The Influence of New and Artificial Aged Microplastic and Leachates on the Germination of Lepidium sativum L.
Abstract
1. Introduction
2. Results and Discussion
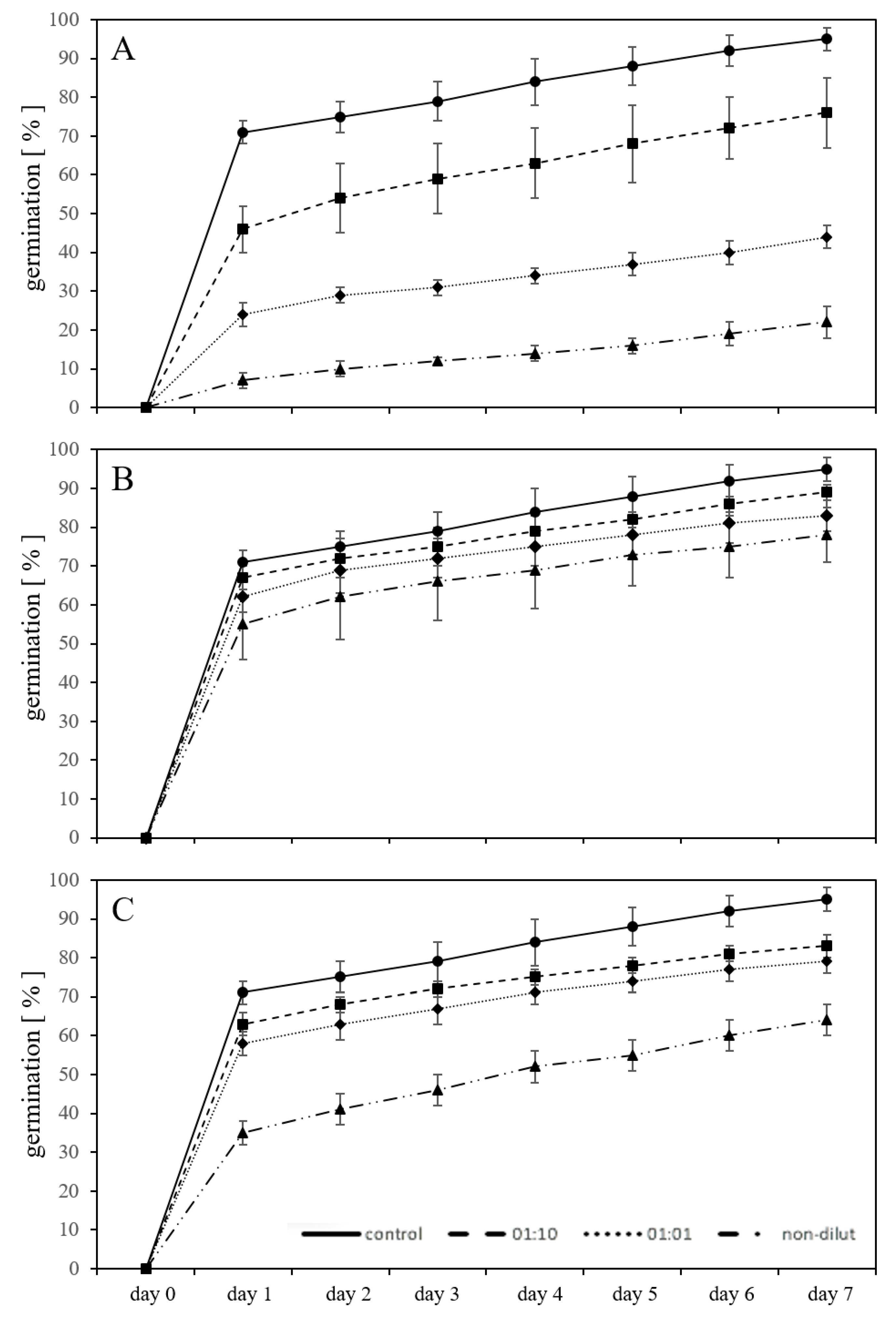
2.1. Germination in Substrate-Free System with Leachates
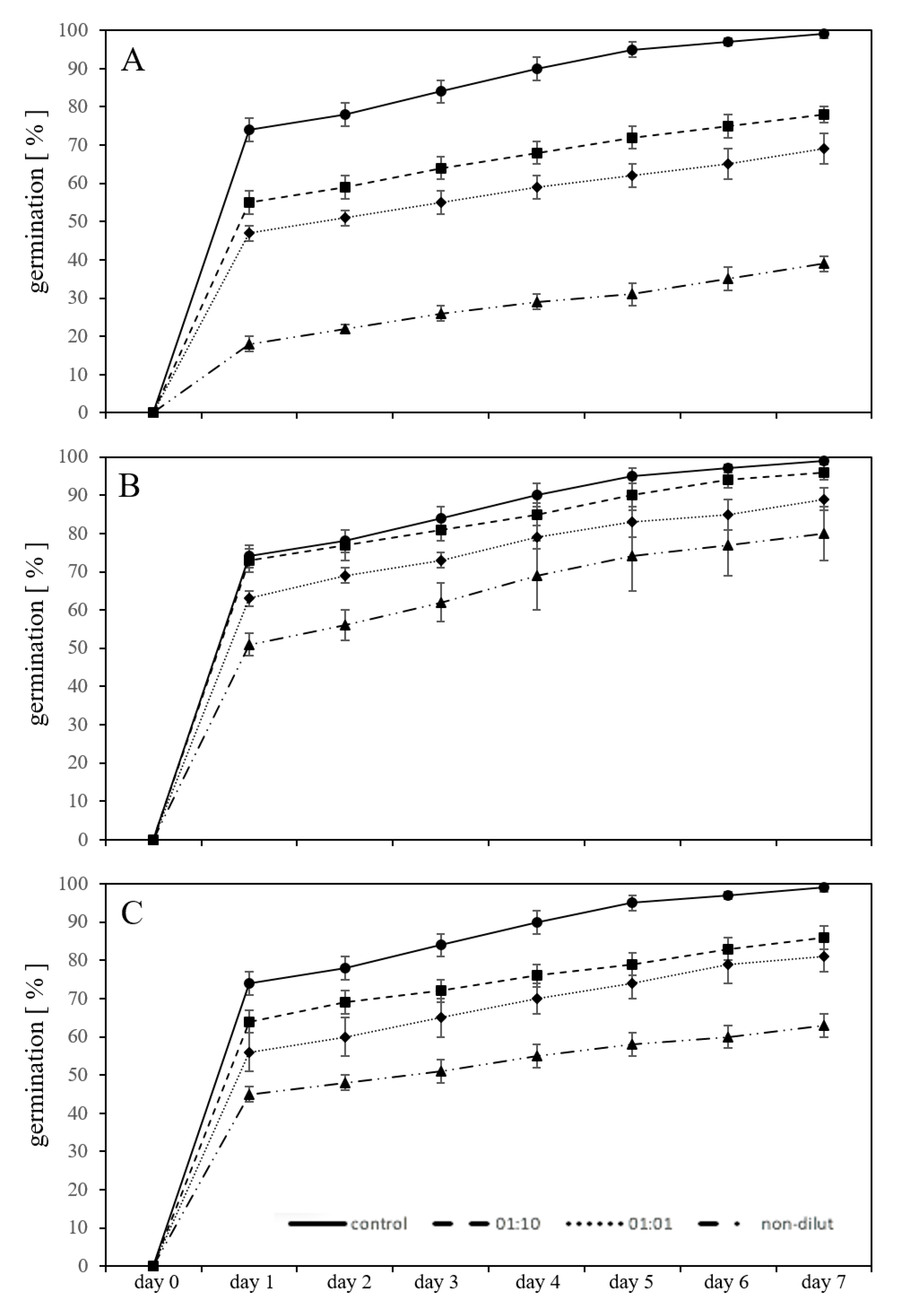
2.2. Germination in a Substrate-System with Exposure to Leachates
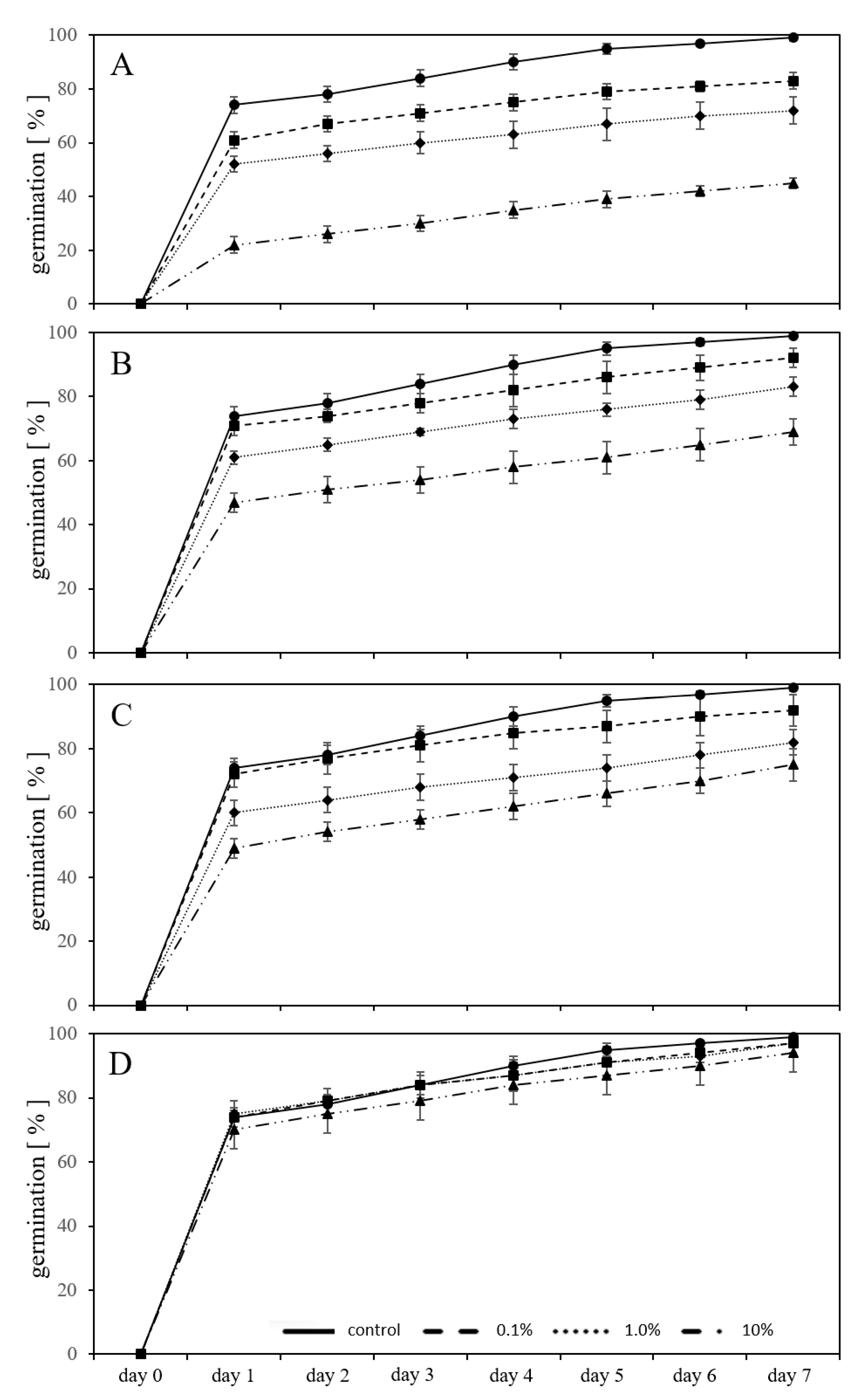
2.3. Germination in a Substrate-System with MP Granules
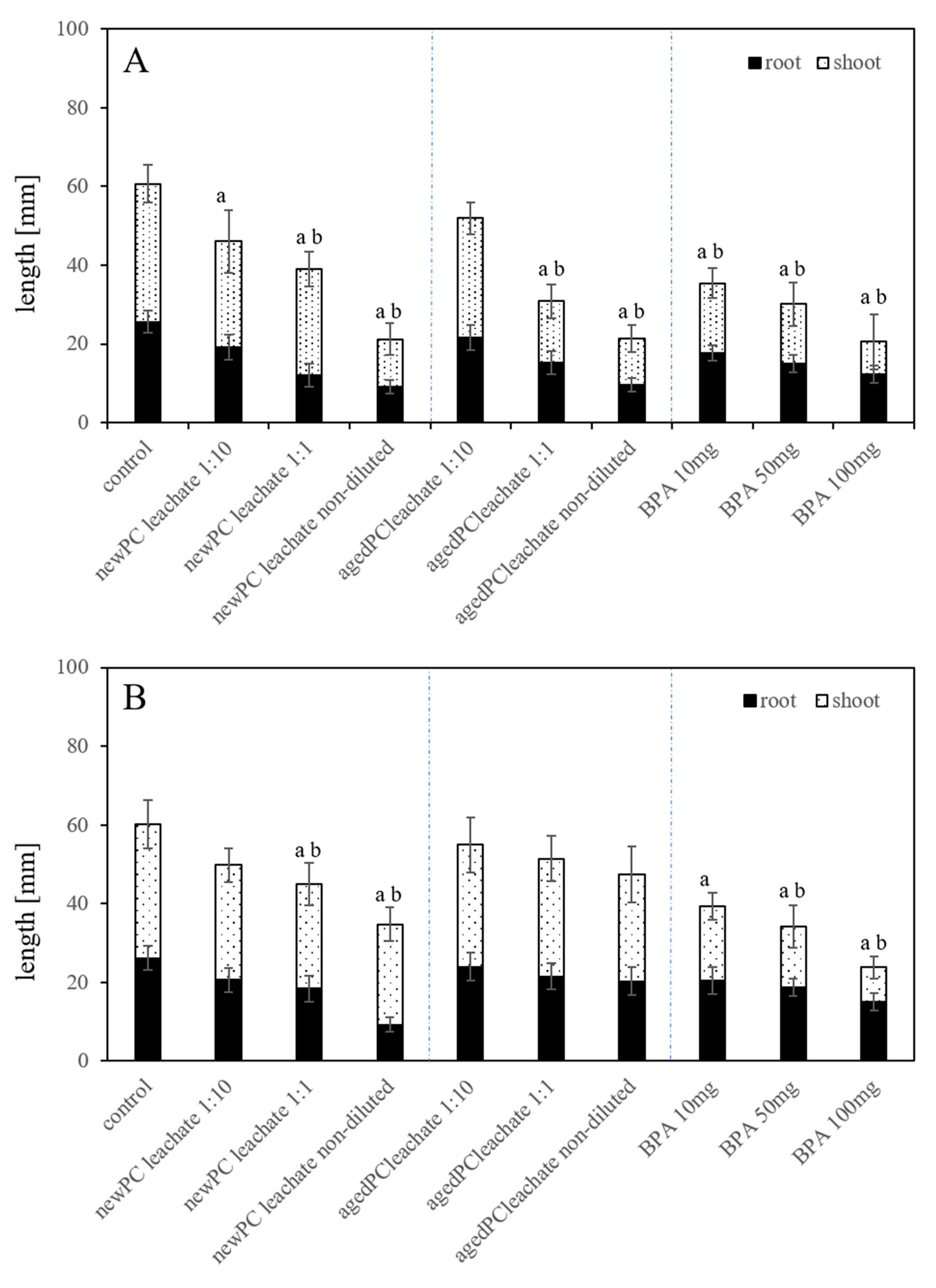
2.4. Root and Seedling Length in Substrate-Free Exposures
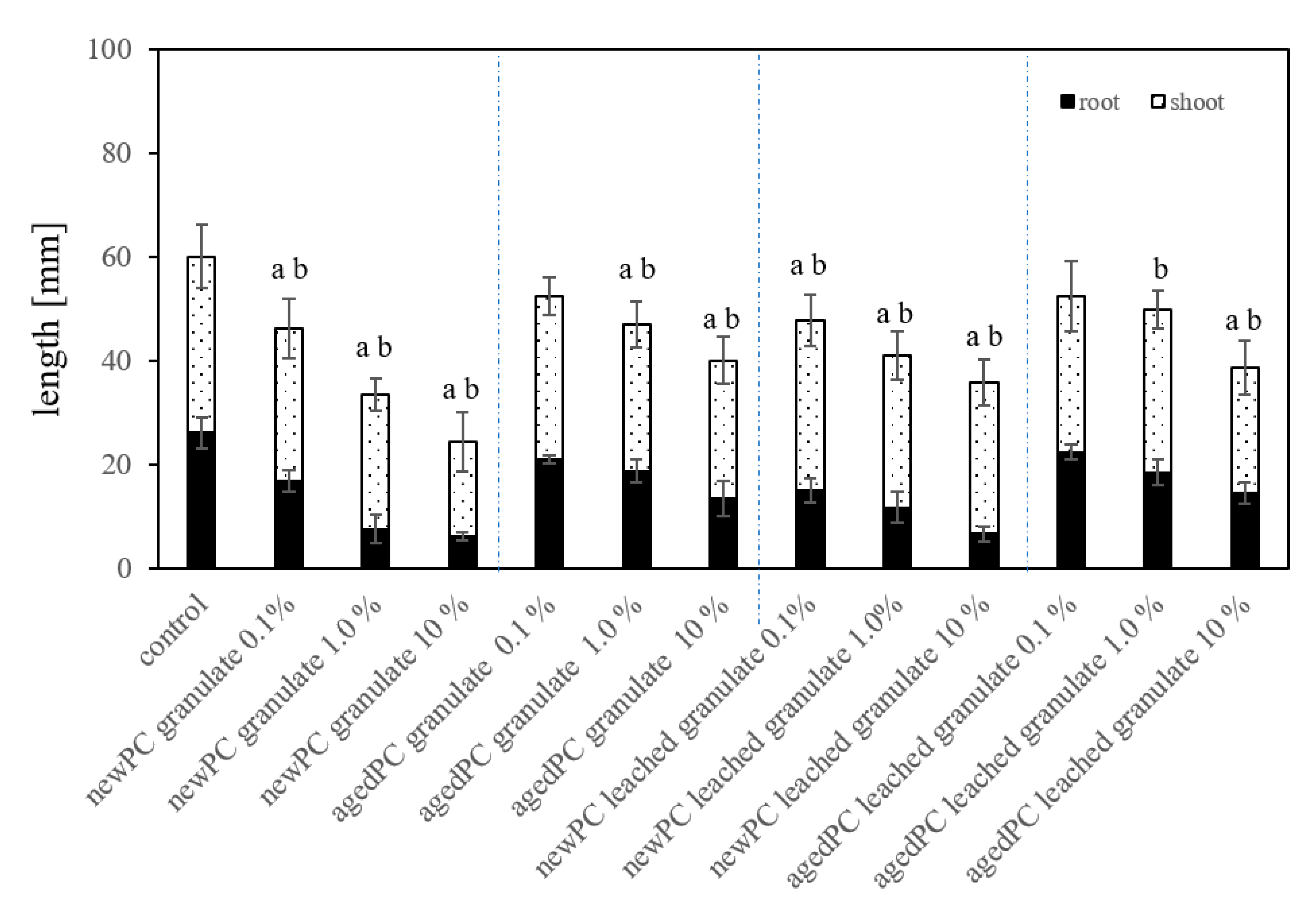
2.5. Root and Seedling Length in Substrate Exposures
2.6. Germination Rate Index Calculation
3. Materials and Methods
3.1. Lepidium sativum (L.) Seeds
3.2. Microplastics, Aged Microplastics and Plastic Leachates
3.3. Bisphenol A
3.4. Control for Plastic Leachate and BPA Exposures
3.5. Experimental Design
3.5.1. Substrate-Free Experiments
3.5.2. Substrate Experiments
3.6. Experimental End Points
- (a)
- Germination percentage (GP) is determined by calculating the level of cumulative seed germination percentage as shown in Equation (1):
- (b)
- Mean total seedlings length, and radicle length was measured in mm after seven days of germination with a digital caliper.
- (c)
- Germination rate index(Speed of germination; GRI) is expressed as the germination in terms of the total number of seeds germinating in a time interval in%/time as per Equation (2):
3.7. Statistics
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, use, and fate of all plastics ever made. Sci. Adv. 2017, 3, e1700782. [Google Scholar] [CrossRef] [PubMed]
- Horton, A.A.; Walton, A.; Spurgeon, D.J.; Lahive, E.; Svendsen, C. Microplastics in freshwater and terrestrial environments: Evaluating the current understanding to identify the knowledge gaps and future priorities. Sci. Total Environ. 2017, 586, 127–141. [Google Scholar] [CrossRef] [PubMed]
- Wallace, H.; Jan, A.; Barregård, L.; Bignami, M.; Ceccatelli, S.; Cottrill, B.; Dinovi, M.; Edler, L.; Grasl-Kraupp, B.; Hogstrand, C.; et al. Presence of microplastics and nanoplastics in food, with particular focus on seafood. EFSA Journal 2016, 14, 1–30. [Google Scholar] [CrossRef]
- Scopetani, C.; Chelazzi, D.; Cincinelli, A.; Esterhuizen-Londt, M. Assessment of microplastic pollution: Occurrence and characterisation in Vesijarvi lake and Pikku Vesijarvi pond, Finland. Environ. Monit. Assess. 2019, 191, 652. [Google Scholar] [CrossRef]
- Hanke, G. Guidance on Monitoring of Marine Litter in European Seas; A guidance document within the common implementation strategy for the marine strategy framework directive. J.R.C. MSDF Technical Subgroup on Marine Litter, Report No. EUR 26113 EN; European Commission: Ispra, Italy, 2013; 128p. [Google Scholar]
- Koelmans, A.A. Modeling the role of microplastics in bioaccumulation of organic chemicals to marine aquatic organisms. A Critical Review. In Marine anthropogenic litter; Bergmann, M., Gutow, L., Klages, M., Eds.; Springer: Berlin, Germany, 2015; pp. 313–328. [Google Scholar]
- Ng, E.-L.; Lwanga, E.H.; Eldridge, S.M.; Johnston, P.; Hu, H.-W.; Geissen, V.; Chen, D. An overview of microplastic and nanoplastic pollution in agroecosystems. Sci. Total Environ. 2018, 627, 1377–1388. [Google Scholar] [CrossRef]
- Nizzetto, L.; Futter, M.; Langaas, S. Are agricultural soils dumps for microplastics of urban origin? Environ. Sci. Technol. 2016, 50, 10777–10779. [Google Scholar] [CrossRef]
- Nizzetto, L.; Langaas, S.; Futter, M. Pollution: Do microplastics spill on to farm soils? Nature 2016, 537, 488. [Google Scholar] [CrossRef]
- Besseling, E.; Quik, J.T.K.; Sun, M.; Koelmams, A.A. Fate of nano- and microplastic in freshwater systems: A modeling study. Environ. Pollut. 2016, 220, 540–548. [Google Scholar] [CrossRef]
- Piehl, S.; Leibner, A.; Löder, M.G.J.; Dris, R.; Bogner, C.; Laforsch, C. Identification and quantification of macro- and microplastics on agricultural farmland. Sci. Rep. 2018, 8, 17950. [Google Scholar] [CrossRef]
- Rillig, M. Microplastic in terrestrial ecosystems and the soil. Environ. Sci. Technol. 2012, 46, 6453–6454. [Google Scholar] [CrossRef]
- Weithmann, N.; Möller, J.N.; Löder, M.G.J.; Piehl, S.; Laforsch, C.; Freitag, R. Organic fertilizer as a vehicle for the entry of microplastic into the environment. Sci. Adv. 2018, 4, eaap8060. [Google Scholar] [CrossRef] [PubMed]
- de Souza Machado, A.A.; Kloas, W.; Zarfl, C.; Hempel, S.; Rillig, M.C. Microplastics as an emerging threat to terrestrial ecosystems. Glob. Change Biol. 2018, 24, 1405–1416. [Google Scholar] [CrossRef] [PubMed]
- de Souza Machado, A.A.; Lau, C.W.; Till, J.; Kloas, W.; Lehmann, A.; Becker, R.; Rillig, M.C. Impacts of microplastics on the soil biophysical environment. Environ. Sci. Technol. 2018, 52, 9656–9665. [Google Scholar] [CrossRef] [PubMed]
- Pflugmacher, S.; Huttunen, J.H.; von Wolff, M.A.; Penttinen, O.P.; Kim, Y.J.; Kim, S.; Mitrovic, S.M.; Esterhuizen-Londt, M. Enchytraeus crypticus avoid soil spiked with microplastic. Toxics 2020, 8, 10. [Google Scholar] [CrossRef] [PubMed]
- Lithner, D.; Larsson, A.; Dave, G. Environmental and health ranking and assessment of plastic polymers based on chemical composition. Sci. Total Environ. 2011, 409, 3309–3324. [Google Scholar] [CrossRef]
- Hermabessiere, L.; Dehaut, A.; Paul-Pont, I.; Lacroix, C.; Jezequel, R.; Soudant, P.; Duflos, G. Occurrence and effects of plastic additives on marine environments and organisms: A review. Chemosphere 2017, 182, 781–793. [Google Scholar] [CrossRef]
- Crompton, T. Additive migration from plastics into foods. A guide for analytical chemistry; Smithers Rapra Technology Publishing: Shrewsbury, UK, 2007. [Google Scholar]
- Møller, L. Environmental Project no. 901, 2004. Retrieved 2010, from Evaluation of Alternatives for Compounds under Risk Assessment in the EU, Bisphenol A. Available online: http://www.miljoindflydelse.dk/common/Udgivramme/Frame.asp?http://www.miljoindflydelse.dk/udgiv/publications/2004/87-7614-181-0/html/helepubl_eng.htm (accessed on 4 February 2020).
- Labouriau, L.G. A germinação das sementes. Organização dos Estados Americanos. Programa Regional de Desenvolvimento Científico e Tecnoloógico. Série de Biologia., Monografia 1983, 24, 174. [Google Scholar]
- Parkpian, P.; Leong, S.T.; Laortanakul, P.; Juntaramitree, J. An environmentally sound method for disposal of both ash and sludge wastes by mixing with soil: A case study of Bangkok plain. Environ. Monit. Assess. 2002, 74, 27–43. [Google Scholar] [CrossRef]
- Staples, C.A.; Dome, P.B.; Klecka, G.M.; Oblock, S.T.; Harris, L.R. A review of the environmental fate, effects, and exposures of bisphenol A. Chemosphere 1998, 36, 2149–2173. [Google Scholar] [CrossRef]
- Dogan, M.; Yumrutas, O.; Saygideger, S.D.; Korkunc, M.; Gulnaz, O.; Sokmen, A. Effects of bisphenol A and tetrabromobisphenol A on chickpea roots in germination stage. Am. Eurasian, J. Agric. Environ. Sci. 2010, 9, 186–192. [Google Scholar]
- Dogan, M.; Korkung, M.; Yumrutas, O. Effects of bisphenol A and tetrabromobisphenol A on bread and durum wheat varieties. Ekoloji 2012, 21, 114–122. [Google Scholar] [CrossRef]
- Al-Hiyasat, A. The effect of bisphenol A on root development and chlorophyll a:b ratio in Lens culinaris. Int. J. Sci.: Basic Appl. Res. 2017, 34, 115–123. [Google Scholar]
- Rillig, M.C.; Lehmann, A.; de Souza Machado, A.A.; Yang, G. Microplastic effects on plants. New Phytol. 2019, 223, 1066–1070. [Google Scholar] [CrossRef] [PubMed]
- Maila, M.P.; Cloete, T.E. Germination of Lepidium sativum as a method to evaluate polycyclic aromatic hydrocarbons (PAHs) removal from contaminated soil. Int. Biodeter. Biodegr. 2002, 50, 107–113. [Google Scholar] [CrossRef]
- Bosker, T.; Bouwman, L.J.; Brun, N.R.; Behrens, P.; Vijver, M.G. Microplastics accumulate on pores in seed capsule and delay germination and root growth of the terrestrial vascular plant Lepidium sativum. Chemosphere 2019, 226, 774–781. [Google Scholar] [CrossRef]
- Wan, Y.; Wu, C.C.; Xue, Q.; Hui, X. Effects of plastic contamination on water evaporation and desiccation cracking in soil. Sci. Total Environ. 2019, 654, 576–582. [Google Scholar] [CrossRef]
- Ferrara, G.; Loffredo, E.; Senesi, N. Phytotoxic, clastogenic and bioaccumulation effects of the environmental endocrine disruptor bisphenol A in various crops grown hydroponically. Planta 2006, 223, 910–916. [Google Scholar] [CrossRef]
- Sun, H.; Wang, L.; Zhou, Q. Effects of bisphenol A on growth and nitrogen nutrition of roots of soybean seedlings. Environ. Toxicol. Chem. 2013, 32, 174–180. [Google Scholar] [CrossRef]
- Nicola, S. Understanding root systems to improve seedling quality. Hort. Technol. 1998, 8, 544–549. [Google Scholar] [CrossRef]
- Leskovar, D.I.; Stoffella, P.J. Vegetable seedling root systems: Morphology, development and importance. HortScience 1995, 30, 1153–1159. [Google Scholar] [CrossRef]
- Zimmermann, R.P.; Kardos, L.T. Effect of bulk density on root growth. Soil Sci. 1961, 91, 280–288. [Google Scholar] [CrossRef]
- Esechie, H. Interaction of salinity and temperature on the germination of sorghum. J. Agron. Crop Sci. 1994, 172, 194–199. [Google Scholar] [CrossRef]
- Sommer, F.; Dietze, V.; Baum, A.; Sauer, J.; Gilge, S.; Maschowski, C.; Giere, R. Tire abrasion as a major source of microplastics in the environment. Aerosol Air Qual. Res. 2018, 18, 2014–2028. [Google Scholar] [CrossRef]
- International Organization for Standardization (ISO). Water Quality - Determination of the Acute Lethal Toxicity of Substances to a Freshwater Fish [Brachydanio Rerio Hamilton-Buchanan (Teleostei, Cyprinidae)]. ISO 7346-3: Flow-through Method; ISO: Paris, France, 1996; Available online: http://www.iso.org (accessed on 6 February 2020).
- Fejdyś, M.; Landwijt, M.; Strusczyk, M.H. Effects of accelerated ageing conditions on the degradation process of dyneema ® polyethylene composites. Fibres Text. East Eur. 2011, 19, 60–65. [Google Scholar]
- Swedish Standards Institute (SIS). Characterization of Waste - Leaching - Compliance Test for Leaching of Granular Waste Materials and Sludges; Svensk Standard SS-EN 12457-3; SIS: Stockholm, Sweden, 2003; Available online: https://www.sis.se (accessed on 6 February 2020).
- Wypych, G. Weathering of plastics: Testing to mirror life performance, 1st ed.; ChemTec Publishing: New York, NY, USA, 1999; ISBN 1-884207-75-8. [Google Scholar]
- Vela-Soria, F.; Ballesteros, O.; Zafra-Gòmez, A.; Ballesteros, L.; Navalòn, A. UHPLC-MS/MS method for the determination of bisphenol A and its chlorinated derivatives, bisphenol S, parabens, and benzophenones in human urine samples. Anal. Bioanal. Chem. 2014, 406, 3773–3875. [Google Scholar] [CrossRef]
- Scopetani, C.; Esterhuizen-Londt, M.; Chelazzi, D.; Cincinelli, A.; Setälä, H.; Pflugmacher, S. Self-contamination from clothing in microplastics research. Ecotoxicol. Environ. Saf. 2020, 189, 110036. [Google Scholar] [CrossRef]
- Janssen, J.G.M. A method of recording germination curves. Ann. Bot. 1973, 37, 705–708. [Google Scholar] [CrossRef]
- Scott, S.J.; Jones, R.A.; Williams, W.A. Review of data analysis for seed germination. Crop Sci. 1984, 24, 1192–1199. [Google Scholar] [CrossRef]
- International Seed Testing Association (ISTA). International rules for seed testing. Seed Sci. Technol. 1985, 13, 307–513. [Google Scholar]
- International Seed Testing Association (ISTA). Chapter 5: The germination test. International Rules for Seed Testing; International Seed Testing Association: Zurich, Switzerland, 2015; p. i–5–56. [Google Scholar] [CrossRef]
- Throneberry, G.O.; Smith, F.G. Relation of respiratory and enzymatic activity to corn seed viability. Plant Physiol. 1955, 30, 337–343. [Google Scholar] [CrossRef]
- Wardle, D.A.; Ahmed, M.; Nicholson, K.S. Allelopathic influence of nodding thistle (Carduus nutans L.) seeds on germination and radicle growth of pasture plants. New Zeal. J. Agr. Res. 1991, 34, 185–191. [Google Scholar] [CrossRef]
- Sokal, R.R.; Rohlf, F.J. Biometry. The Principles and Practice of Statistic in Biological Research; WH Freeman and Company: New York, NY, USA, 1997. [Google Scholar]

| MP Concentration | GRI Substrate-Free Exposures | GRI Substrate Exposures | |
|---|---|---|---|
| control | 1.59 ± 0.06 | 1.71 ± 0.03 | |
| new-PC-Leachate | 0.1% 1.0% 10% | 1.23 ± 0.06 0.62 ± 0.03 0.22 ± 0.04 | 1.29 ± 0.05 1.12 ± 0.06 0.50 ± 0.06 |
| aged-PC-Leachate | 0.1% 1.0% 10% | 1.50 ± 0.06 1.37 ± 0.05 1.11 ± 0.04 | 1.49 ± 0.06 1.27 ± 0.10 1.05 ± 0.04 |
| BPA | 10 mg/L 50 mg/L 100 mg/L | 1.46 ± 0.05 1.33 ± 0.06 0.93 ± 0.05 | 1.63 ± 0.70 1.49 ± 0.04 1.30 ± 0.03 |
| new-PC granules | 0.1% 1.0% 10% | 1.45 ± 0.05 1.20 ± 0.08 0.61 ± 0.06 | |
| aged-PC granules | 0.1% 1.0% 10% | 1.61 ± 0.07 1.37 ± 0.05 1.11 ± 0.03 | |
| new-PC-leached granules | 0.1% 1.0% 10% | 1.58 ± 0.06 1.38 ± 0.05 1.14 ± 0.02 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pflugmacher, S.; Sulek, A.; Mader, H.; Heo, J.; Noh, J.H.; Penttinen, O.-P.; Kim, Y.; Kim, S.; Esterhuizen, M. The Influence of New and Artificial Aged Microplastic and Leachates on the Germination of Lepidium sativum L. Plants 2020, 9, 339. https://doi.org/10.3390/plants9030339
Pflugmacher S, Sulek A, Mader H, Heo J, Noh JH, Penttinen O-P, Kim Y, Kim S, Esterhuizen M. The Influence of New and Artificial Aged Microplastic and Leachates on the Germination of Lepidium sativum L. Plants. 2020; 9(3):339. https://doi.org/10.3390/plants9030339
Chicago/Turabian StylePflugmacher, Stephan, Amalia Sulek, Hannah Mader, Jeongin Heo, Ji Hyeon Noh, Olli-Pekka Penttinen, YoungJun Kim, Sanghun Kim, and Maranda Esterhuizen. 2020. "The Influence of New and Artificial Aged Microplastic and Leachates on the Germination of Lepidium sativum L." Plants 9, no. 3: 339. https://doi.org/10.3390/plants9030339
APA StylePflugmacher, S., Sulek, A., Mader, H., Heo, J., Noh, J. H., Penttinen, O.-P., Kim, Y., Kim, S., & Esterhuizen, M. (2020). The Influence of New and Artificial Aged Microplastic and Leachates on the Germination of Lepidium sativum L. Plants, 9(3), 339. https://doi.org/10.3390/plants9030339





