Systematic Medication Review in General Practice by an Interdisciplinary Team: A thorough but Laborious Method to Address Polypharmacy among Elderly Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. General Practice in Denmark
2.2. The Setting
2.3. Inclusion of Patients
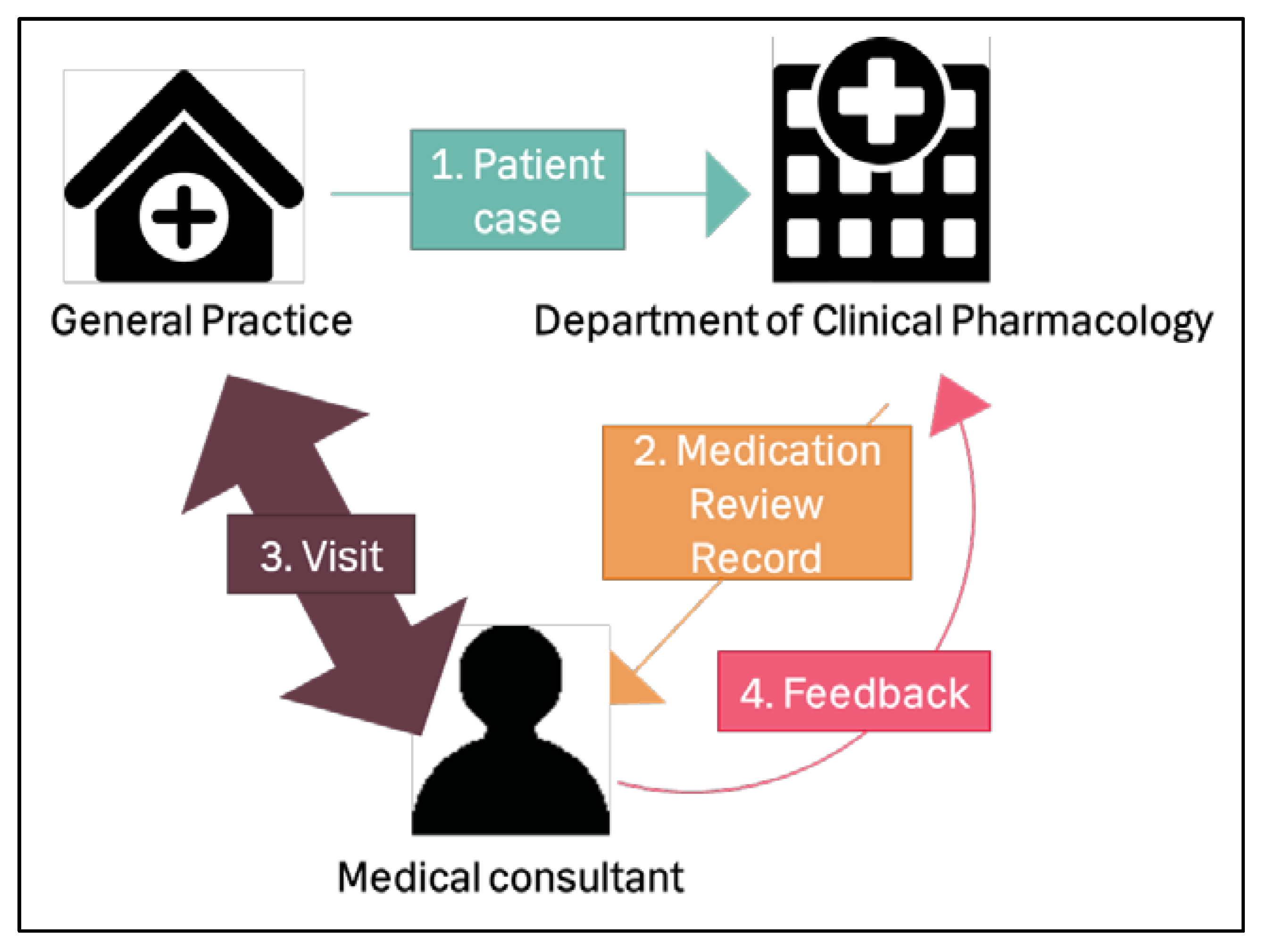
2.4. Design
- Each GP sent one patient case (consisting of the patient’s medical and health information) to the medication team at the Department of Clinical Pharmacology, Copenhagen University Hospital Bispebjerg (Figure 1). The medication team consisted of a group of pharmacists and physicians. Each medication review was conducted by one pharmacist and one physician from the medication team. An interdisciplinary medication team was chosen because we considered different healthcare professionals may find different medication-related problems [4]; this would improve the quality of the medication review. They registered the recommended changes for the medication in a medication review record.
- This record was sent electronically to the medical consultant from KAP-H.
- The medical consultant then visited the GP in person, and they discussed the medication review.
- The medical consultant reported feedback electronically to the medication team about which of the recommended changes the GP accepted.
2.5. Patient Case
2.6. Medication Review
2.7. Feedback
2.8. Data Analysis/Statistical Method
2.9. Ethics
3. Results
3.1. Patients
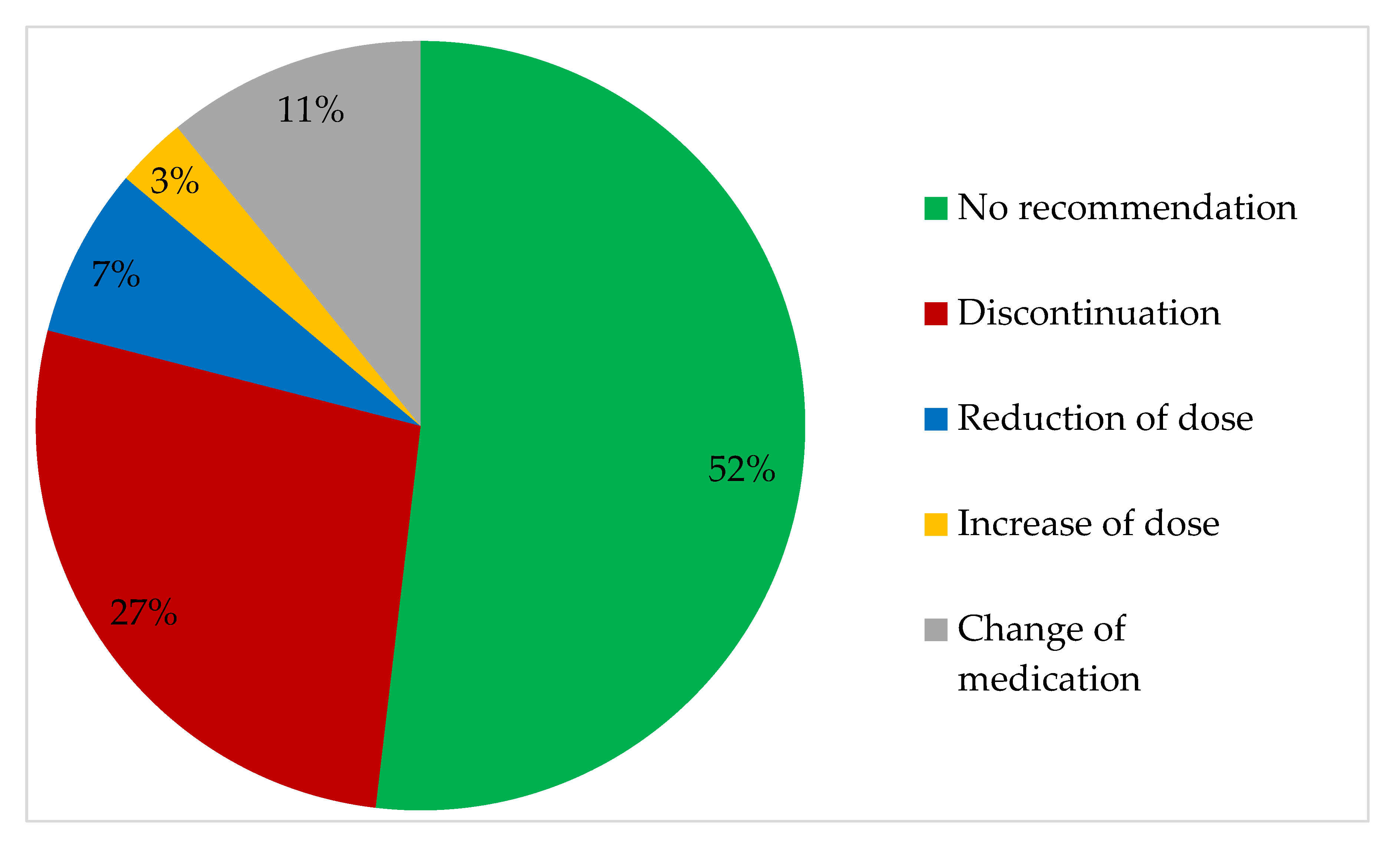
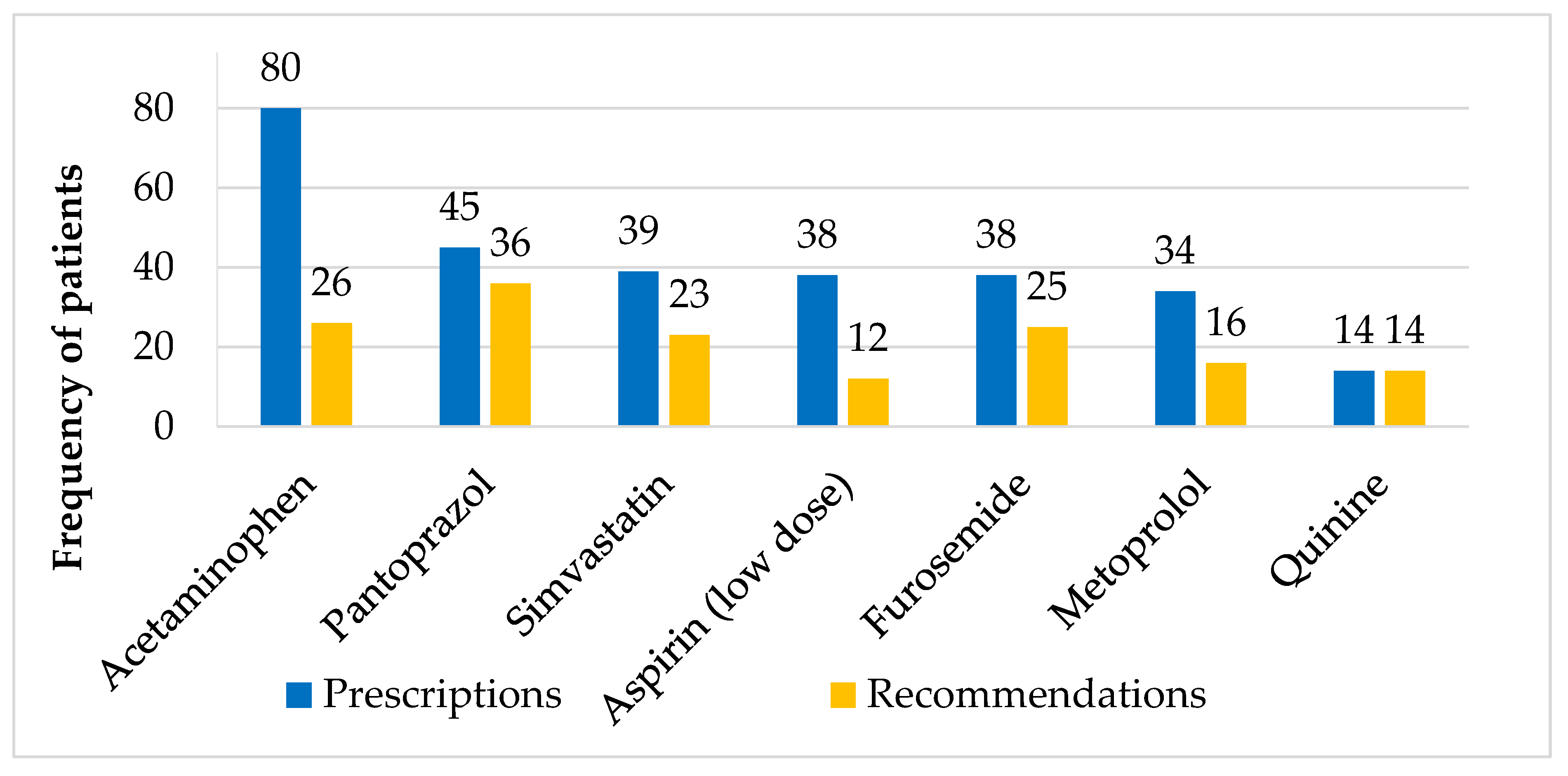
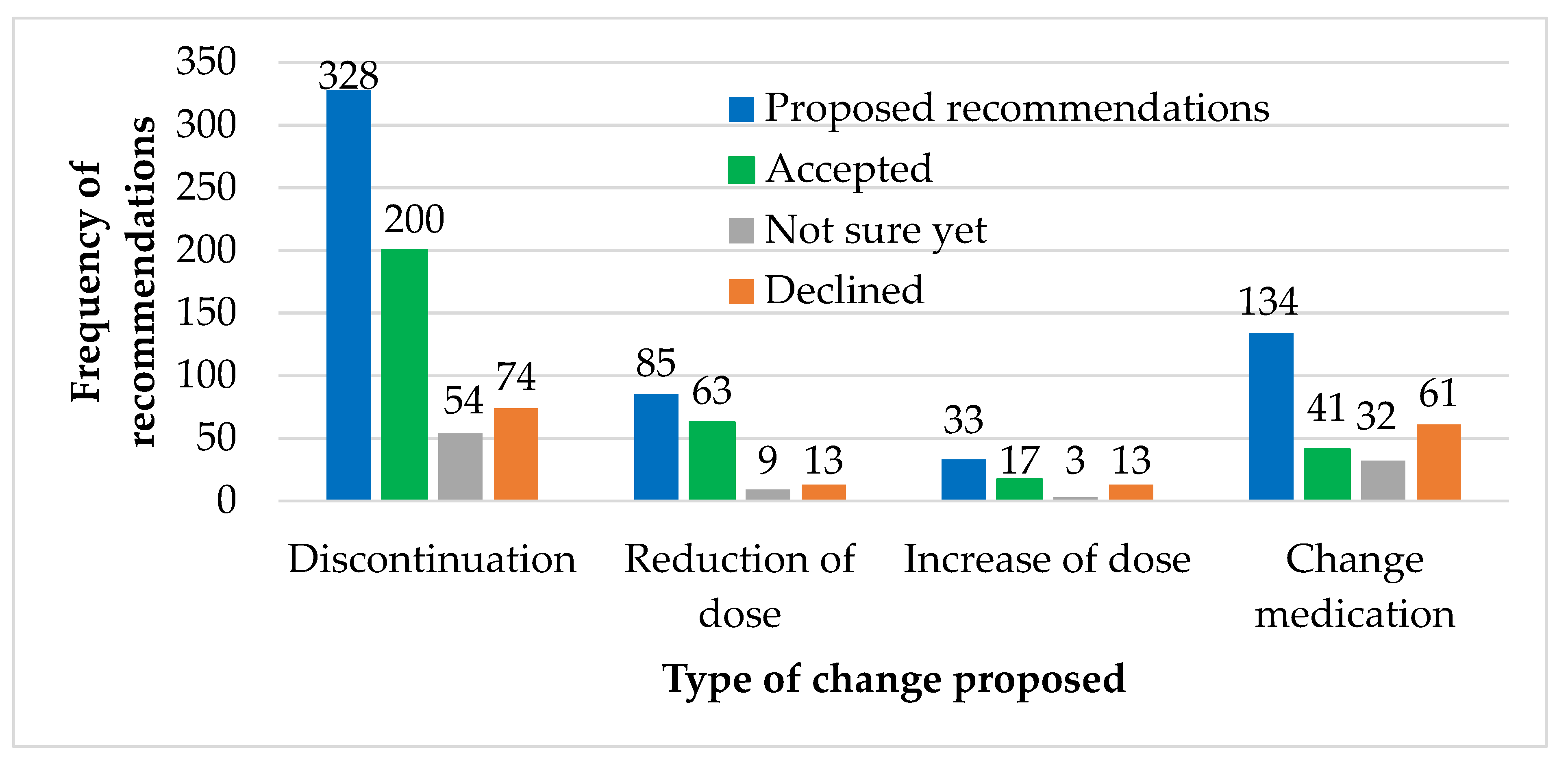
3.2. Prescriptions and Recommendations
3.3. Method—Quality of the Patient Cases
4. Discussion
4.1. Primary Findings
4.2. Recommended Changes to Medication
4.3. The Method
4.4. Patient Case
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Oscanoa, T.J.; Lizaraso, F.; Carvajal, A. Hospital admissions due to adverse drug reactions in the elderly. A meta-analysis. Eur. J. Clin. Pharmacol. 2017, 73, 759–770. [Google Scholar] [CrossRef] [PubMed]
- Hallas, J.; Gram, L.; Grodum, E.; Damsbo, N.; Brosen, K.; Haghfelt, T.; Harvald, B.; Nielsen, J.B.; Worm, J.; Jensen, K.; et al. Drug related admissions to medical wards: A population based survey. Br. J. Clin. Pharmacol. 1992, 33, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Geurts, M.M.; Talsma, J.; Brouwers, J.R.; de Gier, J.J. Medication review and reconciliation with cooperation between pharmacist and general practitioner and the benefit for the patient: A systematic review. Br. J. Clin. Pharmacol. 2012, 74, 16–33. [Google Scholar] [CrossRef] [PubMed]
- Hurkens, K.P.; Mestres-Gonzalvo, C.; De Wit, H.; Van Der Kuy, P.; Janknegt, R.; Verhey, F.; Schols, J.; Stehouwer, C.; Winkens, B.; Mulder, W. Usually available clinical and laboratory data are insufficient for a valid medication review: A crossover study. J. Nutr. Health Aging 2016, 20, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Krska, J.; Gill, D.; Hansford, D. Pharmacist-supported medication review training for general practitioners: Feasibility and acceptability. Med. Educ. 2006, 40, 1217–1225. [Google Scholar] [CrossRef] [PubMed]
- The Danish Health Authority. Styrket Indsats for Den Ældre Medicinske Patient [Improved Effort for the Older Medicated Patient]; Sundhedsstyrelsen: København, Denmark, 2016; Available online: https://www.sst.dk/-/media/Udgivelser/2016/Styrket-indsats-for-den-aeldre-medicinske-patient.ashx?la=da&hash=8F69A91EB3B59BC7AF2ABA646A7089D99BCD660E (accessed on 30 January 2020).
- Lee, L.; Patel, T.; Hillier, L.M.; Maulkhan, N.; Slonim, K.; Costa, A. Identifying frailty in primary care: A systematic review. Geriatr. Gerontol. Int. 2017, 17, 1358–1377. [Google Scholar] [CrossRef] [PubMed]
- Danish Nephrology Society; Danish Pædiatric Society; Danish Society for Clinical Biochemistry. Kronisk Nyresygdom: Analysemetoder Og Klinisk Evaluering [Chronic Kidney Disiease: Analitical Methods and Clinical Evaluation]; Danish Nephrology Society; Danish Pædiatric Society; Danish Society for Clinical Biochemistry: Copenhagen, Denmark, 2015; Available online: http://www.nephrology.dk/Publikationer/Kronisk%20nyresygdom%202015%20endelig,%2014-08-15.pdf (accessed on 30 January 2020).
- Snorgaard, O.; Kristensen, J.K.; Balasubramaniam, K.; Breum, L.; Charles, M.; Højlund, K.; Madsen, G.K.; Bruun, J.M.; Navntoft, D.; Rungby, J.; et al. Dansk Endokrinologisk Selskab [Danish Society of Endocrinology]. In National Behandlingsvejledning: Behandling Og Kontrol af Type 2 Diabetes [National Guidelines for Treatment of Diabetes Type 2]; Danish Society of Endocrinology, Copenhagen, Danish Society of Endocrinology: Copenhagen, Denmark, 2018; Available online: http://www.endocrinology.dk/index.php/1-diabetes-mellitus/nbv-endokrinologi-behandling-og-kontrol-af-type-2-diabetes-t2d-diabetes-arskontrol-nyopdaget-diabetes-2-peroral-behandling-insulin-behandling-kolesterolbehandling-blodtryksbehandling-glp1-og-dpp4 (accessed on 30 January 2020).
- Dansk Cardiologisk Selskab [Danish Cardiology Society]. National Cardiologisk Behandlingsvejledning: Arteriel Hypertension [National Cardiology Guideline: Arterial Hypertension]; Danish Cardiology Society: Copenhagen, Denmark, 2018; Available online: https://www.nbv.cardio.dk/hypertension (accessed on 30 January 2020).
- Dansk Cardiologisk Selskab [Danish Cardiology Society]. National Kardiologisk Behandlingsvejledning (NBV) [National Cardiology Guideline]; Danish Cardiology Society: Copenhagen, Denmark, 2017; Available online: https://www.nbv.cardio.dk/nbv (accessed on 30 January 2020).
- Dansk Endokrinologisk Selskab [Danish Society of Endocrinology]. Nationale Behandlingsvejledninger for Endokrinologi [National Guidelines of Endocrinology]; Danish Society of Endocrinology: Copenhagen, Denmark, 2017; Available online: http://www.endocrinology.dk/index.php/baggrund/forord (accessed on 30 January 2020).
- Dansk Neurologisk Selskab [Danish Society of Neurology]. Neurologisk National BehandlingsVejledning (NNBV) [Neurological National Guideline]; Danish Society of Neurology: Copenhagen, Denmark, 2016; Available online: https://neuro.dk/wordpress/nnbv/ (accessed on 30 January 2020).
- Dansk Selskab for Almen Medicin [Danish College of General Pratitioners]. KOL [COPD] 2017. Available online: https://vejledninger.dsam.dk/kol/ (accessed on 30 January 2020).
- Dansk Selskab for Gastroenterologi og Hepatologi [Danish Society of Gastroenterology and Hepatology]. Guidelines 2017. Available online: https://www.dsgh.dk/index.php/guidelines/guidelines (accessed on 30 January 2020).
- Global Initiative for Asthma (GINA). Global Strategy for Asthma Management and Prevention; Global Initiative for Asthma: Fontana, WI, USA, 2016; Available online: https://ginasthma.org/wp-content/uploads/2016/04/wms-GINA-2016-main-report-final.pdf (accessed on 30 January 2020).
- Global Initiative for Chronic Obstructive Lung Disease (GOLD). GOLD 2017 Global Stategy for the Diagnosis, Management and Prevention of COPD 2017. Available online: https://goldcopd.org/gold-2017-global-strategy-diagnosis-management-prevention-copd/ (accessed on 30 January 2020).
- Medicinfunktionen Region Hovedstaden [Medicine Unit, Capital Region of Denmark], KAP-H [Quality in General Practice in the Capital Region of Denmark]. Basislisten [Basic list]. 2017. Available online: https://www.sundhed.dk/sundhedsfaglig/information-til-praksis/hovedstaden/almen-praksis/laegemidler/basislisten-hovedstaden/ (accessed on 30 January 2020).
- Danish Health Authority; The Capitol Region of Denmark; Region Zealand. The Region of Southern Denmark, Central Denmark Region, The North Denmark Region; Seponeringslisten [The Deprescribing List]: Danish Health Authority: Copenhagen, Denmark, 2019; Available online: https://www.sst.dk/da/rationel-farmakoterapi/medicingennemgang/seponeringsliste (accessed on 30 January 2020).
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Dansk Cardiologisk Selskab [Danish Cardiology Society]. National Cardiologisk Behandlingsvejledning: Dyslipidæmi [National Cardology Guideline: Dyslididemia]; Danish Cardiology Society: Copenhagen, Denmark, 2018; Available online: https://www.nbv.cardio.dk/dyslipidaemi (accessed on 30 January 2020).
- Castelino, R.L.; Bajorek, B.V.; Chen, T.F. Are interventions recommended by pharmacists during Home Medicines Review evidence-based? J. Eval. Clin. Pract. 2011, 17, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Khera, S.; Abbasi, M.; Dabravolskaj, J.; Sadowski, C.A.; Yua, H.; Chevalier, B. Appropriateness of medications in older adults living with frailty: Impact of a pharmacist-led structured medication review process in primary care. J. Prim. Care Community Health 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Brulhart, M.I.; Wermeille, J.P. Multidisciplinary medication review: Evaluation of a pharmaceutical care model for nursing homes. Int. J. Clin. Pharm. 2011, 33, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Dolovich, L.; Pottie, K.; Kaczorowski, J.; Farrell, B.; Austin, Z.; Rodriguez, C.; Gaebel, K.; Sellors, C. Integrating family medicine and pharmacy to advance primary care therapeutics. Clin. Pharmacol. Ther. 2008, 83, 913–917. [Google Scholar] [CrossRef] [PubMed]
- Doucette, W.R.; McDonough, R.P.; Klepser, D.; McCarthy, R. Comprehensive medication therapy management: Identifying and resolving drug-related issues in a community pharmacy. Clin. Ther. 2005, 27, 1104–1111. [Google Scholar] [CrossRef]
- Gilbert, A.L.; Roughead, E.E.; Beilby, J.; Mott, K.; Barratt, J.D. Collaborative medication management services: Improving patient care. Med. J. Aust. 2002, 177, 189–192. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Soendergaard, B.; Kirkeby, B.; Dinsen, C.; Herborg, H.; Kjellberg, J.; Staehr, P. Drug-related problems in general practice: Results from a development project in Denmark. Pharm. World Sci. 2006, 28, 61–64. [Google Scholar] [CrossRef] [PubMed]
- Lenander, C.; Bondesson, Å.; Viberg, N.; Beckman, A.; Midlöv, P. Effects of medication reviews on use of potentially inappropriate medications in elderly patients; a cross-sectional study in Swedish primary care. BMC Health Serv. Res. 2018, 18, 616. [Google Scholar] [CrossRef] [PubMed]
- Nishtala, P.S.; McLachlan, A.J.; Bell, J.S.; Chen, T.F. A retrospective study of drug-related problems in Australian aged care homes: Medication reviews involving pharmacists and general practitioners. J. Eval. Clin. Pract. 2011, 17, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Niquille, A.; Bugnon, O. Relationship between drug-related problems and health outcomes: A cross-sectional study among cardiovascular patients. Pharm. World Sci. 2010, 32, 512–519. [Google Scholar] [CrossRef] [PubMed]
- Freeman, C.R.; Cottrell, W.N.; Kyle, G.; Williams, I.D.; Nissen, L. An evaluation of medication review reports across different settings. Int. J. Clin. Pharm. 2013, 35, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Clinical Pharmacists in General Practice: National Health Service. Available online: https://www.england.nhs.uk/contact-us/privacy-notice/how-we-use-your-information/public-and-partners/clinical-pharmacists-in-general-practice/ (accessed on 3 March 2020).
| Examination of Whether Provision of the Medication is Rational on the Basis of the Following Criteria | Comparison with the Following Guidelines/References |
|---|---|
| Indication matching diagnoses | Guidelines on specific diseases from Danish or international professional associations [11,12,13,14,15,16,17] |
| Contraindication | |
| Basislisten (The list of recommended medication in the Capital Region of Denmark) [18] | |
| Dose | |
| The Deprescribing List [19] of medications that can often be discontinued, including information on when and how to discontinue. This is published jointly by the Danish Health Authority and the regions of Denmark. | |
| Form of dosage | |
| Drug-drug interactions using two interaction databases (http://interaktionsdatabasen.dk and http://www.micromedexsolutions.com) | |
| Time of dosage | |
| Price |
| Information | Median | Interquartile Range | Range | Patients (n) |
| Age (years) | 79 | 72–83 | 65–98 | 94 |
| eGFR (mL/min) | 63 | 46–80 | 19–92 | 82 |
| Sex | Percentage of study population | Patients (n) | ||
| Female | 56.4 | 53 | ||
| Male | 43.6 | 41 | ||
| Stated diagnosis | Percentage of study population | Patients (n) | ||
| Cardiovascular | 90 | 85 | ||
| Musculoskeletal | 68 | 64 | ||
| Lung | 52 | 49 | ||
| Diabetes | 50 | 47 | ||
| Other | 45 | 42 | ||
| Gastrointestinal | 33 | 31 | ||
| Psychiatric | 26 | 24 | ||
| Pain | 24 | 23 | ||
| Metabolism | 20 | 19 | ||
| Neurologic | 19 | 18 | ||
| Cancer | 16 | 15 | ||
| Kidney | 13 | 12 | ||
| Infection | 3 | 3 | ||
| Laboratory data | Mean | Min-max | Goal-of-care | Patients (n) |
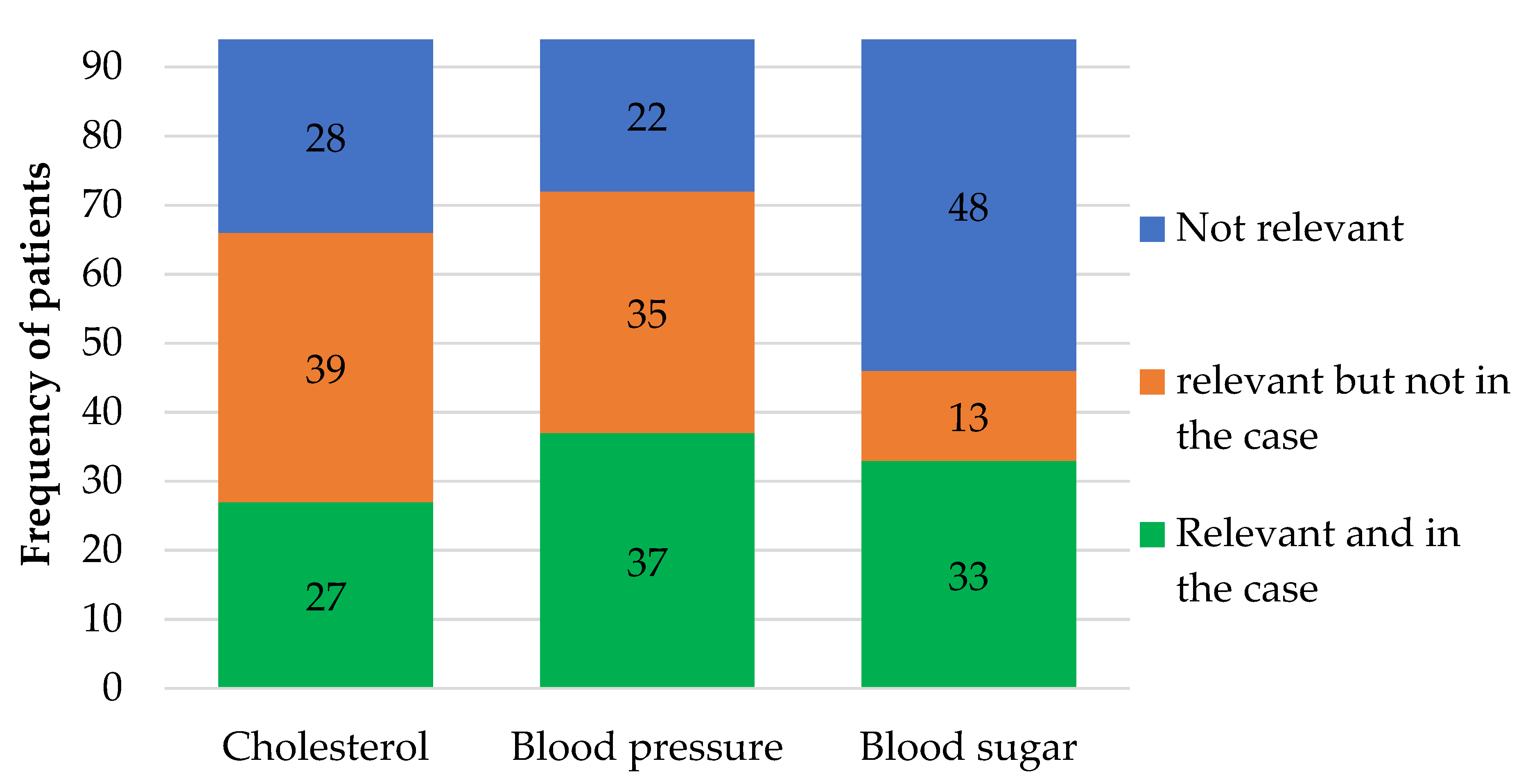
| Blood sugar HbA1c (mmol/mol) | 50.32 | 30.00–82.00 | Individual < 48–75 [9] | 49 |
| Cholesterol LDL (mmol/l) | 2.33 | 0.2–6.7 | Individual < 1.8–3.0 [21] | 52 |
| Blood pressure (mmHg) | 136/74 | 100/40–180/97 | Individual < 130/80–145/85 [10] | 61 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dalin, D.A.; Vermehren, C.; Jensen, A.K.; Unkerskov, J.; Andersen, J.T. Systematic Medication Review in General Practice by an Interdisciplinary Team: A thorough but Laborious Method to Address Polypharmacy among Elderly Patients. Pharmacy 2020, 8, 57. https://doi.org/10.3390/pharmacy8020057
Dalin DA, Vermehren C, Jensen AK, Unkerskov J, Andersen JT. Systematic Medication Review in General Practice by an Interdisciplinary Team: A thorough but Laborious Method to Address Polypharmacy among Elderly Patients. Pharmacy. 2020; 8(2):57. https://doi.org/10.3390/pharmacy8020057
Chicago/Turabian StyleDalin, Dagmar Abelone, Charlotte Vermehren, Anette Kobberø Jensen, Janne Unkerskov, and Jon Trærup Andersen. 2020. "Systematic Medication Review in General Practice by an Interdisciplinary Team: A thorough but Laborious Method to Address Polypharmacy among Elderly Patients" Pharmacy 8, no. 2: 57. https://doi.org/10.3390/pharmacy8020057
APA StyleDalin, D. A., Vermehren, C., Jensen, A. K., Unkerskov, J., & Andersen, J. T. (2020). Systematic Medication Review in General Practice by an Interdisciplinary Team: A thorough but Laborious Method to Address Polypharmacy among Elderly Patients. Pharmacy, 8(2), 57. https://doi.org/10.3390/pharmacy8020057





