A Case Study of Ozone Diurnal Variation in the Convective Boundary Layer in the Southeastern United States Using Multiple Observations and Large-Eddy Simulation
Abstract
1. Introduction
2. Measurements and Model
2.1. Measurements
2.2. Dutch Atmospheric Large-Eddy Simulation (DALES)
2.3. Methodology
2.3.1. Physics Settings
2.3.2. Chemistry Settings
3. Results and Discussions
3.1. Meteorological Analysis
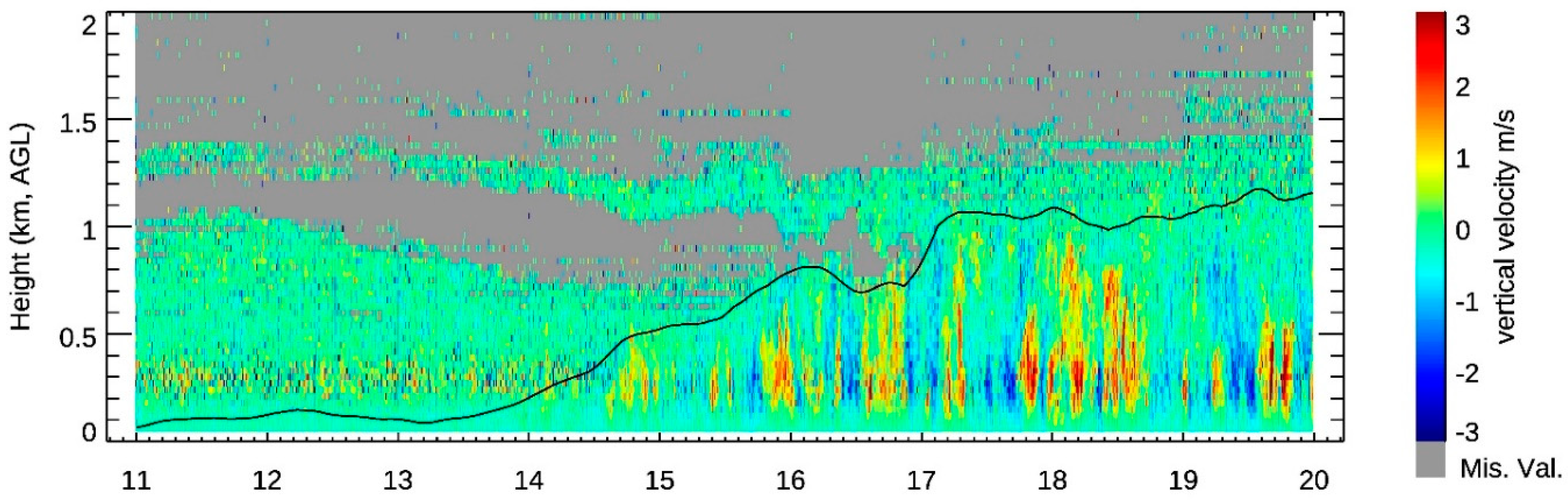
3.2. The CBL Height
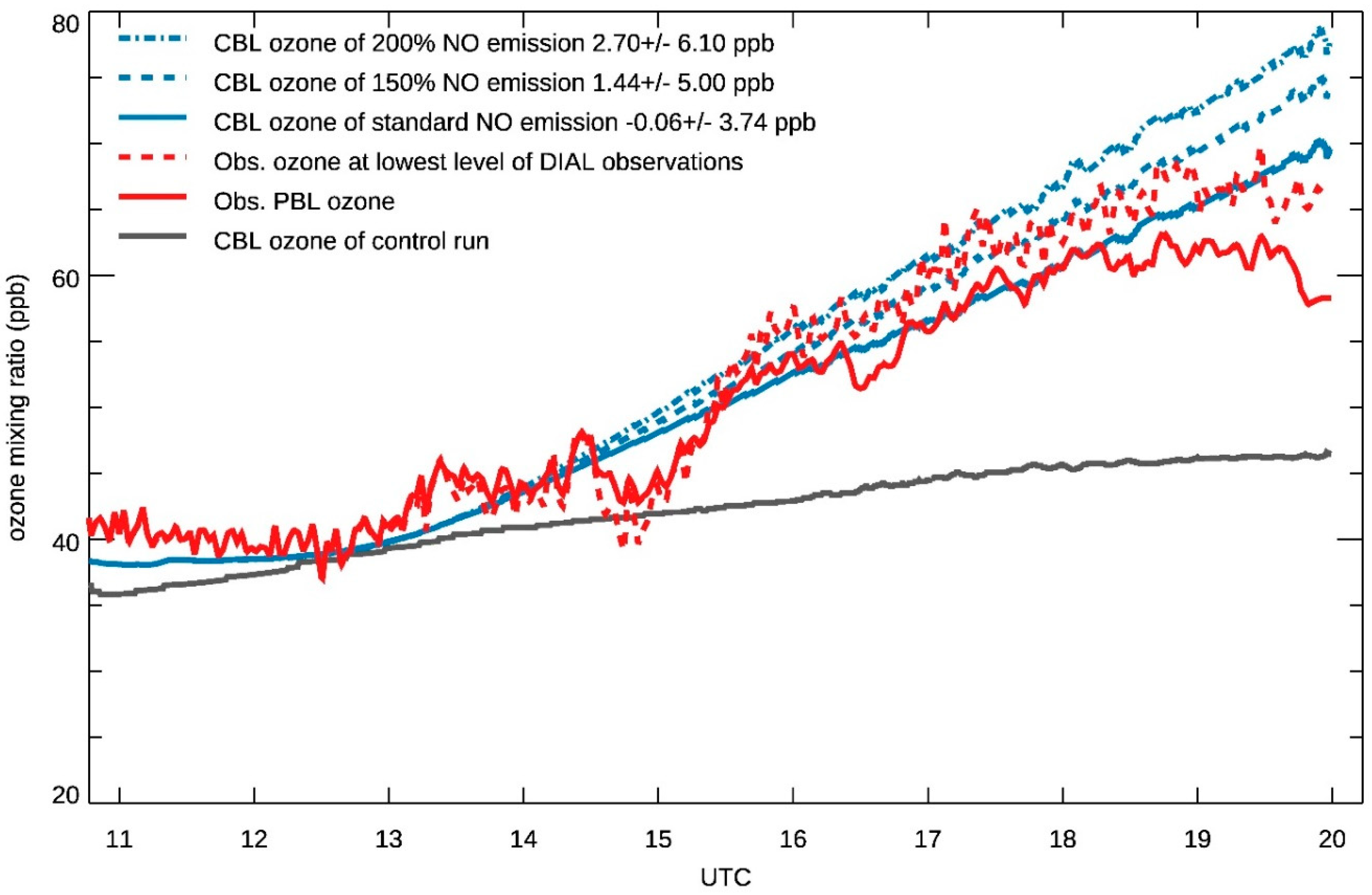
3.3. Ozone Variation in the CBL
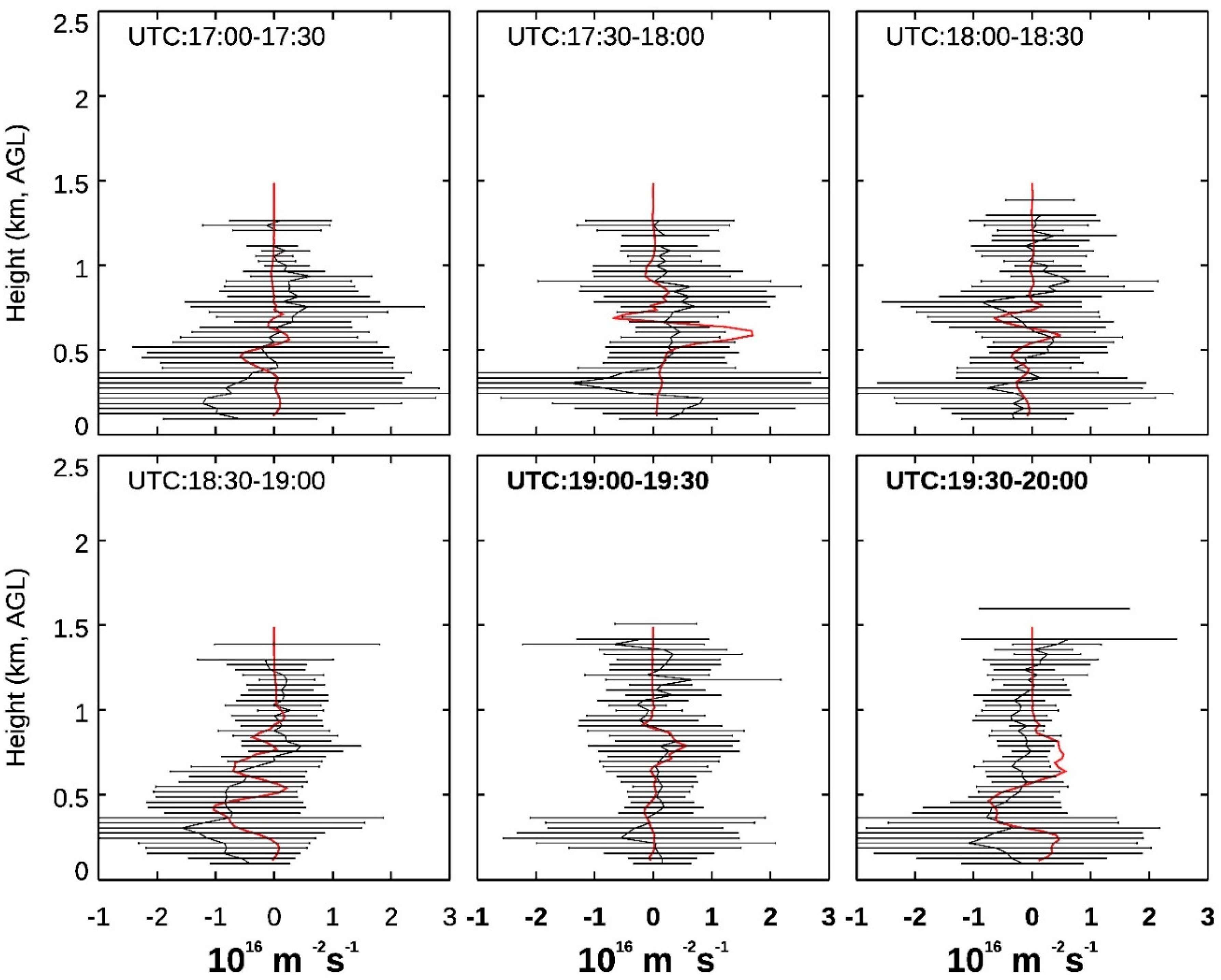
3.4. Ozone Transport within the CBL
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jenkin, M.E.; Clemitshaw, K.C. Ozone and other secondary photochemical pollutants: chemical processes governing their formation in the planetary boundary layer. Atmos. Environ. 2000, 34, 2499–2527. [Google Scholar] [CrossRef]
- EPA (Ed.) EPA’s Reports on the Environment; United States Environmental Protection Agency: Washington, DC, USA, 2008; p. 366.
- Kuang, S.; Newchurch, M.J.; Burris, J.; Wang, L.; Buckley, P.I.; Johnson, S.; Knupp, K.; Huang, G.; Phillips, D.; Cantrell, W. Nocturnal ozone enhancement in the lower troposphere observed by lidar. Atmos. Environ. 2011, 45, 6078–6084. [Google Scholar] [CrossRef]
- Huang, G.; Newchurch, M.J.; Kuang, S.; Buckley, P.I.; Cantrell, W.; Wang, L. Definition and determination of ozone laminae using Continuous Wavelet Transform (CWT) analysis. Atmos. Environ. 2015, 104, 125–131. [Google Scholar] [CrossRef]
- Kuang, S.; Newchurch, M.J.; Burris, J.; Wang, L.; Knupp, K.; Huang, G. Stratosphere-to-troposphere transport revealed by ground-based lidar and ozonesonde at a midlatitude site. J. Geophys. Res.-Atmos. 2012, 117. [Google Scholar] [CrossRef]
- Tong, N.O.; Leung, D.C.; Liu, C.-H. A Review on Ozone Evolution and Its Relationship with Boundary Layer Characteristics in Urban Environments. Water Air Soil Pollut. 2011, 214, 13–36. [Google Scholar] [CrossRef]
- Jaffe, D.A.; Wigder, N.L. Ozone production from wildfires: A critical review. Atmos. Environ. 2012, 51, 1–10. [Google Scholar] [CrossRef]
- Langford, A.O.; Brioude, J.; Cooper, O.R.; Senff, C.J.; Alvarez, R.J., II; Hardesty, R.M.; Johnson, B.J.; Oltmans, S.J. Stratospheric influence on surface ozone in the Los Angeles area during late spring and early summer of 2010. J. Geophys. Res. 2012, 117, D00V06. [Google Scholar] [CrossRef]
- Duncan, B.N.; Yoshida, Y.; Olson, J.R.; Sillman, S.; Martin, R.V.; Lamsal, L.; Hu, Y.; Pickering, K.E.; Retscher, C.; Allen, D.J.; et al. Application of OMI observations to a space-based indicator of NOx and VOC controls on surface ozone formation. Atmos. Environ. 2010, 44, 2213–2223. [Google Scholar] [CrossRef]
- Sillman, S.; Al-Wali, K.I.; Marsik, F.J.; Nowacki, P.; Samson, P.J.; Rodgers, M.O.; Garland, L.J.; Martinez, J.E.; Stoneking, C.; Imhoff, R.; et al. Photochemistry of ozone formation in Atlanta, GA-Models and measurements. Atmos. Environ. 1995, 29, 3055–3066. [Google Scholar] [CrossRef]
- Blanchard, C.L.; Hidy, G.M.; Tanenbaum, S. Ozone in the southeastern United States: An observation-based model using measurements from the SEARCH network. Atmos. Environ. 2014, 88, 192–200. [Google Scholar] [CrossRef]
- Hidy, G.M. Ozone process insights from field experiments—Part I: overview. Atmos. Environ. 2000, 34, 2001–2022. [Google Scholar] [CrossRef]
- Zhang, Y.; Bocquet, M.; Mallet, V.; Seigneur, C.; Baklanov, A. Real-time air quality forecasting, part II: State of the science, current research needs, and future prospects. Atmos. Environ. 2012, 60, 656–676. [Google Scholar] [CrossRef]
- Yegorova, E.A.; Allen, D.J.; Loughner, C.P.; Pickering, K.E.; Dickerson, R.R. Characterization of an eastern U.S. severe air pollution episode using WRF/Chem. J. Geophys. Res.-Atmos. 2011, 116, D17306. [Google Scholar] [CrossRef]
- Castellanos, P.; Marufu, L.T.; Doddridge, B.G.; Taubman, B.F.; Schwab, J.J.; Hains, J.C.; Ehrman, S.H.; Dickerson, R.R. Ozone, oxides of nitrogen, and carbon monoxide during pollution events over the eastern United States: An evaluation of emissions and vertical mixing. J. Geophys. Res.-Atmos. 2011, 116, D16307. [Google Scholar] [CrossRef]
- So, K.L.; Wang, T. On the local and regional influence on ground-level ozone concentrations in Hong Kong. Environ. Pollut. 2003, 123, 307–317. [Google Scholar] [CrossRef]
- Shao, M.; Zhang, Y.; Zeng, L.; Tang, X.; Zhang, J.; Zhong, L.; Wang, B. Ground-level ozone in the Pearl River Delta and the roles of VOC and NOx in its production. J. Environ. Manag. 2009, 90, 512–518. [Google Scholar] [CrossRef]
- Chance, K.; Liu, X.; Suleiman, R.M.; Flittner, D.E.; Al-Saadi, J.; Janz, S.J. Tropospheric Emissions: Monitoring of Pollution (TEMPO); SPIE: Bellingham, WA, USA, 2013; Volume 8866. [Google Scholar]
- Zoogman, P.; Liu, X.; Suleiman, R.M.; Pennington, W.F.; Flittner, D.E.; Al-Saadi, J.A.; Hilton, B.B.; Nicks, D.K.; Newchurch, M.J.; Carr, J.L.; et al. Tropospheric emissions: Monitoring of pollution (TEMPO). J. Quant. Spectrosc. Radiat. Transf. 2017, 186, 17–39. [Google Scholar] [CrossRef]
- Martins, D.K.; Stauffer, R.M.; Thompson, A.M.; Halliday, H.S.; Kollonige, D.; Joseph, E.; Weinheimer, A.J. Ozone correlations between mid-tropospheric partial columns and the near-surface at two mid-atlantic sites during the DISCOVER-AQ campaign in July 2011. J. Atmos. Chem. 2013, 1–19. [Google Scholar] [CrossRef]
- Goldberg, D.L.; Loughner, C.P.; Tzortziou, M.; Stehr, J.W.; Pickering, K.E.; Marufu, L.T.; Dickerson, R.R. Higher surface ozone concentrations over the Chesapeake Bay than over the adjacent land: Observations and models from the DISCOVER-AQ and CBODAQ campaigns. Atmos. Environ. 2014, 84, 9–19. [Google Scholar] [CrossRef]
- Peterson, D.A.; Hyer, E.J.; Campbell, J.R.; Fromm, M.D.; Hair, J.W.; Butler, C.F.; Fenn, M.A. The 2013 Rim Fire: Implications for Predicting Extreme Fire Spread, Pyroconvection, and Smoke Emissions. Bull. Am. Meteorol. Soc. 2014. [Google Scholar] [CrossRef]
- Duncan, B.N.; Prados, A.I.; Lamsal, L.N.; Liu, Y.; Streets, D.G.; Gupta, P.; Hilsenrath, E.; Kahn, R.A.; Nielsen, J.E.; Beyersdorf, A.J.; et al. Satellite data of atmospheric pollution for U.S. air quality applications: Examples of applications, summary of data end-user resources, answers to FAQs, and common mistakes to avoid. Atmos. Environ. 2014, 94, 647–662. [Google Scholar] [CrossRef]
- Fishman, J.; Iraci, L.T.; Al-Saadi, J.; Chance, K.; Chavez, F.; Chin, M.; Coble, P.; Davis, C.; DiGiacomo, P.M.; Edwards, D.; et al. The United States’ Next Generation of Atmospheric Composition and Coastal Ecosystem Measurements: NASA’s Geostationary Coastal and Air Pollution Events (GEO-CAPE) Mission. Bull. Am. Meteorol. Soc. 2012, 93, 1547–1566. [Google Scholar] [CrossRef]
- van Stratum, B.J.H.; Vilà-Guerau de Arellano, J.; Ouwersloot, H.G.; van den Dries, K.; van Laar, T.W.; Martinez, M.; Lelieveld, J.; Diesch, J.M.; Drewnick, F.; Fischer, H.; et al. Case study of the diurnal variability of chemically active species with respect to boundary layer dynamics during DOMINO. Atmos. Chem. Phys. 2012, 12, 5329–5341. [Google Scholar] [CrossRef]
- Chamecki, M.; Meneveau, C.; Parlange, M.B. Large eddy simulation of pollen transport in the atmospheric boundary layer. J. Aerosol Sci. 2009, 40, 241–255. [Google Scholar] [CrossRef]
- Ouwersloot, H.G.; Vilà-Guerau de Arellano, J.; Nölscher, A.C.; Krol, M.C.; Ganzeveld, L.N.; Breitenberger, C.; Mammarella, I.; Williams, J.; Lelieveld, J. Characterization of a boreal convective boundary layer and its impact on atmospheric chemistry during HUMPPA-COPEC-2010. Atmos. Chem. Phys. 2012, 12, 9335–9353. [Google Scholar] [CrossRef]
- Langford, A.O.; Tucker, S.C.; Senff, C.J.; Banta, R.M.; Brewer, W.A.; Alvarez, R.J., II; Hardesty, R.M.; Lerner, B.M.; Williams, E.J. Convective venting and surface ozone in Houston during TexAQS 2006. J. Geophys. Res. 2010, 115, D16305. [Google Scholar] [CrossRef]
- Senff, C.J.; Alvarez, R.J., II; Hardesty, R.M.; Banta, R.M.; Langford, A.O. Airborne lidar measurements of ozone flux downwind of Houston and Dallas. J. Geophys. Res. 2010, 115, D20307. [Google Scholar] [CrossRef]
- Kuang, S.; Newchurch, M.J.; Johnson, M.S.; Wang, L.; Burris, J.; Pierce, R.B.; Eloranta, E.W.; Pollack, I.B.; Graus, M.; de Gouw, J.; et al. Summertime tropospheric ozone enhancement associated with a cold front passage due to stratosphere-to-troposphere transport and biomass burning: simultaneous ground-based lidar and airborne measurements. J. Geophys. Res.-Atmos. 2017. [Google Scholar] [CrossRef]
- Kuang, S.; Newchurch, M.J.; Burris, J.; Liu, X. Ground-based lidar for atmospheric boundary layer ozone measurements. Appl. Opt. 2013, 52, 3557–3566. [Google Scholar] [CrossRef]
- Kuang, S.; Burris, J.F.; Newchurch, M.J.; Johnson, S.; Long, S. Differential Absorption Lidar to Measure Subhourly Variation of Tropospheric Ozone Profiles. IEEE Trans. Geosci. Remote Sens. 2011, 49, 557–571. [Google Scholar] [CrossRef]
- Karan, H.; Knupp, K. Mobile Integrated Profiler System (MIPS) Observations of Low-Level Convergent Boundaries during IHOP. Mon. Weather Rev. 2006, 134, 92–112. [Google Scholar] [CrossRef]
- Wingo, S.M.; Knupp, K.R. Multi-platform Observations Characterizing the Afternoon-to-Evening Transition of the Planetary Boundary Layer in Northern Alabama, USA. Bound. -Layer Meteorol. 2014, 155, 29–53. [Google Scholar] [CrossRef]
- Busse, J.; Knupp, K. Observed Characteristics of the Afternoon–Evening Boundary Layer Transition Based on Sodar and Surface Data. J. Appl. Meteorol. Climatol. 2012, 51, 571–582. [Google Scholar] [CrossRef]
- Knupp, K.R.; Coleman, T.; Phillips, D.; Ware, R.; Cimini, D.; Vandenberghe, F.; Vivekanandan, J.; Westwater, E. Ground-Based Passive Microwave Profiling during Dynamic Weather Conditions. J. Atmos. Ocean. Technol. 2009, 26, 1057–1073. [Google Scholar] [CrossRef]
- Heus, T.; van Heerwaarden, C.C.; Jonker, H.J.J.; Pier Siebesma, A.; Axelsen, S.; van den Dries, K.; Geoffroy, O.; Moene, A.F.; Pino, D.; de Roode, S.R.; et al. Formulation of the Dutch Atmospheric Large-Eddy Simulation (DALES) and overview of its applications. Geosci. Model Dev. 2010, 3, 415–444. [Google Scholar] [CrossRef]
- Böing, S.J.; Jonker, H.J.J.; Siebesma, A.P.; Grabowski, W.W. Influence of the Subcloud Layer on the Development of a Deep Convective Ensemble. J. Atmos. Sci. 2012, 69, 2682–2698. [Google Scholar] [CrossRef]
- Vilà-Guerau de Arellano, J.; Kim, S.W.; Barth, M.C.; Patton, E.G. Transport and chemical transformations influenced by shallow cumulus over land. Atmos. Chem. Phys. 2005, 5, 3219–3231. [Google Scholar] [CrossRef]
- Ouwersloot, H.G. The Impact of Dynamic Processes on Chemistry in Atmospheric Boundary Layers over Tropical and Boreal Forest; Wageningen University: Wageningen, The Netherlands, 2013. [Google Scholar]
- Aan de Brugh, J.M.J.; Ouwersloot, H.G.; Vilà-Guerau de Arellano, J.; Krol, M.C. A large-eddy simulation of the phase transition of ammonium nitrate in a convective boundary layer. J. Geophys. Res.-Atmos. 2013, 118, 826–836. [Google Scholar] [CrossRef]
- Ouwersloot, H.G.; Vilà-Guerau de Arellano, J.; van Heerwaarden, C.C.; Ganzeveld, L.N.; Krol, M.C.; Lelieveld, J. On the segregation of chemical species in a clear boundary layer over heterogeneous land surfaces. Atmos. Chem. Phys. 2011, 11, 10681–10704. [Google Scholar] [CrossRef]
- Vilà-Guerau de Arellano, J.; van den Dries, K.; Pino, D. On inferring isoprene emission surface flux from atmospheric boundary layer concentration measurements. Atmos. Chem. Phys. 2009, 9, 3629–3640. [Google Scholar] [CrossRef]
- Blay-Carreras, E.; Pino, D.; Vilà-Guerau de Arellano, J.; van de Boer, A.; De Coster, O.; Darbieu, C.; Hartogensis, O.; Lohou, F.; Lothon, M.; Pietersen, H. Role of the residual layer and large-scale subsidence on the development and evolution of the convective boundary layer. Atmos. Chem. Phys. 2014, 14, 4515–4530. [Google Scholar] [CrossRef]
- Dlugi, R.; Berger, M.; Zelger, M.; Hofzumahaus, A.; Rohrer, F.; Holland, F.; Lu, K.; Kramm, G. The balances of mixing ratios and segregation intensity: A case study from the field (ECHO 2003). Atmos. Chem. Phys. 2014, 14, 10333–10362. [Google Scholar] [CrossRef][Green Version]
- Vilà-Guerau de Arellano, J.; Patton, E.G.; Karl, T.; van den Dries, K.; Barth, M.C.; Orlando, J.J. The role of boundary layer dynamics on the diurnal evolution of isoprene and the hydroxyl radical over tropical forests. J. Geophys. Res. 2011, 116, D07304. [Google Scholar] [CrossRef]
- Wesely, M.L.; Hicks, B.B. A review of the current status of knowledge on dry deposition. Atmos. Environ. 2000, 34, 2261–2282. [Google Scholar] [CrossRef]
- Seinfeld, J.H.; Pandis, S.N. Atmospheric Chemistry and Physics: From Air Pollution to Climate Change, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2006. [Google Scholar]
- Biazar, A.P. The Role of Natural Nitrogen Oxides in Ozone Production in the Southeastern Environment. Ph.D. Thesis, The University of Alabama in Huntsville, Ann Arbor, MI, USA, 1995. [Google Scholar]
- Stull, R.B. An Introduction to Boundary Layer Meteorology; Springer: Berlin, Germany, 1988; Volume 13. [Google Scholar]
- Dentener, F.; Vet, R.; Dennis, R.L.; Du, E.; Kulshrestha, U.C.; Galy-Lacaux, C. Progress in Monitoring and Modelling Estimates of Nitrogen Deposition at Local, Regional and Global Scales. In Nitrogen Deposition, Critical Loads and Biodiversity; Sutton, M.A., Mason, K.E., Sheppard, L.J., Sverdrup, H., Haeuber, R., Hicks, W.K., Eds.; Springer: Dordrecht, The Netherlands, 2014; pp. 7–22. [Google Scholar]
- Erisman, J.W.; Van Pul, A.; Wyers, P. Parametrization of surface resistance for the quantification of atmospheric deposition of acidifying pollutants and ozone. Atmos. Environ. 1994, 28, 2595–2607. [Google Scholar] [CrossRef]
- Ganzeveld, L.; Lelieveld, J. Dry deposition parameterization in a chemistry general circulation model and its influence on the distribution of reactive trace gases. J. Geophys. Res. 1995, 100, 20999. [Google Scholar] [CrossRef]
- Pleim, J.E.; Xiu, A.; Finkelstein, P.L.; Otte, T.L. A Coupled Land-Surface and Dry Deposition Model and Comparison to Field Measurements of Surface Heat, Moisture, and Ozone Fluxes. Water Air Soil Pollut. Focus 2001, 1, 243–252. [Google Scholar] [CrossRef]
- Zhang, L.; Moran, M.D.; Makar, P.A.; Brook, J.R.; Gong, S. Modelling gaseous dry deposition in AURAMS: A unified regional air-quality modelling system. Atmos. Environ. 2002, 36, 537–560. [Google Scholar] [CrossRef]
- Zhang, L.; Brook, J.R.; Vet, R. A revised parameterization for gaseous dry deposition in air-quality models. Atmos. Chem. Phys. 2003, 3, 2067–2082. [Google Scholar] [CrossRef]
- Sickles Ii, J.E.; Shadwick, D.S. Air quality and atmospheric deposition in the eastern US: 20 years of change. Atmos. Chem. Phys. 2015, 15, 173–197. [Google Scholar] [CrossRef]
- Yi, C.; Davis, K.J.; Berger, B.W.; Bakwin, P.S. Long-Term Observations of the Dynamics of the Continental Planetary Boundary Layer. J. Atmos. Sci. 2001, 58, 1288–1299. [Google Scholar] [CrossRef]
- Geron, C.D.; Nie, D.; Arnts, R.R.; Sharkey, T.D.; Singsaas, E.L.; Vanderveer, P.J.; Guenther, A.; Sickles, J.E.; Kleindienst, T.E. Biogenic isoprene emission: Model evaluation in a southeastern United States bottomland deciduous forest. J. Geophys. Res. 1997, 102, 18889. [Google Scholar] [CrossRef]
- Kesselmeier, J.; Staudt, M. Biogenic volatile organic compounds (VOC): An overview on emission, physiology and ecology. J. Atmos. Chem. 1999, 33, 23–88. [Google Scholar] [CrossRef]
- Sullivan, L.J.; Moore, T.C.; Aneja, V.P.; Robarge, W.P.; Pierce, T.E.; Geron, C.; Gay, B. Environmental variables controlling nitric oxide: emissions from agricultural soils in the southeast united states. Atmos. Environ. 1996, 30, 3573–3582. [Google Scholar] [CrossRef]
- Solomon, P.; Cowling, E.; Hidy, G.; Furiness, C. Comparison of scientific findings from major ozone field studies in North America and Europe. Atmos. Environ. 2000, 34, 1885–1920. [Google Scholar] [CrossRef]
- Davis, K.J.; Gamage, N.; Hagelberg, C.R.; Kiemle, C.; Lenschow, D.H.; Sullivan, P.P. An Objective Method for Deriving Atmospheric Structure from Airborne Lidar Observations. J. Atmos. Ocean. Technol. 2000, 17, 1455–1468. [Google Scholar] [CrossRef]
- Senff, C.J.; Bösenberg, J.; Peters, G.; Schaberl, T. Remote Sensing of Turbulent Ozone Fluxes and the Ozone Budget in the Convective Boundary Layer with DIAL and Radar-RASS: A Case Study. Beitr. Phys. Atmos. 1996, 69, 161–176. [Google Scholar]
- Patton, E.G.; Sullivan, P.P.; Moeng, C.-H. The Influence of Idealized Heterogeneity on Wet and Dry Planetary Boundary Layers Coupled to the Land Surface. J. Atmos. Sci. 2005, 62, 2078–2097. [Google Scholar] [CrossRef]
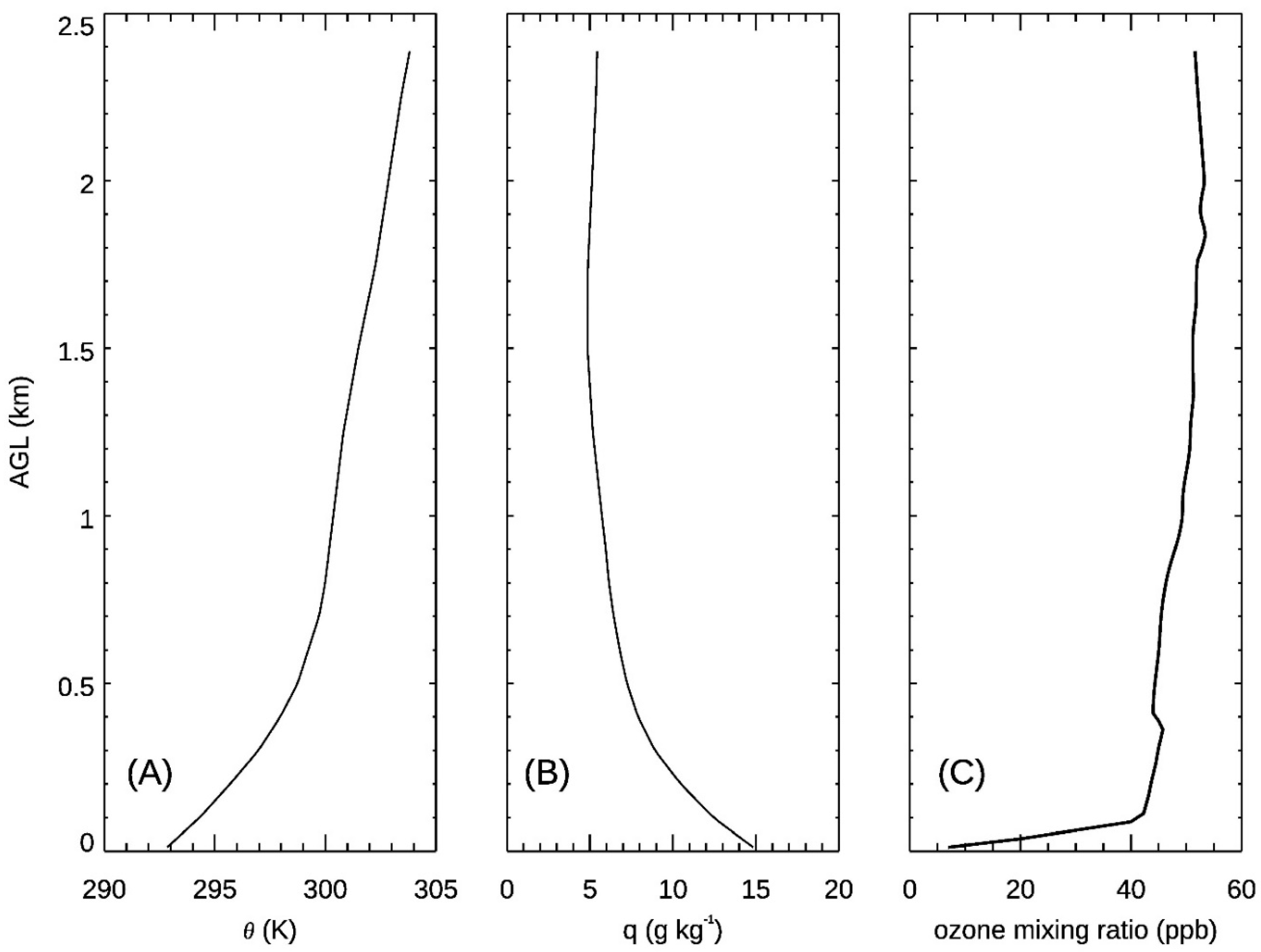
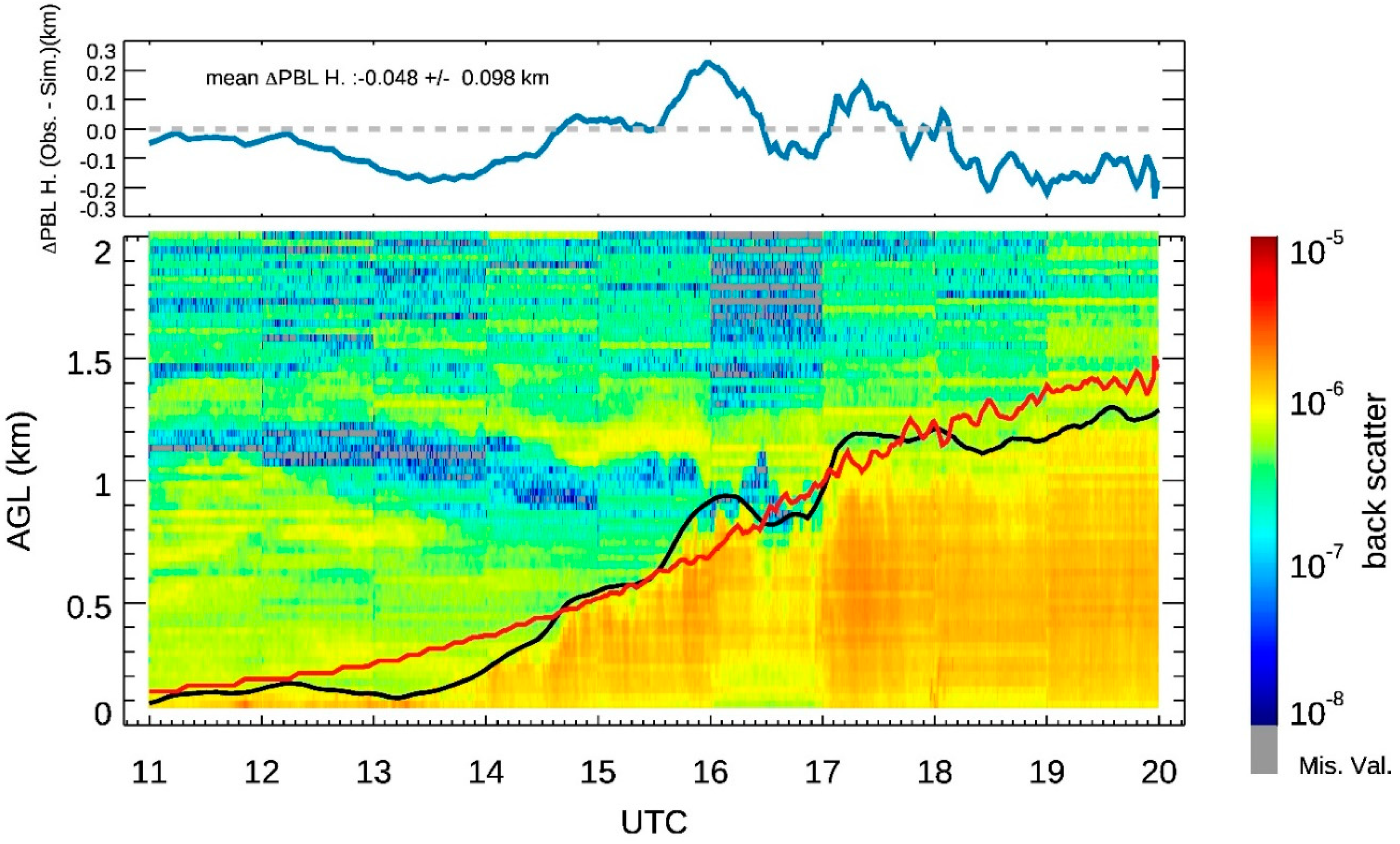
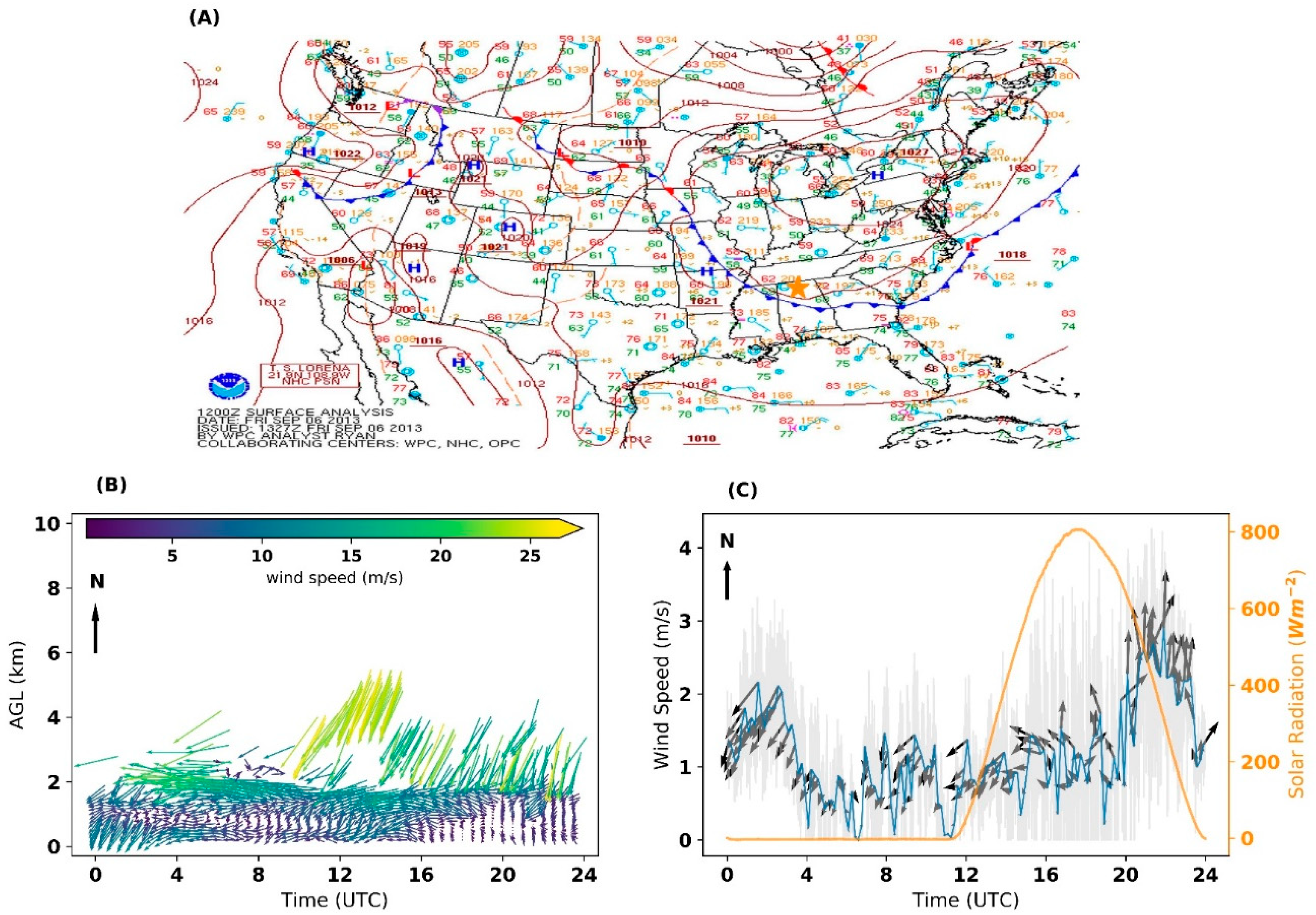

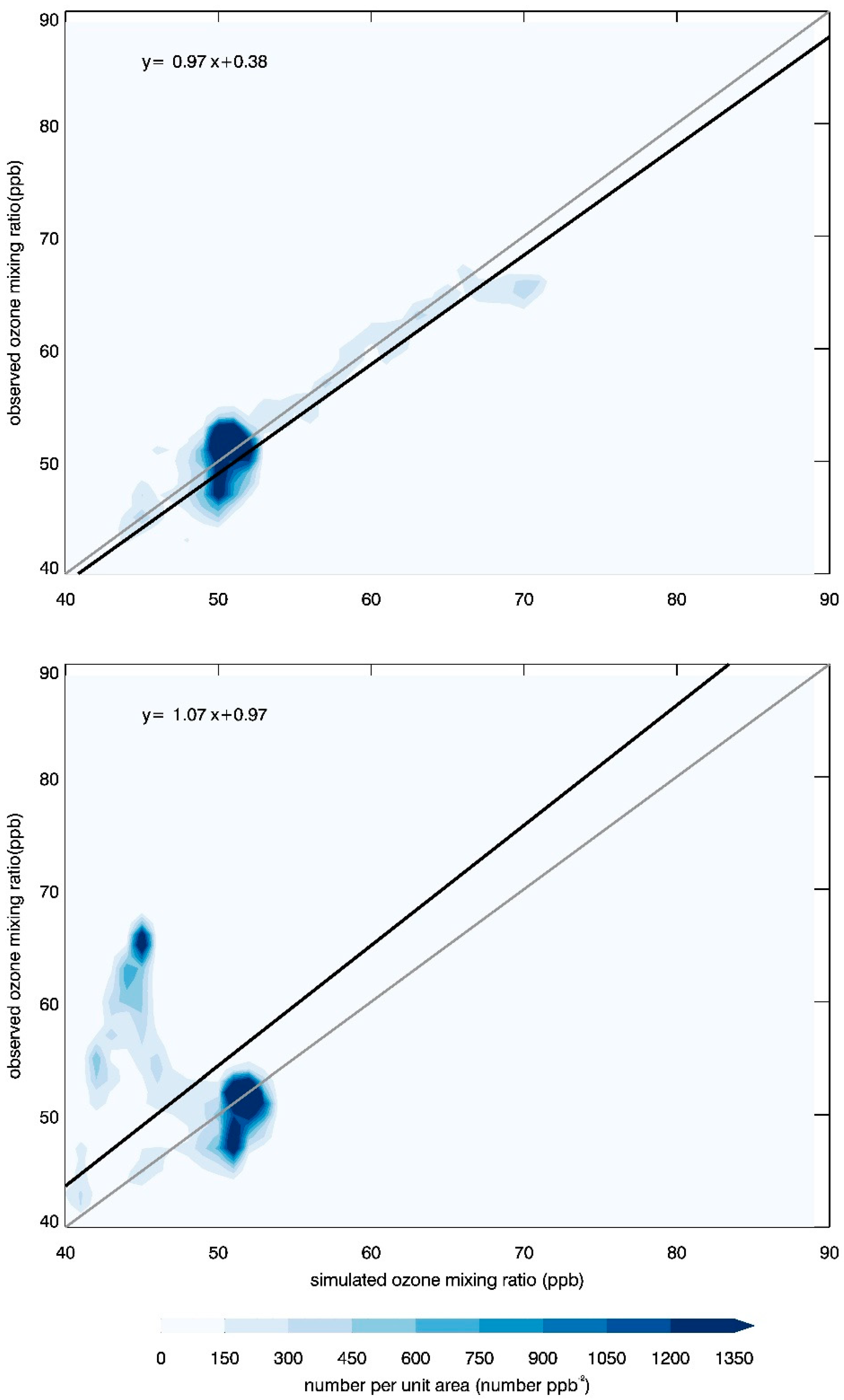
| Instrument | Measurements | Vertical Range | Vertical Resolution | Temporal Resolution |
|---|---|---|---|---|
| 915 MHz wind profiler | Vertical motion, | 0.19–4 km | 60 or 106 m | 60 s |
| horizontal wind, | ||||
| spectral width | ||||
| Ceilometer | Backscatter, | 0.3–10+ km | 30 m | 15 s |
| cloud base | ||||
| MPR | Temperature, | Surface–10 km | 100 m from surface to 1 km | 1–14 min |
| integrated water vapor | 250 m above 1 km | |||
| Ozone differential absorption lidar (DIAL) | Ozone | Surface–10+ km | 30 m (sampling resolution) | 2–10 min |
| compact wind and aerosol lidar (CWAL) | Aerosol, wind velocity | 0.75–10 km | 30 m | 0.1–30 s |
| Surface | Temperature, wind velocity, | 2 m | N/A | 5 s |
| solar radiation |
| Reaction Number | Reaction | Reaction Rate |
|---|---|---|
| R1 | O3 + hv → O(1D) + O2 | |
| R2 | O(1D) + H2O → 2OH | |
| R3 | O(1D) + N2 → O3 + PRODUCT | |
| R4 | O(1D) + O2 →O3 + PRODUCT | |
| R5 | NO2 + hv → NO + O3 | |
| R6 | CH2O + hv → HO2 | |
| R7 | OH + CO → HO2 + CO2 | |
| R8 | OH + CH4 → CH3O2 | |
| R9 | OH + ISO → RO2 | |
| R10 | OH + MVK → HO2 + CH2O | |
| R11 | HO2 + NO → OH + NO2 | |
| R12 | CH3O2 + NO → HO2 + NO2 + CH2O | |
| R13 | RO2 + NO → HO2 + NO2 + CH2O + MVK | |
| R14 | OH + CH2O + O2 → HO2 + CO + H2O | |
| R15 | HO2 + HO2 → H2O2 + O2 | k* |
| R16 | CH3O2 + HO2 → PRODUCT | |
| R17 | RO2 + HO2 → nOH product | |
| R18 | OH + NO2→ HNO3 | |
| R19 | NO + O3 → NO2 + O2 | |
| k* = (k1 + k2)∙k3. | ||
| k1 = ; k2 = ; k3 = | ||
| Mixing Ratio (ppb, z < 200 m) | Mixing Ratio (ppb, z > 200 m) | |
|---|---|---|
| Ozone | as Figure 1 | as Figure 1 |
| NO | 0 | 0 |
| NO2 | 1 | 0 |
| ISO | 2 | 0 |
| HO2 | 0 | 0 |
| OH | 0 | 0 |
| MVK | 1.3 | 1.3 |
| CH4 | 1724 | 1724 |
| CO | 124 | 124 |
| Name | Dynamics | Chemistry | Dry Depo. |
|---|---|---|---|
| Control | on | off | off |
| Std. NO emis. | on | on | on |
| 150% NO emis. | on | on | on |
| 200% NO emis. | on | on | on |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, G.; Newchurch, M.J.; Kuang, S.; Ouwersloot, H.G. A Case Study of Ozone Diurnal Variation in the Convective Boundary Layer in the Southeastern United States Using Multiple Observations and Large-Eddy Simulation. Climate 2019, 7, 53. https://doi.org/10.3390/cli7040053
Huang G, Newchurch MJ, Kuang S, Ouwersloot HG. A Case Study of Ozone Diurnal Variation in the Convective Boundary Layer in the Southeastern United States Using Multiple Observations and Large-Eddy Simulation. Climate. 2019; 7(4):53. https://doi.org/10.3390/cli7040053
Chicago/Turabian StyleHuang, Guanyu, M.J. Newchurch, Shi Kuang, and Huug G. Ouwersloot. 2019. "A Case Study of Ozone Diurnal Variation in the Convective Boundary Layer in the Southeastern United States Using Multiple Observations and Large-Eddy Simulation" Climate 7, no. 4: 53. https://doi.org/10.3390/cli7040053
APA StyleHuang, G., Newchurch, M. J., Kuang, S., & Ouwersloot, H. G. (2019). A Case Study of Ozone Diurnal Variation in the Convective Boundary Layer in the Southeastern United States Using Multiple Observations and Large-Eddy Simulation. Climate, 7(4), 53. https://doi.org/10.3390/cli7040053






