Somatic Embryogenesis from Mature Embryos of Olea europaea L. cv. ‘Galega Vulgar’ and Long-Term Management of Calli Morphogenic Capacity
Abstract
1. Introduction
2. Results and Discussion
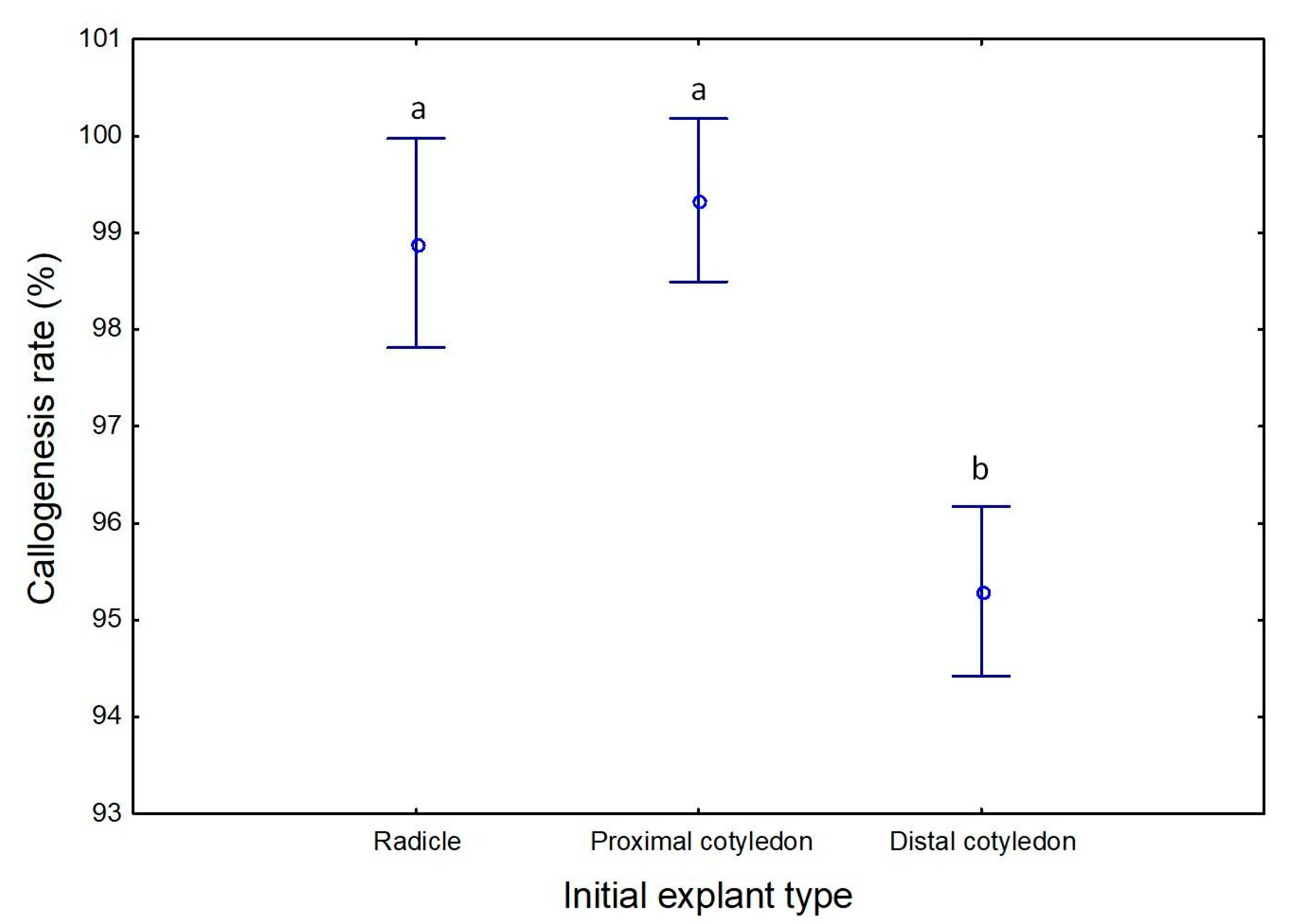
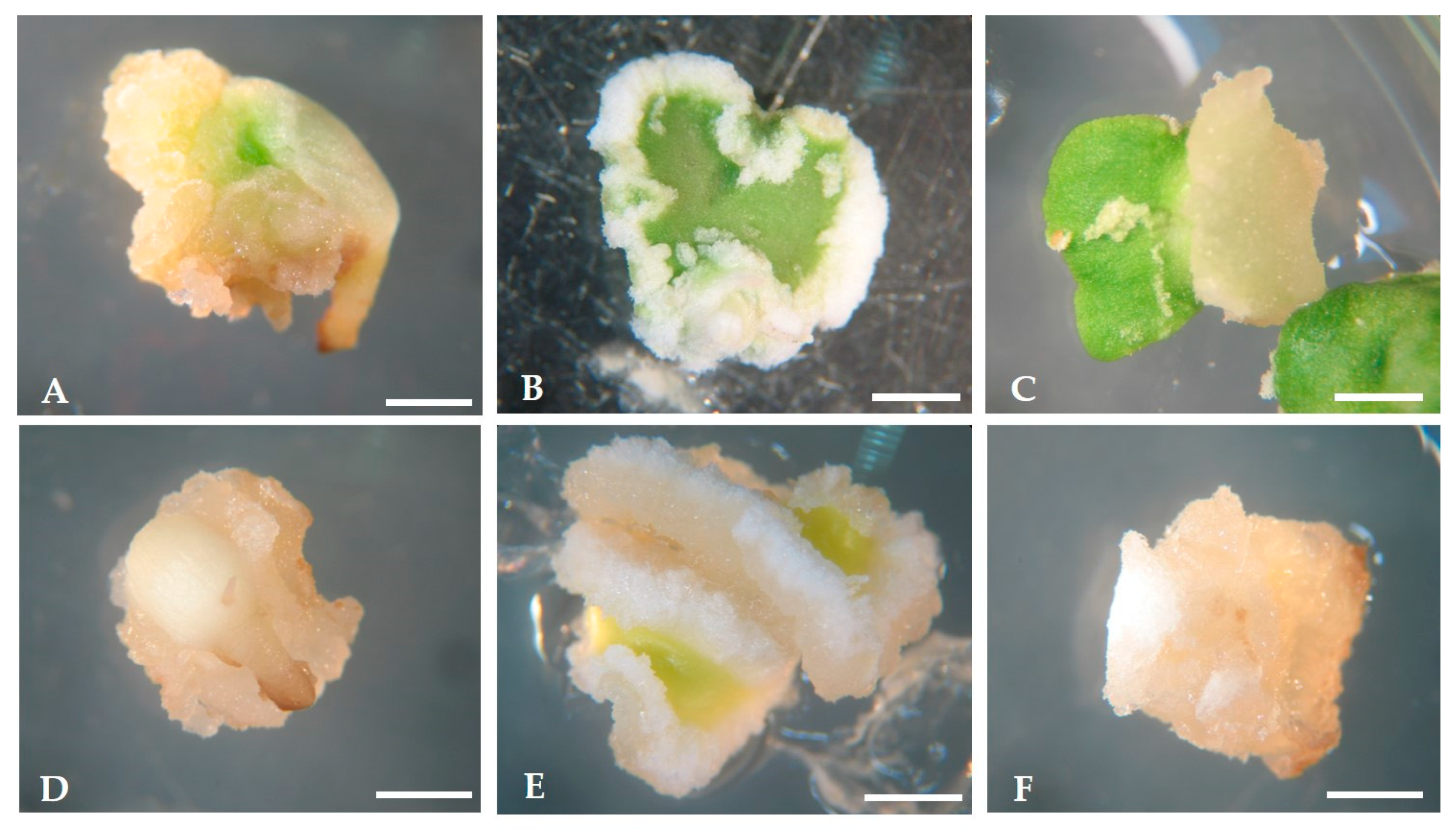
2.1. Induction of Somatic Embryogenesis
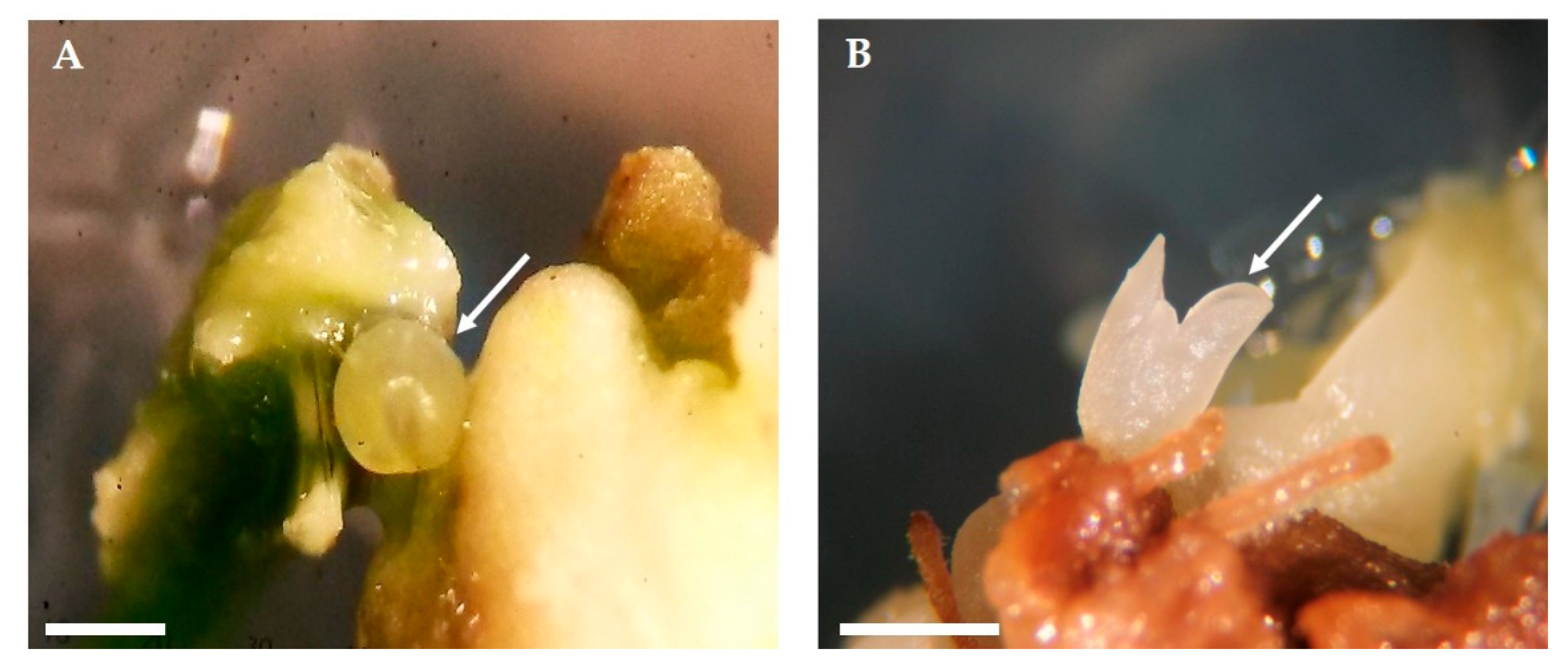
2.2. Expression of Somatic Embryogenesis
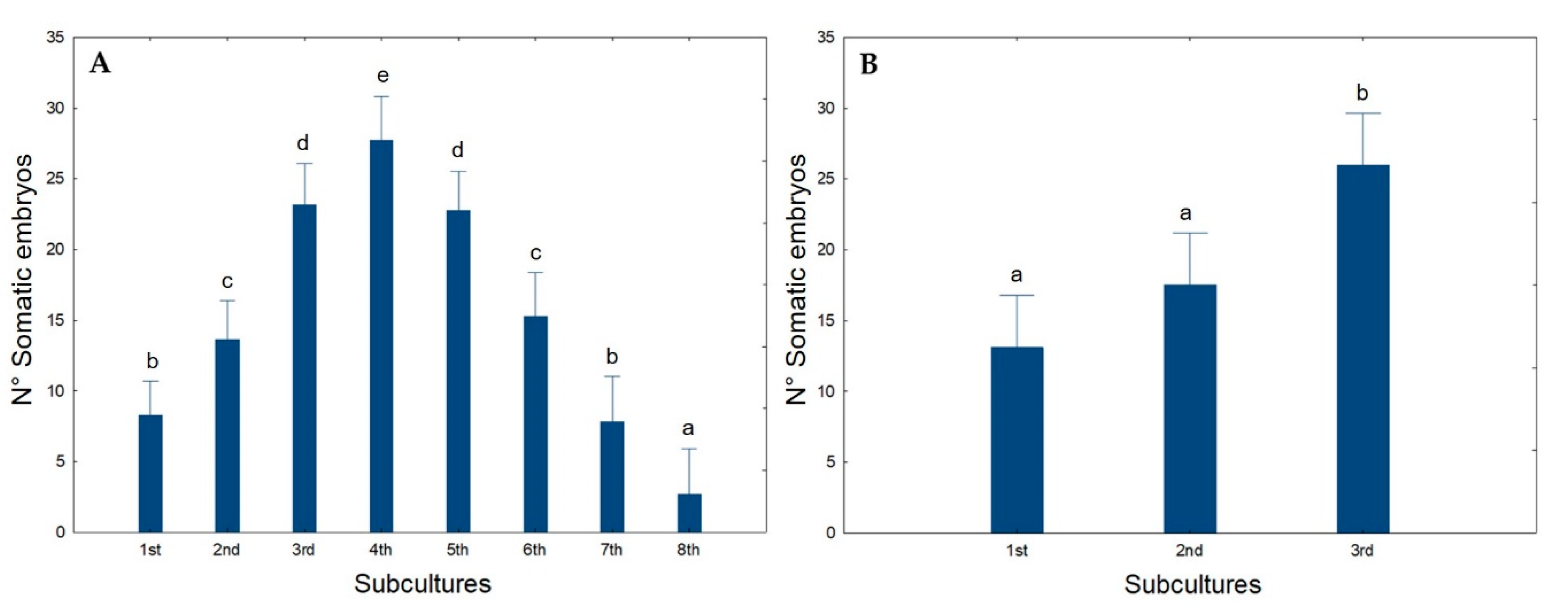
2.3. Management of Embryogenic Potential-Cyclic Embryogenesis Induction and Maintenance
2.4. Embryo Conversion and Plant Acclimatization
3. Materials and Methods
3.1. Plant Material
3.2. Seed Surface Disinfection and Explant Preparation
3.3. Embryogenesis Induction and Expression
3.4. Cyclic Embryogenesis
3.5. Recover of Calli Embryogenic Capacity
3.6. Embryos Conversion and Plants Acclimatation
3.7. Experimental Design and Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rallo, L.; Barranco, D.; Díez, C.M.; Rallo, P.; Suárez, M.P.; Trapero, C.; Pliego-Alfaro, F. Strategies for Olive (Olea europaea L.) Breeding: Cultivated Genetic Resources and Crossbreeding. In Advances in Plant Breeding Strategies: Fruits; Al-Khayri, J.M., Jain, S.M., Johnson, D.V., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 535–600. ISBN 978-3-319-91943-0. [Google Scholar]
- Besnard, G.; Bervillé, A. Multiple origins for Mediterranean olive (Olea europaea L. ssp. europaea) based upon mitochondrial DNA polymorphisms. Comptes Rendus l’Académie Sci. Ser. III Sci. 2000, 323, 173–181. [Google Scholar] [CrossRef]
- Besnard, G.; Khadari, B.; Baradat, P.; Bervillé, A. Olea europaea (Oleaceae) phylogeography based on chloroplast DNA polymorphism. Theor. Appl. Genet. 2002, 104, 1353–1361. [Google Scholar] [CrossRef]
- Khdari, B.; Bakkali, A.E.; Essalouh, L.; Tollon, C.; Pinatel, C.; Besnard, G. Cultivated Olive Diversification at Local and Regional Scales: Evidence From the Genetic Characterization of French Genetic Resources. Front. Plant Sci. 2019, 10, 1593. [Google Scholar] [CrossRef] [PubMed]
- Miazzi, M.; di Rienzo, V.; Mascio, I.; Montemurro, C.; Sion, S.; Sabetta, W.; Vivaldi, G.; Camposeo, S.; Caponio, F.; Squeo, G.; et al. Re.Ger. OP: Integrated Project for the Recovery of Ancient and Rare Olive Germplasm. Front. Plant Sci. 2020, 11, 73. [Google Scholar] [CrossRef] [PubMed]
- Muleo, R.; Morgante, M.; Velasco, R.; Cavallini, A.; Perrota, G.; Baldoni, L. Olive Tree Genomic. In Olive Germplasm—The Olive Cultivation, Table Olive and Olive Oil Industry in Italy; Muzzalupo, I., Ed.; InTech: Rijeka, Croatia, 2012; ISBN 978-953-51-0883-2. [Google Scholar]
- Rugini, E.; Cristofori, V.; Silvestri, C. Genetic improvement of olive (Olea europaea L.) by conventional and in vitro biotechnology methods. Biotechnol. Adv. 2016, 34, 687–696. [Google Scholar] [CrossRef] [PubMed]
- Sebastiani, L.; Busconi, M. Recent developments in olive (Olea europaea L.) genetics and genomics: Applications in taxonomy, varietal identification, traceability and breeding. Plant Cell Rep. 2017, 36, 1345–1360. [Google Scholar] [CrossRef]
- Alagna, F.; D’Agostino, N.; Torchia, L.; Servili, M.; Rao, R.; Pietrella, M.; Giuliano, G.; Chiusano, M.; Baldoni, L.; Perrotta, G. Comparative 454 pyrosequencing of transcripts from two olive genotypes during fruit development. BMC Genom. 2009, 10, 399. [Google Scholar] [CrossRef]
- Alagna, F.; Cirilli, M.; Galla, G.; Carbone, F.; Daddiego, L.; Facella, P.; Lopez, L.; Colao, C.; Mariotti, R.; Cultrera, N.; et al. Transcript Analysis and Regulative Events during Flower Development in Olive (Olea europaea L.). PLoS ONE 2016, 11, e0152943. [Google Scholar] [CrossRef]
- Jiménez-Ruiz, J.; Leyva-Pérez, M.O.; Schilirò, E.; Barroso, J.B.; Bombarely, A.; Mueller, L.; Mercado-Blanco, J.; Luque, F. Transcriptomic Analysis of Olea europaea L. Roots during the Verticillium dahliae Early Infection Process. Plant Genome 2017, 10. [Google Scholar] [CrossRef]
- Roka, L.; Koudounas, K.; Daras, G.; Zoidakis, J.; Vlahou, A.; Kalaitzis, P.; Hatzopoulos, P. Proteome of olive non-glandular trichomes reveals protective protein network against (a)biotic challenge. J. Plant Physiol. 2018, 231, 210–218. [Google Scholar] [CrossRef]
- D’Angeli, S.; Altamura, M.M. Osmotin induces cold protection in olive trees by affecting programmed cell death and cytoskeleton organization. Planta 2007, 225, 1147–1163. [Google Scholar] [CrossRef] [PubMed]
- Haberman, A.; Bakhshian, O.; Cerezo-Medina, S.; Paltiel, J.; Adler, C.; Ben-Ari, G.; Mercado, J.A.; Pliego-Alfaro, F.; Lavee, S.; Samach, A. A possible role for flowering locus T-encoding genes in interpreting environmental and internal cues affecting olive (Olea europaea L.) flower induction: Olive flowering. Plant Cell Environ. 2017, 40, 1263–1280. [Google Scholar] [CrossRef] [PubMed]
- Narváez, I.; Khayreddine, T.; Pliego, C.; Cerezo, S.; Jiménez-Díaz, R.M.; Trapero-Casas, J.L.; López-Herrera, C.; Arjona-Girona, I.; Martín, C.; Mercado, J.A.; et al. Usage of the Heterologous Expression of the Antimicrobial Gene afp From Aspergillus giganteus for Increasing Fungal Resistance in Olive. Front. Plant Sci. 2018, 9, 680. [Google Scholar] [CrossRef] [PubMed]
- Arnholdt-Schmitt, B.; Ragonezi, C.; Cardoso, H. Do Mitochondria Play a Central Role in Stress-Induced Somatic Embryogenesis? In In Vitro Embryogenesis in Higher Plants; Germana, M.A., Lambardi, M., Eds.; Methods in Molecular Biology; Springer New York: New York, NY, USA, 2016; Volume 1359, pp. 87–100. ISBN 978-1-4939-3060-9. [Google Scholar]
- Gray, D.J.; Mortensen, J.A. Initiation and maintenance of long term somatic embryogenesis from anthers and ovaries of Vitis longii ‘Microsperma’. Plant Cell Tissue Organ Cult. 1987, 9, 73–80. [Google Scholar] [CrossRef]
- Matsuta, N.; Hirabayashi, T. Embryogenic cell lines from somatic embryos of grape (Vitis vinifera L.). Plant Cell Rep. 1989, 7, 684–687. [Google Scholar] [CrossRef]
- Martinelli, L.; Bragagna, P.; Poletti, V.; Scienza, A. Somatic embryogenesis from leaf- and petiole-derived callus of Vitis rupestris. Plant Cell Rep. 1993, 12. [Google Scholar] [CrossRef]
- Perl, A.; Saad, S.; Sahar, N.; Holland, D. Establishment of long-term embryogenic cultures of seedless Vitis vinifera cultivars—A synergistic effect of auxins and the role of abscisic acid. Plant Sci. 1995, 104, 193–200. [Google Scholar] [CrossRef]
- Toonen, M.A.J.; de Vries, S.C. Initiation of somatic embryos from single cells. In Embryogenesis: The Generation of a Plant; Wang, T.L., Cuming, A., Eds.; Bios Scientific Publishers: Oxford, UK, 1996; pp. 173–189. [Google Scholar]
- Rugini, E.; Pannelli, G.; Ceccarelli, M.; Muganu, M. Isolation of triploid and tetraploid olive (Olea europaea L.) plants from mixoploid cv. “Frantoio” and “Leccino” mutants by in vivo and in vitro selection. Plant Breed. 1996, 115, 23–27. [Google Scholar] [CrossRef]
- Rugini and Tarini. Somatic embryogenesis in olive (Olea europaea L.). In Proceeding Conference Fruit Tree Biotechnology; Moët-Hennessy: Monte Carlo, Monaco, 1986; p. 62.
- Rugini, E. Somatic embryogenesis and plant regeneration in olive (Olea europaea L.). Plant Cell Tissue Organ Cult. 1988, 14, 207–214. [Google Scholar] [CrossRef]
- Mitrakos, K.; Alexaki, A.; Papadimitriou, P. Dependence of Olive Morphogenesis on Callus Origin and Age. J. Plant Physiol. 1992, 139, 269–273. [Google Scholar] [CrossRef]
- Leva, A.; Muleo, R.; Petruccelli, R. Long-term somatic embryogenesis from immature olive cotyledons. J. Hortic. Sci. 1995, 70, 417–421. [Google Scholar] [CrossRef]
- Brhadda, N.; Abousalim, A. Loudyi Dou Elmacane Walali Effets du milieu de culture et de la lumière sur l’embryogenèse somatique de l’olivier (Olea europaea L.) cv. Picholine marocaine. Fruits 2003, 58, 167–174. [Google Scholar] [CrossRef]
- Trabelsi, E.B.; Bouzid, S.; Bouzid, M.; Elloumi, N.; Belfeleh, Z.; Benabdallah, A.; Ghezel, R. In-vitro regeneration of olive tree by somatic embryogenesis. J. Plant Biol. 2003, 46, 173–180. [Google Scholar] [CrossRef]
- Pritsa, T.S.; Voyiatzis, D.G. The in vitro morphogenetic capacity of olive embryo explants at different developmental stages, as affected by L-Glutamine, L-Arginine and 2,4-D. J. Biol. Res. 2004, 1, 55–61. [Google Scholar]
- Cerezo, S.; Mercado, J.A.; Pliego-Alfaro, F. An efficient regeneration system via somatic embryogenesis in olive. Plant Cell Tissue Organ Cult. 2011, 106, 337–344. [Google Scholar] [CrossRef]
- Jafarzadeh-Bajestani, M.; Khodai-Kalaki, M.; Motamed, N. Genetic transformation of olive somatic embryos through Agrobacterium tumefaciens and regeneration of transgenic plants. Afr. J. Biotechnol. 2011, 10, 5468–5475. [Google Scholar] [CrossRef]
- Cardoso, H.; Figueiredo, A.; Serrazina, S.; Pires, R.; Peixe, A. Genome modification approaches to improve performance, quality and stress tolerance of important Mediterranean fruit species (Olea europaea L., Vitis vinifera L., and Quercus suber L.). In Advances in Plant Transgenics: Methods and Applications; Sathishkumar, R., Kumar, S.R., Hema, J., Baskar, V., Eds.; Springer: Singapore, 2019; pp. 273–312. [Google Scholar] [CrossRef]
- Sánchez-Romero, C. Somatic embryogenesis in Olea spp. Plant Cell Tissue Organ Cult. 2019, 138, 403–426. [Google Scholar] [CrossRef]
- Rugini, E.; Caricato, G. Somatic embryogenesis and plant recovery from mature tissues of olive cultivars (Olea europaea L.) ‘Canino’ and ‘Moraiolo’. Plant Cell Rep. 1995, 14. [Google Scholar] [CrossRef]
- Trabelsi, E.B.; Naija, S.; Elloumi, N.; Belfeleh, Z.; Msellem, M.; Ghezel, R.; Bouzid, S. Somatic embryogenesis in cell suspension cultures of olive Olea europaea (L.) ‘Chetoui’. Acta Physiol. Plant. 2011, 33, 319–324. [Google Scholar] [CrossRef]
- Mazri, M.A.; Belkoura, I.; Pliego-Alfaro, F.; Belkoura, M. Somatic embryogenesis from leaf and petiole explants of the Moroccan olive cultivar Dahbia. Sci. Hortic. 2013, 159, 88–95. [Google Scholar] [CrossRef]
- Toufik, I.; Guenoun, F.; Belkoura, I. Embryogenesis expression from somatic explants of olive (Olea europaea L.) cv. Picual. Moroc. J. Biol. 2014, 11, 25. [Google Scholar]
- Capelo, A.M.; Silva, S.; Brito, G.; Santos, C. Somatic embryogenesis induction in leaves and petioles of a mature wild olive. Plant Cell Tissue Organ Cult. 2010, 103, 237–242. [Google Scholar] [CrossRef]
- Narváez, I.; Martín, C.; Jiménez-Díaz, R.M.; Mercado, J.A.; Pliego-Alfaro, F. Plant Regeneration via Somatic Embryogenesis in Mature Wild Olive Genotypes Resistant to the Defoliating Pathotype of Verticillium dahliae. Front. Plant Sci. 2019, 10, 1471. [Google Scholar] [CrossRef]
- Cardoso, H.G.; Campos, M.C.; Pais, M.S.; Peixe, A. Use of morphometric parameters for tracking ovule and evolution in grapevine (Vitis vinifera L., cv. “Aragonez”) and evaluation of their potential to improve in vitro somatic embryogenesis efficiency from gametophyte tissues. In Vitro Cell. Dev. Biol. Plant 2010, 46, 499–508. [Google Scholar] [CrossRef][Green Version]
- Cardoso, H.G.; Campos, M.C.; Pais, M.S.; Peixe, A. Somatic Embryogenesis in Iberian Grapevine (Vitis vinifera) Cultivars Using Carpels as Initial Explants: Protocol Establishment and Histological Evaluation. JAST-B 2019, 9. [Google Scholar] [CrossRef]
- Parrott, W.A.; Merkle, S.A.; Williams, E.G. Somatic embryogenesis: Potential for use in propagation and gene transfer systems. In Advanced Methods in Plant Breeding and Biotechnology; Murray, D.R., Ed.; CAB International: Wallingford, UK, 1991. [Google Scholar]
- Canhoto, J.M. Biotecnologia Vegetal: Da Clonagem de Plantas à Transformação Genética; Imprensa da Universidade de Coimbra: Coimbra, Portugal, 2010; ISBN 978-989-26-0404-6. [Google Scholar]
- Loyola-Vargas, V.M. The History of Somatic Embryogenesis. In Somatic Embryogenesis: Fundamental Aspects and Applications; Loyola-Vargas, V.M., Ochoa-Alejo, N., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 11–22. ISBN 978-3-319-33704-3. [Google Scholar]
- Merkle, S.A.; Parrott, W.A.; Flinn, B.S. Morphogenic Aspects of Somatic Embryogenesis. In In Vitro Embryogenesis in Plants; Thorpe, T.A., Ed.; Springer: Dordrecht, The Netherlands, 1995; Volume 20, pp. 155–203. ISBN 978-94-010-4217-8. [Google Scholar]
- Cheong, E.J.; Pooler, M.R. Factors affecting somatic embryogenesis in Prunus incisa cv. February Pink. Plant Cell Rep. 2004, 22, 810–815. [Google Scholar] [CrossRef] [PubMed]
- Montalbán, I.A.; García-Mendiguren, O.; Goicoa, T.; Ugarte, M.D.; Moncaleán, P. Cold storage of initial plant material affects positively somatic embryogenesis in Pinus radiata. New For. 2015, 46, 309–317. [Google Scholar] [CrossRef]
- Corredoira, E.; Ballester, A.; Ibarra, M.; Vieitez, A.M. Induction of somatic embryogenesis in explants of shoot cultures established from adult Eucalyptus globulus and E. saligna x E. maidenii trees. Tree Physiol. 2015, 35, 678–690. [Google Scholar] [CrossRef]
- Rugini, E. In vitro propagation of some olive (Olea europaea sativa L.) cultivars with different root-ability, and medium development using analytical data from developing shoots and embryos. Sci. Hortic. 1984, 24, 123–134. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A Revised Medium for Rapid Growth and Bio Assays with Tobacco Tissue Cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Pérez-Barranco, G.; Mercado, J.A.; Pliego-Alfaro, F.; Sánchez-Romero, C. Genetic Transformation of Olive Somatic Embryos Through Biolistics. Acta Hortic. 2007, 473–477. [Google Scholar] [CrossRef]
- Pérez-Barranco, G.; Torreblanca, R.; Padilla, I.M.G.; Sánchez-Romero, C.; Pliego-Alfaro, F.; Mercado, J.A. Studies on genetic transformation of olive (Olea europaea L.) somatic embryos: I. Evaluation of different aminoglycoside antibiotics for nptII selection; II. Transient transformation via particle bombardment. Plant Cell Tissue Organ Cult. 2009, 97, 243–251. [Google Scholar] [CrossRef]
- Velada, I.; Grzebelus, D.; Lousa, D.; Soares, C.M.; Santos Macedo, E.; Peixe, A.; Arnholdt-Schmitt, B.; Cardoso, G.H. AOX1-Subfamily Gene Members in Olea europaea cv. “Galega Vulgar”—Gene Characterization and Expression of Transcripts during IBA-Induced in Vitro Adventitious Rooting. Int. J. Mol. Sci. 2018, 19, 597. [Google Scholar] [CrossRef] [PubMed]
- Velada, I.; Cardoso, H.; Porfirio, S.; Peixe, A. Expression Profile of PIN-Formed Auxin Efflux Carrier Genes during IBA-Induced In Vitro Adventitious Rooting in Olea europaea L. Plants 2020, 9, 185. [Google Scholar] [CrossRef]
- Orinos, T.; Mitrakos, K. Rhizogenesis and somatic embryogenesis in calli from wild olive (Olea europaea var. sylvestris (Miller) Lehr) mature zygotic embryos. Plant Cell Tissue Organ Cult. 1991, 27, 183–187. [Google Scholar] [CrossRef]
- Rugini, E.; Silvestri, C. Somatic Embryogenesis in Olive (Olea europaea L. subsp. europaea var. sativa and var. sylvestris). In In vitro Embryogenesis in Higher Plants, Methods in Molecular Biology; Maria, A.G., Maurizio, L., Eds.; Springer: New York, NY, USA, 2016; pp. 341–349. [Google Scholar] [CrossRef]
- Mouaad Amine Mazri Embryogenic competence of calli and embryos regeneration from various explants of Dahbia cv, a Moroccan olive tree (Olea europaea L.). Afr. J. Biotechnol. 2011, 10. [CrossRef]
- D’Onofrio, C.; Morini, S.; Bellocchi, G. Effect of light quality on somatic embryogenesis of quince leaves. Plant Cell Tissue Organ Cult. 1998, 53, 91–98. [Google Scholar] [CrossRef]
- Corredoira, E. Proliferation, Maturation and Germination of Castanea sativa Mill. Somatic Embryos Originated from Leaf Explants. Ann. Bot. 2003, 92, 129–136. [Google Scholar] [CrossRef]
- De-la-Peña, C.; Galaz-Ávalos, R.M.; Loyola-Vargas, V.M. Possible Role of Light and Polyamines in the Onset of Somatic Embryogenesis of Coffea canephora. Mol. Biotechnol. 2008, 39, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Hernández, I.; Cuenca, B.; Carneros, E.; Alonso-Blázquez, N.; Ruiz, M.; Celestino, C.; Ocaña, L.; Alegre, J.; Toribio, M. Application of Plant Regeneration of Selected Cork Oak Trees by Somatic Embryogenesis to Implement Multivarietal Forestry for Cork Production. Tree For. Sci. Biotechnol. 2011, 5, 19–26. [Google Scholar]
- Park, S.-Y.; Yeung, E.C.; Paek, K.-Y. Endoreduplication in Phalaenopsis is affected by light quality from light-emitting diodes during somatic embryogenesis. Plant Biotechnol. Rep. 2010, 4, 303–309. [Google Scholar] [CrossRef]
- Ramakrishna, D.; Shasthree, T. High efficient somatic embryogenesis development from leaf cultures of Citrullus colocynthis (L.) Schrad for generating true type clones. Physiol. Mol. Biol. Plants 2016, 22, 279–285. [Google Scholar] [CrossRef]
- Ali, M.; Mujib, A.; Tonk, D.; Zafar, N. Plant regeneration through somatic embryogenesis and genome size analysis of Coriandrum sativum L. Protoplasma 2017, 254, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Frederico, A.M.; Campos, M.D.; Cardoso, H.G.; Imani, J.; Arnholdt-Schmitt, B. Alternative oxidase involvement in Daucus carota somatic embryogenesis. Physiol. Plant. 2009, 137, 498–508. [Google Scholar] [CrossRef] [PubMed]
- Bhojwani, S.S.; Razdan, M.K. Plant Tissue Culture: Theory and Practice, a Revised Edition; (Studies in Plant Science, Vol. 5); Elsevier: Amsterdam, The Netherlands, 1996; ISBN 978-0-444-81623-8. [Google Scholar]
- Bradaï, F.; Pliego-Alfaro, F.; Sánchez-Romero, C. Long-term somatic embryogenesis in olive (Olea europaea L.): Influence on regeneration capability and quality of regenerated plants. Sci. Hortic. 2016, 199, 23–31. [Google Scholar] [CrossRef]
- Leva, A.R.; Petruccelli, R.; Rinaldi, L.M.R. Somaclonal Variation in Tissue Culture: A Case Study with Olive. In Recent advances in plant in vitro culture; Leva, A.R., Rinaldi, L.M.R., Eds.; INTECH Open Access Publisher: Rijeka, Croatia, 2012; pp. 123–150. ISBN 978-953-51-0787-3. [Google Scholar]
- Bradaï, F.; Pliego-Alfaro, F.; Sánchez-Romero, C. Somaclonal variation in olive (Olea europaea L.) plants regenerated via somatic embryogenesis: Influence of genotype and culture age on phenotypic stability. Sci. Hortic. 2016, 213, 208–215. [Google Scholar] [CrossRef]
- Brar, D.S.; Jain, S.M. Somac1onal Variation: Mechanism and Applications in Crop Improvement. In Somaclonal variation and induced mutations in crop improvement; Jain, S.M., Brar, B.S., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1998; pp. 15–37. [Google Scholar]
- Bradaï, F.; Sánchez-Romero, C.; Martín, C. Somaclonal variation in olive (Olea europaea L.) plants regenerated via somatic embryogenesis: Influence of genotype and culture age on genetic stability. Sci. Hortic. 2019, 251, 260–266. [Google Scholar] [CrossRef]
- Benelli, C.; Fabbri, A.; Grassi, S.; Lambardi, M.; Rugini, E. Histology of somatic embryogenesis in mature tissues of olive (Olea europaea L.). J. Hortic. Sci. Biotechnol. 2001, 76, 112–119. [Google Scholar] [CrossRef]
- Junaid, A.; Mujib, A.; Bhat, M.A.; Sharma, M.P. Somatic embryo proliferation, maturation and germination in Catharanthus roseus. Plant Cell Tissue Organ Cult. 2006, 84, 325–332. [Google Scholar] [CrossRef]
- Toufik, I.; Belkoura, I.; Guenoun, F. Improving germination conditions of somatic embryos derived from radicle explants of Moroccan olive cultivar Dahbia. J. Biotechnol. Biosci. 2017, 5, 51–56. [Google Scholar]
- Rugini, E.; Mencuccini, M.; Biasi, R.; Altamura, M.M. Olive (Olea Europaea L.). In Protocol for Somatic Embryogenesis in Woody Plants; Jain, S.M., Gupta, P.K., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 245–360. [Google Scholar]
- Benzekri, H.; Sánchez-Romero, C. Maturation of Olive Somatic Embryos. Acta Hortic. 2012, 441–447. [Google Scholar] [CrossRef]
- Guzmám-Garcia, E. Avocado Somatic Embryogenesis: Maturation and Germination of Somatic Embryos, Characterization and Cryopreservation of Embryogenic Cultures. Ph.D. Thesis, University of Cordoba, Cordoba, Spain, 2012. [Google Scholar]
- Cañas, L.A.; Benbadis, A. In vitro plant regeneration from cotyledon fragments of the olive tree (Olea europaea L.). Plant Sci. 1988, 54, 65–74. [Google Scholar] [CrossRef]
- Torreblanca, R.; Cerezo, S.; Palomo-Ríos, E.; Mercado, J.A.; Pliego-Alfaro, F. Development of a high throughput system for genetic transformation of olive (Olea europaea L.) plants. Plant Cell Tissue Organ Cult. 2010, 103, 61–69. [Google Scholar] [CrossRef]



| Variables | Sum of Squares | Degrees of freedom | Medium Square | F | p |
|---|---|---|---|---|---|
| Explant type | 619 | 2 | 310 | 6.23 | 0.002 |
| Photoperiod | 15 | 1 | 15 | 0.30 | 0.583 |
| Explant type × Photoperiod | 49 | 2 | 25 | 0.49 | 0.619 |
| ERROR | 8689 | 175 | 50 |
| 0 h | 16 h | |||||
|---|---|---|---|---|---|---|
| Radicle | Proximal | Distal | Radicle | Proximal | Distal | |
| Number of explants in culture | 97 | 259 | 132 | 127 | 135 | 92 |
| Embryogenic calli (%) | 17 b | 16 b | 12 c | 22 a | 7 d | 16 b |
| Average number of embryos per calli | 1.66 b | 1.25 b | 1.55 b | 2.54 a | 1.50 b | 1.55 b |
| Subculture I | Subculture II | |||
|---|---|---|---|---|
| 0 h | 16 h | 0 h | 16 h | |
| Number of explants in culture | 70 | 74 | 75 | 83 |
| Embryogenic calli (%) | 29 d | 50 b | 44 c | 63 a |
| Average number of embryos per calli | 2.7 c | 7.5 b | 3.95 c | 13.6 a |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pires, R.; Cardoso, H.; Ribeiro, A.; Peixe, A.; Cordeiro, A. Somatic Embryogenesis from Mature Embryos of Olea europaea L. cv. ‘Galega Vulgar’ and Long-Term Management of Calli Morphogenic Capacity. Plants 2020, 9, 758. https://doi.org/10.3390/plants9060758
Pires R, Cardoso H, Ribeiro A, Peixe A, Cordeiro A. Somatic Embryogenesis from Mature Embryos of Olea europaea L. cv. ‘Galega Vulgar’ and Long-Term Management of Calli Morphogenic Capacity. Plants. 2020; 9(6):758. https://doi.org/10.3390/plants9060758
Chicago/Turabian StylePires, Rita, Hélia Cardoso, Augusto Ribeiro, Augusto Peixe, and António Cordeiro. 2020. "Somatic Embryogenesis from Mature Embryos of Olea europaea L. cv. ‘Galega Vulgar’ and Long-Term Management of Calli Morphogenic Capacity" Plants 9, no. 6: 758. https://doi.org/10.3390/plants9060758
APA StylePires, R., Cardoso, H., Ribeiro, A., Peixe, A., & Cordeiro, A. (2020). Somatic Embryogenesis from Mature Embryos of Olea europaea L. cv. ‘Galega Vulgar’ and Long-Term Management of Calli Morphogenic Capacity. Plants, 9(6), 758. https://doi.org/10.3390/plants9060758







