Evaluation of Peak Inspiratory Flow Rate in Hospitalized Palliative Care Patients with COPD
Abstract
1. Introduction
2. Materials and Methods
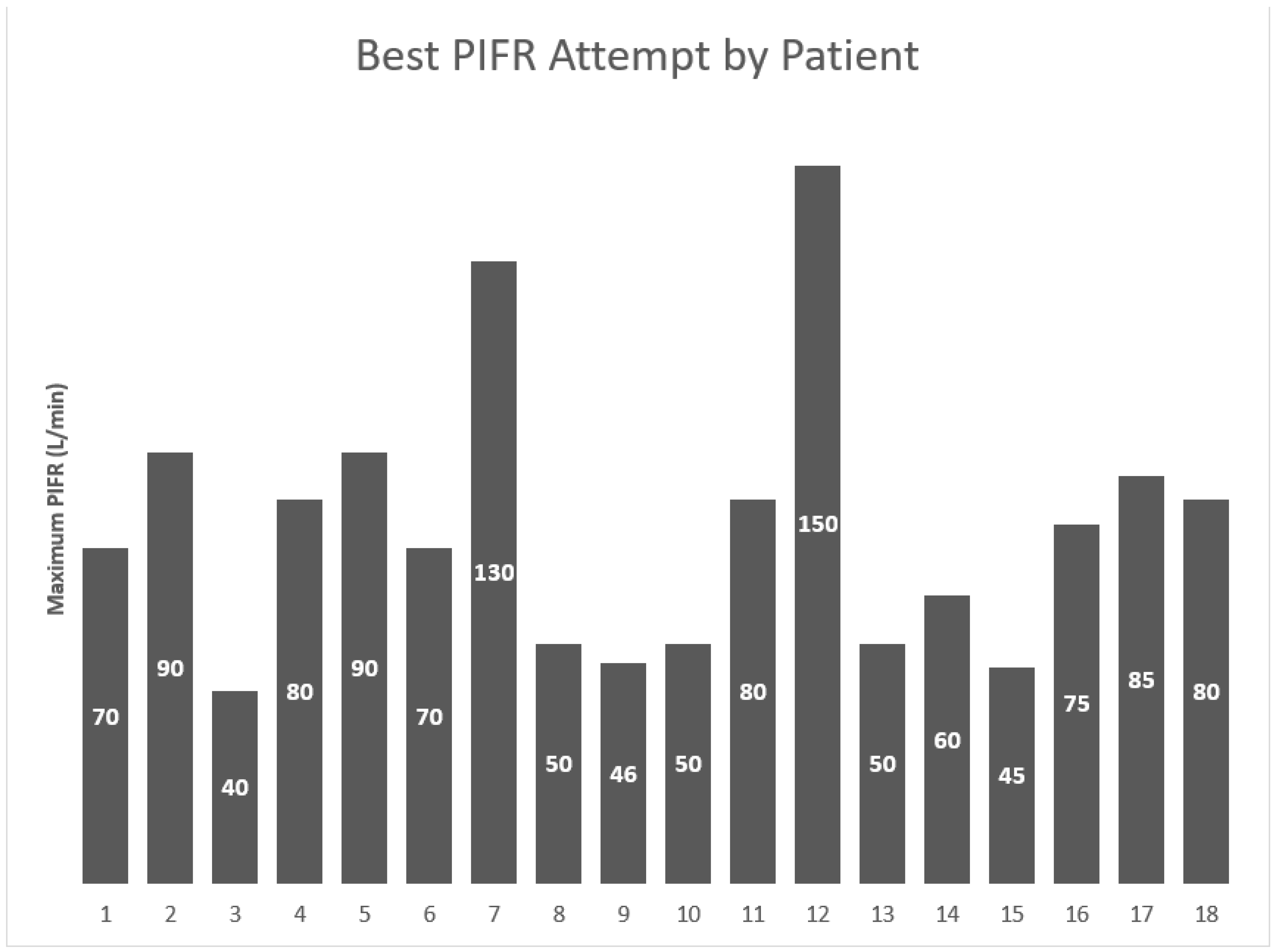
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Guarascio, A.J.; Ray, S.M.; Finch, C.K.; Self, T.H. The clinical and economic burden of chronic obstructive pulmonary disease in the USA. Clin. Outcomes Res. CEOR 2013, 5, 235–245. [Google Scholar] [CrossRef]
- Iheanacho, I.; Zhang, S.; King, D.; Rizzo, M.; Ismaila, A.S. Economic Burden of Chronic Obstructive Pulmonary Disease (COPD): A Systematic Literature Review. Int. J. Chronic Obstr. Pulm. Dis. 2020, 15, 439–460. [Google Scholar] [CrossRef]
- Chronic Obstructive Pulmonary Disease (COPD). Data and Statistics. Centers for Disease Control and Prevention. Available online: https://www.cdc.gov/copd/data.html (accessed on 6 March 2022).
- WHO. Palliative Care. World Health Organization. Available online: https://www.who.int/news-room/fact-sheets/detail/palliative-care (accessed on 6 March 2022).
- Brocklebank, D.; Ram, F.; Wright, J.; Barry, P.; Cates, C.; Davies, L.; Douglas, G.; Muers, M.; Smith, D.; White, J. Comparison of the effectiveness of inhaler devices in asthma and chronic obstructive airways disease: A systematic review of the literature. Health Technol Assess Winch Engl. 2001, 5, 1–149. [Google Scholar] [CrossRef]
- Albuterol/Ipratropium Prices, Coupons & Patient Assistance Programs. Drugs.com. Available online: https://www.drugs.com/price-guide/albuterol-ipratropium (accessed on 6 March 2022).
- Advair Diskus Prices, Coupons & Patient Assistance Programs. Drugs.com. Available online: https://www.drugs.com/price-guide/advair-diskus (accessed on 6 March 2022).
- Ghosh, S.; Ohar, J.A.; Drummond, M.B. Peak Inspiratory Flow Rate in Chronic Obstructive Pulmonary Disease: Implications for Dry Powder Inhalers. J. Aerosol. Med. Pulm. Drug Deliv. 2017, 30, 381–387. [Google Scholar] [CrossRef] [PubMed]
- GlaxoSmithKline. Serevent Diskus; GlaxoSmithKline: Brentford, UK, 2006. [Google Scholar]
- Merck & Co., Inc. Foradil Aerolizer; Merck & Co., Inc.: New York, NY, USA, 2011. [Google Scholar]
- GlaxoSmithKline. Incruse Ellipta; GlaxoSmithKline: Brentford, UK, 2016. [Google Scholar]
- Almirall. Tudorza Pressair; Almirall: Wilmington, DE, USA, 2019. [Google Scholar]
- Boehringer Ingelheim International GmbH. Spiriva Handihaler; Boehringer Ingelheim International GmbH: Ridgefield, CT, USA, 2006. [Google Scholar]
- Novartis AG. Utibron Neohaler; Novartis AG: East Hanover, NJ, USA, 2015. [Google Scholar]
- Yokoyama, H.; Yamamura, Y.; Ozeki, T.; Iga, T.; Yamada, Y. Analysis of Relationship between Peak Inspiratory Flow Rate and Amount of Drug Delivered to Lungs Following Inhalation of Fluticasone Propionate with a Diskhaler. Biol. Pharm. Bull. 2007, 30, 162–164. [Google Scholar] [CrossRef] [PubMed]
- Weers, J.; Clark, A. The Impact of Inspiratory Flow Rate on Drug Delivery to the Lungs with Dry Powder Inhalers. Pharm. Res. 2017, 34, 507–528. [Google Scholar] [CrossRef]
- Harb, H.S.; Laz, N.I.; Rabea, H.; Abdelrahim, M.E.A. Prevalence and predictors of suboptimal peak inspiratory flow rate in COPD patients. Eur. J. Pharm. Sci. 2020, 147, 105298. [Google Scholar] [CrossRef]
- Ghosh, S.; Pleasants, R.A.; Ohar, J.A.; Donohue, J.F.; Drummond, M.B. Prevalence and factors associated with suboptimal peak inspiratory flow rates in COPD. Int. J. Chronic Obstr. Pulm. Dis. 2019, 14, 585–595. [Google Scholar] [CrossRef]
- Mahler, D.A. The role of inspiratory flow in selection and use of inhaled therapy for patients with chronic obstructive pulmonary disease. Respir. Med. 2020, 161, 105857. [Google Scholar] [CrossRef]
- Connor, S.R.; Bermedo, M.C.S. Worldwide Palliative Care Alliance, World Health Organization. Global Atlas of Palliative Care at the End of Life. Worldwide Palliative Care Alliance. 2014. Available online: https://www.who.int/nmh/Global_Atlas_of_Palliative_Care.pdf (accessed on 6 March 2022).
- Soler-Cataluña, J.J.; Martínez-García, M.A.; Román Sánchez, P.; Salcedo, E.; Navarro, M.; Ochando, R. Severe acute exacerbations and mortality in patients with chronic obstructive pulmonary disease. Thorax 2005, 60, 925–931. [Google Scholar] [CrossRef]
- Global Initiative for Chronic Obstructive Lung Disease. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease. Available online: https://goldcopd.org/wp-content/uploads/2020/11/GOLD-REPORT-2021-v1.1-25Nov20_WMV.pdf (accessed on 6 March 2022).
- Alliance Tech Medical. In-Check. Alliance Tech Medical. Available online: https://alliancetechmedical.com/check-inspiratory-flow-measurement-device/ (accessed on 6 March 2022).
- Faul, F.; Erdfelder, E.; Lang, A.G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef]
- Sharma, G.; Mahler, D.A.; Mayorga, V.M.; Harshaw, Q.; Deering, K.L.; Ganapathy, V. Prevalence of Low Peak Inspiratory Flow Rate at Discharge in Patients Hospitalized for COPD Exacerbation. Chronic Obstr. Pulm. Dis. J. COPD Found. 2017, 4, 217–224. [Google Scholar] [CrossRef]
- Samarghandi, A.; Ioachimescu, O.C.; Qayyum, R. Association between peak inspiratory flow rate and hand grip muscle strength in hospitalized patients with acute exacerbation of chronic obstructive pulmonary disease. PLoS ONE 2020, 15, e0227737. [Google Scholar] [CrossRef] [PubMed]
- Loh, C.H.; Peters, S.P.; Lovings, T.M.; Ohar, J.A. Suboptimal Inspiratory Flow Rates Are Associated with Chronic Obstructive Pulmonary Disease and All-Cause Readmissions. Ann. Am. Thorac. Soc. 2017, 14, 1305–1311. [Google Scholar] [CrossRef] [PubMed]
- Alqahtani, J.S.; Aldabayan, Y.S.; Aldhahir, A.M.; Al Rajeh, A.M.; Mandal, S.; Hurst, J.R. Predictors of 30- and 90-Day COPD Exacerbation Readmission: A Prospective Cohort Study. Int. J. Chronic Obstr. Pulm. Dis. 2021, 16, 2769–2781. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.Y.; Huang, C.K.; Peng, H.C.; Tsai, H.C.; Huang, S.Y.; Yu, C.J.; Chien, J.Y. Peak-Inspiratory-Flow-Rate Guided Inhalation Therapy Reduce Severe Exacerbation of COPD. Front Pharmacol. 2021, 12, 704316. [Google Scholar] [CrossRef]
- Shireman, T.; Han, L.; Davidson, H.E.; Radlowski, P.; Dembek, C.; Gravenstein, S. Assessment of Peak Inspiratory Flow Rates and Medical Management of COPD in Nursing Facility Residents. Chest 2019, 156, A1748–A1749. [Google Scholar] [CrossRef]
- Duarte, A.G.; Tung, L.; Zhang, W.; Hsu, E.S.; Kuo, Y.F.; Sharma, G. Spirometry Measurement of Peak Inspiratory Flow Identifies Suboptimal Use of Dry Powder Inhalers in Ambulatory Patients with COPD. Chronic Obstr. Pulm. Dis. J. COPD Found. 2019, 6, 246–255. [Google Scholar] [CrossRef]
- Mahler, D.A.; Waterman, L.A.; Ward, J.; Gifford, A.H. Comparison of dry powder versus nebulized beta-agonist in patients with COPD who have suboptimal peak inspiratory flow rate. J. Aerosol Med. Pulm. Drug Deliv. 2014, 27, 103–109. [Google Scholar] [CrossRef]
- Newman, S. Improving inhaler technique, adherence to therapy and the precision of dosing: Major challenges for pulmonary drug delivery. Expert Opin. Drug Deliv. 2014, 11, 365–378. [Google Scholar] [CrossRef]
- Afrane, M.; Sera, L.; Holmes, H.M.; McPherson, M.L. Commonly Prescribed Medications Among Patients in Hospice Care for Chronic Obstructive Pulmonary Disease. Am. J. Hosp. Palliat. Med. 2016, 33, 638–643. [Google Scholar] [CrossRef] [PubMed]
| Dry Powder Inhaler (DPI) | Optimal PIFR Recommended | Minimum PIFR Required |
|---|---|---|
| Salmeterol Serevent Diskus® (Brentford, UK) | 60 L/min | 30 L/min |
| Formoterol Aerolizer® (New York, NY, USA) | 60 L/min | 30 L/min |
| Vilanterol and/or umeclidinium Ellipta® (Brentford, UK) | 60 L/min | 30 L/min |
| Aclidinium Genuair/PressAir® (Wilmington, DE, USA) | 63 L/min | 30 L/min |
| Tiotropium HandiHaler® (Ridgefield, CT, USA) | 39 L/min | 20 L/min |
| Indacaterol and/or glycopyrrolate Breezhaler/Neohaler® (East Hanover, NJ, USA) | 90 L/min | 30 L/min |
| Demographic | n = 18 (%) |
|---|---|
| Median age, years (range) | 69.5 (46–91) |
| Sex, female | 12 (66%) |
| Inhaler prior to admission | |
| DPI MDI Nebulized | 11 (61%) 6 (33%) 5 (28%) |
| Number of hospitalizations due to COPD exacerbations within past 12 months | |
| 1 2 >3 | 5 (28%) 5 (28%) 8 (44%) |
| mMRC score | |
| 0–1 2 3–4 | 0 (0%) 5 (28%) 13 (72%) |
| GOLD classification | |
| C D | 0 (0%) 18 (100%) |
| Inpatient steroid use | |
| Oral IV | 9 (50%) 10 (56%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borris, J.; Cook, H.; Chae, S.; Walker, K.A.; McPherson, M.L. Evaluation of Peak Inspiratory Flow Rate in Hospitalized Palliative Care Patients with COPD. Pharmacy 2023, 11, 113. https://doi.org/10.3390/pharmacy11040113
Borris J, Cook H, Chae S, Walker KA, McPherson ML. Evaluation of Peak Inspiratory Flow Rate in Hospitalized Palliative Care Patients with COPD. Pharmacy. 2023; 11(4):113. https://doi.org/10.3390/pharmacy11040113
Chicago/Turabian StyleBorris, Joshua, Heather Cook, Sulgi Chae, Kathryn A. Walker, and Mary Lynn McPherson. 2023. "Evaluation of Peak Inspiratory Flow Rate in Hospitalized Palliative Care Patients with COPD" Pharmacy 11, no. 4: 113. https://doi.org/10.3390/pharmacy11040113
APA StyleBorris, J., Cook, H., Chae, S., Walker, K. A., & McPherson, M. L. (2023). Evaluation of Peak Inspiratory Flow Rate in Hospitalized Palliative Care Patients with COPD. Pharmacy, 11(4), 113. https://doi.org/10.3390/pharmacy11040113






