A Robust Reacting Flow Solver with Computational Diagnostics Based on OpenFOAM and Cantera
Abstract
:1. Introduction
2. Methodology
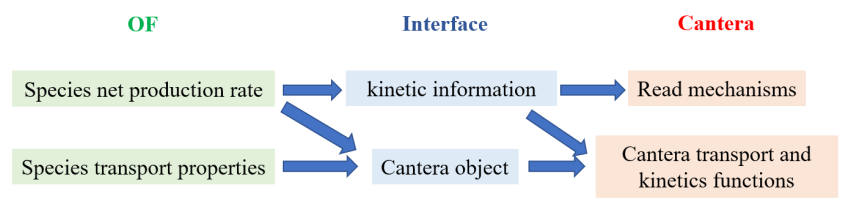
2.1. Transport Models
2.2. Chemistry Models and ODE Solvers
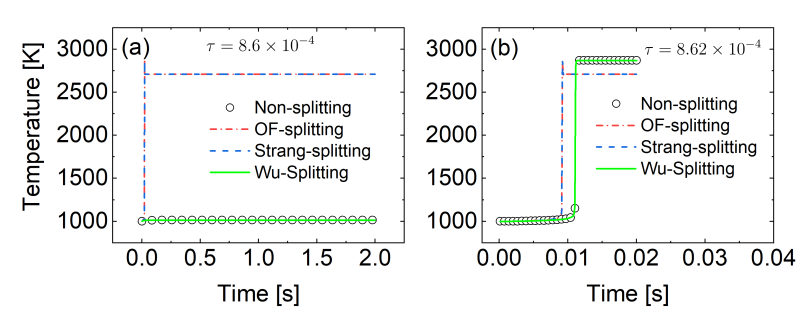
2.3. Splitting Schemes
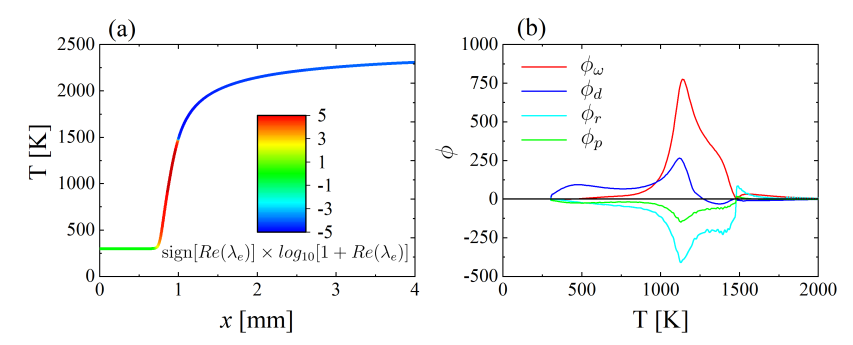
2.4. Conservative Chemical Explosive Mode Analysis (CCEMA)
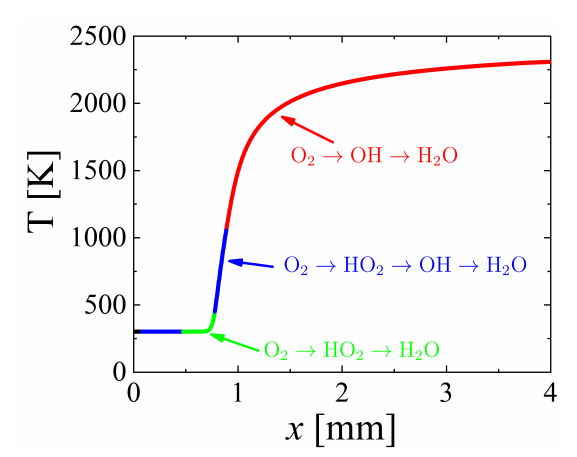
2.5. Global Pathway Analysis (GPA)
3. Results and Analysis
3.1. Zero-Dimensional Auto-Ignition: Chemistry Readers and Models, ODE Solvers
3.2. Zero-Dimensional Perfectly Stirred Reactor (PSR): Splitting Schemes
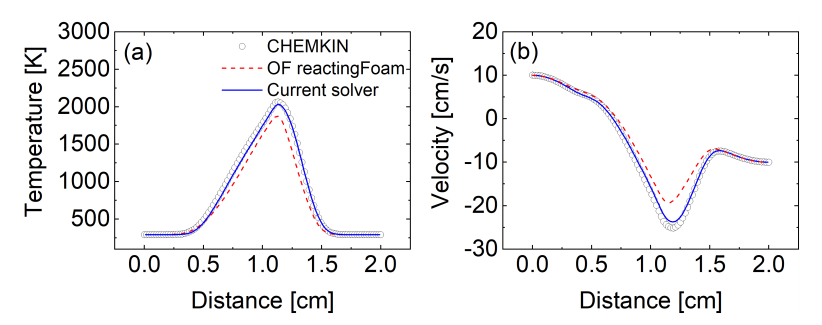
3.3. One-Dimensional Premixed Flame: CCEMA and GPA
3.4. Two-Dimensional Counter-Flow Diffusion Flame: Molecular Transport Models
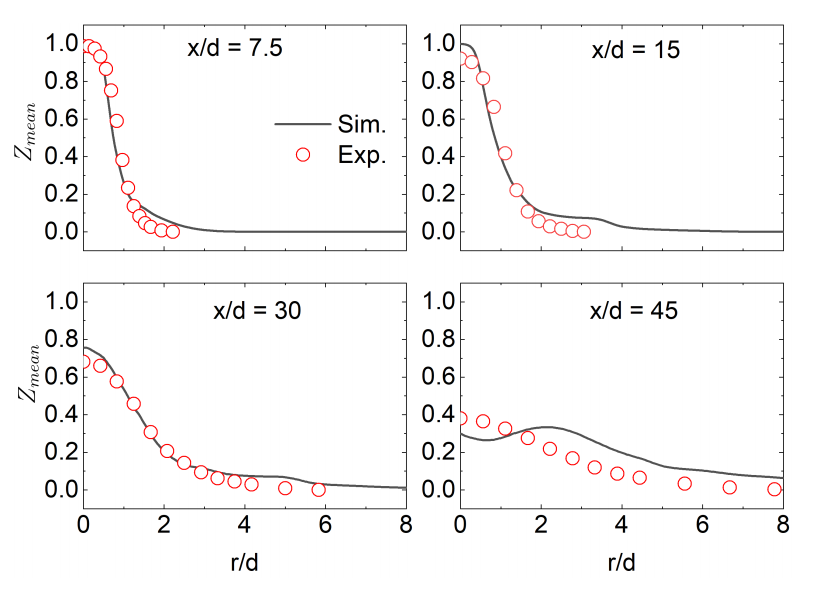
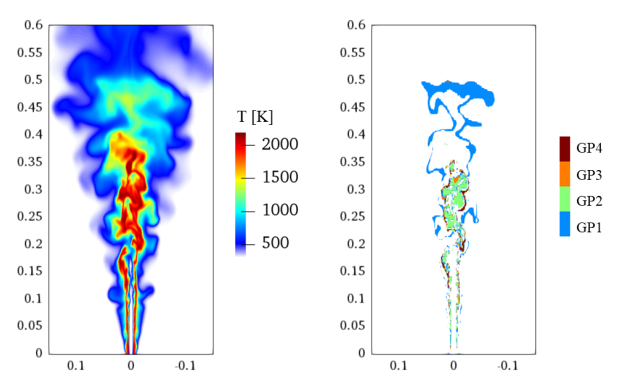
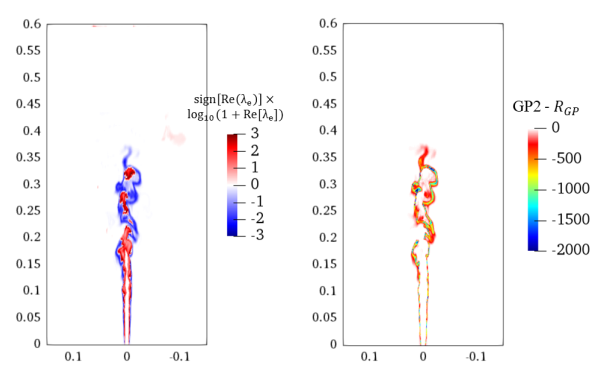
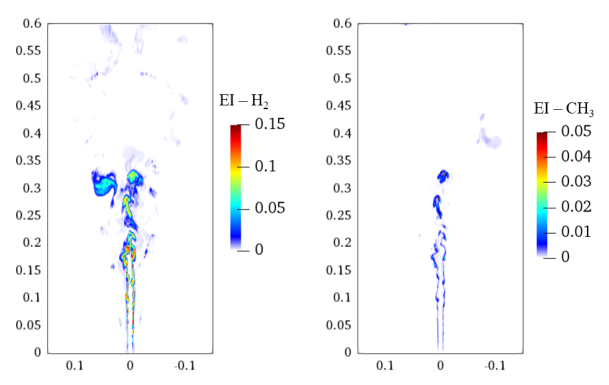
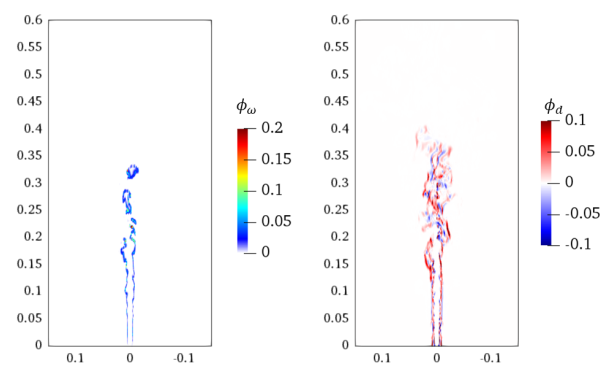
3.5. Three-Dimensional Turbulent Partially Premixed Flame: Integrated CCEMA and GPA
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ihme, M.; Pitsch, H. Prediction of extinction and reignition in nonpremixed turbulent flames using a flamelet/progress variable model: 2. Application in LES of Sandia flames D and E. Combust. Flame 2008, 155, 90–107. [Google Scholar] [CrossRef]
- Jasak, H. OpenFOAM: Open source CFD in research and industry. Int. J. Nav. Archit. Ocean. Eng. 2009, 1, 89–94. [Google Scholar]
- Chen, Z.X.; Swaminathan, N.; Stöhr, M.; Meier, W. Interaction between self-excited oscillations and fuel–air mixing in a dual swirl combustor. Proc. Combust. Inst. 2019, 37, 2325–2333. [Google Scholar] [CrossRef] [Green Version]
- Huang, Z.; Zhao, M.; Xu, Y.; Li, G.; Zhang, H. Eulerian-Lagrangian modelling of detonative combustion in two-phase gas-droplet mixtures with OpenFOAM: Validations and verifications. Fuel 2021, 286, 119402. [Google Scholar] [CrossRef]
- Yang, Q.; Zhao, P.; Ge, H. reactingFoam-SCI: An open source CFD platform for reacting flow simulation. Comput. Fluids 2019, 190, 114–127. [Google Scholar] [CrossRef]
- Dixon-Lewis, G.N. Flame structure and flame reaction kinetics II. Transport phenomena in multicomponent systems. Proc. R. Soc. Lond. Ser. A Math. Phys. Sci. 1968, 307, 111–135. [Google Scholar]
- Bird, R.B.; Stewart, W.E.; Lightfoot, E.N. Transport Phenomena; John Wiley & Sons: Hoboken, NJ, USA, 2007. [Google Scholar]
- Burali, N.; Lapointe, S.; Bobbitt, B.; Blanquart, G.; Xuan, Y. Assessment of the constant non-unity Lewis number assumption in chemically-reacting flows. Combust. Theory Model. 2016, 20, 632–657. [Google Scholar] [CrossRef]
- Zhang, F.; Bonart, H.; Zirwes, T.; Habisreuther, P.; Bockhorn, H.; Zarzalis, N. Direct numerical simulation of chemically reacting flows with the public domain code OpenFOAM. In High Performance Computing in Science and Engineering’14; Springer: Berlin/Heidelberg, Germany, 2015; pp. 221–236. [Google Scholar]
- Kee, R.J.; Rupley, F.M.; Miller, J.A. Chemkin-II: A Fortran Chemical Kinetics Package for the Analysis of Gas-Phase Chemical Kinetics; Technical Report; Sandia National Lab. (SNL-CA): Livermore, CA, USA, 1989. [Google Scholar]
- Troe, J. Theory of Thermal Unimolecular Reactions in the Fall-off Range. I. Strong Collision Rate Constants. Berichte Bunsenges. FüR Phys. Chem. 1983, 87, 169–177. [Google Scholar] [CrossRef]
- Hairer, E. Solving Ordinary Differential Equations II: Stiff and Differential—Algebraic Problems, 1st ed.; Springer Series in Computational Mathematics Ser; Springer: Berlin/Heidelberg, Germany, 1991. [Google Scholar]
- Cohen, S.D.; Hindmarsh, A.C.; Dubois, P.F. CVODE, A Stiff/Nonstiff ODE Solver in C. Comput. Phys. 1996, 10, 138–143. [Google Scholar] [CrossRef] [Green Version]
- Wu, H.; Ma, P.C.; Ihme, M. Efficient time-stepping techniques for simulating turbulent reactive flows with stiff chemistry. Comput. Phys. Commun. 2019, 243, 81–96. [Google Scholar] [CrossRef]
- Lu, Z.; Zhou, H.; Li, S.; Ren, Z.; Lu, T.; Law, C.K. Analysis of operator splitting errors for near-limit flame simulations. J. Comput. Phys. 2017, 335, 578–591. [Google Scholar] [CrossRef]
- Strang, G. On the construction and comparison of difference schemes. SIAM J. Numer. Anal. 1968, 5, 506–517. [Google Scholar] [CrossRef]
- Gao, Y.; Liu, Y.; Ren, Z.; Lu, T. A dynamic adaptive method for hybrid integration of stiff chemistry. Combust. Flame 2015, 162, 287–295. [Google Scholar] [CrossRef]
- Goodwin, D.G.; Speth, R.L.; Moffat, H.K.; Weber, B.W. Cantera: An Object-oriented Software Toolkit for Chemical Kinetics, Thermodynamics, and Transport Processes. Version 2.4.0. 2018. Available online: https://www.cantera.org (accessed on 20 May 2019). [CrossRef]
- Zhou, D.; Zhang, H.; Yang, S. Computational Diagnostics for Reacting Flows with Global Pathway Analysis Aided by Chemical Explosive Mode Analysis. In Proceedings of the AIAA Scitech 2021 Forum, Nashville, TN, USA, 19–21 January 2021; p. 1368. [Google Scholar]
- Wu, B.; Zhao, X.; Xu, C.; Lu, T. Analysis of the chemical states of a bluff-body stabilized premixed flame near blowoff. In Proceedings of the AIAA Scitech 2019 Forum, San Diego, CA, USA, 7–11 January 2019; p. 0185. [Google Scholar]
- Wu, W.; Piao, Y.; Xie, Q.; Ren, Z. Flame diagnostics with a conservative representation of chemical explosive mode analysis. AIAA J. 2019, 57, 1355–1363. [Google Scholar] [CrossRef]
- Gao, X.; Gou, X.; Sun, W. Global Pathway Analysis: A hierarchical framework to understand complex chemical kinetics. Combust. Theory Model. 2018, 23, 1–23. [Google Scholar] [CrossRef]
- Yang, S.; Gao, X.; Sun, W. Global pathway analysis of the extinction and re-ignition of a turbulent non-premixed flame. In Proceedings of the 53rd AIAA/SAE/ASEE Joint Propulsion Conference, Atlanta, GA, USA, 10–12 July 2017; p. 4850. [Google Scholar]
- Gou, J.A.X.; Miller, W.S.; Ju, Y. 2011. Available online: http://engine.princeton.edu (accessed on 20 May 2019).
- Li, Y.; Zhou, C.W.; Somers, K.P.; Zhang, K.; Curran, H.J. The oxidation of 2-butene: A high pressure ignition delay, kinetic modeling study and reactivity comparison with isobutene and 1-butene. Proc. Combust. Inst. 2017, 36, 403–411. [Google Scholar] [CrossRef] [Green Version]
- Cloney, C.T.; Ripley, R.C.; Pegg, M.J.; Amyotte, P.R. Laminar burning velocity and structure of coal dust flames using a unity Lewis number CFD model. Combust. Flame 2018, 190, 87–102. [Google Scholar] [CrossRef]
- Serban, R.; Hindmarsh, A.C. CVODES: An ODE Solver with Sensitivity Analysis Capabilities; Technical report, Technical Report UCRL-JP-200039; Lawrence Livermore National Laboratory: Livermore, CA, USA, 2003. [Google Scholar]
- Hindmarsh, A.C.; Brown, P.N.; Grant, K.E.; Lee, S.L.; Serban, R.; Shumaker, D.E.; Woodward, C.S. SUNDIALS. ACM Trans. Math. Softw. 2005, 31, 363–396. [Google Scholar] [CrossRef]
- Speth, R.L.; Green, W.H.; MacNamara, S.; Strang, G. Balanced splitting and rebalanced splitting. SIAM J. Numer. Anal. 2013, 51, 3084–3105. [Google Scholar] [CrossRef]
- Lu, T.; Yoo, C.S.; Chen, J.; Law, C.K. Three-dimensional direct numerical simulation of a turbulent lifted hydrogen jet flame in heated coflow: A chemical explosive mode analysis. J. Fluid Mech. 2010, 652, 45–64. [Google Scholar] [CrossRef]
- Xu, C.; Park, J.W.; Yoo, C.S.; Chen, J.H.; Lu, T. Identification of premixed flame propagation modes using chemical explosive mode analysis. Proc. Combust. Inst. 2019, 37, 2407–2415. [Google Scholar] [CrossRef]
- Wu, K.; Zhang, P.; Yao, W.; Fan, X. Numerical investigation on flame stabilization in DLR hydrogen supersonic combustor with strut injection. Combust. Sci. Technol. 2017, 189, 2154–2179. [Google Scholar] [CrossRef] [Green Version]
- Shan, R.; Yoo, C.S.; Chen, J.H.; Lu, T. Computational diagnostics for n-heptane flames with chemical explosive mode analysis. Combust. Flame 2012, 159, 3119–3127. [Google Scholar] [CrossRef]
- Xu, C.; Poludnenko, A.Y.; Zhao, X.; Wang, H.; Lu, T. Structure of strongly turbulent premixed n-dodecane–air flames: Direct numerical simulations and chemical explosive mode analysis. Combust. Flame 2019, 209, 27–40. [Google Scholar] [CrossRef]
- Gao, X.; Yang, S.; Sun, W. A global pathway selection algorithm for the reduction of detailed chemical kinetic mechanisms. Combust. Flame 2016, 167, 238–247. [Google Scholar] [CrossRef]
- Zhou, D.; Yang, S. Soot-based Global Pathway Analysis: Soot formation and evolution at elevated pressures in co-flow diffusion flames. Combust. Flame 2021, 227, 255–270. [Google Scholar] [CrossRef]
- Smith, G.P.; Golden, D.M.; Frenklach, M.; Moriarty, N.W.; Eiteneer, B.; Goldenberg, M.; Bowman, C.T.; Hanson, R.K.; Song, S.; Gardiner, W.C., Jr.; et al. GRI-MECH 3.0. 1999. Available online: http://www.me.berkeley.edu/gri_mech/ (accessed on 20 May 2019).
- Ó Conaire, M.; Curran, H.J.; Simmie, J.M.; Pitz, W.J.; Westbrook, C.K. A comprehensive modeling study of hydrogen oxidation. Int. J. Chem. Kinet. 2004, 36, 603–622. [Google Scholar] [CrossRef]
- Li, J.; Zhao, Z.; Kazakov, A.; Dryer, F.L. An updated comprehensive kinetic model of hydrogen combustion. Int. J. Chem. Kinet. 2004, 36, 566–575. [Google Scholar] [CrossRef]
- Barlow, R.; Frank, J. Effects of turbulence on species mass fractions in methane/air jet flames. Symp. (Int.) Combust. 1998, 27, 1087–1095. [Google Scholar] [CrossRef]
- Pitsch, H.; Steiner, H. Large-eddy simulation of a turbulent piloted methane/air diffusion flame (Sandia flame D). Phys. Fluids 2000, 12, 2541–2554. [Google Scholar] [CrossRef]
- Jones, W.; Prasad, V. Large Eddy Simulation of the Sandia Flame Series (D–F) using the Eulerian stochastic field method. Combust. Flame 2010, 157, 1621–1636. [Google Scholar] [CrossRef]
- Yang, S.; Wang, X.; Huo, H.; Sun, W.; Yang, V. An efficient finite-rate chemistry model for a preconditioned compressible flow solver and its comparison with the flamelet/progress-variable model. Combust. Flame 2019, 210, 172–182. [Google Scholar] [CrossRef]
- Smagorinsky, J. General circulation experiments with the primitive equations: I. The basic experiment. Mon. Weather. Rev. 1963, 91, 99–164. [Google Scholar] [CrossRef]
- Yifan, D.; Zhixun, X.; Likun, M.; Zhenbing, L.; Huang, X.; Xiong, D. LES of the Sandia flame series DF using the Eulerian stochastic field method coupled with tabulated chemistry. Chin. J. Aeronaut. 2020, 33, 116–133. [Google Scholar]
- Golovitchev, V.I.; Nordin, N.; Jarnicki, R.; Chomiak, J. 3-D diesel spray simulations using a new detailed chemistry turbulent combustion model. SAE Trans. 2000, 109, 1391–1405. [Google Scholar]
- Mathey, F.; Cokljat, D.; Bertoglio, J.P.; Sergent, E. Assessment of the vortex method for large eddy simulation inlet conditions. Prog. Comput. Fluid Dyn. Int. J. 2006, 6, 58–67. [Google Scholar] [CrossRef]




| Fuel Jet | Piloted Flame | Co-Flow | |
|---|---|---|---|
| Composition | 25% CH4/75% air (by volume) | CH4/air equilibrium mixture () | Air |
| Inner diameter (mm) | 7.2 | 7.7 | 18.9 |
| Outer diameter (mm) | 7.7 | 18.2 | N.A. |
| Bulk velocity (m/s) | 49.6 | 11.4 | 0.9 |
| Temperature (K) | 294 | 1880 | 291 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, D.; Zhang, H.; Yang, S. A Robust Reacting Flow Solver with Computational Diagnostics Based on OpenFOAM and Cantera. Aerospace 2022, 9, 102. https://doi.org/10.3390/aerospace9020102
Zhou D, Zhang H, Yang S. A Robust Reacting Flow Solver with Computational Diagnostics Based on OpenFOAM and Cantera. Aerospace. 2022; 9(2):102. https://doi.org/10.3390/aerospace9020102
Chicago/Turabian StyleZhou, Dezhi, Hongyuan Zhang, and Suo Yang. 2022. "A Robust Reacting Flow Solver with Computational Diagnostics Based on OpenFOAM and Cantera" Aerospace 9, no. 2: 102. https://doi.org/10.3390/aerospace9020102
APA StyleZhou, D., Zhang, H., & Yang, S. (2022). A Robust Reacting Flow Solver with Computational Diagnostics Based on OpenFOAM and Cantera. Aerospace, 9(2), 102. https://doi.org/10.3390/aerospace9020102








