A System for Monitoring Breathing Activity Using an Ultrasonic Radar Detection with Low Power Consumption
Abstract
1. Introduction
2. Anatomy of the Respiratory System and Abnormality
3. Materials and Methods
3.1. Subjects and Experimental Setup
3.2. Measurement System
3.2.1. Ultrasonic PING Sensor
3.2.2. Microcontroller PIC18F452
3.2.3. Smart G100 GSM
3.3. Power Consumption Model
3.4. Software
- -
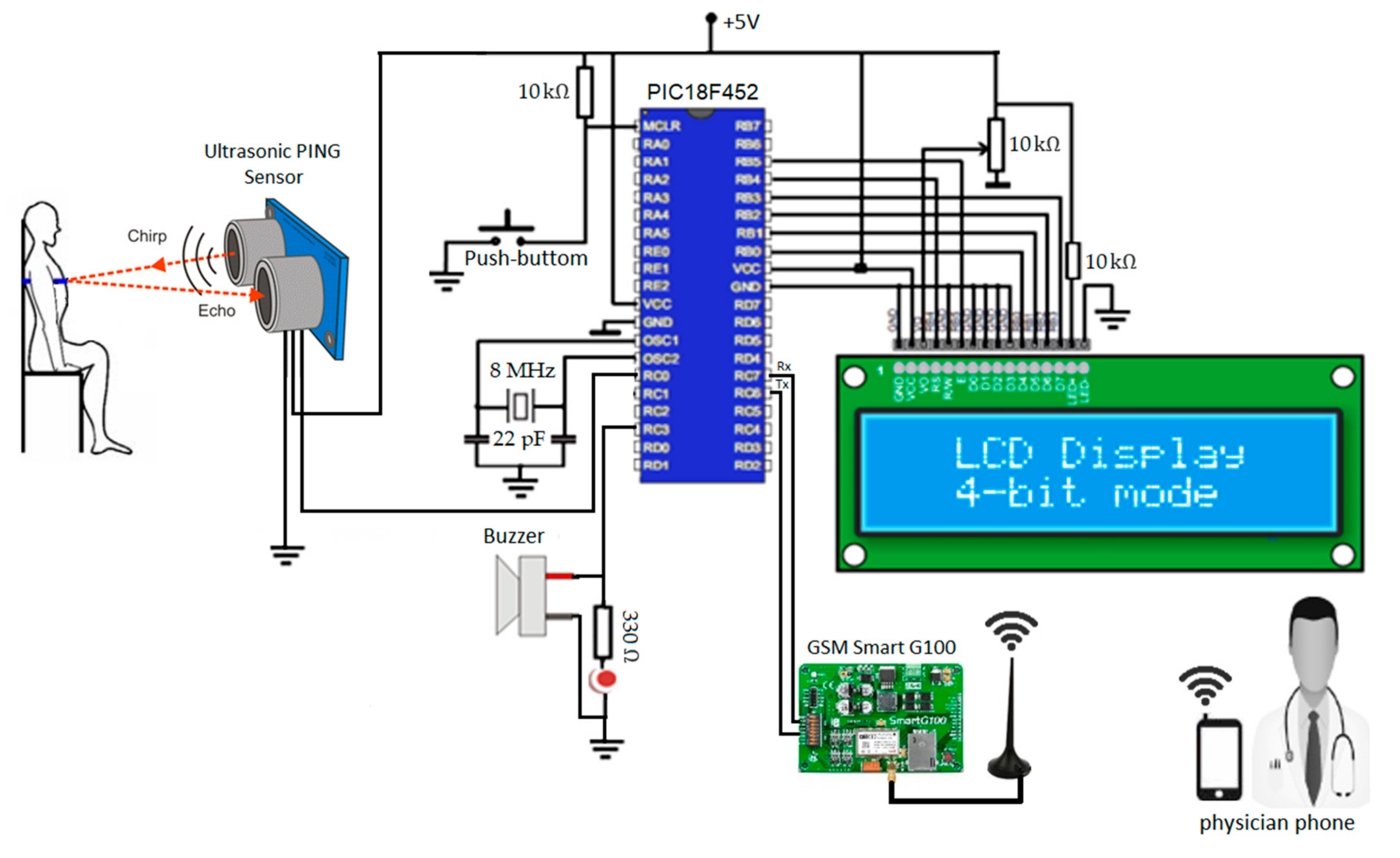
- The I/O port pins of the PIC18F452 were connected to the ultrasonic PING sensor, smart G100 GSM, buzzer alarm circuit and 16 × 2 liquid crystal display (LCD) as shown in Figure 2. The ultrasonic sensor was connected to the PIC microcontroller through the pin (RC0), where the PIC triggers the sensor with 100 μs pulse-width and then measures the amount of time elapsing until the arrival of the reflected signal measured by the same pin. The audible alarm circuit was connected to the output pin of the PIC (RC3) with a light emitting diode (LED) and a sound buzzer. The LCD display unit was connected to the output port of the PIC (PORT B) to display the subject’s name and their breathing state. The RXD/TXD (receive/transmit data) DIP pins of the smart G100 GSM modem was interfaced with the PIC via a serial protocol UART (RC6) as transmitting pin (Tx) and RC7 as receiving pin (Rx).
- -
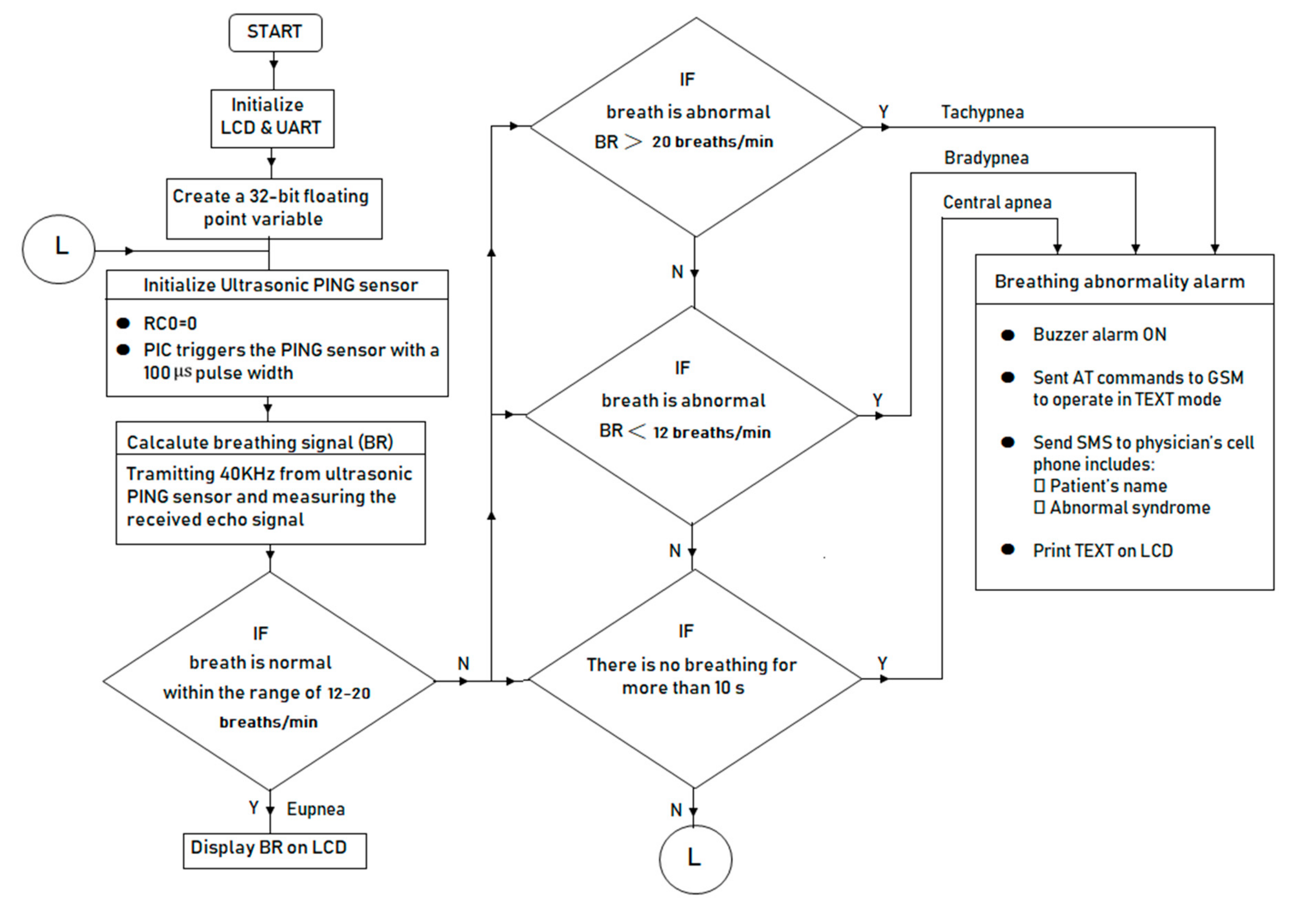
- The microcontroller initializes PING, LCD, and UART and creates a 32-bits floating point variable.
- -
- The ultrasonic PING sensor senses the motion in the thorax and detects the propagation delay and amplitude of the return signal.
- -
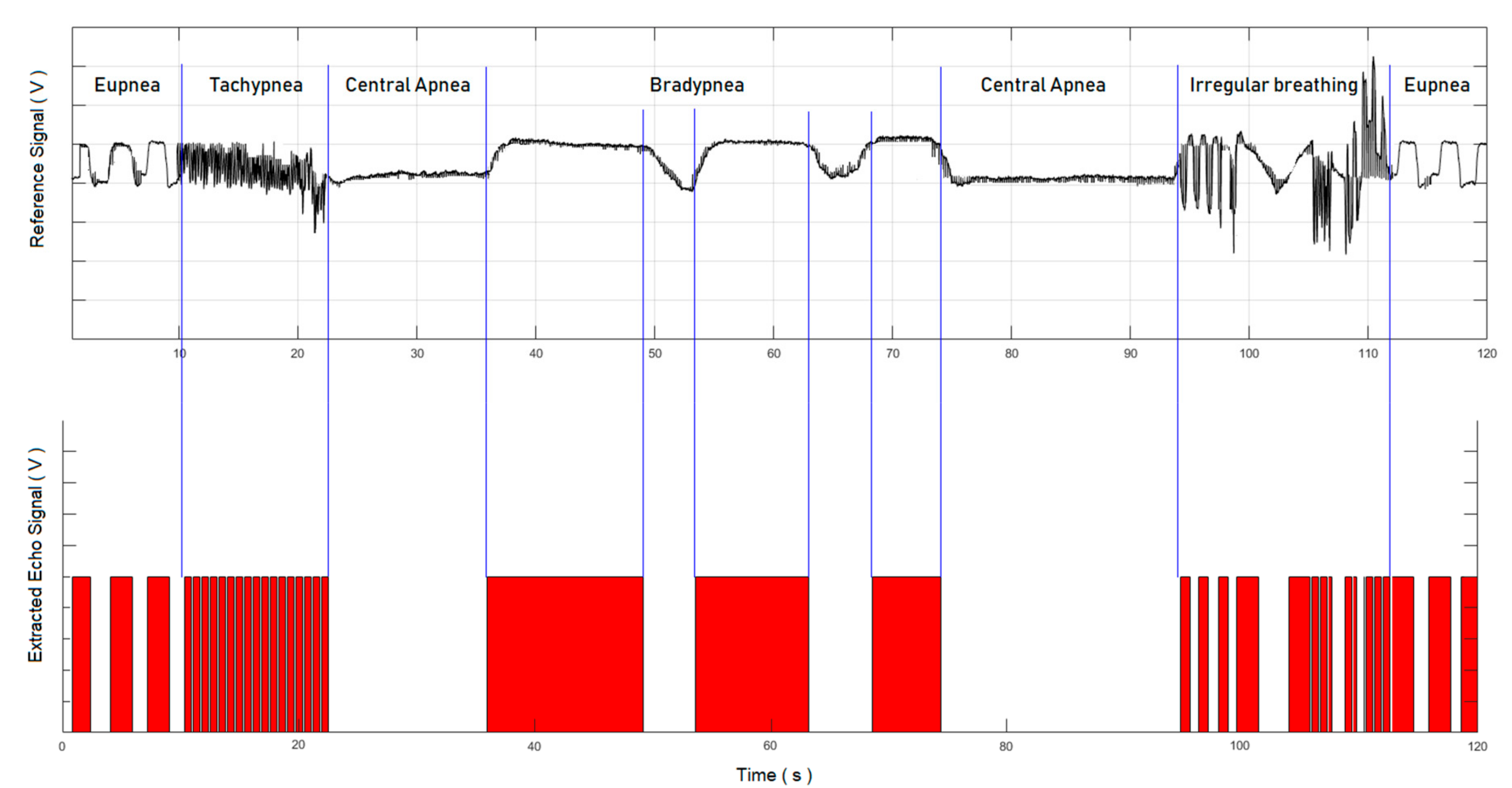
- The microcontroller reads the return signal and calcalutes the distance and breathing cycle using Equations (1) and (2). The microcontroller sends a text message alert to physician’s phone number when the BR falls outside the normal range (tachypnea for BR greater than 20 breaths/min and bradypnea for BR less than 12 breaths/min) [49] or when no breathing exists for more than 10 s.
- -
- A sleep/wake scheme has been implemented inside the microcontroller code based on a duty cycle. All components are in sleep mode until the microcontroller wakes up, which based on an internal timer interrupt.
4. Results and Discussion
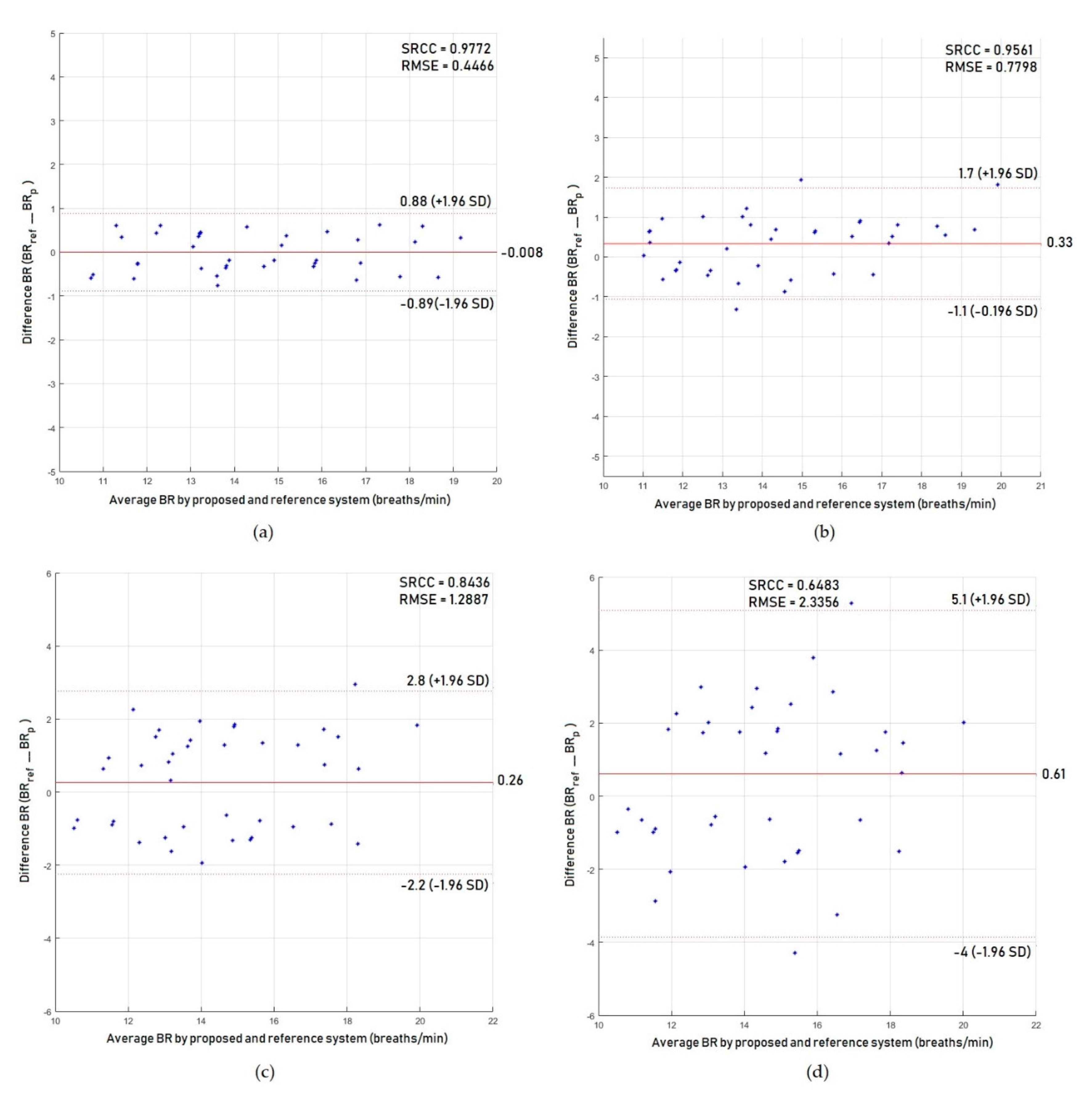
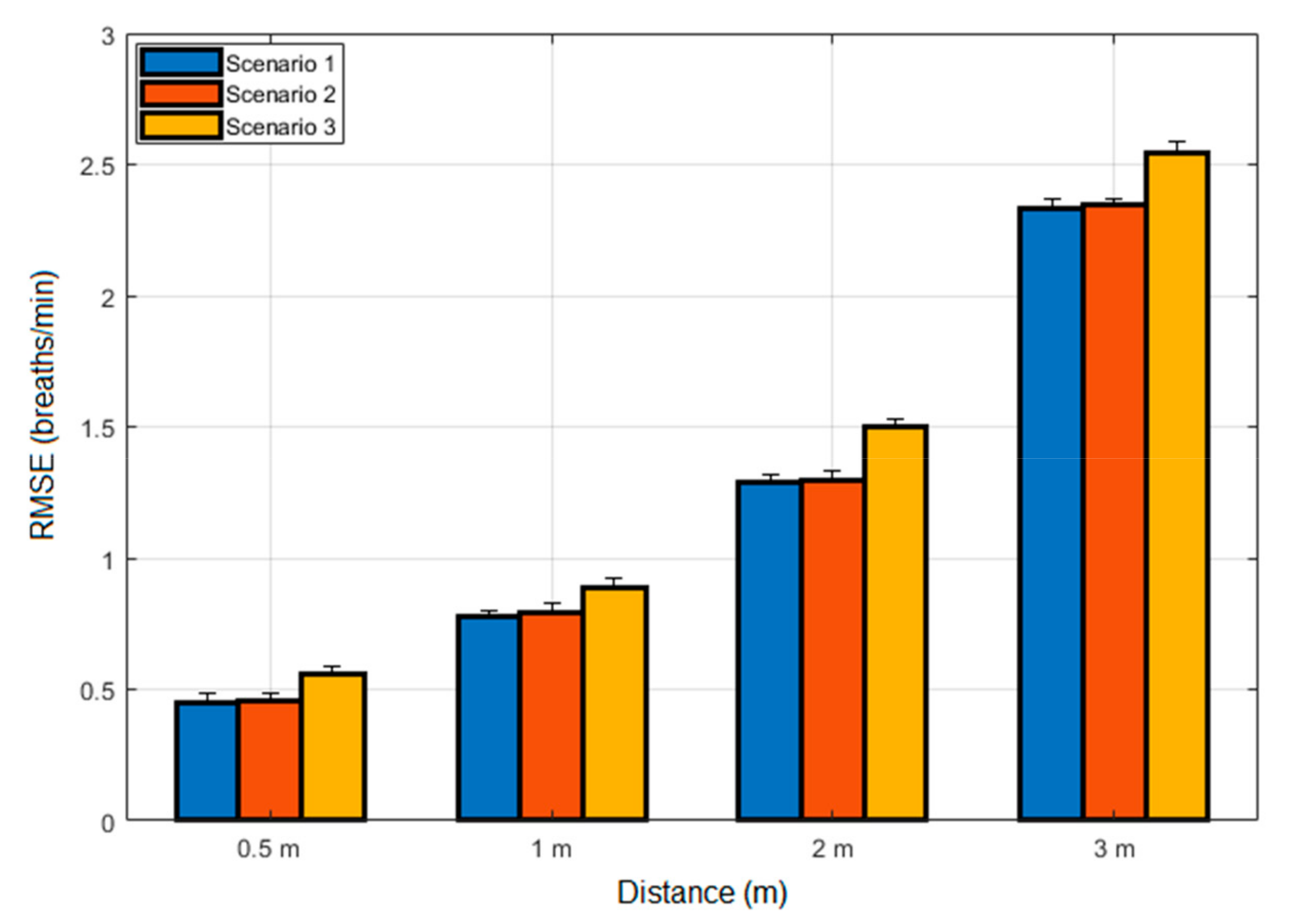
4.1. Results for Abnormal Breathing Syndromes
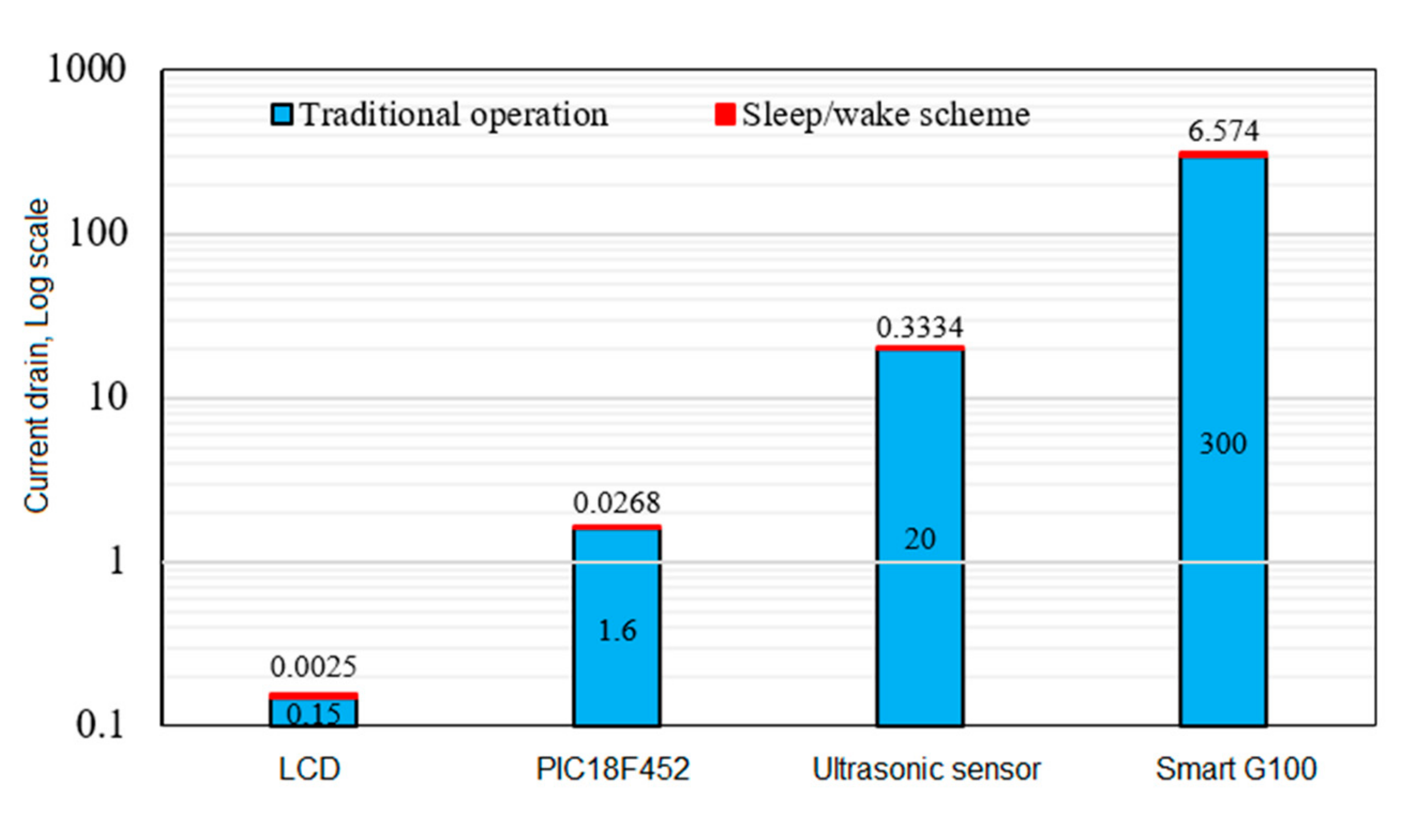
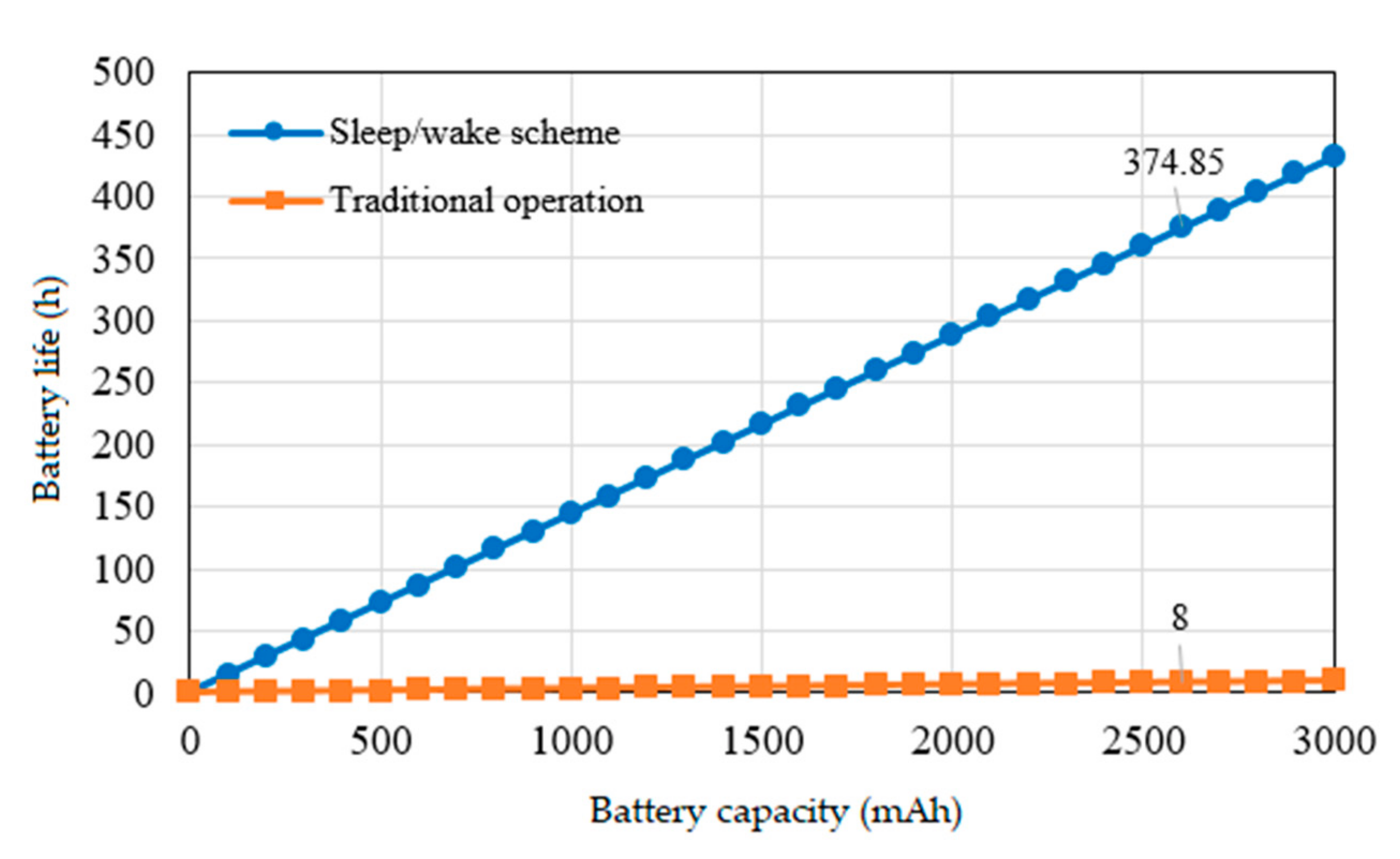
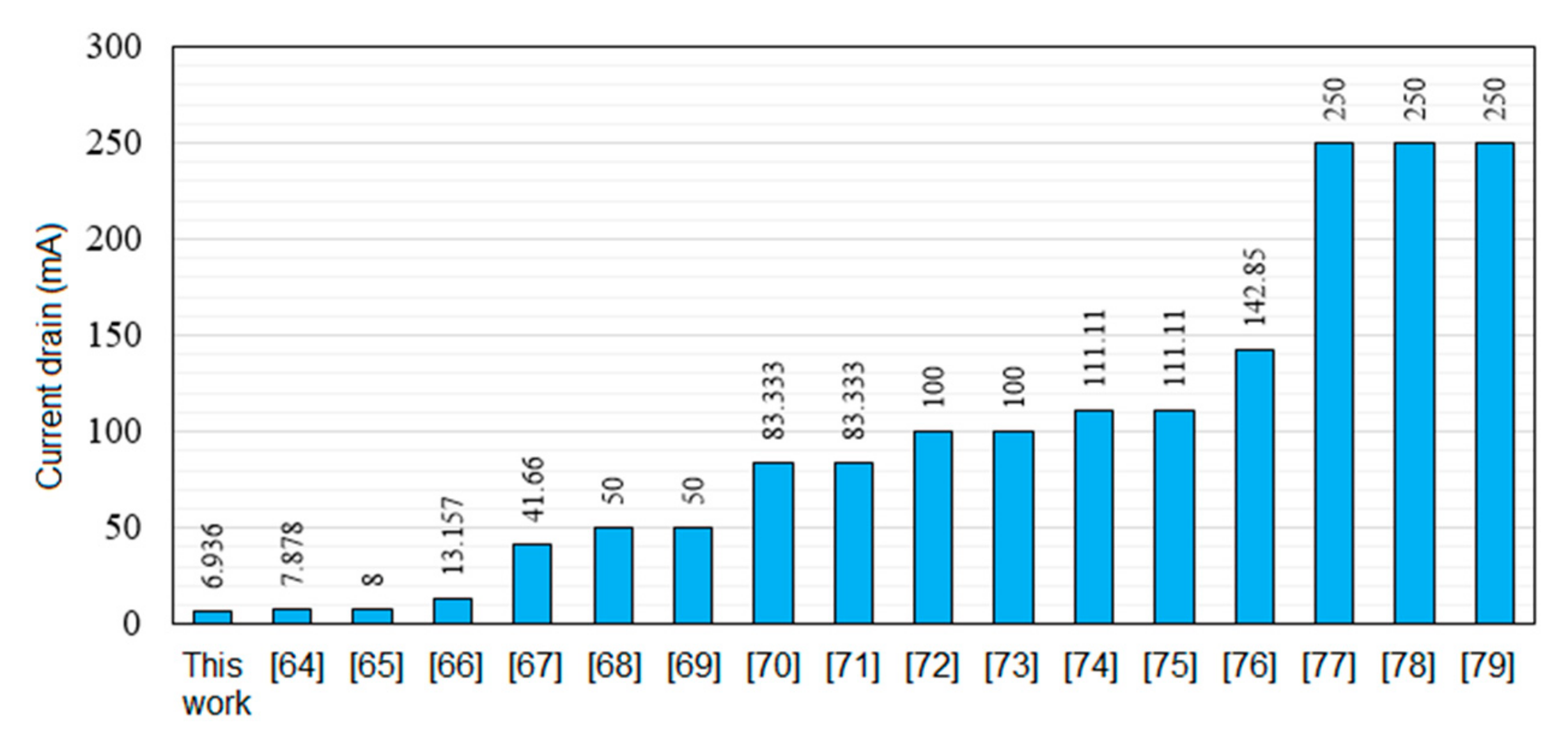
4.2. Results for Power Consumption
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yuan, G.; Drost, N.A.; McIvor, R.A. Respiratory rate and breathing pattern. McMaster Univ. Med. J. 2013, 10, 23–25. [Google Scholar]
- Reeves, W.C.; Heim, C.; Maloney, E.M.; Youngblood, L.S.; Unger, E.R.; Decker, M.J.; Jones, J.F.; Rye, D.B. Sleep characteristics of persons with chronic fatigue syndrome and non-fatigued controls: Results from a population-based study. BMC Neurol. 2006, 6, 41. [Google Scholar] [CrossRef] [PubMed]
- Kjelsberg, F.N.; Ruud, E.A.; Stavem, K. Predictors of symptoms of anxiety and depression in obstructive sleep apnea. Sleep Med. 2005, 6, 341–346. [Google Scholar] [CrossRef]
- Owens, R.L.; Malhotra, A. Sleep-disordered breathing and COPD: The overlap syndrome. Respir. Care 2010, 55, 1333–1346. [Google Scholar]
- Tomasic, I.; Tomasic, N.; Trobec, R.; Krpan, M.; Kelava, T. Continuous remote monitoring of COPD patients—Justification and explanation of the requirements and a survey of the available technologies. Med. Biol. Eng. Comput. 2018, 56, 1–23. [Google Scholar] [CrossRef]
- Hetzenecker, A.; Escourrou, P.; Kuna, S.T.; Series, F.; Lewis, K.; Birner, C.; Pfeifer, M.; Arzt, M. Treatment of sleep apnea in chronic heart failure patients with auto-servo ventilation improves sleep fragmentation: A randomized controlled trial. Sleep Med. 2016, 17, 25–31. [Google Scholar] [CrossRef]
- Wooten, W.I., III; Muenzer, J.; Vaughn, B.V.; Muhlebach, M.S. Relationship of sleep to pulmonary function in mucopolysaccharidosis II. J. Pediatr. 2013, 162, 1210–1215. [Google Scholar] [CrossRef] [PubMed]
- Kaditis, A.; Kheirandish-Gozal, L.; Gozal, D. Pediatric OSAS: Oximetry can provide answers when polysomnography is not available. Sleep Med. Rev. 2016, 27, 96–105. [Google Scholar] [CrossRef]
- Alnaji, A.; Gibson, K.; Lee, S.-H.; Chahl, J. Real time apnoea monitoring of children using the Microsoft Kinect sensor: A pilot study. Sensors 2017, 17, 286. [Google Scholar] [CrossRef]
- Sinharay, A.; Rakshit, R.; Khasnobish, A.; Chakravarty, T.; Ghosh, D.; Pal, A. The ultrasonic directional tidal breathing pattern sensor: Equitable design realization based on phase information. Sensors 2017, 17, 1853. [Google Scholar] [CrossRef]
- Alnaji, A.; Gibson, K.; Chahl, J. Remote sensing of physiological signs using a machine vision system. J. Med. Eng. Technol. 2017, 41, 396–405. [Google Scholar] [CrossRef] [PubMed]
- Kranjec, J.; Beguš, S.; Geršak, G.; Drnovšek, J. Non-contact heart rate and heart rate variability measurements: A review. Biomed. Signal Process. Control 2014, 13, 102–112. [Google Scholar] [CrossRef]
- Fu, L.Y.; Moon, R.Y. Apparent life-threatening events (ALTEs) and the role of home monitors. Pediatr. Rev. 2007, 28, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Teichmann, D.; Foussier, J.; Leonhardt, S. Respiration monitoring based on magnetic induction using a single coil. In Proceedings of the 2010 IEEE UbiComp Biomedical Circuits and Systems Conference (BioCAS), Paphos, Cyprus, 3–5 November 2010; IEEE: Piscataway, NJ, USA, 2010; pp. 37–40. [Google Scholar]
- Teichmann, D.; Foussier, J.; Buscher, M.; Walter, M.; Leonhardt, S. Textile integration of a magnetic induction sensor for monitoring of cardiorespiratory activity. In Proceedings of the World Congress on Medical Physics and Biomedical Engineering, Beijing, China, 26–31 May 2012; Springer: New York, NY, USA, 2013; pp. 1350–1353. [Google Scholar]
- Gursoy, D.; Scharfetter, H. Magnetic induction pneumography: A planar coil system for continuous monitoring of lung function via contactless measurements. J. Electr. Bioimpedance 2010, 1, 56–62. [Google Scholar] [CrossRef]
- Milici, S.; Lázaro, A.; Villarino, R.; Girbau, D.; Magnarosa, M. Wireless wearable magnetometer-based sensor for sleep quality monitoring. IEEE Sens. J. 2018, 18, 2145–2152. [Google Scholar] [CrossRef]
- Sun, X.; Lu, Z.; Hu, W.; Cao, G. SymDetector: Detecting sound-related respiratory symptoms using smartphones. In Proceedings of the 2015 ACM International Joint Conference on Pervasive and Ubiquitous Computing (UbiComp’15), Osaka, Japan, 7–11 September 2015; pp. 97–108. [Google Scholar]
- Nam, Y.; Reyes, B.A.; Chon, K.H. Estimation of respiratory rates using the built-in microphone of a smartphone or headset. IEEE J. Biomed. Health Inform. 2016, 20, 1493–1501. [Google Scholar] [CrossRef]
- Wartzek, T.; Weyer, S.; Leonhardt, S. A differential capacitive electrical field sensor array for contactless measurement of respiratory rate. Physiol. Meas. 2011, 32, 1575. [Google Scholar] [CrossRef]
- Oum, J.; Lee, S.; Kim, D.; Hong, S. Non-contact heartbeat and respiration detector using capacitive sensor with Colpitts oscillator. Electron. Lett. 2008, 44, 1. [Google Scholar] [CrossRef]
- Naranjo-Hernández, D.; Talaminos-Barroso, A.; Reina-Tosina, J.; Roa, L.; Barbarov-Rostan, G.; Cejudo-Ramos, P.; Márquez-Martín, E.; Ortega-Ruiz, F. Smart vest for respiratory rate monitoring of COPD patients based on non-contact capacitive sensing. Sensors 2018, 18, 2144. [Google Scholar] [CrossRef]
- Min, S.D.; Yun, Y.; Shin, H. Simplified structural textile respiration sensor based on capacitive pressure sensing method. IEEE Sens. J. 2014, 14, 3245–3251. [Google Scholar]
- Birsan, N.; Munteanu, D.-P. Non-contact cardiopulmonary monitoring algorithm for a 24 GHz Doppler radar. In Proceedings of the 34th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), San Diego, CA, USA, 28 August–1 September 2012; pp. 3227–3230. [Google Scholar]
- Droitcour, A.; Lubecke, V.; Lin, J.; Boric-Lubecke, O. A microwave radio for Doppler radar sensing of vital signs. In Proceedings of the 2001 IEEE MTT-S International Microwave Symposium Digest, Phoenix, AZ, USA, 20–24 May 2001; Volume 1, pp. 175–178. [Google Scholar]
- Kagawa, M.; Yoshida, Y.; Kubota, M.; Kurita, A.; Matsui, T. Non-contact heart rate monitoring method for elderly people in bed with random body motions using 24 GHz dual radars located beneath the mattress in clinical settings. J. Med. Eng. Technol. 2012, 36, 344–350. [Google Scholar] [CrossRef]
- Lee, Y.S.; Pathirana, P.N.; Evans, R.J.; Steinfort, C.L. Separation of Doppler radar-based respiratory signatures. Med. Biol. Eng. Comput. 2016, 54, 1169–1179. [Google Scholar] [CrossRef] [PubMed]
- Lie, D.; Ichapurapu, R.; Jain, S.; Lopez, J.; Banister, R.; Nguyen, T.; Griswold, J. A 2.4 GHz non-contact biosensor system for continuous monitoring of vital-signs. In Telemedicine Techniques and Applications; InTech: London, UK, 2011; pp. 211–238. [Google Scholar]
- Lubecke, O.B.; Ong, P.-W.; Lubecke, V.M. 10 GHz Doppler radar sensing of respiration and heart movement. In Proceedings of the 28th Annual Bioengineering Conference, Philadelphia, PA, USA, 21 April 2002; pp. 55–56. [Google Scholar]
- Scalise, L.; Marchionni, P.; Ercoli, I. Optical method for measurement of respiration rate. In Proceedings of the IEEE International Workshop on Medical Measurements and Applications (MeMeA), Ottawa, ON, Canada, 30 April–1 May 2010; IEEE: Hoboken, NJ, USA, 2010; pp. 19–22. [Google Scholar]
- Scalise, L.; Ercoli, I.; Marchionni, P.; Tomasini, E.P. Measurement of respiration rate in preterm infants by laser Doppler vibrometry. In Proceedings of the IEEE International Workshop on Medical Measurement and Applications (MEMEA), Bari, Italy, 30–31 May 2011; pp. 657–661. [Google Scholar]
- Morbiducci, U.; Scalise, L.; De Melis, M.; Grigioni, M. Optical vibrocardiography: A novel tool for the optical monitoring of cardiac activity. Annal. Biomed. Eng. 2007, 35, 45–58. [Google Scholar] [CrossRef] [PubMed]
- Marchionni, P.; Scalise, L.; Ercoli, I.; Tomasini, E. An optical measurement method for the simultaneous assessment of respiration and heart rates in preterm infants. Rev. Sci. Instrum. 2013, 84, 121705. [Google Scholar] [CrossRef]
- Min, S.D.; Yoon, D.J.; Yoon, S.W.; Yun, Y.H.; Lee, M. A study on a non-contacting respiration signal monitoring system using Doppler ultrasound. Med. Biol. Eng. Comput. 2007, 45, 1113–1119. [Google Scholar] [CrossRef]
- Min, S.D.; Kim, J.K.; Shin, H.S.; Yun, Y.H.; Lee, C.K.; Lee, J.-H. Noncontact respiration rate measurement system using an ultrasonic proximity sensor. Sens. J. IEEE 2010, 10, 1732–1739. [Google Scholar]
- Heldt, G.P.; Ward, R.J., III. Evaluation of Ultrasound-Based Sensor to Monitor Respiratory and Nonrespiratory Movement and Timing in Infants. IEEE Trans. Biomed. Eng. 2016, 63, 619–629. [Google Scholar] [CrossRef]
- Arlotto, P.; Grimaldi, M.; Naeck, R.; Ginoux, J.-M. An ultrasonic contactless sensor for breathing monitoring. Sensors 2014, 14, 15371–15386. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Liu, Q.; Turner, T.; Wu, Y. Vital sign estimation from passive thermal video. In Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition (CVPR), Anchorage, AK, USA, 23–28 June 2008; pp. 1–8. [Google Scholar]
- Fei, J.; Pavlidis, I. Analysis of breathing air flow patterns in thermal imaging. In Proceedings of the 28th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBS’06), New York, NY, USA, 29–30 August 2006; pp. 946–952. [Google Scholar]
- Chekmenev, S.Y.; Rara, H.; Farag, A.A. Non-contact, wavelet-based measurement of vital signs using thermal imaging. In Proceedings of the First International Conference on Graphics, Vision, and Image Processing (GVIP), Cairo, Egypt, 19 December 2005; pp. 107–112. [Google Scholar]
- Abbas, A.K.; Heimann, K.; Jergus, K.; Orlikowsky, T.; Leonhardt, S. Neonatal non-contact respiratory monitoring based on real-time infrared thermography. Biomed. Eng. Online 2011, 10, 93. [Google Scholar] [CrossRef]
- Procházka, A.; Charvátová, H.; Vaseghi, S.; Vyšata, O. Machine learning in rehabilitation assessment for thermal and heart rate data processing. IEEE Trans. Neural Syst. Rehabil. Eng. 2018, 26, 1209–1214. [Google Scholar] [CrossRef]
- Hu, M.-H.; Zhai, G.-T.; Li, D.; Fan, Y.-Z.; Chen, X.-H.; Yang, X.-K. Synergetic use of thermal and visible imaging techniques for contactless and unobtrusive breathing measurement. J. Biomed. Opt. 2017, 22, 036006. [Google Scholar] [CrossRef]
- Zhao, F.; Li, M.; Qian, Y.; Tsien, J.Z. Remote measurements of heart and respiration rates for telemedicine. PLoS ONE 2013, 8, e71384. [Google Scholar] [CrossRef]
- Alnaji, A.; Chahl, J. Remote optical cardiopulmonary signal extraction with noise artifact removal, multiple subject detection & long-distance. IEEE Access 2018, 6, 11573–11595. [Google Scholar]
- Ali, A.-N.; Javaan, C. Detection of Cardiopulmonary Activity and Related Abnormal Events Using Microsoft Kinect Sensor. Sensors 2018, 18, 920. [Google Scholar]
- Alnaji, A.; Chahl, J. Simultaneous tracking of cardiorespiratory signals for multiple persons using a machine vision system with noise artifact removal. IEEE J. Transl. Eng. Health Med. 2017, 5, 1–10. [Google Scholar] [CrossRef] [PubMed]
- De Haan, G.; van Leest, A. Improved motion robustness of remote-PPG by using the blood volume pulse signature. Physiol. Meas. 2014, 35, 1913. [Google Scholar] [CrossRef]
- Aarts, L.A.; Jeanne, V.; Cleary, J.P.; Lieber, C.; Nelson, J.S.; Bambang Oetomo, S.; Verkruysse, W. Non-contact heart rate monitoring utilizing camera photoplethysmography in the neonatal intensive care unit—A pilot study. Early Hum. Dev. 2013, 89, 943–948. [Google Scholar] [CrossRef]
- Massaroni, C.; Lopes, D.S.; Lo Presti, D.; Schena, E.; Silvestri, S. Contactless Monitoring of Breathing Patterns and Respiratory Rate at the Pit of the Neck: A Single Camera Approach. J. Sens. 2018, 2018, 4567213. [Google Scholar] [CrossRef]
- Alnaji, A.; Gibson, K.; Lee, S.H.; Chahl, J. Monitoring of Cardiorespiratory Signal: Principles of Remote Measurements and Review of Methods. IEEE Access 2017, 99, 1. [Google Scholar]
- Hall, T.; Lie, D.; Nguyen, T.; Mayeda, J.; Lie, P.; Lopez, J.; Banister, R. Non-contact sensor for long-term continuous vital signs monitoring: A review on intelligent phased-array Doppler sensor design. Sensors 2017, 17, 2632. [Google Scholar] [CrossRef]
- Massaroni, C.; Nicolò, A.; Lo Presti, D.; Sacchetti, M.; Silvestri, S.; Schena, E. Contact-based methods for measuring respiratory rate. Sensors 2019, 19, 908. [Google Scholar] [CrossRef]
- Costa, C.; Oliveira, N.; Júnior, E.S.; Melo, P. A noncontact instrument based on ultrasound for the evaluation of asynchronous thoracoabdominal movement in respiratory diseases. In Journal of Physics: Conference Series; IOP Publishing: Bristol, UK, 2018; Volume 1044, p. 012002. [Google Scholar]
- Breteler, M.J.; Huizinga, E.; van Loon, K.; Leenen, L.P.; Dohmen, D.A.; Kalkman, C.J.; Blokhuis, T.J. Reliability of wireless monitoring using a wearable patch sensor in high-risk surgical patients at a step-down unit in the Netherlands: A clinical validation study. BMJ Open 2018, 8, e020162. [Google Scholar] [CrossRef]
- Paralax Inc. PING Ultrasonic Distance Sensor; Paralax Inc.: Rocklin, CA, USA, 2006. [Google Scholar]
- Mazidi, M.A.; McKinlay, R.D.; Causey, D. PIC Microcontroller and Embedded Systems: Using Assembly and C for PIC18; Pearson International: Hudson, NY, USA, 2008. [Google Scholar]
- MikroElektronika. Available online: https://www.mikroe.com/ (accessed on 22 April 2019).
- Gharghan, S.K. Energy-efficient remote temperature monitoring system for patients based on GSM modem and microcontroller. J. Commun. 2017, 12, 433–442. [Google Scholar] [CrossRef]
- Gharghan, S.K.; Nordin, R.; Ismail, M. Energy efficiency of ultra-low-power bicycle wireless sensor networks based on a combination of power reduction techniques. J. Sens. 2016, 2016, 7314207. [Google Scholar] [CrossRef]
- Jawad, H.; Nordin, R.; Gharghan, S.; Jawad, A.; Ismail, M.; Abu-AlShaeer, M. Power reduction with sleep/wake on redundant data (SWORD) in a wireless sensor network for energy-efficient precision agriculture. Sensors 2018, 18, 3450. [Google Scholar] [CrossRef]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Int. J. Nurs. Stud. 2010, 47, 931–936. [Google Scholar] [CrossRef]
- Gharghan, S.; Mohammed, S.; Al-Naji, A.; Abu-AlShaeer, M.; Jawad, H.; Jawad, A.; Chahl, J. Accurate fall detection and localization for elderly people based on neural network and energy-efficient wireless sensor network. Energies 2018, 11, 2866. [Google Scholar] [CrossRef]
- Magno, M.; Polonelli, T.; Casamassima, F.; Gomez, A.; Farella, E.; Benini, L. Energy-efficient context aware power management with asynchronous protocol for body sensor network. Mobile Netw. Appl. 2017, 22, 814–824. [Google Scholar] [CrossRef]
- Fakhri, A.B.; Gharghan, S.K.; Mohammed, S.L. Statistical validation of patient vital signs based on energy-efficient wireless sensor network monitoring system. ARPN J. Eng. Appl. Sci. 2018, 13, 8258–8270. [Google Scholar]
- Gia, T.N.; Sarker, V.K.; Tcarenko, I.; Rahmani, A.M.; Westerlund, T.; Liljeberg, P.; Tenhunen, H. Energy efficient wearable sensor node for IoT-based fall detection systems. Microprocess. Microsyst. 2018, 56, 34–46. [Google Scholar]
- Kantoch, E.; Grochala, D.; Kajor, M. Bio-inspired topology of wearable sensor fusion for telemedical application. In Proceedings of the International Conference on Artificial Intelligence and Soft Computing, Zurich, Switzerland, 2–3 January 2017; Springer: New York, NY, USA, 2017; pp. 658–667. [Google Scholar]
- Di Rosa, M.; Hausdorff, J.M.; Stara, V.; Rossi, L.; Glynn, L.; Casey, M.; Burkard, S.; Cherubini, A. Concurrent validation of an index to estimate fall risk in community dwelling seniors through a wireless sensor insole system: A pilot study. Gait Posture 2017, 55, 6–11. [Google Scholar] [CrossRef]
- Wu, F.; Zhao, H.; Zhao, Y.; Zhong, H. Development of a wearable-sensor-based fall detection system. Int. J. Telemed. Appl. 2015, 2015, 2. [Google Scholar] [CrossRef]
- Miramontes, R.; Aquino, R.; Flores, A.; Rodríguez, G.; Anguiano, R.; Ríos, A.; Edwards, A. PlaIMoS: A remote mobile healthcare platform to monitor cardiovascular and respiratory variables. Sensors 2017, 17, 176. [Google Scholar] [CrossRef]
- Ngu, A.; Wu, Y.; Zare, H.; Polican, A.; Yarbrough, B.; Yao, L. Fall detection using smartwatch sensor data with accessor architecture. In Proceedings of the International Conference on Smart Health, Hong Kong, China, 26–27 June 2017; Springer: New York, NY, USA, 2017; pp. 81–93. [Google Scholar]
- Aziz, K.; Tarapiah, S.; Ismail, S.H.; Atalla, S. Smart real-time healthcare monitoring and tracking system using GSM/GPS technologies. In Proceedings of the 3rd MEC International Conference on Big Data and Smart City (ICBDSC), Muscat, Oman, 15–16 March 2016; IEEE: Hoboken, NJ, USA, 2016; pp. 1–7. [Google Scholar]
- Tarapiah, S.; Daadoo, M.; Atalla, S. Android-based real-time healthcare system. Int. J. Med. Eng. Inform. 2017, 9, 253–268. [Google Scholar] [CrossRef]
- Sunehra, D.; Ramakrishna, P. Web based patient health monitoring system using Raspberry Pi. In Proceedings of the 2nd International Conference on Contemporary Computing and Informatics (IC3I), Noida, India, 14–17 December 2016; IEEE: Hoboken, NJ, USA, 2016; pp. 568–574. [Google Scholar]
- Liang, Y.; Zhou, X.; Yu, Z.; Guo, B. Energy-efficient motion related activity recognition on mobile devices for pervasive healthcare. Mobile Netw. Appl. 2014, 19, 303–317. [Google Scholar] [CrossRef]
- He, J.; Bai, S.; Wang, X. An unobtrusive fall detection and alerting system based on Kalman filter and Bayes network classifier. Sensors 2017, 17, 1393. [Google Scholar] [CrossRef]
- Prakash, R.; Ganesh, A.B.; Girish, S.V. Cooperative wireless network control based health and activity monitoring system. J. Med. Syst. 2016, 40, 206. [Google Scholar] [CrossRef] [PubMed]
- Megalingam, R.K.; Veliyara, P.S.; Prabhu, R.M.; Krishna, R.R.; Katoch, R. Wheeled patient monitoring system. In Intelligent Computing, Communication and Devices. Advances in Intelligent Systems and Computing; Springer: New Delhi, India, 2015; Volume 305, pp. 779–786. [Google Scholar]
- Ijaz, U.; Ameer, U.; Tarar, H.; Ilyas, A.; Ijaz, A. E-health acquistion, transmission & monitoring system. In Proceedings of the 2nd IEEE Workshop on Recent Trends in Telecommunications Research, Palmerston North, New Zealand, 10 February 2017. [Google Scholar]


| Methods | Region of Interest | No. of Subjects | Measurements | Abnormalities | Distance (m) | Clothing Scenarios | Communication with Care Providers | Power Consumption |
|---|---|---|---|---|---|---|---|---|
| Min et al. [34] | Thorax | 1 | Normal breathing | Apnea | 0.1–0.2 | Subject was naked | None | None |
| Min et al. [35] | Abdomen | 10 | Normal breathing | None | 1 | Naked and semi-clothed | None | None |
| Arlotto et al. [37] | Head (nose) | 1 | Normal breathing | Apnea | 0.5 | None | None | None |
| Heldt et al. [36] | Torso | 11 | Normal breathing | None | 0.15–0.5 | Fully-clothed | None | None |
| Sinharay et al. [10] | Head (mouth) | 20 | Normal breathing | None | None | None | None | None |
| Costa et al. [54] | Thorax | 12 | Normal breathing | Modifications associated with simulated breathing disease | ~1 | Semi-clothed | None | None |
| The current study | Thorax | 12 | Normal breathing | Tachypnea, bradypnea, central apnea and irregular breathing | 3 | Naked, semi-clothes and fully-clothed | Included | Included |
| Parameter | Ultrasonic PING Sensor | LCD | PIC18F452 | Smart G100 GSM |
|---|---|---|---|---|
| Active current ()/mA | 20 | 0.15 | 1.6 @ 4 MHz | 300 |
| Sleep current or standby state ()/mA | 0 | 0 | 0.2 µA (standby) | 1.6 |
| Active time (Tactive)/min | 24 | 24 | 24 | 24 |
| Sleep time (Tsleep or tstandby)/min | 1416 (1440−24) | 1416 | 1416 | 1416 |
| Total period (Ttotal)/min | 1440 | 1440 | 1440 | 1440 |
| Subtotal average current drain | ||||
| CAvg/mA, for each component | 0.3334 | 0.0025 | 0.0268 | 6.574 |
| CAvg/mA, based on sleep/wake scheme | 6.936 | |||
| Cactive/mA, based on traditional operation | 321.75 | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Naji, A.; Al-Askery, A.J.; Gharghan, S.K.; Chahl, J. A System for Monitoring Breathing Activity Using an Ultrasonic Radar Detection with Low Power Consumption. J. Sens. Actuator Netw. 2019, 8, 32. https://doi.org/10.3390/jsan8020032
Al-Naji A, Al-Askery AJ, Gharghan SK, Chahl J. A System for Monitoring Breathing Activity Using an Ultrasonic Radar Detection with Low Power Consumption. Journal of Sensor and Actuator Networks. 2019; 8(2):32. https://doi.org/10.3390/jsan8020032
Chicago/Turabian StyleAl-Naji, Ali, Ali J. Al-Askery, Sadik Kamel Gharghan, and Javaan Chahl. 2019. "A System for Monitoring Breathing Activity Using an Ultrasonic Radar Detection with Low Power Consumption" Journal of Sensor and Actuator Networks 8, no. 2: 32. https://doi.org/10.3390/jsan8020032
APA StyleAl-Naji, A., Al-Askery, A. J., Gharghan, S. K., & Chahl, J. (2019). A System for Monitoring Breathing Activity Using an Ultrasonic Radar Detection with Low Power Consumption. Journal of Sensor and Actuator Networks, 8(2), 32. https://doi.org/10.3390/jsan8020032







