Radiation Interception, Conversion and Partitioning Efficiency in Potato Landraces: How Far Are We from the Optimum?
Abstract
1. Introduction
2. Results
2.1. Relationship among Efficiencies and Physiological and Yield Traits
2.2. Characterization of the Potato Diversity Mini Core with Assessed Parameters
2.3. Efficiencies Diversity in Other Crops
3. Discussion
3.1. Relationship among Efficiencies and Other Plant Traits
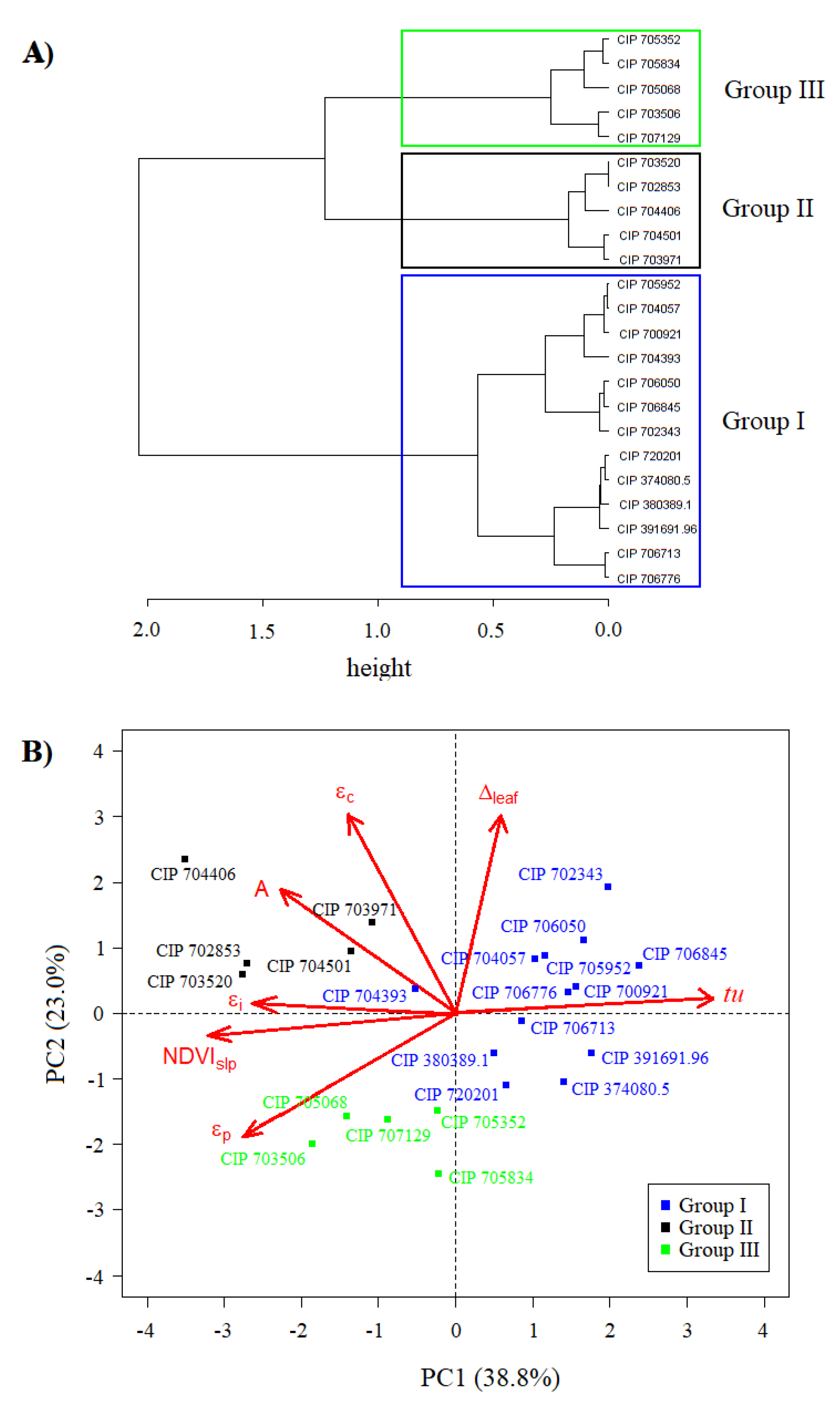
3.2. Defining the Functional Diversity of Potato Mini Core
3.3. Efficiencies in Relation to Other Crops
4. Materials and Methods
4.1. Study Site and Crop Management
4.2. Plant Material and Experimental Design
4.3. Data Acquisition
4.3.1. Remote Sensing Imagery
4.3.2. Physiological Assessment
4.4. Radiation-Related Parameters Calculation
4.4.1. Radiation Interception Efficiency ()
4.4.2. Conversion Efficiency ()
4.4.3. Partitioning Efficiency ()
4.5. Senescence Delay and Precocity Proxies
4.6. Data Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stratonovitch, P.; Semenov, M. Heat tolerance around flowering in wheat identified as a key trait for increased yield potential in Europe under climate change. J. Exp. Bot. 2015, 66, 3599–3609. [Google Scholar] [CrossRef] [PubMed]
- IPCC. Adaptation climate change of improved genetic material. In Climate Change 2014: Impacts, Adaptation, and Vulnerability. Part A: Global and Sectoral Aspects. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Field, C.B., Barros, V.R., Eds.; Cambridge University Press: New York, NY, USA, 2014; pp. 899–943. [Google Scholar]
- South, P.F.; Cavanagh, A.P.; Liu, H.W.; Ort, D.R. Synthetic glycolate metabolism pathways stimulate crop growth and productivity in the field. Plant Sci. 2019, 363, eaat9077. [Google Scholar] [CrossRef] [PubMed]
- Long, S.P.; Zhu, X.-G.; Naidu, S.L.; Ort, D.R. Can improvement in photosynthesis increase crop yields? Plant Cell Environ. 2006, 29, 315–330. [Google Scholar] [CrossRef] [PubMed]
- Monteith, J.L. Solar Radiation and Productivity in Tropical Ecosystems. J. Appl. Ecol. 1972, 9, 747–766. [Google Scholar] [CrossRef]
- Spitters, C.J.T. An analysis of variation in yield among potato cultivars in terms of light absorption, light utilization and dry matter partitioning. Agrometeorol. Potato Crop 1987, 214, 71–84. [Google Scholar] [CrossRef]
- Condori, B.; Hijmans, R.J.; Quiroz, R.; Ledent, J.F. Quantifying the expression of potato genetic diversity in the high Andes through growth analysis and modeling. Field Crop. Res. 2010, 119, 135–144. [Google Scholar] [CrossRef]
- Devaux, A.; Kromann, P.; Ortiz, O. Potatoes for Sustainable Global Food Security. Potato Res. 2014, 57, 185–199. [Google Scholar] [CrossRef]
- Huamán, Z.; Salas, A.; Gomez, R.; Panta, A.; Toledo, J. Conservation of potato genetic resources at CIP. In Potato, Global Research and Development; Khurana, S.M.P., Shekhawat, G.S., Singh, B.P., Pandey, S.K., Eds.; Indian Potato Association: Shimba, Indian, 2000; Volume 1, pp. 102–112. [Google Scholar]
- Quiroz, R.; Loayza, H.; Barreda, C.; Gavilán, C.; Posadas, A.; Ramírez, D.A. Linking process-based potato models with light reflectance data: Does model complexity enhance yield prediction accuracy? Eur. J. Agron. 2017, 82, 104–112. [Google Scholar] [CrossRef]
- Burstall, L.; Harris, P.M. The estimation of percentage light interception from leaf area index and percentage ground cover in potatoes. J. Agric. Sci. 1983, 100, 241–244. [Google Scholar] [CrossRef]
- Haverkort, A.J.; Harris, P.M. Conversion coefficients between intercepted solar radiation and tuber yields of potato crops under tropical highland conditions. Potato Res. 1986, 29, 529–533. [Google Scholar] [CrossRef]
- Borrás, L.; Maddonni, G.A.; Otegui, M.E. Leaf senescence in maize hybrids: Plant population, row spacing and kernel set effects. Field Crop. Res. 2003, 82, 13–26. [Google Scholar] [CrossRef]
- O’Connell, M.G.; O’leary, G.J.; Whitfield, D.M.; Connor, D.J. Interception of photosynthetically active radiation and radiation-use efficiency of wheat, field pea and mustard in a semi-arid environment. Field Crop. Res. 2004, 85, 111–124. [Google Scholar] [CrossRef]
- Arkebauer, T.J.; Weiss, A.; Sinclair, T.R.; Blum, A. In defense of radiation use efficiency: A response to Demetriades-Shah et al. (1992). Agric. For. Meteorol. 1994, 68, 221–227. [Google Scholar] [CrossRef]
- Stockle, C.O.; Kiniry, J.R. Variability in crop radiation-use efficiency associated with vapor-pressure deficit. Field Crop. Res. 1990, 25, 171–181. [Google Scholar] [CrossRef]
- Bouman, B.A.M.; van Kasteren, H.W.J.; Uenk, D. Standard relations to estimate ground cover and LAI of agricultural crops from reflectance measurements. Eur. J. Agron. 1992, 1, 249–262. [Google Scholar] [CrossRef]
- De Souza, A.P.; Massenburg, L.N.; Jaiswal, D.; Cheng, S.; Shekar, R.; Long, S.P. Rooting for cassava: Insights into photosynthesis and associated physiology as a route to improve yield potential. New Phytol. 2017, 213, 50–65. [Google Scholar] [CrossRef]
- Sauer, T.J.; Singer, J.W.; Prueger, J.H.; DeSutter, T.M.; Hatfield, J.L. Radiation balance and evaporation partitioning in a narrow-row soybean canopy. Agric. For. Meteorol. 2007, 145, 206–214. [Google Scholar] [CrossRef]
- Horton, P. Prospects for crop improvement through the genetic manipulation of photosynthesis: Morphological and biochemical aspects of light capture. J. Exp. Bot. 2000, 51, 475–485. [Google Scholar] [CrossRef]
- Lopes, M.S.; Reynolds, M.P. Stay-green in spring wheat can be determined by spectral reflectance measurements (normalized difference vegetation index) independently from phenology. J. Exp. Bot. 2012, 10, 3789–3798. [Google Scholar] [CrossRef]
- Ramírez, D.A.; Yactayo, W.; Gutiérrez, R.; Mares, V.; De Mendiburu, F.; Posadas, A.; Quiroz, R. Chlorophyll concentration in leaves is an indicator of potato tuber yield in water-shortage conditions. Sci. Hortic. 2014, 168, 202–209. [Google Scholar] [CrossRef]
- Li, X.; Ramírez, D.A.; Qin, J.; Dormatey, R.; Bi, Z.; Sun, C.; Wang, H.; Bai, J. Water restriction scenarios and their effects on traits in potato with different degrees of drought tolerance. Sci. Hortic. 2019, 256, 108525. [Google Scholar] [CrossRef]
- Rolando, J.L.; Ramírez, D.A.; Yactayo, W.; Monneveux, P.; Quiroz, R. Leaf greenness as a drought tolerance related trait in potato (Solanum tuberosum L.). Environ. Exp. Bot. 2015, 110, 27–35. [Google Scholar] [CrossRef]
- Thomas, H.; Smart, C.M. Crops that stay green 1. Ann. Appl. Biol. 1993, 123, 193–219. [Google Scholar] [CrossRef]
- Ramírez, D.A.; Gavilán, C.; Barreda, C.; Condori, B.; Rossel, G.; Mwanga, R.O.M.; Andrade, M.; Monneveux, P.; Anglin, N.L.; Ellis, D.; et al. Characterizing the diversity of sweetpotato through growth parameters and leaf traits: Precocity and light use efficiency as important ordination factors. S. Afr. J. Bot. 2017, 113, 192–199. [Google Scholar] [CrossRef]
- CIP Genebank Set. Available online: http://genebank.cipotato.org/gringlobal/search.aspx (accessed on 10 May 2020).
- Potato Mini Core Subset. Available online: http://genebank.cipotato.org/gringlobal/methodaccession.aspx (accessed on 10 May 2020).
- Silva, C.; Ramirez, D.; Loayza, H.; Rinza, J.; Ninanya, J. Dataset for: Ecophysiological and Remotely Sensed Measurements Taken in a Trial Testing Potential Growth of the Potato Minicore; V2. International Potato Center: Lima, Peru, 2017. [Google Scholar]
- Zhu, X.-G.; Long, S.P.; Ort, D.R. Improving Photosynthetic Efficiency for Greater Yield. Ann. Rev. Plant Biol. 2010, 61, 235–261. [Google Scholar] [CrossRef]
- Beukema, H.P.; van der Zaag, D.E. (Eds.) A Method for calculating tuber yield. In In Introduction to Potato Production; Pudoc: Wageningen, NL, USA, 1990; pp. 61–68. [Google Scholar]
- Singels, A.; Smit, M.A.; Redshaw, K.A.; Donaldson, R.A. The effect of crop start date, crop class and cultivar on sugarcane canopy development and radiation interception. Field Crop. Res. 2005, 92, 249–260. [Google Scholar] [CrossRef]
- Muchow, R.C.; Evensen, C.I.; Osgood, R.V.; Robertson, M.J. Yield Accumulation in Irrigated Sugarcane: II. Utilization of Intercepted Radiation. Agron. J. 1997, 89, 646–652. [Google Scholar] [CrossRef]
- Kapur, R.; Duttamajumder, S.K.; Srivastava, B.L.; Madhok, H.L.; Kumar, R. Harvest index and the components of biological yield in sugarcane. Indian J. Genet. 2013, 73, 386–391. [Google Scholar] [CrossRef]
- Muchow, R.C. Effect of nitrogen on yield determination in irrigated maize in tropical and subtropical environments. Field Crop. Res. 1994, 38, 1–13. [Google Scholar] [CrossRef]
- Lindquist, J.L.; Arkebauer, T.J.; Walters, D.T.; Cassman, K.G.; Dobermann, A. Maize Radiation Use Efficiency under Optimal Growth Conditions. Agron. J. 2005, 97, 72–78. [Google Scholar] [CrossRef]
- Chimonyo, V.G.; Modi, A.T.; Mabhaudhi, T. Sorghum radiation use efficiency and biomass partitioning in intercrop systems. S. Afr. J. Bot. 2018, 118, 76–84. [Google Scholar] [CrossRef]
- Kiniry, J.R.; Landivar, J.A.; Witt, M.; Gerik, T.J.; Cavero, J.; Wade, L.J. Radiation-use efficiency response to vapor pressure deficit for maize and sorghum. Field Crop. Res. 1998, 3, 265–270. [Google Scholar] [CrossRef]
- Ali, S.; Xu, Y.; Ahmad, I.; Jia, Q.; Ma, X.; Sohail, A.; Manzoor, A.; Arif, M.; Ren, X.; Cai, T.; et al. The ridge-furrow system combined with supplemental irrigation strategies to improves radiation use efficiency and winter wheat productivity in semi-arid regions of China. Agric. Water Manag. 2019, 213, 76–86. [Google Scholar] [CrossRef]
- Kemanian, A.R.; Stöckle, C.O.; Huggins, D.R. Variability of Barley Radiation-Use Efficiency. Crop Sci. 2004, 44, 1662–1672. [Google Scholar] [CrossRef]
- Li, H.L.; Luo, Y.; Ma, J.H. Radiation-use efficiency and the harvest index of winter wheat at different nitrogen levels and their relationships to canopy spectral reflectance. Crop Pasture Sci. 2011, 62, 208–217. [Google Scholar] [CrossRef]
- Mitchell, P.L.; Sheehy, J.E.; Woodward, F.I. Potential yields and the efficiency of radiation use in rice. IRRI Discuss. Pap. Ser. 1998, 32, 69. [Google Scholar]
- Danso, E.O.; Yakubu, A.; Darrah, Y.O.K.; Arthur, E.; Manevski, K.; Sabi, E.B.; Abenney-Mickson, S.; Ofori, K.; Plauborg, F.; Andersen, M.N. Impact of rice straw biochar and irrigation on maize yield, intercepted radiation and water productivity in a tropical sandy clay loam. Field Crop. Res. 2019, 243, 107628. [Google Scholar] [CrossRef]
- Koester, R.P.; Skoneczka, J.A.; Cary, T.R.; Diers, B.W.; Ainsworth, E.A. Historical gains in soybean (Glycine max Merr.) seed yield are driven by linear increases in light interception, energy conversion, and partitioning efficiencies. J. Exp. Bot. 2014, 65, 3311–3321. [Google Scholar] [CrossRef]
- Martin, R.J. Radiation interception and growth of sugar beet at different sowing dates in Canterbury. N. Z. J. Agric. Res. 1986, 29, 381–390. [Google Scholar] [CrossRef]
- Malnou, C.S.; Jaggard, K.W.; Sparkes, D.L. Nitrogen fertilizer and the efficiency of the sugar beet crop in late sum-mer. Eur. J. Agron. 2008, 28, 47–56. [Google Scholar] [CrossRef]
- Lamani, K.D.; Halikatti, S.I. Performance of Sugar Beet (Beeta vulgaris) to Different Dates of Sowing under Temperature Regime. Int. J. Plant Soil Sci. 2019, 27, 1–12. [Google Scholar] [CrossRef]
- Leepipatpaiboon, S.; Boonyawat, S.; Sarobol, E. Estimation of solar radiation use efficiency in paddy and cassava fields. Nat. Sci. 2009, 43, 642–649. [Google Scholar]
- Okpara, D.A.; Okon, O.E.; Ekeleme, F. Optimizing nitrogen fertilization for production of white and orange-fleshed sweet potato in Southeast Nigeria. J. Plant Nutr. 2009, 32, 878–891. [Google Scholar] [CrossRef]
- Nwankwo, I.I.M.; Akinbo, O.K.; Ikoro, A.I.; Orji, N.A.C.; Njoku, T.C. Evaluation of selected sweet potato landraces for high harvest index and high root yield indices for parental selection. Int. J. Agric. Policy Res. 2018, 6, 90–97. [Google Scholar]
- Dermody, O.; Long, S.P.; McConnaugghay, K.; DeLucia, E.H. How do elevated CO2 and O3 affect the interception and utilization of radiation by a soybean canopy? Glob. Chang. Biol. 2008, 14, 556–564. [Google Scholar] [CrossRef]
- Tourneux, C.; Devaux, A.; Camacho, M.; Mamani, P.; Ledent, J.F. Effects of water shortage on six potato genotypes in the highlands of Bolivia (I): Morphological parameters, growth and yield. Agronomie 2003, 23, 169–179. [Google Scholar] [CrossRef]
- Kooman, P.L.; Fahem, M.; Tegera, P.; Haverkort, A.J. Effects of climate on different potato genotypes 1. Radiation interception, total and tuber dry matter production. Eur. J. Agron. 1996, 5, 193–205. [Google Scholar] [CrossRef]
- Rinza, J.; Ramírez, D.A.; García, J.; de Mendiburu, F.; Yactayo, W.; Barreda, C.; Velasquez, T.; Mejía, A.; Quiroz, R. Infrared Radiometry as a Tool for Early Water Deficit Detection: Insights into Its Use for Establishing Irrigation Calendars for Potatoes Under Humid Conditions. Potato Res. 2019, 62, 109–122. [Google Scholar] [CrossRef]
- Burstall, L.; Harris, P.M. The physiological basis for mixing varieties and seed ’ages’ in potato crops. J. Agric. Sci. 1986, 106, 411–418. [Google Scholar] [CrossRef]
- de la Casa, A.; Ovando, G.; Bressanini, L.; Martínez, J.; Rodríguez, A. Eficiencia en el uso de la radiación en papa, estimada a partir de la cobertura del follaje. Agriscientia 2011, 28, 21–30. [Google Scholar] [CrossRef]
- Van Der Zaag, D.E.; Doornbos, J.H. An atempt to explain differences in the yielding ability of potato cultivars based on differences in cumulative light interception, utilization efficiency of foliage and harvest index. Potato Res. 1987, 30, 551–568. [Google Scholar] [CrossRef]
- Levy, D.; Livesku, L.; Van Der Zaag, D.E. Double cropping of potatoes in a semi-arid environment: The association of ground cover with tuber yields. Potato Res. 1987, 29, 437–449. [Google Scholar] [CrossRef]
- Condori, B.; Hijmans, R.J.; Ledent, J.F.; Quiroz, R. Managing potato biodiversity to cope with frost risk in the high Andes: A modeling perspective. PLoS ONE 2014, 9, e81510. [Google Scholar] [CrossRef] [PubMed]
- Haverkort, A.J.; Uenk, D.; Veroude, H.; Van De Waart, M. Relationships between ground cover, intercepted solar radiation, leaf area index and infrared reflectance of potato crops. Potato Res. 1991, 34, 113–121. [Google Scholar] [CrossRef]
- Ramírez, D.A.; Rolando, J.L.; Yactayo, W.; Monneveux, P.; Quiroz, R. Is discrimination of 13C in potato leaflets and tubers an appropriate trait to describe genotype responses to restrictive and well-watered conditions? J. Agron. Crop Sci. 2015, 201, 410–418. [Google Scholar] [CrossRef]
- Ramírez, D.A.; Yactayo, W.; Rens, L.R.; Rolando, J.L.; Palacios, S.; de Mendiburu, F.; Mares, V.; Barreda, C.; Loayza, H.; Monneveux, P.; et al. Defining biological thresholds associated to plant water status for monitoring water restriction effects: Stomatal conductance and photosynthesis recovery as key indicators in potato. Agric. Water Manag. 2016, 117, 369–378. [Google Scholar] [CrossRef]
- Paula, F.L.M.D.; Streck, N.A.; Heldwein, A.B.; Bisognin, D.A.; Paula, A.L.D.; Dellai, J. Thermal time of some developmental phases in potato. (Solanum tuberosum L.). Ciência Rural 2005, 35, 1034–1042. [Google Scholar] [CrossRef]
- Yactayo, W.; Ramírez, D.A.; Gutiérrez, R.; Mares, V.; Posadas, A.; Quiroz, R. Effect of partial root-zone drying irrigation timing on potato tuber yield and water use efficiency. Agric. Water Manag. 2013, 123, 65–70. [Google Scholar] [CrossRef]
- Abdelrahman, M.; El-Sayed, M.; Jogaiah, S.; Burritt, D.J.; Tran, L.-S.P. The “STAY-GREEN” trait and phytohormone signaling networks in plants under heat stress. Plant Cell Rep. 2017, 36, 1009–1025. [Google Scholar] [CrossRef]
- Huamán, Z.; Ortiz, R.; Gomez, R. Selecting a Solanum tuberosum subsp. andigena core collection using morphological, geographical, disease and pest descriptors. Am. J. Potato Res. 2000, 77, 183–190. [Google Scholar] [CrossRef]
- Ochoa, C.M. Potato landraces in Peru. In Las Papas del Peru, Base de Datos 1947-1997 (No. F70 O23); Centro Internacional de la Papa (CIP), UNALM and COSUDE: Lima, Peru, 2003; 185p. [Google Scholar]
- Faostat. Available online: http://www.fao.org/faostat/en/ (accessed on 6 May 2020).
- Cucho-Padin, G.; Loayza, H.; Palacios, S.; Balcazar, M.; Carbajal, M.; Quiroz, R. Development of low-cost remote sensing tools and methods for supporting smallholder agriculture. Appl. Geomat. 2019, 1–7. [Google Scholar] [CrossRef]
- Jefferies, R.A.; Mackerron, D.K.L. Radiation interception and growth of irrigated and droughted potato (Solanum tuberosum). Field Crop. Res. 1989, 22, 101–112. [Google Scholar] [CrossRef]
- Condori, B.; Quiroz, R.; Barreda, C.; Gavilan, C.; Guerrero, J.; Osorio, J. Solanum: A Potato Production Simulation Model; Version, 2.4.3; International Potato Center: Lima, Peru, 2017. [Google Scholar]
- Harahagazwe, D.; Condori, B.; Barreda, C.; Bararyenya, A.; Byarugaba, A.A.; Kude, D.A.; Lung’aho, C.; Martinho, C.; Mbiri, D.; Nasona, B.; et al. How big is the potato (Solanum tuberosum L.) yield gap in Sub-Saharan Africa and why? A participatory approach. Open Agric. 2018, 3, 180–189. [Google Scholar] [CrossRef]
- Yin, X. A Flexible Sigmoid Function of Determinate Growth. Ann. Bot. Lond. 2003, 91, 361–371. [Google Scholar] [CrossRef]
- Sinclair, T.R.; Muchow, R.C. Radiation Use Efficiency. In Advances in Agronomy; Sparks, D.L., Ed.; Academic Press: Cambridge, MA, USA, 1999; pp. 215–265. [Google Scholar]
- de Mendiburu, F.; Package ’Agricolae’. R Package Version 1.2-2. Available online: http://tarwi.lamolina.edu.pe/~fmendiburu (accessed on 22 November 2019).
- Husson, F.; Josse, J.; Le, S.; Mazet, J.; Husson, M.F. Package ’FactoMineR’. R Package Version, 2.3. Available online: http://factominer.free.fr (accessed on 27 February 2020).
- R Core Team. R Software. Version, 5.3.3. Available online: https://www.r-project.org/ (accessed on 22 November 2019).
- Scopus. Available online: https://www.scopus.com/ (accessed on 8 May 2020).
- Google Scholar. Available online: https://scholar.google.com/ (accessed on 8 May 2020).
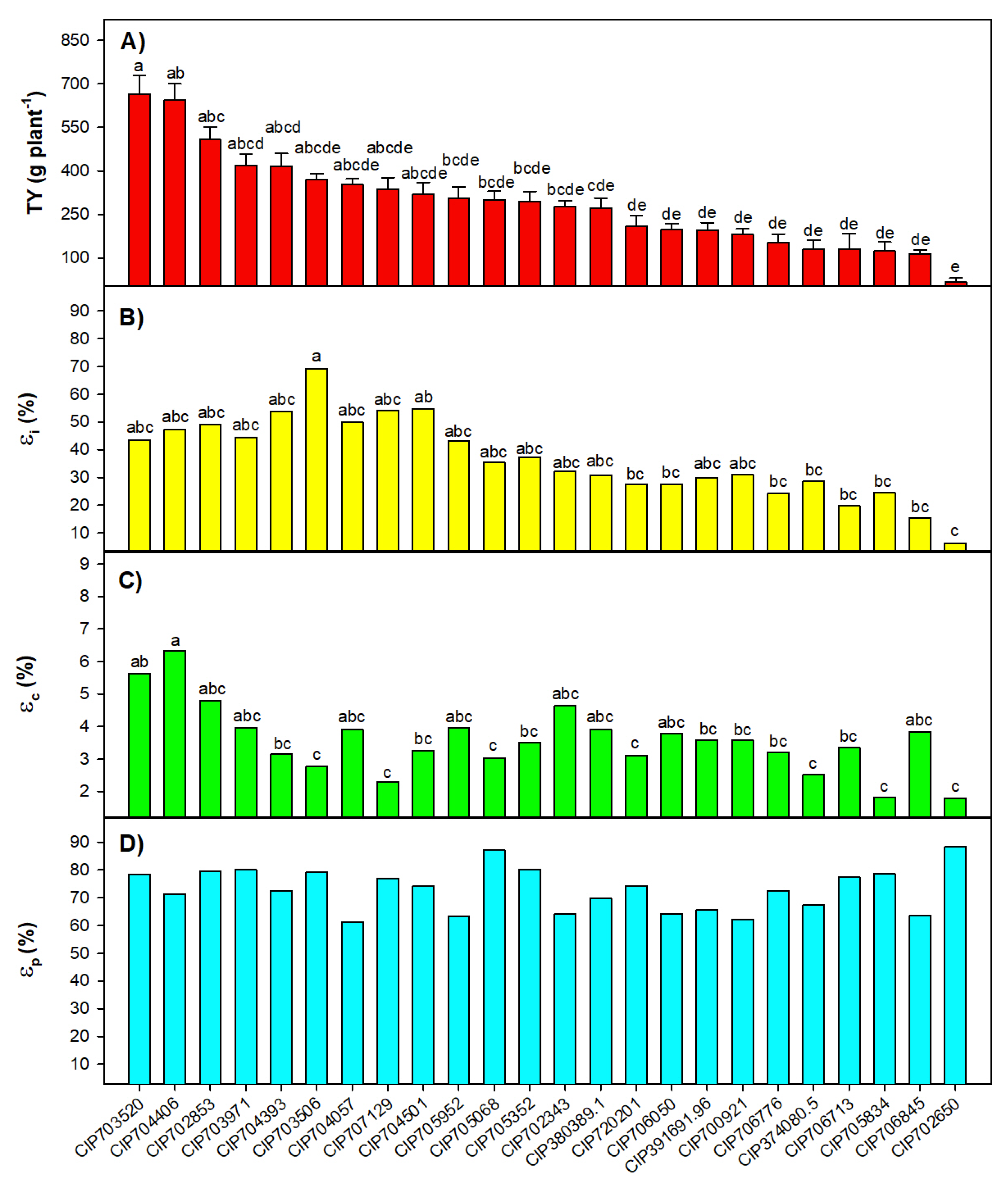
| Trait | Check | Augmented | Check × Augmented |
|---|---|---|---|
| TY (g plant) | 5.24 * | 11.7 *** | 38.1 *** |
| (%) | 0.17 n.s. | 4.62 * | 14.5 ** |
| (%) | 7.80 ** | 5.78 ** | 5.01 n.s. |
| (%) | 1.53 n.s. | 1.35 n.s. | 3.78 n.s. |
| A (mol CO m s) | 3.20 n.s. | 4.9 ** | 15.3 ** |
| (‰) | 0.88 n.s. | 2.02 n.s. | 15.8 ** |
| NDVI | 1.74 n.s. | 2.31 n.s. | 3.04 n.s. |
| (C d) | 0.30 n.s. | 1.41 n.s. | 3.5 n.s. |
| Accession Number | A (mol CO m s) | (‰) | NDVI () | (C day) |
|---|---|---|---|---|
| CIP 700921 | 20.4 ± 2.36 | 22 ± 0.4 | −0.41 | 531 |
| CIP 702343 | 21.0 ± 2.08 | 23 ± 0.2 | −0.51 | 556 |
| CIP 702650 | - | - | −0.50 | 376 |
| CIP 702853 | 24.7 ± 1.14 | 22 ± 0.2 | −0.31 | 391 |
| CIP 703506 | 19.1 ± 1.58 | 21 ± 0.0 | −0.31 | 431 |
| CIP 703520 | 24.9 ± 0.82 | 21 ± 0.1 | −0.31 | 396 |
| CIP 703971 | 26.2 ± 1.24 | 23 ± 0.6 | −0.44 | 476 |
| CIP 704057 | 21.3 ± 1.94 | 22 ± 0.4 | −0.44 | 556 |
| CIP 704393 | 20.7 ± 2.25 | 23 ± 0.0 | −0.34 | 476 |
| CIP 704406 | 26.6 ± 0.58 | 22 ± 0.1 | −0.24 | 356 |
| CIP 704501 | 24.4 ± 1.83 | 23 ± 0.3 | −0.44 | 396 |
| CIP 705068 | 23.6 ± 1.56 | 21 ± 0.2 | −0.44 | 416 |
| CIP 705352 | 19.5 ± 2.28 | 21 ± 0.5 | −0.41 | 471 |
| CIP 705834 | 20.3 ± 1.83 | 21 ± 0.1 | −0.34 | 436 |
| CIP 705952 | 21.7 ± 1.79 | 22 ± 0.2 | −0.41 | 556 |
| CIP 706050 | 24.3 ± 1.85 | 22 ± 0.5 | −0.51 | 551 |
| CIP 706713 | 22.8 ± 1.31 | 22 ± 0.4 | −0.54 | 476 |
| CIP 706776 | 24.1 ± 2.48 | 22 ± 0.3 | −0.61 | 496 |
| CIP 706845 | 17.0 ± 1.59 | 23 ± 0.4 | −0.51 | 456 |
| CIP 707129 | 22.4 ± 2.02 | 21 ± 0.2 | −0.34 | 496 |
| CIP 374080.5 | 21.7 ± 2.30 | 21 ± 0.1 | −0.50 | 495 |
| CIP 380389.1 | 20.3 ± 2.20 | 21 ± 0.0 | −0.40 | 490 |
| CIP 391691.96 | 19.5 ± 1.50 | 21 ± 0.6 | −0.50 | 520 |
| CIP 720201 | 21.7 ± 1.60 | 21 ± 0.4 | −0.45 | 500 |
| TY | |||||||
|---|---|---|---|---|---|---|---|
| 0.69 | |||||||
| 0.70 | 0.12 | ||||||
| 0.27 | 0.24 | −0.21 | |||||
| 0.55 | 0.23 | 0.44 | 0.27 | ||||
| 0.06 | 0.04 | 0.29 | −0.46 | 0.16 | |||
| NDVI | 0.74 | 0.69 | 0.25 | 0.38 | 0.21 | −0.13 | NDVI |
| −0.54 | −0.03 | −0.26 | −0.62 | −0.37 | 0.08 | −0.59 |
| Trait | PC1 | PC2 | PC3 |
|---|---|---|---|
| −0.66 | 0.04 | −0.60 | |
| −0.35 | 0.77 | 0.00 | |
| −0.69 | −0.46 | 0.42 | |
| A | −0.56 | 0.46 | 0.47 |
| 0.15 | 0.76 | −0.03 | |
| NDVI | −0.80 | −0.09 | −0.47 |
| 0.83 | 0.06 | −0.25 | |
| Eigenvalue | 2.72 | 1.61 | 1.03 |
| TCV (%) | 38.8 | 61.8 | 76.5 |
| Plant Type | Crop Type | Crop | Ref | Ref | Ref | |||
|---|---|---|---|---|---|---|---|---|
| Perennial grass | Sugarcane | 95.0 | [32] | 8.0 | [33] | 81.2 | [34] | |
| C4 | Grain | Maize | 55.0 | [35] | 8.7 | [36] | 47.1 | [35] |
| Grain | Sorghum | 50.1 | [37] | 8.0 | [38] | 20.0 | [37] | |
| Grain | Winter wheat | 97.8 | [39] | 7.5 | [40] | 44.0 | [41] | |
| Grain | Rice | 80.0 | [43] | 6.6 | [42] | 62.0 | [38] | |
| Leguminous | Soybean | 89.0 | [30] | 4.3 | [44] | 60.0 | [30] | |
| C3 | Tuber root | Sugar beet | 90.0 | [45] | 6.2 | [46] | 86.0 | [47] |
| Tuber root | Cassava | 64.0 | [48] | 1.4 | [18] | 70.5 | [18] | |
| Tuber root | Sweetpotato | 91.0 | [49] | 3.4 | [26] | 46.0 | [50] | |
| Tuber | Potato | 69.3 | 6.4 | 87.0 |
| Meteorological Variables | July | August | September | October | November |
|---|---|---|---|---|---|
| Maximum temperature (C) | 19.8 ± 0.29 | 19.2 ± 0.27 | 19.0 ± 0.38 | 21.5 ± 0.30 | 22.1 ± 0.26 |
| Minimum temperature (C) | 15.3 ± 0.14 | 14.3 ± 0.10 | 14.0 ± 0.08 | 14.6 ± 0.08 | 15.2 ± 0.19 |
| Average temperature (C) | 16.8 ± 0.10 | 15.8 ± 0.10 | 15.6 ± 0.12 | 16.8 ± 0.15 | 17.7 ± 0.18 |
| Average relative humidity (%) | 85.4 ± 0.61 | 88.7 ± 0.57 | 91.2 ± 0.59 | 89.6 ± 0.46 | 86.2 ± 0.57 |
| Solar radiation (MJ m day) | 8.5 ± 0.77 | 8.8 ± 0.69 | 9.4 ± 0.96 | 16.8 ± 0.68 | 16.1 ± 0.73 |
| PAR (MJ m day) | 3.7 ± 0.27 | 3.6 ± 0.27 | 3.8 ± 0.36 | 7.2 ± 0.27 | 6.8 ± 0.29 |
| Average VPD (kPa) | 0.24 ± 0.01 | 0.18 ± 0.01 | 0.14 ± 0.01 | 0.17 ± 0.01 | 0.24 ± 0.01 |
| Maximum VPD (kPa) | 0.68 ± 0.03 | 0.60 ± 0.03 | 0.56 ± 0.05 | 0.84 ± 0.03 | 0.92 ± 0.03 |
| Accession Number | Species | Ploidy | DOI | Contry of Origin | Harvest Time (dap) | Groups |
|---|---|---|---|---|---|---|
| CIP 700921 | Solanum tuberosum subsp. andigenum | 4× | 10.18730/91RP | Peru | 121 | I |
| CIP 702343 | Solanum tuberosum subsp. andigenum | 4× | 10.18730/9CJ= | Peru | 121 | I |
| CIP 702650 | Solanum ajanhuiri | 2× | 10.18730/9EY0 | Bolivia | 121 | - |
| CIP 702853 | Solanum tuberosum subsp. andigenum | 4× | 10.18730/9GB8 | Peru | 141 | II |
| CIP 703506 | Solanum phureja | 2× | 10.18730/9R4= | Colombia | 121 | III |
| CIP 703520 | Solanum tuberosum subsp. andigenum | 4× | 10.18730/9RHB | Colombia | 141 | II |
| CIP 703971 | Solanum tuberosum subsp. andigenum | 4× | 10.18730/A4X0 | Peru | 141 | II |
| CIP 704057 | Solanum tuberosum subsp. andigenum | 4× | 10.18730/A7G9 | Ecuador | 121 | I |
| CIP 704393 | Solanum stenotomum subsp. goniocalyx | 2× | 10.18730/AGC$ | Peru | 141 | I |
| CIP 704406 | Solanum tuberosum subsp. andigenum | 4× | 10.18730/AGR9 | Peru | 141 | II |
| CIP 704501 | Solanum tuberosum subsp. andigenum | 4× | 10.18730/AKKT | Peru | 121 | II |
| CIP 705068 | Solanum tuberosum subsp. tuberosum | 4× | 10.18730/B40∼ | Chile | 141 | III |
| CIP 705352 | Solanum tuberosum subsp. andigenum | 4× | 10.18730/BC0Y | Ecuador | 141 | III |
| CIP 705834 | Solanum stenotomum subsp. stenotomum | 2× | 10.18730/BTDA | Peru | 141 | III |
| CIP 705952 | Solanum stenotomum subsp. stenotomum | 2× | 10.18730/BXK1 | Bolivia | 121 | I |
| CIP 706050 | Solanum juzepczukii | 3× | 10.18730/C09D | Peru | 141 | I |
| CIP 706713 | Solanum tuberosum subsp. andigenum | 4× | 10.18730/CJ0S | Argentina | 121 | I |
| CIP 706776 | Solanum curtilobum | 5× | 10.18730/CKS8 | Bolivia | 121 | I |
| CIP 706845 | Solanum stenotomum subsp. stenotomum | 2× | 10.18730/CNTU | Bolivia | 121 | I |
| CIP 707129 | Solanum chaucha | 3× | 10.18730/CS5* | Peru | 141 | III |
| Checks | ||||||
| CIP 374080.5 | Variety Perricholi | 4× | 10.18730/2BRK | Peru | 121 | I |
| CIP 380389.1 | Variety Canchan | 4× | 10.18730/P5MJZ | Peru | 141 | I |
| CIP 391691.96 | Variety Serranita | 4× | 10.18730/P5P8B | Peru | 121 | I |
| CIP 720201 | Variety Yungay | 4× | 10.18730/D72∼ | Peru | 141 | I |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva-Díaz, C.; Ramírez, D.A.; Rinza, J.; Ninanya, J.; Loayza, H.; Gómez, R.; Anglin, N.L.; Eyzaguirre, R.; Quiroz, R. Radiation Interception, Conversion and Partitioning Efficiency in Potato Landraces: How Far Are We from the Optimum? Plants 2020, 9, 787. https://doi.org/10.3390/plants9060787
Silva-Díaz C, Ramírez DA, Rinza J, Ninanya J, Loayza H, Gómez R, Anglin NL, Eyzaguirre R, Quiroz R. Radiation Interception, Conversion and Partitioning Efficiency in Potato Landraces: How Far Are We from the Optimum? Plants. 2020; 9(6):787. https://doi.org/10.3390/plants9060787
Chicago/Turabian StyleSilva-Díaz, Cecilia, David A. Ramírez, Javier Rinza, Johan Ninanya, Hildo Loayza, René Gómez, Noelle L. Anglin, Raúl Eyzaguirre, and Roberto Quiroz. 2020. "Radiation Interception, Conversion and Partitioning Efficiency in Potato Landraces: How Far Are We from the Optimum?" Plants 9, no. 6: 787. https://doi.org/10.3390/plants9060787
APA StyleSilva-Díaz, C., Ramírez, D. A., Rinza, J., Ninanya, J., Loayza, H., Gómez, R., Anglin, N. L., Eyzaguirre, R., & Quiroz, R. (2020). Radiation Interception, Conversion and Partitioning Efficiency in Potato Landraces: How Far Are We from the Optimum? Plants, 9(6), 787. https://doi.org/10.3390/plants9060787










