Effects of Soil Fertilization on Terpenoids and Other Carbon-Based Secondary Metabolites in Rosmarinus officinalis Plants: A Comparative Study
Abstract
1. Introduction
2. Material and Methods
2.1. Characteristics of the Soil and of the Composts Used as Organic Amendments
2.2. Experimental Design
2.3. Nitrogen, Phosphorus and Potassium Contents in Leaves and Stems
2.4. Terpenoid Analysis
2.5. Total Phenolics and Flavonoids
2.6. Structural Carbohydrates
2.7. Statistical Analysis
3. Results
3.1. Total N, P and K in Rosmarinus Leaves and Stems
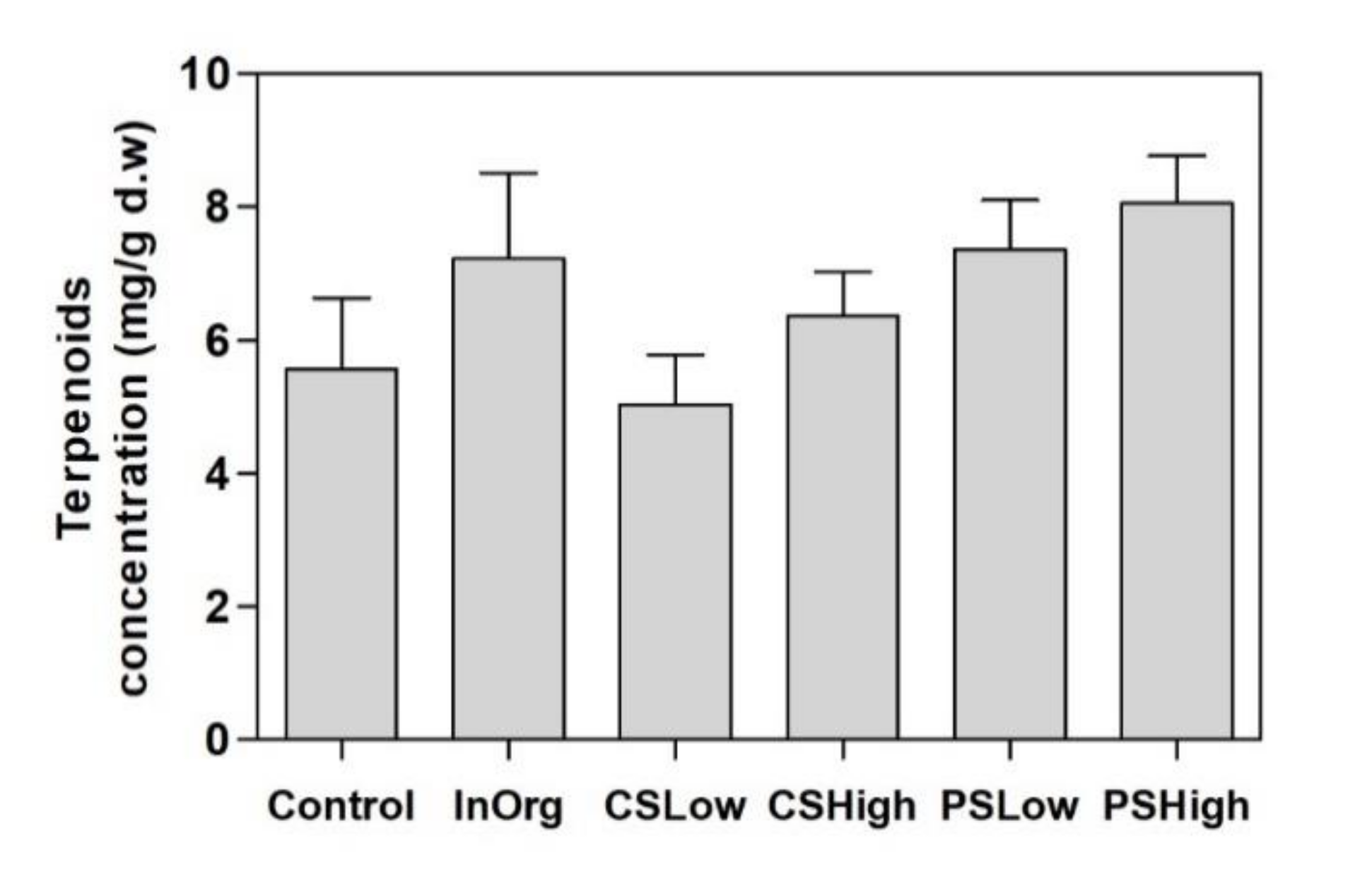
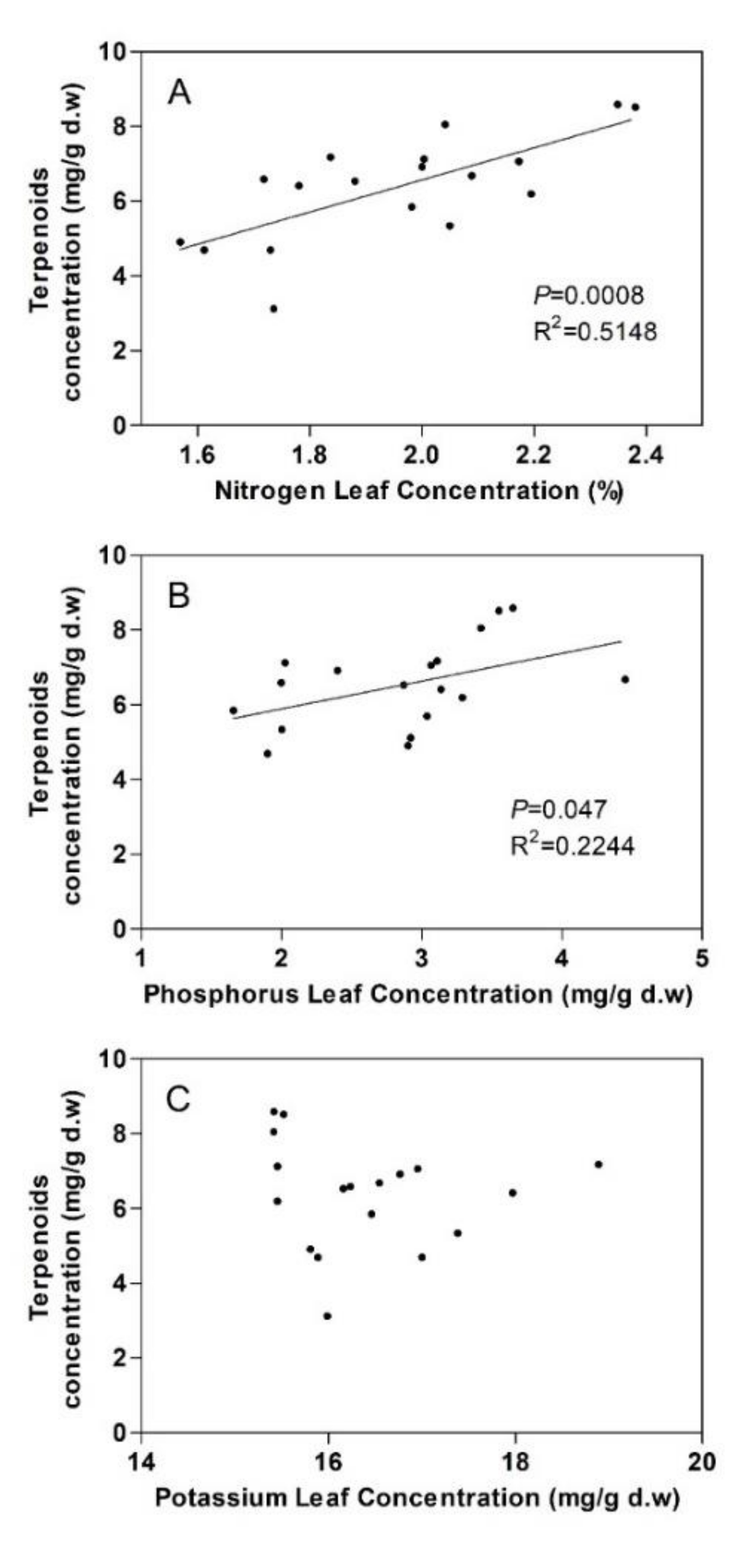
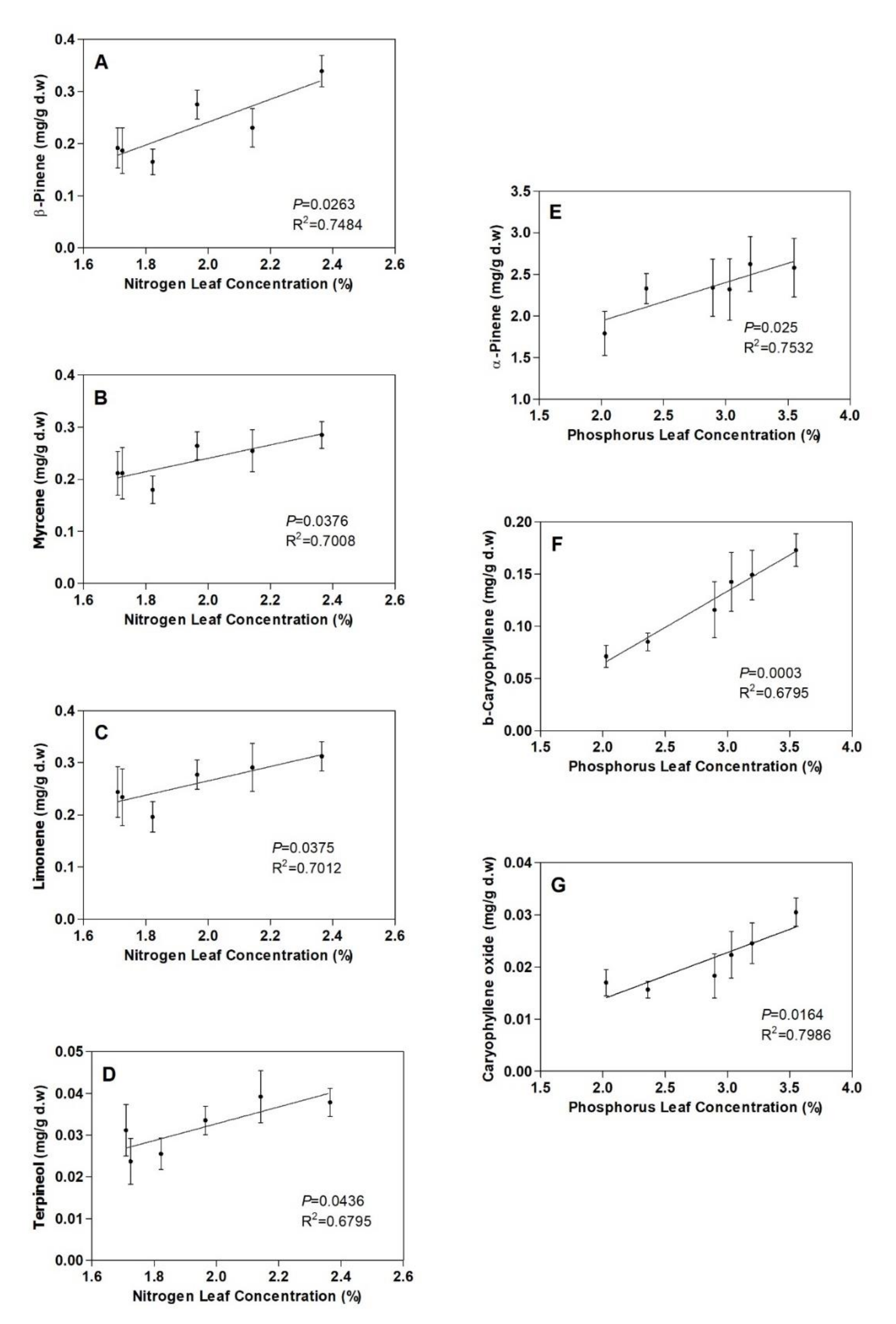
3.2. Terpenoids
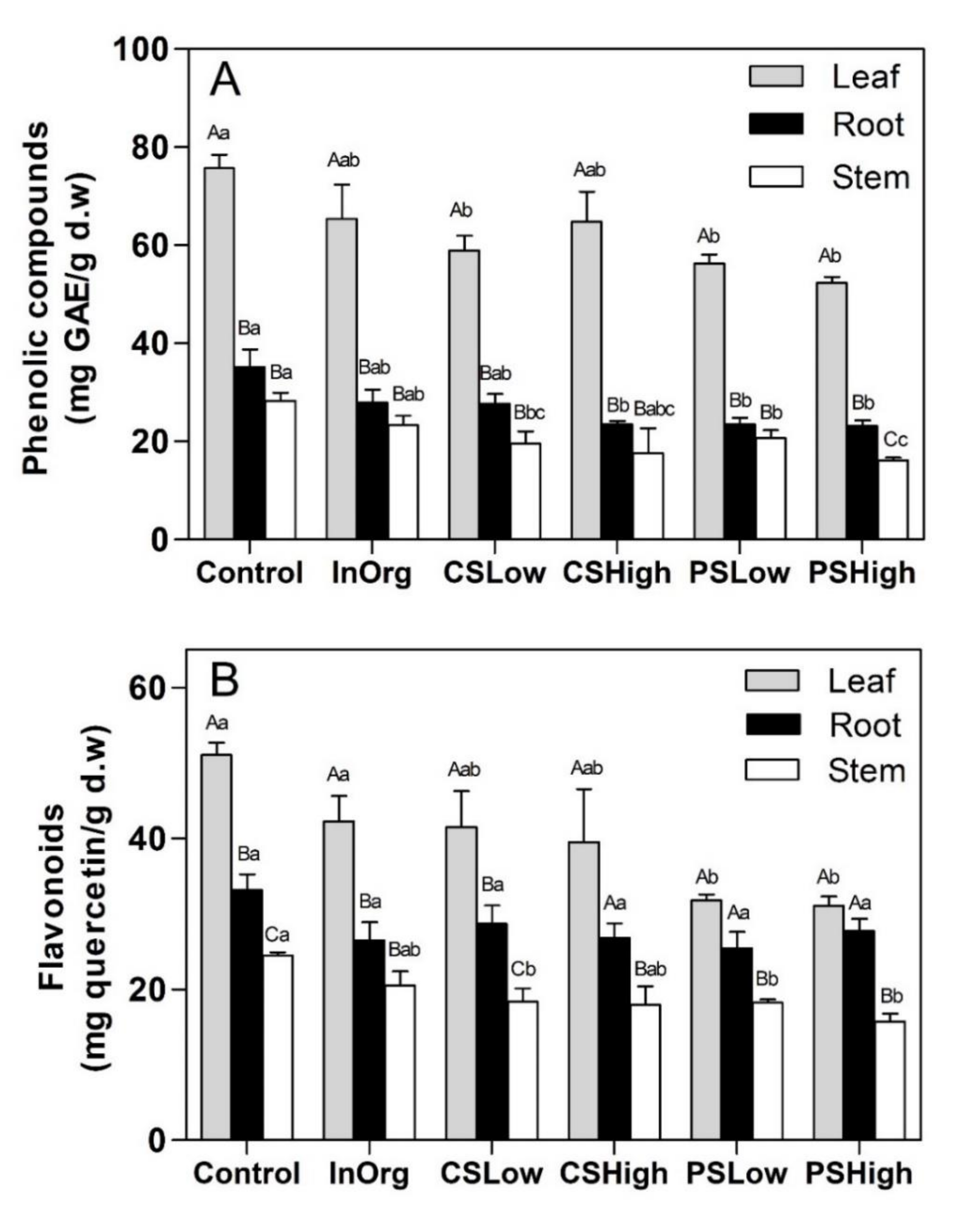
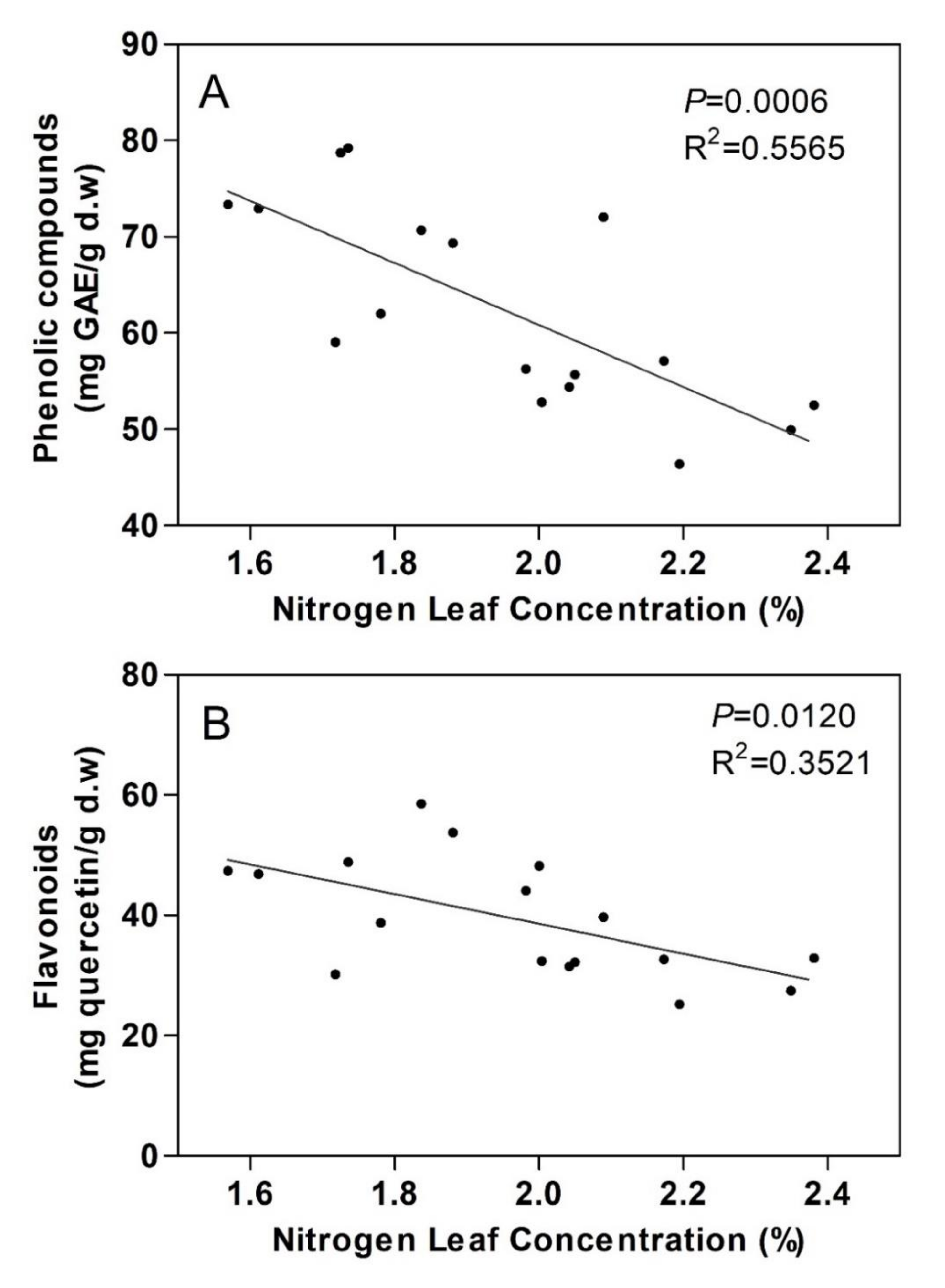
3.3. Total Phenolic and Flavonoid Content
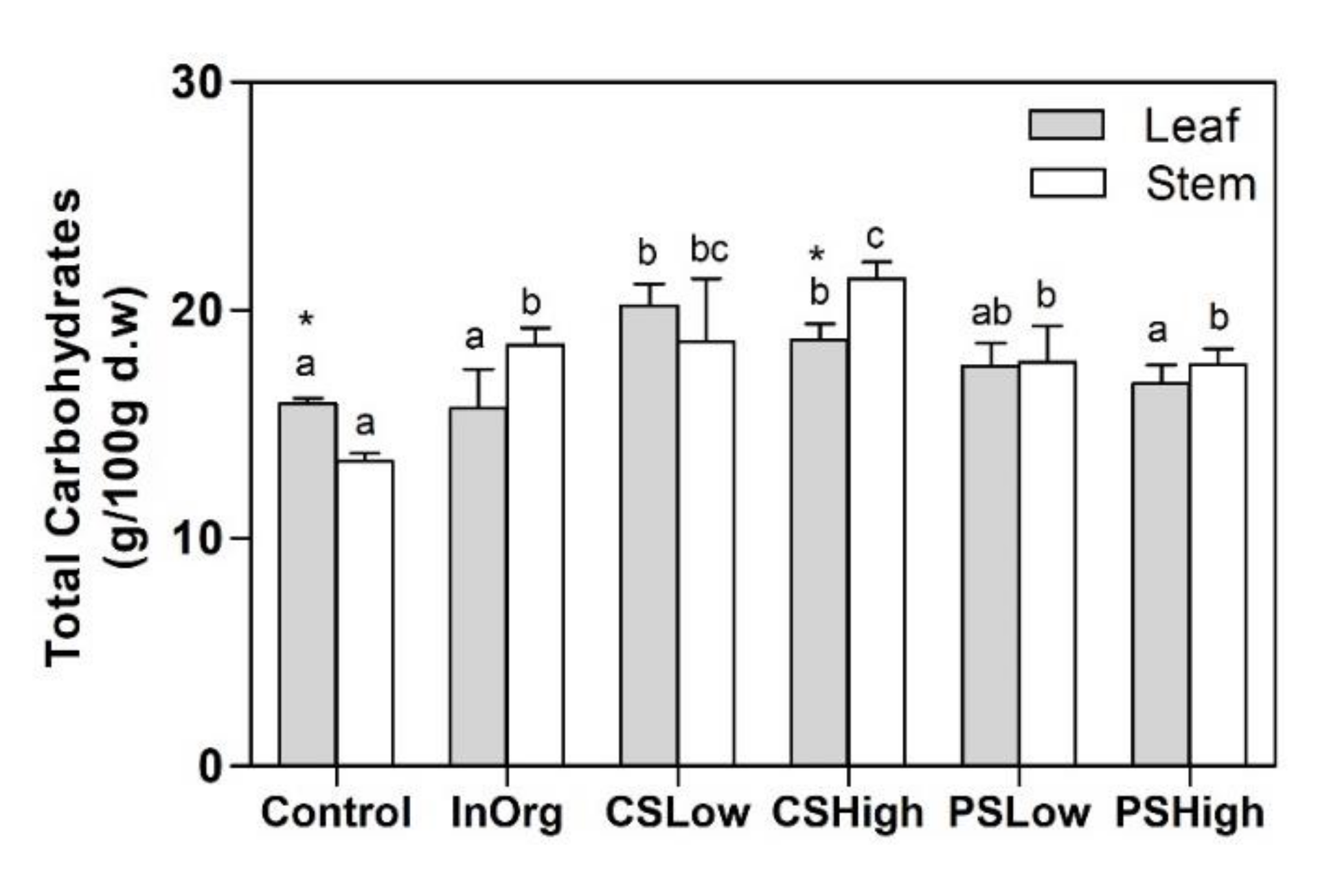
3.4. Structural Carbohydrates
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Loreto, F.; Schnitzler, J.-P. Abiotic stresses and induced BVOCs. Trends Plant. Sci. 2010, 15, 154–166. [Google Scholar] [CrossRef] [PubMed]
- Niinemets, Ü.; Kännaste, A.; Copolovici, L. Quantitative patterns between plant volatile emissions induced by biotic stresses and the degree of damage. Front. Plant Sci. 2013, 4, 262. [Google Scholar] [CrossRef] [PubMed]
- Gershenzon, J.; Dudareva, N. The function of terpene natural products in the natural world. Nat. Chem. Biol. 2007, 3, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Gouinguené, S.P.; Turlings, T.C.J. The Effects of Abiotic Factors on Induced Volatile Emissions in Corn Plants. Plant Physiol. 2002, 129, 1296–1307. [Google Scholar] [CrossRef]
- Boege, K.; Marquis, R.J. Facing herbivory as you grow up: The ontogeny of resistance in plants. Trends Ecol. Evol. 2005, 20, 441–448. [Google Scholar] [CrossRef]
- Niinemets, Ü. Mild versus severe stress and BVOCs: Thresholds, priming and consequences. Trends Plant Sci. 2010, 35, 77–99. [Google Scholar] [CrossRef]
- Loreto, F.; Pollastri, S.; Fineschi, S.; Velikova, V. Volatile isoprenoids and their importance for protection against environmental constraints in the Mediterranean area. Env. Exp. Bot. 2014, 103, 99–106. [Google Scholar] [CrossRef]
- Loreto, F.; Pinelli, P.; Manes, F.; Kollist, H. Impact of ozone on monoterpene emissions and evidence for an isoprene-like antioxidant action of monoterpenes emitted by Quercus ilex leaves. Tree Physiol. 2004, 24, 361–367. [Google Scholar] [CrossRef]
- Grote, R.; Niinemets, Ü. Modeling volatile isoprenoid emissions—A story with split ends. Plant Biol. 2008, 10, 8–28. [Google Scholar] [CrossRef]
- Vickers, C.E.; Gershenzon, J.; Lerdau, M.T.; Loreto, F. A unified mechanism of action for volatile isoprenoids in plant abiotic stress. Nat. Chem. Biol. 2009, 5, 283–291. [Google Scholar] [CrossRef]
- Gershenzon, J.; Croteau, R. Terpenoids. In Herbivores: Their Interactions with Secondary Plant Metabolites; Rosenthal, G.A., Berenbaum, M., Eds.; Academic Press: San Diego, CA, USA, 1991; pp. 165–219. ISBN 9780125971836. [Google Scholar]
- Sharifi-Rad, J.; Ezzat, S.M.; El Bishbishy, M.H.; Mnayer, D.; Sharopov, F.; Kılıç, C.S.; Neagu, M.; Constantin, C.; Sharifi-Rad, M.; Atanassova, M.; et al. Rosmarinus plants: Key farm concepts towards food applications. Phyther. Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Boix, Y.F.; Victório, C.P.; Defaveri, A.C.A.; Arruda, R.D.C.D.O.; Sato, A.; Lage, C.L.S. Glandular trichomes of Rosmarinus officinalis L.: Anatomical and phytochemical analyses of leaf volatiles. Plant Biosyst. Int. J. Deal. All Asp. Plant Biol. 2011, 145, 848–856. [Google Scholar] [CrossRef]
- Andrade, J.M.; Faustino, C.; Garcia, C.; Ladeiras, D.; Reis, C.P.; Rijo, P. Rosmarinus officinalis L.: An update review of its phytochemistry and biological activity. Futur. Sci. OA 2018, 4, FSO283. [Google Scholar] [CrossRef] [PubMed]
- Francenia Santos-Sánchez, N.; Salas-Coronado, R.; Hernández-Carlos, B.; Villanueva-Cañongo, C. Shikimic Acid Pathway in Biosynthesis of Phenolic Compounds. In Plant Physiological Aspects of Phenolic Compounds; Soto-Hernández, M., García-Mateos, R., Palma-Tenango, M., Eds.; IntechOpen: London, UK, 2019; p. 15. [Google Scholar]
- Arneth, A.; Monson, R.K.; Schurgers, G.; Niinemets, Ü.; Palmer, P.I. Why are estimates of global terrestrial isoprene emissions so similar (and why is this not so for monoterpenes)? Atmos. Chem. Phys. 2008, 8, 4605–4620. [Google Scholar] [CrossRef]
- Guenther, A.; Karl, T.; Harley, P.; Wiedinmyer, C.; Palmer, P.I.; Geron, C. Estimates of global terrestrial isoprene emissions using MEGAN (Model of Emissions of Gases and Aerosols from Nature). Atmos. Chem. Phys. 2006, 6, 3181–3210. [Google Scholar] [CrossRef]
- Monson, R.K.; Grote, R.; Niinemets, Ü.; Schnitzler, J.-P. Modeling the isoprene emission rate from leaves. New Phytol. 2012, 195, 541–559. [Google Scholar] [CrossRef]
- Nogués, I.; Fares, S.; Oksanen, E.; Loreto, F. Ozone Effects on the Metabolism and the Antioxidant System of Poplar Leaves at Different Stages of Development. In Photosynthesis. Energy from the Sun; Springer: Dordrecht, The Netherlands, 2008; pp. 1317–1321. ISBN 978-1-4020-6709-9. [Google Scholar]
- Staudt, M.; Bertin, N.; Frenzel, B.; Seufert, G. Seasonal variation in amount and composition of monoterpenes emitted by young Pinus pinea trees—Implications for emission modeling. J. Atmos. Chem. 2000, 35, 77–99. [Google Scholar] [CrossRef]
- Staudt, M.; Rambal, S.; Joffre, R.; Kesselmeier, J. Impact of drought on seasonal monoterpene emissions from Quercus ilex in southern France. J. Geophys. Res. Atmos. 2002, 107, ACH 15-1–ACH 15-9. [Google Scholar] [CrossRef]
- Plaza, J. Field monoterpene emission of Mediterranean oak (Quercus ilex) in the central Iberian Peninsula measured by enclosure and micrometeorological techniques: Observation of drought stress effect. J. Geophys. Res. 2005, 110, D03303. [Google Scholar] [CrossRef]
- Blanch, J.-S.; Peñuelas, J.; Llusià, J. Sensitivity of terpene emissions to drought and fertilization in terpene-storing Pinus halepensis and non-storing Quercus ilex. Physiol. Plant 2007, 131, 211–225. [Google Scholar] [CrossRef]
- Curci, G.; Beekmann, M.; Vautard, R.; Smiatek, G.; Steinbrecher, R.; Theloke, J.; Friedrich, R. Modelling study of the impact of isoprene and terpene biogenic emissions on European ozone levels. Atmos. Env. 2009, 43, 1444–1455. [Google Scholar] [CrossRef]
- Peñuelas, J.; Staudt, M. BVOCs and global change. Trends Plant Sci. 2010, 15, 133–144. [Google Scholar] [CrossRef] [PubMed]
- McKinney, K.A.; Lee, B.H.; Vasta, A.; Pho, T.V.; Munger, J.W. Emissions of isoprenoids and oxygenated biogenic volatile organic compounds from a New England mixed forest. Atmos. Chem. Phys. 2011, 11, 4807–4831. [Google Scholar] [CrossRef]
- Holopainen, J.K.; Gershenzon, J. Multiple stress factors and the emission of plant VOCs. Trends Plant Sci. 2010, 15, 176–184. [Google Scholar] [CrossRef]
- Bohlmann, J.; Meyer-Gauen, G.; Croteau, R. Plant terpenoid synthases: Molecular biology and phylogenetic analysis. Proc. Natl. Acad. Sci. USA 1998, 95, 4126–4133. [Google Scholar] [CrossRef] [PubMed]
- Fischbach, R.J.; Staudt, M.; Zimmer, I.; Rambal, S.; Schnitzler, J.-P. Seasonal pattern of monoterpene synthase activities in leaves of the evergreen tree Quercus ilex. Physiol. Plant 2002, 114, 354–360. [Google Scholar] [CrossRef]
- Dudareva, N.; Pichersky, E.; Gershenzon, J. Biochemistry of Plant Volatiles. Plant Physiol. 2004, 135, 1893–1902. [Google Scholar] [CrossRef]
- Ghirardo, A.; Wright, L.P.; Bi, Z.; Rosenkranz, M.; Pulido, P.; Rodríguez-Concepción, M.; Niinemets, Ü.; Brüggemann, N.; Gershenzon, J.; Schnitzler, J.-P. Metabolic Flux Analysis of Plastidic Isoprenoid Biosynthesis in Poplar Leaves Emitting and Nonemitting Isoprene. Plant Physiol. 2014, 165, 37–51. [Google Scholar] [CrossRef]
- Wright, L.P.; Rohwer, J.M.; Ghirardo, A.; Hammerbacher, A.; Ortiz-Alcaide, M.; Raguschke, B.; Schnitzler, J.-P.; Gershenzon, J.; Phillips, M.A. Deoxyxylulose 5-Phosphate Synthase Controls Flux through the Methylerythritol 4-Phosphate Pathway in Arabidopsis. Plant Physiol. 2014, 165, 1488–1504. [Google Scholar] [CrossRef]
- Kleiber, A.; Duan, Q.; Jansen, K.; Verena Junker, L.; Kammerer, B.; Rennenberg, H.; Ensminger, I.; Gessler, A.; Kreuzwieser, J. Drought effects on root and needle terpenoid content of a coastal and an interior Douglas fir provenance. Tree Physiol. 2017, 37, 1648–1658. [Google Scholar] [CrossRef]
- Llusià, J.; Peñuelas, J.; Ogaya, R.; Alessio, G. Annual and seasonal changes in foliar terpene content and emission rates in Cistus albidus L. submitted to soil drought in Prades forest (Catalonia, NE Spain). Acta Physiol. Plant. 2010, 32, 387–394. [Google Scholar]
- Hanover, J.W. Applications of terpene analysis in forest genetics. New For. 1992, 6, 159–178. [Google Scholar] [CrossRef]
- Langenheim, J.H. Higher plant terpenoids: A phytocentric overview of their ecological roles. J. Chem. Ecol. 1994, 20, 1223–1280. [Google Scholar] [CrossRef] [PubMed]
- Plomion, C.; Yani, A.; Marpeau, A. Genetic determinism of δ3-carene in maritime pine using RAPD markers. Genome 1996, 39, 1123–1127. [Google Scholar] [CrossRef]
- Madmony, A.; Tognetti, R.; Zamponi, L.; Capretti, P.; Michelozzi, M. Monoterpene responses to interacting effects of drought stress and infection by the fungus Heterobasidion parviporum in two clones of Norway spruce (Picea abies). Env. Exp. Bot. 2018, 152, 137–148. [Google Scholar] [CrossRef]
- Silvestrini, E.; Michelozzi, M.; Skroppa, T.; Brancaleoni, E.; Ciccioli, P. Characterisation of different clones of Picea abies (L.) Karst using head-space sampling of cortical tissues combined with enantioselective capillary gas chromatography for the separation of chiral and non-chiral monoterpenes. J. Chromatogr. A 2004, 1034, 183–189. [Google Scholar] [CrossRef]
- Elzinga, S.; Fischedick, J.; Podkolinski, R.; Raber, J.C. Cannabinoids and Terpenes as Chemotaxonomic Markers in Cannabis. Nat. Prod. Chem. Res. 2015, 3, 1000181. [Google Scholar]
- Barbero, F.; Maffei, M. Biodiversity and chemotaxonomic significance of specialized metabolites. In Plant Specialized Metabolism: Genomics, Biochemistry and Biological Functions; Arimura, G., Maffei, M., Eds.; CRC Press: Boca Raton, FL, USA, 2016; pp. 24–46. ISBN 9781498726283. [Google Scholar]
- Sardans, J.; Peñuelas, J.; Estiarte, M. Warming and drought alter soil phosphatase activity and soil P availability in a Mediterranean shrubland. Plant Soil 2006, 289, 227–238. [Google Scholar] [CrossRef]
- Bustamante, M.A.; Said-Pullicino, D.; Agulló, E.; Andreu, J.; Paredes, C.; Moral, R. Application of winery and distillery waste composts to a Jumilla (SE Spain) vineyard: Effects on the characteristics of a calcareous sandy-loam soil. Agric. Ecosyst. Env. 2011, 140, 80–87. [Google Scholar] [CrossRef]
- Turbé, A.; De Toni, A.; Benito, P.; Lavelle, P.; Ruiz, N.; der Putten, W.H.; Van Labouze, E.; Mudgal, S. Soil Biodiversity: Functions, Threats and Tools for Policy Makers; Report for European Commission (DG Environment); Bio Intelligence Service, IRD, and NIOO: Paris, France, 2010; ISBN 978-92-79-20668-9. [Google Scholar]
- Tejada, M.; Hernandez, M.T.; Garcia, C. Application of Two Organic Amendments on Soil Restoration: Effects on the Soil Biological Properties. J. Env. Qual. 2006, 35, 1010–1017. [Google Scholar] [CrossRef]
- Bustamante, M.A.; Alburquerque, J.A.; Restrepo, A.P.; de la Fuente, C.; Paredes, C.; Moral, R.; Bernal, M.P. Co-composting of the solid fraction of anaerobic digestates, to obtain added-value materials for use in agriculture. Biomass Bioenergy 2012, 43, 26–35. [Google Scholar] [CrossRef]
- Larchevêque, M.; Ballini, C.; Baldy, V.; Korboulewsky, N.; Ormeño, E.; Montès, N. Restoration of a Mediterranean Postfire Shrubland: Plant Functional Responses to Organic Soil Amendment. Restor. Ecol. 2010, 18, 729–741. [Google Scholar] [CrossRef]
- Niinemets, U.; Hauff, K.; Bertin, N.; Tenhunen, J.D.; Steinbrecher, R.; Seufert, G. Monoterpene emissions in relation to foliar photosynthetic and structural variables in Mediterranean evergreen Quercus species. New Phytol. 2002, 153, 243–256. [Google Scholar] [CrossRef]
- Lerdau, M.; Matson, P.; Fall, R.; Monson, R. Ecological Controls over Monoterpene Emissions from Douglas-Fir (Pseudotsuga Menziesii). Ecology 1995, 76, 2640–2647. [Google Scholar] [CrossRef]
- Bryant, J.P.; Chapin, F.S.; Klein, D.R. Carbon/Nutrient Balance of Boreal Plants in Relation to Vertebrate Herbivory. Oikos 1983, 40, 357. [Google Scholar] [CrossRef]
- Ormeño, E.; Fernandez, C. Effect of Soil Nutrient on Production and Diversity of Volatile Terpenoids from Plants. Curr. Bioact. Compd. 2012, 8, 71–79. [Google Scholar]
- Lorio, P.L. Growth-differentiation balance: A basis for understanding southern pine beetle-tree interactions. For. Ecol. Manag. 1986, 14, 259–273. [Google Scholar] [CrossRef]
- Kainulainen, P.; Utriainen, J.; Holopainen, J.K.; Oksanen, J.; Holopainen, T. Influence of elevated ozone and limited nitrogen availability on conifer seedlings in an open-air fumigation system: Effects on growth, nutrient content, mycorrhiza, needle ultrastructure, starch and secondary compounds. Glob. Chang. Biol. 2000, 6, 345–355. [Google Scholar] [CrossRef]
- Kainulainen, P.; Holopainen, J.; Palomäki, V.; Holopainen, T. Effects of nitrogen fertilization on secondary chemistry and ectomycorrhizal state of Scots pine seedlings and on growth of grey pine aphid. J. Chem. Ecol. 1996, 22, 617–636. [Google Scholar] [CrossRef]
- Heyworth, C.J.; Iason, G.R.; Temperton, V.; Jarvis, P.G.; Duncan, A.J. The effect of elevated CO2 concentration and nutrient supply on carbon-based plant secondary metabolites in Pinus sylvestris L. Oecologia 1998, 115, 344–350. [Google Scholar] [CrossRef]
- Ormeño, E.; Baldy, V.; Ballini, C.; Fernandez, C. Production and Diversity of Volatile Terpenes from Plants on Calcareous and Siliceous Soils: Effect of Soil Nutrients. J. Chem. Ecol. 2008, 34, 1219–1229. [Google Scholar] [CrossRef] [PubMed]
- Blanch, J.-S.; Peñuelas, J.; Sardans, J.; Llusià, J. Drought, warming and soil fertilization effects on leaf volatile terpene concentrations in Pinus halepensis and Quercus ilex. Acta Physiol. Plant 2009, 31, 207–218. [Google Scholar] [CrossRef]
- Massad, T.J.; Dyer, L.A.; Vega, C.G. Costs of Defense and a Test of the Carbon-Nutrient Balance and Growth-Differentiation Balance Hypotheses for Two Co-Occurring Classes of Plant Defense. PLoS ONE 2012, 7, e47554. [Google Scholar] [CrossRef]
- Bustamante, M.A.; Restrepo, A.P.; Alburquerque, J.A.; Pérez-Murcia, M.D.; Paredes, C.; Moral, R.; Bernal, M.P. Recycling of anaerobic digestates by composting: Effect of the bulking agent used. J. Clean. Prod. 2013, 47, 61–69. [Google Scholar] [CrossRef]
- Vico, A.; Sáez, J.A.; Pérez-Murcia, M.D.; Martinez-Tomé, J.; Andreu-Rodríguez, J.; Agulló, E.; Bustamante, M.A.; Sanz-Cobena, A.; Moral, R. Production of spinach in intensive Mediterranean horticultural systems can be sustained by organic-based fertilizers without yield penalties and with low environmental impacts. Agric. Syst. 2020, 178, 102765. [Google Scholar] [CrossRef]
- Velikova, V.; La Mantia, T.; Lauteri, M.; Michelozzi, M.; Nogues, I.; Loreto, F. The impact of winter flooding with saline water on foliar carbon uptake and the volatile fraction of leaves and fruits of lemon (Citrus × limon) trees. Funct. Plant Biol. 2012, 39, 199. [Google Scholar] [CrossRef]
- Fares, S.; Brilli, F.; Noguès, I.; Velikova, V.; Tsonev, T.; Dagli, S.; Loreto, F. Isoprene emission and primary metabolism in Phragmites australis grown under different phosphorus levels. Plant Biol. 2008, 10, 38–43. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Chang, C.C.; Yang, M.H.; Wen, H.M.; Chern, J.C. Estimation of total flavonoid content in propolis by two complementary colometric methods. J. Food Drug Anal. 2002, 10, 178–182. [Google Scholar]
- Nordlund, E.; Lille, M.; Silventoinen, P.; Nygren, H.; Seppänen-Laakso, T.; Mikkelson, A.; Aura, A.-M.; Heiniö, R.-L.; Nohynek, L.; Puupponen-Pimiä, R.; et al. Plant cells as food—A concept taking shape. Food Res. Int. 2018, 107, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Llusia, J.; Penuelas, J.; Alessio, G.A.; Estiarte, M. Seasonal contrasting changes of foliar concentrations of terpenes and other volatile organic compound in four dominant species of a Mediterranean shrubland submitted to a field experimental drought and warming. Physiol. Plant 2006, 127, 632–649. [Google Scholar] [CrossRef]
- Satyal, P.; Jones, T.; Lopez, E.; McFeeters, R.; Ali, N.; Mansi, I.; Al-kaf, A.; Setzer, W. Chemotypic Characterization and Biological Activity of Rosmarinus officinalis. Foods 2017, 6, 20. [Google Scholar] [CrossRef] [PubMed]
- Litvak, M.E.; Loreto, F.; Harley, P.C.; Sharkey, T.D.; Monson, R.K. The response of isoprene emission rate and photosynthetic rate to photon flux and nitrogen supply in aspen and white oak trees. Plant Cell Env. 1996, 19, 549–559. [Google Scholar] [CrossRef]
- King, D.J.; Gleadow, R.M.; Woodrow, I.E. Terpene deployment in Eucalyptus polybractea; relationships with leafstructure, environmental stresses, and growth. Funct. Plant Biol. 2004, 31, 451. [Google Scholar] [CrossRef]
- Sardans, J.; Alonso, R.; Janssens, I.A.; Carnicer, J.; Vereseglou, S.; Rillig, M.C.; Fernández-Martínez, M.; Sanders, T.G.M.; Peñuelas, J. Foliar and soil concentrations and stoichiometry of nitrogen and phosphorous across European Pinus sylvestris forests: Relationships with climate, N deposition and tree growth. Funct. Ecol. 2016, 30, 676–689. [Google Scholar] [CrossRef]
- Ganjali, A.; Kaykhaii, M. Investigating the Essential Oil Composition of Rosmarinus officinalis Before and After Fertilizing with Vermicompost. J. Essent. Oil Bear. Plants 2017, 20, 1413–1417. [Google Scholar] [CrossRef]
- Frescura, V.D.-S.; Boligon, A.A.; Barbosa, F.M.; De Souza, J.M.; Lerner, M.A.; Laughinghouse, H.D.; Da Silva, C.D.B.; Andriolo, J.L.; Lopes, S.J.; Tedesco, S.B. Nutrient solution concentration and collection time in phytomass production, content, yield and chemical composition of essential oil of rosemary. J. Plant Nutr. 2018, 41, 1293–1302. [Google Scholar] [CrossRef]
- Barra Caracciolo, A.; Bustamante, M.A.; Nogues, I.; Di Lenola, M.; Luprano, M.L.; Grenni, P. Changes in microbial community structure and functioning of a semiarid soil due to the use of anaerobic digestate derived composts and rosemary plants. Geoderma 2015, 245, 89–97. [Google Scholar] [CrossRef]
- Langenkämper, G.; Zörb, C.; Seifert, M.; Mäder, P.; Fretzdorff, B.; Betsche, T. Nutritional quality of organic and conventional wheat. J. Appl. Bot. Food Qual. 2006, 80, 150–154. [Google Scholar]
- Bénard, C.; Bourgaud, F.; Gautier, H. Impact of Temporary Nitrogen Deprivation on Tomato Leaf Phenolics. Int. J. Mol. Sci. 2011, 12, 7971–7981. [Google Scholar] [CrossRef]
- Jones, C.G.; Hartley, S.E. A Protein Competition Model of Phenolic Allocation. Oikos 1999, 86, 27. [Google Scholar] [CrossRef]
- Feller, I.C. Effects of Nutrient Enrichment on Growth and Herbivory of Dwarf Red Mangrove (Rhizophora mangle). Ecol. Monogr. 1995, 65, 477–505. [Google Scholar] [CrossRef]
- Zhang, W.; Xu, M.; Wang, X.; Huang, Q.; Nie, J.; Li, Z.; Li, S.; Hwang, S.W.; Lee, K.B. Effects of organic amendments on soil carbon sequestration in paddy fields of subtropical China. J. Soils Sediments 2012, 12, 457–470. [Google Scholar] [CrossRef]
- Wright, D.M.; Jordan, G.J.; Lee, W.G.; Duncan, R.P.; Forsyth, D.M.; Coomes, D.A. Do leaves of plants on phosphorus-impoverished soils contain high concentrations of phenolic defence compounds? Funct. Ecol. 2010, 24, 52–61. [Google Scholar] [CrossRef]
- Chrysargyris, A.; Antoniou, O.; Athinodorou, F.; Vassiliou, R.; Papadaki, A.; Tzortzakis, N. Deployment of olive-stone waste as a substitute growing medium component for Brassica seedling production in nurseries. Env. Sci. Pollut. Res. 2019, 26, 35461–35472. [Google Scholar] [CrossRef] [PubMed]
- Rioba, N.B.; Itulya, F.M.; Saidi, M.; Dudai, N.; Bernstein, N. Effects of nitrogen, phosphorus and irrigation frequency on essential oil content and composition of sage (Salvia officinalis L.). J. Appl. Res. Med. Aromat. Plants 2015, 2, 21–29. [Google Scholar] [CrossRef]
- Crocoll, C.; Asbach, J.; Novak, J.; Gershenzon, J.; Degenhardt, J. Terpene synthases of oregano (Origanum vulgare L.) and their roles in the pathway and regulation of terpene biosynthesis. Plant Mol. Biol. 2010, 73, 587–603. [Google Scholar] [CrossRef]
- Tholl, D.; Boland, W.; Hansel, A.; Loreto, F.; Röse, U.S.R.; Schnitzler, J.-P. Practical approaches to plant volatile analysis. Plant J. 2006, 45, 540–560. [Google Scholar] [CrossRef]
- Kleine, S.; Müller, C. Intraspecific plant chemical diversity and its relation to herbivory. Oecologia 2011, 166, 175–186. [Google Scholar] [CrossRef]
- Flade, J.; Beschow, H.; Wensch-Dorendorf, M.; Plescher, A.; Wätjen, W. Occurrence of Nine Pyrrolizidine Alkaloids in Senecio vulgaris L. Depending on Developmental Stage and Season. Plants 2019, 8, 54. [Google Scholar] [CrossRef]
- Fritz, C.; Palacios-Rojas, N.; Feil, R.; Stitt, M. Regulation of secondary metabolism by the carbon-nitrogen status in tobacco: Nitrate inhibits large sectors of phenylpropanoid metabolism. Plant J. 2006, 46, 533–548. [Google Scholar] [CrossRef]
- Saviranta, N.M.M.; Julkunen-Tiitto, R.; Oksanen, E.; Karjalainen, R.O. Leaf phenolic compounds in red clover (Trifolium pratense L.) induced by exposure to moderately elevated ozone. Env. Pollut. 2010, 158, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Pichersky, E.; Raguso, R.A. Why do plants produce so many terpenoid compounds? New Phytol. 2018, 220, 692–702. [Google Scholar] [CrossRef] [PubMed]
- Mote, T.E.; Villalba, J.J.; Provenza, F.D. Relative availability of Tannins-and terpene-containing foods affects food intake and preference by lambs. J. Chem. Ecol. 2007, 33, 1197–1206. [Google Scholar] [CrossRef] [PubMed]
- Chávez-González, M.L.; Rodríguez-Herrera, R.; Aguilar, C.N. Essential Oils: A Natural Alternative to Combat Antibiotics Resistance. In Antibiotic Resistance-Mechanisms and New Antimicrobial Approaches; Kon, K., Rai, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 227–237. [Google Scholar]

| Plant Part | Treatment | Total N (%) | P (g kg−1) | K (g kg−1) |
|---|---|---|---|---|
| Stem | Control | 0.69 a* ± 0.06 | 2.04 a ± 0.26 | 16.40 b ± 0.04 |
| InOrg | 0.75 a* ± 0.02 | 2.17 a ± 0.06 | 14.86 ab ± 1.71 | |
| CSLow | 0.77 ab* ± 0.13 | 2.04 a ± 0.01 | 13.80 a ± 0.24 | |
| CSHigh | 0.99 ab* ±0.10 | 2.30 a ± 0.33 | 16.61 ab ± 1.57 | |
| PSLow | 0.84 ab* ± 0.14 | 2.36 a ± 0.39 | 15.68 ab ± 0.63 | |
| PSHigh | 1.11 b* ± 0.05 | 2.64 a ± 0.05 | 19.00 c*± 1.53 | |
| Leaf | Control | 1.72 ab ± 0.16 | 2.90 a ± 0.01 | 15.95 ab* ± 0.11 |
| InOrg | 1.71 a ± 0.05 | 3.03 a* ± 0.06 | 16.99 bc ± 0.57 | |
| CSLow | 1.82 ab ± 0.20 | 2.03 a ± 0.37 | 16.87 c*± 0.27 | |
| CSHigh | 2.14 b ±0.11 | 3.20 a ± 0.09 | 16.96 abc ± 1.01 | |
| PSLow | 1.96 ab ± 0.13 | 2.36 a ± 0.35 | 16.24 abc ± 0.41 | |
| PSHigh | 2.36 b ± 0.11 | 3.55 b* ± 0.05 | 15.42 a ± 0.33 |
| Control | InOrg | CSLow | CSHigh | PSLow | PSHigh | |
|---|---|---|---|---|---|---|
| (+)-α-pinene | 39.6 ± 1.0 | 34.9 ± 0.6 | 35.7 ± 1.5 | 35.4 ± 0.8 | 37.8 ± 1.2 | 35.1 ± 1.6 |
| camphene | 8.3 ± 0.3 | 7.5 ± 0.3 | 8.0 ± 0.5 | 8.4 ± 0.5 | 7.6 ± 0.7 | 9.0 ± 0.2 |
| unknown 5 | 0.4 ± 0.1 | 0.4 ± 0 | 0.4 ± 0 | 0.4 ± 0.1 | 0.5 ± 0 | 0.6 ± 0 |
| sabinene | 0.4 ± 0.1 | 0.3 ± 0 | 0.4 ± 0 | 0.3 ± 0.1 | 0.3 ± 0 | 0.3 ± 0 |
| (+)-β-pinene | 3.2 ± 0.3 | 3.0 ± 0.3 | 3.3 ± 0.2 | 3.1 ± 0.5 | 3.7 ± 0.4 | 4.2 ± 0.3 |
| myrcene | 3.6 ± 0.1 | 3.3 ± 0.1 | 3.6 ± 0.1 | 3.4 ± 0.1 | 3.6 ± 0.1 | 3.5 ± 0.2 |
| limonene | 4.4 ± 0.2 | 4.2 ± 0.1 | 4.3 ± 0.1 | 4.3 ± 0.1 | 4.2 ± 0.1 | 4.1 ± 0.2 |
| p-cymene | 2.7 ± 0.2 | 2.8 ± 0.3 | 2.4 ± 0.2 | 2.4 ± 0.3 | 1.9 ± 0.4 | 2.3 ± 0.5 |
| cineole | 7.7 ± 0.3 | 7.9 ± 0.2 | 7.8 ± 0.2 | 7.9 ± 0.3 | 8.4 ± 0.2 | 8.5 ± 0.3 |
| γ-terpinene | 0.5 ± 0 | 0.5 ± 0 | 0.5 ± 0 | 0.5 ± 0 | 0.7 ± 0 | 0.5 ± 0 |
| terpinolene | 0.6 ± 0 | 0.8 ± 0.1 | 0.7 ± 0 | 0.6 ± 0.1 | 0.6 ± 0 | 0.7 ± 0.1 |
| linalool | 1.1 ± 0.2 | 1.2 ± 0.1 | 1.1 ± 0 | 1.2 ± 0.1 | 1.1 ± 0.2 | 1.0 ± 0.1 |
| camphor | 8.9 ± 0.6 | 9.1 ± 0.3 | 9.1 ± 0.4 | 9.6 ± 0.5 | 9.0 ± 0.6 | 9.9 ± 0.4 |
| terpinen-4-ol | 0.4 ± 0 | 0.5 ± 0 | 0.5 ± 0 | 0.5 ± 0 | 0.5 ± 0.1 | 0.5 ± 0 |
| unknown 30 | 1.0 ± 0 | 1.2 ± 0 | 1.2 ± 0 | 1.2 ± 0.1 | 1.1 ± 0.1 | 1.3 ± 0.1 |
| borneol | 2.3 ± 0.5 | 2.4 ± 0.3 | 2.7 ± 0.2 | 3.6 ± 0.5 | 1.9 ± 0.2 | 2.2 ± 0.3 |
| bornylacetate | 0.6 ± 0.2 | 0.9 ± 0 | 1.0 ± 0 | 0.9 ± 0.1 | 0.9 ± 0.1 | 0.8 ± 0.1 |
| verbenone | 11.3 ± 2.9 | 15.4 ± 0.7 | 13.6 ± 2.8 | 12.7 ± 2.6 | 13.6 ± 2.8 | 11.7 ± 3 |
| geraniol | 0.3 ± 0.1 | 0.5 ± 0 | 0.4 ± 0.1 | 0.4 ± 0.1 | 0.4 ± 0.1 | 0.4 ± 0.1 |
| unknown 37 | 0.4 ± 0.1 | 0.6 ± 0 | 0.5 ± 0.1 | 0.5 ± 0.2 | 0.4 ± 0.1 | 0.6 ± 0 |
| β-caryophyllene | 2.0 ± 0.6 | 2.2 ± 0.4 | 1.4 ± 0.5 | 2.0 ± 0.4 | 1.2 ± 0.4 | 2.1 ± 0.4 |
| caryophyllene oxide | 0.3 ± 0.1 | 0.3 ± 0.1 | 0.3 ± 0.1 | 0.3 ± 0.1 | 0.2 ± 0.1 | 0.4 ± 0.1 |
| Plant Part | Treatment | Glucose | Arabinose | Galactose | Mannose | Xylose | Rhamnose | Fucose |
|---|---|---|---|---|---|---|---|---|
| Leaf | Control | 8.18 ± 0.25 | 3.32 ± 0.26 | 2.17 ± 0.06 | 1.12 ± 0.07 | 1.05 ± 0.05 | 0.15 ± 0.05 | <0.1 |
| InOrg | 8.06 ± 0.89 | 3.16 ± 0.29 | 2.23 ± 0.29 | 1.20 ± 0.17 | 1.03 ± 0.20 | 0.55 ± 0.25 | <0.1 | |
| CSLow | 11.05 ± 1.16 | 3.17 ± 0.20 | 2.32 ± 0.31 | 1.47 ± 0.20 | 1.15 ± 0.13 | 0.20 ± 0.00 | <0.1 | |
| CSHigh | 10.45 ± 0.70 | 3.25 ± 0.09 | 2.27 ± 0.09 | 1.52 ± 0.07 | 1.17 ± 0.06 | 0.20 ± 0.00 | <0.1 | |
| PSLow | 8.87 ± 0.30 | 3.67 ± 0.33 | 2.50 ± 0.25 | 1.37 ± 0.13 | 1.05 ± 0.05 | 0.20 ± 0.10 | <0.1 | |
| PSHigh | 8.62 ± 0.38 | 3.42 ± 0.22 | 2.27 ±0.13 | 1.32 ± 0.07 | 1.15 ± 0.09 | 0.20 ± 0.10 | <0.1 | |
| Stem | Control | 21.00 ±0.70 | 1.50 ± 0.09 | 1.00 ± 0.10 | 0.85 ± 0.03 | 0.85 ± 0.09 | 0.15 ± 0.05 | <0.1 |
| InOrg | 21.40 ± 0.80 | 1.90 ± 0.11 | 1.20 ± 0.01 | 1.40 ± 0.06 | 1.22 ± 0.06 | 0.25 ± 0.05 | <0.1 | |
| CSLow | 21.80 ± 0.30 | 1.90 ± 0.33 | 1.50 ± 0.11 | 1.35 ± 0.14 | 1.22 ± 0.06 | 0.30 ± 0.10 | <0.1 | |
| CSHigh | 21.80 ± 0.30 | 2.15 ± 0.04 | 1.40 ± 0.02 | 1.70 ± 0.01 | 1.34 ± 0.04 | 0.40 ± 0.00 | <0.1 | |
| PSLow | 20.65 ± 2.35 | 2.15 ± 0.25 | 1.40 ± 0.23 | 1.40 ± 0.11 | 1.09 ± 0.05 | 0.30 ± 0.10 | <0.1 | |
| PSHigh | 20.10 ±0.02 | 1.90 ± 0.03 | 1.50 ± 0.20 | 1.40 ± 0.06 | 1.18 ± 0.04 | 0.35 ± 0.05 | <0.1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bustamante, M.Á.; Michelozzi, M.; Barra Caracciolo, A.; Grenni, P.; Verbokkem, J.; Geerdink, P.; Safi, C.; Nogues, I. Effects of Soil Fertilization on Terpenoids and Other Carbon-Based Secondary Metabolites in Rosmarinus officinalis Plants: A Comparative Study. Plants 2020, 9, 830. https://doi.org/10.3390/plants9070830
Bustamante MÁ, Michelozzi M, Barra Caracciolo A, Grenni P, Verbokkem J, Geerdink P, Safi C, Nogues I. Effects of Soil Fertilization on Terpenoids and Other Carbon-Based Secondary Metabolites in Rosmarinus officinalis Plants: A Comparative Study. Plants. 2020; 9(7):830. https://doi.org/10.3390/plants9070830
Chicago/Turabian StyleBustamante, Maria Ángeles, Marco Michelozzi, Anna Barra Caracciolo, Paola Grenni, Janine Verbokkem, Peter Geerdink, Carl Safi, and Isabel Nogues. 2020. "Effects of Soil Fertilization on Terpenoids and Other Carbon-Based Secondary Metabolites in Rosmarinus officinalis Plants: A Comparative Study" Plants 9, no. 7: 830. https://doi.org/10.3390/plants9070830
APA StyleBustamante, M. Á., Michelozzi, M., Barra Caracciolo, A., Grenni, P., Verbokkem, J., Geerdink, P., Safi, C., & Nogues, I. (2020). Effects of Soil Fertilization on Terpenoids and Other Carbon-Based Secondary Metabolites in Rosmarinus officinalis Plants: A Comparative Study. Plants, 9(7), 830. https://doi.org/10.3390/plants9070830










