Hypersensitivity Reactions in Serious Adverse Events Reported for Paracetamol in the EudraVigilance Database, 2007–2018
Abstract
:1. Introduction
2. Materials and Methods
3. Statistical analysis
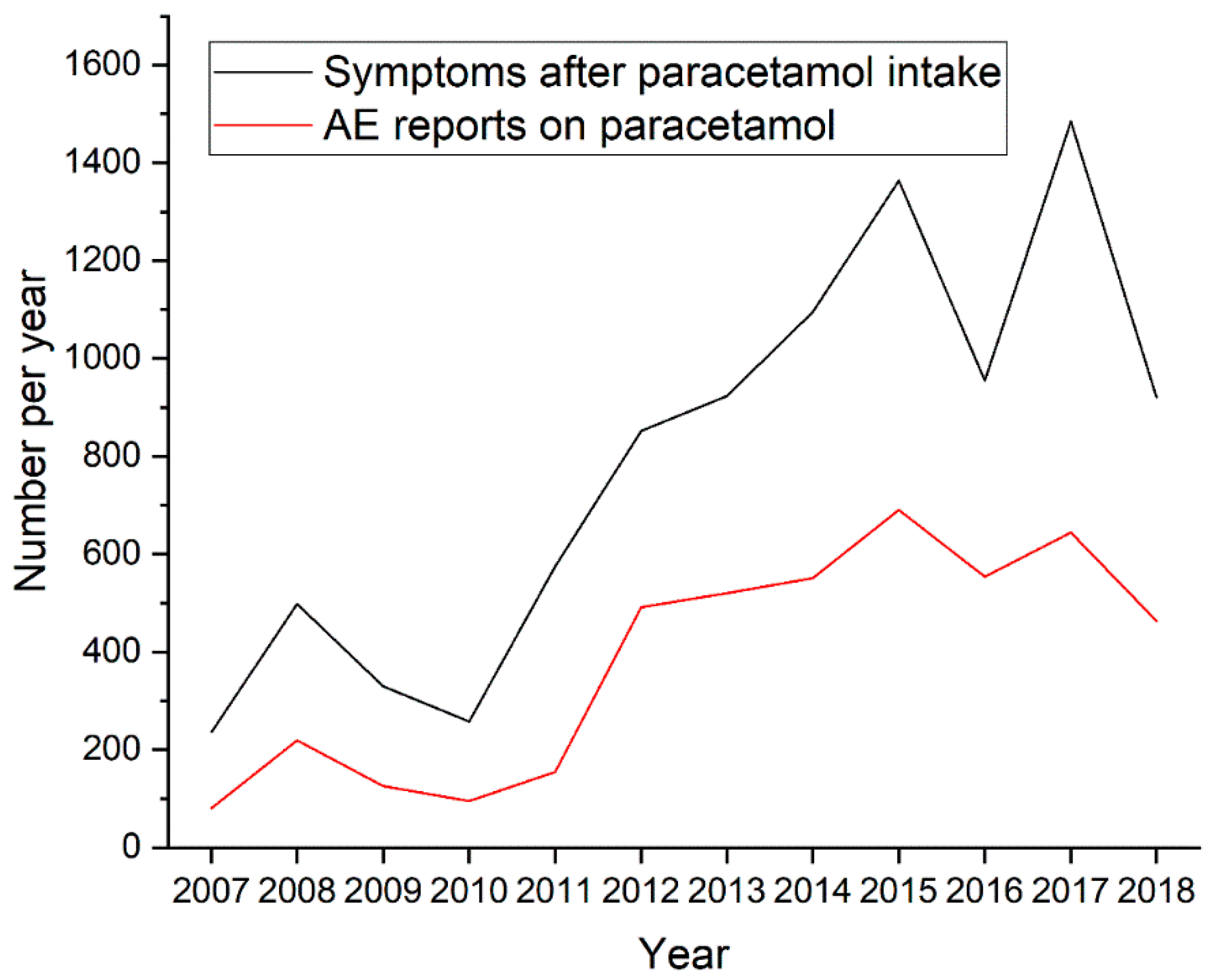
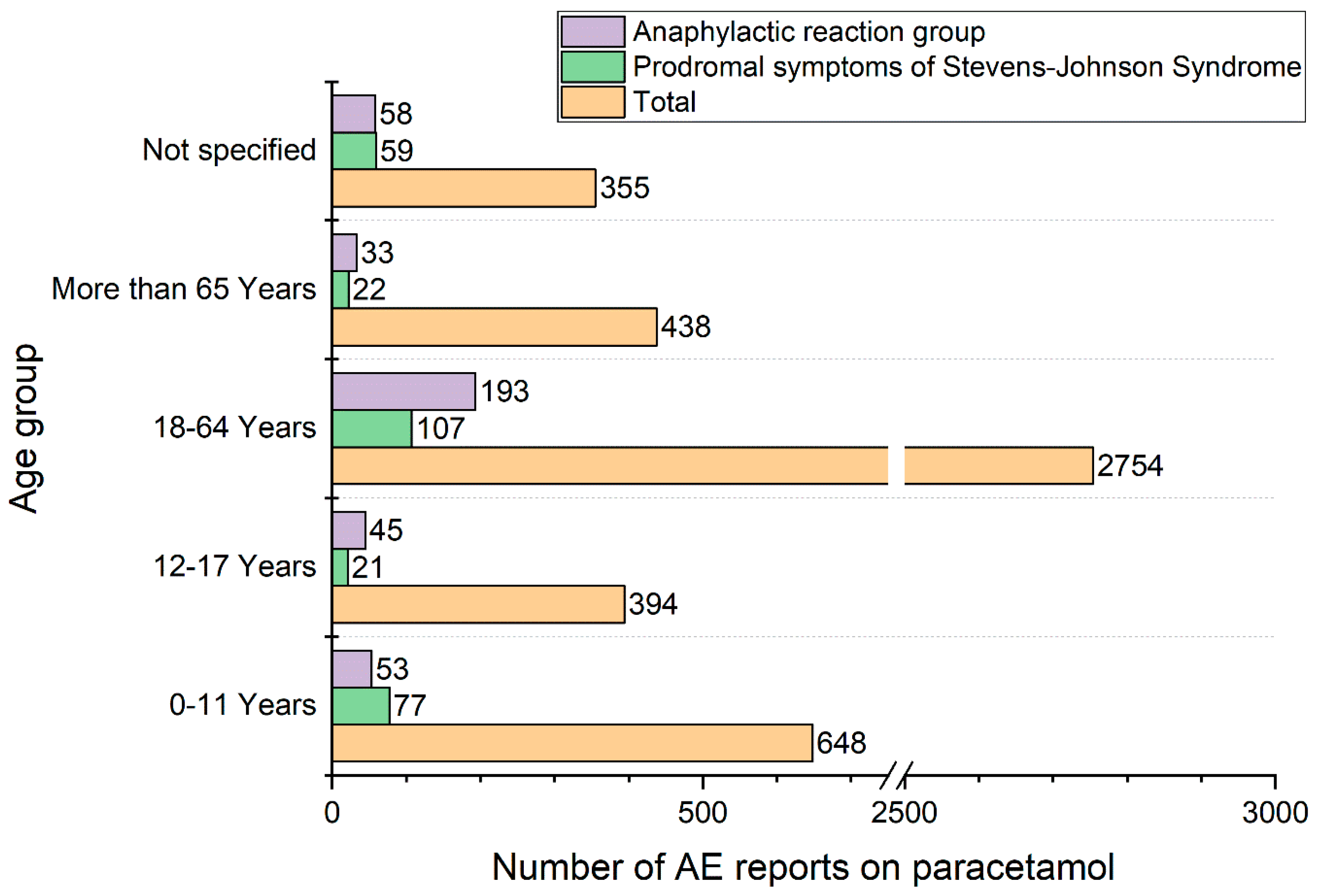
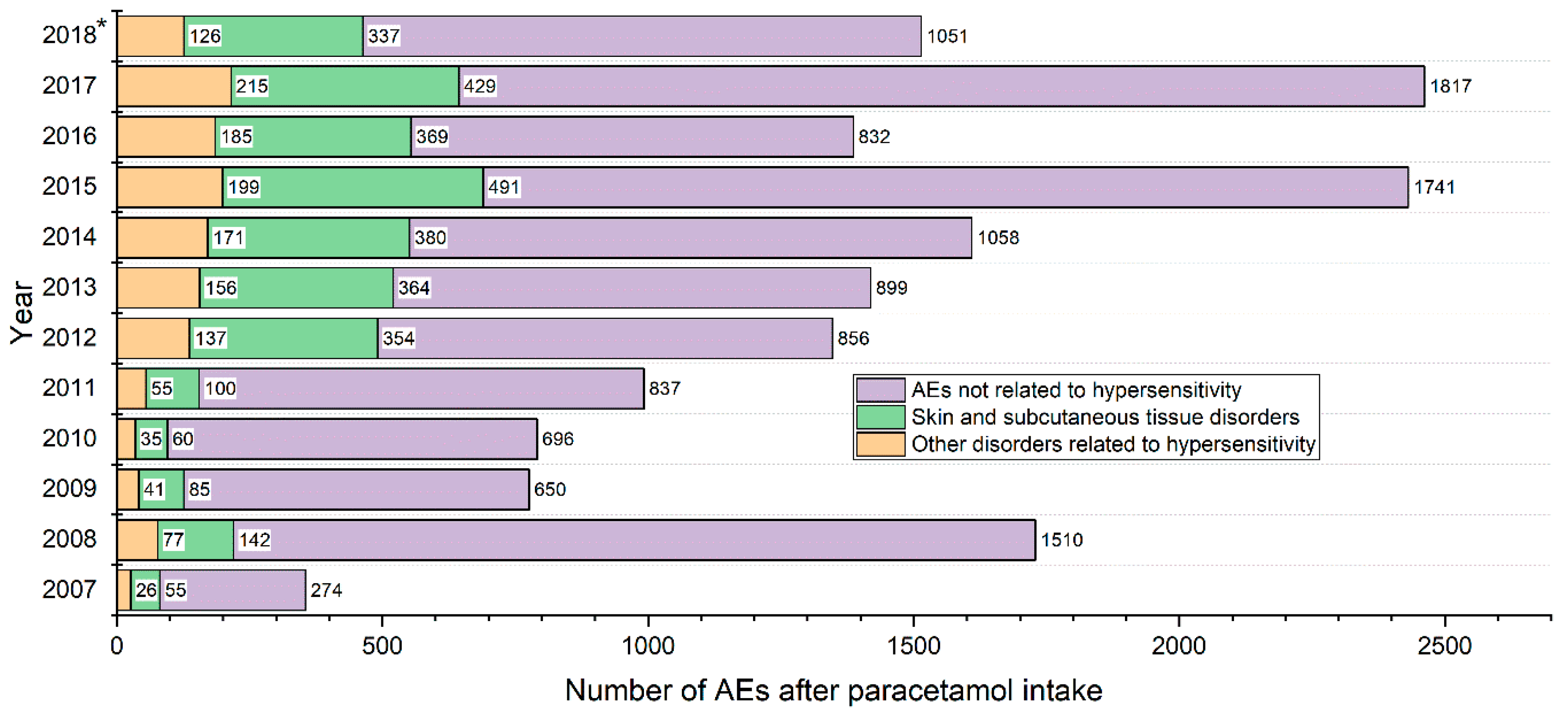
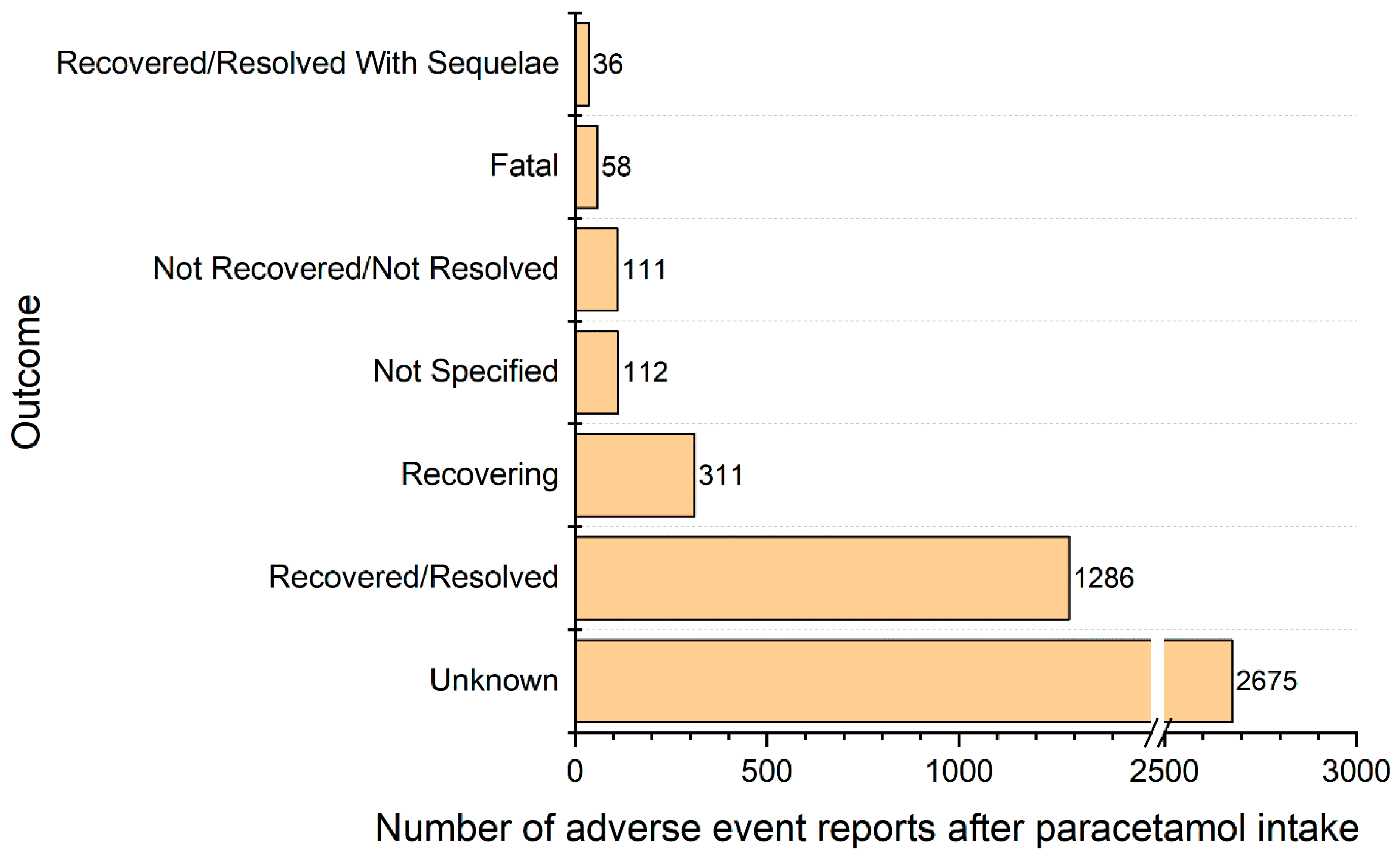
4. Results
Population Characteristic, Presentation of Results
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jóźwiak-Bebenista, M.; Nowak, J.Z. Paracetamol: Mechanism of action, applications and safety concern. Acta Pol. Pharm. 2014, 71, 11–23. [Google Scholar] [PubMed]
- Dear, J.W.; Antoine, D.J.; Park, B.K. Where are we now with paracetamol? BMJ 2015, 351, h3705. [Google Scholar] [CrossRef]
- Sharma, C.V.; Mehta, V. Paracetamol: Mechanisms and updates. Contin. Educ. Anaesth. Crit. Care Pain 2014, 14, 153–158. [Google Scholar] [CrossRef]
- Drugs.com Paracetamol Side Effects. Available online: https://www.drugs.com/sfx/paracetamol-side-effects.html (accessed on 27 December 2018).
- Craig, D.G.N.; Bates, C.M.; Davidson, J.S.; Martin, K.G.; Hayes, P.C.; Simpson, K.J. Overdose pattern and outcome in paracetamol-induced acute severe hepatotoxicity: Outcome following unintentional paracetamol overdose. Br. J. Clin. Pharmacol. 2011, 71, 273–282. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency EudraVigilance. Available online: https://www.ema.europa.eu/en/human-regulatory/research-development/pharmacovigilance/eudravigilance (accessed on 27 December2018).
- The European Parliament and The Council Directive 2001/83/EC of 6 November 2001 on the Community Code Relating to Medicinal Products for Human Use. 2011. Available online: https://ec.europa.eu/health/sites/health/files/files/eudralex/vol–1/dir_2001_83_consol_2012/dir_2001_83_cons_2012_en.pdf (accessed on 27 December 2018).
- European Medicines Agency 2017 Annual Report on EudraVigilance for the European Parliament, the Council and the Commission. Available online: https://www.ema.europa.eu/documents/report/2017-annual-report-eudravigilance-european-parliament-council-commission-reporting-period-1-january_en.pdf (accessed on 27 December 2018).
- Celik, G.; Pichler, W.; Adkinson, N. Drug Allergy. In Middleton’s Allergy: Principles & Practice; Mosby/Elsevier: Philadelphia, PA, USA, 2009; pp. 1205–1226. ISBN 978-0-323-05659-5. [Google Scholar]
- Aronson, J.K.; Ferner, R.E. Clarification of terminology in drug safety. Drug Saf. 2005, 28, 851–870. [Google Scholar] [CrossRef] [PubMed]
- Yoon, E.; Babar, A.; Choudhary, M.; Kutner, M.; Pyrsopoulos, N. Acetaminophen-Induced Hepatotoxicity: A Comprehensive Update. J. Clin. Transl. Hepatol. 2016, 4, 131–142. [Google Scholar]
- Rutkowski, K.; Nasser, S.M.; Ewan, P.W. Paracetamol hypersensitivity: Clinical features, mechanism and role of specific IgE. Int. Arch. Allergy Immunol. 2012, 159, 60–64. [Google Scholar] [CrossRef]
- Rajput, R.; Sagari, S.; Durgavanshi, A.; Kanwar, A. Paracetamol induced Steven-Johnson syndrome: A rare case report. Contemp. Clin. Dent. 2015, 6, 278. [Google Scholar] [CrossRef] [PubMed]
- FDA Drug Safety Communication FDA Warns of Rare but Serious Skin Reactions with the Pain Reliever/Fever Reducer Acetaminophen. Available online: https://www.fda.gov/Drugs/DrugSafety/ucm363041.htm (accessed on 29 November 2018).
- Gyamlani, G.G.; Parikh, C.R. Acetaminophen toxicity: Suicidal vs. accidental. Crit Care 2002, 6, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Blieden, M.; Paramore, L.C.; Shah, D.; Ben-Joseph, R. A perspective on the epidemiology of acetaminophen exposure and toxicity in the United States. Expert Rev. Clin. Pharmacol. 2014, 7, 341–348. [Google Scholar] [CrossRef]
- Allanore, L.; Roujeau, J.-C. Clinic and Pathogenesis of Severe Bullous Skin Reactions: Stevens-Johnson Syndrome, Toxic Epidermal Necrolysis. In Drug Hypersensitivity; Pichler, W.J., Ed.; Karger: Basel, Switzerlands, 2007; pp. 267–277. ISBN 978-3-8055-8269-8. [Google Scholar]
- Sánchez-Borges, M.; Caballero-Fonseca, F.; Capriles-Hulett, A.; González-Aveledo, L. Hypersensitivity Reactions to Nonsteroidal Anti-Inflammatory Drugs: An Update. Pharmaceuticals 2010, 3, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Simons, F.E.R.; Ebisawa, M.; Sanchez-Borges, M.; Thong, B.Y.; Worm, M.; Tanno, L.K.; Lockey, R.F.; El-Gamal, Y.M.; Brown, S.G.; Park, H.-S.; et al. 2015 update of the evidence base: World Allergy Organization anaphylaxis guidelines. World Allergy Organ. J. 2015, 8, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Numata, T.; Fukushi, R.; Ito, T.; Tsuboi, R.; Harada, K. Acetaminophen anaphylaxis diagnosed by skin prick test. Allergol. Int. 2016, 65, 490–491. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Settipane, R.; Schrank, P.; Simon, R.; Mathison, D.; Christiansen, S.; Stevenson, D. Prevalence of cross-sensitivity with acetaminophen in aspirin-sensitive asthmatic subjects. J. Allergy Clin. Immunol. 1995, 96, 480–485. [Google Scholar] [CrossRef]
- Lee, Q. Hypersensitivity to antipyretics: Pathogenesis, diagnosis and management. Hong Kong Med. J. 2017, 23, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Rademaker, M. Do Women Have More Adverse Drug Reactions? Am. J. Clin. Dermatol. 2001, 2, 349–351. [Google Scholar] [CrossRef] [PubMed]
- Kando, J.C.; Yonkers, K.A.; Cole, J.O. Gender as a Risk Factor for Adverse Events to Medications. Drugs 1995, 50, 1–6. [Google Scholar] [CrossRef]
- Rydberg, D.M.; Mejyr, S.; Loikas, D.; Schenck-Gustafsson, K.; von Euler, M.; Malmström, R.E. Sex differences in spontaneous reports on adverse drug events for common antihypertensive drugs. Eur. J. Clin. Pharmacol. 2018, 74, 1165–1173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Toki, T.; Ono, S. Spontaneous Reporting on Adverse Events by Consumers in the United States: An Analysis of the Food and Drug Administration Adverse Event Reporting System Database. Drugs Real World Outcomes 2018, 5, 117–128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stergiopoulos, S.; Brown, C.A.; Felix, T.; Grampp, G.; Getz, K.A. A Survey of Adverse Event Reporting Practices Among US Healthcare Professionals. Drug Saf. 2016, 39, 1117–1127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cranswick, N.; Coghlan, D. Paracetamol efficacy and safety in children: The first 40 years. Am. J. Ther. 2000, 7, 135–141. [Google Scholar] [CrossRef]
- Eccles, R. Efficacy and safety of over-the-counter analgesics in the treatment of common cold and flu. J. Clin. Pharmacy Ther. 2006, 31, 309–319. [Google Scholar] [CrossRef] [Green Version]
- Rojas-Pérez-Ezquerra, P.; Sánchez-Morillas, L.; Gómez-Traseira, C.; Gonzalez-Mendiola, R.; Alcorta Valle, A.R.; Laguna-Martinez, J. Selective hypersensitivity reactions to acetaminophen: A 13-case series. J. Allergy Clin. Immunol. Pract. 2014, 2, 343–345. [Google Scholar] [CrossRef] [PubMed]
- Muraro, A.; Roberts, G.; Worm, M.; Bilò, M.B.; Brockow, K.; Fernández Rivas, M.; Santos, A.F.; Zolkipli, Z.Q.; Bellou, A.; Beyer, K.; et al. Anaphylaxis: Guidelines from the European Academy of Allergy and Clinical Immunology. Allergy 2014, 69, 1026–1045. [Google Scholar] [CrossRef]
- Creamer, D.; Walsh, S.A.; Dziewulski, P.; Exton, L.S.; Lee, H.Y.; Dart, J.K.G.; Setterfield, J.; Bunker, C.B.; Ardern-Jones, M.R.; Watson, K.M.T.; et al. U.K. guidelines for the management of Stevens-Johnson syndrome/toxic epidermal necrolysis in adults 2016. Br. J. Dermatol. 2016, 174, 1194–1227. [Google Scholar] [CrossRef] [Green Version]
- Feldmeyer, L.; Heidemeyer, K.; Yawalkar, N. Acute Generalized Exanthematous Pustulosis: Pathogenesis, Genetic Background, Clinical Variants and Therapy. Int. J. Mol. Sci. 2016, 17, 1214. [Google Scholar] [CrossRef]
- Sidoroff, A. Acute Generalized Exanthematous Pustulosis. In Chemical Immunology and Allergy; French, L.E., Ed.; S. KARGER AG: Basel, Switzerlands, 2012; Volume 97, pp. 139–148. ISBN 978-3-8055-9970-2. [Google Scholar]
- Chen, Y.-C.; Cho, Y.-T.; Chang, C.-Y.; Chu, C.-Y. Drug reaction with eosinophilia and systemic symptoms: A drug-induced hypersensitivity syndrome with variable clinical features. Dermatol. Sin. 2013, 31, 196–204. [Google Scholar] [CrossRef] [Green Version]
- Porebski, G. In Vitro Assays in Severe Cutaneous Adverse Drug Reactions: Are They Still Research Tools or Diagnostic Tests Already? Int. J. Mol. Sci. 2017, 18, 1737. [Google Scholar] [CrossRef]
- Porębski, G.; Czarnobilska, E.; Bosak, M. Cytotoxic-based assays in delayed drug hypersensitivity reactions induced by antiepileptic drugs. Pol. Arch. Med. Wewn. 2015, 125, 823–834. [Google Scholar] [CrossRef]
- Rolfes, L.; van Hunsel, F.; van der Linden, L.; Taxis, K.; van Puijenbroek, E. The Quality of Clinical Information in Adverse Drug Reaction Reports by Patients and Healthcare Professionals: A Retrospective Comparative Analysis. Drug Saf. 2017, 40, 607–614. [Google Scholar] [CrossRef]
- Inácio, P.; Cavaco, A.; Airaksinen, M. The value of patient reporting to the pharmacovigilance system: A systematic review: The value of patient reporting to the pharmacovigilance system. Br. J. Clin. Pharmacol. 2017, 83, 227–246. [Google Scholar] [CrossRef]
| Adverse Event | n | % of Reports | Adverse Event | n | % of Reports |
|---|---|---|---|---|---|
| Angioedema | 1108 | 24.1% | Stevens-Johnson syndrome | 129 | 2.8% |
| Rash | 847 | 18.5% | Anaphylactic shock | 115 | 2.5% |
| Urticaria | 480 | 10.5% | Fixed eruption | 83 | 1.8% |
| Orbital or periorbital oedema | 390 | 8.5% | Rash generalised | 76 | 1.7% |
| Head, neck or respiratory tract oedema excluding orbital or periorbital area | 319 | 7.0% | Lip swelling | 75 | 1.6% |
| Hypersensitivity; Drug hypersensitivity | 316 | 6.9% | Swelling face | 72 | 1.6% |
| Eye swelling | 263 | 5.7% | Rash erythematous | 65 | 1.4% |
| Anaphylactic reaction | 246 | 5.4% | Cough | 65 | 1.4% |
| Erythema | 173 | 3.8% | Blister | 58 | 1.3% |
| Face oedema | 163 | 3.6% | Maculo-papular rash | 53 | 1.2% |
| Oedema in other than head or unspecified localisation | 97 | 2.1% |
| Reaction Groups | Healthcare Professional | Non-Healthcare Professional | Not Specified/Missing | Total |
|---|---|---|---|---|
| Skin and subcutaneous tissue disorders | 2657 | 493 | 16 | 3166 |
| Eye disorders | 532 | 116 | 2 | 650 |
| Immune system disorders | 559 | 159 | 4 | 722 |
| General disorders and administration site conditions | 230 | 0 | 0 | 230 |
| Vascular disorders | 178 | 37 | 1 | 216 |
| Respiratory, thoracic and mediastinal disorders | 172 | 53 | 0 | 225 |
| Gastrointestinal disorders | 136 | 29 | 2 | 167 |
| Reproductive system and breast disorders | 9 | 15 | 0 | 24 |
| Ear and labyrinth disorders | 4 | 4 | 0 | 8 |
| Investigations | 2 | 1 | 0 | 3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Popiołek, I.; Piotrowicz-Wójcik, K.; Porebski, G. Hypersensitivity Reactions in Serious Adverse Events Reported for Paracetamol in the EudraVigilance Database, 2007–2018. Pharmacy 2019, 7, 12. https://doi.org/10.3390/pharmacy7010012
Popiołek I, Piotrowicz-Wójcik K, Porebski G. Hypersensitivity Reactions in Serious Adverse Events Reported for Paracetamol in the EudraVigilance Database, 2007–2018. Pharmacy. 2019; 7(1):12. https://doi.org/10.3390/pharmacy7010012
Chicago/Turabian StylePopiołek, Iwona, Katarzyna Piotrowicz-Wójcik, and Grzegorz Porebski. 2019. "Hypersensitivity Reactions in Serious Adverse Events Reported for Paracetamol in the EudraVigilance Database, 2007–2018" Pharmacy 7, no. 1: 12. https://doi.org/10.3390/pharmacy7010012
APA StylePopiołek, I., Piotrowicz-Wójcik, K., & Porebski, G. (2019). Hypersensitivity Reactions in Serious Adverse Events Reported for Paracetamol in the EudraVigilance Database, 2007–2018. Pharmacy, 7(1), 12. https://doi.org/10.3390/pharmacy7010012





