Nitric Oxide Pre-Treatment Advances Seed Germination and Alleviates Copper-Induced Photosynthetic Inhibition in Indian Mustard
Abstract
1. Introduction
2. Materials and Methods
2.1. Treatments
2.2. Determination of H2O2 Content and Lipid Peroxidation
2.3. Assay of Antioxidant Enzymes
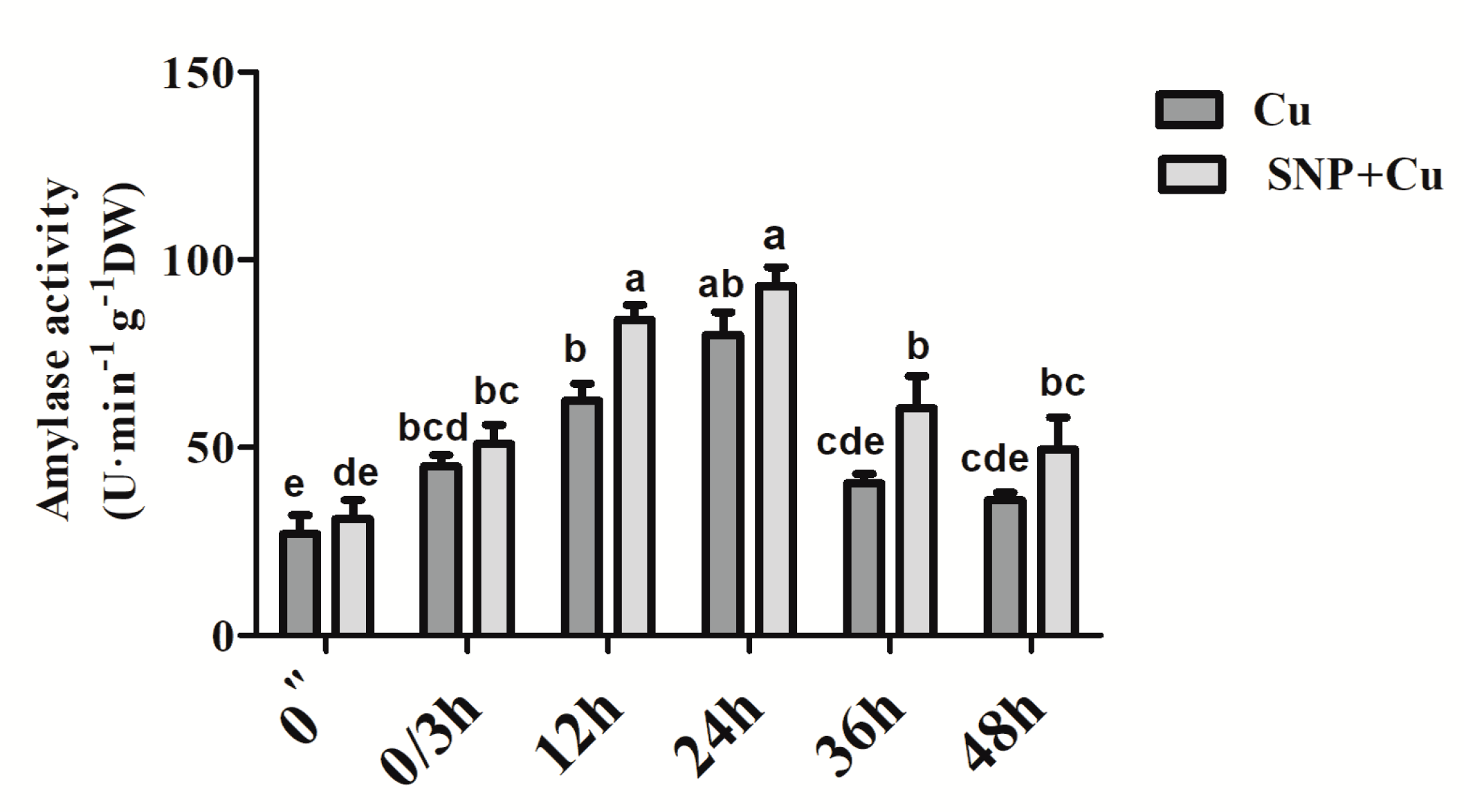
2.4. Assay of Amylase Activity
2.5. Determination of GSH Content
2.6. Histochemical Detection of Reactive Oxygen Species
2.7. Confocal Laser Microscopy Study for ROS Imaging and Cell Viability Determination
2.8. Determination of Cu Concentration
2.9. Photosynthetic Characteristics
2.10. Determination of Growth Parameters
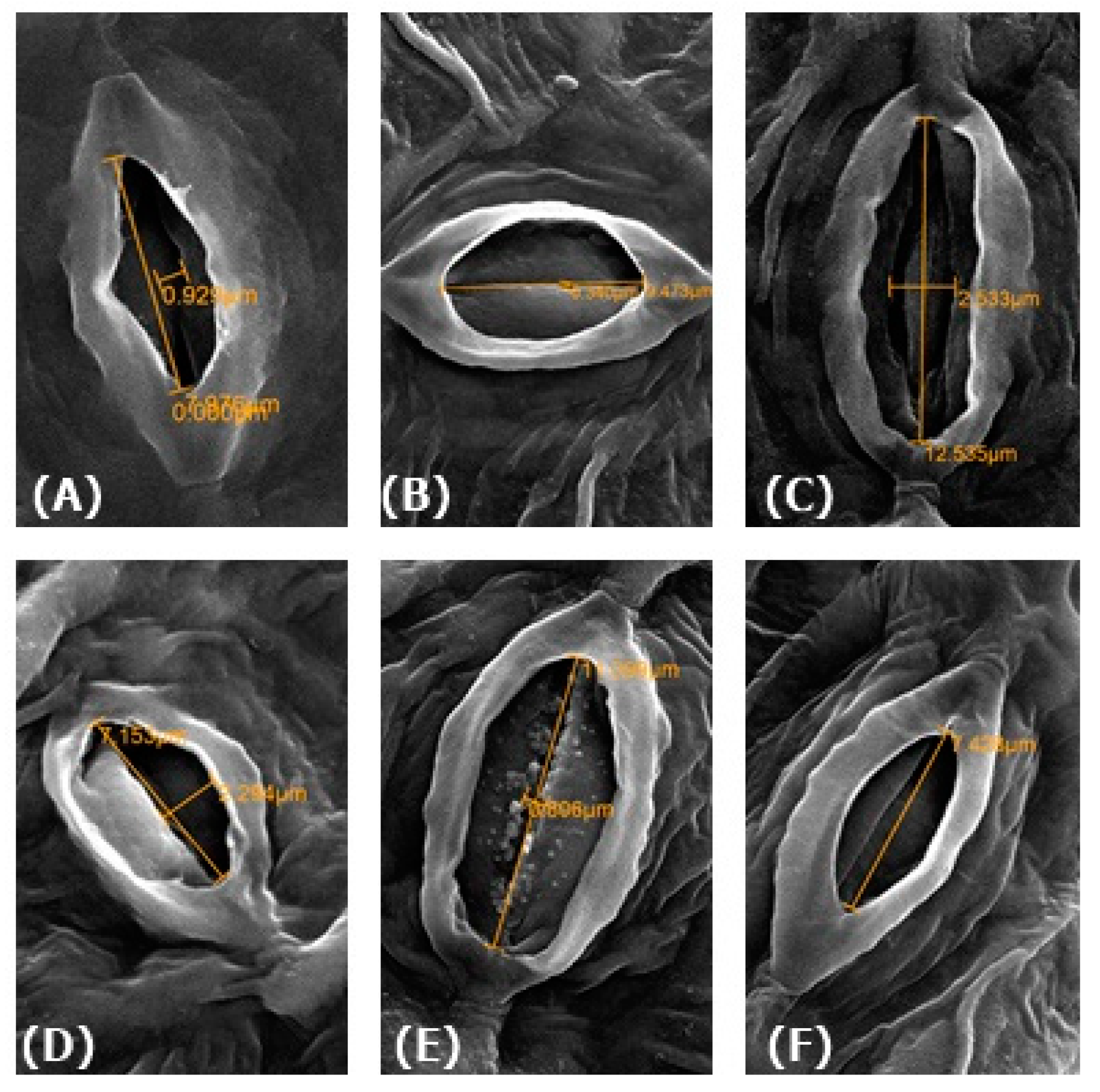
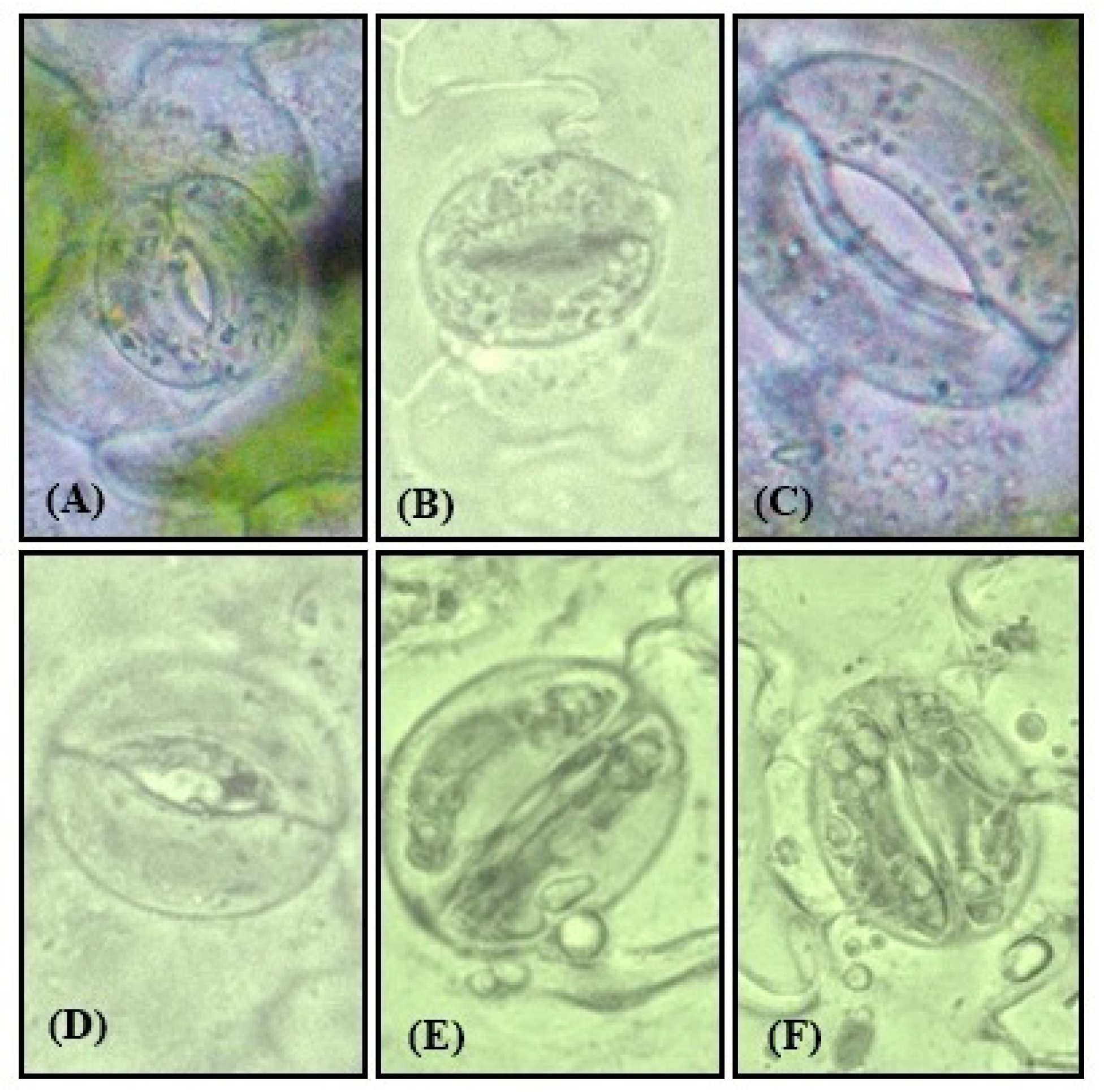
2.11. Physiological Measurements of Guard Cells
2.12. NO Generation
2.13. Statistical Analysis
3. Results
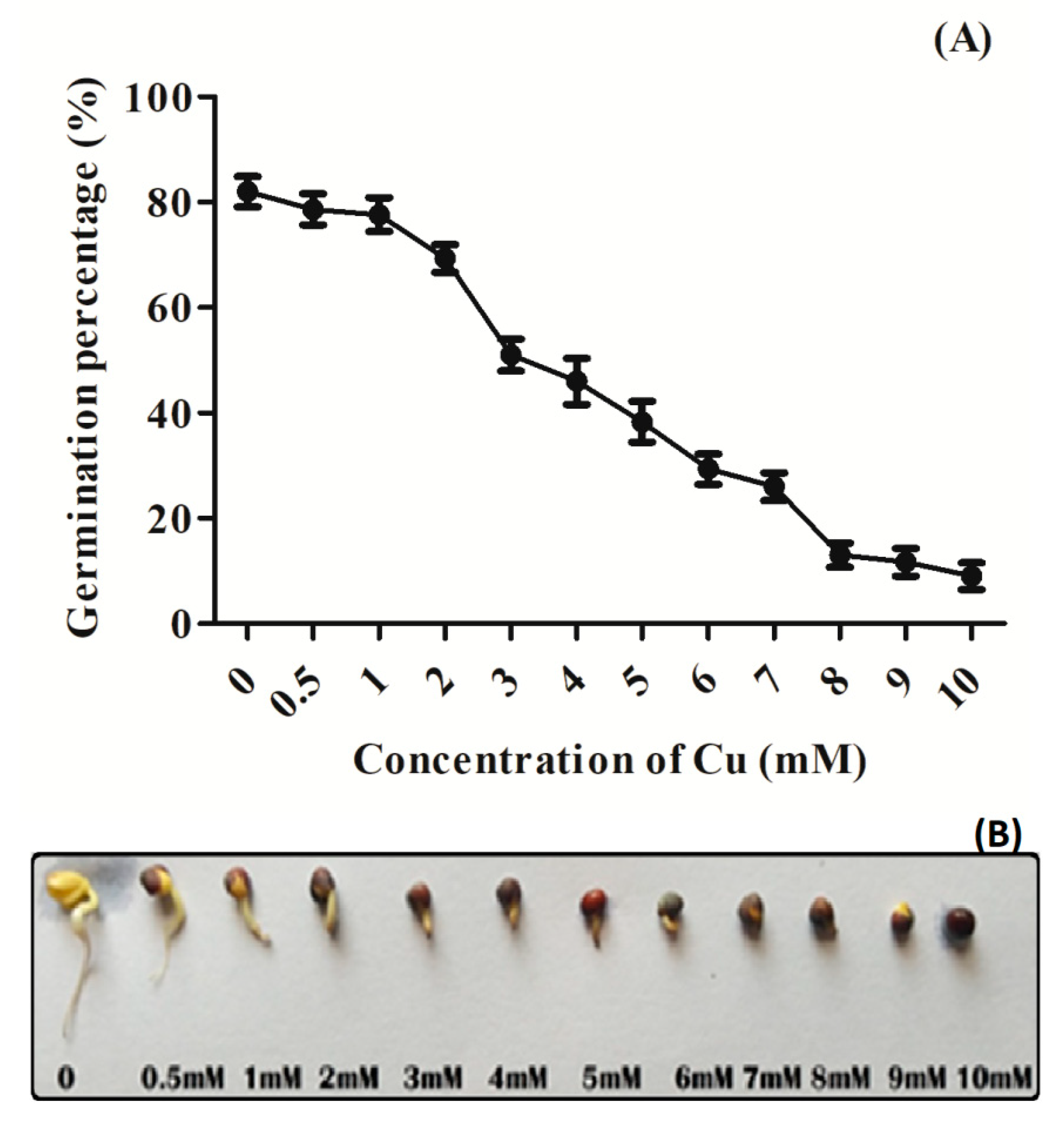
3.1. Effect of Cu Stress on Seed Germination Percentage
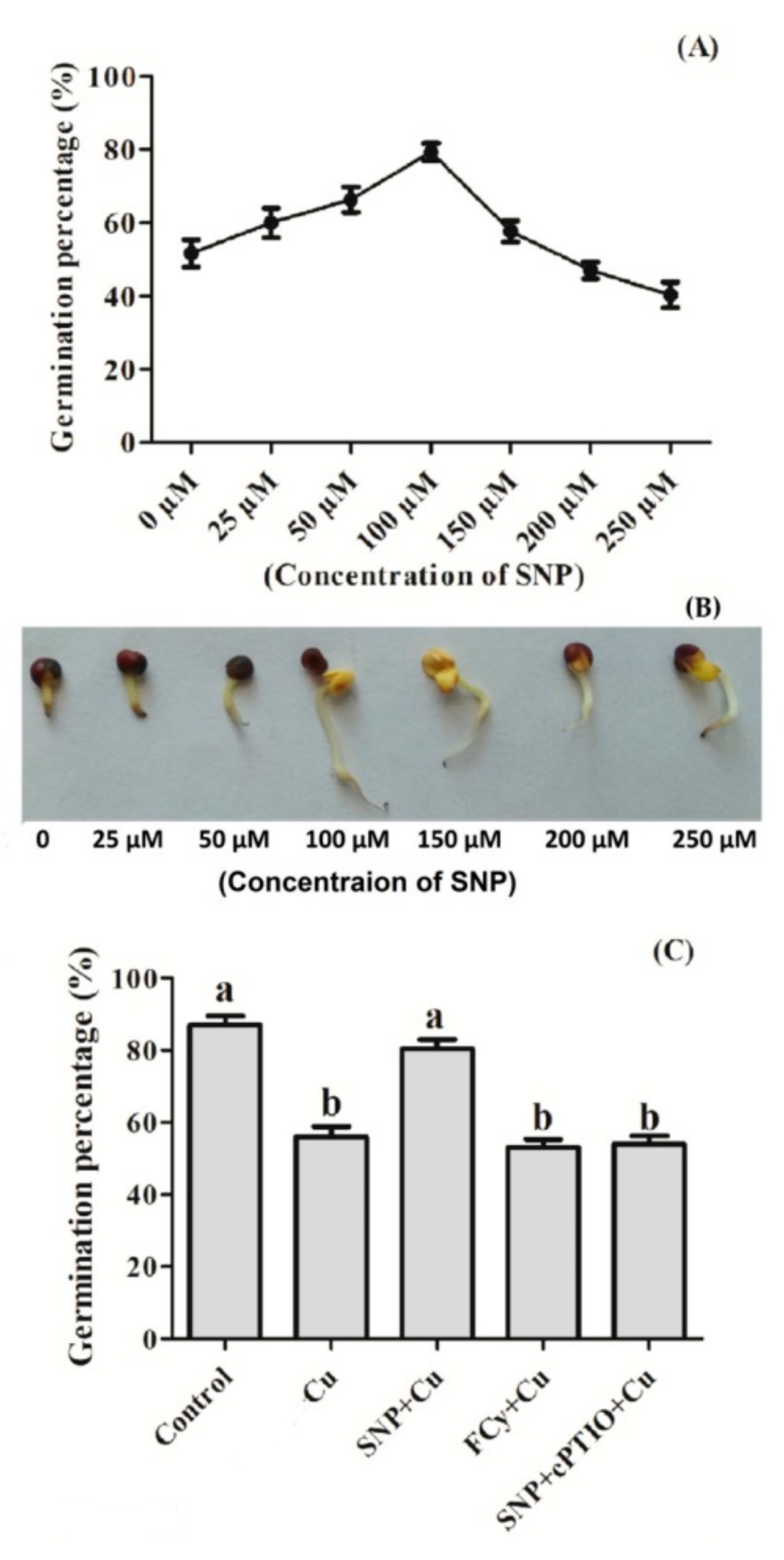
3.2. Effect of NO Pre-treatment on Germination of Cu Stressed Seeds
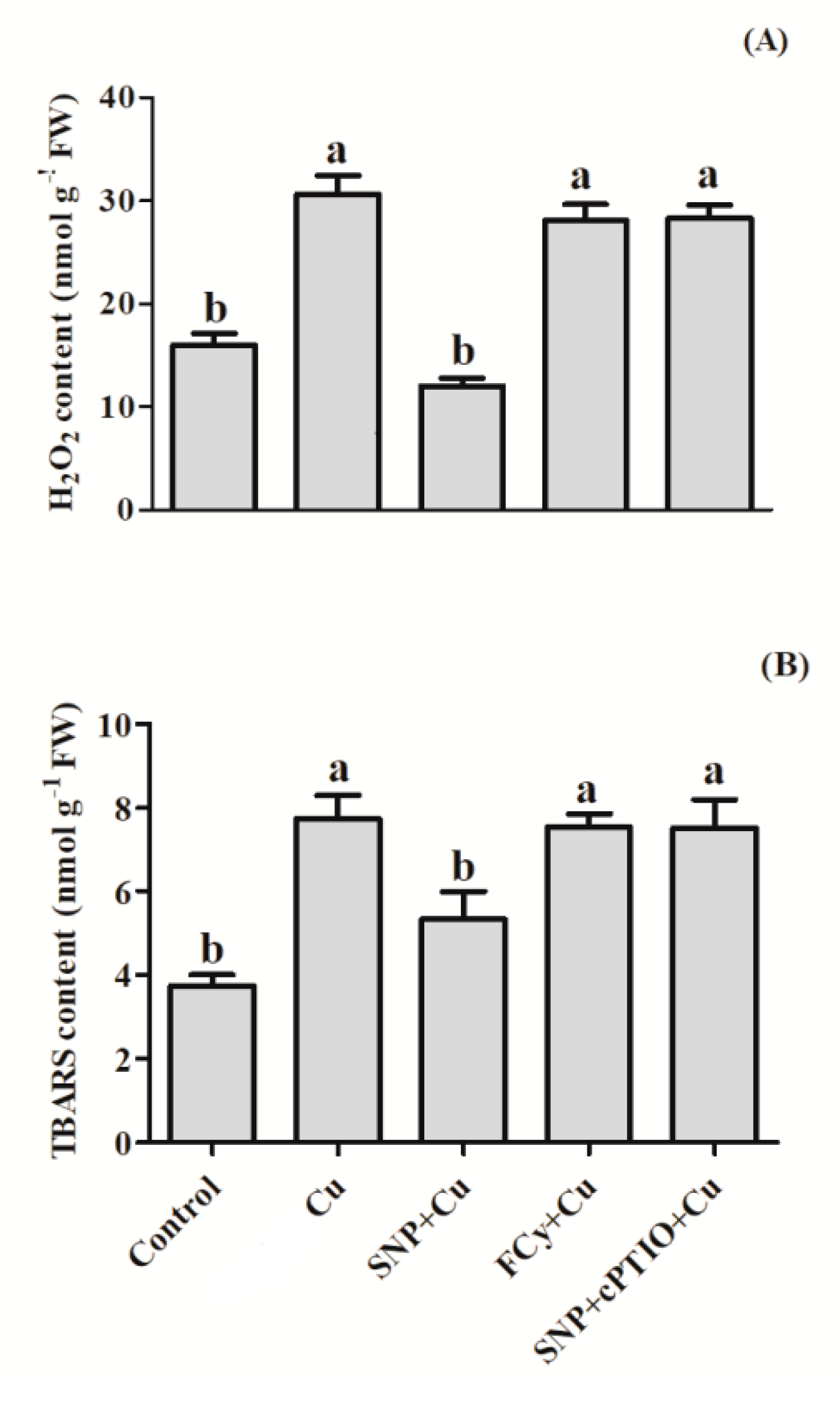
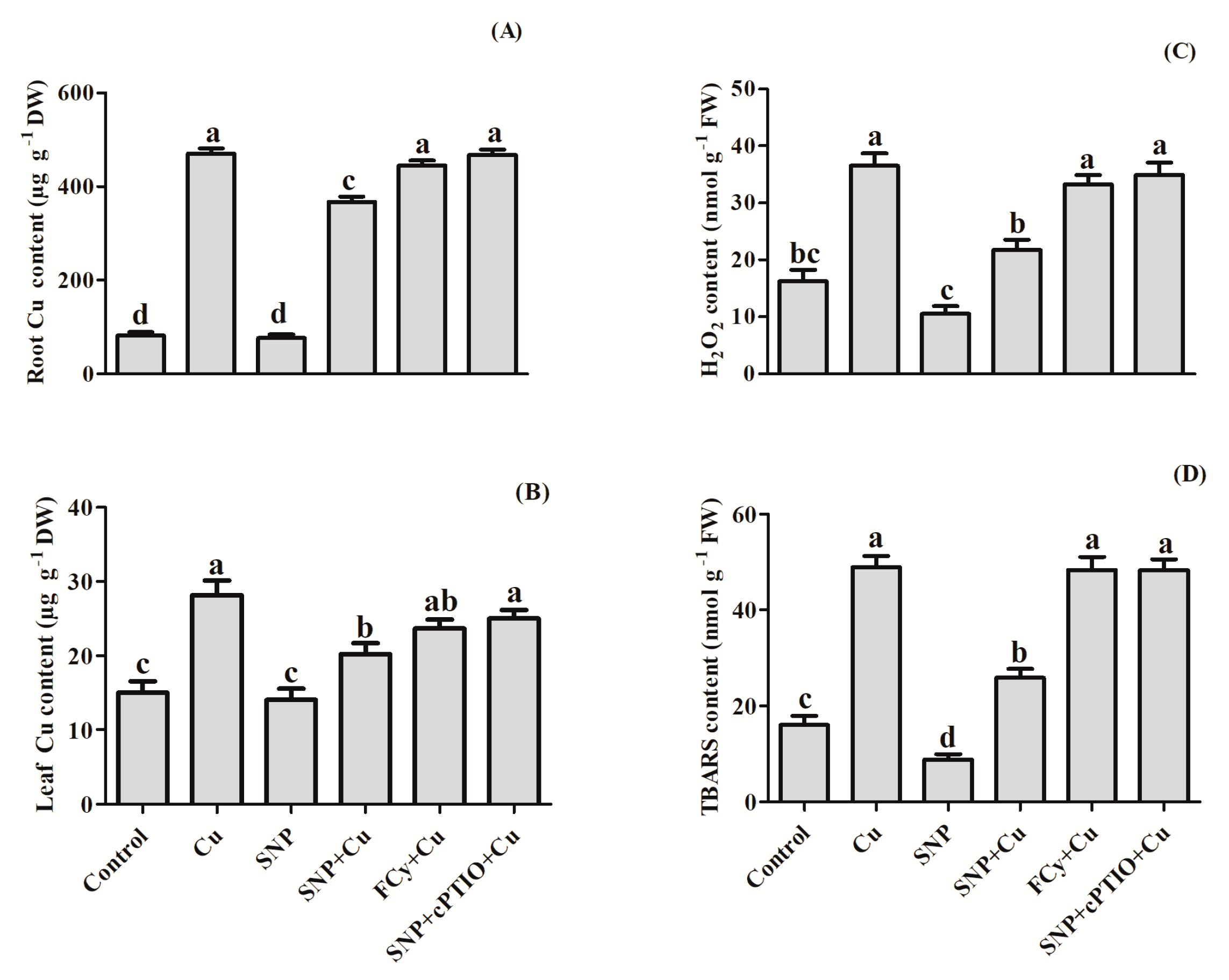
3.3. Effect of NO on H2O2 and TBARS Contents in Germinating Seeds under Cu Stress
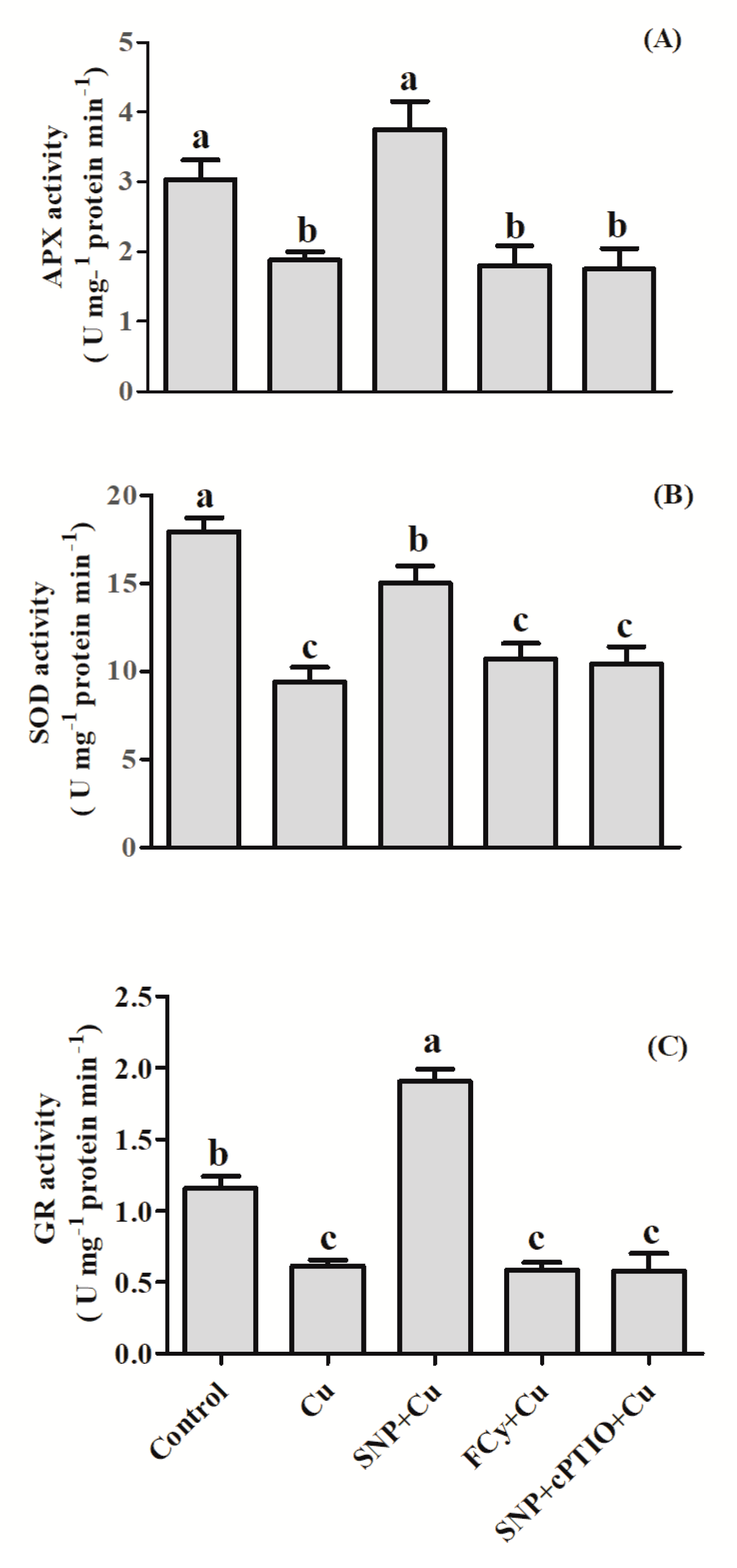
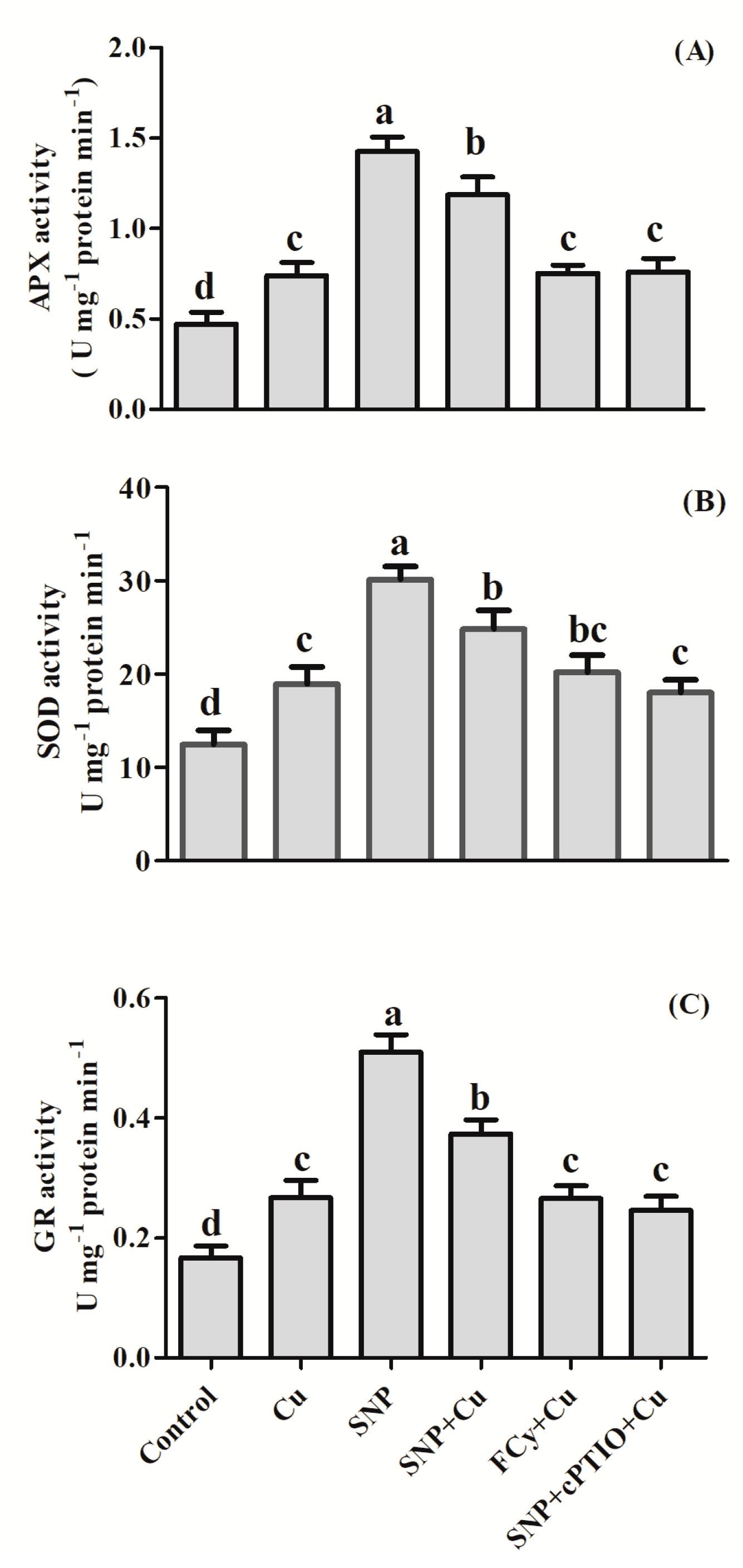
3.4. Effect of NO Application on Activities of Antioxidant Enzymes under Cu Stress during Seed Germination
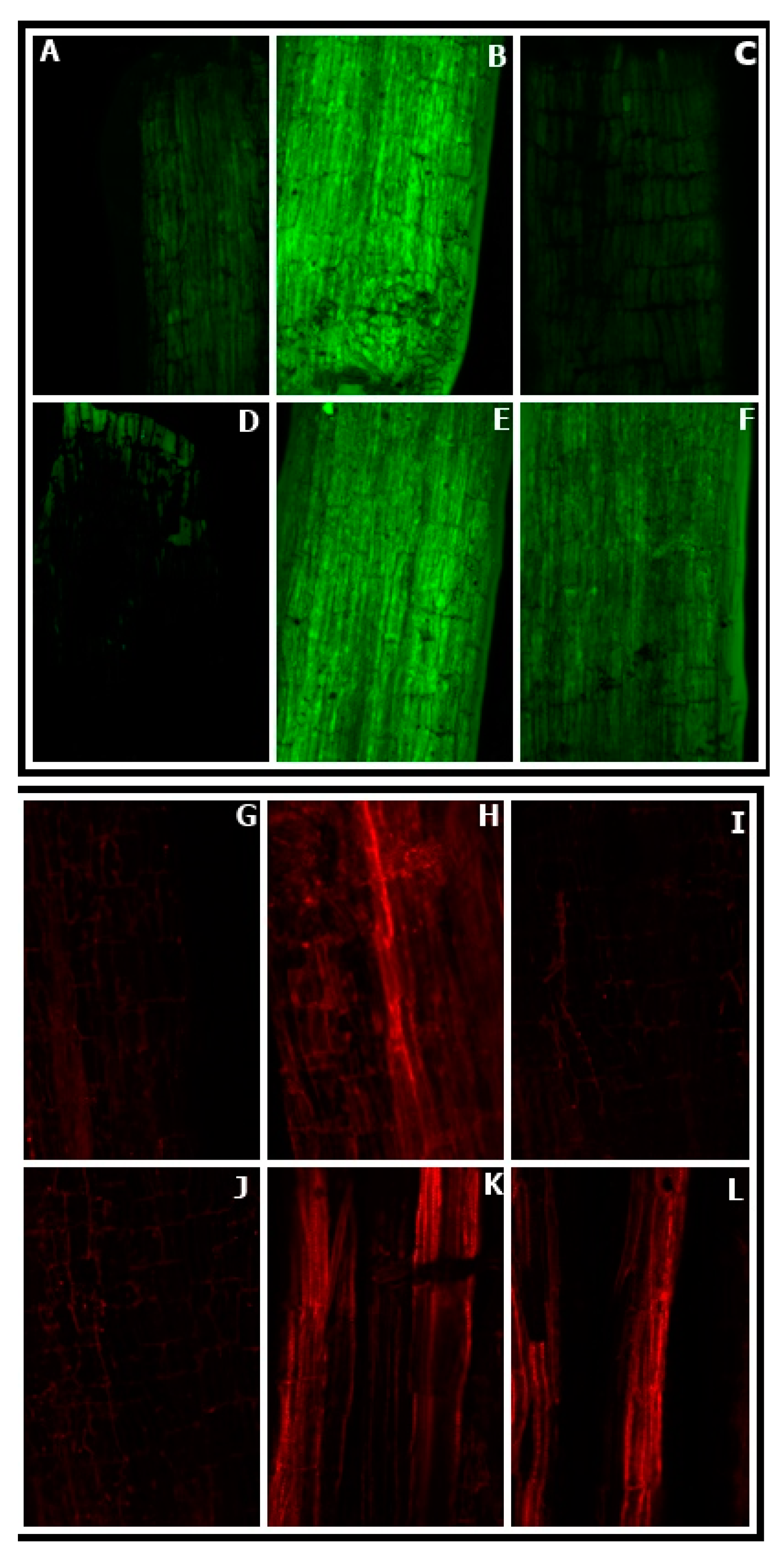
3.5. Effect of NO Application on ROS Accumulation in Germinating Seeds
3.6. Effect of NO Application on the Activity of Amylase
3.7. Effect of NO Application on Cu Uptake and Levels of H2O2 and TBARS
3.8. Effect of NO Treatment on ROS Accumulation by Leaves
3.9. Confocal Laser Scanning Microscopy
3.10. Effect of NO on the Activity of Antioxidant Enzymes
3.11. Impact of NO on Photosynthetic Performance
3.12. Effect of NO on Physiology of Guard Cells
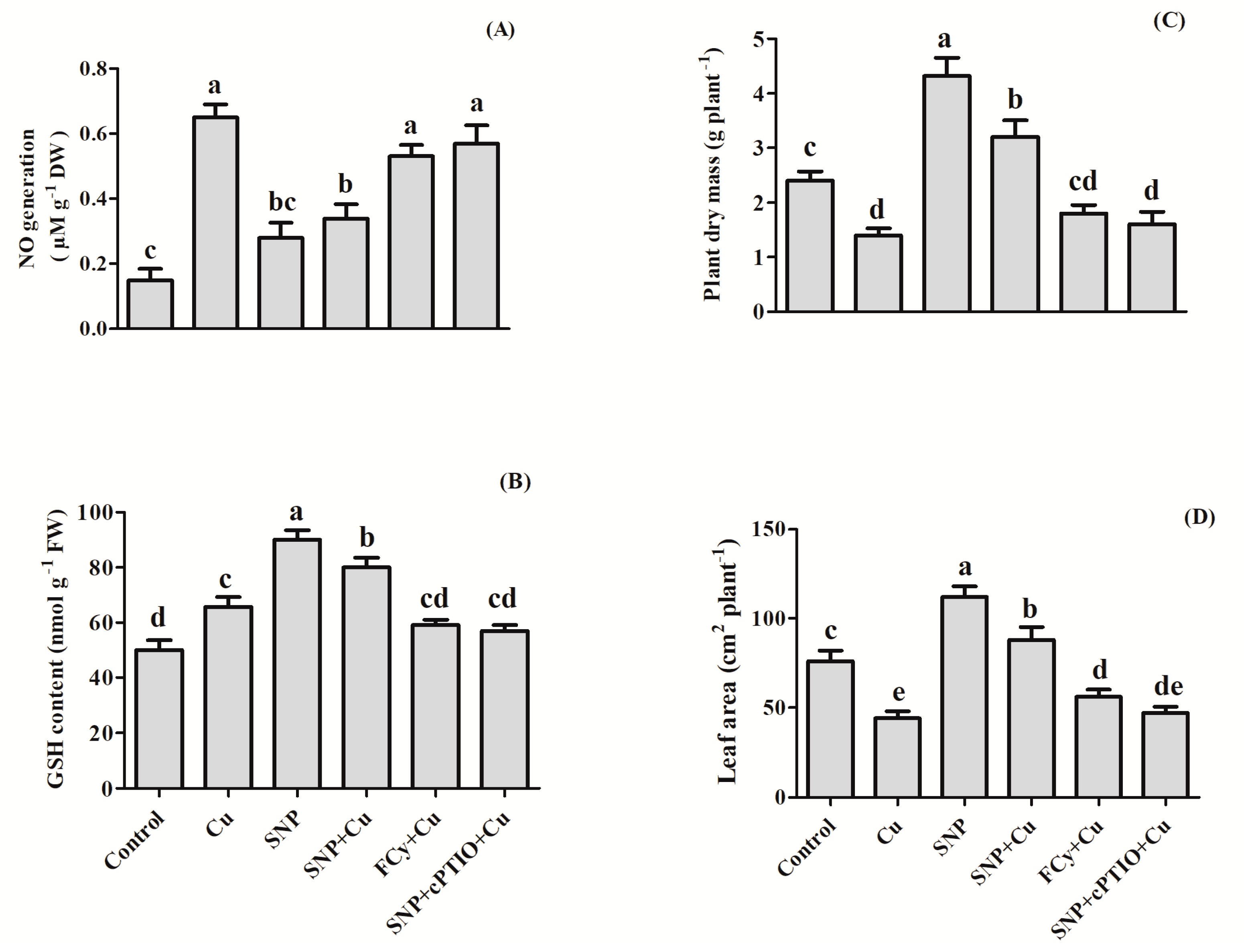
3.13. Effect of NO on NO Generation, GSH Content, Plant Dry Mass and Leaf Area
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mostofa, M.G.; Hossain, M.A.; Fujita, M.; Tran, L.-S.P. Physiological and biochemical mechanisms associated with trehalose-induced copper-stress tolerance in rice. Sci. Rep. 2015, 5, 11433. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Sharma, A.; Kaur, P.; Sidhu, G.P.S.; Bali, A.S.; Bhardwaj, R.; Thukral, A.R.; Cerda, A. Pollution assessment of heavy metals in soils of India and ecological risk assessment: A state-of-the-art. Chemosphere 2019, 216, 449–462. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, D.L.; Johnston, K.M.; Dietrich, A.M. Fate and transport of copper-based crop protectants in plasticulture runoff and the impact of sedimentation as a best management practice. Water Res. 2001, 35, 2984–2994. [Google Scholar] [CrossRef]
- Adrees, M.; Ali, S.; Rizwan, M.; Ibrahim, M.; Abbas, F.; Farid, M.; Zia-Ur-Rehman, M.; Irshad, M.K.; Bharwana, S.A. The effect of excess copper on growth and physiology of important food crops: A review. Environ. Sci. Pollut. Res. 2015, 22, 8148–8162. [Google Scholar] [CrossRef]
- Ravet, K.; Pilon, M. Copper and iron homeostasis in plants: The challenges of oxidative stress. Antiox. Redox Sign. 2013, 19, 919–932. [Google Scholar] [CrossRef]
- Yruela, I. Copper in plants: Acquisition, transport and interactions. Funct. Plant Biol. 2009, 36, 409–430. [Google Scholar] [CrossRef]
- Alaoui-Sossé, B.; Genet, P.; Vinit-Dunand, F.; Toussaint, M.L.; Epron, D.; Badot, P.M. Effect of copper on growth in cucumber plants (Cucumis sativus) and its relationships with carbohydrate accumulation and changes in ion contents. Plant Sci. 2004, 166, 1213–1218. [Google Scholar] [CrossRef]
- Purakayastha, T.J.; Viswanath, T.; Bhadraray, S.; Chhonkar, P.K.; Adhikari, P.P.; Suribabu, K. Phytoextraction of zinc, copper, nickel and lead from a contaminated soil by different species of Brassica. Int. J. Phytorem. 2008, 10, 61–72. [Google Scholar] [CrossRef]
- Mourato, M.P.; Moreira, I.N.; Leitão, I.; Pinto, F.R.; Sales, J.R.; Martins, L.L. Effect of heavy metals in plants of the genus Brassica. Int. J. Mol. Sci. 2015, 16, 17975–17998. [Google Scholar] [CrossRef]
- Rahman, M.M.; Azirun, S.M.; Boyce, A.N. Enhanced accumulation of copper and lead in amaranth (Amaranthus paniculatus), Indian mustard (Brassica juncea) and sunflower (Helianthus annuus). PLoS ONE 2013, 8, e62941. [Google Scholar] [CrossRef]
- Küpper, H.; Götz, B.; Mijovilovich, A.; Küpper, F.C.; Meyer-Klaucke, W. Complexation and toxicity of copper in higher plants. I. Characterization of copper accumulation, speciation, and toxicity in Crassula helmsii as a new copper accumulator. Plant Physiol. 2009, 151, 702–714. [Google Scholar] [CrossRef] [PubMed]
- Maksymiec, W.; Russa, R.; Urbanik-Sypniewska, T.; Baszyński, T. Effect of excess Cu on the photosynthetic apparatus of runner bean leaves treated at two different growth stages. Physiol. Plant. 1994, 91, 715–725. [Google Scholar] [CrossRef]
- Emamverdian, A.; Ding, Y.; Mokhberdoran, F.; Xie, Y. Heavy metal stress and some mechanisms of plant defense response. Sci. World J. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.-L.; Huo, S.-T.; Wang, L.-T.; Meng, J.-F.; Zhang, Z.-W.; Xi, Z.-M. Exogenous 24-Epibrassinolide alleviates oxidative damage from copper stress in grape (Vitis vinifera L.) cuttings. Plant Physiol. Biochem. 2018, 130, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Contreras, L.; Mella, D.; Moenne, A.; Correa, J.A. Differential responses to copper-induced oxidative stress in the marine macro algae Lessonia nigrescens and Scytosiphon lomentaria (Phaeophyceae). Aquat. Toxic. 2009, 94, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Ke, W.; Xiong, Z.-T.; Chen, S.; Chen, J. Effects of copper and mineral nutrition on growth, copper accumulation and mineral element uptake in two Rumex japonicus populations from a copper mine and an uncontaminated field sites. Environ. Exp. Bot. 2007, 59, 59–67. [Google Scholar] [CrossRef]
- Puig, S.; Andrés-Colás, N.; García-Molina, A.; Penarrubia, L. Copper and iron homeostasis in Arabidopsis: Responses to metal deficiencies, interactions and biotechnological applications. Plant Cell Environ. 2007, 30, 271–290. [Google Scholar] [CrossRef]
- Gonzalez-Mendoza, D.; Moreno, A.Q.; Zapata-Perez, O. Coordinated responses of phytochelatin synthase and metallothionein genes in black mangrove, Avicennia germinans, exposed to cadmium and copper. Aquat. Toxicol. 2007, 83, 306–314. [Google Scholar] [CrossRef]
- Navarrete, A.; González, A.; Gómez, M.; Contreras, R.A.; Díaz, P.; Lobos GMoenne, A. Copper excess detoxification is mediated by a coordinated and complementary induction of glutathione, phytochelatins and metallothioneins in the green seaweed Ulva compressa. Plant Physiol. Biochem. 2019, 135, 423–431. [Google Scholar] [CrossRef]
- Karmous, I.; Trevisan, R.; El Ferjani, E.; Chaoui, A.; Sheehan, D. Redox biology response in germinating Phaseolus vulgaris seeds exposed to copper: Evidence for differential redox buffering in seedlings and cotyledon. PLoS ONE 2017, 12, e0184396. [Google Scholar] [CrossRef]
- Ouzounidou, G. Effect of copper on germination and seedling growth of Minuartia, Silene, Alyssum and Thlaspi. Biol. Plant. 1995, 37, 411. [Google Scholar] [CrossRef]
- Bes, C.M.; Jaunatre, R.; Mench, M. Seed bank of Cu-contaminated top soils at a wood preservation site: Impacts of copper and compost on seed germination. Environ. Monit. Assess. 2013, 185, 2039–2053. [Google Scholar] [CrossRef] [PubMed]
- Ashagre, H.; Almaw, D.; Feyisa, T. Effect of copper and zinc on seed germination, phytotoxicity, tolerance and seedling vigor of tomato (Lycopersicon esculentum L. cultivar Roma VF). Int. J. Agric. Sci. Res. 2013, 2, 312–317. [Google Scholar]
- Adhikari, T.; Kundu, S.; Biswas, A.K.; Tarafdar, J.C.; Rao, A.S. Effect of copper oxide nanoparticle on seed germination of selected crops. J. Agric. Sci. Technol. 2012, 2, 815. [Google Scholar]
- Muccifora, S.; Bellani, L.M. Effects of copper on germination and reserve mobilization in Vicia sativa L. seeds. Environ. Pollut. 2013, 179, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Gould, K.; Lamotte, O.; Klinguer, A.; Pugin, A.; Wendehenne, D. Nitric oxide production in tobacco leaf cells: A generalized stress response? Plant Cell Environ. 2003, 26, 1851–1862. [Google Scholar] [CrossRef]
- Del Castello, F.; Nejamkin, A.; Cassia, R.; Correa-Aragunde, N.; Fernández, B.; Foresi, N.; Lombardo, C.; Ramirez, L.; Lamattina, L. The era of nitric oxide in plant biology: Twenty years tying up loose ends. Nitric Oxide 2019, 85, 17–27. [Google Scholar] [CrossRef]
- Corpas, F.J.; Palma, J.M. Assessing nitric oxide (NO) in higher plants: An outline. Nitrogen 2018, 1, 12–20. [Google Scholar] [CrossRef]
- Santisree, P.; Adimulam, S.S.; Sharma, K.; Bhatnagar-Mathur, P.; Sharma, K.K. Insights into the Nitric Oxide Mediated Stress Tolerance in Plants. In Plant Signaling Molecules; Khan, M.I.R., Reddy, P.S., Ferrante, A., Khan, N.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 385–406. [Google Scholar] [CrossRef]
- Lamattina, L.; García-Mata, C.; Graziano, M.; Pagnussat, G. Nitric oxide: The versatility of an extensive signal molecule. Ann. Rev. Plant Biol. 2003, 54, 109–136. [Google Scholar] [CrossRef]
- Simontacchi, M.; Jasid, S.; Puntarulo, S. Nitric oxide generation during early germination of sorghum seeds. Plant Sci. 2004, 167, 839–847. [Google Scholar] [CrossRef]
- Beligni, M.V.; Lamattina, L. Nitric oxide stimulates seed germination and de-etiolation, and inhibits hypocotyl elongation, three light-inducible responses in plants. Planta 2000, 210, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Shen, W.-B.; Xu, L.-L. Effects of nitric oxide on the germination of wheat seeds and its reactive oxygen species metabolisms under osmotic stress. Acta Bot. Sin. 2003, 45, 901–905. [Google Scholar]
- Bethke, P.C.; Gubler, F.; Jacobsen, J.V.; Jones, R.L. Dormancy of Arabidopsis seeds and barley grains can be broken by nitric oxide. Planta 2004, 219, 847–855. [Google Scholar] [CrossRef] [PubMed]
- Sarath, G.; Bethke, P.C.; Jones, R.; Baird, L.M.; Hou, G.; Mitchell, R.B. Nitric oxide accelerates seed germination in warm-season grasses. Planta 2006, 223, 1154–1164. [Google Scholar] [CrossRef] [PubMed]
- Hu, K.-D.; Hu, L.-Y.; Li, Y.-H.; Zhang, F.-Q.; Zhang, H. Protective roles of nitric oxide on germination and antioxidant metabolism in wheat seeds under copper stress. Plant Growth Regul. 2007, 53, 173–183. [Google Scholar] [CrossRef]
- Fatma, M.; Masood, A.; Per, T.S.; Rasheed, F.; Khan, N.A. Interplay between nitric oxide and sulfur assimilation in salt tolerance in plants. Crop J. 2016, 4, 153–161. [Google Scholar] [CrossRef]
- Jahan, B.; Al Ajmi, M.F.; Rehman, M.T.; Khan, N.A. Treatment of nitric oxide supplemented with nitrogen and sulfur regulates photosynthetic performance and stomatal behavior in mustard under salt stress. Physiol. Plant. 2020, 168, 490–510. [Google Scholar] [CrossRef] [PubMed]
- Fatma, M.; Masood, A.; Per, T.S.; Khan, N.A. Nitric oxide alleviates salt stress inhibited photosynthetic performance by interacting with sulfur assimilation in mustard. Front. Plant Sci. 2016, 7, 521. [Google Scholar] [CrossRef]
- Wang, J.; Yu, S.X.; Zhang, M.; Cui, X.M. Exogenous nitric oxide-mediated GSH-PC synthesis pathway in tomato under copper stress. Russ. J. Plant Physiol. 2015, 62, 349–359. [Google Scholar] [CrossRef]
- Per, T.S.; Masood, A.; Khan, N.A. Nitric oxide improves S-assimilation and GSH production to prevent inhibitory effects of cadmium stress on photosynthesis in mustard (Brassica juncea L.). Nitric Oxide 2017, 68, 111–124. [Google Scholar] [CrossRef]
- Okuda, T.; Matsuda, Y.; Yamanaka, A.; Sagisaka, S. Abrupt increase in the level of hydrogen peroxide in leaves of winter wheat is caused by cold treatment. Plant Physiol. 1991, 9, 1265–1267. [Google Scholar] [CrossRef] [PubMed]
- Dhindsa, R.S.; Plumb-Dhindsa, P.; Thorpe, T.A. Leaf senescence: Correlated with increased levels of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J. Exp. Bot. 1981, 32, 93–101. [Google Scholar] [CrossRef]
- Beyer, W.F., Jr.; Fridovich, I. Assaying for superoxide dismutase activity: Some large consequences of minor changes in conditions. Anal. Biochem. 1987, 161, 559–566. [Google Scholar] [CrossRef]
- Giannopolitis, C.N.; Ries, S.K. Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol. 1977, 59, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar] [CrossRef]
- Foyer, C.H.; Halliwell, B. The presence of glutathione and glutathione reductase in chloroplasts: A proposed role in ascorbic acid metabolism. Planta 1976, 133, 21–25. [Google Scholar] [CrossRef]
- Collins, G.; Jenner, C.; Paleg, L. The metabolism of soluble nucleotides in wheat aleurone layers treated with gibberellic acid. Plant Physiol. 1972, 49, 404–410. [Google Scholar] [CrossRef][Green Version]
- Anderson, M.E. Determination of glutathione and glutathione disulfide in biological samples. Methods Enzymol. 1985, 113, 548–555. [Google Scholar] [CrossRef]
- Fatma, M.; Khan, N.A. Nitric oxide protects photosynthetic capacity inhibition by salinity in Indian mustard. J. Funct. Environ. Bot. 2014, 4, 106–116. [Google Scholar] [CrossRef]
- Kumar, D.; Yusuf, M.A.; Singh, P.; Sardar, M.; Sarin, N.B. Histochemical detection of superoxide and H2O2 accumulation in Brassica juncea seedlings. Biol. Protoc. 2014, 4, e1108. [Google Scholar] [CrossRef]
- Usuda, H. The activation state of ribulose 1, 5-bisphosphate carboxylase in maize leaves in dark and light. Plant Cell Physiol. 1985, 26, 1455–1463. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Zhou, B.; Guo, Z.; Xing, J.; Huang, B. Nitric oxide is involved in abscisic acid-induced antioxidant activities in Stylosanthes guianensis. J. Exp. Bot. 2005, 56, 3223–3228. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Hu, L.Y.; Hu, K.D.; He, Y.D.; Wang, S.H.; Luo, J.P. Hydrogen sulfide promotes wheat seed germination and alleviates oxidative damage against copper stress. J. Integr. Plant Biol. 2008, 50, 1518–1529. [Google Scholar] [CrossRef] [PubMed]
- Delledonne, M.; Xia, Y.; Dixon, R.A.; Lamb, C. Nitric oxide functions as a signal in plant disease resistance. Nature 1998, 394, 585. [Google Scholar] [CrossRef] [PubMed]
- Wimalasekera, R.; Tebartz, F.; Scherer, G.F. Polyamines, polyamine oxidases and nitric oxide in development, abiotic and biotic stresses. Plant Sci. 2011, 181, 593–603. [Google Scholar] [CrossRef]
- Manai, J.; Kalai, T.; Gouia, H.; Corpas, F. Exogenous nitric oxide (NO) ameliorates salinity-induced oxidative stress in tomato (Solanum lycopersicum) plants. J. Soil. Sci. 2014, 14, 433–446. [Google Scholar] [CrossRef]
- Mur, L.A.; Mandon, J.; Persijn, S.; Cristescu, S.M.; Moshkov, I.E.; Novikova, G.V.; Hall, M.A.; Harren, F.J.M.; Hebelstrup, K.H.; Gupta, K.J. Nitric oxide in plants: An assessment of the current state of knowledge. AoB Plants 2013, 5. [Google Scholar] [CrossRef]
- Bethke, P.C.; Libourel, I.G.; Reinöhl, V.; Jones, R.L. Sodium nitroprusside, cyanide, nitrite, and nitrate break Arabidopsis seed dormancy in a nitric oxide-dependent manner. Planta 2006, 223, 805–812. [Google Scholar] [CrossRef]
- He, J.; Ren, Y.; Chen, X.; Chen, H. Protective roles of nitric oxide on seed germination and seedling growth of rice (Oryza sativa L.) under cadmium stress. Ecotoxicol. Environ. Saf. 2014, 108, 114–119. [Google Scholar] [CrossRef]
- Liu, J.; Xue, T.; Shen, Y. Effect of Nitric Oxide on Seed Germination and Dormancy in Empress Trees. HortTechnology 2019, 29, 271–275. [Google Scholar] [CrossRef]
- Ren, Y.; Wang, W.; He, J.; Zhang, L.; Wei, Y.; Yang, M. Nitric oxide alleviates salt stress in seed germination and early seedling growth of pakchoi (Brassica chinensis L.) by enhancing physiological and biochemical parameters. Ecotoxicol. Environ. Saf. 2020, 187, 109785. [Google Scholar] [CrossRef]
- Zhang, H.; Shen, W.-B.; Zhang, W.; Xu, L.-L. A rapid response of β-amylase to nitric oxide but not gibberellin in wheat seeds during the early stage of germination. Planta 2005, 220, 708–716. [Google Scholar] [CrossRef] [PubMed]
- Patel, P.; Kadur Narayanaswamy, G.; Kataria, S.; Baghel, L. Involvement of nitric oxide in enhanced germination and seedling growth of magneto primed maize seeds. Plant. Signal. Behav. 2017, 12, e1293217. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Terrón-Camero, L.C.; Peláez-Vico, M.Á.; Del-Val, C.; Sandalio, L.M.; Romero-Puertas, M.C. Role of nitric oxide in plant responses to heavy metal stress: Exogenous application versus endogenous production. J. Exp. Bot. 2019, 70, 4477–4488. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Zhang, J.; Wang, C.; Liao, W. Recent progress in the knowledge on the alleviating effect of nitric oxide on heavy metal stress in plants. Plant Physiol. Biochem. 2020, 147, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Baudhouin, E. The language of nitric oxide signaling. Plant Biol. 2011, 13, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Pető, A.; Lehotai, N.; Feigl, G.; Tugyi, N.; Ördög, A.; Gémes, K.; Kolbert, Z. Nitric oxide contributes to copper tolerance by influencing ROS metabolism in Arabidopsis. Plant Cell Rep. 2013, 32, 1913–1923. [Google Scholar] [CrossRef]
- Wang, H.; Hou, J.L.Y.; Zhang, Y.; Huang, J.; Liang, W. Nitric oxide-mediated cytosolic glucose-6-phosphate dehydrogenase is involved in aluminum toxicity of soybean under high aluminum concentration. Plant Soil 2017, 416, 39–52. [Google Scholar] [CrossRef]
- Singh, A.P.; Dixit, G.; Kumar, A.; Mishra, S.; Kumar, N.; Dixit, S.; Dhankher, O.P. A protective role for nitric oxide and salicylic acid for arsenite phytotoxicity in rice (Oryza sativa L.). Plant Physiol. Biochem. 2017, 115, 163–173. [Google Scholar] [CrossRef]
- Gong, B.; Nie, W.; Yan, Y.; Gao, Z.; Shi, Q. Unraveling cadmium toxicity and nitric oxide induced tolerance in Cucumis sativus: Insight into regulatory mechanisms using proteomics. J. Hazard. Mater. 2017, 336, 202–213. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.J.; Hu, M.M.; Cui, X.M.; Lou, Y.H.; Zhuge, Y.P. Exogenous NO mediated the detoxification pathway of tomato seedlings under different stress of Cu and Cd. J. Appl. Ecol. 2018, 29, 4199. [Google Scholar] [CrossRef]
- Mostofa, M.G.; Seraj, Z.I.; Fujita, M. Exogenous sodium nitroprusside and glutathione alleviate copper toxicity by reducing copper uptake and oxidative damage in rice (Oryza sativa L.) seedlings. Protoplasma 2014, 251, 1373–1386. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Han, X.; Chen, X.; Jin, H.; Cui, X. Exogenous nitric oxide on antioxidative system and ATPase activities from tomato seedlings under copper stress. Sci. Hortic. 2009, 123, 217–223. [Google Scholar] [CrossRef]
- Rizwan, M.; Mostofa, M.G.; Ahmad, M.Z.; Imtiaz, M.; Mehmood, S.; Adeel, M.; Dai, Z.; Li, Z.; Aziz, O.; Zhang, Y.; et al. Nitric oxide induces rice tolerance to excessive nickel by regulating nickel uptake, reactive oxygen species detoxification and defense-related gene expression. Chemosphere 2018, 191, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Jinc, S.; Liu, S.; Xu, L.; Kong, J. Effects of exogenous nitric oxide on growth of cotton seedlings under NaCl stress. J. Soil Sci. Plant Nutr. 2014, 14, 1–13. [Google Scholar] [CrossRef][Green Version]
- Dong, Y.X.; Wang, X.F.; Cui, X.M. Exogenous nitric oxide involved in subcellular distribution and chemical forms of Cu2+ under copper stress in tomato seedlings. J. Integr. Agric. 2013, 12, 1783–1790. [Google Scholar] [CrossRef]
- Dong, Y.; Xu, L.; Wang, Q.; Fan, Z.; Kong, J.; Bai, X. Effects of exogenous nitric oxide on photosynthesis, antioxidative ability, and mineral element contents of perennial ryegrass under copper stress. J. Plant Interact. 2014, 9, 402–411. [Google Scholar] [CrossRef]
- Hattab, S.; Chouba, L.; Benkheder, M.; Mahouachi, T.; Boussetta, H. Cadmium- and copper-induced DNA damage in Pisum sativum roots and leaves as determined by the Comet assay. Plant Biosyst. 2009, 143, 6–11. [Google Scholar] [CrossRef]
- Xiong, J.; An, L.; Lu, H.; Yhu, C. Exogenous nitric oxide enhances cadmium tolerance of rice by increasing pectin and hemicellulose contents in root cell wall. Planta 2009, 230, 755–765. [Google Scholar] [CrossRef]
- Wang, Q.H.; Liang, X.; Dong, Y.J.; Xu, L.L.; Zhang, X.W.; Hou, J.; Fan, Z.Y. Effects of exogenous nitric oxide on cadmium toxicity, element contents and antioxidative system in perennial ryegrass. Plant Growth Regul. 2013, 69, 11–20. [Google Scholar] [CrossRef]
- Fecht-Christoffers, M.M.; Braun, H.P.; Lemaitre-Guillier, C.; van Dorsselaer, A.; Horst, W.J. Effect of manganese toxicity on the proteome of the leaf apoplast in cowpea. Plant Physiol. 2003, 133, 1935–1946. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Yang, R.; Pan, Y.; Ren, B.; Chen, Q.; Li, X.; Ma, M. Beneficial behavior of nitric oxide in copper-treated medicinal plants. J. Hazard. Mater. 2016, 314, 140–154. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Singh, H.P.; Batish, D.R.; Mahajan, P.; Kohli, R.K.; Rishi, V. Exogenous nitric oxide (NO) interferes with lead (Pb)-induced toxicity by detoxifying reactive oxygen species in hydroponically grown wheat (Triticumaestivum) roots. PLoS ONE 2015, 10, e0138713. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, D.K.; Singh, S.; Singh, S.; Srivastava, P.K.; Singh, V.P.; Singh, S.; Prasad, S.M.; Singh, P.K.; Dubey, N.K.; Pandey, A.C.; et al. Nitric oxide alleviates silver nanoparticles (AgNps)-induced phytotoxicity in Pisumsativum seedlings. Plant Physiol. Biochem. 2017, 110, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, P.; Ahanger, M.A.; Alyemeni, M.N.; Wijaya, L.; Alam, P. Exogenous application of nitric oxide modulates osmolyte metabolism, antioxidants, enzymes of ascorbate-glutathione cycle and promotes growth under cadmium stress in tomato. Protoplasma 2018, 255, 79–93. [Google Scholar] [CrossRef]
- Sehar, Z.; Masood, A.; Khan, N.A. Nitric oxide reverses glucose-mediated photosynthetic repression in wheat (Triticum aestivum L.) under salt stress. Environ. Exp. Bot. 2019, 161, 277–289. [Google Scholar] [CrossRef]
- Laspina, N.; Groppa, M.; Tomaro, M.; Benavides, M. Nitric oxide protects sunflower leaves against Cd-induced oxidative stress. Plant Sci. 2005, 169, 323–330. [Google Scholar] [CrossRef]
- Ruan, H.-H.; Shen, W.-B.; Xu, L.-L. Nitric oxide involved in the abscisic acid induced proline accumulation in wheat seedling leaves under salt stress. Acta Bot. Sin. 2004, 46, 1307–1315. [Google Scholar]
- Fan, H.; Guo, S.; Jiao, Y.; Zhang, R.; Li, J. Effects of exogenous nitric oxide on growth, active oxygen species metabolism, and photosynthetic characteristics in cucumber seedlings under NaCl stress. Front. Agric. China 2007, 1, 308–314. [Google Scholar] [CrossRef]
- Feigl, G.; Kumar, D.; Lehotai, N.; Tugyi, N.; Molnár, Á.; Ördög, A.; Szepesi, A.; Gemes, K.; Laskay, G.; Erdei, L.; et al. Physiological and morphological responses of the root system of Indian mustard (Brassica juncea L. Czern.) and rapeseed (Brassica napus L.) to copper stress. Ecotoxicol. Environ. Saf. 2013, 94, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Khairy, A.I.H.; Oh, M.J.; Lee, S.M.; Roh, K.S. Nitric oxide overcomes Cd and Cu toxicity in in vitro-grown tobacco plants through increasing contents and activities of rubisco and rubisco activase. Biochim. Open 2016, 2, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Dong, Y.; Wang, Q.; Xu, L.; Kong, J.; Liu, S. Effects of lead and nitric oxide on photosynthesis, antioxidative ability, and mineral element content of perennial ryegrass. Biol. Plant 2015, 59, 163–170. [Google Scholar] [CrossRef]
- Tewari, R.K.; Hahn, E.J.; Paek, K.Y. Modulation of copper toxicity-induced oxidative damage by nitric oxide supply in the adventitious roots of Panax ginseng. Plant Cell Rep. 2008, 27, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yang, L.; Yang, F.; Li, X.; Song, Y.; Wang, X.; Hu, X. Involvements of H2O2 and metallothionein in NO-mediated tomato tolerance to copper toxicity. J. Plant Physiol. 2010, 167, 1298–1306. [Google Scholar] [CrossRef] [PubMed]


| Treatments | Details |
|---|---|
| Control | Seeds pre-treated with water and raised without Cu stress |
| Cu | Seeds pre-treated with water and raised with Cu stress |
| SNP | Seeds pre-treated with SNP and raised without Cu stress |
| SNP + Cu | Seeds pre-treated with SNP and raised with Cu stress |
| FCy + Cu | Seeds pre-treated with FCy and raised with Cu stress |
| SNP + cPTIO + Cu | Seeds pre-treated with SNP+cPTIO and raised with Cu stress |
| Treatments | Net Photosynthesis (µmol CO2 m−2 s−1) | Internal CO2 Concentration (µmol CO2 mol−1) | Maximal PSII Photochemical Efficiency | Stomatal Conductance (mmol CO2 m−2 s−1) | Rubisco Activity (µmol CO2 mg1 protein min−1) |
|---|---|---|---|---|---|
| Control | 16.67 ± 0.89 c | 203 ± 5.57 c | 0.6 ± 0.09 c | 288 ± 9.54 b | 0.82 ± 0.06 b |
| Cu | 10.27 ± 0.92 e | 159 ± 5.51 d | 0.34 ± 0.06 d | 203 ± 8.54 c | 0.49 ± 0.05 c |
| SNP | 26.16 ± 0.72 a | 303 ± 4.04 a | 0.8 ± 0.08 a | 392.67 ± 8.19 a | 1.06 ± 0.07 a |
| SNP + Cu | 22 ± 0.58 b | 278 ± 6.08 b | 0.7 ± 0.06 b | 374.33 ± 8.68 a | 0.92 ± 0.09 ab |
| FCy + Cu | 12.96 ± 0.80 d | 168 ± 5.29 d | 0.38 ± 0.07 d | 221 ± 6.66 c | 0.52 ± 0.04 c |
| SNP + cPTIO + Cu | 11.97 ± 58 de | 162 ± 4.51 d | 0.35 ± 0.05 d | 208 ± 5.51 c | 0.51 ± 0.04 c |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rather, B.A.; Mir, I.R.; Masood, A.; Anjum, N.A.; Khan, N.A. Nitric Oxide Pre-Treatment Advances Seed Germination and Alleviates Copper-Induced Photosynthetic Inhibition in Indian Mustard. Plants 2020, 9, 776. https://doi.org/10.3390/plants9060776
Rather BA, Mir IR, Masood A, Anjum NA, Khan NA. Nitric Oxide Pre-Treatment Advances Seed Germination and Alleviates Copper-Induced Photosynthetic Inhibition in Indian Mustard. Plants. 2020; 9(6):776. https://doi.org/10.3390/plants9060776
Chicago/Turabian StyleRather, Bilal A., Iqbal R. Mir, Asim Masood, Naser A. Anjum, and Nafees A. Khan. 2020. "Nitric Oxide Pre-Treatment Advances Seed Germination and Alleviates Copper-Induced Photosynthetic Inhibition in Indian Mustard" Plants 9, no. 6: 776. https://doi.org/10.3390/plants9060776
APA StyleRather, B. A., Mir, I. R., Masood, A., Anjum, N. A., & Khan, N. A. (2020). Nitric Oxide Pre-Treatment Advances Seed Germination and Alleviates Copper-Induced Photosynthetic Inhibition in Indian Mustard. Plants, 9(6), 776. https://doi.org/10.3390/plants9060776









