Heterosis Breeding in Eggplant (Solanum melongena L.): Gains and Provocations
Abstract
1. Introduction
2. Origin, Evolution and Domestication
3. Breeding for Eggplant
Wide Hybridization, Embryo Rescue
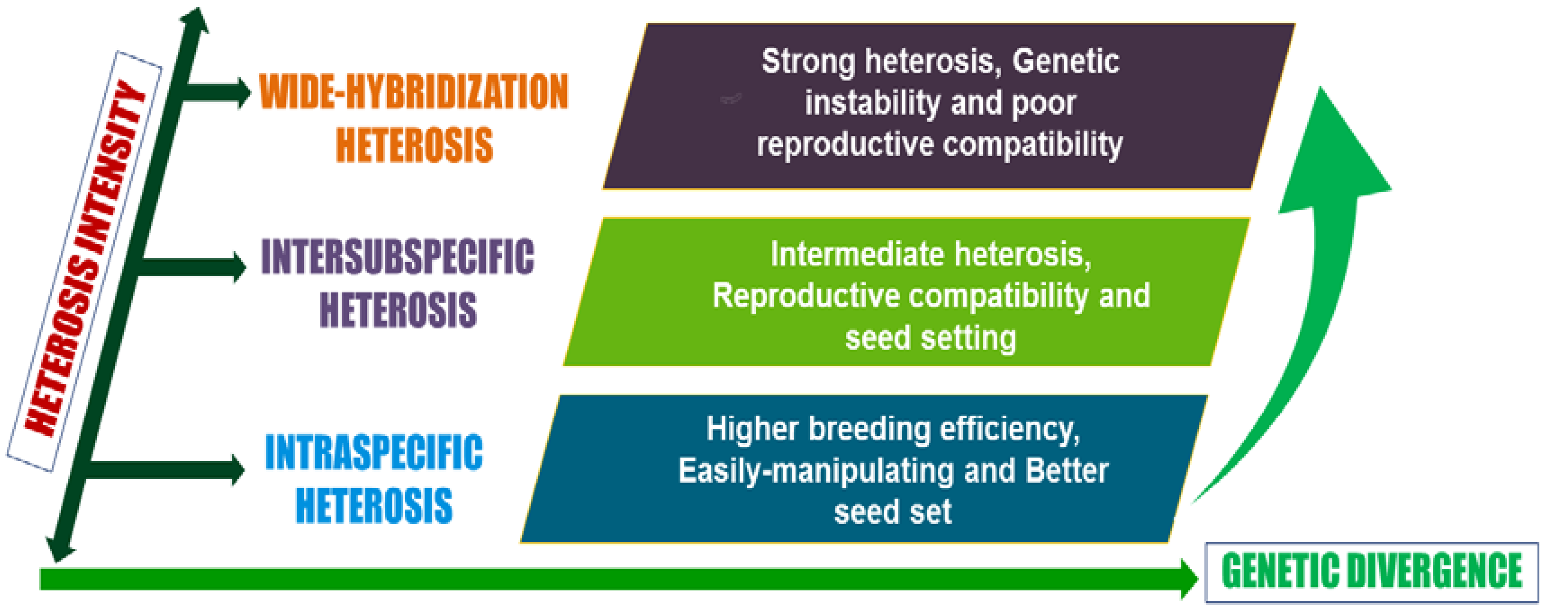
4. Heterosis Prediction
5. Genetic and Molecular Basis of Heterosis
6. Heterosis in Eggplant
7. Prospects of Male Sterility in Eggplant Heterosis
8. Epigenetic Regulation of Heterosis in Eggplant
9. Application of Transgenic Approaches and Genome Editing
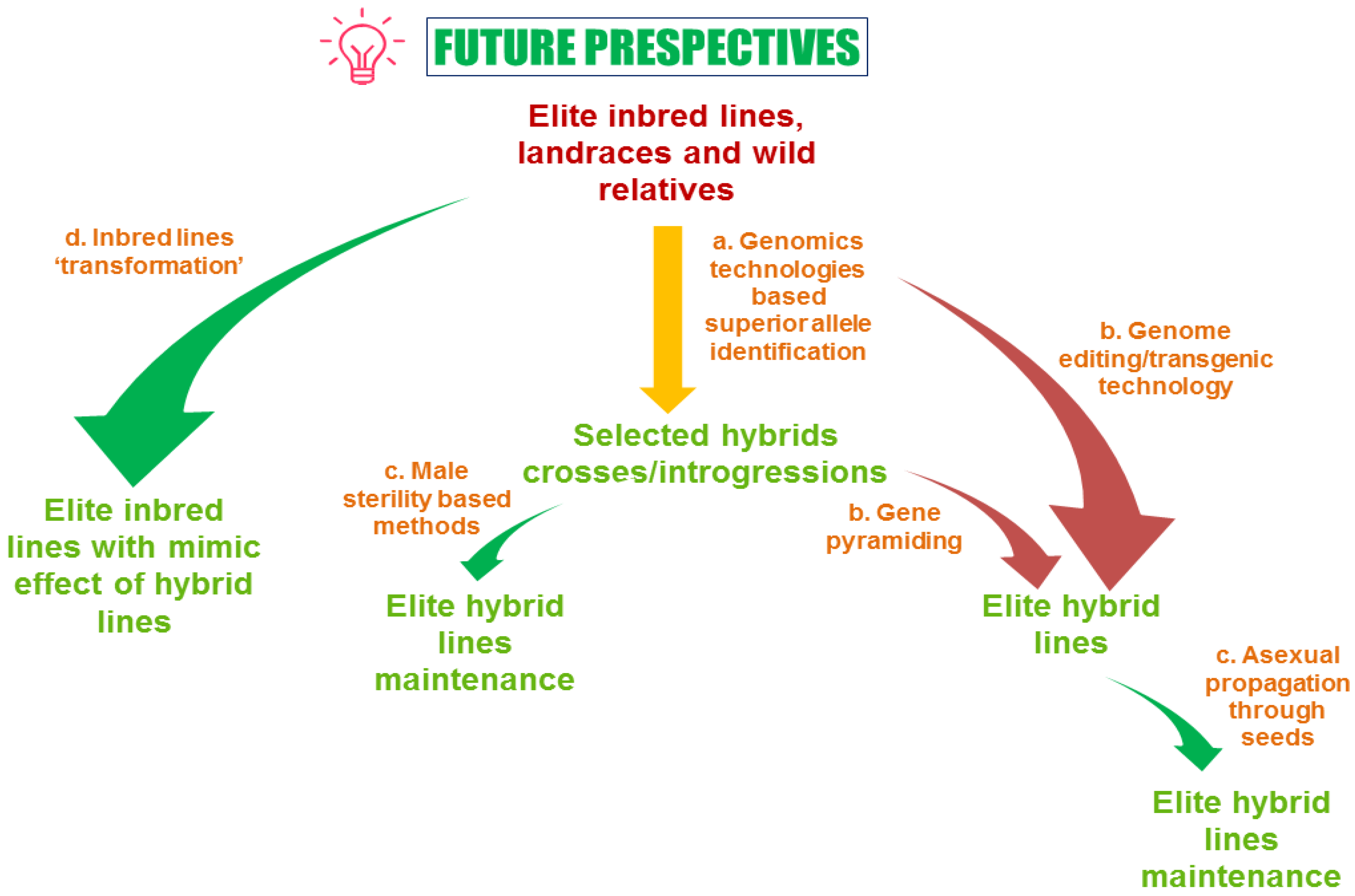
10. Conclusions and Future Perspective
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Saini, D.K.; Kaushik, P. Visiting eggplant from a biotechnological perspective: A review. Sci. Hortic. 2019, 253, 327–340. [Google Scholar] [CrossRef]
- FAOSTAT. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 11 January 2019).
- Chen, N.C.; Li, H.M. Cultivation and breeding of eggplant. In Proceedings of the Training Workshop on Vegetable Cultivation and Seed Production, Shanhua, Tainan, 11–30 November 1996; Volume 26. [Google Scholar]
- Kaushik, P.; Plazas, M.; Prohens, J.; Vilanova, S.; Gramazio, P. Diallel genetic analysis for multiple traits in eggplant and assessment of genetic distances for predicting hybrids performance. PLoS ONE 2018, 13, e0199943. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, P. Line × Tester Analysis for Morphological and Fruit Biochemical Traits in Eggplant (Solanum melongena L.) Using Wild Relatives as Testers. Agronomy 2019, 9, 185. [Google Scholar] [CrossRef]
- Birchler, J.A.; Yao, H.; Chudalayandi, S.; Vaiman, D.; Veitia, R.A. Heterosis. Plant Cell 2010, 22, 2105–2112. [Google Scholar] [CrossRef] [PubMed]
- Ellstrand, N.C.; Prentice, H.C.; Hancock, J.F. Gene flow and introgression from domesticated plants into their wild relatives. Ann. Rev. Ecol. Syst. 1999, 30, 539–563. [Google Scholar] [CrossRef]
- Lippman, Z.B.; Zamir, D. Heterosis: Revisiting the magic. Trends Genet. 2007, 23, 60–66. [Google Scholar] [CrossRef]
- Kakizaki, Y. Hybrid vigor in egg-plants and its practical utilization. Genetics 1931, 16, 1–25. [Google Scholar]
- Krommydas, K.S.; Tzikalios, Z.; Madesis, P.; Bletsos, F.A.; Mavromatis, A.; Roupakias, D.G. Development and fertility restoration of CMS eggplant lines carrying the cytoplasm of Solanum violaceum. J. Agric. 2016, 8, 10–26. [Google Scholar] [CrossRef]
- Stuber, C.W. Heterosis in plant breeding. Plant Breed. Rev. 1994, 12, 227–251. [Google Scholar]
- Luo, L.J.; Li, Z.-K.; Mei, H.W.; Shu, Q.Y.; Tabien, R.; Zhong, D.B.; Ying, C.S.; Stansel, J.W.; Khush, G.S.; Paterson, A.H. Overdominant epistatic loci are the primary genetic basis of inbreeding depression and heterosis in rice. II. Grain yield components. Genetics 2001, 158, 1755–1771. [Google Scholar]
- Birchler, J.A.; Yao, H.; Chudalayandi, S. Unraveling the genetic basis of hybrid vigor. Proc. Natl. Acad. Sci. USA 2006, 103, 12957–12958. [Google Scholar] [CrossRef]
- O’Sullivan, D.M.; Edwards, K.J. 6 The impact of plant genomics on maize improvement. Plant Mol. Breed. 2003, 12, 152. [Google Scholar]
- Taher, D.; Solberg, S.Ø.; Prohens, J.; Chou, Y.; Rakha, M.; Wu, T. World Vegetable Center Eggplant Collection: Origin, Composition, Seed Dissemination and Utilization in Breeding. Front. Plant Sci. 2017, 8, 1484. [Google Scholar] [CrossRef] [PubMed]
- Davidar, P.; Snow, A.A.; Rajkumar, M.; Pasquet, R.; Daunay, M.C.; Mutegi, E. The potential for crop to wild hybridization in eggplant (Solanum melongena; Solanaceae) in southern India. Am. J. Bot. 2015, 102, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.C. Eggplant Seed Production; AVRDC International Cooperators’ Training Guide Shanhua: Shanhua, Taiwan, 2001; pp. 1–14. Available online: https://afghanag.ucdavis.edu/fruits-nuts-vegetables/Vegetables/files/eggplant/Man_Veg_Eggplant_Seed_Prod_AVRDC.pdf (accessed on 23 January 2020).
- Legesse, B.W.; Pixley, K.V.; Botha, A.-M. Combining ability and heterotic grouping of highland transition maize inbred lines. Maydica 2009, 54, 9–14. [Google Scholar]
- Hunziker, A.T. South American Solanaceae: A Synoptic Survey; Linnean Society symposium series; Universidad Nacional de Coìrdoba, Facultad de Ciencias Exactas: Bogota, Colombia, 1979. [Google Scholar]
- Vorontsova, M.S.; Knapp, S. A new species of Solanum (Solanaceae) from South Africa related to the cultivated eggplant. PhytoKeys 2012, 8, 1–11. [Google Scholar] [CrossRef]
- Frodin, D.G. History and concepts of big plant genera. Taxon 2004, 53, 753–776. [Google Scholar] [CrossRef]
- Smith, C.E. The New World Centers of Origin of Cultivated Plants and the Archaeological Evidence. Economic Botany 1968, 22, 253–266. [Google Scholar] [CrossRef]
- Vavilov, N.I. TheOrigin, Variation, Immunity and Breeding of Cultivated Plants; Chronica Botanica: New York, NY, USA, 1951. [Google Scholar]
- Knapp, S.; Vorontsova, M.S.; Prohens, J. Wild relatives of the eggplant (Solanum melongena L. Solanaceae): New understanding of species names in a complex group. PLoS ONE 2013, 8, e57039. [Google Scholar] [CrossRef]
- Meyer, R.S.; Karol, K.G.; Little, D.P.; Nee, M.H.; Litt, A. Phylogeographic relationships among Asian eggplants and new perspectives on eggplant domestication. Mol. Phylogenet. Evol. 2012, 63, 685–701. [Google Scholar] [CrossRef]
- Prohens, J.; Blanca, J.M.; Nuez, F. Morphological and molecular variation in a collection of eggplants from secondary center of diversity: Implications for conservation and breeding. J. Am. Soc. Hort. Sci. 2005, 130, 54–63. [Google Scholar] [CrossRef]
- Bohs, L. Major clades in Solanum based on ndhF sequences. In A festschrift for William G. D’Arcy: The legacy of a taxonomist, Monographs in systematic botany from the Missouri Botanical Garden; Keating, R.C., Hollowell, V.C., Croat, T.B., Eds.; Missouri Botanical Garden Press: St. Louis, MO, USA, 2005; Volume 104, pp. 27–49. [Google Scholar]
- Echeverria-Londoño, S.; Särkinen, T.; Fenton, I.S.; Knapp, S.; Purvis, A. Dynamism and context dependency in diversification of a megadiverse plant genus Solanum L. (Solanaceae). bioRxiv 2018, 348961. [Google Scholar] [CrossRef]
- Särkinen, T.; Bohs, L.; Olmstead, R.G.; Knapp, S. A phylogenetic framework for evolutionary study of the nightshades (Solanaceae): A dated 1000-tip tree. BMC Evol. Biol. 2013, 13, 214. [Google Scholar] [CrossRef] [PubMed]
- Robinson, R.W.; Shail, J.W.; Gao, Y.X.; Doganlar, S. Interspecific hybridization of eggplant for Verticillium wilt resistance and other useful traits. In Solanaceae V. Advances in Taxonomy and Utilization; Nijmegen University Press: Nijmegen, The Netherlands, 2001; pp. 279–291. [Google Scholar]
- Acciarri, N.; Rotino, G.L.; Valentino, D.; Vitelli, G.; Sunseri, F.; Martelli, G.; Tamietti, G. Genetic improvement of eggplant for resistance to Verticillium through interspecific hybridization with Solanum sodomeum L. In Proceedings of the 11th Eucarpia Meeting on Genetics and Breeding of Capsicum and Eggplant, Antalya, Turkey, 9–13 April 2001; pp. 298–302. [Google Scholar]
- Nunome, T.; Ishiguro, K.; Yoshida, T.; Hirai, M. Mapping of fruit shape and color development traits in eggplant (Solanum melongena L.) based on RAPD and AFLP markers. Breed. Sci. 2001, 51, 19–26. [Google Scholar] [CrossRef][Green Version]
- Dempewolf, H.; Baute, G.; Anderson, J.; Kilian, B.; Smith, C.; Guarino, L. Past and future use of wild relatives in crop breeding. Crop. Sci. 2017, 57, 1070–1082. [Google Scholar] [CrossRef]
- Gramazio, P.; Prohens, J.; Plazas, M.; Mangino, G.; Herraiz, F.J.; García-fortea, E.; Vilanova, E. Genomic tools for the enhancement of vegetable crops: A case in eggplant. Notulae Botanicae Horti Agrobotanici Cluj-Napoca Cluj-Napoca 2018, 46, 1–13. [Google Scholar] [CrossRef]
- Kaushik, P.; Prohens, J.; Vilanova, S.; Gramazio, P.; Plazas, M. Phenotyping of eggplant wild relatives and interspecific hybrids with conventional and phenomics descriptors provides insight for their potential utilization in breeding. Front. Plant Sci. 2016, 7, 677. [Google Scholar] [CrossRef]
- Kouassi, B.; Prohens, J.; Gramazio, P.; Kouassi, A.B.; Vilanova, S.; Galan-Avila, A.; Herraiz, F.J.; Kouassi, A.; Segui-Simarro, J.M.; Plazas, M. Development of backcross generations and new interspecific hybrid combinations for introgression breeding in eggplant (Solanum melongena). Sci. Hortic. 2016, 213, 199–207. [Google Scholar] [CrossRef]
- Kaushik, P.; Gramazio, P.; Vilanova, S.; Raigón, M.D.; Prohens, J.; Plazas, M. Phenolics content, fruit flesh colour and browning in cultivated eggplant, wild relatives and interspecific hybrids and implications for fruit quality breeding. Food Res. Int. 2017, 102, 392–401. [Google Scholar] [CrossRef]
- Rajendrakumar, P.; Hariprasanna, K.; Seetharama, N. Prediction of Heterosis in Crop Plants–Status and Prospects. J. Exp. Agric. 2015, 1–16. [Google Scholar] [CrossRef]
- Oettler, G.; Tams, S.H.; Utz, H.F.; Bauer, E.; Melchinger, A.E. Prospects for hybrid breeding in winter triticale. I. Heterosis and combining ability for agronomic traits in European elite germplasm. Crop. Sci. 2005, 45, 1476–1482. [Google Scholar] [CrossRef]
- Kaushik, P. Application of Conventional, Biotechnological and Genomics Approaches for Eggplant (Solanum melongena.L). Breeding with a Focus on Bioactive Phenolics; Universitat Politècnica de València: Valencia, Spain, 2019. [Google Scholar] [CrossRef]
- Lee, E.A.; Ash, M.J.; Good, B. Re-examining the relationship between degree of relatedness, genetic effects and heterosis in maize. Crop. Sci. 2007, 47, 629–635. [Google Scholar] [CrossRef]
- Verma, P.S.; Singh, S.B. Heterosis in relation to per se performance and effects of general combining ability in sugarcane. Sugar Tech. 2004, 6, 181–185. [Google Scholar] [CrossRef]
- Zhang, T.; Ni, X.L.; Jiang, K.F.; Deng, H.F.; He, Q.; Yang, Q.H.; Li, Y.A.N.G.; Xian-Qi, W.A.N.; Cao, Y.J.; Zheng, J.K. Relationship between heterosis and parental genetic distance based on molecular markers for functional genes related to yield traits in rice. Rice Sci. 2010, 17, 288–295. [Google Scholar] [CrossRef]
- Smith, O.S.; Smith, J.S.C.; Bowen, S.L.; Tenborg, R.A.; Wall, S.J. Similarities among a group of elite maize inbreds as measured by pedigree, F1 grain yield, grain yield heterosis and RFLPs. Theor. Appl. Genet. 1990, 80, 833–840. [Google Scholar] [CrossRef]
- Corbellini, M.; Perenzin, M.; Accerbi, M.; Vaccino, P.; Borghi, B. Genetic diversity in bread wheat, as revealed by coefficient of parentage and molecular markers, and its relationship to hybrid performance. Euphytica 2002, 123, 273–285. [Google Scholar] [CrossRef]
- Cheres, M.T.; Miller, J.F.; Crane, J.M.; Knapp, S.J. Genetic distance as a predictor of heterosis and hybrid performance within and between heterotic groups in sunflower. Theor. Appl. Genet. 2000, 100, 889–894. [Google Scholar] [CrossRef]
- Diers, B.W.; McKetty, P.B.E.; Osborn, T.C. Relationship between heterosis and genetic distance based on restriction fragment length polymorphism markers in oilseed rape (Brassica napus L). Crop. Sci. 1996, 36, 79–83. [Google Scholar] [CrossRef]
- Zenke-Philippi, C.; Thiemann, A.; Seifert, F.; Schrag, T.; Melchinger, A.E.; Scholten, S.; Frisch, M. Prediction of hybrid performance in maize with a ridge regression model employed to DNA markers and mRNA transcription profiles. BMC Genom. 2016, 17, 262. [Google Scholar] [CrossRef]
- Frisch, M.; Thiemann, A.; Fu, J.; Schrag, T.A.; Scholten, S.; Melchinger, A.E. Transcriptome-based distance measures for grouping of germplasm and prediction of hybrid performance in maize. Theor. Appl. Genet. 2010, 120, 441–450. [Google Scholar] [CrossRef]
- Fu, J.; Falke, K.C.; Thiemann, A.; Schrag, T.A.; Melchinger, A.E.; Scholten, S.; Frisch, M. Partial least squares regression, support vector machine regression, and transcriptome-based distances for prediction of maize hybrid performance with gene expression data. Theor. Appl. Genet. 2012, 124, 825–833. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, T.; Wimmer, V.; Auinger, H.J.; Erbe, M.; Knaak, C.; Ouzunova, M.; Simianer, H.; Schön, C.C. Genome-based prediction of testcross values in maize. Theor. Appl. Genet. 2011, 123, 339. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, P. Genetic Analysis for Fruit Phenolics Content, Flesh Color, and Browning Related Traits in Eggplant (Solanum melongena). Int. J. Mol. Sci. 2019, 20, 2990. [Google Scholar] [CrossRef]
- Riedelsheimer, C.; Czedik-Eysenberg, A.; Grieder, C.; Lisec, J.; Technow, F.; Sulpice, R.; Altmann, T.; Stitt, M.; Willmitzer, L.; Melchinger, A.E. Genomic and metabolic prediction of complex heterotic traits in hybrid maize. Nat. Genet. 2012, 44, 217–220. [Google Scholar] [CrossRef] [PubMed]
- Vacher, M.; Small, I. Simulation of heterosis in a genome-scale metabolic network provides mechanistic explanations for increased biomass production rates in hybrid plants. NPJ Syst. Biol. Appl. 2019, 5, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Bruce, A.B. The Mendelian theory of heredity and the augmentation of vigor. Science 1910, 32, 627–628. [Google Scholar] [CrossRef]
- Jones, D.F. Dominance of linked factors as a means of accounting for heterosis. Genetics 1917, 2, 466–475. [Google Scholar] [CrossRef]
- Hallauer, A.R.; Carena, M.J.; Miranda Filho, J.D. Quantitative genetics in maize breeding (Vol. 6); Springer Science & Business Media: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Moll, R.; Lindsey, M.F.; Robinson, H.F. Estimates of genetic variances and level of dominance in maize. Genetics 1964, 49, 411. [Google Scholar]
- Fiévet, J.B.; Dillmann, C.; de Vienne, D. Systemic properties of metabolic networks lead to an epistasis-based model for heterosis. Theor. Appl. Genet. 2010, 120, 463–473. [Google Scholar] [CrossRef]
- Buckler, E.S.; Holland, J.B.; Bradbury, P.J.; Acharya, C.B.; Brown, P.J.; Browne, C.; Ersoz, E.; Flint-Garcia, S.; Garcia, A.; Glaubitz, J.C.; et al. The genetic architecture of maize flowering time. Science 2009, 325, 714–718. [Google Scholar] [CrossRef]
- Cook, J.P.; McMullen, M.D.; Holland, J.B.; Tian, F.; Bradbury, P.; Ross-Ibarra, J.; Buckler, E.S.; Flint-Garcia, S.A. Genetic architecture of maize kernel composition in the nested association mapping and inbred association panels. Plant Physiol. 2012, 158, 824–834. [Google Scholar] [CrossRef] [PubMed]
- Tian, F.; Bradbury, P.J.; Brown, P.J.; Hung, H.; Sun, Q.; Flint-Garcia, S.; Rocheford, T.R.; McMullen, M.D.; Holland, J.B.; Buckler, E.S. Genome-wide association study of leaf architecture in the maize nested association mapping population. Nat. Genet. 2011, 43, 159–162. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Yang, S.; Gong, J.; Zhao, Q.; Feng, Q.; Zhan, Q.; Zhao, Y.; Li, W.; Cheng, B.; Xia, J.; et al. Genomic architecture of heterosis for yield traits in rice. Nature 2016, 537, 629–633. [Google Scholar] [CrossRef]
- Liu, J.; Li, M.; Zhang, Q.; Wei, X.; Huang, X. Exploring the molecular basis of heterosis for plant breeding. J. Integr. Plant Biol. 2019, 62, 287–298. [Google Scholar] [CrossRef]
- Dhankhar, B.S.; Mehrotra, N.; Kirti, S. Heterosis in relation to yield components and shoot/fruit borer (Leucinodes orbonalis Gn.) in brinjal (Solanum melongena L.). Genet. Agr. 1980, 34, 215–220. [Google Scholar]
- Chadha, M.L.; Sidhu, S. Studies on hybrid vigour in brinjal (Solanum melongena L.). Indian J. Hort. 1982, 39, 233–238. [Google Scholar]
- Munson, W.M. Notes on eggplants. Maine Agr. Expt. St. Ann. Rep. 1892, 1892, 76–89. [Google Scholar]
- Nagai, K.; Kida, M. An experiment with some crosses of eggplant. Jpn. J. Gene 1926, 4, 10–30. [Google Scholar]
- Mistry, C.R.; Kathiria, K.B.; Sabolu, S.; Kumar, S. Heterosis and inbreeding depression for fruit yield attributing traits in eggplant. Curr. Plant Biol. 2018, 16, 27–31. [Google Scholar] [CrossRef]
- Daunay, M.C.; Hazra, P. Eggplant. In Handbook of Vegetables; Peter, K.V., Hazra, P., Eds.; Studium Press: Houston, TX, USA, 2012; pp. 257–322. [Google Scholar]
- Ansari, A.M.; Singh, Y.V. Heterosis studies for fruit characters in Brinjal (Solanum melongena L.). Electron. J. Plant Breed. 2016, 7, 197–208. [Google Scholar] [CrossRef]
- Ramya, A.R.; Ahamed, M.; Satyavathi, C.T.; Rathore, A.; Katiyar, P.; Raj, A.G.; Kumar, S.; Gupta, R.; Mahendrakar, M.D.; Yadav, R.S.; et al. Towards de- fining heterotic gene pools in pearl millet [Pennisetum glaucum (L.) R. Br.]. Front. Plant Sci. 2018, 8, 1934. [Google Scholar] [CrossRef] [PubMed]
- Virmani, S.S.; Pandey, M.P.; Singh, I.S.; Xu, W.J. Classical and molecular concepts of heterosis. In Plant Breeding; Springer: Dordrecht, The Netherlands, 2004; pp. 407–418. [Google Scholar]
- Birchler, J.A. Heterosis: The genetic basis of hybrid vigour. Nat. Plants 2015, 1, 15020. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Sidhu, A.S.; Kumar, A. Heterosis for fruit yield and its components in brinjal (Solanum melongena L.). J. Hortic. Sci. 2012, 7, 142–144. [Google Scholar]
- Acquaah, G. Principles of Plant Genetics and Breeding; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Shattuck, V.I.; Christie, B.; Corso, C. Principles for Griffing’s combining ability analysis. Genetica 1993, 90, 73–77. [Google Scholar] [CrossRef]
- Rodríguez-Burruezo, A.; Prohens, J.; Nuez, F. Performance of hybrids between local varieties of eggplant (Solanum melongena) and its relation to the mean of parents and to morphological and genetic distances among parents. Eur. J. Hortic. Sci. 2008, 73, 76. [Google Scholar]
- Geleta, L.F.; Labuschagne, M.T.; Viljoen, C.D. Relationship between heterosis and genetic distance based on morphological traits and AFLP markers in pepper. Plant Breed. 2004, 123, 467–473. [Google Scholar] [CrossRef]
- Zhang, J.F.; Abdelraheem, A. Combining ability, heterosis, and genetic distance among nine elite American Pima cotton genotypes (Gossypium barbadense). Euphytica 2017, 213, 240. [Google Scholar] [CrossRef]
- Hernández-Bautista, A.; Lobato-Ortiz, R.; García-Zavala, J.J.; Rocandio-Rodríguez, M.; Mejía-Contreras, J.A.; Chávez-Servia, J.L.; García-Velazquez, J.A. Relationship of parental genetic distance with agronomic performance, specific combining ability, and predicted breeding values of raspberry families. Euphytica 2018, 214, 37. [Google Scholar] [CrossRef]
- Singh, B.; Kumar, N. Studies on hybrid vigour and combining ability in brinjal (Solanum melongena L.). Veg. Sci. 1988, 15, 72–78. [Google Scholar]
- Saha, M.G.; Hossain, A.K.M.A.; Hoque, K.R.; Bhowmik, A. Genetic analysis of plant height and number of branches in brinjal (Solanum melongena L.). Ann. Bangl. Agric. 1991, 1, 91–97. [Google Scholar]
- Das, G.; Barua, N.S. Heterosis and combining ability for yield and its components in brinjal. Ann. Agrl. Res. 2001, 22, 399–403. [Google Scholar]
- Kaur, J.; Patel, J.A.; Patel, M.J.; Bhanvadia, A.S.; Acharya, R.R. Heterosis for fruit yield and its components in brinjal (Solanum melongena L.). Capsicum Egg News 2001, 20, 102–105. [Google Scholar]
- Patil, S.D.; Warade, S.D.; Badgujar, C.D. Heterosis studies in brinjal (Solanum melongena L.). J. Soil Crop 2001, 11, 47–51. [Google Scholar]
- Hamada, M.S.; Hamaiel, A.F.; Farid, S.M.; El-Kady, M.M. Heterosis and Combining Ability For Some Traits of Intraspecific and Interspecific Hybridization Between Solanum melongena and Solanum macrocarpon. J. Agric. Res. Kafr El-Sheikh Univ. 2016, 42, 34–45. [Google Scholar]
- Akpan, N.M.; Ogbonna, P.E.; Onyla, V.N.; Okechukwn, E.C.; Dominic, I.O.I.; Atugwu, A.I. Genetic Control and Heterosis of Quantitative Traits in Several Local Eggplant Genotypes. Not. Sci. Biol. 2017, 9, 520–524. [Google Scholar] [CrossRef]
- Shahjahan, M.; Kabir, K.; Zomo, S.A.; Sarkar, M.D.; Fazlullah, M.U. Evaluation of heterosis in exotic eggplant. Int. J. Environ. Agric. Res. 2016, 1, 23–32. [Google Scholar]
- Kalaiyarasi, G.; Ram, S.R.R.; Saravanan, K. Studies on Heterosis for yield in Brinjal (Solanum melongena. L). Horti. Biotechnol. Res. 2018, 4, 35–38. [Google Scholar]
- Patel, A.A.; Gohil, D.P.; Dhruve, J.J.; Damor, H.I. Heterosis for fruit yield and its quality characters in brinjal (Solanum melongena L.). J. Pharmacogn. Phytochem. 2017, 6, 975–978. [Google Scholar]
- Khan, M.M.R.; Isshiki, S. Cytoplasmic male sterility in eggplant. Hortic. J. 2016, 85, 1–7. [Google Scholar] [CrossRef]
- Phatak, S.C.; Liu, J.; Jaworski, C.A.; Sultanbawa, A.F. Functional male sterility in eggplant: Inheritance and linkage to the purple fruit color gene. J. Hered. 1991, 82, 81–83. [Google Scholar] [CrossRef]
- Khan, M.M.R.; Isshiki, S. Development of a cytoplasmic male-sterile line of eggplant (Solanum melongena L.) with the cytoplasm of Solanum anguivi. Plant Breed. 2011, 130, 256–260. [Google Scholar] [CrossRef]
- Hasnunnahar, M.; Khan, M.R.; Isshiki, S. Inheritance analysis of fertility restoration genes (Rf) in a male sterile system of eggplant using cytoplasm of Solanum grandifolium. AJCS 2012, 6, 475–479. [Google Scholar]
- Cao, B.; Huang, Z.; Chen, G.; Lei, J. Restoring pollen fertility in transgenic male-sterile eggplant by Cre/loxp-mediated site-specific recombination system. Genet. Mol. Biol. 2010, 33, 298–307. [Google Scholar] [CrossRef]
- Toppino, L.; Kooiker, M.; Lindner, M.; Dreni, L.; Rotino, G.L.; Kater, M.M. Reversible male sterility in eggplant (Solanum melongena L.) by artificial microRNA-mediated silencing of general transcription factor genes. Plant. Biotechnol. J. 2011, 9, 684–692. [Google Scholar] [CrossRef]
- Budar, F.; Pelletier, G. Male sterility in plants: Occurrence, determinism, significance and use. Comptes Rendus de l’Académie Des Sciences, Series III, Sciences de La Vie. Eur PMC 2001, 324, 543–550. [Google Scholar]
- Hazra, P.; Roy, T.; Choudhury, J. Characterization of genic functional male sterility in brinjal and its expression in different genetic backgrounds. Indian J. Genet. 2008, 68, 47–51. [Google Scholar]
- Costa-Font, M.; Gil, J.M.; Traill, W.B. Consumer acceptance, valuation of and attitudes towards genetically modified food: Review and implications for food policy. Food Policy 2008, 33, 99–111. [Google Scholar] [CrossRef]
- Bentolila, S.; Alfonso, A.A.; Hanson, M.R. A pentatricopeptide repeat-containing gene restores fertility to cytoplasmic male-sterile plants. Proc. Natl. Acad. Sci. USA 2002, 99, 10887–10892. [Google Scholar] [CrossRef]
- Yoshimi, M.; Kitamura, Y.; Isshiki, S.; Saito, T.; Yasumoto, K.; Terachi, T.; Yamagishi, H. Variations in the structure and transcription of the mitochondrial atp and cox genes in wild Solanum species that induce male sterility in eggplant (S.melongena). Theor. Appl. Genet. 2013, 126, 1851–1859. [Google Scholar] [CrossRef]
- Yang, Y.; Bao, S.; Zhou, X.; Liu, J.; Zhuang, Y. The key genes and pathways related to male sterility of eggplant revealed by comparative transcriptome analysis. BMC Plant Biol. 2018, 18, 209. [Google Scholar] [CrossRef]
- Kapazoglou, A.; Ganopoulos, I.; Tani, E.; Tsaftaris, A. Epigenetics, epigenomics and crop improvement. Adv. Bot. Res. 2018, 86, 287–324. [Google Scholar]
- Kenchanmane Raju, S.K.; Niederhuth, C.E. Epigenetic diversity and application to breeding. Adv. Bot. Res 2018, 88, 49–86. [Google Scholar]
- Dapp, M.; Reinders, J.; Bédiée, A.; Balsera, C.; Bucher, E.; Theiler, G.; Granier, C.; Paszkowski, J. Heterosis and inbreeding depression of epigenetic Arabidopsis hybrids. Nat. Plants 2015, 1, 15092. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Chen, L.; Zhang, C.; Hao, P.; Jing, X.; Li, X. Global transcriptome analysis reveals extensive gene remodeling, alternative splicing and differential transcription profiles in non-seed vascular plant Selaginella moellendorffii. BMC Genom. 2017, 18, 1042. [Google Scholar] [CrossRef] [PubMed]
- Greaves, I.K.; Gonzalez-Bayon, R.; Wang, L.; Zhu, A.; Liu, P.C.; Groszmann, M.; Peacock, W.J.; Dennis, E.S. Epigenetic changes in hybrids. Plant Physiol. 2015, 168, 1197–1205. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Sun, S.; Hua, S.; Shen, E.; Ye, C.Y.; Cai, D.; Timko, M.P.; Zhu, Q.H.; Fan, L. Analysis of transcriptional and epigenetic changes in hybrid vigor of allopolyploid Brassica napus uncovers key roles for small RNAs. Plant J. 2017, 91, 874–893. [Google Scholar] [CrossRef] [PubMed]
- Zhu, A.; Greaves, I.K.; Dennis, E.S.; Peacock, W.J. Genome-wide analyses of four major histone modifications in Arabidopsis hybrids at the germinating seed stage. BMC Genom. 2017, 18, 137. [Google Scholar] [CrossRef]
- Chapman, E.J.; Carrington, J.C. Specialization and evolution of endogenous small RNA pathways. Nat. Rev. Genet. 2007, 8, 884–896. [Google Scholar] [CrossRef]
- Herr, A.J.; Jensen, M.B.; Dalmay, T.; Baulcombe, D.C. RNA polymerase IV directs silencing of endogenous DNA. Science 2005, 308, 118–120. [Google Scholar] [CrossRef]
- Onodera, Y.; Haag, J.R.; Ream, T.; Nunes, P.C.; Pontes, O.; Pikaard, C.S. Plant nuclear RNA polymerase IV mediates siRNA and DNA methylation-dependent heterochromatin formation. Cell 2005, 120, 613–622. [Google Scholar] [CrossRef]
- Law, J.A.; Jacobsen, S.E. Establishing, maintaining and modifying DNA methylation patterns in plants and animals. Nat. Rev. Genet. 2010, 11, 204–220. [Google Scholar] [CrossRef]
- Haag, J.R.; Pikaard, C.S. Multisubunit RNA polymerases IV and V: Purveyors of non-coding RNA for plant gene silencing. Nat. Rev. Mol. Cell Biol. 2011, 12, 483–492. [Google Scholar] [CrossRef] [PubMed]
- Wassenegger, M.; Heimes, S.; Riedel, L.; Sanger, H.L. RNA-directed de novo methylation of genomic sequences in plants. Cell 1994, 76, 567–576. [Google Scholar] [CrossRef]
- He, G.; Zhu, X.; Elling, A.A.; Chen, L.; Wang, X.; Guo, L.; Liang, M.; He, H.; Zhang, H.; Chen, F.; et al. Global epigenetic and transcriptional trends among two rice subspecies and their reciprocal hybrids. Plant Cell 2010, 22, 17–33. [Google Scholar] [CrossRef] [PubMed]
- Barber, W.T.; Zhang, W.; Win, H.; Varala, K.K.; Dorweiler, J.E.; Hudson, M.E.; Moose, S.P. Repeat associated small RNAs vary among parents and following hybridization in maize. Proc. Natl Acad. Sci. USA 2012, 109, 10444–10449. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Kim, M.; Han, J.; Yeom, K.H.; Lee, S.; Baek, S.H.; Kim, V.N. MicroRNA genes are transcribed by RNA polymerase II. EMBO J. 2004, 23, 4051–4060. [Google Scholar] [CrossRef] [PubMed]
- Park, W.; Li, J.; Song, R.; Messing, J.; Chen, X. Carpel Factory, a Dicer homolog, and HEN1, a novel protein, act in microRNA metabolism in Arabidopsis thaliana. Curr. Biol. 2002, 12, 1484. [Google Scholar] [CrossRef]
- Shivaprasad, P.V.; Dunn, R.M.; Santos, B.A.; Bassett, A.; Baulcombe, D.C. Extraordinary transgressive phenotypes of hybrid tomato are influenced by epigenetics and small silencing RNAs. EMBO J. 2012, 31, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.; He, H.; Li, J.; Chen, W.; Wang, X.; Guo, L.; Peng, Z.; He, G.; Zhong, S.; Qi, Y.; et al. Genome-wide analysis of DNA methylation and gene expression changes in two Arabidopsis ecotypes and their reciprocal hybrids. Plant Cell 2012, 24, 875–892. [Google Scholar] [CrossRef]
- Greaves, I.K.; Groszmann, M.; Ying, H.; Taylor, J.M.; Peacock, W.J.; Dennis, E.S. Trans chromosomal methylation in Arabidopsis hybrids. Proc. Natl Acad. Sci. USA 2012, 109, 3570–3575. [Google Scholar] [CrossRef]
- Chodavarapu, R.K.; Feng, S.; Ding, B.; Simon, S.A.; Lopez, D.; Jia, Y.; Wang, G.L.; Meyers, B.C.; Jacobsen, S.E.; Pellegrini, M. Transcriptome and methylome interactions in rice hybrids. Proc. Natl Acad. Sci. USA 2012, 109, 12040–12045. [Google Scholar] [CrossRef] [PubMed]
- Ferguson-Smith, A.C. Genomic imprinting: The emergence of an epigenetic paradigm. Nat. Rev. Genet. 2011, 12, 565–575. [Google Scholar] [CrossRef]
- Mosher, R.A.; Melnyk, C.W.; Kelly, K.A.; Dunn, R.M.; Studholme, D.J.; Baulcombe, D.C. Uniparental expression of PolIV-dependent siRNAs in developing endosperm of Arabidopsis. Nature 2009, 460, 283–286. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Zhang, C.; Baulcombe, D.C.; Chen, Z.J. Maternal siRNAs as regulators of parental genome imbalance and gene expression in endosperm of Arabidopsis seeds. Proc. Natl Acad. Sci. USA 2012, 109, 5529–5534. [Google Scholar] [CrossRef]
- Ng, D.W.; Lu, J.; Chen, Z.J. Big roles for small RNAs in polyploidy, hybrid vigor, and hybrid incompatibility. Curr. Opin. Plant Biol. 2012, 15, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.J. Genomic and epigenetic insights into the molecular bases of heterosis. Nat. Rev. Genet. 2013, 14, 471–482. [Google Scholar] [CrossRef]
- James, C. Global review of commercialized transgenic crops. Curr. Sci. 2003, 84, 303–309. [Google Scholar]
- Liu, B.; Zeng, Q.; Yan, F.M.; Xu, H.G.; Xu, C.R. Effects of transgenic plants on soil microorganisms. Plant Soil 2005, 271, 1–13. [Google Scholar] [CrossRef]
- Groot, A.T.; Dicke, M. Insect-resistant transgenic plants in a multi-trophic context. Plant J. 2002, 31, 387–406. [Google Scholar] [CrossRef]
- Hung, Y.H.; Liu, F.; Zhang, X.Q.; Xiao, W.; Hsieh, T.F. Sexual and Non-sexual Reproduction: Inheritance and Stability of Epigenetic Variations and Consequences for Breeding Application. Adv. Bot. Res. 2018, 88, 117–163. [Google Scholar]

| Descriptors | S. insanum | S. anguivi | S. incanum | S. lichtensteinii | S. linnaeanum | S. tomentosum |
|---|---|---|---|---|---|---|
| Plant height (cm) | 16.7 ± 4.6 | 34.4 ± 7.1 | 36.8 ± 11.3 | 38.1 ± 4.4 | 2.3 | 23.3 ± 4.2 |
| Stem diameter (mm) | 10.5 ± 4.3 | 10.4 ± 3.8 | 29.1 ± 11.0 | 39.8 ± 10.3 | −18.7 | 23.8 ± 3.8 |
| Leaf prickles (upper surface) | 155.1 ± 34.5 | 260.0 ±173.9 | 733.3 ± 100.0 | 144.4 ± 92.9 | 100 | 800.0 ± 800.0 |
| Leaf pedicel length (cm) | 39.7 ± 6.5 | 22.5 ± 7.8 | 19.5 ± 2.7 | 24.9 ± 9.2 | −13.3 | 56.3 ± 23.9 |
| Leaf blade length (cm) | 24.9 ± 4.1 | 22.2 ± 5.5 | 47.6 ± 6.6 | 30.6 ± 6.3 | 3.9 | 22.8 ± 1.6 |
| Leaf blade width (cm) | 27.7 ± 4.5 | 38.2 ± 9.5 | 67.7 ± 9.6 | 41.7 ± 8.5 | 7.1 | 22.4 ± 14.0 |
| Number of flowers per inflorescence | 70.1 ± 16.0 | 75.9 ± 16.3 | 21.0 ± 9.4 | 42.7 ± 15.7 | −1.8 | 87.7 ± 35.5 |
| Corolla color | 15.9 ± 4.3 | −2.5 ± 4.6 | 19.2 ± 3.0 | 16.2 ± 4.8 | 7.5 | −0.1 ± 8.6 |
| Number of petals | 1.3 ± 2.1 | −4.8 ± 1.6 | −4.4 ± 2.4 | −2.2 ± 3.4 | −3.2 | −1.0 ± 1.0 |
| Fruit weight (g) | −5.5 ± 6.9 | −98.2 ± 0.3 | −86.6 ± 2.8 | −89.4 ± 1.5 | −89.9 | −98.6 ± 0.3 |
| Fruit calyx prickles | 32.9 ± 25.2 | −100.0 ± 0.0 | 27.1 ± 42.4 | 56.9 ± 27.6 | 80 | 29.1 ± 104.1 |
| Dry matter (%) | −2.9 ± 3.6 | −35.8 ± 6.6 | 12.4 ± 12.8 | −16.6 ± 7.4 | 41.6 | −31.6 ± 14.1 |
| Total phenolics (g/kgDW) | −23.3 ± 5.2 | 16.9 ± 10.2 | 52.5 ± 17.1 | 19.9 ± 9.3 | 21.8 | 73.9 ± 25.1 |
| Chlorogenic acid(mg/g) | −21.8 ± 5.7 | 76.5 ± 17.8 | 1.5 ± 6.9 | 11.5 ± 3.5 | 4.9 | 38.5 ± 8.2 |
| Polyphenol oxidase activity | 16.5 ± 17.8 | 65.6 ± 27.9 | 50.3 ± 81.1 | 23.8 ± 32.5 | −13.7 | 187.3 ± 42.1 |
| Degree of browning | −16.8 ± 12.8 | 85.9 ± 20.2 | 224.6 ± 48.1 | 138.6 ± 26.6 | 143.9 | −9.1 ± 8.5 |
| Trait | Types of Crosses | Range | References |
|---|---|---|---|
| Fruit Yield (q ha-1) | di allel, | 50.48–62.20 | [87,88] |
| Plant Height (cm) | di allel, Line × Tester, F1 Crosses | 6.09–57.77 | [69,87,88,89] |
| Fruit Yield/ Plant(kg) | half diallel, Line × Tester, F1 Crosses | 28.95–63.54 | [69,88,90,91] |
| Number of Fruits/Plant | half diallel, Line × Tester, F1 Crosses | 14.56–158.90 | [69,88,90] |
| Fruit Weight (g) | F1 Crosses | 19.8 | [69] |
| Fruit Length (cm) | Line × Tester, F1 Crosses | 21.81–47.08 | [69,90] |
| Fruit Girth (cm) | Line × Tester, F1 Crosses | 19.15–29.05 | [69,90] |
| Ascorbic acid content (mg/100) | half diallel | 22.39 | [91] |
| Total Phenolic Content (mg/100) | half diallel | 7.97 | [91] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, A.; Sharma, V.; Jain, B.T.; Kaushik, P. Heterosis Breeding in Eggplant (Solanum melongena L.): Gains and Provocations. Plants 2020, 9, 403. https://doi.org/10.3390/plants9030403
Kumar A, Sharma V, Jain BT, Kaushik P. Heterosis Breeding in Eggplant (Solanum melongena L.): Gains and Provocations. Plants. 2020; 9(3):403. https://doi.org/10.3390/plants9030403
Chicago/Turabian StyleKumar, Ashish, Vinay Sharma, Bharat Taindu Jain, and Prashant Kaushik. 2020. "Heterosis Breeding in Eggplant (Solanum melongena L.): Gains and Provocations" Plants 9, no. 3: 403. https://doi.org/10.3390/plants9030403
APA StyleKumar, A., Sharma, V., Jain, B. T., & Kaushik, P. (2020). Heterosis Breeding in Eggplant (Solanum melongena L.): Gains and Provocations. Plants, 9(3), 403. https://doi.org/10.3390/plants9030403







