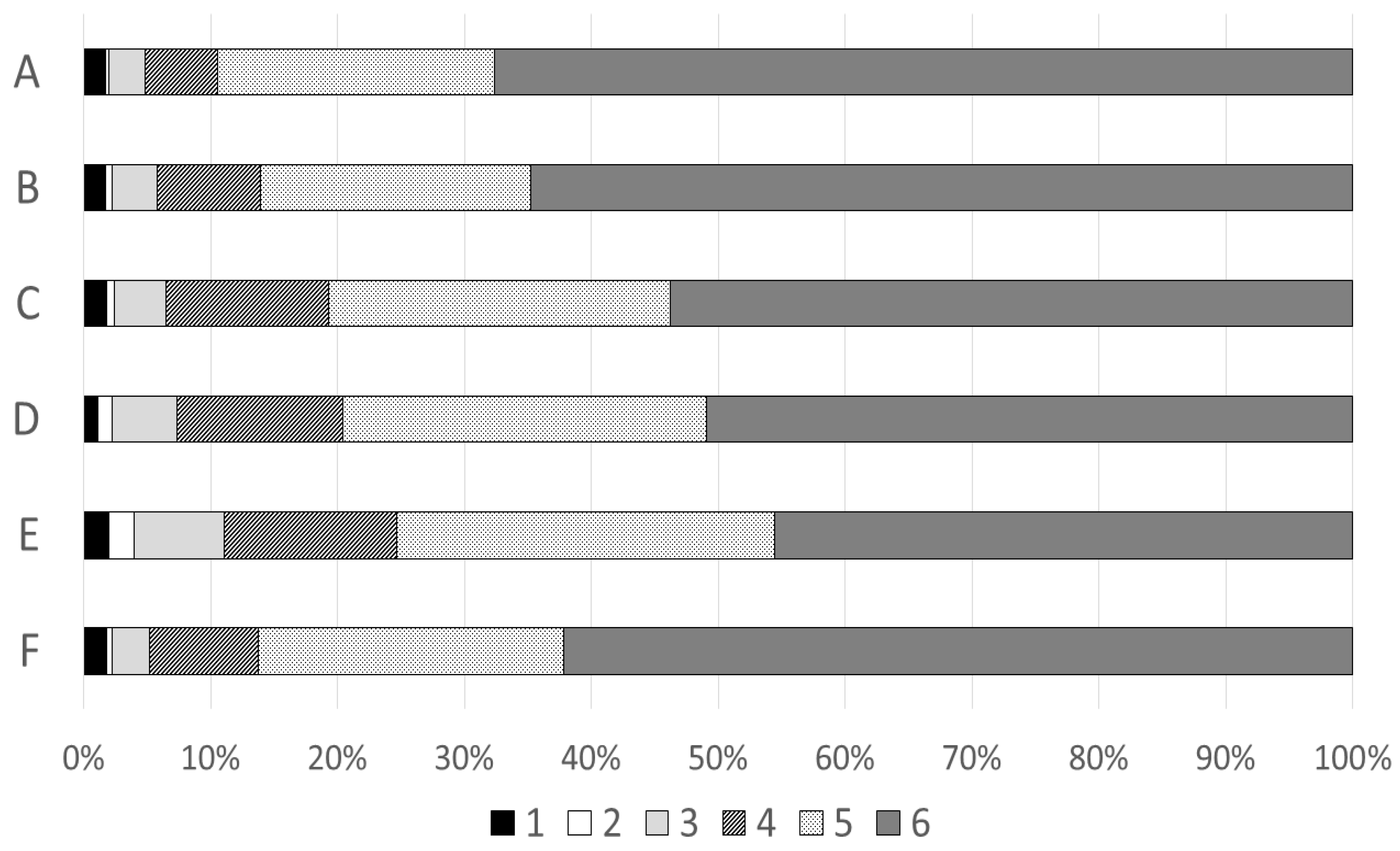
3.3. Pharmacists’ Perceptions of EES and The Intervention
4. Discussion
4.1. Effects of The National Intervention
4.2. Alerts Being Resolved
4.3. Pharmacists’ Perceptions and Experiences with EES
4.4. Using CDSSs to Improve Medication Safety
4.5. Method Discussion (Strengths and Weaknesses)
4.6. Future Research
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- James, M.; Ritter, L.D.L.; Timothy, G.K.; Mant, A.F. A Textbook of Clinical Pharmacology and Therapeutics; Hodden Education: London, UK, 2008. [Google Scholar]
- Bemt, P.M.V.D.; Egberts, T.; Berg, L.T.D.J.-V.D.; Brouwers, J.R.; Bemt, P.M.V.D. Drug-related problems in hospitalised patients. Drug Saf. 2000, 22, 321–333. [Google Scholar] [CrossRef] [PubMed]
- Avery, A.; Sheikh, A.; Hurwitz, B.; Smeaton, L.; Chen, Y.-F.; Howard, R.; Cantrill, J.; Royal, S. Safer medicines management in primary care. Br. J. Gen. Pract. 2002, 52, S17–S22. [Google Scholar] [PubMed]
- Salvi, F.; Marchetti, A.; D’Angelo, F.; Boemi, M.; Lattanzio, F.; Cherubini, A. Adverse drug events as a cause of hospitalization in older adults. Drug Saf. 2012, 35, 29–45. [Google Scholar] [CrossRef]
- Topinková, E.; Baeyens, J.P.; Lang, P.-O.; Topinková, P.E.; Michel, J.P. Evidence-based strategies for the optimization of pharmacotherapy in older people. Drugs Aging 2012, 29, 477–494. [Google Scholar] [CrossRef]
- Spinewine, A.; Schmader, K.E.; Barber, N.; Hughes, C.; Lapane, K.L.; Swine, C.; Hanlon, J.T. Appropriate prescribing in elderly people: How well can it be measured and optimised? Lancet 2007, 370, 173–184. [Google Scholar] [CrossRef]
- McKibbon, K.A.; Lokker, C.; Handler, S.; Dolovich, L.; Holbrook, A.M.; O’Reilly, D.; Tamblyn, R.; Hemens, B.J.; Basu, R.; Troyan, S.; et al. The effectiveness of integrated health information technologies across the phases of medication management: A systematic review of randomized controlled trials. J. Am. Med. Inform. Assoc. 2011, 19, 22–30. [Google Scholar] [CrossRef]
- Eiermann, B.; Bastholm, R.P.; Korkmaz, S.; Landberg, C.; Lilja, B.; Shemeikka, T.; Veg, A.; Wettermark, B.; Gustafsson, L.L. Knowledge bases for clinical decision support in drug prescribing—Development, quality assurance, management, integration, implementation and evaluation of clinical value. In Decision Support Systems; Jao, C.S., Ed.; InTech: Rijeka, Croatia, 2010. [Google Scholar]
- Coleman, J.J.; van der Sijs, H.; Haefeli, W.E.; Slight, S.P.; McDowell, S.E.; Seidling, H.M.; Eiermann, B.; Aarts, J.; Ammenwerth, E.; Ferner, R.E.; et al. On the alert: Future priorities for alerts in clinical decision support for computerized physician order entry identified from a European workshop. BMC Med. Inform. Decis. Mak. 2013, 13, 1–8. [Google Scholar]
- Garg, A.X.; Adhikari, N.K.J.; McDonald, H.; Rosas-Arellano, M.P.; Devereaux, P.J.; Beyene, J.; Sam, J.; Haynes, R.B. Effects of computerized clinical decision support systems on practitioner performance and patient outcomes. JAMA 2005, 293, 1223–1238. [Google Scholar] [CrossRef]
- Black, A.D.; Car, J.; Pagliari, C.; Anandan, C.; Cresswell, K.; Bokun, T.; McKinstry, B.; Procter, R.; Majeed, A.; Sheikh, A. The impact of ehealth on the quality and safety of health care: A systematic overview. PLoS Med. 2011, 8, e1000387. [Google Scholar] [CrossRef]
- Kuperman, G.; Bobb, A.; Payne, T.H.; Avery, A.; Gandhi, T.K.; Burns, G.; Classen, D.C.; Bates, D.W. Medication-related clinical decision support in computerized provider order entry systems: A review. J. Am. Med. Inform. Assoc. 2007, 14, 29–40. [Google Scholar] [CrossRef]
- Robertson, J.; Walkom, E.; Pearson, S.-A.; Hains, I.; Williamsone, M.; Newby, D.A. The impact of pharmacy computerised clinical decision support on prescribing, clinical and patient outcomes: A systematic review of the literature. Int. J. Pharm. Pract. 2010, 18, 69–87. [Google Scholar]
- Coiera, E.; Westbrook, J.; Wyatt, J. The safety and quality of decision support systems. Yearb. Med. Inform. 2006, 15, 20–25. [Google Scholar]
- Hemens, B.J.; Holbrook, A.; Tonkin, M.; Mackay, J.A.; Weise-Kelly, L.; Navarro, T.; Wilczynski, N.L.; Haynes, R.B. Computerized clinical decision support systems for drug prescribing and management: A decision-maker-researcher partnership systematic review. Implement. Sci. 2011, 6, 89. [Google Scholar] [CrossRef] [PubMed]
- Jaspers, M.; Smeulers, M.; Vermeulen, H.; Dusseljee-Peute, L.W. Effects of clinical decision-support systems on practitioner performance and patient outcomes: A synthesis of high-quality systematic review findings. J. Am. Med. Inform. Assoc. 2011, 18, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Mulder-Wildemors, L.G.M.; Heringa, M.; Floor-Schreudering, A.; Jansen, P.A.F.; Bouvy, M.L. Reducing inappropriate drug use in older patients by use of clinical decision support in community pharmacy: A mixed-methods evaluation. Drugs Aging 2019, 37, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Mandt, I.; Horn, A.M.; Ekedahl, A.; Granas, A.G. Community pharmacists’ prescription intervention practices—Exploring variations in practice in Norwegian pharmacies. Res. Soc. Adm. Pharm. 2010, 6, 6–17. [Google Scholar] [CrossRef]
- Teinilä, T.; Kaunisvesi, K.; Airaksinen, M. Primary care physicians’ perceptions of medication errors and error prevention in cooperation with community pharmacists. Res. Soc. Adm. Pharm. 2011, 7, 162–179. [Google Scholar] [CrossRef]
- Seidling, H.M.; Send, A.F.J.; Bittmann, J.; Renner, K.; Dewald, B.; Lange, D.; Bruckner, T.; Haefeli, W.E. Medication review in German community pharmacies—Post-hoc analysis of documented drug-related problems and subsequent interventions in the ATHINA-project. Res. Soc. Adm. Pharm. 2017, 13, 1127–1134. [Google Scholar] [CrossRef]
- Saverno, K.R.; Hines, L.E.; Warholak, T.L.; Grizzle, A.J.; Babits, L.; Clark, C.; Taylor, A.M.; Malone, D.C. Ability of pharmacy clinical decision-support software to alert users about clinically important drug-drug interactions. J. Am. Med. Inform. Assoc. 2011, 18, 32–37. [Google Scholar] [CrossRef]
- Campmans, Z.; Van Rhijn, A.; Dull, R.M.; Santen-Reestman, J.; Taxis, K.; Borgsteede, S.D. Preventing dispensing errors by alerting for drug confusions in the pharmacy information system—A survey of users. PLoS ONE 2018, 13, e0197469. [Google Scholar] [CrossRef]
- Scott, I.; Pillans, P.I.; Barras, M.; Morris, C. Using EMR-enabled computerized decision support systems to reduce prescribing of potentially inappropriate medications: A narrative review. Ther. Adv. Drug Saf. 2018, 9, 559–573. [Google Scholar] [CrossRef] [PubMed]
- Galt, K.A.; Fuji, K.T.; Kaufman, T.K.; Shah, S.R. Health information technology use and patient safety: Study of pharmacists in Nebraska. Pharmacy 2019, 7, 7. [Google Scholar] [CrossRef] [PubMed]
- Hammar, T.; Hovstadius, B.; Lidström, B.; Petersson, G.; Eiermann, B. Potential drug related problems detected by electronic expert support system in patients with multi-dose drug dispensing. Int. J. Clin. Pharm. 2014, 36, 943–952. [Google Scholar]
- Hammar, T.; Lidström, B.; Petersson, G.; Gustafson, Y.; Eiermann, B. Potential drug-related problems detected by electronic expert support system: Physicians’ views on clinical relevance. Int. J. Clin. Pharm. 2015, 37, 941–948. [Google Scholar] [CrossRef] [PubMed]
- Hammar, T.; Ohlson, M.; Hanson, E.; Petersson, G. Implementation of information systems at pharmacies—A case study from the re-regulated pharmacy market in Sweden. Res. Soc. Adm. Pharm. 2015, 11, e85–e99. [Google Scholar] [CrossRef]
- Watkins, K.; Wood, H.; Schneider, C.R.; Clifford, R. Effectiveness of implementation strategies for clinical guidelines to community pharmacy: A systematic review. Implement. Sci. 2015, 10, 151. [Google Scholar] [CrossRef]
- Chauhan, B.F.; Jeyaraman, M.M.; Mann, A.S.; Lys, J.; Skidmore, B.; Sibley, K.M.; Abou-Setta, A.M.; Zarychanski, R.; Zarychanksi, R. Behavior change interventions and policies influencing primary healthcare professionals’ practice—An overview of reviews. Implement. Sci. 2017, 12, 3. [Google Scholar] [CrossRef]
- Seidling, H.M.; Schmitt, S.P.W.; Bruckner, T.; Kaltschmidt, J.; Pruszydlo, M.G.; Senger, C.; Bertsche, T.; Walter-Sack, I.; Haefeli, W.E. Patient-specific electronic decision support reduces prescription of excessive doses. BMJ Qual. Saf. 2010, 19, e15. [Google Scholar] [CrossRef][Green Version]
- Scott, G.P.T.; Shah, P.; Wyatt, J.C.; Makubate, B.; Cross, F.W. Making electronic prescribing alerts more effective: Scenario-based experimental study in junior doctors. J. Am. Med. Inform. Assoc. 2011, 18, 789–798. [Google Scholar] [CrossRef]
- Mollon, B.; Chong, J.J.; Holbrook, A.; Sung, M.; Thabane, L.; Foster, G. Features predicting the success of computerized decision support for prescribing: A systematic review of randomized controlled trials. BMC Med. Inform. Decis. Mak. 2009, 9, 11. [Google Scholar] [CrossRef]
- Horsky, J.; Phansalkar, S.; Desai, A.; Bell, U.; Middleton, B. Design of decision support interventions for medication prescribing. Int. J. Med. Inform. 2013, 82, 492–503. [Google Scholar] [CrossRef] [PubMed]
- Riedmann, D.; Jung, M.; Hackl, W.; Ammenwerth, E. How to improve the delivery of medication alerts within computerized physician order entry systems: An international Delphi study. J. Am. Med. Inform. Assoc. 2011, 18, 760–766. [Google Scholar] [CrossRef][Green Version]
- Ranji, S.R.; Rennke, S.; Wachter, R.M. Computerised provider order entry combined with clinical decision support systems to improve medication safety: A narrative review. BMJ Qual. Saf. 2014, 23, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Jani, Y.H.; Barber, N.; Wong, I.C. Characteristics of clinical decision support alert overrides in an electronic prescribing system at a tertiary care paediatric hospital. Int. J. Pharm. Pract. 2011, 19, 363–366. [Google Scholar] [CrossRef]
- Nanji, K.C.; Slight, S.P.; Seger, D.L.; Cho, I.; Fiskio, J.M.; Redden, L.M.; Volk, L.A.; Bates, D.W. Overrides of medication-related clinical decision support alerts in outpatients. J. Am. Med. Inform. Assoc. 2014, 21, 487–491. [Google Scholar] [CrossRef]
- Van Der Sijs, H.; Aarts, J.; Vulto, A.; Berg, M. Overriding of drug safety alerts in computerized physician order entry. J. Am. Med. Inform. Assoc. 2006, 13, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Van Der Sijs, H.; Mulder, A.; Van Gelder, T.; Aarts, J.; Berg, M.; Vulto, A. Drug safety alert generation and overriding in a large Dutch university medical centre. Pharmacoepidemiol. Drug Saf. 2009, 18, 941–947. [Google Scholar] [CrossRef]
- Payne, T.H.; Hines, L.E.; Chan, R.C.; Hartman, S.; Kapusnik-Uner, J.; Russ, A.L.; Chaffee, B.W.; Hartman, C.; Tamis, V.; Galbreth, B.; et al. Recommendations to improve the usability of drug-drug interaction clinical decision support alerts. J. Am. Med. Inform. Assoc. 2015, 22, 1243–1250. [Google Scholar] [CrossRef]
- Shorten, A.; Smith, J. Mixed methods research: Expanding the evidence base. Évid. Based Nurs. 2017, 20, 74–75. [Google Scholar] [CrossRef]
- Böttiger, Y.; Laine, K.; Andersson, M.L.; Korhonen, T.; Molin, B.; Ovesjö, M.-L.; Tirkkonen, T.; Rane, A.; Gustafsson, L.L.; Eiermann, B.; et al. SFINX—A drug-drug interaction database designed for clinical decision support systems. Eur. J. Clin. Pharmacol. 2009, 65, 627–633. [Google Scholar] [CrossRef]
- Kontopantelis, E.; Doran, T.; A Springate, D.A.; Buchan, I.; Reeves, D. Regression based quasi-experimental approach when randomisation is not an option: Interrupted time series analysis. BMJ 2015, 350, h2750. [Google Scholar] [CrossRef] [PubMed]
- Bernal, J.L.; Cummins, S.; Gasparrini, A. Interrupted time series regression for the evaluation of public health interventions: A tutorial. Int. J. Epidemiol. 2017, 46, 348–355. [Google Scholar]
- Linden, A. Conducting interrupted time-series analysis for single- and multiple-group comparisons. Stata J. 2015, 15, 480–500. [Google Scholar] [CrossRef]
- Sandelowski, M. Whatever happened to qualitative description? Res. Nurs. Health 2000, 23, 334–340. [Google Scholar] [CrossRef]
- Patwardhan, P.D.; Amin, M.E.K.; Chewning, B. Intervention research to enhance community pharmacists’ cognitive services: A systematic review. Res. Soc. Adm. Pharm. 2014, 10, 475–493. [Google Scholar] [CrossRef] [PubMed]
- Cresswell, K.; Bates, D.W.; Sheikh, A. Ten key considerations for the successful implementation and adoption of large-scale health information technology. J. Am. Med. Inform. Assoc. 2013, 20, e9–e13. [Google Scholar] [CrossRef] [PubMed]
- Cresswell, K.; Sheikh, A. Organizational issues in the implementation and adoption of health information technology innovations: An interpretative review. Int. J. Med. Inform. 2013, 82, e73–e86. [Google Scholar] [CrossRef] [PubMed]
- Swedish eHealth Agency. Nationella läkemedelslistan [In Swedish]. Available online: https://www.ehalsomyndigheten.se/tjanster/yrkesverksam/nationella-lakemedelslistan/ (accessed on 7 July 2020).
- Hammar, T.; Ekedahl, A.; Petersson, G. Implementation of a shared medication list: Physicians’ views on availability, accuracy and confidentiality. Int. J. Clin. Pharm. 2014, 36, 933–942. [Google Scholar] [CrossRef]
- Manskow, U.S.; Lind, K.F.; Bergmo, T.S. Digital solutions for a shared medication list—A narrative literature review. In Proceedings of the 17th Scandinavian Conference on Health Informatics, Oslo, Norway, 12–13 November 2019. [Google Scholar]
- Pontefract, S.K.; Coleman, J.J.; Vallance, H.K.; Hirsch, C.A.; Shah, S.; Marriott, J.F.; Redwood, S. The impact of computerised physician order entry and clinical decision support on pharmacist-physician communication in the hospital setting: A qualitative study. PLoS ONE 2018, 13, e0207450. [Google Scholar] [CrossRef]
- Krähenbühl, J.-M.; Kremer, B.; Guignard, B.; Bugnon, O. Practical evaluation of the drug-related problem management process in Swiss community pharmacies. Pharm. World Sci. 2008, 30, 777–786. [Google Scholar] [CrossRef]
- Hermansyah, A.; Sainsbury, E.; Krass, I. The operation of a Research and Development (R&D) program and its significance for practice change in community pharmacy. PLoS ONE 2017, 12, e0184954. [Google Scholar]
| Data | Description | Time Period |
|---|---|---|
| Number of EES analyses | Statistics from eHealth Agency Number of times EES is used, active choice to press the EES button. (only calculated once/unique individual/pharmacy/day). | Total population Per year 2014–2017 Per week from week 1 of 2017 to week 11 of year 2019. |
| Number of EES analyses | Statistics from eHealth Agency Number of times EES is used, active choice to press the EES button. (only calculated once/unique individual/pharmacy/day). | 75 years or older Per week (week 11,15, 21, 36 of 2018 and week 11 of 2019) |
| Individuals having prescriptions dispensed | Statistics from eHealth Agency Number of individuals having prescriptions dispensed (only calculated once /unique individual/pharmacy/day). | Per week (week 11, 15, 21, 36 of 2018 and week 11 of 2019) |
| Proportion of individuals getting an EES analysis | Calculation Number of EES analyses/Individuals having prescriptions dispensed (%). | Per week (week 11, 15, 21, 36 of 2018 and week 11 of 2019) |
| Number of EES alerts | Statistics from eHealth Agency Total number of alerts from EES. Each time a pharmacist utilises EES, EES analyses the patient’s prescriptions in the prescription repository and may generate a number of alerts. | Per week (week 11, 15, 21, 36 of 2018 and week 11 of 2019) |
| Average number of alerts per EES analysis | Calculation Number of EES alerts/number of EES analysis. How many alerts are generated on average each time EES is used. | Per week (week 11, 15, 21, 36 of 2018 and week 11 of 2019) |
| Closed (resolved) EES alerts | Statistics from eHealth Agency In EES the pharmacist can close an alert after it has been resolved. When an alert is closed the pharmacist can provide the reason for closing the alert from a number of alternatives in the system. | Per week (week 11, 15, 21, 36 of 2018 and week 11 of 2019) |
| Proportion of alerts being closed | Calculation Closed alerts/number of alerts (%). | Per week (week 11, 15, 21, 36 of 2018 and week 11 of 2019) |
| Type of alert generated, resolved, and documented action | Statistics from eHealth Agency According to eHealth Agencies alert categories and reasons for closing alert available in the system. | Week 15 of 2018 |
| Active pharmacies | Statistics from eHealth Agency Number of pharmacies actively using EES, e.g., at least one EES analysis during the week. | Per week (week 11, 15, 21, 36 of 2018 and week 11 of 2019) |
| Question | Description | |
|---|---|---|
| 1 | Year of birth | YYYY |
| 2 | Gender | Multiple choice: |
| 3 | Education | Multiple choice |
| 4 | Years as a pharmacist at a community pharmacy | Multiple choice |
| 5 | What pharmacy chain do you work at? | Multiple choice |
| 6 | How do you characterise the vicinity of the pharmacy where you most often work? | Multiple choice |
| 7 | How do you characterise the city or region where the pharmacy where you most often work is located? | Multiple choice |
| 8 | How often have you on average used EES during the past year? | Multiple choice |
| 9 | Five questions about the intervention (here called focus week) Have you received information about the focus week (before this questionnaire)? Has your work been affected by the focus week? Have you used EES more actively in general during the focus week? Have you used EES more actively for patients aged 75 years or older during the focus week? Have you used EES less than usual for patients younger than 75 years during the focus week? | Multiple choice: yes/no/do not know Before the questions, the questionnaire had one sentence about the focus week for clarification |
| 10 | Have you gone through any education regarding EES? (Web-based education from eHealth Agency/Education with a supervisor from eHealth Agency/Meetings or days of education at your pharmacy chain/ Reading material on your own/Other education/ No education/ Do not know) | Multiple choice Several answers possible. Possible to answer in free text |
| 11 | Six statements about the pharmacist’s perceptions about using EES | Degree of agreement on a six-point Likert scale (1 = do not agree at all, 6 = totally agree) |
| 12 | Which EES alert category do you perceive provides you with the best support in pharmacological control (for pharmacy customers all ages) | Multiple choice. Several answers possible. |
| 13 | Have you ever handled or taken actions related to an EES alert, for example, having a dialogue with the customer or contacting the prescriber, without closing the alert? | Multiple choice |
| 14 | If you answered yes to 13, what was the reason? | Multiple choice. Several answers possible. Possible to answer in free text. |
| 15 | For the times you do not use EES for pharmacy customers aged 75 years or older, what is the reason? | Multiple choice. Several answers possible. Possible to answer in free text. |
| 16 | What would you need to use EES more often? | Multiple choice. Several answers possible. Possible to answer in free text. |
| 17 | Do you perceive any needs for improvement or development in EES? Describe these. | Free text |
| 18 | What other decision support or sources of information do you use regularly (at least once a month) in your work to improve use of medication? | Multiple choice. Several answers possible. Possible to answer in free text. |
| 19 | What would facilitate or make it easier for you in your work with improving the use of medications for pharmacy customers aged 75 years or older? | Free text |
| 20 | Do you have any other comments about EES, the focus week, or the questionnaire? | Free text |
| Week 11 2018 | Week 15 2018 | Week 21 2018 | Week 36 2018 | Week 11 2019 | ||
|---|---|---|---|---|---|---|
| 75 years or older | Number of EES analyses | 27,829 | 55,180 | 28,348 | 31,635 | 51,953 |
| Individuals having prescriptions dispensed | 253,938 | 257,783 | 258,449 | 253,678 | 261,990 | |
| Proportion of individuals getting an EES analysis | 11% | 21% | 11% | 12% | 20% | |
| Number of EES alerts | 139,648 | 329,138 | 137,708 | 207,583 | 339,289 | |
| Average number of alerts per EES analysis | 5.0 | 6.0 | 4.9 | 6.6 | 6.5 | |
| Number of closed EES alerts | 4601 | 21,952 | 3579 | 3662 | 6327 | |
| Proportion of alerts being closed (%) | 3.3% | 6.7% | 2.6% | 1.8% | 1.9% | |
| Total population | Number of EES analyses | 92,312 | 144,423 | 93,175 | 103,153 | 186,557 |
| Proportion of individuals getting an EES analysis | 10% | 15% | 10% | 11% | 19% | |
| Number of EES alerts | 587,323 | 837,572 | 585,236 | 630,909 | 1,104,063 | |
| Average number of alerts per EES analysis | 6.7 | 5.8 | 6.3 | 6.1 | 5.9 | |
| Number of closed EES alerts | 14,906 | 46,634 | 12,606 | 11,715 | 23,620 | |
| Proportion of alerts being closed (%) | 2.5% | 5.6% | 2.2% | 1.9% | 2.1% | |
| Number of active pharmacies | 1245 | 1358 | 1301 | 1342 | 1390 |
| Alert Categories | Number of Alerts | Proportion of All Alerts (%) | Number of Closed Alerts | Proportion being Closed (%) |
|---|---|---|---|---|
| B interaction | 103,722 | 31.5 | 1828 | 1.8 |
| C interaction | 97,710 | 29.7 | 9040 | 9.3 |
| Therapy duplication | 54,512 | 16.5 | 5843 | 10.7 |
| High dose for elderly | 19,716 | 6.0 | 1948 | 9.9 |
| A interaction | 17,231 | 5.2 | 37 | 0.21 |
| Supplementary rules | 8226 | 2.5 | 428 | 5.2 |
| D interaction | 7140 | 2.2 | 1107 | 15.5 |
| Drug-disease inferred | 6714 | 2.0 | 464 | 7.0 |
| High dose | 2680 | 0.8 | 187 | 7.0 |
| Drug gender warning | 74 | 0.0 | 6 | 8.1 |
| Total | 329,138 | 21,952 | 6.7 |
| Reason for Closing Alert | n | % |
|---|---|---|
| Dialogue with patient—verification of treatment | 18,055 | 82.2 |
| Only pharmaceutical assessment | 1989 | 9.1 |
| Dialogue with patient—referring to prescriber | 869 | 4.0 |
| Dialogue with patient—maculation of prescription | 255 | 1.2 |
| Contact with prescriber—without change of prescription | 143 | 0.7 |
| Contact with prescriber—change of prescription | 44 | 0.2 |
| Other action/measure | 597 | 2.7 |
| Total | 21,952 | 100 |
| Background Characteristics | n | % | |
|---|---|---|---|
| Gender | Female | 398 | 87.1 |
| Male | 55 | 12.0 | |
| Other | 4 | 0.9 | |
| Year of birth | 1959 or before | 66 | 14.4 |
| 1960–1969 | 100 | 21.9 | |
| 1970–1979 | 125 | 27.4 | |
| 1980–1989 | 125 | 27.4 | |
| 1990 or later | 41 | 9.0 | |
| Education * | Pharmacist (5–year education) | 157 | 34.4 |
| Pharmacist (or prescriptionist, 3–year education) | 297 | 65.0 | |
| Other | 3 | 0.7 | |
| Years as pharmacist at a community pharmacy | < 5 years | 99 | 21.7 |
| 5–10 years | 123 | 26.9 | |
| 11–20 years | 127 | 27.8 | |
| > 20 years | 108 | 23.6 | |
| Pharmacy chain | Apotea | 10 | 2.2 |
| Apotek Hjärtat | 178 | 38.9 | |
| Apoteket AB | 124 | 27.1 | |
| Apoteksgruppen | 61 | 13.3 | |
| Kronans Apotek | 68 | 14.9 | |
| Lloyds Apotek | 11 | 2.4 | |
| Other pharmacy company | 5 | 1.1 | |
| Pharmacy area/vicinity | Pharmacy in connection with hospital | 39 | 8.5 |
| Pharmacy in connection with primary healthcare centre | 107 | 23.4 | |
| Pharmacy in connection with commercial streets/major shopping mall/supermarket | 230 | 50.3 | |
| Pharmacy in connection with junction/public transport | 36 | 7.9 | |
| None of the options above | 45 | 9.8 | |
| City or region where the pharmacy is located | Larger city (more than 100,000 inhabitants) | 141 | 30.9 |
| Small town (10,000–100,000 inhabitants) | 229 | 50.1 | |
| Village/sparsely populated region (< 10,000 inhabitants) | 80 | 17.5 | |
| None of the options above | 7 | 1.5 | |
| Question | Answer (Multiple Choice) | n | % |
|---|---|---|---|
| How often have you used EES during the past year? | Daily | 269 | 58.9 |
| Several times a week | 132 | 28.9 | |
| Several times a month | 32 | 7.0 | |
| A few times | 20 | 4.4 | |
| Never | 2 | 0.4 | |
| Do not know | 2 | 0.4 | |
| Have you had any education regarding EES? | Yes | 446 | 97.6 |
| No | 11 | 2.4 | |
| Which alert category gives you the best support in the pharmaceutical control? ** | Drug—drug interactions | 421 | 92.1 |
| Elderly warnings | 258 | 56.5 | |
| Paediatric warnings | 253 | 55.4 | |
| High dose | 237 | 51.9 | |
| Drug—disease inferred | 213 | 46.6 | |
| Therapy duplication | 197 | 43.1 | |
| Drug gender warning | 45 | 9.8 | |
| Do not know | 5 | 1.1 | |
| What are the reasons for not using EES to analyse prescriptions for patients 75 years or older? ** | Difficult with relatives or caregivers collecting the medication | 335 | 73.3 |
| The requirement for consent is an obstacle | 209 | 45.7 | |
| I feel that it takes too much time | 144 | 31.5 | |
| I see no need | 133 | 29.1 | |
| I do not have enough experience with the system | 19 | 4.2 | |
| I lack sufficient knowledge | 10 | 2.2 | |
| I think EES is difficult to use | 6 | 1.3 | |
| Other reason * | 72 | 15.8 | |
| Do not know | 9 | 2.0 | |
| What do you need to use EES more? ** | More time for the patient | 326 | 71.3 |
| More knowledge among patients about EES | 146 | 31.9 | |
| More experience | 141 | 30.9 | |
| More user-friendly system | 74 | 16.2 | |
| Changed working procedures at the pharmacy | 61 | 13.3 | |
| Other functionality in EES | 43 | 9.4 | |
| More education | 43 | 9.4 | |
| Different leadership/management at the pharmacy | 16 | 3.5 | |
| Nothing, do not want to use | 2 | 0.4 | |
| Other * | 37 | 8.1 | |
| Do not know | 19 | 4.2 | |
| Have you ever resolved a potential problem from an EES alert without closing it? | Yes, many times | 172 | 37.9 |
| Yes, a few times | 215 | 47.4 | |
| No, never | 48 | 10.6 | |
| Do not know | 19 | 4,2 | |
| If you have (resolved without closing), what is the reason for not closing the alert? | Lack of time | 270 | 59.1 |
| Want other pharmacists to see the alert | 187 | 40.9 | |
| Not sure I selected/chose the right action | 70 | 15.3 | |
| Unclear when to close alerts | 54 | 11.8 | |
| Have not seen the meaning | 16 | 3.5 | |
| Other reason * | 51 | 11.2 | |
| Do not know | 21 | 4.6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hammar, T.; Hellström, L.; Ericson, L. The Use of a Decision Support System in Swedish Pharmacies to Identify Potential Drug-Related Problems—Effects of a National Intervention Focused on Reviewing Elderly Patients’ Prescriptions. Pharmacy 2020, 8, 118. https://doi.org/10.3390/pharmacy8030118
Hammar T, Hellström L, Ericson L. The Use of a Decision Support System in Swedish Pharmacies to Identify Potential Drug-Related Problems—Effects of a National Intervention Focused on Reviewing Elderly Patients’ Prescriptions. Pharmacy. 2020; 8(3):118. https://doi.org/10.3390/pharmacy8030118
Chicago/Turabian StyleHammar, Tora, Lina Hellström, and Lisa Ericson. 2020. "The Use of a Decision Support System in Swedish Pharmacies to Identify Potential Drug-Related Problems—Effects of a National Intervention Focused on Reviewing Elderly Patients’ Prescriptions" Pharmacy 8, no. 3: 118. https://doi.org/10.3390/pharmacy8030118
APA StyleHammar, T., Hellström, L., & Ericson, L. (2020). The Use of a Decision Support System in Swedish Pharmacies to Identify Potential Drug-Related Problems—Effects of a National Intervention Focused on Reviewing Elderly Patients’ Prescriptions. Pharmacy, 8(3), 118. https://doi.org/10.3390/pharmacy8030118





