Effect of Ammonia and Indole-3-acetic Acid Producing Endophytic Klebsiella pneumoniae YNA12 as a Bio-Herbicide for Weed Inhibition: Special Reference with Evening Primroses
Abstract
1. Introduction
2. Materials and Methods
2.1. Isolation and Identification of Endophytic Bacteria
2.2. Preliminary Biochemical Assessment of Endophytic Bacteria
2.2.1. IAA Detection
2.2.2. Citrate Utilization Test
2.2.3. Catalase Test
2.3. Biochemical Analysis
2.3.1. Quantification of IAA Production by GC/MS-SIM
2.3.2. Production of Ammonia
2.3.3. Catalase Activity
2.4. Screening of Plant Growth Inhibition by Endophytic Bacteria via Germination Test
2.4.1. Effect of YNA12 on the Abscisic Acid (ABA) Modulation of Evening Primroses
2.4.2. Analysis of Macronutrient Regulation in Plants by Inductively Coupled Plasma Mass Spectrometry (ICP-MS)
2.5. Statistical Analysis
3. Results
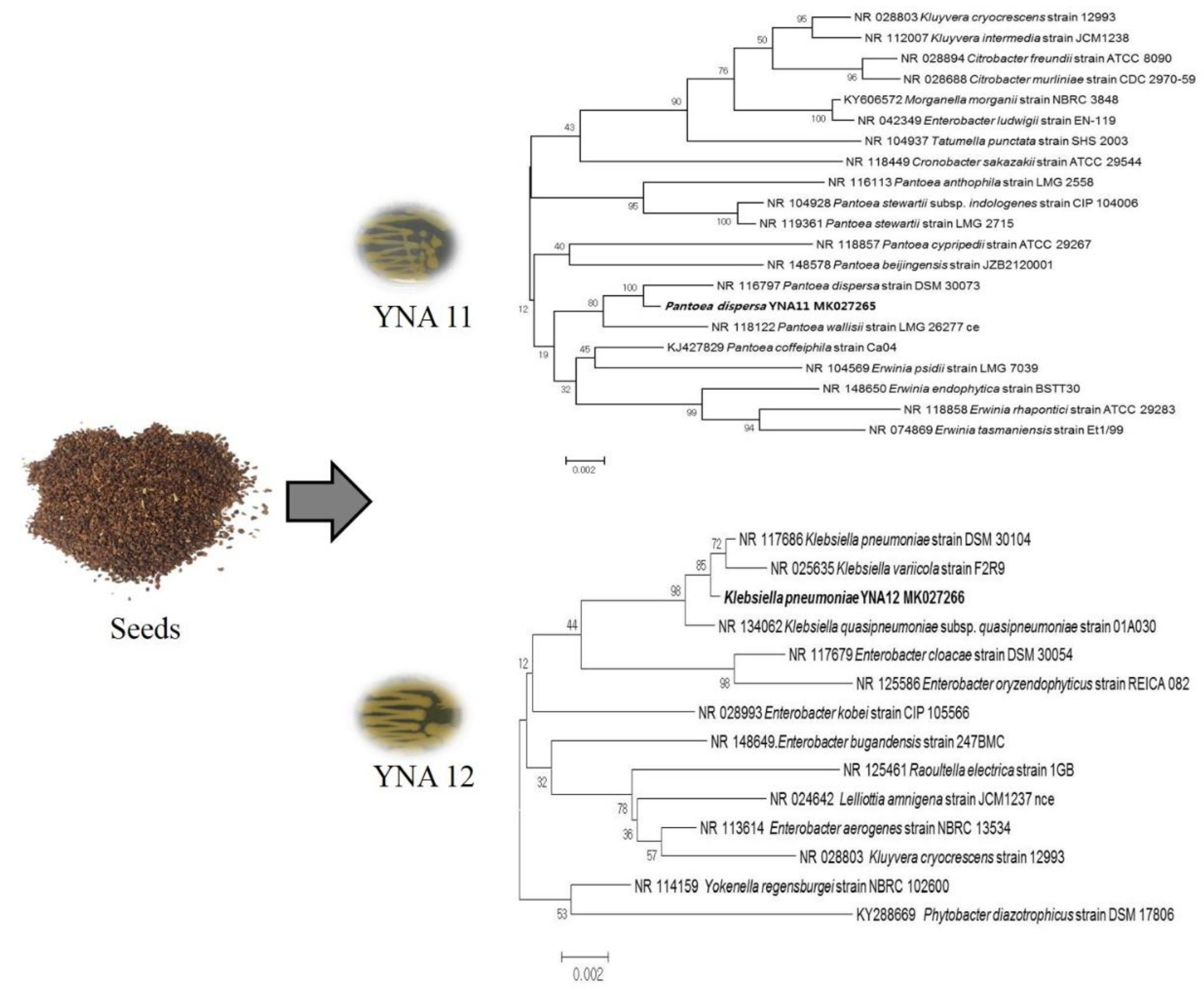
3.1. Identification and Phylogenetic Analysis of YNA11 and YNA12
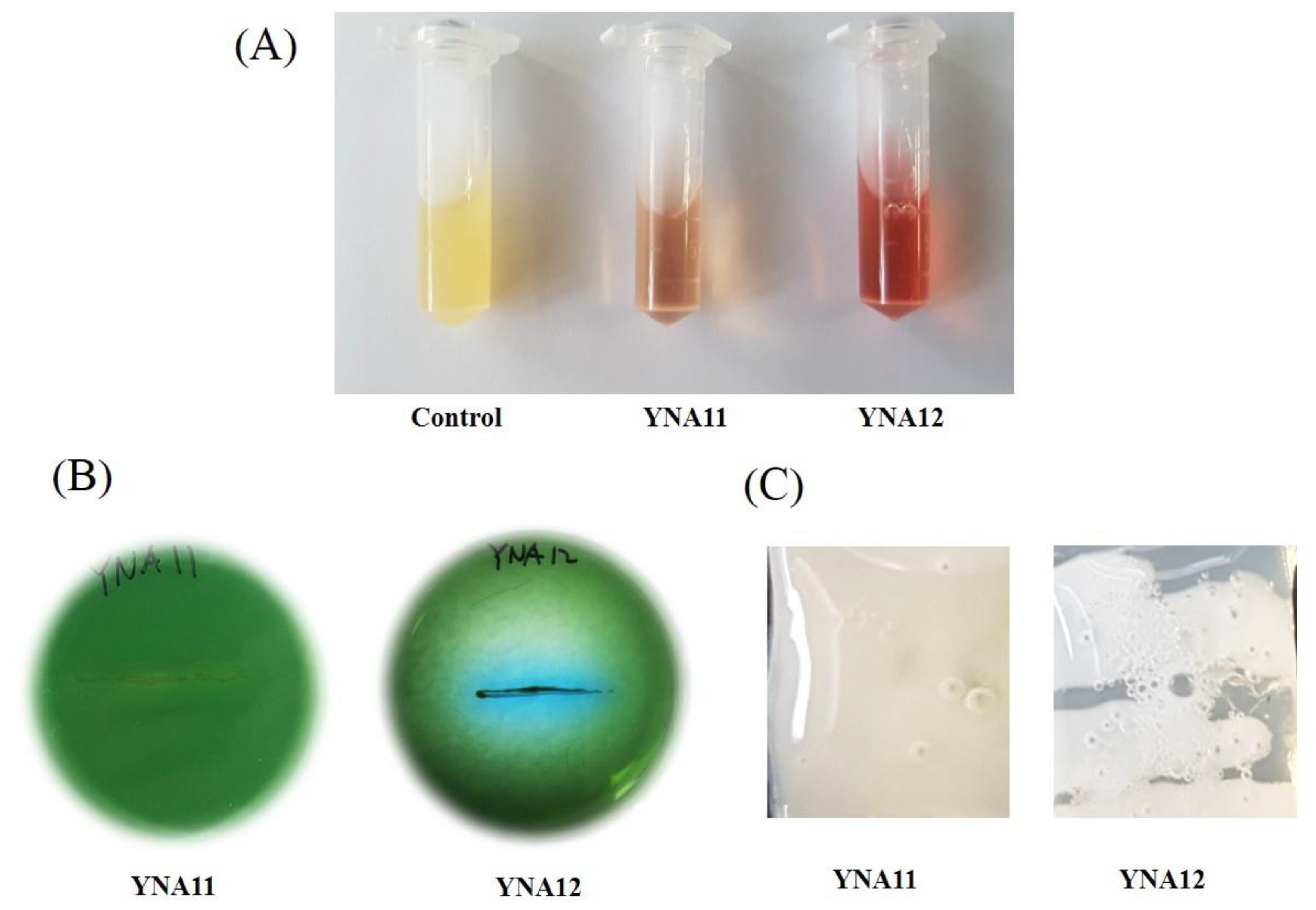
3.2. Biochemical Characterization
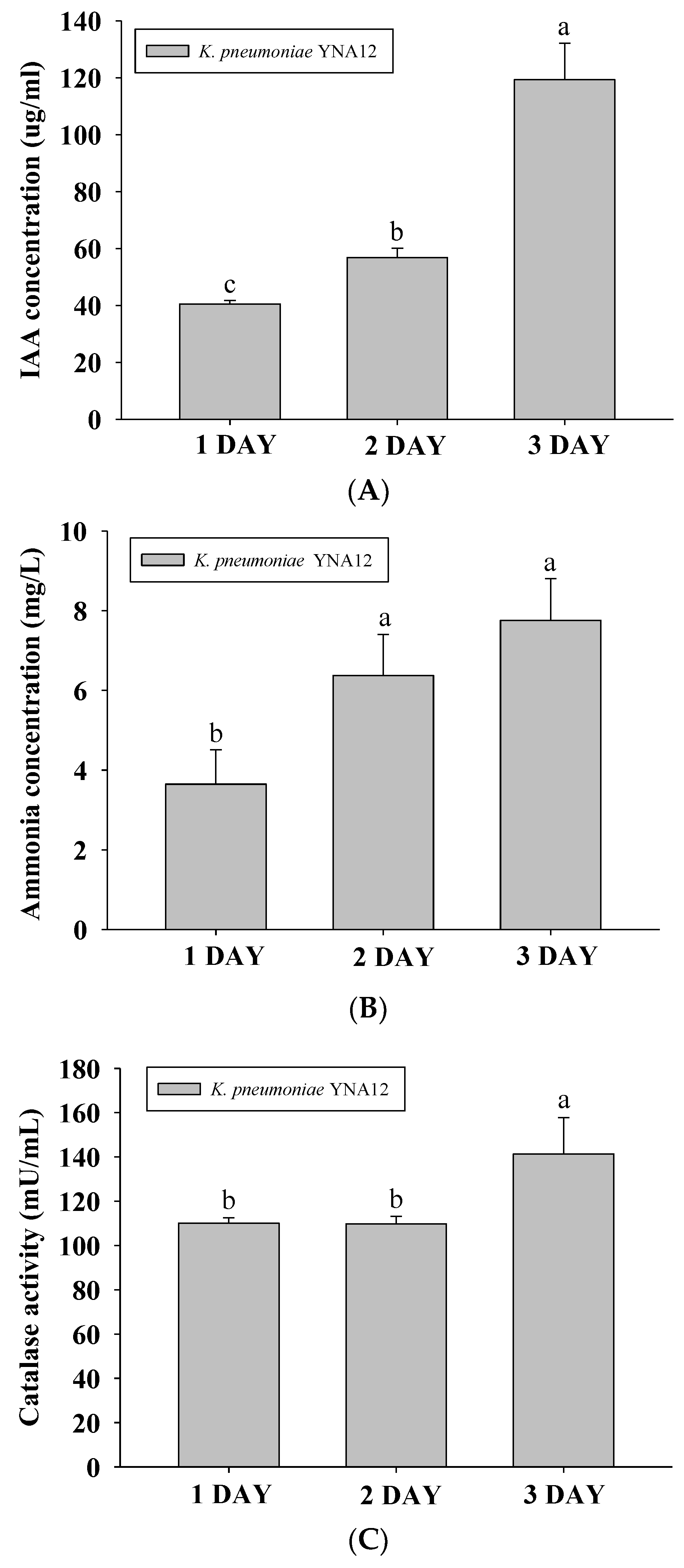
3.3. Quantification of Metabolite Production by YNA12
3.4. Adverse Effects of Endophytic YNA12 on Seed Germination of Evening Primrose
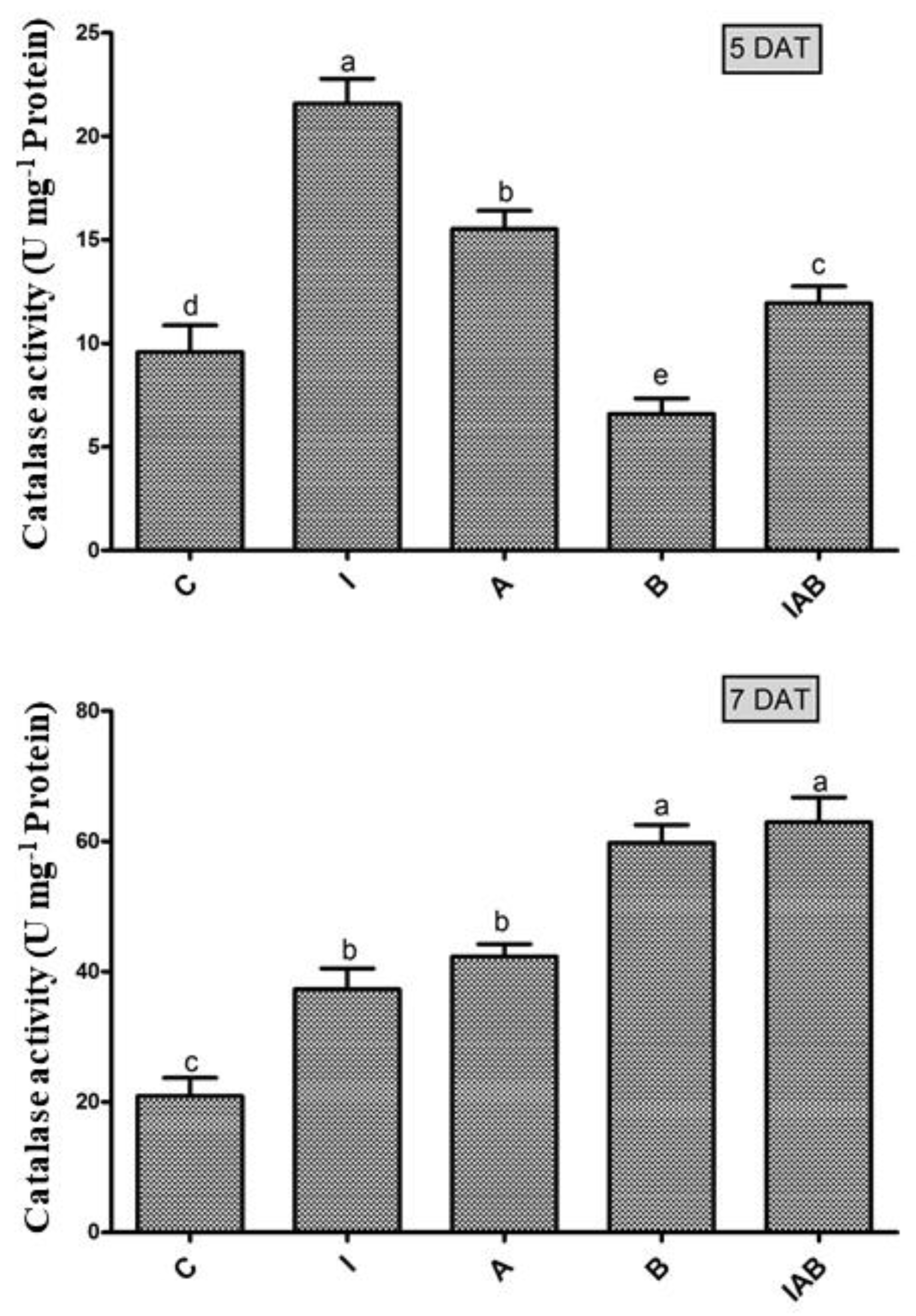
3.5. Effect of YNA12 on the Catalase Activity of Evening Primroses
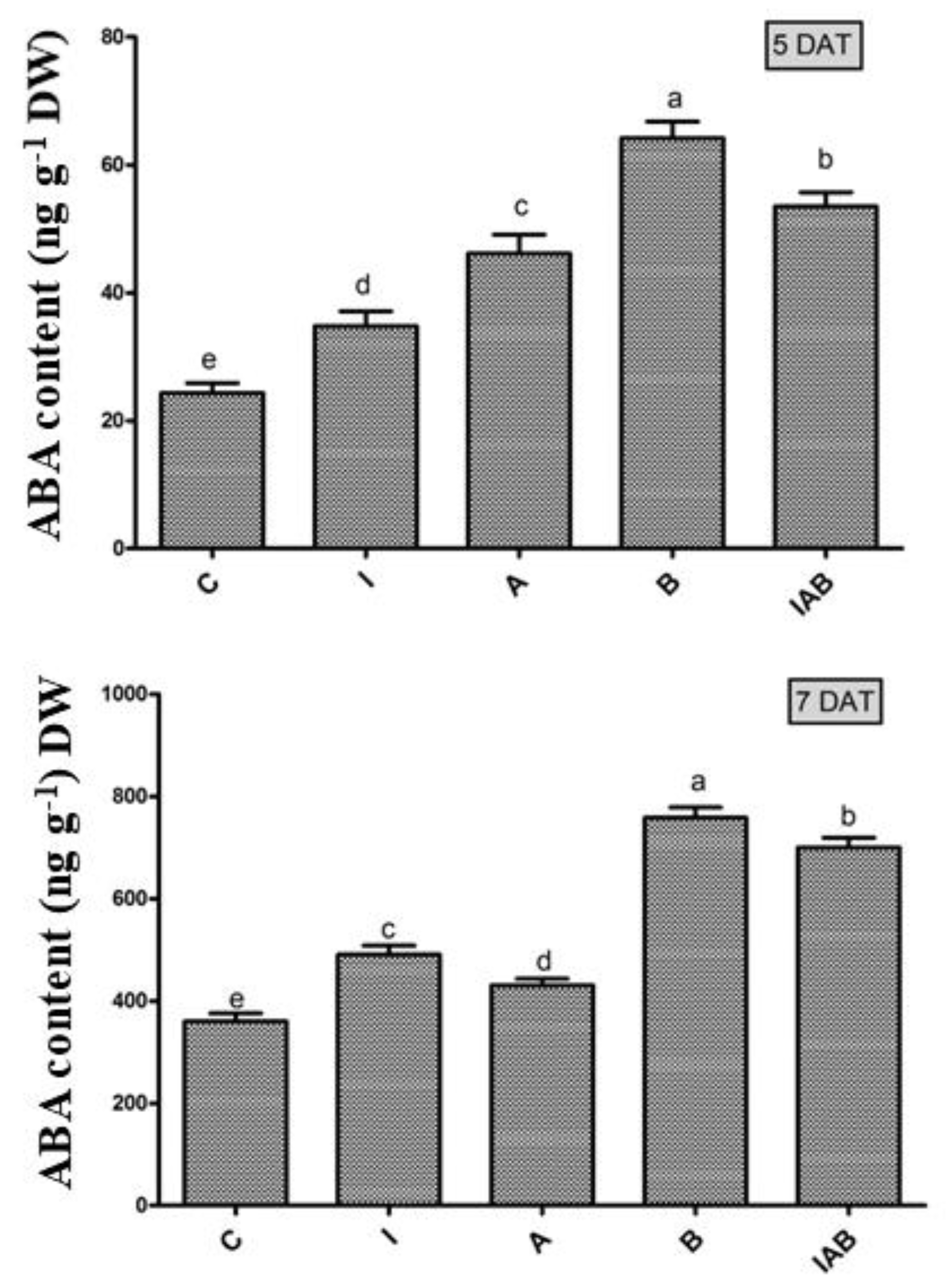
3.6. Effect of YNA12 on the Regulation of the Endogenous ABA of Evening Primroses
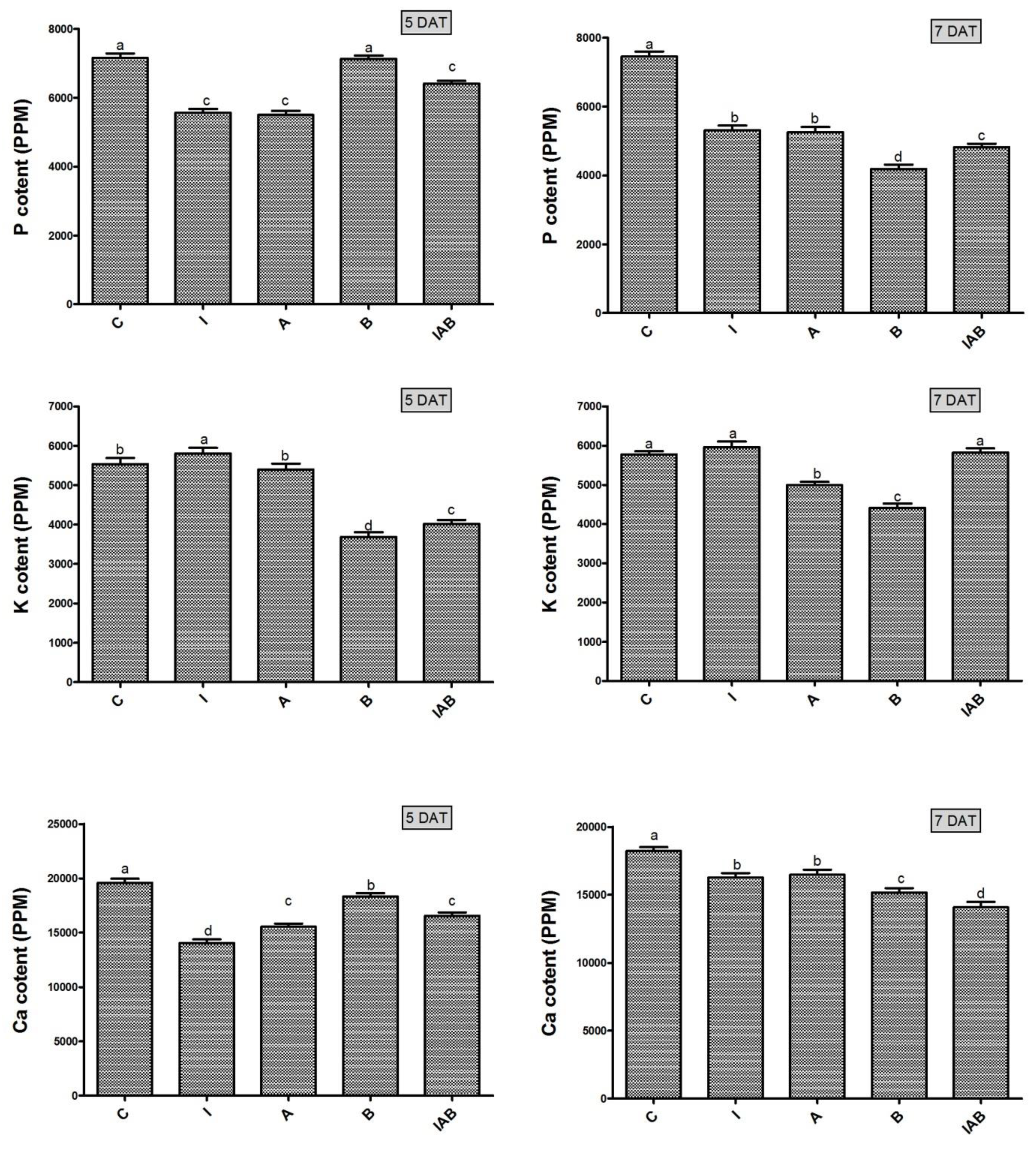
3.7. Effect of YNA12 on the Macronutrient Regulation of Evening Primroses
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Bennett, J.A.; Klironomos, J. Mechanisms of plant–soil feedback: Interactions among biotic and abiotic drivers. New Phytol. 2019, 222, 91–96. [Google Scholar] [CrossRef]
- Prasad, M.; Srinivasan, R.; Chaudhary, M.; Choudhary, M.; Jat, L.K. Plant Growth Promoting Rhizobacteria (PGPR) for Sustainable Agriculture: Perspectives and Challenges. In PGPR Amelioration in Sustainable Agriculture; Elsevier: Amsterdam, The Netherlands, 2019; pp. 129–157. [Google Scholar]
- Shahzad, R.; Khan, A.L.; Bilal, S.; Asaf, S.; Lee, I.-J. What is there in seeds? Vertically transmitted endophytic resources for sustainable improvement in plant growth. Front. Plant Sci. 2018, 9, 24. [Google Scholar] [CrossRef] [PubMed]
- Behrend, K.; Noohi, B. Moduli of non-commutative polarized schemes. Math. Ann. 2018, 371, 1375–1408. [Google Scholar] [CrossRef]
- Shahzad, R.; Khan, A.L.; Bilal, S.; Asaf, S.; Lee, I.-J. Plant growth-promoting endophytic bacteria versus pathogenic infections: An example of Bacillus amyloliquefaciens RWL-1 and Fusarium oxysporum f. sp. lycopersici in tomato. PeerJ 2017, 5, e3107. [Google Scholar] [CrossRef] [PubMed]
- Brimecombe, M.J.; De Leij, F.; Lynch, J.M. Rhizodeposition and microbial populations. In The Rhizosphere: Biochemistry and Organic Substances at the Soil-Plant Interface; CRC Press: Boca Raton, FL, USA; Taylor & Francis Group: London, UK, 2007; pp. 73–109. [Google Scholar]
- Park, J.-M.; Radhakrishnan, R.; Kang, S.-M.; Lee, I.-J. IAA producing Enterobacter sp. I-3 as a potent bio-herbicide candidate for weed control: A special reference with lettuce growth inhibition. Indian J. Microbiol. 2015, 55, 207–212. [Google Scholar] [CrossRef]
- Lakshmi, V.; Kumari, S.; Singh, A.; Prabha, C. Isolation and characterization of deleterious Pseudomonas aeruginosa KC1 from rhizospheric soils and its interaction with weed seedlings. J. King Saud Univ.-Sci. 2015, 27, 113–119. [Google Scholar] [CrossRef]
- Kai, M.; Crespo, E.; Cristescu, S.M.; Harren, F.J.; Francke, W.; Piechulla, B. Serratia odorifera: Analysis of volatile emission and biological impact of volatile compounds on Arabidopsis thaliana. Appl. Microbiol. Biotechnol. 2010, 88, 965–976. [Google Scholar] [CrossRef]
- Udeigwe, T.K.; Teboh, J.M.; Eze, P.N.; Stietiya, M.H.; Kumar, V.; Hendrix, J.; Mascagni, H.J., Jr.; Ying, T.; Kandakji, T. Implications of leading crop production practices on environmental quality and human health. J. Environ. Manag. 2015, 151, 267–279. [Google Scholar] [CrossRef]
- Ramankutty, N.; Mehrabi, Z.; Waha, K.; Jarvis, L.; Kremen, C.; Herrero, M.; Rieseberg, L.H. Trends in global agricultural land use: Implications for environmental health and food security. Ann. Rev. Plant Biol. 2018, 69, 789–815. [Google Scholar] [CrossRef]
- Zhang, L.; Yan, C.; Guo, Q.; Zhang, J.; Ruiz-Menjivar, J. The impact of agricultural chemical inputs on environment: Global evidence from informetrics analysis and visualization. Int. J. Low-Carbon Technol. 2018, 13, 338–352. [Google Scholar] [CrossRef]
- Thomashow, L.S.; Kwak, Y.S.; Weller, D.M. Root-associated microbes in sustainable agriculture: Models, metabolites and mechanisms. Pest Manag. Sci. 2019, 75, 2360–2367. [Google Scholar] [CrossRef] [PubMed]
- Trognitz, F.; Hackl, E.; Widhalm, S.; Sessitsch, A. The role of plant–microbiome interactions in weed establishment and control. FEMS Microbiol. Ecol. 2016, 92, 10. [Google Scholar] [CrossRef]
- Elshafie, H.-S.; Sakr, S.; Bufo, S.-A.; Camele, I. An attempt of biocontrol the tomato-wilt disease caused by Verticillium dahliae using Burkholderia gladioli pv. agaricicola and its bioactive secondary metabolites. Int. J. Plant Biol. 2017, 8, 7263. [Google Scholar] [CrossRef]
- Kang, S.-M.; Shahzad, R.; Bilal, S.; Khan, A.L.; You, Y.-H.; Lee, W.-H.; Ryu, H.-L.; Lee, K.-E.; Lee, I.-J. Metabolism-mediated induction of zinc tolerance in Brassica rapa by Burkholderia cepacia CS2-1. J. Microbiol. 2017, 55, 955–965. [Google Scholar] [CrossRef]
- Khan, A.L.; Bilal, S.; Halo, B.A.; Al-Harrasi, A.; Khan, A.R.; Waqas, M.; Al-Thani, G.S.; Al-Amri, I.; Al-Rawahi, A.; Lee, I.-J. Bacillus amyloliquefaciens BSL16 improves phytoremediation potential of Solanum lycopersicum during copper stress. J. Plant Interact. 2017, 12, 550–559. [Google Scholar] [CrossRef]
- Kang, S.-M.; Waqas, M.; Hamayun, M.; Asaf, S.; Khan, A.L.; Kim, A.-Y.; Park, Y.-G.; Lee, I.-J. Gibberellins and indole-3-acetic acid producing rhizospheric bacterium Leifsonia xyli SE134 mitigates the adverse effects of copper-mediated stress on tomato. J. Plant Interact. 2017, 12, 373–380. [Google Scholar] [CrossRef]
- MacWilliams, M.P. Citrate Test Protocol. Available online: https://www.asmscience.org/content/education/protocol/protocol.3203# (accessed on 8 December 2009).
- Tiwari, P.; Singh, J.S. A plant growth promoting rhizospheric Pseudomonas aeruginosa strain inhibits seed germination in Triticum aestivum (L) and Zea mays (L). Microbiol. Res. 2017, 8, 2. [Google Scholar] [CrossRef]
- Lee, K.-E.; Adhikari, A.; Kang, S.-M.; You, Y.-H.; Joo, G.-J.; Kim, J.-H.; Kim, S.-J.; Lee, I.-J. Isolation and characterization of the high silicate and phosphate solubilizing novel strain Enterobacter ludwigii GAK2 that promotes growth in rice plants. Agronomy 2019, 9, 144. [Google Scholar] [CrossRef]
- Park, Y.-G.; Mun, B.-G.; Kang, S.-M.; Hussain, A.; Shahzad, R.; Seo, C.-W.; Kim, A.-Y.; Lee, S.-U.; Oh, K.Y.; Lee, D.Y. Bacillus aryabhattai SRB02 tolerates oxidative and nitrosative stress and promotes the growth of soybean by modulating the production of phytohormones. PLoS ONE 2017, 12, e0173203. [Google Scholar] [CrossRef]
- Qi, Q.; Rose, P.A.; Abrams, G.D.; Taylor, D.C.; Abrams, S.R.; Cutler, A.J. (+)-Abscisic acid metabolism, 3-ketoacyl-coenzyme a synthase gene expression, and very-long-chain monounsaturated fatty acid biosynthesis inBrassica napus embryos. Plant Physiol. 1998, 117, 979–987. [Google Scholar] [CrossRef]
- Bilal, S.; Khan, A.L.; Shahzad, R.; Asaf, S.; Kang, S.-M.; Lee, I.-J. Endophytic Paecilomyces formosus LHL10 augments Glycine max L. adaptation to Ni-contamination through affecting endogenous phytohormones and oxidative stress. Front. Plant Sci. 2017, 8, 870. [Google Scholar] [CrossRef] [PubMed]
- Bilal, S.; Shahzad, R.; Khan, A.L.; Kang, S.-M.; Imran, Q.M.; Al-Harrasi, A.; Yun, B.-W.; Lee, I.-J. Endophytic microbial consortia of phytohormones-producing fungus Paecilomyces formosus LHL10 and bacteria Sphingomonas sp. LK11 to Glycine max L. regulates physio-hormonal changes to attenuate aluminum and zinc stresses. Front. Plant Sci. 2018, 9, 1273. [Google Scholar] [CrossRef] [PubMed]
- Harding, D.P.; Raizada, M.N. Controlling weeds with fungi, bacteria and viruses: A review. Front. Plant Sci. 2015, 6, 659. [Google Scholar] [CrossRef] [PubMed]
- Gomes, R.; Glienke, C.; Videira, S.; Lombard, L.; Groenewald, J.; Crous, P. Diaporthe: A genus of endophytic, saprobic and plant pathogenic fungi. Pers. Mol. Phyl. Evolut. Fungi 2013, 31, 1. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, A.; Naveed, M.; Saeed, Q.; Ashraf, M.-N.; Hussain, A.; Abbas, T.; Kamran, M.; Minggang, X. Application potentials of plant growth promoting rhizobacteria and fungi as an alternative to conventional weed control methods. InCrop Prod. 2019. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, S.-Q.; Tang, H.-Y.; Li, X.-J.; Zhang, L.; Xiao, J.; Gao, Y.-Q.; Zhang, A.-L.; Gao, J.-M. Potential allelopathic indole diketopiperazines produced by the plant endophytic Aspergillus fumigatus using the one strain–many compounds method. J. Agric. Food Chem. 2013, 61, 11447–11452. [Google Scholar] [CrossRef]
- Barghouthi, S.; Salman, M. Bacterial inhibition of Orobanche aegyptiaca and Orobanche cernua radical elongation. Biocontrol Sci. Technol. 2010, 20, 423–435. [Google Scholar] [CrossRef]
- Weise, T.; Kai, M.; Piechulla, B. Bacterial ammonia causes significant plant growth inhibition. PLoS ONE 2013, 8, e63538. [Google Scholar] [CrossRef]
- Tabatabaei, S.; Ehsanzadeh, P.; Etesami, H.; Alikhani, H.A.; Glick, B.R. Indole-3-acetic acid (IAA) producing Pseudomonas isolates inhibit seed germination and α-amylase activity in durum wheat (Triticum turgidum L.). Span. J. Agric. Res. 2016, 14, 15. [Google Scholar] [CrossRef]
- Shu, K.; Liu, X.-D.; Xie, Q.; He, Z.-H. Two faces of one seed: Hormonal regulation of dormancy and germination. Mol. Plant 2016, 9, 34–45. [Google Scholar] [CrossRef]
- Radhakrishnan, R.; Park, J.-M.; Lee, I.-J. Enterobacter sp. I-3, a bio-herbicide inhibits gibberellins biosynthetic pathway and regulates abscisic acid and amino acids synthesis to control plant growth. Microbiol. Res. 2016, 193, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Kong, D.; Ju, C.; Parihar, A.; Kim, S.; Cho, D.; Kwak, J.M. Arabidopsis glutamate receptor homolog3. 5 modulates cytosolic Ca2+ level to counteract effect of abscisic acid in seed germination. Plant Physiol. 2015, 167, 1630–1642. [Google Scholar] [CrossRef] [PubMed]
- Barazani, O.-Z.; Friedman, J. Allelopathic bacteria and their impact on higher plants. Crit. Rev. Plant Sci. 1999, 18, 741–755. [Google Scholar] [CrossRef]
- Mantovani, C.; Prado, R.M.; Pivetta, K.F. Impact of Nitrate and Ammonium ratio on Nutrition and Growth of two Epiphytic Orchids. An. Acad. Bras. Ciênc. 2018, 90, 3423–3431. [Google Scholar] [CrossRef] [PubMed]
| Treatments | GR(%) | SL(cm) | SW(mg) | CDF(%) | ||||
|---|---|---|---|---|---|---|---|---|
| 5D | 7D | 5D | 7D | 5D | 7D | 5D | 7D | |
| Control | 38.3 ± 3.5a | 50.0 ± 8.66a | 1.54 ± 0.09a | 2.10 ± 0.27a | 2.58 ± 0.28a | 4.1 ± 0.8a | 52.2 | 73.3 |
| IAA 100 ppm | 29.0 ± 4.0b | 36.6 ± 2.88b | 0.84 ± 0.05b | 1.20 ± 0.13b | 1.57 ± 0.30b | 2.1 ± 0.22b | 13.5 | 17.6 |
| Ammonia 10 ppm | 16.0 ± 1.7c | 29.0 ± 1.73c | 0.58 ± 0.08c | 1.08 ± 0.18b | 1.00 ± 0.04c | 1.38 ± 0.13c | 11.5 | 15.6 |
| YNA 12 Culture Filtrate | 11.0 ± 2.6d | 22.3 ± 2.65d | 0.30 ± 0.04d | 0.40 ± 0.029c | 0.84 ± 0.03d | 1.12 ± 0.15c | 5.2 | 13.6 |
| A+I+YNA12 | 10.0 ± 1.0d | 16.7 ± 1.53e | 0.20 ± 0.04e | 0.34 ± 0.041c | 0.76 ± 0.05d | 1.06 ± 0.14c | 3.2 | 10.0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, S.-M.; Bilal, S.; Shahzad, R.; Kim, Y.-N.; Park, C.-W.; Lee, K.-E.; Lee, J.-R.; Lee, I.-J. Effect of Ammonia and Indole-3-acetic Acid Producing Endophytic Klebsiella pneumoniae YNA12 as a Bio-Herbicide for Weed Inhibition: Special Reference with Evening Primroses. Plants 2020, 9, 761. https://doi.org/10.3390/plants9060761
Kang S-M, Bilal S, Shahzad R, Kim Y-N, Park C-W, Lee K-E, Lee J-R, Lee I-J. Effect of Ammonia and Indole-3-acetic Acid Producing Endophytic Klebsiella pneumoniae YNA12 as a Bio-Herbicide for Weed Inhibition: Special Reference with Evening Primroses. Plants. 2020; 9(6):761. https://doi.org/10.3390/plants9060761
Chicago/Turabian StyleKang, Sang-Mo, Saqib Bilal, Raheem Shahzad, Yu-Na Kim, Chang-Wook Park, Ko-Eun Lee, Jeong-Ran Lee, and In-Jung Lee. 2020. "Effect of Ammonia and Indole-3-acetic Acid Producing Endophytic Klebsiella pneumoniae YNA12 as a Bio-Herbicide for Weed Inhibition: Special Reference with Evening Primroses" Plants 9, no. 6: 761. https://doi.org/10.3390/plants9060761
APA StyleKang, S.-M., Bilal, S., Shahzad, R., Kim, Y.-N., Park, C.-W., Lee, K.-E., Lee, J.-R., & Lee, I.-J. (2020). Effect of Ammonia and Indole-3-acetic Acid Producing Endophytic Klebsiella pneumoniae YNA12 as a Bio-Herbicide for Weed Inhibition: Special Reference with Evening Primroses. Plants, 9(6), 761. https://doi.org/10.3390/plants9060761






