Chemical Constituents of the Leaves of Campanula takesimana (Korean Bellflower) and Their Inhibitory Effects on LPS-induced PGE2 Production
Abstract
1. Introduction
2. Results
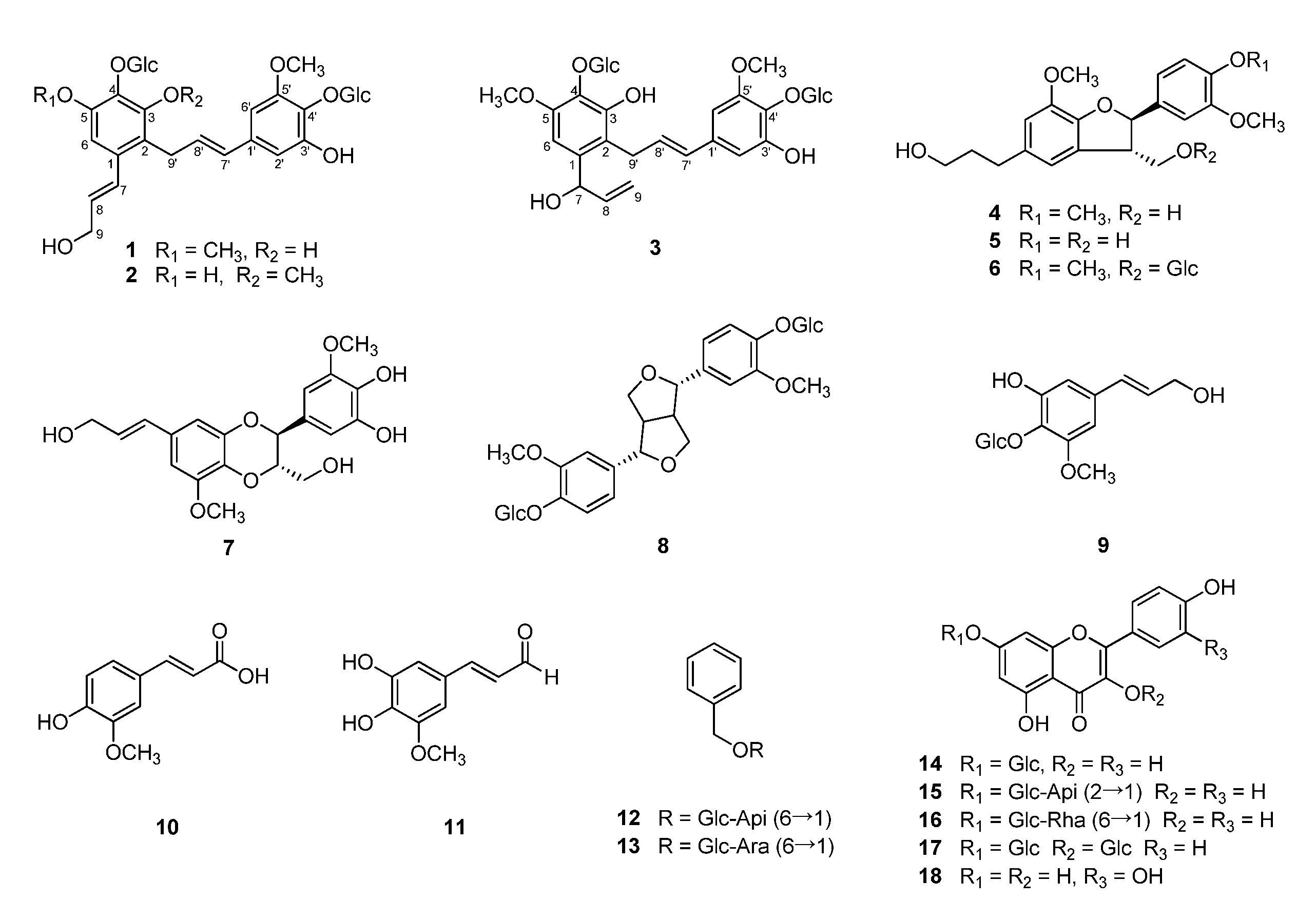
2.1. Structure Elucidation of New Compounds 1–3
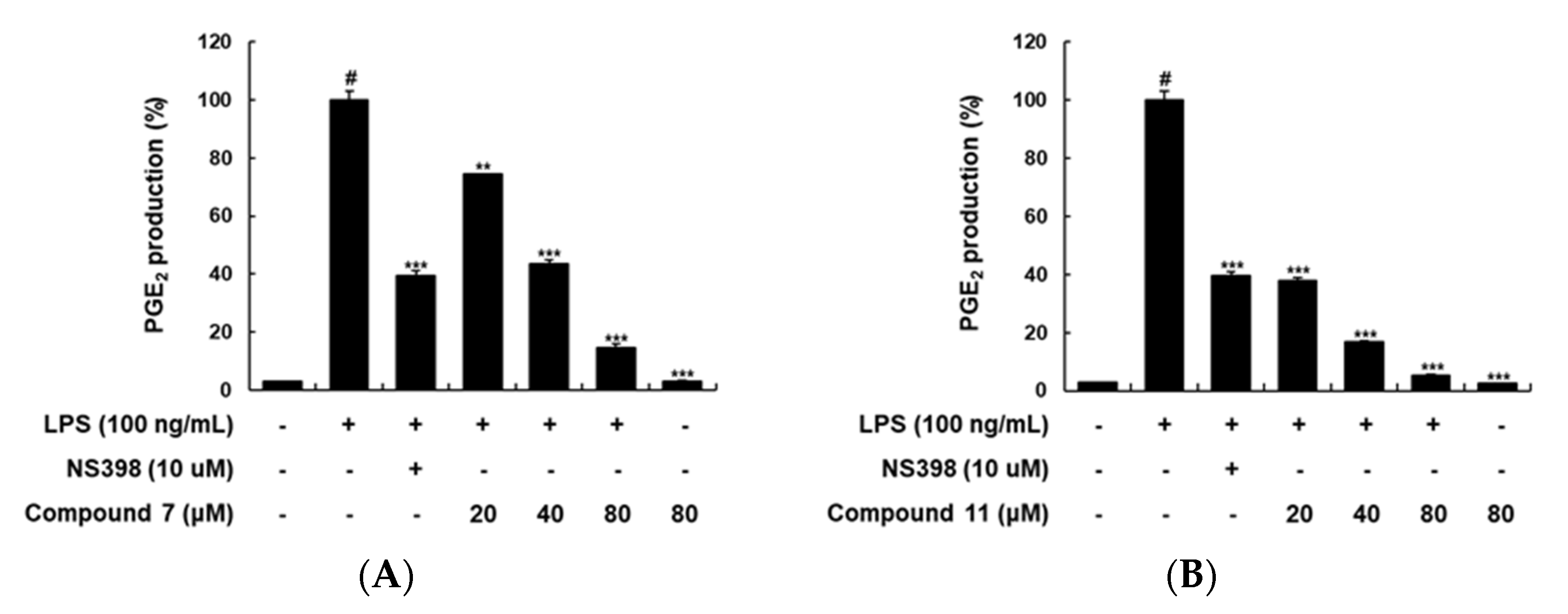
2.2. Inhibitory Effects of Compounds on LPS-Induced PGE2 Production in RAW 264.7 Macrophages
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. General Experimental Procedures
4.3. Extraction and Isolation
4.3.1. Campanulalignan A (1)
4.3.2. Campanulalignan B (2)
4.3.3. Campanulalignan C (3)
4.4. Enzymatic Hydrolysis of Compound 1
4.5. Absolute Configurations of β-glucose in Compound 1
4.6. Cell Culture
4.7. Cell Viability Test
4.8. Measurement of PGE2 Production
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Park, K.R.; Jung, H.J. Isozyme and morphological variation in Campanula punctata and C. takesimana (Campanulaceae). Korean J. Plant Taxon. 2000, 30, 1–16. [Google Scholar] [CrossRef]
- Kim, M.; Kim, K.; Yook, H. Antioxidative effects of Campanula takesimana Nakai extract. J. Korean Soc. Food Sci. Nutr. 2012, 41, 1331–1337. [Google Scholar] [CrossRef]
- Kim, H.J.; Kang, S.H. Ethnobotany, Phytochemistry, pharmacology of the Korean Campanulaceae: A comprehensive review. Korean J. Plant Res. 2017, 30, 240–264. [Google Scholar] [CrossRef]
- Yang, G.; Lee, K.; Lee, M.; Ham, I.; Choi, H.Y. Inhibition of lipopolysaccharide-induced nitric oxide and prostaglandin E 2 production by chloroform fraction of Cudrania tricuspidata in RAW 264.7 macrophages. BMC Complement. Altern. Med. 2012, 12, 250. [Google Scholar] [CrossRef]
- Agrawal, P.K. NMR spectroscopy in the structural elucidation of oligosaccharides and glycosides. Phytochemistry 1992, 31, 3307–3330. [Google Scholar] [CrossRef]
- Yuda, M.; Ohtani, K.; Mizutani, K.; Kasai, R.; Tanaka, O.; Jia, M.R.; Ling, Y.R.; Pu, X.F.; Saruwatari, Y.I. Neolignan glycosides from roots of Codonopsis tangshen. Phytochemistry 1990, 29, 1989–1993. [Google Scholar] [CrossRef]
- Mizutani, K.; Yuda, M.; Tanaka, O.; Saruwatari, Y.I.; Jia, M.R.; Ling, Y.K.; Pu, X.F. Tanghenosides i and ii from chuan-dangshen, the root of Codonopsis tangshen oliv. Chem. Pharm. Bull. 1998, 36, 2726–2729. [Google Scholar] [CrossRef]
- Pieters, L.; de Bruyne, T.; Claeys, M.; Vlietinck, A.; Calomme, M.; vanden Berghe, D. Isolation of a dihydrobenzofuran lignan from South American dragon’s blood (Croton spp.) as an inhibitor of cell proliferation. J. Nat. Prod. 1993, 56, 899–906. [Google Scholar] [CrossRef]
- Li, L.; Seeram, N.P. Maple syrup phytochemicals include lignans, coumarins, a stilbene, and other previously unreported antioxidant phenolic compounds. J. Agric. Food Chem. 2010, 58, 11673–11679. [Google Scholar] [CrossRef]
- Calis, I.; Kirmizibekmez, H.; Beutler, J.A.; Donmez, A.A.; Yalçin, F.N.; Kilic, E.; Ozalp, M.; Ruedi, P.; Tasdemir, D. Secondary metabolites of Phlomis viscosa and their biological activities. Turk. J. Chem. 2005, 29, 71–81. [Google Scholar]
- Son, Y.K.; Lee, M.H.; Han, Y.N. A New antipsychotic effective neolignan from Firmiana simplex. Arch. Pharm. Res. 2005, 28, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, B.; Scholle, S.; Hölzl, J.; Khudeir, N.; Hess, S.; Müller, C.E. Lignans isolated from valerian: Identification and characterization of a new olivil derivative with partial agonistic mactivity at A1 adenosine receptors. J. Nat. Prod. 2002, 65, 1479–1485. [Google Scholar] [CrossRef] [PubMed]
- Tan, R.X.; Ma, W.G.; Wei, H.X.; Zhang, L.X. Glycosides from Wahlenbergia marginata. Phytochemistry 1998, 48, 1245–1250. [Google Scholar] [CrossRef]
- Yawadio, R.; Tanimori, S.; Morita, N. Identification of phenolic compounds isolated from pigmented rices and their aldose reductase inhibitory activities. Food Chem. 2007, 101, 1616–1625. [Google Scholar] [CrossRef]
- Sakakibara, N.; Nakatsubo, T.; Suzuki, S.; Shibata, D.; Shimada, M.; Umezawa, T. Metabolic analysis of the cinnamate/monolignol pathway in Carthamus tinctorius seeds by a stable-isotope-dilution method. Org. Biomol. Chem. 2007, 5, 802–815. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.; Fu, X.; Li, Y.; Xiong, Z.; Shi, X.; Zhang, F.; Huo, G.; Li, B. Phytochemical study of stem and leaf of Clausena lansium. Molecules 2019, 24, 3124. [Google Scholar] [CrossRef]
- Lee, S.Y.; Kim, K.H.; Lee, I.K.; Lee, K.H.; Choi, S.U.; Lee, K.R. A new flavonol glycoside from Hylomecon vernalis. Arch. Pharm. Res. 2012, 35, 415–442. [Google Scholar] [CrossRef]
- Chiang, H.; Lo, Y.; Lu, F. Xanthine oxidase inhibitors from the leaves of Alsophila spinulosa (Hook) Tryon. J. Enzym. Inhib. 1994, 8, 61–71. [Google Scholar] [CrossRef]
- Cheng, X.; Tang, X.; Guo, C.; Zhang, C.; Yang, Q. New flavonol glycosides from the seeds of Desmodium styracifolium. Chem. Nat. Compd. 2018, 54, 846–850. [Google Scholar] [CrossRef]
- Kazuma, K.; Noda, N.; Suzuki, M. Malonylated flavonol glycosides from the petals of Clitoria ternatea. Phytochemistry 2003, 62, 229–237. [Google Scholar] [CrossRef]
- Markham, K.R.; Ternai, B.; Stanley, R.; Geiger, H.; Mabry, T.J. Carbon-13 NMR Studies of flavonoids—III: Naturally occurring flavonoid glycosides and their acylated derivatives. Tetrahedron 1978, 34, 1389–1397. [Google Scholar] [CrossRef]
- Aisyah, L.S.; Yun, Y.F.; Herlina, T.; Julaeha, E.; Zainuddin, A.; Nurfarida, I.; Hidayat, A.T.; Supratman, U.; Shiono, Y. Flavonoid compounds from the leaves of Kalanchoe prolifera and their cytotoxic activity against P-388 murine leukimia cells. Nat. Prod. Sci. 2017, 23, 139–145. [Google Scholar] [CrossRef]
- Nathan, C. Points of control in inflammation. Nature 2002, 420, 846–852. [Google Scholar] [CrossRef] [PubMed]
- Hotamisligil, G.S. Inflammation and metabolic disorders. Nature 2006, 444, 860–867. [Google Scholar] [CrossRef]
- Hammer, K.D.; Hillwig, M.L.; Solco, A.K.; Dixon, P.M.; Delate, K.; Murphy, P.A.; Wurtele, E.S.; Birt, D.F. Inhibition of prostaglandin E2 production by anti-inflammatory Hypericum perforatum extracts and constituents in RAW264. 7 mouse macrophage cells. J. Agric. Food Chem. 2007, 55, 7323–7331. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Huang, X.; Bai, M.; Chang, X.; Yan, X.; Zhu, T.; Zhao, W.; Peng, Y.; Song, S. Antioxidant and anti-inflammatory active dihydrobenzofuran neolignans from the Seeds of Prunus tomentosa. J. Agric. Food Chem. 2014, 62, 7796–7803. [Google Scholar] [CrossRef]
- Yao, J.; Zou, Z.; Wang, X.; Ji, X.; Yang, J. Pinoresinol diglucoside alleviates oxLDL-induced dysfunction in human umbilical vein endothelial cells. Evid. Based Complement. Alternat. Med. 2016, 2016, 10. [Google Scholar] [CrossRef]
- Yoo, S.; Jeong, S.; Lee, N.; Shin, H.; Seo, C. Simultaneous determination and anti-inflammatory effects of four phenolic compounds in Dendrobii herba. Nat. Prod. Res. 2017, 31, 2923–2926. [Google Scholar] [CrossRef]
- Wu, X.; Zhang, L.; He, J.; Li, G.; Ding, L.; Gao, X.; Dong, L.; Song, L.; Li, Y.; Zhao, Q. Two new diterpenoids from Excoecaria acerifolia. J. Asian Nat. Prod. Res. 2013, 15, 151–157. [Google Scholar] [CrossRef]
- Li, L.; Popko, J.L.; Umezawa, T.; Chiang, V.L. 5-hydroxyconiferyl aldehyde modulates enzymatic methylation for syringyl monolignol formation, a new view of monolignol biosynthesis in angiosperms. J. Biol. Chem. 2000, 275, 6537–6545. [Google Scholar] [CrossRef]
- Tanaka, T.; Tatsuya, N.; Toshihisa, U.; Kenji, T.; Isao, K. Facile discrimination of aldose enantiomers by reversed-phase HPLC. Chem. Pharm. Bull. 2007, 55, 899–901. [Google Scholar] [CrossRef] [PubMed]
 ) correlations of compounds 1–3.
) correlations of compounds 1–3.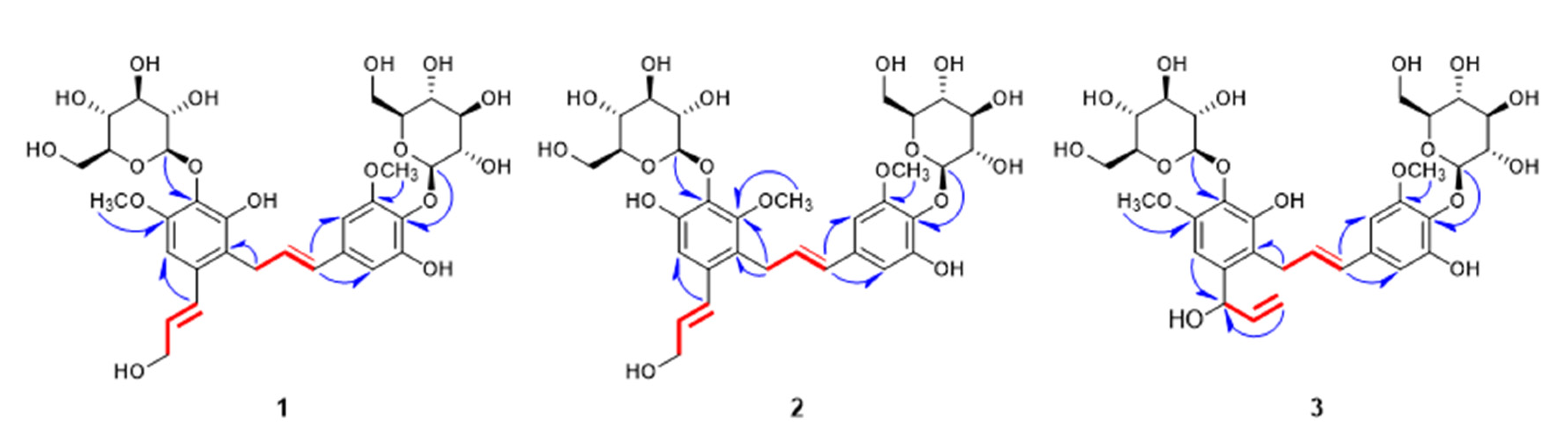
| Position a | 1 | 2 | 3 | |||
|---|---|---|---|---|---|---|
| δC | δHb | δC | δHb | δC | δHb | |
| 1 | 134.9 | 135.4 | 140.0 | |||
| 2 | 119.5 | 123.9 | 119.0 | |||
| 3 | 152.4 | 153.0 | 150.1 | |||
| 4 | 134.9 | 139.3 | 134.6 | |||
| 5 | 150.1 | 150.8 | 152.5 | |||
| 6 | 102.5 | 6.71 s | 110.8 | 6.87 s | 102.9 | 6.71 s |
| 7 | 129.3 | 6.86 d (15.5) | 128.9 | 6.81 d (15.5) | 72.1 | 5.40 d (5.5) |
| 8 | 132.0 | 6.24 dt (15.0, 5.0) | 130.0 | 6.17 dt (15.5, 6.0) | 141.7 | 6.01 ddd (16.5, 10.0, 5.0) |
| 9 | 62.2 | 4.24 dd (5.5, 2.0) | 63.9 | 4.21 dd (5.5, 2.0) | 115.2 | 5.26 d (17.0) |
| 5.12 d (10.5) | ||||||
| 1′ | 136.9 | 136.6 | 136.9 | |||
| 2′ | 108.2 | 6.48 d (2.0) | 108.2 | 6.49 d (2.0) | 103.1 | 6.47 d (1.5) |
| 3′ | 151.9 | 152.0 | 152.5 | |||
| 4′ | 134.5 | 134.6 | 134.6 | |||
| 5′ | 154.1 | 154.3 | 154.3 | |||
| 6′ | 103.0 | 6.47 d (2.0) | 103.1 | 6.48 d (2.0) | 108.4 | 6.48 d (1.5) |
| 7′ | 130.9 | 6.14 (15.5) | 130.4 | 6.13 d (15.5) | 131.0 | 6.18 d (16.0) |
| 8′ | 129.9 | 6.20 dt (15.0, 5.0) | 131.4 | 6.24 dt (15.5, 5.5) | 130.1 | 6.24 dt (15.5, 6.0) |
| 9′ | 29.9 | 3.55 d (5.5) | 30.2 | 3.55 d (5.5) | 29.5 | 3.62 d (7.0) |
| 3.54 d (6.0) | ||||||
| Glc-1 | 107.3 | 4.67 d (8.0) | 107.3 | 4.81 d (8.0) | 107.3 | 4.67 d (8.0) |
| Glc-2 | 71.0 | 3.45 overlap | 71.0 | 3.49 m | 71.0 | 3.51 overlap |
| Glc-3 | 77.8 | 3.40 overlap | 78.0 | 3.43 overlap | 77.8 | 3.45 overlap |
| Glc-4 | 75.5 | 3.49 overlap | 75.5 | 3.43 overlap | 75.5 | 3.42 overlap |
| Glc-5 | 78.5 | 3.22 m | 78.6 | 3.22 overlap | 78.7 | 3.23 overlap |
| Glc-6 | 62.0 | 3.68/3.82 overlap | 62.4 | 3.82 overlap | 62.3 | 3.86 m |
| Glc-1′ | 107.0 | 4.63 d (8.0) | 107.0 | 4.64 d (8.0) | 107.1 | 4.63 d (7.5) |
| Glc-2′ | 71.0 | 3.47 overlap | 71.0 | 3.47 m | 71.0 | 3.51 overlap |
| Glc-3′ | 77.8 | 3.43 overlap | 77.9 | 3.84 overlap | 77.8 | 3.45 overlap |
| Glc-4′ | 75.5 | 3.51 m | 75.5 | 3.44 m | 75.5 | 3.42 overlap |
| Glc-5′ | 78.5 | 3.30 m | 78.5 | 3.29 m | 78.7 | 3.23 overlap |
| Glc-6′ | 62.0 | 3.68/3.82 overlap | 62.2 | 3.22 overlap | 62.3 | 3.78 m |
| OCH3-5 | 56.8 | 3.86 s | 62.7 | 3.87s | 56.8 | 3.84 s |
| OCH3-5′ | 56.8 | 3.81 s | 56.8 | 3.81s | 56.7 | 3.81 s |
| Compound | IC80 (μM) Cytotoxicitiy | Inhibition Rate (%) a | Compound | IC80 (μM) Cytotoxicitiy | Inhibition Rate (%) a |
|---|---|---|---|---|---|
| 1 | >50 | 23.92 | 10 | >50 | 62.53 |
| 2 | 33.66 | 33.07 | 11 | >50 | 85.17 |
| 3 | - | - | 12 | >50 | 31.07 |
| 4 | 30.69 | 7.89 | 13 | >50 | 33.12 |
| 5 | >50 | 69.35 | 14 | >50 | 23.55 |
| 6 | >50 | 20.01 | 15 | >50 | 43.49 |
| 7 | >50 | 60.01 | 16 | >50 | 36.73 |
| 8 | >50 | 58.08 | 17 | >50 | 34.51 |
| 9 | >50 | 37.44 | 18 | >50 | 97.01 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qi, Y.; Choi, S.-I.; Son, S.-R.; Han, H.-S.; Ahn, H.S.; Shin, Y.-K.; Lee, S.H.; Lee, K.-T.; Kwon, H.C.; Jang, D.S. Chemical Constituents of the Leaves of Campanula takesimana (Korean Bellflower) and Their Inhibitory Effects on LPS-induced PGE2 Production. Plants 2020, 9, 1232. https://doi.org/10.3390/plants9091232
Qi Y, Choi S-I, Son S-R, Han H-S, Ahn HS, Shin Y-K, Lee SH, Lee K-T, Kwon HC, Jang DS. Chemical Constituents of the Leaves of Campanula takesimana (Korean Bellflower) and Their Inhibitory Effects on LPS-induced PGE2 Production. Plants. 2020; 9(9):1232. https://doi.org/10.3390/plants9091232
Chicago/Turabian StyleQi, Yutong, Se-In Choi, So-Ri Son, Hee-Soo Han, Hye Shin Ahn, Yu-Kyong Shin, Sun Hee Lee, Kyung-Tae Lee, Hak Cheol Kwon, and Dae Sik Jang. 2020. "Chemical Constituents of the Leaves of Campanula takesimana (Korean Bellflower) and Their Inhibitory Effects on LPS-induced PGE2 Production" Plants 9, no. 9: 1232. https://doi.org/10.3390/plants9091232
APA StyleQi, Y., Choi, S.-I., Son, S.-R., Han, H.-S., Ahn, H. S., Shin, Y.-K., Lee, S. H., Lee, K.-T., Kwon, H. C., & Jang, D. S. (2020). Chemical Constituents of the Leaves of Campanula takesimana (Korean Bellflower) and Their Inhibitory Effects on LPS-induced PGE2 Production. Plants, 9(9), 1232. https://doi.org/10.3390/plants9091232






