1. Introduction
Potato (
Solanum tuberosum L.) is a valuable food crop with great importance in ensuring food security worldwide [
1]. One of the most acute problems in modern agriculture and food industries is the loss of potato tubers (about 50–60% of the total harvest) during storage from diseases [
2]. Fusarium dry rot of potato that is caused by
Fusarium spp. is a major devastating disease that causes postharvest rotting in storage and seed tubers decay [
3,
4]. About thirteen different
Fusarium spp. have been identified as agents that are responsible for potato dry rot worldwide. Among them, the most common species are
F. oxysporum,
F. solani,
F. sambucinum,
F. avenaceum,
F. culmorum,
F. acuminatum,
F. equiseti, and
F. crookwellense [
4,
5]. Pathogens often infect plants and live in their tissues without any typical disease symptoms at growing time and are not always visible at the time of harvest, but may rapidly appear after harvesting and become the major decay factor. Initially, lesions appear as brown or black spots on the surface of the tuber. Lesions later form large, hollow cavities. Often, the affected areas appear wrinkled on the surface of the tuber with numerous white bundles of mycelium. Losses from
Fusarium-associated dry rot has been estimated to reduce up to 25% crop establishment by affecting the development of potato sprouts and more than 60% of tubers can be infected during long-term storage [
3,
5]. Moreover,
Fusarium spp. might contaminate food products with mycotoxins (of which the most common groups are trichothecenes, zearalenones, fumonisins, enniatins, moniliformin, and beauvericin, etc.), which threatens animal health and can cause a wide range of infections of humans [
6,
7,
8]. Potatoes may be infected by pathogens any time during the growing season or through wounds inflicted during harvesting, sorting, transportation, and handling for storage [
9]. Traditionally applied chemicals for control of postharvest decay are hazardous to human, animal, and environmental health due to the toxicological risk, and its application is prohibited in many countries [
10]. Environmentally friendly and safe approaches, which might induce the natural defense mechanisms of the plant organisms against diseases, are important alternative strategies [
2,
9,
10]. The most effective approaches to control dry rot are the utilization of resistant potato cultivars, although better handling of potatoes at harvest and during postharvest operations would sensibly reduce the impact of such diseases as well [
2,
9].
Beneficial antagonistic bacteria
Bacillus subtilis (with a well-known role in plant growth promotion and anti-stress physiological programs induction), generally recognized as safe microorganisms (GRAS) to use in the food industry, are considered a bio-active and eco-friendly agent for controlling postharvest decay [
9,
10,
11]. To date, numerous examples of successful
B. subtilis application in the control of various postharvest-emerged pathogens of different fruits/vegetables during handling, transportation, and storage have been described in the literature [
9,
12,
13,
14]. Their mechanisms of action are still largely unclear; however, it is suggested that they include competition for space/nutrients with pathogens, production of various bio-active substances with antibiotic activity and cell wall-degrading compounds, and the induction of systemic resistance in whole host-plant organisms [
9,
11,
15]. These features, together with the sporulation ability of
B. subtilis, make them ideal for the development of commercial bioproducts [
9]. With that,
Bacillus efficiency might depend on various factors, including strain characteristics (epiphytes or endophytes), application methods (before or after harvest/storage), type of pathogens/hosts, etc. [
9,
16]. Additionally, there is interest in the co-application of
B. subtilis alongside other biological agents in an integrated vision of disease management due to the lack of individual strains with broad spectrum activity against a wide range of pathogens. Salicylic acid (SA) is a safe signaling molecule that can be used for the development of complex bioproducts with wide spectrums of action in both preharvest and postharvest strategies, while having high commercial potential for enhancing nutritional quality along with the extension of the shelf-life of fruits/vegetables, including potato [
9]. Numerous studies have indicated the potential of SA postharvest application for reducing chilling injury and decay, delaying ripening, and enhancing the health benefits of fruit and vegetable consumption by increasing antioxidant capacity [
17]. However, there is little information regarding the influence of the joint application of endophytic
B. subtilis with SA on plant growth, development, and postharvest physiology of healthy and pathogens-infected potatoes during long-time storage [
16,
18,
19]. In our recent work, we generated data about endophytic
B. subtilis (strains 10-4, 26D), alone or in compositions with SA, increasing the resistance of potato tubers to
Phytophthora infestans and
Fusarium oxysporum during long-time storage, which was accompanied with prolonging shelf-life of potatoes and preserving their appearance [
16]. The findings suggest
B. subtilis decrease as disease incidence in potato due to colonizing internal tissues and protect cells inside against pathogen development and stress-induced cell damages, therewith, controlling aging processes [
16]. However, the mechanisms underlying
B. subtilis actions both alone and in compositions with SA on potato under postharvest pathogen’s infection are largely unknown and they require further detailed investigations to fully realize their potential in agricultural/food industries.
The main mode of action driving the antifungal effect of
B. subtilis might be due to the disruption of the cellular membrane structure of pathogens by hydrolases, capable of destroying the structural polysaccharides of the fungal cell wall and lysing the hyphae of fungi [
9,
20]. A correlation between antagonistic activity to various pathogenic fungi and the synthesis of hydrolases, such as amylases (AMY) and proteases (PRO), by a number of bacteria has been established [
9,
21]. The protective response of plants is an increase in the content of hydrolase inhibitors, which make a significant contribution to the regulation of the activity of hydrolytic enzymes by suppressing the activity of their own native enzymes, and those from pathogenic fungi and bacteria [
20,
22,
23]. Previously, on model plants of potato and sugar beet, we showed that
B. subtilis-based bioproducts promoted PRO inhibitors synthesis and protected growing plants from the penetration/development of pathogenic microorganisms [
18,
24,
25]. With that, it is an interest in the character of changing in hydrolytic enzymes and their inhibitors in harvested potato tubers that are treated with
B. subtilis upon pathogen infection during long-term storage.
As many other stresses, pathogens attack leads to the synthesis of a range of defense compounds in plant cells to protect them from damages [
26,
27,
28]. As an important biochemical marker for the formation of plants resistance can be the accumulation of malondialdehyde (MDA) (the final product of stress-caused lipid peroxidation) and a multifunctional stress metabolite proline, which acts as an antioxidant, osmolyte [
29], and a low molecular weight chaperone [
23], which is involved in maintaining the native structure of enzymes [
29,
30]. Many studies reported an increase in proline content in plants in response to different stresses and its importance for plant survival in extreme situations [
29,
30]. The formation of potatoes resistance can also be assayed by the changes in reducing sugars (RS), total dry matter (TDM), and starch contents in healthy and pathogen-infected stored tubers [
31,
32], which also largely determine the consumer and table qualities of potatoes and uses to judge the best further options for products processing [
31,
33]. An increase in the content of RS and starch decomposition into sugar in potato tubers during storage negatively affects the culinary qualities (during heat treatment, the flesh darkens, the taste worsens, and becomes sweet-starchy-viscous) [
31,
33]. Tubers with higher TDM and starch concentration exhibited the highest resistance to mechanical impacts [
34]. Under unfavorable storage conditions (i.e. pathogen attack), normal metabolic pathways change in potato, which leads to the synthesis of abnormal amounts of numerous novel compounds and secondary metabolites, including glycoalkaloids (GA) (α-Solanine and α-Chaconine are 95% of the total GA), which is believed are associated with antimicrobial, antifungal activities [
35,
36], and play an important role in the disease resistance of tubers [
37]. With that, elevated GA levels in potatoes (200 mg/kg of tubers are considered as critical) are toxic compounds that pose a serious potential hazard to humans [
38,
39]. The highest accumulation of toxic GA levels in potatoes typically occurs in the storage period. However, the correlation between the content of GA in potatoes during storage and under conditions of infection by
F. oxysporum, and the use of endophytic bacteria
B. subtilis and
B. subtilis + SA in the available literature has not been established at the moment of the beginning of our work.
In this work, we investigated the effect of endophytic B. subtilis (strains 10-4, 26D) and compositions of B. subtilis (10-4, 26D) with SA on the development of Fusarium dry rot, the activity of hydrolytic enzymes and their inhibitors, the level of lipid peroxidation and proline content (as markers of oxidative and osmotic stresses development, respectively), reducing sugars, starch, total dry matter, and glycoalkaloids accumulation in healthy (non-infected) and F. oxysporum-infected potato tubers after long-term storage.
3. Discussion
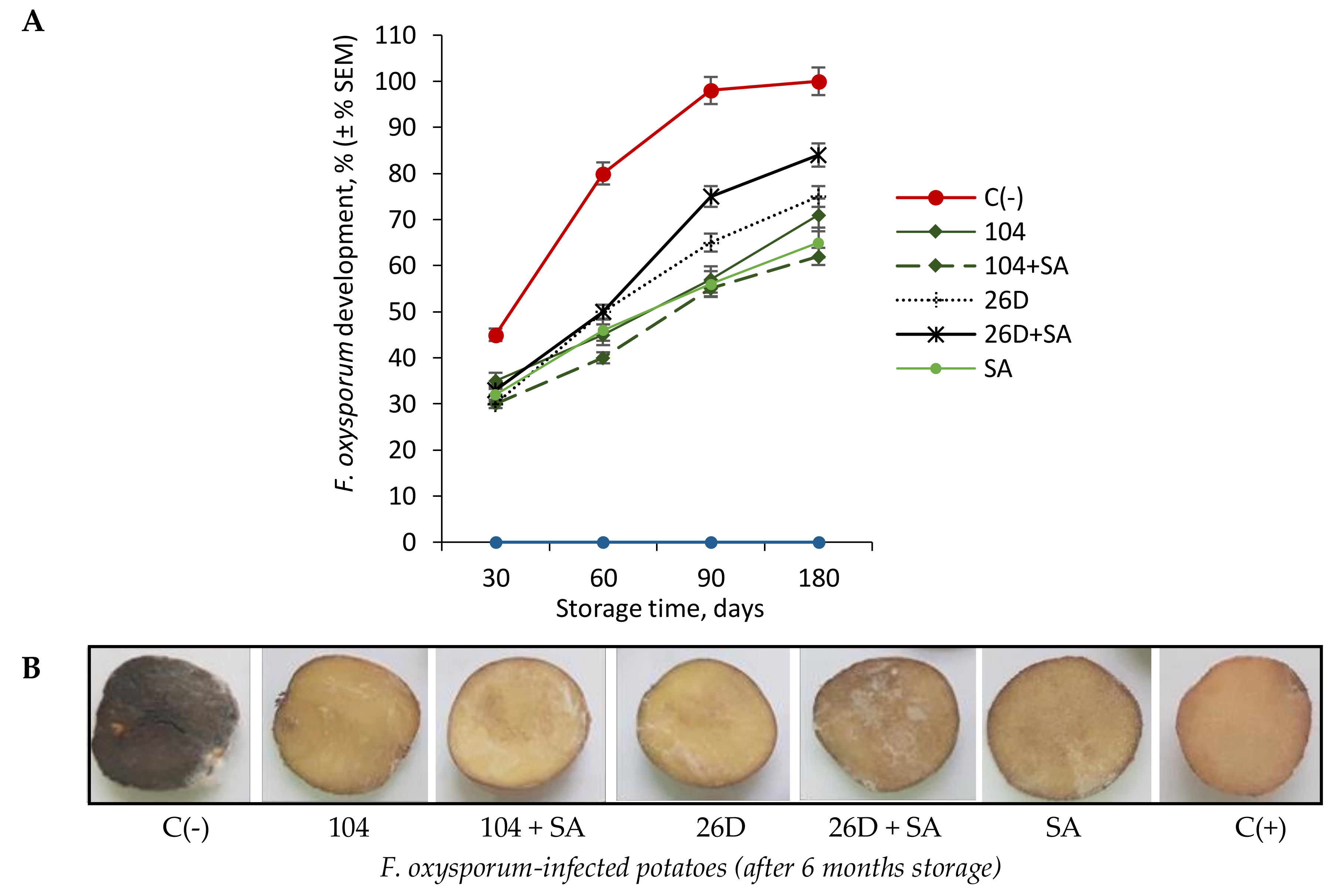
The infection of tubers with phytopathogenic fungi
F. oxysporum resulted in typical disease symptom development, manifested as the form of gray (brown) putrefied depressed spots with a white coating, under which the flesh became dry, rotten, the skin wrinkled, and white spores of the fungus appeared on it, which can dissipate and infect neighboring tubers. This becomes visible after 6 months of storage at a temperature of 4 °C, at which point tubers are completely infected (
Figure 1A,B). The bacterization with
B. subtilis 10-4 and 26D reduced the intensity of fusarium disease development, manifested as a decrease in lesion area or even in their absence (
Figure 1A,B). Among the tested treatments, the composition of
B. subtilis 10-4 + SA most effectively decreased the incidence of dry rot caused by
F. oxysporum in stored potatoes (
Figure 1A,B). Moreover, by the sixth months of storage following fusarium infection, tubers that were treated with 10-4 + SA looked healthier and fresher, while, in variants with
B. subtilis 26D + SA treatment, the picture was different, and traces of damage were became visible on the tubers (
Figure 1B). It can be assumed that the mechanisms of action differ between strains 10-4 and 26D and that in the compositions with SA, the protective effect against
Fusarium is enhanced in one case (strain 10-4 + SA), and not the other (strain 26D + SA) (
Figure 1A,B). This finding allows for the possibility that this effect might be associated with the ability of the strains to produce their own phytohormones [
13] and activate different pathways that are responsible for suppressing pathogen development, inhibiting the aging process and prolonging shelf-life of stored products. It is likely that, if strain 10-4 produces SA, then the addition of exogenous SA can enhance its protective effect; if strain 26D produces jasmonic acid (JA), the addition of SA to the composition might have the opposite effect, since JA and SA are antagonists. This assumption, of course, requires further detailed study and confirmation.
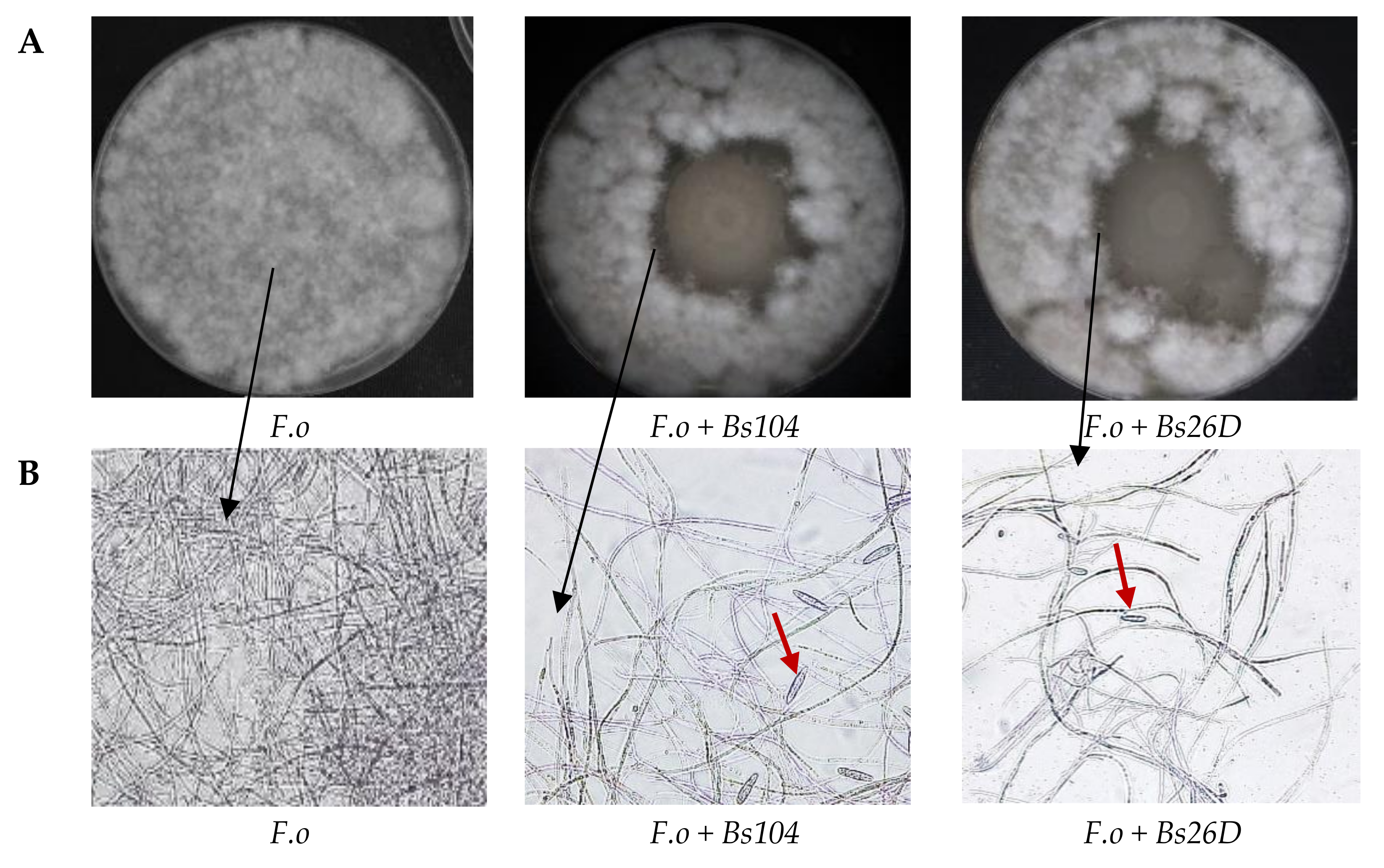
The main mode of action driving the antifungal effect of
B. subtilis might be due to the disruption of the cellular membrane structure of pathogens [
9]. Our in vitro experiments showed that
B. subtilis 10-4 and 26D have antagonistic activity against the phytopathogenic fungus
F. oxysporum (
Figure 2A). The microscopic observation of the
F. oxysporum fungal mycelia clearly revealed morphological variations. The structure of the
F. oxysporum mycelia was well organized in the absence of the bacterial culture medium (
Figure 2B), while numerous gaps of mycelia appeared and macroconidia were produced in the presence of the culture medium of 10-4 and 26D (
Figure 2B). The observed phenomenon of macroconidia production by
F. oxysporum in the presence of 10-4 and 26D (
Figure 2B), obviously, connected with the survival strategy of the pathogen and indicates that they are under adverse conditions and produce macroconidia for species preservation. Demonstrated suppression of
F. oxysporum development in vivo (
Figure 1) and in vitro (
Figure 2) by
B. subtilis 10-4 and 26D might also be connected with the ability of these bacteria to produce compounds with antifungal activity, such as cell wall degrading enzymes, lipopeptides, and hormone-like compounds [
9]. Accordingly, it has been shown that
B. subtilis might produce lipopeptides that are characterized by strong antifungal activity [
9,
11,
15,
40,
41]. It might be proposed that the observed
in vivo antifungal effect of
B. subtilis against
F. oxysporum has its origin in the destabilization of the plasma membrane’s physiological function and that the initial targets could be the functional components, such as the H + -ATPase, which affects fungal integrity. The antifungal effects of
B. subtilis (10-4, 26D) treatment may also promote the maintenance of firmness, because it protects the potato tubers against fungal physiology, which can involve the secretion of cell wall-degrading enzymes. Overall, detailed investigations both of the spectrum of metabolites produced by 10-4 and 26D and changes in physiological, biochemical, and molecular defense responses in cells of stored potatoes for understanding the mechanisms underlying interaction in systems «endophytic
B. subtilis—host plants—pathogens» will be quite interesting.
Significant contribution in the bio-control of pathogens is competition for space and nutrients [
9]. Bacterial inoculants take up nutrients faster than pathogens; this can lead to the inhibition of germination of pathogen spores at the wound site [
9]. A fundamental strategy for nutrient competition might occur via the direct attachment of antagonistic microorganism to pathogen hyphae or via alternative mechanisms, such as the production of a wide range of biologically active molecules, such as antibiotics, biosurfactants, siderophores, hydrogen cyanide, and hydrolases increase their advantage against pathogens, as they compete for a suitable niche for colonization [
9]. The possible mechanisms of the antagonistic function of
B. subtilis can be attributed to the synthesis of extracellular hydrolases that are capable of destroying the structural polysaccharides of the fungal cell wall and lysing the hyphae of fungi [
9,
20,
21]. A correlation between antagonistic activity to various pathogenic fungi and the synthesis of hydrolases such as proteases, cellulases, xylanases, mannanases, and lipases by several bacteria has been established [
9,
22]. For example, among mucolytic enzymes of
B. subtilis strain 739, the most contribution to the lysis of the native mycelium of phytopathogenic fungi
F. culmorum,
Alternaria alternata,
Bipolaris sorokiniana, and
Rhizoctonia solani showed chitinase, chitosanase, β-1,3-glucanases, and proteases [
22].
B. subtilis APEC170 and
Paenibacillus polymyxa APEC136 diminish the symptoms of anthracnose and white rot in harvested apples by the inhibition of mycelial growth of the pathogens, which was attributed to the increased production of chitinase, amylase, and protease by
B. subtilis APEC170 [
14]. In another study, chitosanases and proteases produced by
B. subtilis V26 enhance harvested tomato resistance (up to 79%) to
Botrytis cinerea-caused postharvest diseases [
9]. Our results showed that the application of
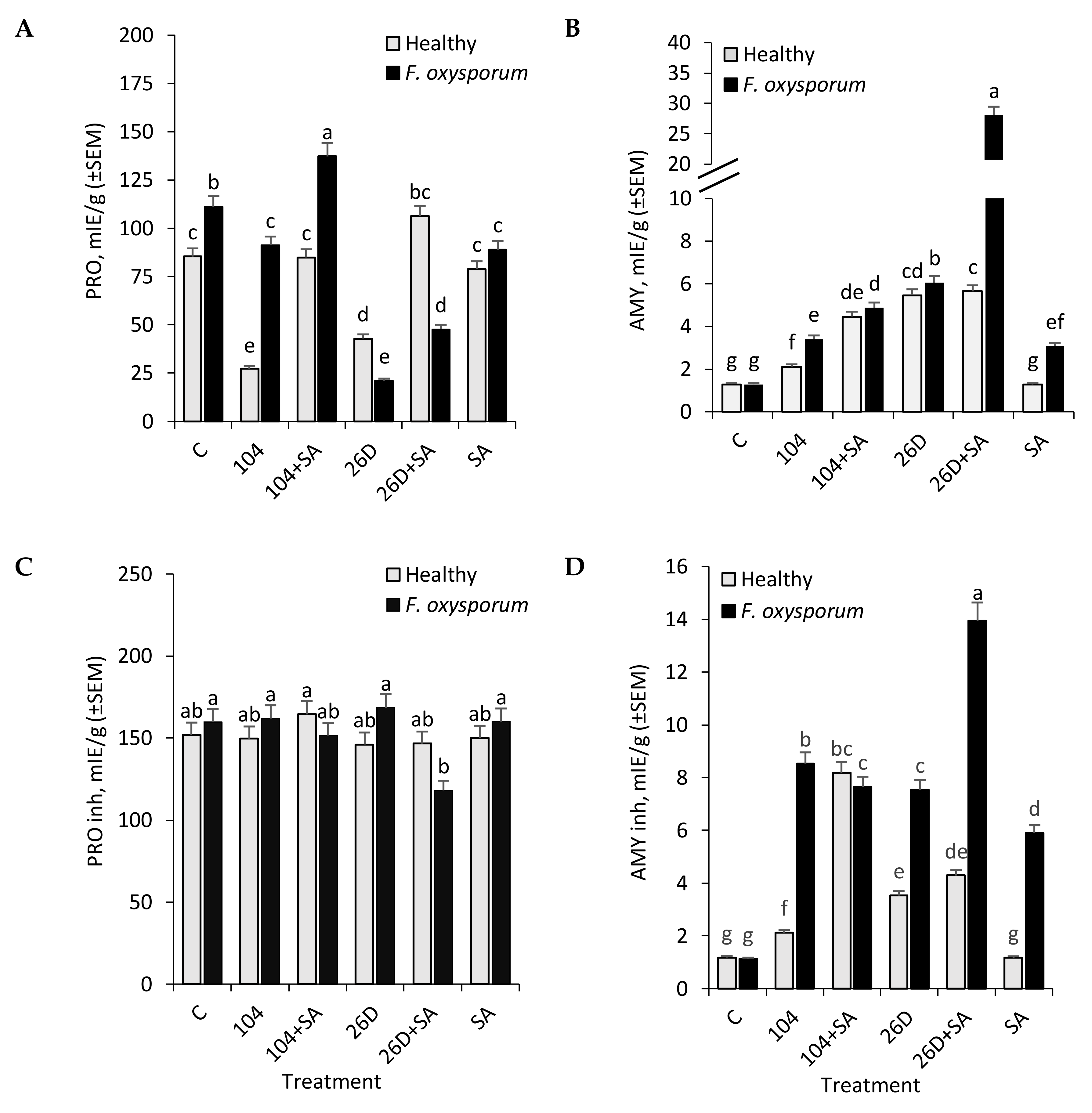
B. subtilis (10-4, 26D) individually and in compositions with SA increased the activity of AMY in stored potato tubers (
Figure 3B). It likely occurs due to bacterial enzymes, rather than potato’s own AMY (in control the activity of AMY is lower), since it is known that the nutrition and development of bacteria require soluble forms of carbohydrates. This might explain the increase in the level of AMY activity during treatment with bacterial preparations. An increase in AMY (during infection) is a factor indicating sensitivity, since starch degradation increases and the pathogen develops more actively on soluble sugars. On the other hand, the activation of AMY can be as a result of the intensification of central metabolism, providing energy and metabolites for the subsequent synthesis of protective compounds (for example, when used only SA, we observe this).
The protective response is an increase in the content of hydrolase inhibitors, which, as a rule, does not occur due to an increase in the concentration of constitutive compounds, but due to the synthesis of new forms of inhibitors [
22,
42]. Plant protein inhibitors make a significant contribution to the regulation of the activity of hydrolytic enzymes by suppressing the activity of their own native enzymes, and those from pathogenic fungi and bacteria [
22,
23]. Earlier, on model plants of potato and sugar beet, we showed that the introduction of biological products that are based on B. subtilis promotes the synthesis of PRO inhibitors and protects growing plants from the penetration and development of pathogenic microorganisms [
18,
24,
25]. In our current study,
B. subtilis 10-4 and 26D had practically no effect or the slight decrease in the activity of PRO inhibitors observed in both non-infected (healthy) and
Fusarium-infected stored potato tubers (
Figure 3C). A slight decrease in the activity of PRO inhibitors was observed for
B. subtilis 26D + SA (upon infection with
F. oxysporum), resulting in the plant’s resistance to the action of pathogen enzymes and their distribution in tissues affecting the appearance of tubers (
Figure 1B). With that, in response to treatment with
B. subtilis strains 10-4, 26D, and their compositions with SA, there was a significant increase in the activity of AMY inhibitors in both healthy and
F. oxysporum-infected tubers during storage (
Figure 3D). In tubers that were infected by
F. oxysporum, there was no significant difference from non-infected control in AMY inhibitors activity, while in tubers treated with
B. subtilis (10-4, 26D) and their compositions with SA
F. oxysporum infection correlated with an increase in activity of AMY inhibitors (
Figure 3D). These findings indicate that
B. subtilis (10-4, 26D), alone and in compositions with SA, activates the synthesis of AMY inhibitors, with the highest activation being observed when
B. subtilis 26D was used in composition with SA. The activity of AMY inhibitors is a resistance factor since it is a regulator of the degree of starch hydrolysis. An increase in the level of inhibitory activity (inhibitors of the commercial
B. subtilis preparation) is explained by an increase in the expression of
B. subtilis amylase inhibitors, which is probably due to the active colonization and development of endophytes into tubers (acting as their number (quantitate amount) regulator). In our previous work, we demonstrated the ability of
B. subtilis intensively to colonize internal tissues of tubers and the ability of SA to enhance this process [
16].
One of the earliest responses of plant cells to infection by a pathogen is an oxidative burst—the formation of reactive oxygen species (ROS) (H
2O
2, superoxide radicals, etc.), resulting in protective reactions of the plant, including the synthesis of the whole spectrum of protective compounds [
24,
26,
27,
28]. In an overall picture, the development of plant protective reactions against pathogen-induced oxidative burst correlates with the degree of accumulation of the final lipid peroxidation product—malondialdehyde (MDA) acting as a marker for oxidative stress [
29,
30]. As a result of oxidative stress in cells denatured proteins (lipid peroxidation products) accumulate in large quantities that act, not only as primary mediators of stress exposure to the stress factor, but also as inducers of the corresponding protective mechanisms of plant cells. Our experiments showed that
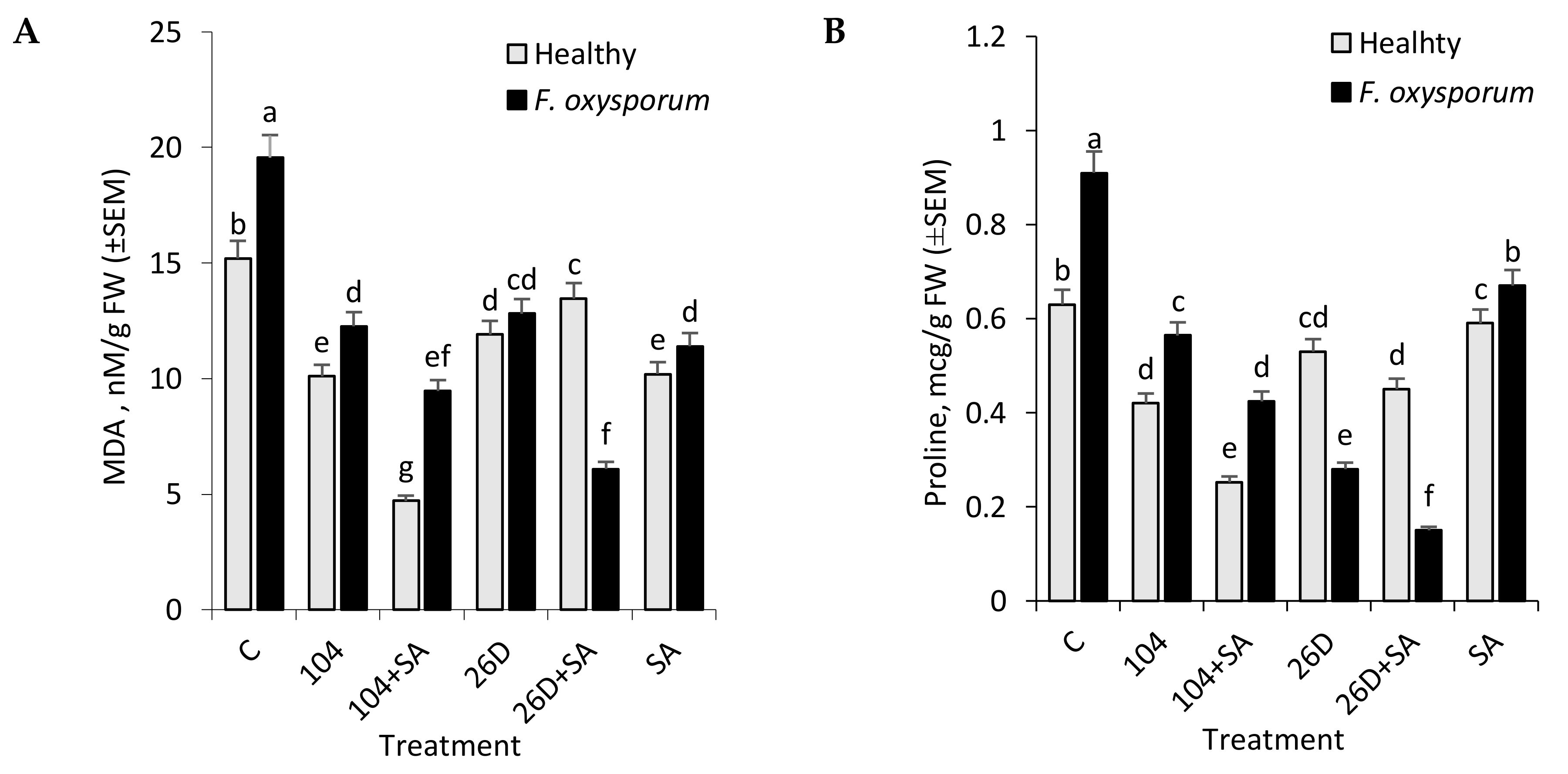
F. oxysporum leads to an increase in MDA level in tubers during long-time storage, while the use of
B. subtilis 10-4 and
B. subtilis 26D, both individually and in compositions with SA, contributed to a reduction of the pathogen-induced increases in lipid peroxidation in stored non-infected (healthy) and infected tubers (
Figure 4A). These findings indicate a reduction of oxidative stress in stored tubers under the treatments before storage with
B. subtilis 10-4,
B. subtilis 10-4 + SA,
B. subtilis 26D and
B. subtilis 26D + SA. These phenomena may be related to the modulation activity of oxidative enzymes under their influence, so that they can control the level of H
2O
2, which induces lipid peroxidation.
An important biochemical marker for the formation of resistance can also be the accumulation of proline, a multifunctional stress metabolite of plants that acts as an antioxidant, osmolyte [
29], and a low molecular weight chaperone [
23], which is involved in maintaining the native structure of enzymes [
29]. Many studies reported an increase in proline content in plants in response to the stresses of various nature and its importance as a factor that is important for plant survival in extreme situations [
29,
30]. Our results show that infection of the potato tubers with
F. oxysporum led to a significant increase in the proline content in the tubers (
Figure 4B). At the same time,
Fusarium-infected tubers that were treated with
B. subtilis 10-4 and 26D, both individually and in compositions with SA, were characterized by a reduced level of pathogen-induced proline accumulation. It should be noted that, under the influence of bacilli in healthy stored tubers, a slight decrease in the amount of proline was also observed, which additionally indicates the important role of this agent in the formation of induced resistance to the causative agent of fusariosis.
Interestingly, based on the results in
Figure 1, SA with 26D has a negative effect when compared with only 26D and other treatments upon
F. oxysporum infection. However, the degree of MDA and proline accumulation is lower than in variants with other treatments. This can be in the case of either blocking the formation of these substances in the plant organism or digesting data compounds with pathocomplex
F. oxysporum and 26D in the presence of SA. Perhaps, this is the so-called cross-talk in metabolism, when some effectors interfere with the work of others and the level of the protective barrier decreases, while the phytopathogen gets the opportunity to develop. Probably, this might also be connected with the ability of exogenous SA (when applied together with 26D) to induce other pathways that lead to cell strengthening in the initial stages of storage and tubers by six months of storage are characterized by a lower degree of cell damages. Additionally, it might be connected with the ability of 26D produce compounds that is not comparable with SA (for example, through JA production and inducing JA-depending signaling pathways); or perhaps if 26D resulted in the production of endogenous SA in tubers in high concentrations and additional exogenous SA resulted in “feedback mechanism”, when the formation of high levels of this compound (along with the additional introduction of exogenous SA) can lead either to the neutralization of their positive effect or even to a negative effect on disease development. These assumptions, of course, require close attention and further detailed investigations.
A biochemical marker for the formation of resistance can also serve the data about the changes in reducing sugar (RS) content in healthy and pathogen-infected tubers during storage [
31,
32]. The results showed that, in healthy stored potato tubers,
B. subtilis and
B. subtilis + SA decreased the RS content (up to 1.2–2.5 times) in comparison with the control (
Figure 5), which indicated that
B. subtilis also feed on sugars, including those that result from pathogen-induced activation of hydrolytic enzymes that break down starch into sugars. It was suggested that discovered changes in RS in
B. subtilis and SA treated potato tubers upon fusarium infection may be due to the pathogens during the attack secrete hydrolytic enzymes, under the influence of which starch breaks down into sugar in the cells of affected tubers, including reducing starch, which is later used by phytopathogens as one of the most available power supplies. Interestingly, in healthy stored tubers
B. subtilis and
B. subtilis + SA decreased RS content (up to 1.2–2.5 times) in comparison with the control (
Figure 5), which indicated that
B. subtilis also feed on sugars, including those that result from pathogen-induced activation of hydrolytic enzymes that break down starch into sugars. These data indicate that
B. subtilis bacteria effectively compete with pathogens for accessible food sources, in particular sugars, and generally create an unfavorable environment for the further development of pathogens inside tubers, which inevitably leads to the inhibition of their growth and, as a result, a decrease in the incidence of tubers during storage. It is likely endophytic
B. subtilis can themselves destroy starch to sugars and/or feed on existing sugars, including those that result from pathogen-induced activation of hydrolytic enzymes that break down starch to sugars. Moreover,
B. subtilis are capable of producing compounds with antibiotic activity and inhibit the propagation of pathogens, compete for available food sources and, in general, create an unfavorable environment for the further development of pathogens inside tubers [
9], which inevitably leads to the inhibition of their growth and how a consequence of a decrease in the incidence of tubers during storage, and the remaining RS can be spent on food by bacteria. Changes in the content of RS in healthy and
Fusarium-infected tubers during storage can serve not only as an important biochemical marker for the formation of resistance [
32], but can also determine the consumer qualities of stored potatoes [
31]. An increase in the RS content in potato tubers during storage negatively affects the culinary qualities (during heat treatment the flesh darkens, the taste worsens and becomes sweet-starchy-viscous). The findings on a decrease in the content of RS in healthy and
Fusarium-infected tubers under the influence of
B. subtilis and
B. subtilis + SA indicate that they positively affect the consumer qualities of stored tubers by this indicator.
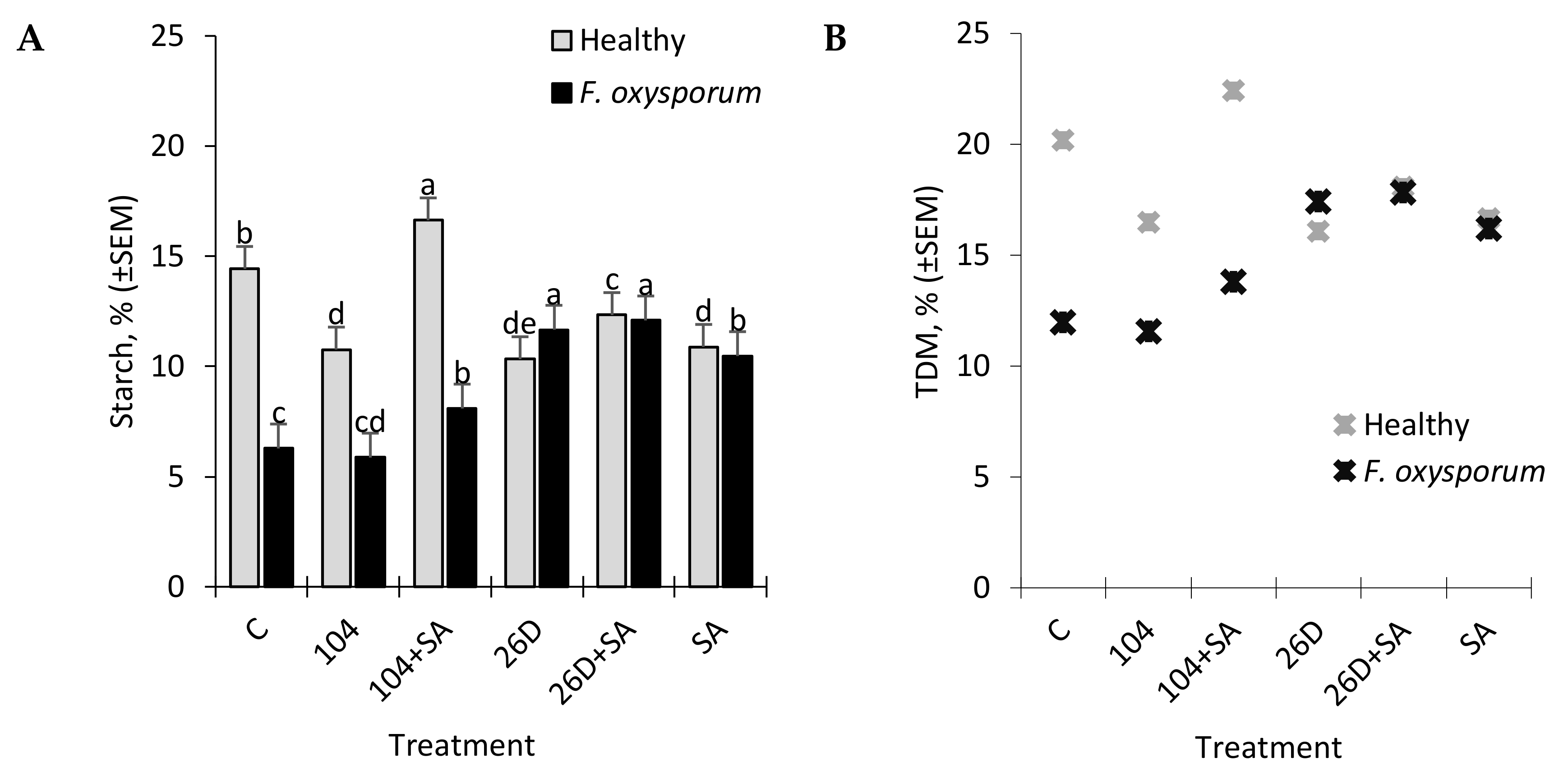
Other important indicators, which largely determine the consumer and table qualities of potatoes, are the content of starch and total dry matter (TDM) in tubers [
31], which can be a judge for the best further options for processing products. The high content of TDM in tubers is a criterion for the low absorption capacity of oil during potato frying [
43]. According to the criterion of starch content, potatoes are divided into 1) starchy (or powdery) (with a high starch content, an average of about 22%), which, when cooked, acquires a granular texture, which is optimal for deep fat, and is also suitable for making mashed potatoes and for baking; 2) wax (potatoes with a lower starch content, on average about 16% and below), which is suitable for cooking and salads, because the tubers maintain the integrity of the fabric. The starch content can vary greatly based on the varietal characteristics of potatoes, the place of growth, climatic conditions, and other factors. At the same time, the process of starch decomposition into sugar during storage, which gives the tubers a sweet taste and negatively affects the taste, is of particular importance. In general, during storage, the decomposition of starch in potato tubers can occur by hydrolytic (with the participation of amylase) and phosphorylithic (with the participation of phosphorylase) pathways. In healthy tubers, amylase activity is often not detectable or is very low. However, the infestation of tubers with phytopathogens and various types of microorganisms, including bacteria, leads to an increase in the activity of hydrolytic enzymes and, accordingly, to an increase in the level of starch decomposition [
44]. At the same time, potato starch granules can be quite resistant to destructive enzymes [
43]. However, as the tubers germinate, the starch granules decompose, which indicates the formation of destructive enzymes into tubers. Some authors reported that
Bacillus circulans capable of breaking potato starch granules [
45] and some bacteria, including
B. subtilis,
B. amyloliquefaciens, and
Microbacterium aurum, are capable of producing AMY [
34,
44,
46,
47]. Storage at 3 °C also increases the activity of AMY in potato tubers, however, an increase can also occur due to the general reaction to stress in tubers, which coincides with cold sweetening, and the enzyme does not directly participate in starch degradation [
44]. Despite current information, the biological process of starch degradation in potato tubers remains unclear [
44,
45] and information on the effect of endophytic
B. subtilis bacteria and their compositions with SA on starch content in healthy and fusarium-infected tubers during long-term storage is not available in the available literature. Our results showed that infestation by
F. oxysporum results in the reduction of starch content in stored potatoes from 14.4% (in non-infected control) to 6.2% (in infected tubers) (
Figure 6A). The application of 26D, 26D + SA, and 10-4 + SA prevented such pathogen-affected starch reduction with maximum positive effects for 26D and 26D + SA. In healthy tubers, 10–4, 26D, and 26D + SA decreased the starch level, while, for 10-4 + SA, a slight increase was observed. In general, these data indicate a positive effect of
B. subtilis and SA on starch safety in tubers that were infected with pathogens during storage. Additionally, the phenomenon of a slight decrease in starch in healthy tubers (not infected with pathogens) under the influence of bacterial treatments might be because
B. subtilis bacteria may use starch as a substrate for nutrition and/or destroy starch due to the production of hydrolytic enzymes. The same pattern of change from
B. subtilis (10-4, 26D) with and without SA were observed for total dry matter (TDM) levels in non-infected (healthy) and
Fusarium-infected tubers (
Figure 6B). In healthy tubers treated with 10-4, 26D, 26D + SA after long-time storage the level of TDM concentration decreased (up to 16.1–18.1%) in comparison with the control, while 10-4 + SA caused an increase (
Figure 6B). The increased levels of starch and TDM percentages in healthy tubers that are treated with
B. subtilis 10-4 + SA (
Figure 5A,B) are most likely to happen due to the trigger of some other additional protective mechanisms by SA in composition with 10-4, while, for the composition 26D + SA the picture was different. Additionally, the phenomenon of a slight decrease in starch in healthy tubers (not infected with pathogens) under the influence of bacterial treatments might be due to the fact that
B. subtilis bacteria use starch as a substrate for nutrition and/or destroy starch due to the production of hydrolytic enzymes. Indeed, it was found that, in healthy stored tubers, under the influence of treatments with the tested strains 10-4, 26D, an increase in AMY activity occurred (
Figure 3B), which is also consistent with the available published data on the ability of a number of non-pathogenic bacteria, including
Bacillus spp., in order to produce AMY [
9,
34,
44,
46,
47]. Earlier, we also demonstrated the ability of the studied bacteria 10-4 and 26D to colonize the internal tissues of both healthy and infected tubers, thereby preventing the spread of the pathogen and reducing the incidence of tubers [
16]. At the same time, in healthy tubers, the highest starch content was found for treatment 10-4 + SA (
Figure 6A). This indicates that the composition 10-4 + SA helps to inhibit the decomposition of starch in tubers during storage. A similar picture was obtained when assessing the changes in the TDM in healthy and
F. oxysporum-infected tubers under the influence of tested treatments (
Figure 6B). Most likely, this nature of the action of strain 10-4 with SA can be a consequence of the fact that SA in compositions with strain 10-4 acts as a trigger of some protective mechanisms, while the picture is different for composition 26D + SA. The revealed difference in the action of the tested strains, both individually and in compositions with SA, can be largely related to the ability of 10-4 and 26D to produce a wide range of protective compounds, including their own phytohormones or other biologically active substances [
9,
11]. Strain 10-4 likely produces SA, and the addition of exogenous SA can enhance its protective effect; if strain 26D produces jasmonic acid (JA), the addition of SA to the composition might have the opposite effect, since JA and SA are antagonists [
9]. This hypothesis, of course, requires additional studies to verify. With that, the results of our analysis allow for us to suggest that
B. subtilis and SA in general impact positive influence in preserving starch and TDM contents in
Fusarium-infected tubers indicating their higher resistance. These findings are consistent with data from the literature regarding tubers, with higher TDM and starch concentrations exhibiting the highest resistance to mechanical impacts [
34]. It might be assumed that the reason for the increased resistance of tubers with a higher concentration of starch and TDM to stresses is associated with a certain cell structure of these tubers; therefore, significant force is required to damage the cell structures.
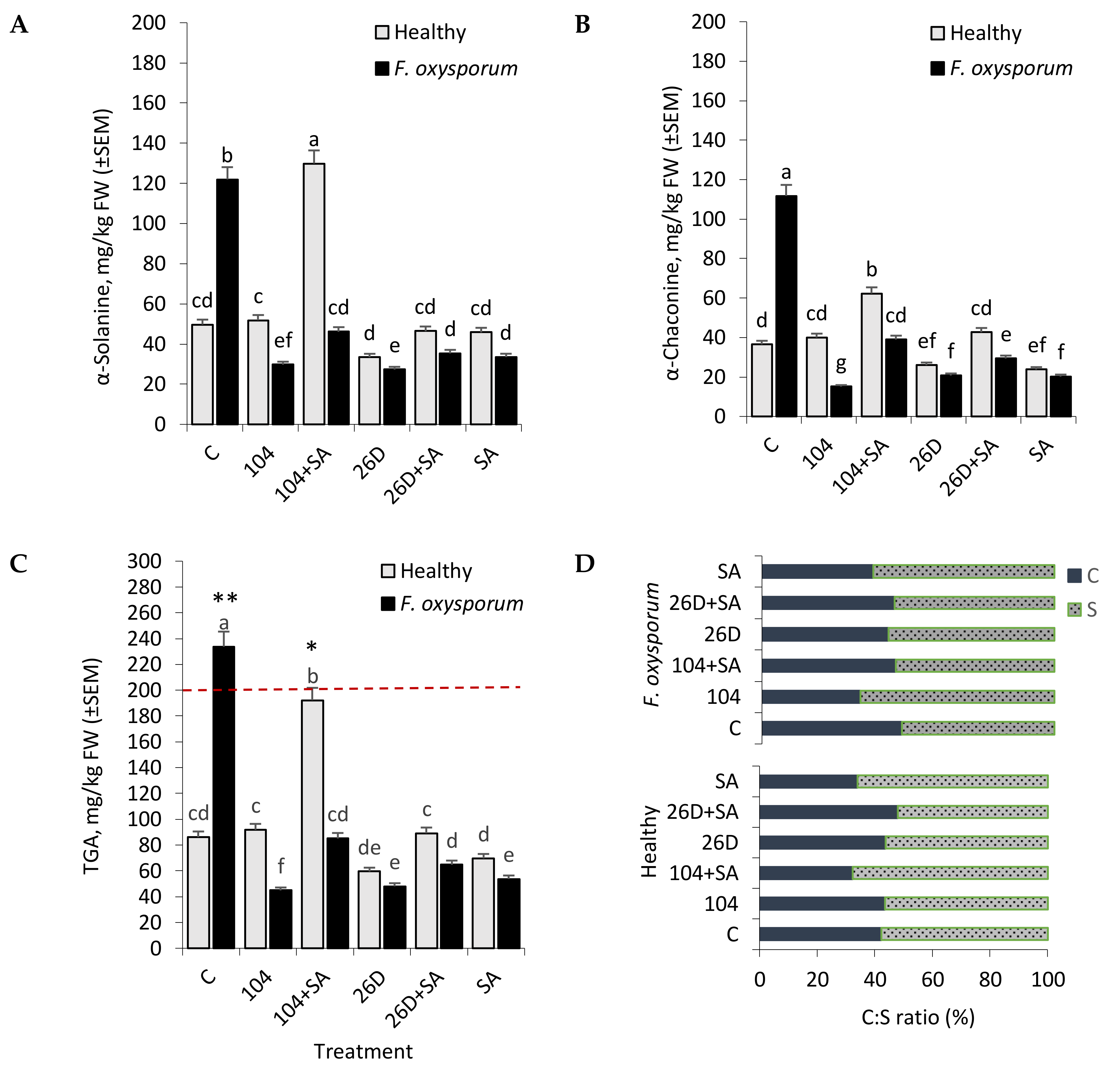
As a result of exposure to different stress factors (light, injuries, microorganisms, extreme temperatures, etc.), normal metabolic pathways change in potato, which lead to the synthesis of abnormal amounts of numerous novel compounds and secondary metabolites, including toxic glycoalkaloids (GA), at levels that exceed the amount found in healthy tissues, also accompanied by the synthesis of other not usually found in the absence of stress [
38]. An important role in the natural disease resistance of tubers is played by GA (in particular, steroid glycosides α-Solanine, and α-Chaconine), being concentrated mainly in their integumentary tissues [
37]. However, GA is also potentially health-threatening toxic compounds in potatoes that pose a serious potential hazard to humans [
38,
39]. The most toxic among GAs of potato are α-Chaconine and α-Solanine, which comprise approximately 95% of the total GA (TGA). Elevated levels of GA (200 mg/kg of tubers are considered to be critical) in potato have a toxic effect on the nervous and gastrointestinal systems of humans [
38]. The literature describes many specific examples of poisoning (sometimes lethal) of people as a result of eating potatoes with a high GA content [
48]. According to some reports, GA content od higher than 100 mg/kg fresh weight (FW) may lead to a bitter flavor in potatoes [
38]. In addition, GA accumulation is associated with the greening of tubers and light-induced processes [
49], but a causal relationship between the two processes has not been established [
50]. The greening of tubers occurs due to the formation of chlorophyll, which is a useful non-toxic indicator that tubers have been exposed to light and should no longer be consumed [
49]. However, GA formation can also occur in the non-green parts of tubers. Therefore, GA formation and the greening of potatoes are physiologically unrelated processes [
51]. The formation of GA can be influenced by many factors, including genotype, growing conditions, transportation, storage, temperature, germination, exposure to light, phytopathogens, herbicides, biostimulants, etc. [
38,
51]. However, the highest accumulation of toxic levels of GA in potatoes typically occurs in the postharvest storage period. Although the mechanisms of action of GA in potatoes are not completely clear, it is believed that they are associated with antimicrobial and antifungal activities [
35,
36]. Several authors have reported an increase in GA content in potato in response to stresses of various nature and its significance as an important factor for plant survival in extreme situations. In general, it is believed that steroidal alkaloids, being secondary metabolites of plants [
52], confer resistance to microbial diseases. However, the correlation between GA content in potato tubers during storage and under conditions of infection by the causative agents of Fusarium dry rot desease, and the use of endophytic bacteria B. subtilis and B. subtilis + SA in the available literature has not been established at the moment of the beginning of our work. Our experiments showed that the infection of tubers with F. oxysporum caused an increase in the level of GA, both α-Solanine and α-Chaconine by the sixth month of storage (
Figure 6A,B). The application of B. subtilis (10-4, 26D), individually and in compositions with SA, significantly decreased such pathogen-induced elevated levels of α-Solanine and α-Chaconine, thereby indicating their involvement in the development of tubers resistance and the positive impact of the studied bacteria on the quality of stored products. Interestingly, in non-infected (healthy) tubers the content of α-Solanine and α-Chaconine had a different profile, depending on the variant of treatment (
Figure 6A,B). Accordingly, in variants with the application of 10-4 and 26D + SA, there was no significant difference from control in the content of α-Solanine and α-Chaconine. In contrast, the application of 10-4 + SA increased (up to 2–2.3 times in comprison with control) both α-Solanine and α-Chaconine contents in healthy tubers. Additionally, the application of 26D resulted in decreased (up to 1.4 times) α-Solanine and α-Chaconine in comparison to the control. These results suggest that B. subtilis, in compositions with and without SA, regulate GA content in stored healthy and Fusarium-infected tubers. However, the joint application of B. subtilis with SA changes GA content (α-Solanine, α-Chaconine) differently in a strain-dependent manner indicating that studied strains (10-4, 26D) use different protective mechanisms, especially when they are applied in combination with SA.