Light and Low Relative Humidity Increase Antioxidants Content in Mung Bean (Vigna radiata L.) Sprouts
Abstract
1. Introduction
2. Results
2.1. Sprouts Development
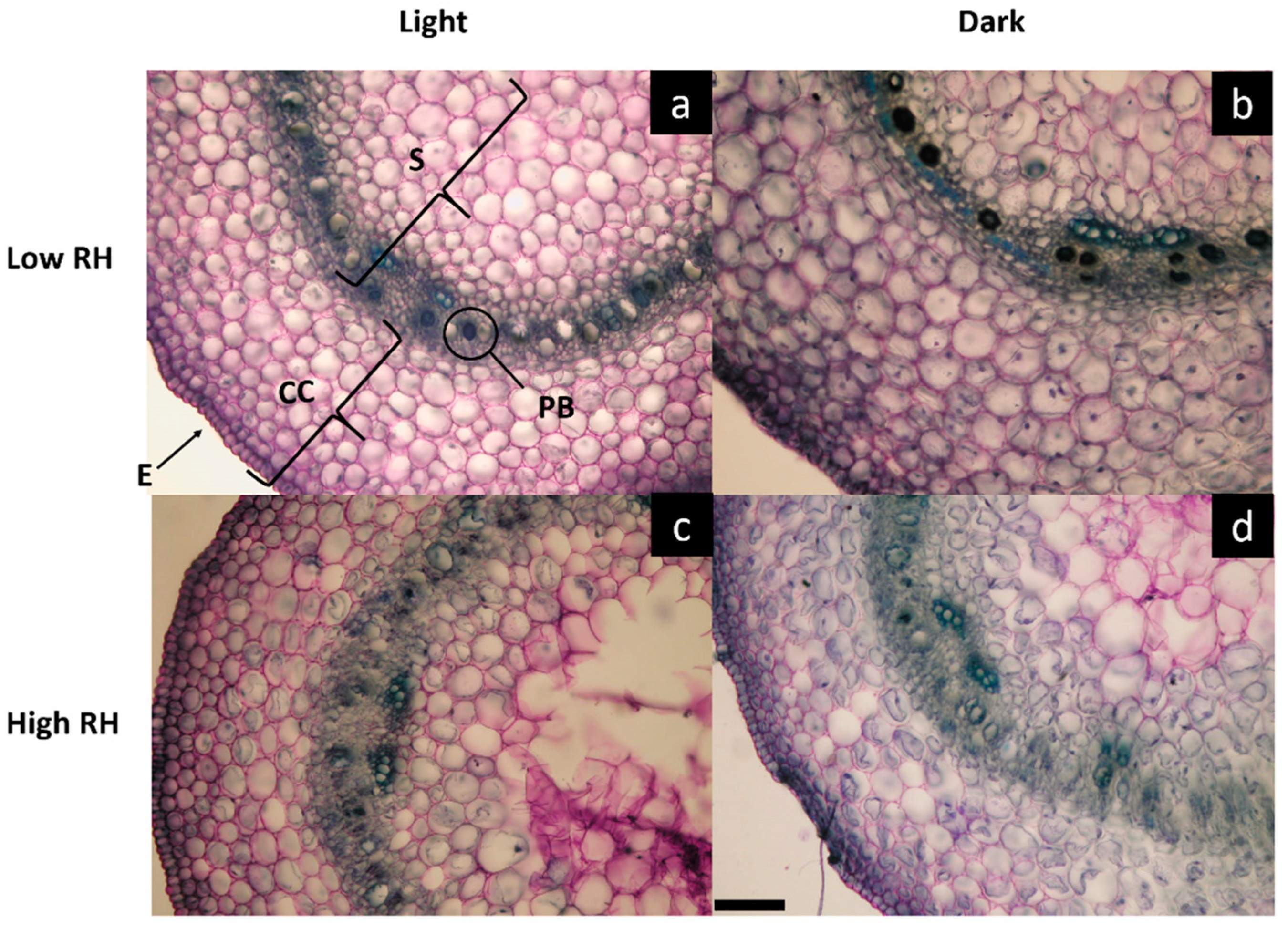
2.2. Morpho-Anatomical Analysis of Hypocotyls
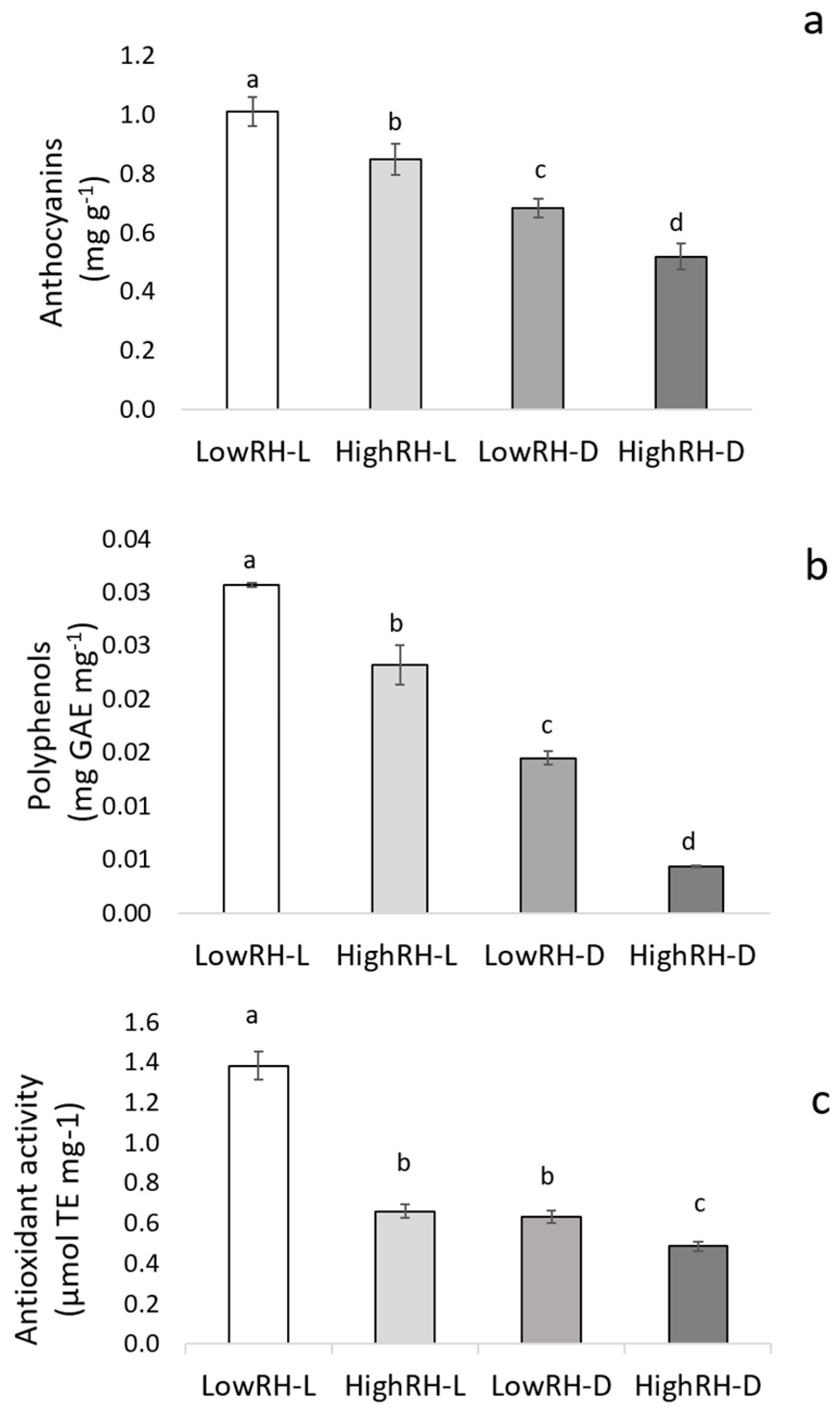
2.3. Content of Anthocyanins and Polyphenols, and FRAP Assay
2.4. Soluble Sugar and Starch Quantification
3. Discussion
4. Materials and Methods
4.1. Experimental Design and Plant Material
4.2. Morpho-Anatomical Analyses
4.3. Polyphenol and Anthocyanin Content
4.4. FRAP Assay
4.5. Soluble Sugar and Starch Quantification
4.6. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- King, B.; Wong, K. The 2017 CGIAR Inspire Challenge: Innovation Strategies for Digital Agriculture. 2017. Available online: https://cgspace.cgiar.org/bitstream/handle/10568/99282/The-2017-CGIAR-InspireChallenge-3.pdf?sequence=1 (accessed on 25 August 2020).
- Amitrano, C.; Chirico, G.B.; De Pascale, S.; Rouphael, Y.; De Micco, V. Crop Management in Controlled Environment Agriculture (CEA) Systems Using Predictive Mathematical Models. Sensors 2020, 20, 3110. [Google Scholar] [CrossRef] [PubMed]
- Derkzen, M.L.; Nagendra, H.; Van Teeffelen, A.J.; Purushotham, A.; Verburg, P.H. Shifts in ecosystem services in deprived urban areas: Understanding people’s responses and consequences for well-being. Ecol. Soc. 2017, 22, 51. [Google Scholar] [CrossRef]
- Weber, C.F. Broccoli microgreens: A mineral-rich crop that can diversify food systems. Front. Nutr. 2017, 4, 7. [Google Scholar] [CrossRef] [PubMed]
- Taylor, R.; Carandang, J.; Alexander, C.; Calleja, J. Making global cities sustainable: Urban rooftop hydroponics for diversified agriculture in emerging economies. OIDA Int. J. Sustain. Dev. 2012, 5, 11–28. [Google Scholar]
- Jim, C.Y. Green-space preservation and allocation for sustainable greening of compact cities. Cities 2004, 21, 311–320. [Google Scholar] [CrossRef]
- Adeyemi, O.; Grove, I.; Peets, S.; Domun, Y.; Norton, T. Dynamic modelling of lettuce transpiration for water status monitoring. Comput. Electron. Agric. 2018, 155, 50–57. [Google Scholar] [CrossRef]
- Mark, J. Digging for the Roots of the Urban Farming Movement. Gastron. J. Food Cult. 2011, 11, 87–91. [Google Scholar] [CrossRef]
- Ulrich, R.S.; Simons, R.F.; Losito, B.D.; Fiorito, E.; Miles, M.A.; Zelson, M. Stress recovery during exposure to natural and urban environments. J. Environ. Psychol. 1991, 11, 201–230. [Google Scholar] [CrossRef]
- Lewis, C.A. Human health and well-being: The psychological, physiological, and sociological effects of plants on people. Hortic. Hum. Life Cult. Environ. 1994, 391, 31–40. [Google Scholar] [CrossRef]
- Lederbogen, F.; Kirsch, P.; Haddad, L.; Streit, F.; Tost, H.; Schuch, P.; Wüst, S.; Pruessner, J.C.; Rietschel, M.; Deuschle, M.; et al. City living and urban upbringing affect neural social stress processing in humans. Nature 2011, 474, 498–501. [Google Scholar] [CrossRef]
- Ebert, A.; Wu, T.; Yang, R. Amaranth sprouts and microgreens—A homestead vegetable production option to enhance food and nutrition security in the rural-urban continuum. In Proceedings of the Regional Symposium on Sustaining Small-Scale Vegetable Production and Marketing Systems for Food and Nutrition Security (SEAVEG 2014), Bangkok, Thailand, 25–27 February 2014; pp. 25–27. [Google Scholar]
- Kyriacou, M.C.; Rouphael, Y.; Di Gioia, F.; Kyratzis, A.; Serio, F.; Renna, M.; De Pascale, S.; Santamaria, P. Micro-scale vegetable production and the rise of microgreens. Trends Food Sci. Technol. 2016, 57, 103–115. [Google Scholar] [CrossRef]
- Adhikari, B.; Dhungana, S.K.; Ali, M.W.; Adhikari, A.; Kim, I.D.; Shin, D.H. Resveratrol, total phenolic and flavonoid contents, and antioxidant potential of seeds and sprouts of Korean peanuts. Food Sci. Biotechnol. 2018, 27, 1275–1284. [Google Scholar] [CrossRef] [PubMed]
- Alekel, D.L.; Germain, A.S.; Peterson, C.T.; Hanson, K.B.; Stewart, J.W.; Toda, T. Isoflavone-rich soy protein isolate attenuates bone loss in the lumbar spine of perimenopausal women. Am. J. Clin. Nutr. 2000, 72, 844–852. [Google Scholar] [CrossRef] [PubMed]
- Tada, O.; Yokogoshi, H. Effect of different dietary protein composition on skeletal muscle atrophy by suspension hypokinesia/hypodynamia in rats. J. Nutr. Sci. Vitaminol. 2002, 48, 115–119. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Faienza, M.F.; D’Amato, G.; Chiarito, M.; Colaianni, G.; Colucci, S.; Grano, M.; Corbo, F.; Brunetti, G. Mechanisms involved in childhood obesity-related bone fragility. Front. Endocrinol. 2019, 10, 269. [Google Scholar] [CrossRef] [PubMed]
- Amitrano, C.; Vitale, E.; De Micco, V.; Arena, C. Light fertilization affects growth and photosynthesis in mung bean (Vigna radiata) plants. J. Environ. Account. Manag. 2018, 6, 295–304. [Google Scholar] [CrossRef]
- Tang, D.; Dong, Y.; Ren, H.; Li, L.; He, C. A review of phytochemistry, metabolite changes, and medicinal uses of the common food mung bean and its sprouts (Vigna radiata). Chem. Cent. J. 2014, 8, 4. [Google Scholar] [CrossRef]
- Chance, E.; Ashton, W.; Pereira, J.; Mulrow, J.; Norberto, J.; Derrible, S.; Guilbert, S. The Plant—An experiment in urban food sustainability. Environ. Prog. Sustain. Energy 2018, 37, 82–90. [Google Scholar] [CrossRef]
- Zhang, Y.; Kacira, M. Enhancing resource use efficiency in plant factory. Acta Hortic. 2020, 1271, 307–314. [Google Scholar] [CrossRef]
- Lefers, R.M.; Bettahalli, N.S.; Fedoroff, N.V.; Ghaffour, N.; Davies, P.A.; Nunes, S.P.; Leiknes, T. Hollow fibre membrane-based liquid desiccant humidity control for controlled environment agriculture. Biosyst. Eng. 2019, 183, 47–57. [Google Scholar] [CrossRef]
- Forney, C.F.; Brandl, D.G. Control of humidity in small controlled-environment chambers using glycerol-water solutions. HortTechnology 1992, 2, 52–54. [Google Scholar] [CrossRef]
- Amitrano, C.; Arena, C.; Rouphael, Y.; De Pascale, S.; De Micco, V. Vapour pressure deficit: The hidden driver behind plant morphofunctional traits in controlled environments. Ann. Appl. Biol. 2019, 175, 313–325. [Google Scholar] [CrossRef]
- Charlebois, S. Microgreens with Big Potential. 2019. Available online: https://static1.squarespace.com/static/59a566808419c2c20ebc2768/t/5bec6f7840ec9a4b55d39143/1542221690715/Microgreens+with+big+potential_CaseStudy.pdf (accessed on 25 August 2020).
- Lal, G.; Shanmugasundaram, S. Mungbean Sprout production. In Training Manual; Asian Vegetable Research and Development Center: Shanhua, Taiwan, 2001. [Google Scholar]
- Miles, A.; Brown, M. Teaching Organic Farming and Gardening: Resources for Instructors; University of California Farm and Garden: Santa Cruz, CA, USA, 2007. [Google Scholar]
- Price, T.V. Seed Sprout Production for Human Consumption—A Review. Can. Inst. Food Sci. Technol. J. 1988, 21, 57–65. [Google Scholar] [CrossRef]
- Samuolienė, G.; Urbonavičiūtė, A.; Brazaitytė, A.; Šabajevienė, G.; Sakalauskaitė, J.; Duchovskis, P. The impact of LED illumination on antioxidant properties of sprouted seeds. Cent. Eur. J. Biol. 2011, 6, 68–74. [Google Scholar] [CrossRef]
- El-Adawy, T.; Rahma, E.; El-Bedawey, A.; El-Beltagy, A. Nutritional potential and functional properties of germinated mung bean, pea and lentil seeds. Plant Foods Hum. Nutr. 2003, 58, 1–13. [Google Scholar] [CrossRef]
- Silva, L.R.; Pereira, M.J.; Azevedo, J.; Gonçalves, R.F.; Valentão, P.; de Pinho, P.G.; Andrade, P.B. Glycine max (L.) Merr., Vigna radiata L. and Medicago sativa L. sprouts: A natural source of bioactive compounds. Food Res. Int. 2013, 50, 167–175. [Google Scholar] [CrossRef]
- Izzo, L.; Arena, C.; De Micco, V.; Capozzi, F.; Aronne, G. Light quality shapes morpho-functional traits and pigment content of green and red leaf cultivars of Atriplex hortensis. Sci. Hortic. 2019, 246, 942–950. [Google Scholar] [CrossRef]
- Paradiso, R.; De Micco, V.; Buonomo, R.; Aronne, G.; Barbieri, G.; De Pascale, S. Soilless cultivation of soybean for Bioregenerative Life-Support Systems: A literature review and the experience of the MELiSSA Project—Food characterisation Phase I. Plant Biol. 2014, 16, 69–78. [Google Scholar] [CrossRef]
- Pérez-Balibrea, S.; Moreno, D.A.; García-Viguera, C. Influence of light on health-promoting phytochemicals of broccoli sprouts. J. Sci. Food Agric. 2008, 88, 904–910. [Google Scholar] [CrossRef]
- Brazaityte, A.; Sakalauskiene, S.; Samuoliene, G.; Jankauskiene, J.; Virsile, A.; Novickovas, A.; Sirtautas, R.; Miliauskiene, J.; Vastakaite, V.; Dabasinskas, L.; et al. The effects of LED illumination spectra and intensity on carotenoid content in Brassicaceae microgreens. Food Chem. 2015, 173, 600–606. [Google Scholar] [CrossRef]
- Ma, M.; Wang, P.; Yang, R.; Gu, Z. Effects of UV-B radiation on the isoflavone accumulation and physiological-biochemical changes of soybean during germination: Physiological-biochemical change of germinated soybean induced by UV-B. Food Chem. 2018, 250, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Raicu, D.; Livardariu, O.; Maximilian, C.; BIra, A. The Evaluation of the Effect of LED-s Irradiation on Wheat Sprouts (Triticum aestivum L.). Rom. Biotechnol. Lett. 2018, 1–8. Available online: https://www.rombio.eu/docs/Raiciu%20et%20al.pdf. (accessed on 25 August 2020).
- Yuan, M.; Jia, X.; Ding, C.; Zeng, H.; Du, L.; Yuan, S.; Liu, J. Effect of fluorescence light on phenolic compounds and antioxidant activities of soybeans (Glycine max L. Merrill) during germination. Food Sci. Biotechnol. 2015, 24, 1859–1865. [Google Scholar] [CrossRef]
- Leyva, R.; Constán-Aguilar, C.; Sánchez-Rodríguez, E.; Romero-Gámez, M.; Soriano, T. Cooling systems in screenhouses: Effect on microclimate, productivity and plant response in a tomato crop. Biosyst. Eng. 2015, 129, 100–111. [Google Scholar] [CrossRef]
- Amitrano, C.; Chirico, G.B.; De Pascale, S.; Rouphael, Y.; De Micco, V. Application of a MEC model for the irrigation control in green and red-leaved lettuce in precision indoor cultivation. In Proceedings of the 2019 IEEE International Workshop on Metrology for Agriculture and Forestry (MetroAgriFor), Portici, Italy, 24–26 October 2019; pp. 196–201. [Google Scholar]
- Du, Q.; Liu, T.; Jiao, X.; Song, X.; Zhang, J.; Li, J. Leaf anatomical adaptations have central roles in photosynthetic acclimation to humidity. J. Exp. Bot. 2019, 70, 4949–4962. [Google Scholar] [CrossRef]
- Lin, P.-Y.; Lai, H.-M. Bioactive compounds in legumes and their germinated products. J. Agric. Food Chem. 2006, 54, 3807–3814. [Google Scholar] [CrossRef]
- Sousa, C.; Lopes, G.; Pereira, D.M.; Taveira, M.; Valentão, P.; Seabra, R.M.; Pereira, J.A.; Baptista, P.; Ferreres, F.; Andrade, P.B. Screening of antioxidant compounds during sprouting of Brassica oleracea L. var. costata DC. Comb. Chem. High Throughput Screen. 2007, 10, 377–386. [Google Scholar] [CrossRef]
- McIntyre, G.I.; Quick, W.A. Control of sprout growth in the potato. Effect of humidity and water supply. Can. J. Bot. 1984, 62, 2140–2145. [Google Scholar] [CrossRef]
- Eissenstat, D.M. Costs and benefits of constructing roots of small diameter. J. Plant Nutr. 1992, 15, 763–782. [Google Scholar] [CrossRef]
- De Micco, V.; Aronne, G. Morpho-Anatomical Traits for Plant Adaptation to Drought. In Plant Responses to Drought Stress: From Morphological to Molecular Features; Springer: Berlin, Germany, 2012; pp. 37–61. [Google Scholar] [CrossRef]
- Fahn, A. Some anatomical adaptations of desert plants. Phytomorphology 1964, 4, 93–101. [Google Scholar]
- Peña-Valdivia, C.B.; Sánchez-Urdaneta, A.B.; Meza Rangel, J.; Juárez Muñoz, J.; García-Nava, R.; Celis Velázquez, R. Anatomical root variations in response to water deficit: Wild and domesticated common bean (Phaseolus vulgaris L). Biol. Res. 2010, 43, 417–427. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Gamir, J.; Intrigliolo, D.S.; Primo-Millo, E.; Forner-Giner, M.A. Relationships between xylem anatomy, root hydraulic conductivity, leaf/root ratio and transpiration in citrus trees on different rootstocks. Physiol. Plant 2010, 139, 159–169. [Google Scholar] [CrossRef] [PubMed]
- McDonald, M.; Galwey, N.; Colmer, T. Similarity and diversity in adventitious root anatomy as related to root aeration among a range of wetland and dryland grass species. Plant Cell Environ. 2002, 25, 441–451. [Google Scholar] [CrossRef]
- Gu, J.; Xu, Y.; Dong, X.; Wang, H.; Wang, Z. Root diameter variations explained by anatomy and phylogeny of 50 tropical and temperate tree species. Tree Physiol. 2014, 34, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Kondo, M.; Aguilar, A.; Abe, J.; Morita, S. Anatomy of nodal roots in tropical upland and lowland rice varieties. Plant Prod. Sci. 2000, 3, 437–445. [Google Scholar] [CrossRef]
- Lux, A.; Luxová, M.; Abe, J.; Morita, S. Root cortex: Structural and functional variability and responses to environmental stress. Root Res. 2004, 13, 117–131. [Google Scholar] [CrossRef]
- Qian, H.; Liu, T.; Deng, M.; Miao, H.; Cai, C.; Shen, W.; Wang, Q. Effects of light quality on main health-promoting compounds and antioxidant capacity of Chinese kale sprouts. Food Chem. 2016, 196, 1232–1238. [Google Scholar] [CrossRef]
- Guo, X.; Li, T.; Tang, K.; Liu, R.H. Effect of germination on phytochemical profiles and antioxidant activity of mung bean sprouts (Vigna radiata). J. Agric. Food Chem. 2012, 60, 11050–11055. [Google Scholar] [CrossRef]
- Boivin, D.; Lamy, S.; Lord-Dufour, S.; Jackson, J.; Beaulieu, E.; Côté, M.; Moghrabi, A.; Barrette, S.; Gingras, D.; Béliveau, R. Antiproliferative and antioxidant activities of common vegetables: A comparative study. Food Chem. 2009, 112, 374–380. [Google Scholar] [CrossRef]
- Kim, J.-H.; Baek, M.-H.; Chung, B.Y.; Wi, S.G.; Kim, J.-S. Alterations in the photosynthetic pigments and antioxidant machineries of red pepper (Capsicum annuum L.) seedlings from gamma-irradiated seeds. J. Plant Biol. 2004, 47, 314–321. [Google Scholar] [CrossRef]
- Nikolova, M.T.; Ivancheva, S.V. Quantitative flavonoid variations of Artemisia vulgaris L. and Veronica chamaedrys L. in relation to altitude and polluted environment. Acta Biol. Szeged. 2005, 49, 29–32. [Google Scholar]
- Arena, C.; Turano, M.; Mele, B.H.; Cataletto, P.; Furia, M.; Pugliese, M.; De Micco, V. Anatomy, photochemical activity, and DNA polymorphism in leaves of dwarf tomato irradiated with X-rays. Biol. Plant. 2017, 61, 305–314. [Google Scholar] [CrossRef]
- De Micco, V.; Arena, C.; Aronne, G. Anatomical alterations of Phaseolus vulgaris L. mature leaves irradiated with X-rays. Plant Biol. 2014, 16, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Selmar, D.; Kleinwachter, M. Stress enhances the synthesis of secondary plant products: The impact of stress-related over-reduction on the accumulation of natural products. Plant Cell Physiol. 2013, 54, 817–826. [Google Scholar] [CrossRef] [PubMed]
- Hara, M.; Oki, K.; Hoshino, K.; Kuboi, T. Enhancement of anthocyanin biosynthesis by sugar in radish (Raphanus sativus) hypocotyl. Plant Sci. 2003, 164, 259–265. [Google Scholar] [CrossRef]
- Steyn, W.; Wand, S.; Holcroft, D.; Jacobs, G. Anthocyanins in vegetative tissues: A proposed unified function in photoprotection. New Phytol. 2002, 155, 349–361. [Google Scholar] [CrossRef]
- Gan, R.-Y.; Wang, M.-F.; Lui, W.-Y.; Wu, K.; Corke, H. Dynamic changes in phytochemical composition and antioxidant capacity in green and black mung bean (Vigna radiata) sprouts. Int. J. Food Sci. Technol. 2016, 51, 2090–2098. [Google Scholar] [CrossRef]
- Vinson, J.A.; Dabbagh, Y.A.; Serry, M.M.; Jang, J. Plant flavonoids, especially tea flavonols, are powerful antioxidants using an in vitro oxidation model for heart disease. J. Agric. Food Chem. 1995, 43, 2800–2802. [Google Scholar] [CrossRef]
- Shi, H.; Nam, P.K.; Ma, Y. Comprehensive profiling of isoflavones, phytosterols, tocopherols, minerals, crude protein, lipid, and sugar during soybean (Glycine max) germination. J. Agric. Food Chem. 2010, 58, 4970–4976. [Google Scholar] [CrossRef]
- Meier, H.; Reid, J. Reserve polysaccharides other than starch in higher plants. In Plant Carbohydrates I; Springer: Berlin/Heidelberg, Germany, 1982; pp. 418–471. [Google Scholar]
- Gill, P.K.; Sharma, A.D.; Singh, P.; Bhullar, S.S. Changes in germination, growth and soluble sugar contents of Sorghum bicolor (L.) Moench seeds under various abiotic stresses. Plant Growth Regul. 2003, 40, 157–162. [Google Scholar] [CrossRef]
- Lu, Y.; Gehan, J.P.; Sharkey, T.D. Daylength and Circadian Effects on Starch Degradation and Maltose Metabolism. Plant Physiol. 2005, 138, 2280–2291. [Google Scholar] [CrossRef] [PubMed]
- Kyriacou, M.C.; Rouphael, Y. Towards a new definition of quality for fresh fruits and vegetables. Sci. Hortic. 2018, 234, 463–469. [Google Scholar] [CrossRef]
- Lei, W.Q.; Khanizadeh, S.; Vigneault, C. Preharvest ways of enhancing the phytochemical content of fruits and vegetables. Stewart Postharvest Rev. 2007, 3, 1–8. [Google Scholar]
- Brückner, B.; Schonhof, I.; Kornelson, C.; Schrödter, R. Multivariate sensory profile of broccoli and cauliflower and consumer preference. Ital. J. Food Sci. 2005, 17, 17–32. [Google Scholar]
- Feder, N.; O’brien, T. Plant microtechnique: Some principles and new methods. Am. J. Bot. 1968, 55, 123–142. [Google Scholar] [CrossRef]
- Schoenwaelder, M.E. The biology of phenolic containing vesicles. Algae 2008, 23, 163–175. [Google Scholar] [CrossRef]
- Fukazawa, K. Ultraviolet microscopy. In Methods in Lignin Chemistry; Springer: Berlin/Heidelberg, Germany, 1992; pp. 110–121. [Google Scholar]
- Ruzin, S.E. Plant Microtechnique and Microscopy; Oxford University Press: New York, NY, USA, 1999; Volume 198. [Google Scholar]
- De Micco, V.; Aronne, G. Combined histochemistry and autofluorescence for identifying lignin distribution in cell walls. Biotech. Histochem. 2007, 82, 209–216. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Costanzo, G.; Iesce, M.R.; Naviglio, D.; Ciaravolo, M.; Vitale, E.; Arena, C. Comparative Studies on Different Citrus Cultivars: A Revaluation of Waste Mandarin Components. Antioxidants 2020, 9, 517. [Google Scholar] [CrossRef]
- Neff, M.M.; Chory, J. Genetic interactions between phytochrome A, phytochrome B, and cryptochrome 1 during Arabidopsis development. Plant Physiol. 1998, 118, 27–35. [Google Scholar] [CrossRef]
- Meng, X.; Xing, T.; Wang, X. The role of light in the regulation of anthocyanin accumulation in Gerbera hybrida. Plant Growth Regul. 2004, 44, 243–250. [Google Scholar] [CrossRef]
- George, B.; Kaur, C.; Khurdiya, D.; Kapoor, H. Antioxidants in tomato (Lycopersium esculentum) as a function of genotype. Food Chem. 2004, 84, 45–51. [Google Scholar] [CrossRef]
- Motta, C.M.; Simoniello, P.; Arena, C.; Capriello, T.; Panzuto, R.; Vitale, E.; Agnisola, C.; Tizzano, M.; Avallone, B.; Ferrandino, I. Effects of four food dyes on development of three model species, Cucumis sativus, Artemia salina and Danio rerio: Assessment of potential risk for the environment. Environ. Pollut. 2019, 253, 1126–1135. [Google Scholar] [CrossRef] [PubMed]
- Marshall, J. Drought and shade interact to cause fine-root mortality in Douglas-fir seedlings. Plant Soil 1986, 91, 51–60. [Google Scholar] [CrossRef]
- Sharma, D.; Pathak, D.; Atwal, A.; Sangha, M. Genetic variation for some chemical and biochemical characteristics in cotton seed oil. J. Cotton Res. 2009, 23, 1–7. [Google Scholar]
- Katoch, R. Analytical Techniques in Biochemistry and Molecular Biology; Springer Science & Business Media: Berlin, Germany, 2011. [Google Scholar]

| Treatments | Hypocotyl Length (cm) | Root Length (cm) | Total Length (cm) | Fresh Weight (g) | Dry Weight (g) |
|---|---|---|---|---|---|
| Relative humidity (RH) | |||||
| Low | 2.53 b | 0.88 b | 3.74 b | 0.22 b | 0.044 a |
| High | 3.47 a | 1.93 a | 3.95 a | 0.46 a | 0.044 a |
| Light regime (LR) | |||||
| Light | 2.69 b | 1.18 b | 3.55 b | 0.32 b | 0.048 a |
| Dark | 3.32a | 1.62 a | 4.13 a | 0.37 a | 0.046 a |
| Interaction | |||||
| LowRH × light | 2.26 a | 0.63 a | 3.40 a | 0.20 a | 0.047 a |
| HighRH × light | 3.12 a | 1.73 a | 3.71 a | 0.44 a | 0.049 a |
| LowRH × dark | 2.80 a | 1.13 a | 4.08 a | 0.25 a | 0.042 a |
| HighRH × dark | 3.83 a | 2.12 a | 4.19 a | 0.49 a | 0.042 a |
| Significance | |||||
| RH | *** | *** | *** | *** | NS |
| LR | *** | *** | *** | * | NS |
| RH × LR | NS | NS | NS | NS | NS |
| Treatments | TCC (µm) | SD (µm) | TD (µm) | CL-CC (n) | CL-S (n) | NC-CC (n mm−2) | NC-S (n mm−2) | NPB (n mm−2) | DPB (µm) |
|---|---|---|---|---|---|---|---|---|---|
| Relative humidity (RH) | |||||||||
| Low | 963.75 a | 1418.13 b | 2381.88 a | 10.05 a | 16.37 a | 25.35 a | 21.10 a | 40.07 a | 4.0 a |
| High | 817.14 b | 1554.28 a | 2371.43 a | 10.02 a | 17.82 a | 23.77 a | 18.02 b | 27.54 b | 3.75 a |
| Light regime (LR) | |||||||||
| Light | 828.46 b | 1429.33 b | 2257.80 b | 9.8 b | 17.45 a | 29.67 a | 23.10 a | 40.76 a | 4.67 a |
| Dark | 952.43 a | 1543.07 a | 2495.51 a | 10.27 a | 16.75 a | 19.45 b | 16.02 b | 26.85 b | 3.07 b |
| Interaction (RH × LR) | |||||||||
| LowRH × light | 886.56 b | 1322.80 b | 2209.36 b | 9.50 b | 16.75 a | 35.45 a | 25.50 a | 45.43 a | 5.1 a |
| HighRH × light | 770.38 c | 1535.87 a | 2306.25 b | 10.10 ab | 18.15 a | 23.90 b | 20.70 b | 36.09 b | 4.25 b |
| LowRH × dark | 1040.94 a | 1513.46 a | 2554.40 a | 10.60 a | 16.50 a | 15.25 c | 16.70 c | 34.71 b | 2.9 c |
| HighRH × dark | 863.92 bc | 1572.69 a | 2436.62 a | 10.06 ab | 17.50 a | 23.65 b | 15.35 c | 18.99 c | 3.25 c |
| Significance | |||||||||
| RH | *** | *** | NS | NS | NS | NS | *** | *** | NS |
| LR | ** | *** | *** | * | NS | *** | *** | *** | *** |
| RH × LR | * | ** | * | * | NS | *** | * | * | ** |
| Treatments | Anthocyanins (mg g−1) | Polyphenols (mg GAE mg−1) | Antioxidant Activity (µmol TE/mg−1) | Soluble Sugars (µmol (glucose) Eq. g−1) | Starch (µmol (glucose) Eq. g−1) |
|---|---|---|---|---|---|
| Relative humidity (RH) | |||||
| Low | 0.85 a | 0.022 a | 1.01 a | 0.24 b | 0.047 a |
| High | 0.68 b | 0.013 b | 0.57 b | 0.53 a | 0.027 b |
| Light regime (LR) | |||||
| Light | 0.93 a | 0.026 a | 1.02 a | 0.33 b | 0.046 a |
| Dark | 0.60 b | 0.011 b | 0.55 b | 0.44 a | 0.028 b |
| Significance | |||||
| RH | *** | *** | *** | *** | * |
| LR | *** | *** | *** | ** | * |
| RH × LR | * | * | *** | ** | * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amitrano, C.; Arena, C.; De Pascale, S.; De Micco, V. Light and Low Relative Humidity Increase Antioxidants Content in Mung Bean (Vigna radiata L.) Sprouts. Plants 2020, 9, 1093. https://doi.org/10.3390/plants9091093
Amitrano C, Arena C, De Pascale S, De Micco V. Light and Low Relative Humidity Increase Antioxidants Content in Mung Bean (Vigna radiata L.) Sprouts. Plants. 2020; 9(9):1093. https://doi.org/10.3390/plants9091093
Chicago/Turabian StyleAmitrano, Chiara, Carmen Arena, Stefania De Pascale, and Veronica De Micco. 2020. "Light and Low Relative Humidity Increase Antioxidants Content in Mung Bean (Vigna radiata L.) Sprouts" Plants 9, no. 9: 1093. https://doi.org/10.3390/plants9091093
APA StyleAmitrano, C., Arena, C., De Pascale, S., & De Micco, V. (2020). Light and Low Relative Humidity Increase Antioxidants Content in Mung Bean (Vigna radiata L.) Sprouts. Plants, 9(9), 1093. https://doi.org/10.3390/plants9091093